Transcriptome Analysis of Leg Muscles and the Effects of ALOX5 on Proliferation and Differentiation of Myoblasts in Haiyang Yellow Chickens
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Experimental Animals and Tissues
2.3. RNA Isolation, Library Preparation, and Sequencing
2.4. Quality Control and Comparative Analysis
2.5. Sequencing Data Analysis and Transcriptome Assembly
2.6. Validation of Gene Expression by Quantitative PCR Analysis
2.7. Isolation and Culture of Chicken Primary Myoblast (CPM)
2.8. RNA Oligonucleotides and Plasmids Construction
2.9. Cell Transfection
2.10. Cell Proliferation Capacity Assay
2.10.1. EdU Assay
2.10.2. CCK-8 Assay
2.11. Cell Differentiation Ability Assay
2.11.1. Immunofluorescence
2.11.2. Changes in the Expression of Genes Related to Cell Differentiation
2.12. Statistical Analyses
3. Results
3.1. Transcriptome Sequencing Results of Tissue RNA Samples
3.2. Comparative Analysis of Reads and Reference Genome
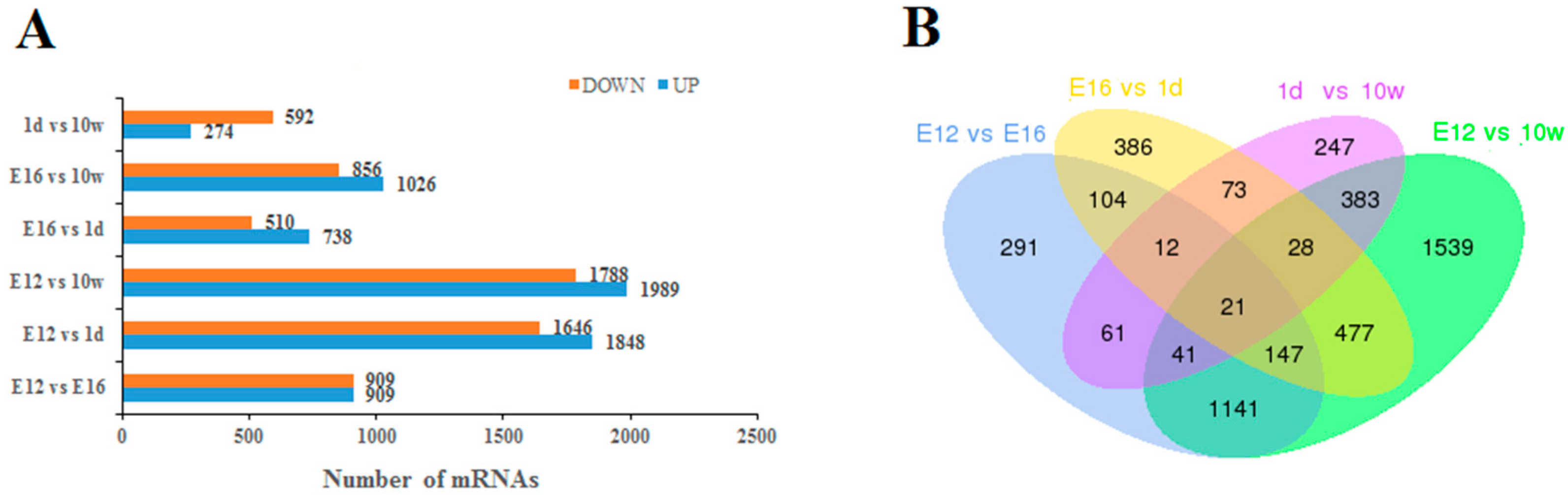
3.3. Screening and Functional Analysis of DEGs
3.3.1. Screening of DEGs
3.3.2. GO Functional Annotation and Enrichment Analysis of DEGs
3.3.3. Pathway Enrichment Analysis of DEGs
3.4. qRT-PCR Validation of DEGs Obtained via RNA-Seq
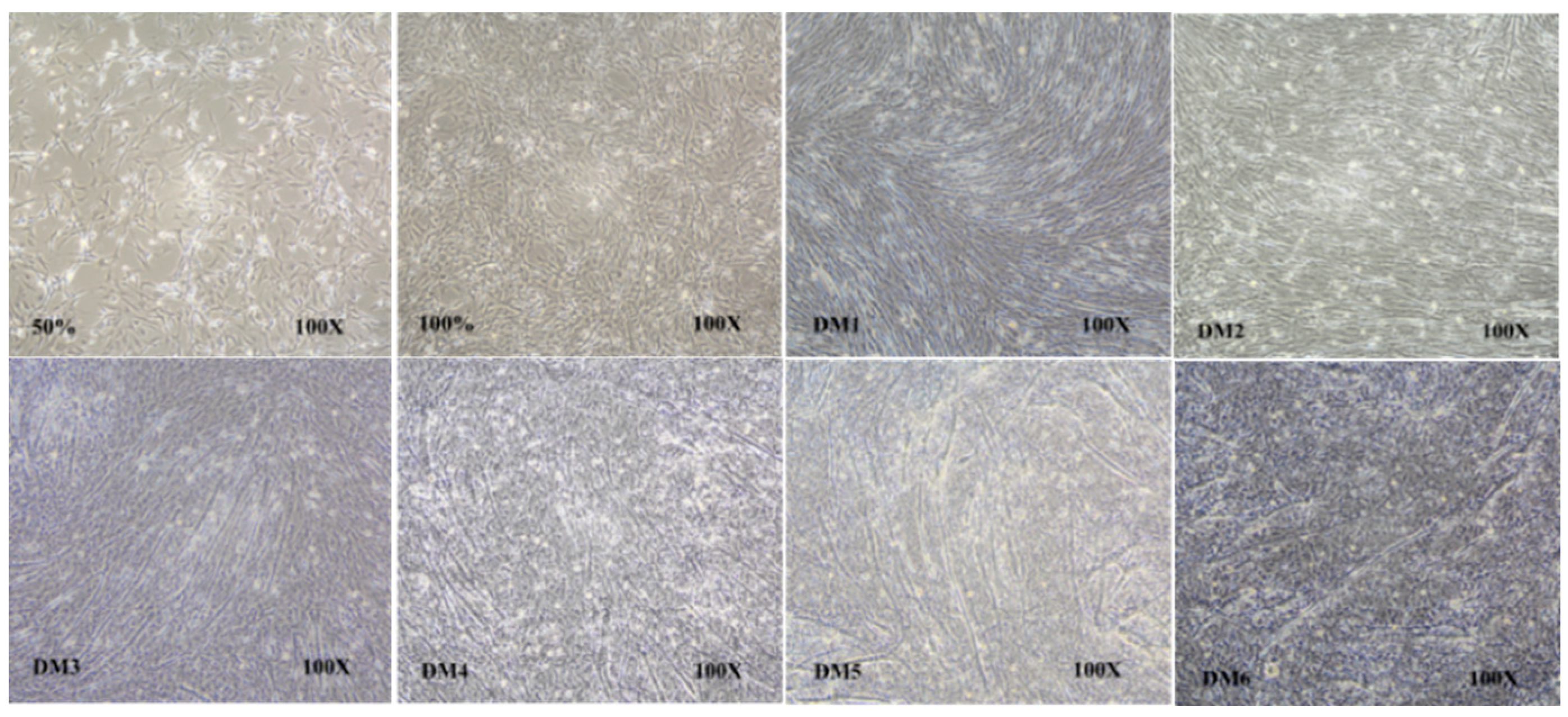
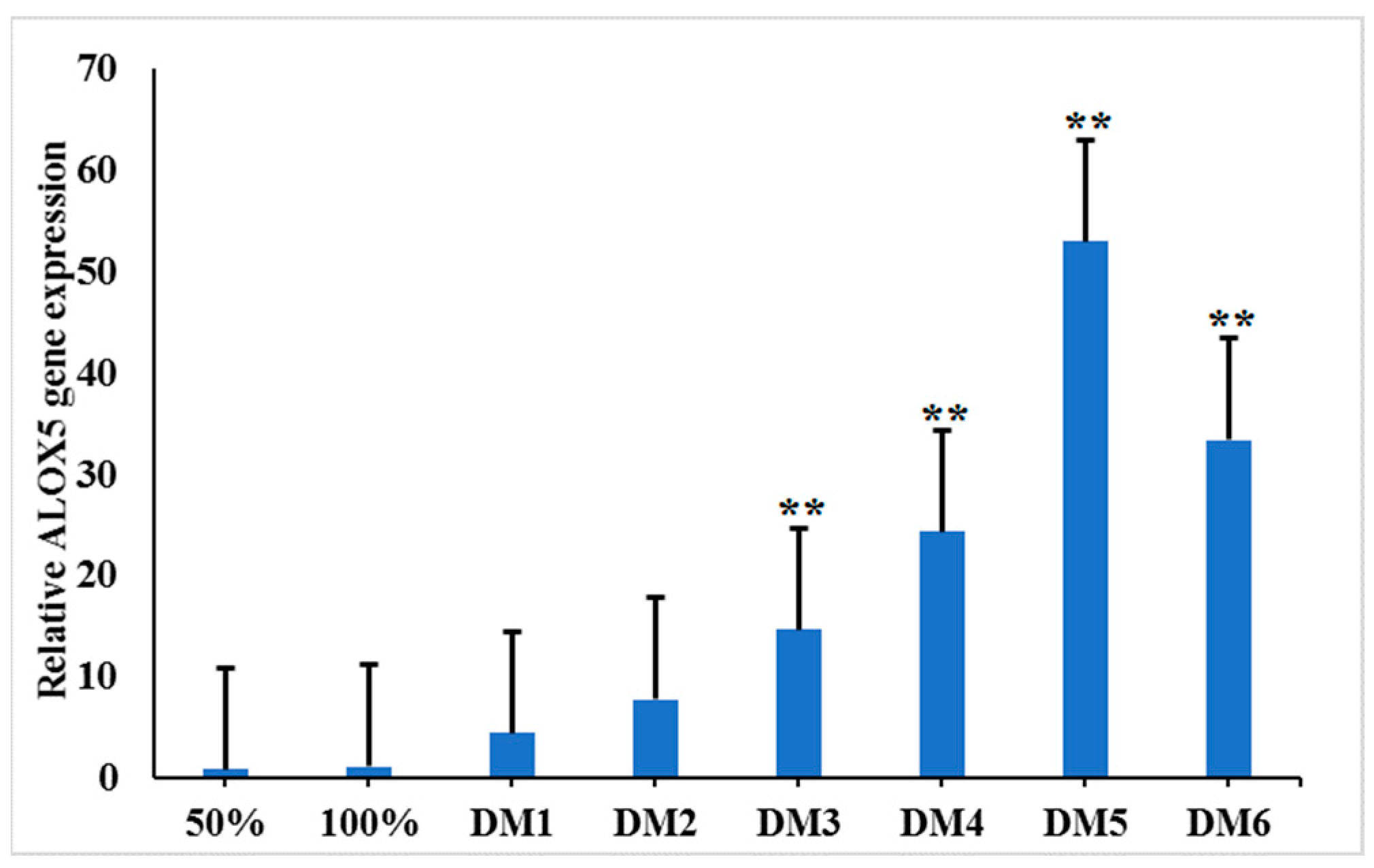
3.5. Spatial and Temporal Expression of the ALOX5 Gene during Myoblast Differentiation
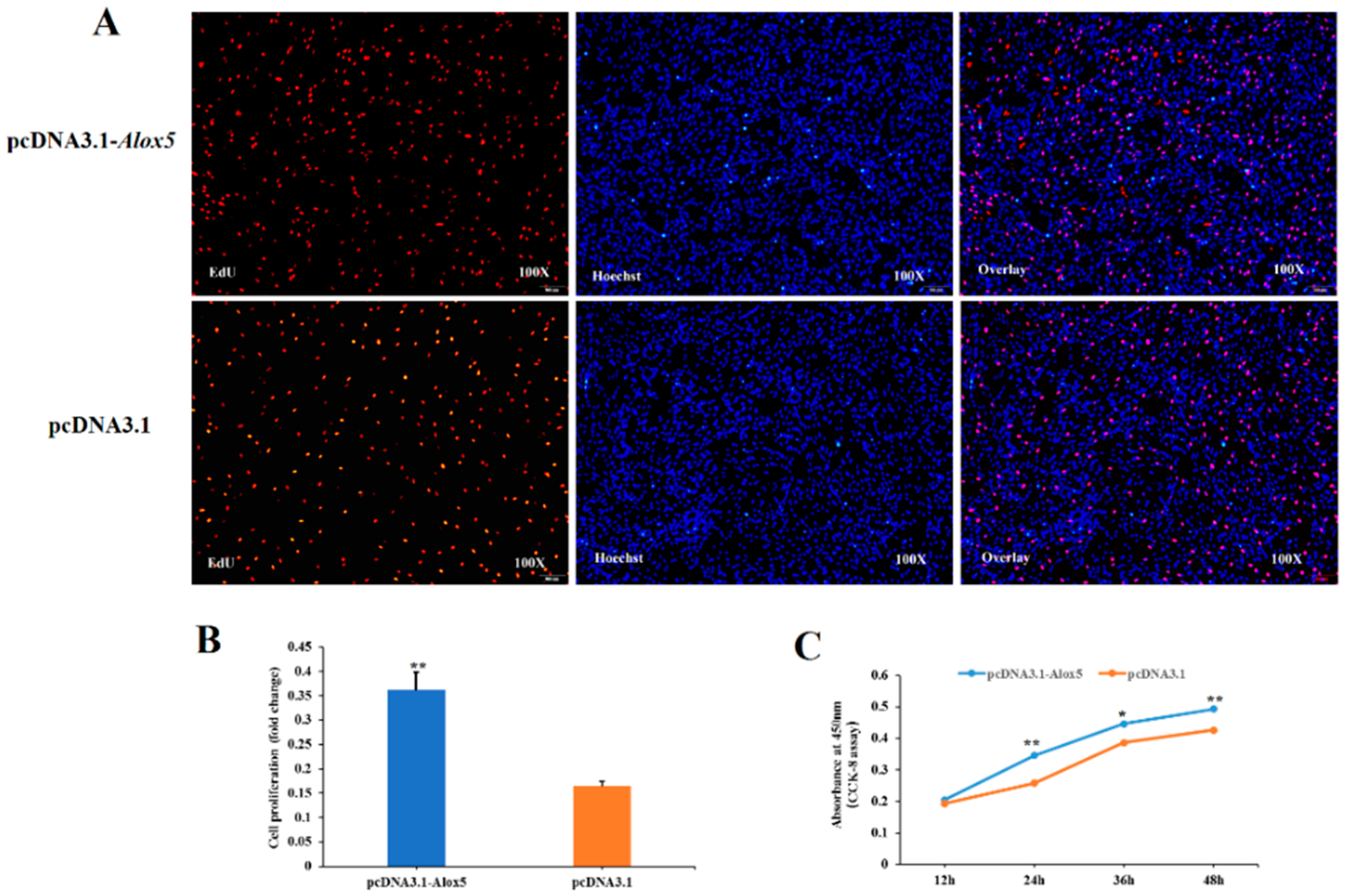
3.6. Effect of ALOX5 Gene on Myoblast Proliferation
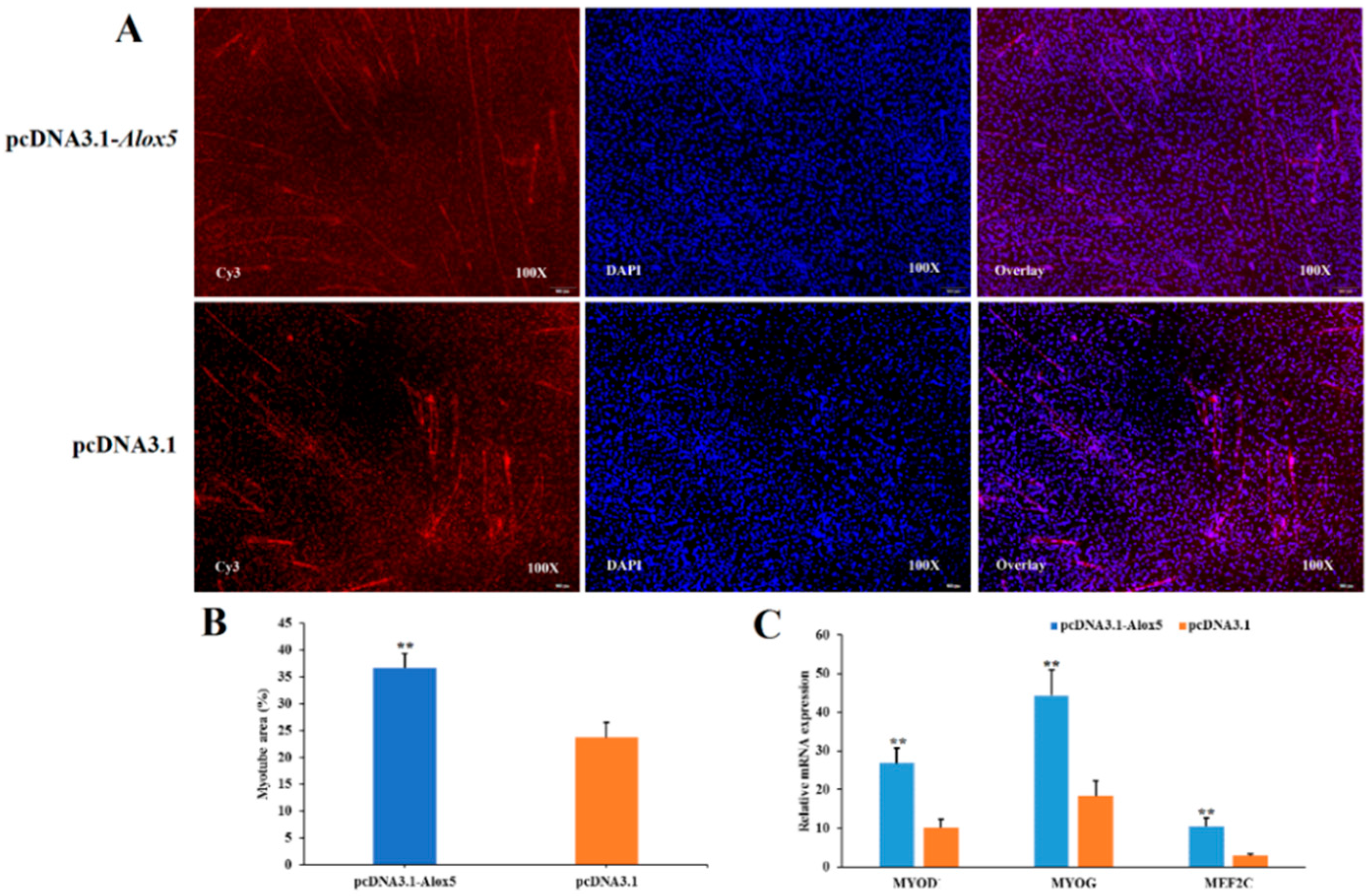
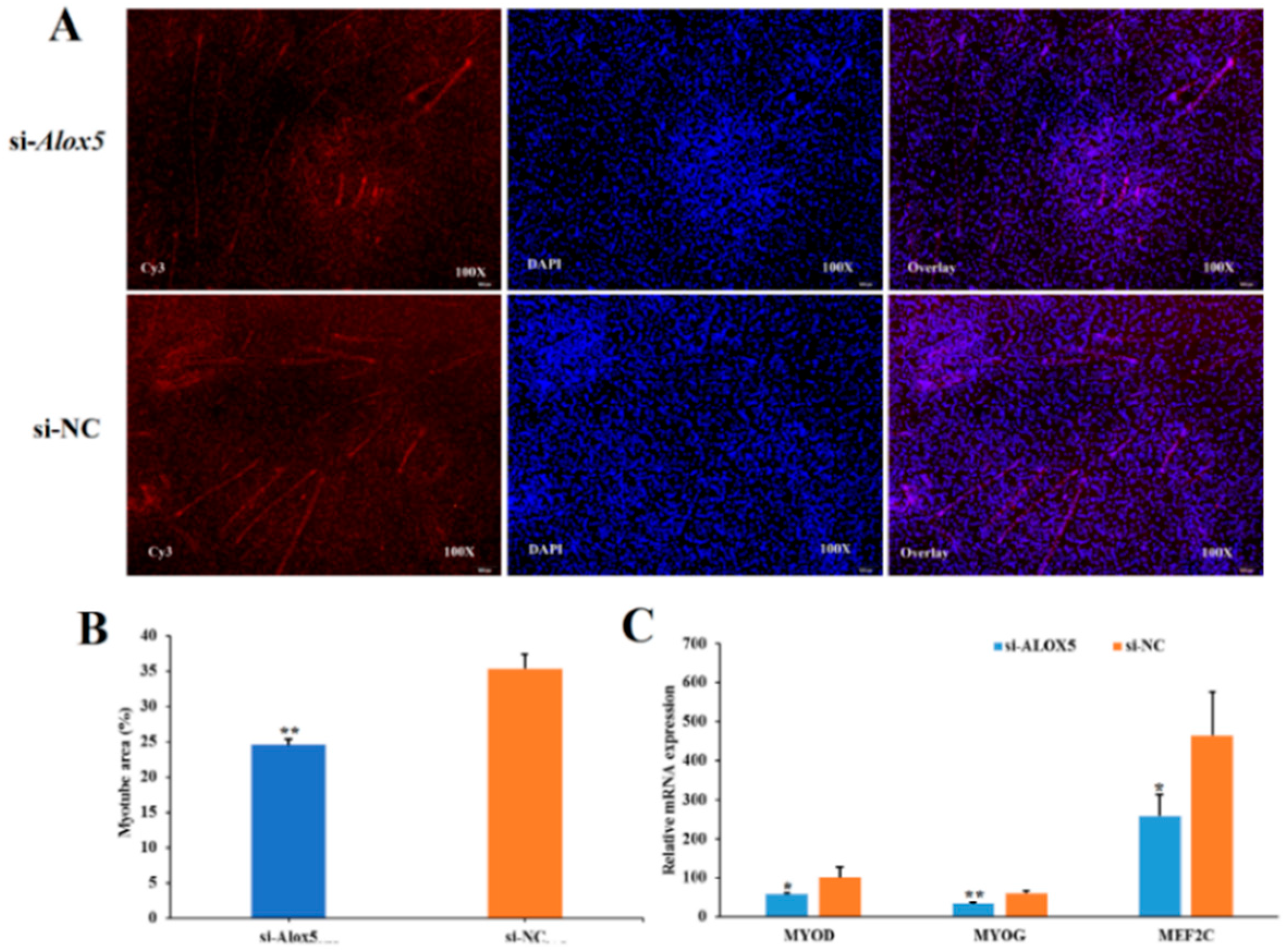
3.7. Effect of ALOX5 Gene on Myoblast Differentiation
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Groothuis, T.G.; Eising, C.M.; Dijkstra, C.; Müller, W. Balancing between costs and benefits of maternal hormone deposition in avian eggs. Biol. Lett. 2005, 1, 78–81. [Google Scholar] [CrossRef]
- Groothuis, T.G.G.; Müller, W.; Engelhardt, N.V.; Carere, C.; Eising, C. Maternal hormones as a tool to adjust offspring phenotype in avian species. Neurosci. Biobehav. Rev. 2005, 29, 329–352. [Google Scholar] [CrossRef]
- Cottrell, E.C.; Ozanne, S.E. Developmental programming of energy balance and the metabolic syndrome. Proc. Nutr. Soc. 2007, 66, 198–206. [Google Scholar] [CrossRef]
- Ge, S.F.; Zhao, R.Q.; Chen, W.H.; Chen, J. Changes of some serum hormone levels during embryonic development in Shaoxing and Gaoyou ducks. J. Nanjing Agric. Univ. 2000, 23, 63–66. [Google Scholar]
- Zhao, R.Q.; Li, S.G.; Chen, W.H.; Lei, Z.H.; Chen, J. A comparative study of pituitary GH gene expression between broiler and layer chickens during embryonic and neonatal development. Acta Vet. Zootech. Sin. 2001, 32, 5–10. [Google Scholar]
- McMillen, I.C.; Edwards, L.J.; Duffield, J.; Muhlhausler, B.S. Regulation of leptin synthesis and secretion before birth: Implications for the early programming of adult obesity. Reproduction 2006, 131, 415–427. [Google Scholar] [CrossRef]
- Bentzinger, C.F.; Wang, Y.X.; Rudnicki, M.A. Building muscle: Molecular regulation of MYOGenesis. Cold Spring Harb. Perspect. Biol. 2012, 4, a008342. [Google Scholar] [CrossRef]
- Gilbreath, H.R.; Castro, D.; Iannaccone, S.T. Congenital myopathies and muscular dystrophies. Neurol. Clin. 2014, 32, 689–703. [Google Scholar] [CrossRef]
- Shen, X.M.; Tang, J.; Jiang, R.; Wang, X.G.; Yang, Z.X.; Huang, Y.Z.; Lan, X.Y.; Lei, C.Z.; Chen, H. CircRILRL 1 promotes muscle proliferation and differentiation via bingding miR-145 to activate IGF1R/PI3K/AKT pathway. Cell Death Dis. 2021, 12, 142. [Google Scholar] [CrossRef]
- Zhang, G.X.; Chen, F.X.; Wu, P.F.; Li, T.T.; He, M.L.; Yin, X.M.; Shi, H.Q.; Duan, Y.J.; Zhang, T.; Wang, J.Y.; et al. MicroRNA-7 targets the KLF4 gene to regulate the proliferation and differentiation of chicken primary myoblasts. Front. Genet. 2020, 11, 842. [Google Scholar] [CrossRef]
- Zhang, G.X.; He, M.L.; Wu, P.F.; Zhang, X.C.; Zhou, K.Z.; Li, T.T.; Zhang, T.; Xie, K.Z.; Dai, G.J.; Wang, J.Y. MicroRNA-27b-3p targets the Myostatin gene to regulate myoblast proliferation and is involved in myoblast differentiation. Cells 2021, 10, 423. [Google Scholar] [CrossRef]
- Hou, D. Biological Effects of CKM Gene on Proliferation and Differentiation of Chicken Myoblasts; Henan Agricultural University: Zhengzhou, China, 2022. [Google Scholar]
- Mo, J.W.; Li, Z.H.; Wang, Z.J.; Nie, Q.H. Study on the regulation mechanism of ApoD on the proliferation and differentiation of chicken myoblasts. China Poult. 2022, 44, 8–13. [Google Scholar]
- Schmieder, R.; Edwards, R. Quality control and preprocessing of metagenomics datasets. Bioinformatics 2011, 27, 863–864. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2 (-Delta Delta C (T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Lu, S.S.; Yang, L.L.; Li, T.B.; Zhang, M.; Jiang, M.S. Based on transcriptome sequencing to explore the relationship between growth and development genes of Guangxi Ma Chickens. China Anim. Husb. Vet. Med. 2022, 49, 3901–3908. [Google Scholar]
- Piórkowska, K.; Żukowski, K.; Nowak, J.; Połtowicz, K.; Ropka-Molik, K.; Gurgul, A. Genome-wide RNA-Seq analysis of breast muscles of two broiler chicken groups differing in shear force. Anim. Genet. 2016, 47, 68–80. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.Y.; Ni, K.; Lv, G.X. Structure and working mechanism of myosin. J. Mol. Catal. 2007, 6, 272–279. [Google Scholar]
- Han, Y.R.; Liu, H.; Zhan, Y.; Wu, W.X.; Zhao, T.J.; Chen, Y.F. Nanomachine—Molecular motors. Bull. Biol. 2010, 45, 6–9. [Google Scholar]
- Li, Y.; Xu, Z.Y.; Li, H.Y.; Xiong, Y.Z.; Zuo, B. Differential transcriptional analysis between red and white skeletal muscle of Chinese Meishan pigs. Int. J. Biol. Sci. 2010, 6, 350–360. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.L. Effects of PHGDH Gene on Meat Traits and Proliferation and Differentiation of MYOGenic Cells in Heying Black Chicken; Yangzhou University: Yangzhou, China, 2021. [Google Scholar]
- Chidgey, M.; Dawson, C. Desmosomes: A role in cancer. Br. J. Cancer 2007, 96, 1783–1787. [Google Scholar] [CrossRef]
- Kamekura, R.; Kolegraff, K.N.; Nava, P.; Hilgarth, R.S.; Feng, M.; Parkos, C.A.; Nusrat, A. Loss of the desmosomal cadherin desmoglein-2 suppresses colon cancer cell proliferation through EGFR signaling. Oncogene 2014, 33, 4531–4536. [Google Scholar] [CrossRef]
- Chen, Y.Y.; Hu, Y.G.; Zhang, H.J.; Peng, C.; Li, S.G. Loss of the ALOX5 gene impairs leukemia stem cells and prevents chronic myeloid leukemia. Nat. Genet. 2009, 41, 783–792. [Google Scholar] [CrossRef]
- Zheng, Y.; Wang, L.S.; Xia, L.; Han, Y.H.; Liao, S.H.; Wang, X.L.; Cheng, J.K.; Chen, G. NDRG1 is down-regulated in the early apoptotic event induced by camptothecin analogs: The potential role in proteolytic activation of PKC delta and apoptosis. Proteomics 2009, 9, 2064–2075. [Google Scholar] [CrossRef]
- Lovly, C.M.; McDonald, N.T.; Chen, H.; Ortiz-Cuaran, S.; Heukamp, L.C.; Yan, Y.J.; Florin, A.; Ozretić, L.; Lim, D.; Wang, L.; et al. Rationale for co-targeting IGF-1R and ALK in ALK fusion–positive lung cancer. Nat. Med. 2014, 20, 1027–1034. [Google Scholar] [CrossRef]
- Aleksic, T.; Verrill, C.; Bryant, R.J.; Han, C.; Worrall, A.R.; Brureau, L.; Larré, S.; Higgins, G.S.; Fazal, F.; Sabbagh, A.; et al. IGF-1R associates with adverse outcomes after radical radiotherapy for prostate cancer. Br. J. Cancer 2017, 117, 1600–1606. [Google Scholar] [CrossRef]
- Gospodarowicz, D. Localisation of a fibroblast growth factor and its effect alone and with hydrocortisone on 3T3 cell growth. Nature 1974, 249, 123–127. [Google Scholar] [CrossRef]
- Smith, H.J. Relation of Body Size to Muscle Cell Size and Number in the Chicken. Poult. Sci. 1963, 42, 283–290. [Google Scholar] [CrossRef]
- Velleman, S.G. Muscle Development in the Embryo and Hatchling. Poult. Sci. 2007, 86, 1050–1054. [Google Scholar] [CrossRef]
- Moss, F.P. The relationship between the dimensions of the fibres and the number of nuclei during restricted growth, degrowth and compensatory growth of skeletal muscle. Am. J. Anat. 1968, 122, 565–571. [Google Scholar] [CrossRef]
- Thomas, K.; Engler, A.J.; Meyer, G.A. Extracellular matrix regulation in the muscle satellite cell niche. Connect. Tissue Res. 2015, 56, 1–8. [Google Scholar] [CrossRef]
- Jenniskens, G.J.; Veerkamp, J.H.; Kuppevelt, T.H.V. Heparan sulfates in skeletal muscle development and physiology. J. Cell Physiol. 2006, 206, 283–294. [Google Scholar] [CrossRef]
- Velleman, S.G. The role of the extracellular matrix in skeletal muscle development. Poult. Sci. 1999, 78, 778–784. [Google Scholar] [CrossRef]
- Velleman, S.; Liu, X.; Eggen, K.; Nestor, K.E. Developmental regulation of proteoglycan synthesis and decorin expression during turkey embryonic skeletal muscle formation. Poult. Sci. 1999, 78, 1619–1626. [Google Scholar] [CrossRef] [PubMed]
- Nucera, C.; Lawler, J.; Parangi, S. BRAF(V600E) and Microenvironment in Thyroid Cancer: A Functional Link to Drive Cancer Progression. Cancer Res. 2011, 71, 2417–2422. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Wu, S.G.; Zhang, H.J.; Yue, H.Y.; Zhai, Y.G.; Qi, G.H. Study on Oxidative Stress-Activated MAPK Signaling Pathway in Broilers Breast Muscle Satellite Cells. Sci. Agric. Sin. 2010, 43, 4286–4294. [Google Scholar]
- Keren, A.; Tamir, Y.; Bengal, E. The p38 MAPK signaling pathway: A major regulator of skeletal muscle development. Mol. Cell Endocrinol. 2006, 252, 224–230. [Google Scholar] [CrossRef]
- Ni, J.H.; Yang, Y.L.; Bai, D.P. Research progress of acetylatio, methylation modification and muscle development and entiation. Xinjiang Agric. Reclam. Technol. 2008, 031, 54–55. [Google Scholar]
- RaDmark, O.; Werz, O.; Steinhilber, D.; Samuelsson, B. 5-Lipoxygenase, a key enzyme for leukotriene biosynthesis in health and disease. Biochim. Biophys. Acta 2015, 1851, 331–339. [Google Scholar] [CrossRef]
- Gillian, M.; Graham, P. Cross-Talk between Cancer Cells and the Tumour Microenvironment: The Role of the 5-Lipoxygenase Pathway. Int. J. Mol. Sci. 2017, 18, 236. [Google Scholar]
- Sinem, T.; Sreeparna, B. Eicosanoid pathway in colorectal cancer:Recent updates. World J. Gastroenterol. 2015, 21, 11748–11766. [Google Scholar]
- Niu, P.F. The Regulation of Mybph Gene in Myoblast Proliferation and Differentiation; Huazhong Agricultural University: Wuhan, China, 2016. [Google Scholar]
- Cai, B.; Ma, M.; Chen, B.; Li, Z.H.; Abdalla, B.A.; Nie, Q.H.; Zhang, X.Q. MiR-16-5p targets SESN1 to regulate the p53 signaling pathway, affecting myoblast proliferation and apoptosis, and is involved in myoblast differentiation. Cell Death Dis. 2018, 9, 367. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.Y.; Wang, F.J.; Liu, R.R.; Zheng, M.Q.; Wen, J.; Fu, R.Q.; Jiang, R.S.; Zhao, G.P. Developmental changes in the expression of the. MYCN gene in chicken myoblast and breast muscle tissues. J. Anhui Agric. Univ. 2015, 42, 110–114. [Google Scholar]
- Gan, W.; Zhao, Y.P.; Zhang, N.N.; Song, Q.; Xiong, X.P.; Li, L. Effect of Leptin on Proliferation and Differentiation of Duck Myoblasts. Chin. J. Anim. Sci. 2017, 53, 50–57. [Google Scholar]
- Wyzykowski, J.C.; Winata, T.I.; Mitin, N.; Taparowsky, E.J.; Konieczny, S.F. Identification of Novel MyoD Gene Targets in Proliferating MYOGenic Stem Cells. Mol. Cell. Biol. 2002, 22, 6199–6208. [Google Scholar] [CrossRef]
- Li, Y.P.; Liang, B.S. Research progress on the mechanism of MyoD muscle formation. Int. J. Bone Sci. 2007, 28, 37–40. [Google Scholar]
- Liu, N.; Ruan, J.X.; Yang, S.L. Regulation of Porcine Muscle Development and Research Progress of Improved Pigs with High Lean Meat Percentage. J. Agric. Biotechnol. 2012, 20, 955–964. [Google Scholar]
- Mcfarland, D.C.; Pesall, J.E.; Gilkerson, K.K.; Swenning, T.A. Comparison of the proliferation and differentiation of MYOGenic satellite cells derived from Merriam’s and commercial varieties of turkeys. Comp. Biochem. Part A Physiol. 1993, 104, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Bowers, D.C.; Fan, S.; Walter, K.A.; Abounader, R.; Williams, J.A.; Rosen, E.M.; Laterra, J. Scatter Factor/Hepatocyte Growth Factor Protects against Cytotoxic Death in Human Glioblastoma via Phosphatidylinositol 3-Kinase- and AKT-dependent Pathways. Cancer Res. 2000, 60, 4277–4283. [Google Scholar]
- Borycki, A.G.; Li, J.; Jin, F.; Emerson, C.P.; Epstein, J.A. Pax3 functions in cell survival and in Pax7 regulation. Development 1999, 126, 1665–1674. [Google Scholar] [CrossRef] [PubMed]


| Primer Name | Primer Sequence (5′-3′) | Sequence Size (bp) |
|---|---|---|
| ALOX5-Gallus-578 | CCAGUUUGACAACGAGAAATT | 21 |
| UUUCUCGUUGUCAAACUGGTT | 21 | |
| ALOX5-Gallus-1000 | CCAUCUGUUUGCUGUACAATT | 21 |
| UUGUACAGCAAACAGAUGGTT | 21 | |
| ALOX5-Gallus-1160 | CCACCAGACGGUGACCCAUTT | 21 |
| AUGGGUCACCGUCUGGUGGTT | 21 | |
| ALOX5-Gallus-NC | UUCUCCGAACGUGUCACGUTT | 21 |
| Annotated | Description | |
|---|---|---|
| ENSGALG00000008601 | AHSG | α 2-HS glycoprotein |
| ENSGALG00000037889 | PTPRT | protein tyrosine phosphatase, receptor type T |
| ENSGALG00000030546 | CLEC19A | C-type lectin domain family 19 member A |
| ENSGALG00000005857 | ALOX5 | arachidonate 5-lipoxygenase |
| ENSGALG00000005043 | ACACB | acetyl-CoA carboxylase β |
| ENSGALG00000015142 | DSG2 | desmoglein 2 |
| ENSGALG00000005171 | AP1S3 | Gallus gallus adaptor-related protein complex 1, sigma 3 subunit (AP1S3) |
| ENSGALG00000003911 | CACNG5 | calcium voltage-gated channel auxiliary subunit γ 5 |
| ENSGALG00000000164 | MYBPH | Gallus gallus myosin binding protein H (MYBPH) |
| ENSGALG00000002988 | PHGDH | phosphoglycerate dehydrogenase |
| ENSGALG00000032404 | MYH1C | Gallus gallus myosin, heavy chain 1C, skeletal muscle |
| ENSGALG00000012594 | NTRK2 | Gallus gallus neurotrophic tyrosine kinase, receptor, type 2 (NTRK2) |
| GO Terms ID | Function Description | Number of DEGs | Partial Genes Associated with Muscle Growth and Development |
|---|---|---|---|
| GO:0032502 | developmental process | 994 | MYBPHL, MYOG, MYOCD, MUSTN1, MYLK3, MYL2, MYL3, GDF2, GDF10, MYOD1, MYLK2, FGF4, FGF8, MYBPC3, FGFR2, MYF6, ACTA1, EGF, IGF-I, MYBPC1, MEF2C, FGF14, MYOC, FGF9, FGF19, IGFBP3, IGF2BP1 |
| GO:0008283 | cell proliferation | 325 | MYOG, MYOCD, MUSTN1, GDF2, MYOD1, FGF4, FGF8, FGFR2, EGF, IGF-I, MEF2C, FGFR3, GAS6, FGF9, FGF19, IGF2BP1 |
| GO:0040007 | growth | 173 | MYOCD, MUSTN1, MYL2, GDF2, MYOD1, FGF8, IGF-I, MEF2C, FGF9, IGFBP3, GDF8 |
| KEGG Term | Number of DEGs | p-Value | Partial genes Associated with Muscle Growth and Development |
|---|---|---|---|
| ECM-receptor interaction | 31 | 0.00009 | LAMB3, COMP, LAMC1, COL9A3, CD36, ITGB8, SPP1, THBS2, VTN, TNR, |
| MAPK signaling pathway | 77 | 0.013 | FGF4, FGF8, FGFR2, EGF, IGF1, FGFR3, TGFA, FGF9, FGF19, FGF22 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yin, X.; Fang, W.; Yuan, M.; Sun, H.; Wang, J. Transcriptome Analysis of Leg Muscles and the Effects of ALOX5 on Proliferation and Differentiation of Myoblasts in Haiyang Yellow Chickens. Genes 2023, 14, 1213. https://doi.org/10.3390/genes14061213
Yin X, Fang W, Yuan M, Sun H, Wang J. Transcriptome Analysis of Leg Muscles and the Effects of ALOX5 on Proliferation and Differentiation of Myoblasts in Haiyang Yellow Chickens. Genes. 2023; 14(6):1213. https://doi.org/10.3390/genes14061213
Chicago/Turabian StyleYin, Xumei, Wenna Fang, Manman Yuan, Hao Sun, and Jinyu Wang. 2023. "Transcriptome Analysis of Leg Muscles and the Effects of ALOX5 on Proliferation and Differentiation of Myoblasts in Haiyang Yellow Chickens" Genes 14, no. 6: 1213. https://doi.org/10.3390/genes14061213




