Highlights
What are the main findings?
- Our study revealed genome-wide alternative splicing of rice protein-coding and long-noncoding RNA (lncRNA) genes in response to light with intron retention as a predominant splicing event.
- This is the first report on light-mediated splicing of genes involved in terpenoid backbone biosynthesis, DNA biosynthesis during cell division, and nitrogen assimilation pathway.
- Our study identified 1709 novel lncRNA transcripts, and a majority of the spliced lncRNA genes are located in the intergenic regions.
What is the implication of the main finding?
- Our data provide insights into the portion of the rice transcriptome undergoing alternative splicing in response to light.
- Our photomorphogenesis transcriptome study reveals novel lncRNAs in rice that may help future studies on the regulation of gene expression.
- Our data contain condition-dependent novel splice events not reported in the reference rice genome annotations, which will help improve genome annotation.
Abstract
Light is one of the most important factors regulating plant gene expression patterns, metabolism, physiology, growth, and development. To explore how light may induce or alter transcript splicing, we conducted RNA-Seq-based transcriptome analyses by comparing the samples harvested as etiolated seedlings grown under continuous dark conditions vs. the light-treated green seedlings. The study aims to reveal differentially regulated protein-coding genes and novel long noncoding RNAs (lncRNAs), their light-induced alternative splicing, and their association with biological pathways. We identified 14,766 differentially expressed genes, of which 4369 genes showed alternative splicing. We observed that genes mapped to the plastid-localized methyl-erythritol-phosphate (MEP) pathway were light-upregulated compared to the cytosolic mevalonate (MVA) pathway genes. Many of these genes also undergo splicing. These pathways provide crucial metabolite precursors for the biosynthesis of secondary metabolic compounds needed for chloroplast biogenesis, the establishment of a successful photosynthetic apparatus, and photomorphogenesis. In the chromosome-wide survey of the light-induced transcriptome, we observed intron retention as the most predominant splicing event. In addition, we identified 1709 novel lncRNA transcripts in our transcriptome data. This study provides insights on light-regulated gene expression and alternative splicing in rice.
1. Introduction
Light is an essential growth factor for sustaining autotrophic plant life. The quality, quantity, direction, and length of light exposure affect plant growth and development [1,2,3]. The natural light–dark cycle maintains the carbon and nitrogen metabolism of rice plants [4]. Light induces transcriptional reprogramming in various plant species [4,5,6,7,8,9,10,11,12] via an array of photoreceptors [13,14]. The photoreceptors phytochrome (red light) and cryptochrome (blue light) are involved in light signaling and photomorphogenesis [15,16]. Genes encoding phytochrome interacting factors (PIFs) promote skotomorphogenesis and development in the dark [17,18,19]. Light-mediated gene expression modulation is also triggered by translational enhancement of the pre-existing mRNA pool instead of an enhanced transcription rate [20,21].
Intron splicing in multiexonic mRNA is a post-transcriptional regulatory process that often produces different mRNA isoforms transcribed from a single gene locus, thus contributing to proteome plasticity. Light-induced alternative splicing (AS) was observed in ~10% of the protein-coding genes of Arabidopsis and Physcomitrella patens [22,23]. A flash of light applied in the middle of the dark or nighttime is sufficient to induce splicing [24]. In contrast to animals, where the most common AS event is exon skipping (ES), intron retention (IR) is the most common AS event in rice [25], Arabidopsis [25,26], poplar [27], and Salvia hispanica (Chia) [28]. It is now well-established that transcriptome modulation via AS is vital for plant growth, development, and stress response [29,30,31].
Compared to the protein-coding genes, long non-coding RNAs (lncRNAs) are transcripts >200 bp in length that do not have protein-coding potential. lncRNAs are classified as follows: (i) sense lncRNAs, (ii) antisense lncRNAs, (iii) intergenic RNAs (lincRNAs), (iv) intronic RNAs, and (v) bidirectional lncRNAs [32]. In addition to the numerous studies coupling gene expression and AS studies on protein-coding genes, it is now known that lncRNAs also play a role in regulating gene expression through transcriptional, post-transcriptional, and chromatin remodeling mechanisms [32,33,34,35]. In rice, lncRNAs regulate biological processes such as ovule development, female gametophyte abortion [36], sexual reproduction [37], stress response [38,39], and meiosis and low fertility in autotetraploids [40]. However, the role of light in regulating lncRNAs is not well studied. Arabidopsis non-coding RNA HID1 is a known positive regulator of photomorphogenesis in continuous red light [41].
Rice is a global staple crop and is a model for studying crop genomics. The transition from skotomorphogenesis under dark conditions to photomorphogenesis under light exposure is critical for seedling survival and requires precise control of gene expression by different regulatory mechanisms. High-throughput gene expression datasets are valuable sources for the detection of alternative transcripts, novel transcript isoforms, protein-coding and noncoding genes, improved annotation of plant transcriptomes, and studying developmental processes [42,43,44].
2. Materials and Methods
2.1. Plant Material, Growth Conditions, and Treatment
Seeds of rice (Oryza sativa spp. japonica cv. Nipponbare) were grown and processed for the experiment by following the growth conditions and sampling described previously [45]. Seeds were sown in the dark and germinated on day 2. After sowing, these germinated seedlings grew in the dark for 8 days. At the end of day 8, three biological replicates of the dark-grown etiolated shoots were harvested. The remaining dark-grown seedlings were exposed to continuous white light at 120 µmol/m2/sec (measured at the soil surface) for 48 h or 2 days (day 9 and 10 after sowing). The shoots of three biological replicates of light-treated green-colored seedling samples were harvested at the end of day 10. Harvested samples were frozen using liquid nitrogen and stored at −80 °C until further processing. Data analysis is described for light regulation.
2.2. Sample Preparation and Sequencing
Total RNA from frozen tissue samples was extracted using RNA Plant reagent (Thermo Fisher Scientific Inc., Waltham, MA, USA) and RNeasy kits (Qiagen Inc., Germantown, MD, USA) and treated with RNase-free DNase (Thermo Fisher Scientific Inc., USA) according to the manufacturer’s protocol. The total RNA quality and concentration were determined using an ND-1000 spectrophotometer (Thermo Fisher Scientific Inc., USA) and Bioanalyzer 2100 (Agilent Technologies Inc., Madison, WI, USA). PolyA-enriched mRNA libraries were prepared from three biological replicates of dark and light samples using the TruSeqTM RNA Sample Preparation Kits (v2) and sequenced as 51 bp single-end reads using the Illumina HiSeq 2000 instrument (Illumina Inc., San Diego, CA, USA) according to the manufacturer’s protocol at the Center for Genomic Research and Biocomputing, Oregon State University. The strand-specific sequencing reads and metadata were deposited at EMBL-EBI ArrayExpress (accession number E-MTAB-5689).
2.3. RNA-seq Data Analysis
The generation of FASTQ files from the RNA-Seq sequences was performed by CASAVA software v1.8.2 (Illumina Inc.). Sequence reads were filtered and trimmed for low quality at a score of 20 using Sickle v1.33 [46]. Clean, high-quality reads from each sample and replicates were aligned to the Osj cv Nipponbare reference genome (IRGSP-1.0.31) using TopHat v2.1.1 [47]. Mapped reads were assembled using Cufflinks, and the reference-guided assembled transcripts from each replicate were merged using Cuffmerge [48]. Assembled transcripts were compared to the reference genome annotation using Cuffcompare. The RSEM software package estimated normalized baseline expression from the aligned sequence reads [49]. For differential gene expression analysis, read count data obtained from RSEM were used in EBSeq [50]. Differentially expressed (DE) genes were filtered based on the false discovery rate-corrected p value ≤ 0.05.
2.4. Functional Annotation
We carried out the Gene Ontology (GO) enrichment analysis tool provided by the GO consortium [51] to determine the biological roles the enriched gene set played. Plant pathway enrichment analysis was performed by mapping the DE genes using the Plant Reactome analysis tool [52,53,54] (http://plantreactome.gramene.org/PathwayBrowser/#TOOL=AT accessed on 16 August 2023).
2.5. Alternative Splicing Analysis
The SpliceGrapher v0.2.5 pipeline identified splicing events in the transcripts from the samples [55]. Sequence reads from each sample, and from replicates, were aligned to the reference rice genome. Splice site-specific classifiers were built using build_classifiers.py script using canonical (GT) and noncanonical (GC) donor sites and acceptor site (AG) for Os genome annotation version 31 (Oryza_sativa.IRGSP-1.0.31). Read alignments in SAM format from each replicate were used as input for SpliceGrapher’s sam_filter.py script to filter out false-positive sites. SpliceGrapher Python scripts were used for the generation of depth files (sam_to_depths.py), splice graph prediction (predict_graphs.py), generating statistics (splicegraph_statistics.py) from a set of splice graphs, gene-by-gene summary (genewise_statistics.py) of splicing events, and splice graph visualization (plotter.py). The Realignment pipeline was used to construct putative transcripts from unresolved exons with sufficient coverage from the alignments [55]. In the following steps, IsoLasso [56], an extension of the SpliceGrapher workflow, was used to predict novel splicing events.
2.6. Prediction of Long Non-Coding RNA (lncRNA)
All transcripts annotated as intergenic transcripts, intron transcripts, antisense exon transcripts overlapping the reference exons, and antisense intron transcripts overlapping the reference introns were considered potential lncRNA candidates. Transcript sequences of length ≤200 nucleotides were filtered out, and the gffread function of Cufflinks was used to extract fasta sequences of potential lncRNA transcripts from the gtf file. CPC2 [57] was used to predict the coding potential of transcripts. Predicted lncRNAs were scanned by InterProScan [58] to ensure the absence of protein-coding domains. To identify novel lncRNAs, a BLASTn [59] search was performed against a custom BLAST database generated using rice lncRNAs downloaded from the CANTATAdb2.0 (http://cantata.amu.edu.pl accessed on 16 August 2023), PNRD [60], GreeNC [61], and RiceLNCPedia [62] databases. Secondary structures for the lncRNA molecules were predicted by the RNAfold software [63,64].
3. Results
3.1. Light-Mediated Differential Gene Expression during Photomorphogenesis
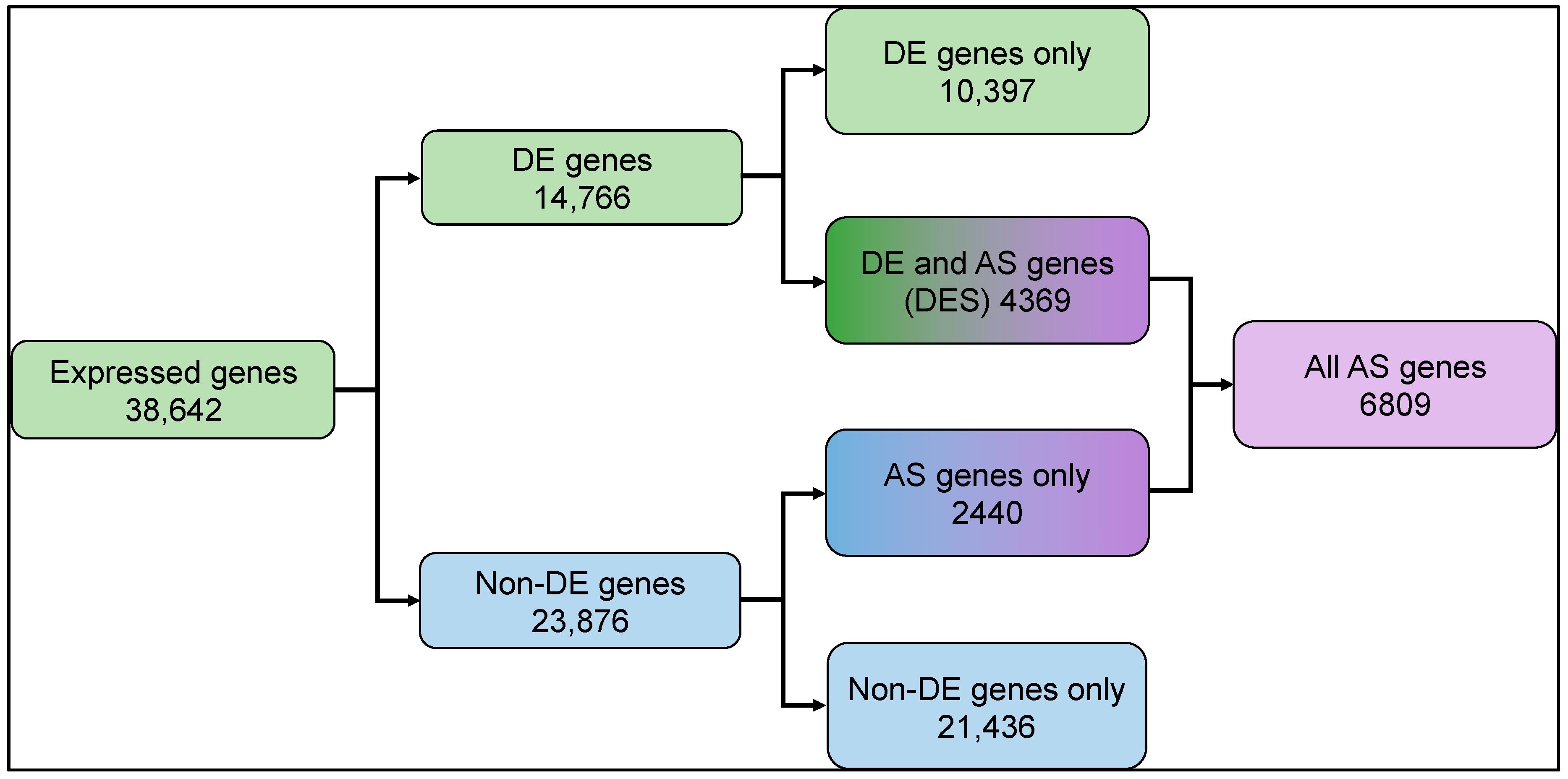
To explore the transcriptome modulation in rice in response to light, we sequenced the strand-specific poly-A enriched RNA fraction isolated from three biological replicates of rice plants grown under dark and light exposure conditions (see methods). A total of ~38 million and ~42 million high-quality reads were generated from the dark- and light-treated samples, respectively. More than 92% of reads from each sample aligned to the rice reference genome (Supplementary Table S1). We found 38,642 genes showed baseline normalized expression in the samples, of which 33,943 genes expressed in the dark vs. 35,772 genes that expressed under light, respectively. Differential expression analysis identified 14,766 light-regulated genes (Figure 1, Supplementary Figure S1, Supplementary Table S2).
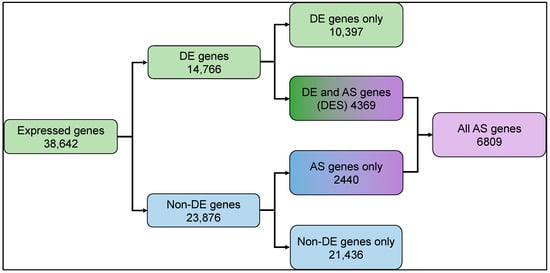
Figure 1.
Summary of differential gene expression and transcript splicing observed in rice seedling shoots undergoing photomorphogenesis. Expressed genes: total number of expressed genes in rice transcriptome; DE genes: differentially expressed genes; Non-DE genes: genes with no significant difference in expression; AS: alternatively spliced; DES: differentially expressed and spliced. Expressed and differentially expressed gene counts are represented by green shaded boxes, non-DE genes in blue shaded boxes, and AS genes in purple boxes. Mixed color boxes represent gene counts from overlapping AS, DE, and non-DE.
3.2. Gene Function and Pathway Enrichment Analyses
Gene Ontology-based functional annotation analysis of the light-regulated differentially expressed gene set (Supplementary Figure S2) revealed enrichment for biological processes (BP), such as chlorophyll and cell wall biosynthesis, besides photosystem light reaction pathways and ribosome assembly. Dominant molecular functions (MF) were rRNA binding, cytoskeletal motor activity, oxidoreductase and metallopeptidase activities, translation elongation, and chaperone binding. As expected, chloroplasts, cytoskeleton, ribosomes, peroxisomes, and nucleus were the major cellular component sites of activity.
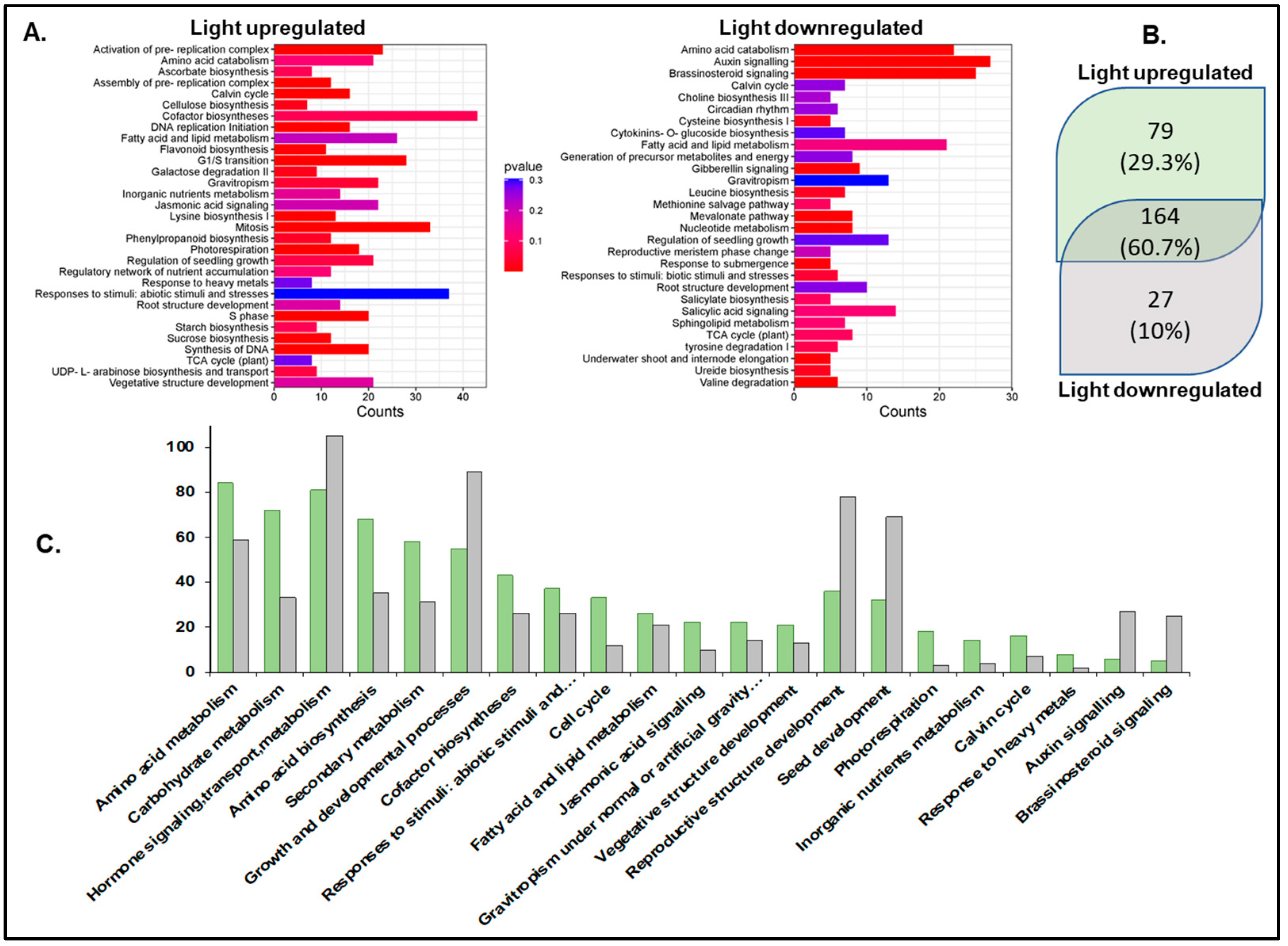
Pathway enrichment analysis using the Plant Reactome pathway analysis tool [65,66] mapped 475 light-upregulated genes to 243 pathways (Supplementary Table S3). A total of 164 pathways overlapped between light-up- and light-downregulated genes. Most pathways showed higher mappings to light-upregulated genes, except hormone auxin and brassinosteroid signaling and reproductive structure development (seed). Pathways unique to light-upregulated genes include those for the biosynthesis of photosynthesis components such as chlorophyll, carotenoid, and phylloquinone, hormones like gibberellin, auxin, abscisic acid, etc. The pathways with unique mapping to light-downregulated genes include polar auxin transport, mevalonate (MVA) pathway, circadian clock, salicylic acid metabolism and signaling, reproductive plant part development, and root-specific gene network of NAC10 transcription factor (Figure 2).
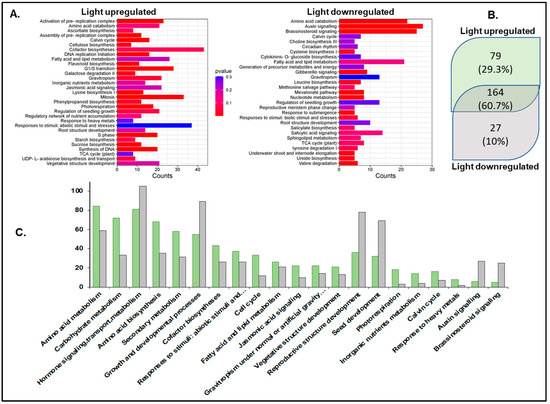
Figure 2.
Pathway enrichment analysis using the Plant Reactome. (A) Plant Reactome pathway enrichment analysis plots. (B) Unique and shared pathways enriched for the light-upregulated and downregulated gene sets. (C) Counts of genes mapped to some of the common Plant Reactome pathways. Light-upregulated (green) and downregulated (grey).
3.3. Identifying Light-Regulated Transcription Factors
To identify the light-regulated transcription factors (TF), a list of rice TFs was downloaded from the Plant Transcription Factor Database [67] and searched against the DE genes. We found 429 light-upregulated and 498 light-downregulated TFs (Supplementary Table S4). We found WRKY, NAC, and orphans were the most abundantly expressed TFs in light, compared to many light-downregulated bHLH, bZIP, and C3H gene family members (Table 1). Highly upregulated (fold change ≥10) TFs belong to the MYB, AP2-EREBP, WRKY, Orphans, NAC, MADS, and bHLH gene families; however, downregulated TFs belong to the AP2-EREBP, C2C2-CO-like, C3H, HB, and NAC gene families (Supplementary Table S4). To investigate whether TFs targeted the MVA and MEP pathway genes, we surveyed the list of TFs and their potential targets identified by the Plant Transcription Factor Database. We identified 23 TFs that potentially bind to the promoter region of six MVA pathway genes. Two MVA pathway genes, hydroxymethylglutaryl-CoA synthase (HMGS; Os08g0544900) and 3-hydroxy-3-methylglutaryl coenzyme A reductase (HMGR; Os08g0512700), were targeted by bZIP and AP2-EREBP factors, respectively, whereas the mevalonate 5-diphosphate decarboxylase (MDD; Os02g0109100) gene was a target of 13 AP2-EREBPs, one C3H protein, and one C2H2 protein (Supplementary Table S5). None of the TFs we found bind to the promoter of MEP pathway genes.

Table 1.
Transcription factor gene families and the number of members showing differential expression in response to light.
3.4. Transcript Splicing during Photomorphogenesis
To study how light exposure modulates transcript splicing (AS) events in the rice seedling shoots, we analyzed the transcriptome data by using SpliceGrapher v0.2.5 [55]. We created highly accurate splice site classifiers for rice with canonical (GT-AG) and semi-canonical (GC-AG) splice sites to filter the splice junctions in the sequence reads aligned to the reference genome. After removing the false positive splice sites, we predicted chromosome-wise splice graphs from the two transcriptome sample datasets. In the dark-treated dataset, 63.9% were true positive junctions, with 5.5% novel splice sites. In the light-treated dataset, 64.3% of splice junctions were true positives, of which 5.8% were predicted as novel sites. We observed 6214 spliced genes with 9685 splicing events in the dark samples compared to the 6809 spliced genes with 10,432 splicing events in the light samples (Figure 1, Table 2). The highest number of spliced genes and events was on chromosome 1, and the lowest was on chromosome 10 (Table 2). Intron retention (IR) was the most prevalent type of splicing event in both samples, followed by exon skipping (ES). Alternative 3′ splicing (Alt.3′) was the least common event. We realigned the sequenced reads from the two samples to the putative transcripts to resolve ambiguous donor and acceptor splice site combinations. We resolved the novel exons to generate splice graphs. Light induced splicing in 2162 unique genes compared to 1567 unique genes that undergo splicing in the dark. Of the 4647 spliced genes shared between the two samples, 165 displayed differential splicing events.

Table 2.
Summary of spliced genes and events under dark and light conditions detected by the SpliceGrapher and the IsoLasso workflows. IR: intron retention; ES: exon skipping; Alt5′: alternative 5′ splice site; Alt3’: alternative 3′ splice site. Color scales: red (large counts) to blue (lower counts).
In our differentially expressed and spliced genes (DES), we found that approximately 30% (4369) differentially expressed (DE) genes also undergo splicing (AS). Approximately 10% (2440) of the non-DE genes undergo splicing (Figure 1). Light-upregulated DES genes show enrichment for various molecular functions like oxidoreductase, hydrolase, isomerase activity, and RNA binding, which play roles in photosynthesis, transmembrane transport, and small molecule metabolic process, and are located in the cellular components chloroplasts and membrane-bound organelles. In contrast, the light-downregulated DES genes are enriched for RNA processing, response to stress, protein binding, and macromolecule metabolic process, and are localized in the cytoplasm and nucleus cellular components (Supplementary Figure S3).
We also surveyed the DES aspects of the 57 known spliced genes in rice [25,68] and observed splicing in 41 genes (Table 3). Of these, seven spliced under dark conditions, three under light conditions, and thirty-one genes spliced under both dark and light conditions. Of the known genes, we also observed that 18 genes were light-upregulated and 24 downregulated. Only 32 genes showed a DES profile. Three light-upregulated genes (Os02g0130600, Os05g0348100, Os12g0567300) show complete splicing in light compared to intron retention in the dark. In contrast, the two light-downregulated genes (Os02g0666200 and Os04g0656100) show intron retention.

Table 3.
Expression and splicing profile of genes in our data that are known to undergo splicing from previous reports.
3.5. Light-Mediated Shift in Gene Expression and Splicing Events
3.5.1. Circadian Clock Pathway Genes
The circadian clock is entrained by light and the diurnal photoperiod-regulated clock genes [69,70]. We investigated the light-regulated DES profile of clock genes. Most of the circadian clock genes spliced under either or both conditions. Four genes, Casein kinase alpha subunit (CK2α-1/Hd6, Os03g0762000), Casein kinase beta subunit (CK2β-2, Os10g0564900), Phytochrome B (PhyB, Os03g0309200), and Timing of CAB Expression 1 (TOC1, Os02g0618200), showed differential splicing (only reference splicing events or complete splicing and no novel isoforms) upon light treatment compared to the presence of an unspliced intron in the absence of light (Supplementary Figure S4). In contrast, only one gene, Phytochrome Interacting Factor 3 (PIF3, Os01g0286100), showed intron retention under light. All five genes were upregulated in light (Supplementary Figure S4). Seven genes, Casein kinase alpha subunit (CK2α-2, Os03g0763000), Casein kinase beta subunit (CK2β-1, Os07g0495100), blue light receptor Cryptochrome 2 (CRY2, Os02g0625000), Gigantea (GI, Os01g0182600), Pseudo-Response Regulator 37 or Heading date 2 (PRR37, Hd2, Os07g0695100), and Pseudo-Response Regulator 73 (PRR73, Os03g0284100), showed differential splicing under both conditions (Supplementary Figures S4 and S5). Exon skipping and alternative 5′ (Alt.5′) splicing events in the CK2α-2 (Os03g0763000) gene were induced in light. Both CK2β-1 (Os07g0495100) and GI (Os01g0182600) displayed incomplete splicing of intron (IR) in the dark. PRR37 showed three types of splicing events (Alt.3′, ES, and IR) under light, whereas the second IR event observed toward the 3’ end under dark was spliced in light. Conversely, PRR73 showed three types of events (Alt.3′, ES, and IR) under dark conditions, whereas under light conditions, two new Alt.5′ events were identified. Expression of both PRR genes (PRR37 and PRR73) was light-downregulated (Supplementary Figure S4). Light-upregulated PRR95 shows an additional unspliced intron in light.
3.5.2. MVA and MEP Pathway Genes
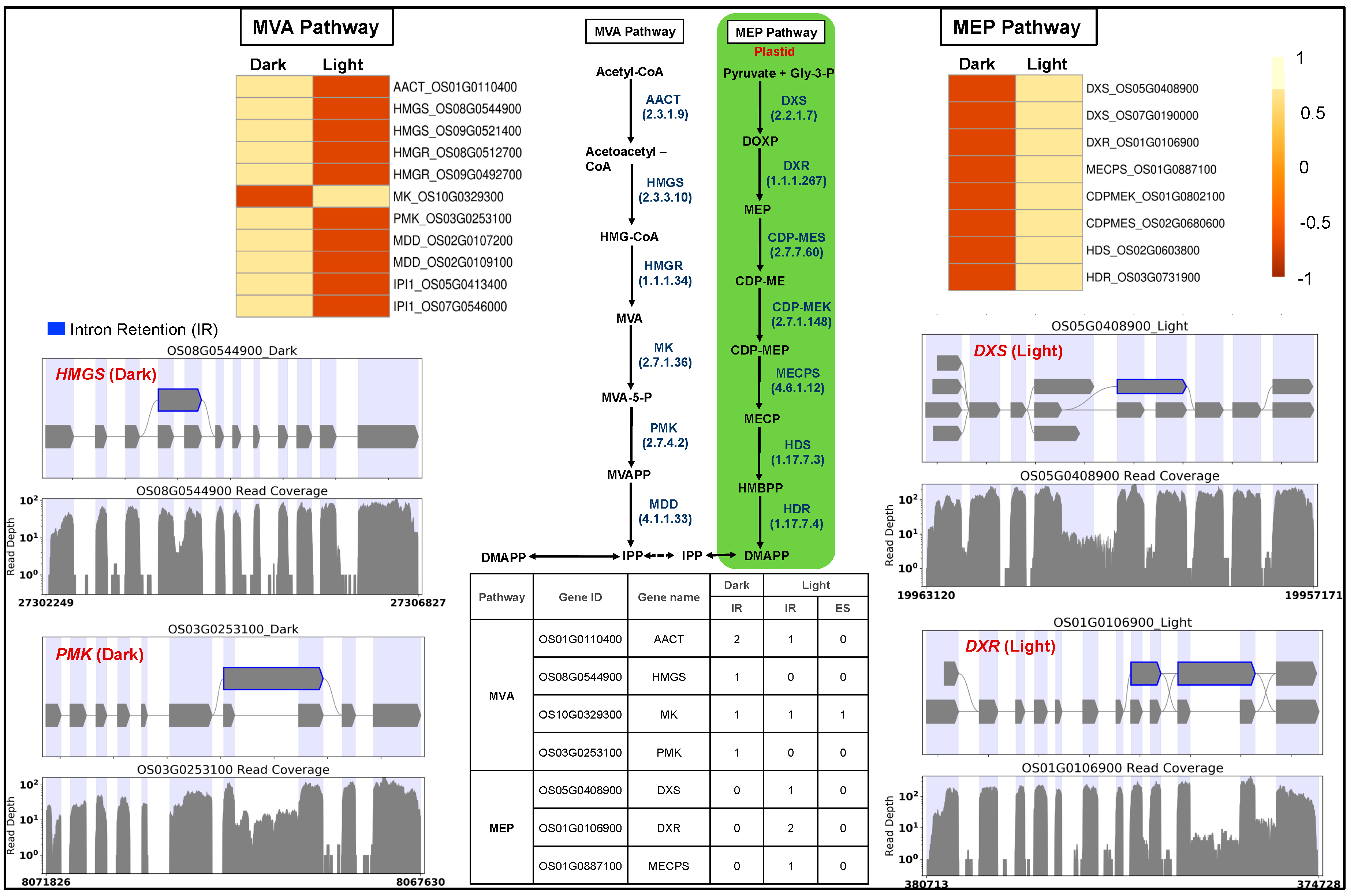
Light regulates the biosynthesis and accumulation of secondary plant products like isoprenoid-derived metabolites required for plant growth and development. Isoprenoid precursors are synthesized via cytosolic MVA and plastid-localized MEP pathways, their fluxes are regulated by light [71,72], and their products are essential for the successful development and function of chloroplast. We observed that MEP pathway genes were light-upregulated compared to the downregulated MVA pathway genes (Figure 3). Light positively regulates MEP pathway activity, whereas MVA pathway activity appears negatively regulated by light [73].
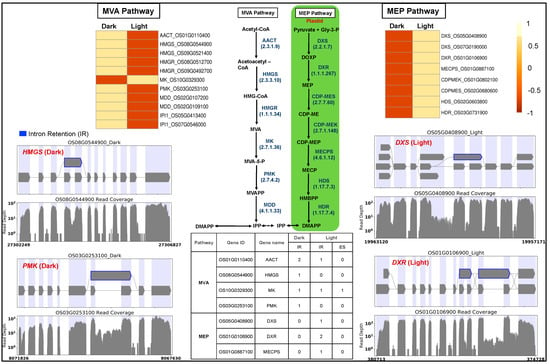
Figure 3.
Gene expression and splicing of cytosolic mevalonate (MVA) and plastid-localized methyl erythritol 4-phosphate (MEP) pathway genes. AACT, acetyl-CoA acetyltransferase; HMGS, hydroxymethyl glutaryl CoA synthase; HMGR, 3-hydroxy-3-methylglutaryl-coenzyme A reductase; MK, mevalonate kinase; PMK, phosphomevalonate kinase; MDD, mevalonate diphosphate decarboxylase; DXS, 1-deoxy-D-xylulose-5-phosphate synthase; DXR, 1-deoxy-D-xylulose-5-phosphate reductoisomerase; CDPMES, 2-C-methyl-D-erythritol4-phosphate cytidylyl transferase; CDPMEK, 4-diphosphocytidyl-2-C-methyl-D-erythritol kinase; MECPS, 2-C-methyl-D-erythritol2,4-cyclodiphosphate synthase; HDS, 4-hydroxy-3-methylbut-2-enyldiphosphate synthase; HDR, 4-hydroxy-3-methylbut-2-enyldiphosphate reductase. Blue: intron retention.
We investigated the light-induced differential splicing events in the MEP and MVA pathway genes. Four MVA pathway genes (acetyl-CoA acetyltransferase: AACT, hydroxymethyl glutaryl CoA synthase: HMGS, mevalonate kinase: MK, phosphomevalonate kinase: PMK) and three MEP pathway genes (1-deoxy-D-xylulose-5-phosphate synthase: DXS, 1-deoxy-D-xylulose-5-phosphate reductoisomerase: DXR, 2-C-methyl-D-erythritol2,4-cyclodiphosphate synthase: MECPS) showed intron retention events in the dark (Figure 3). The gene MK showed a novel light-induced exon-skipping event.
3.6. lncRNA Discovery and Splicing during Photomorphogenesis
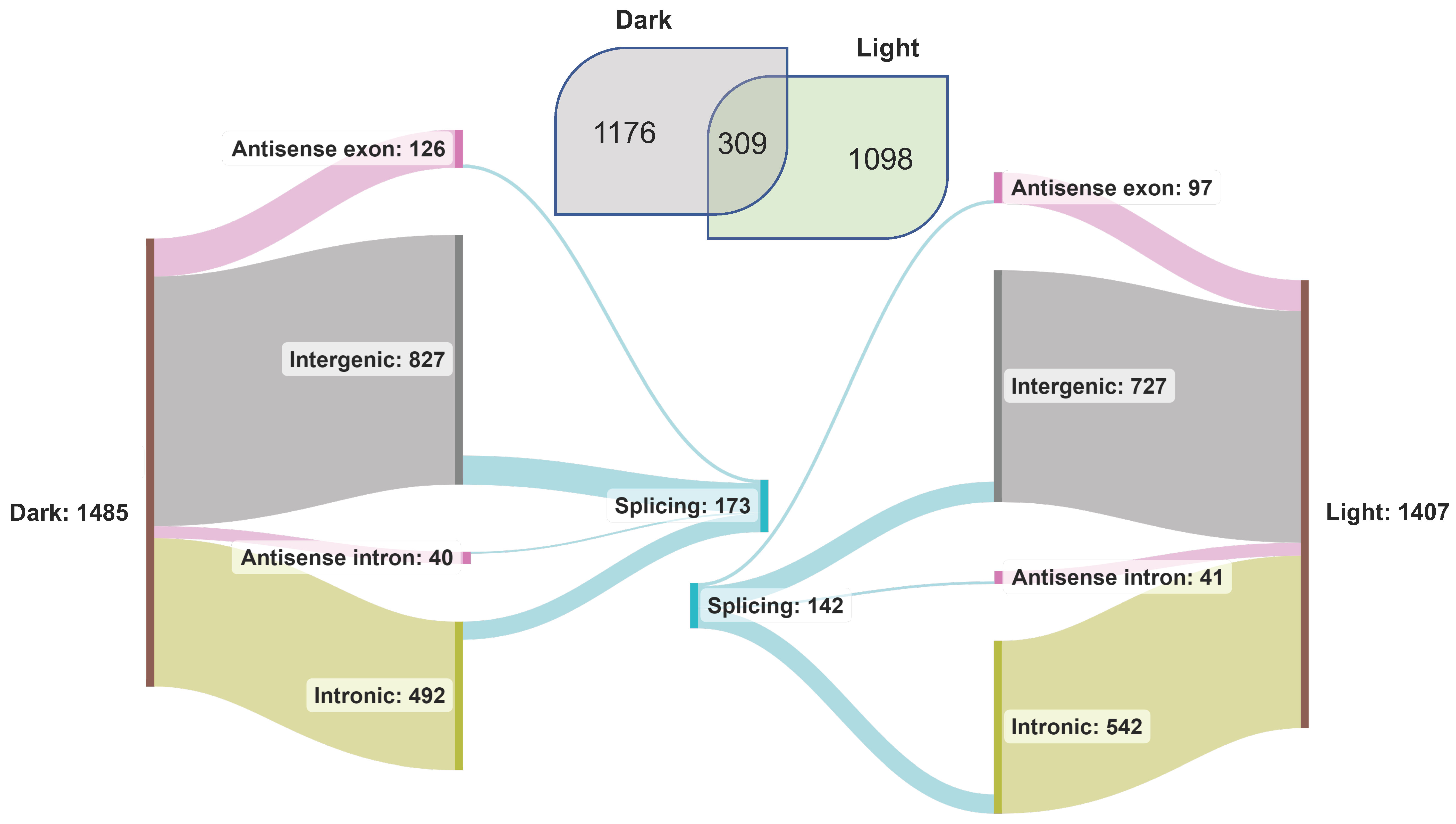
lncRNAs are known to regulate diverse biological processes in plants, including photomorphogenesis [74], and as described above, are categorized into four major types based on their location in the gene space and on the DNA strand. We used our transcriptome data to characterize light-induced rice lncRNAs (Figure 4). We identified 1485 and 1407 lncRNA transcripts in the dark and light samples, of which 309 were common. Many lncRNAs from intergenic and intronic regions were identified (Figure 4). To discover novel lncRNAs, the transcripts from our datasets were queried against the custom BLAST database of rice lncRNAs. About 44% of the total lncRNA transcripts (2583) found a match in the BLAST database (Supplementary Table S6). A total of 173 lncRNAs spliced under dark and 142 spliced under light conditions, most located in the intergenic regions (Figure 4). Interestingly, all the spliced lncRNA genes from both datasets were from Chr1, Chr10, Chr11, and Chr12. The maximum number of spliced lncRNA genes was from Chr1, followed by Chr11, 12, and 10. Among all the splicing events, IR was predominant under both conditions.
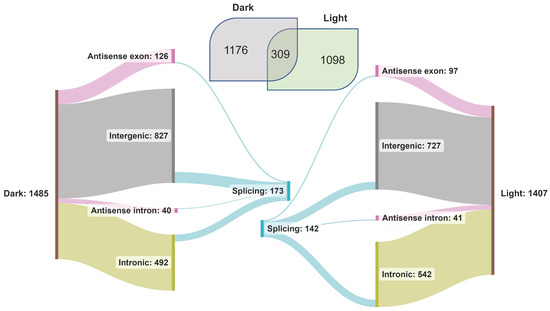
Figure 4.
Classification of lncRNAs identified in the transcriptome of dark- and light-treated rice seedling samples.
We found lncRNA transcripts from light (TCONS_00012152) and dark (TCONS_00011794) datasets that were present on the antisense strand overlapping an exon region of the MADS27 (Os02g0579600) gene. The transcript from the light-treated sample appears longer and showed two alternative 3’ splice sites and one exon skipping event. The TCONS_00011794 may compete with MADS27 to form a complex with miRNA osa-miR444a known to play multiple roles in the nitrate-dependent development pathway [75]. Among the list of lncRNAs present on the antisense strand overlapping annotated genes, we identified a light-induced lncRNA (TCONS_00001515) overlapping ERF99 transcription factor (Os01g0868000) exon that may play a role in its silencing. ERF99 plays a central role in mediating abiotic stress responses [76]. Similarly, a lncRNA transcript (TCONS_00015825) is present exclusively in light and is transcribed from the intronic region of the antisense strand of a B-type response regulator gene (Os03g0224200), known for its involvement in cytokinin signaling, meristem maintenance, and stress response [77,78]. The lncRNA transcript TCONS_00026617, present exclusively in the dark, overlaps the intronic region of a phytochrome-interacting factor gene PIF14 (Os07g0143200). PIF14 is known to bind the active form of phytochrome B and plays a crucial role in cross-talk between light and stress signaling [79].
4. Discussion
Light is known to induce changes in the transcriptome, metabolome, and proteome of plants [80], and thereby not only regulates the development, function, and physiology of the chloroplast but also provides signals for modulating the plant’s morphological, developmental, and physiological adaptions in response to growth environments [7,45,81,82,83]. Though plants experience constantly changing light conditions under the natural environment daily during their lifetime, early development from germination to the seedling stage and acquiring the full autotrophic capability is very much programmed by exposure to light. Therefore, we investigated transcriptome modulation in rice seedlings during photomorphogenesis. Compared to earlier studies in rice and Arabidopsis, where ~20% of the genes were reported to be differentially expressed in dark-grown etiolated seedlings compared to light-exposed green seedlings [6,8,84], we observed that the transition from skotomorphogenesis to photomorphogenesis alters the differential expression of ~38% of the rice genes.
Functional annotation of the differentially expressed genes showed enrichment for roles in secondary metabolism, chloroplast-related biosynthetic pathways, hormone biosynthesis and signaling, amino acid biosynthesis, fatty acid metabolism, etc. (Figure 2, Supplementary Table S3). This result was expected based on the earlier reports on light regulation of such events [24,85].
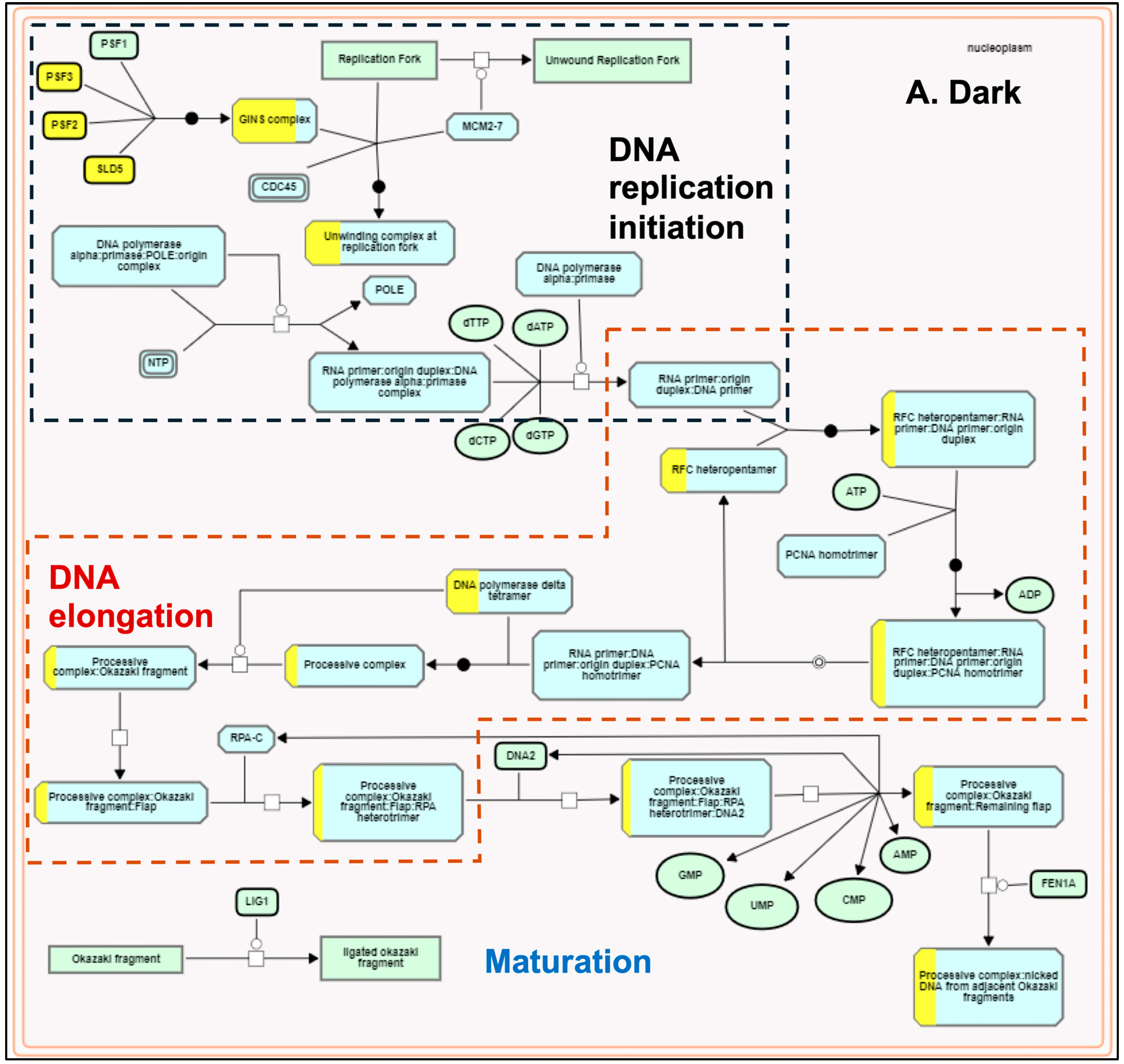
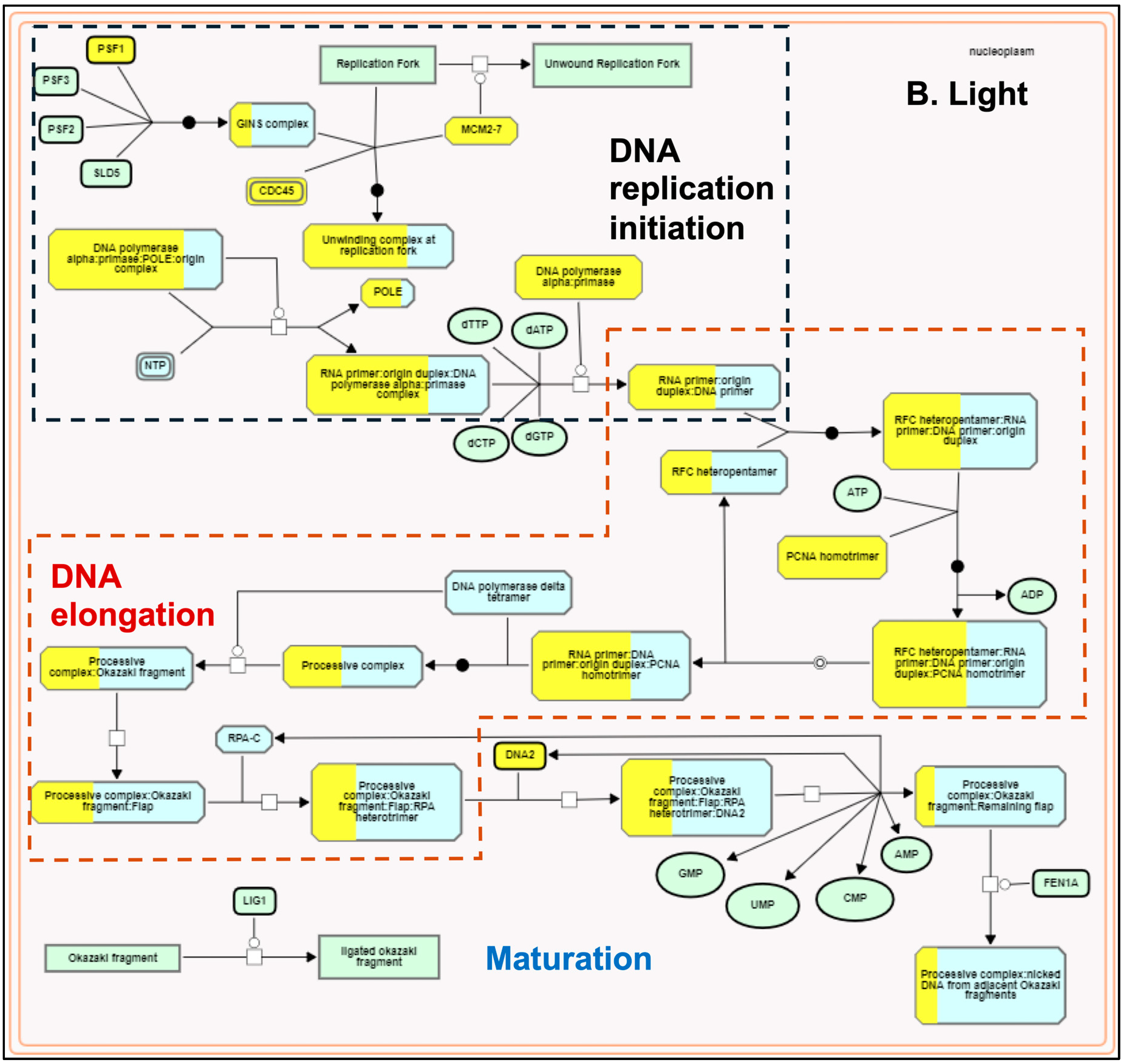
The plastids develop into chloroplasts by following an essential step of developing the thylakoid membrane system and recruiting and assembling components of light and dark reactions to establish a functional photosynthetic process. Many terpenoid compounds are essential to plants’ light-harvesting function and protect against reactive oxygen species (ROS) damage. The basic isoprenoid units for terpenoid biosynthesis, such as tocopherols, plastoquinones, carotenoids, chlorophylls, and precursors of the growth hormones gibberellins and abscisic acid, are synthesized by the plastid-localized MEP pathway [86,87]. At the same time, isoprenoid units synthesized by the MVA pathway contribute to the synthesis of triterpenes, phytosterols, and phytohormones [88]. MEP and MVA pathways complement and contribute to the biosynthesis of chlorophylls and carotenoids required for plastid development [89]. MEP pathway genes were light-upregulated in our transcriptome data compared to the light-downregulated MVA pathway genes (Figure 2). These results are consistent with earlier reports [90,91] and confirm that light acts as a critical regulator in modulating the availability of isoprenoid precursors during photomorphogenesis [92]. Arabidopsis circadian clock genes (LHY, PRR9, CCA1, TOC1) regulate MVA and MEP pathways [73]. AtTOC1 is also known to regulate the G1-to-S phase transition in the mitotic cell cycle in early leaf development [93]. In our dataset, we did not observe any significant change in the expression of LHY/CCA1 (Os08g0157600), PRR95 (Os09g0532400, a homolog of AtPRR9), and TOC1 (Os02g0618200). However, all three genes spliced differently. Under the light, TOC1 showed no splicing, but it retained two introns in the dark. We observed one exon-skipping event of the LHY/CCA1 gene in light, whereas PRR95 showed one additional intron retention (Supplementary Figure S4). Similar to Arabidopsis [93], the functional enrichment analysis also identified the TOC1-regulated DNA biosynthesis pathway of the G1-to-S transition of the mitotic cell cycle (Figure 5) along with a greater coverage of reaction events in the light-upregulated set. To our knowledge, this is the first report on the light regulation of the mitotic cell cycle pathway in rice.
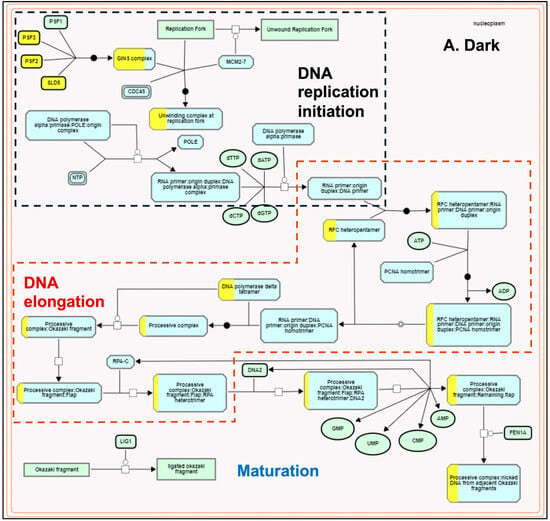
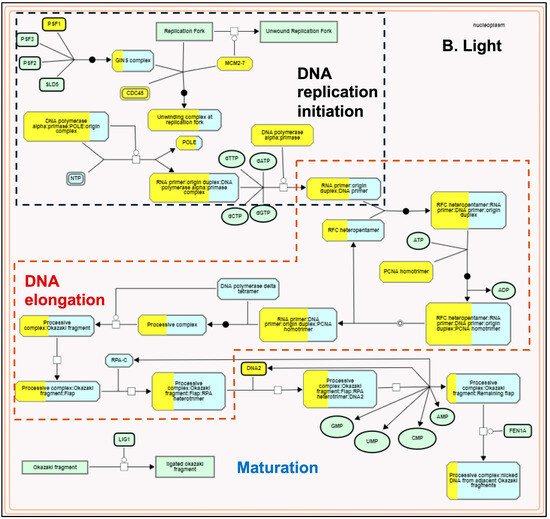
Figure 5.
Plant Reactome pathway enrichment showing light-regulated genes and reaction event coverage for the DNA biosynthesis pathway of the G1-to-S phase mitotic cell cycle. (A) shows enrichment in dark conditions and (B) shows enrichment in light conditions. Each reaction has several input and output entities and may be regulated or catalyzed by a protein and or a protein complex. Solid arrows depict the directionality. The view also shows overlaps between the sub-pathways. The boxes with yellow-painted areas show the gene coverage (the number of genes mapped to the protein or protein complex entity participating in the respective reaction).
Previous transcriptome studies showed that ~42% of Arabidopsis, ~63% of soybean, and ~56% of maize genes undergo alternative splicing events [92,94,95]. Studies on rice by Wang and Brendel [25] reported IR for 69% of 6568 AS genes, and Campbell et al. [96] reported 44.7% of 8772 AS genes. In our dataset, 4369 genes undergo differential expression and alternative splicing, called DES genes (Figure 1A). About 80% (5395) of the spliced genes (6809) show IR events, suggesting that IR is the major splicing event. The more significant number of splicing events in the light-treated samples suggests that light-mediated gene expression and post-transcriptional mRNA splicing play an important role in photomorphogenesis. The splicing events in rice circadian clock genes suggest that IR regulates the expression of PRR genes (Supplementary Figure S4). Many of the light-regulated genes of the MEP and MVA pathways show IR events, suggesting that light plays a positive role in completing the splicing and provides a hypothesis that exposure to light is an adaptive feature and, even if the gene is expressed in dark, it stays unspliced until it encounters light. Our data show the presence of all three states, partially spliced in the dark (GI, Os01g0182600), fully spliced in light (GI, Os01g0182600), and both states in light (CK2β-1, Os07g0495100) (Supplementary Figure S5).
lncRNAs play regulatory functions in essential biological processes such as vernalization, photomorphogenesis, and stress regulation [27,41,97], and display splicing [98]. A total of 6480 long intergenic non-coding RNAs (lincRNAs) were identified from 200 Arabidopsis transcriptome datasets [99]; however, we identified 827 lincRNAs in the dark and 727 lincRNAs in the light datasets. Corona-Gomez and coworkers [100] characterized the splicing conservation of lncRNAs in Brassicaceae, which revealed that ~18% of lincRNAs display splicing conservation in at least one exon. In our dataset, ~10% of lncRNA transcripts undergo splicing, and the majority are of long intergenic non-coding RNA type (Figure 4). Komiya and colleagues reported [101] that phased small interfering RNAs (phasiRNAs) are generated from over 700 lincRNAs, and these phasiRNAs bind to rice argonaute protein MEL1. MEL1 has a specific function in developing pre-meiotic germ cells and the progression of meiosis. In our dataset, many lincRNAs transcribed from MEL1-phasiRNA clusters are apparent.
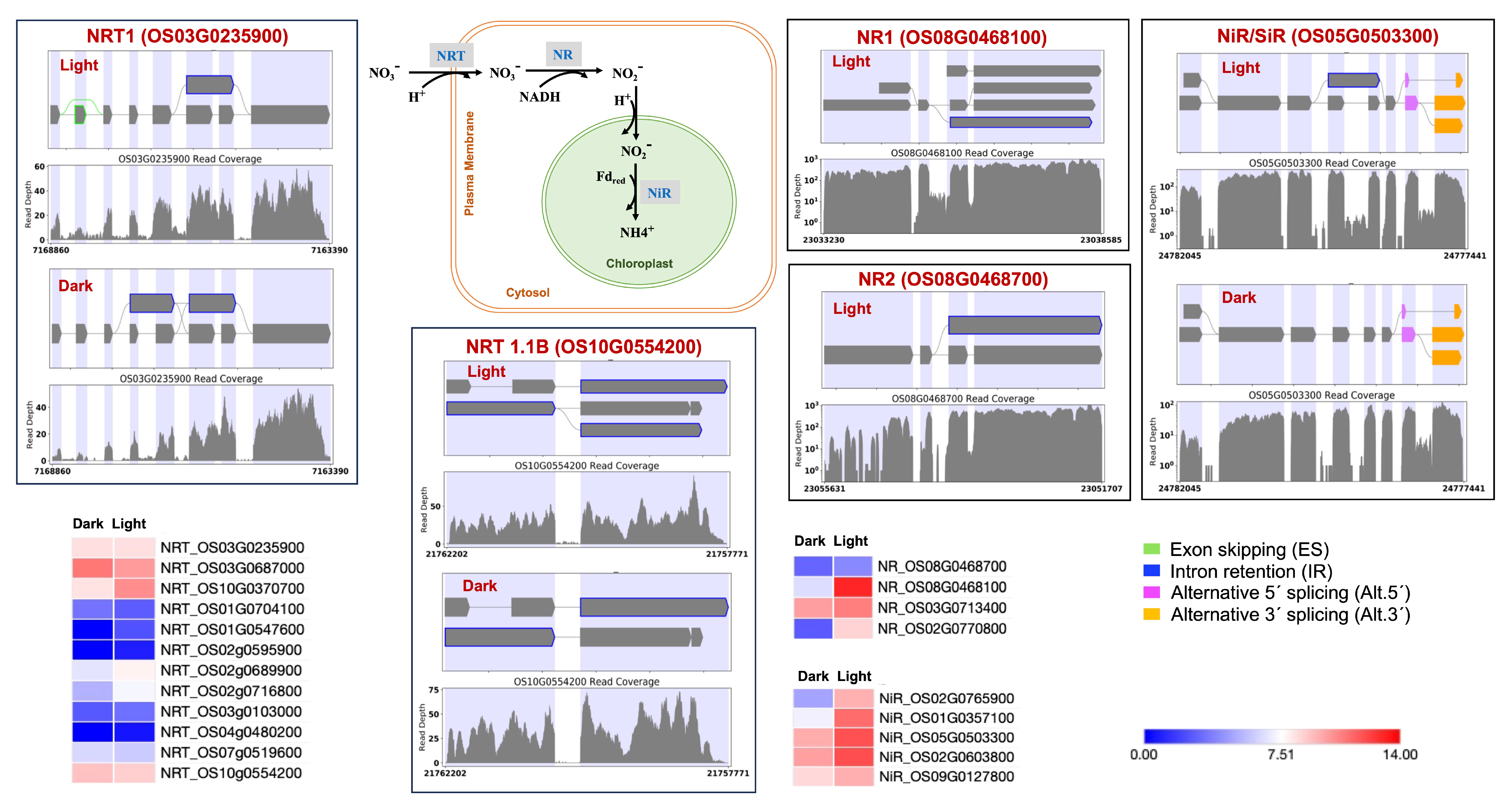
Identification of light-induced lncRNAs antisense to ERF99, B-type response regulator genic region may play a role in transcriptional regulation. ERF99 is known to modulate root architecture and to be downregulated in crown root primordia [102]. The MADS27 gene is targeted by lncRNAs present in both dark and light conditions. However, the dark lncRNA (TCONS_00011794) is longer and, based on its predicted RNA fold structure and lower free energy, is likely more stable than its transcript TCONS_00012152 in light. For the MADS27 gene, we also observed an additional IR event in light. The MADS27-miR444 complex is known to play a role in plant development in a nitrate-dependent manner [103,104,105]. Therefore, we investigated the nitrogen assimilation pathway genes. Nitrogen assimilation is necessary for sustaining plant growth and development. Various nitrogen assimilatory enzymes are known to show isoform and cellular component-specific responses under light and dark conditions [106,107]. We observed light upregulation for the nitrate transporter, nitrate reductase, and nitrite reductase genes. We also observed differential splicing patterns for these genes (Figure 6). We also found light regulation of the recently reported rice abiotic and biotic stress response genes [108,109]. We expect this study’s condition-dependent novel splice events will help improve the annotation of the rice reference genome and pangenome [54,110].
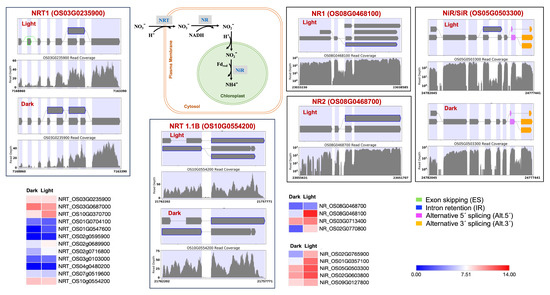
Figure 6.
Splicing and expression pattern of nitrogen assimilation cycle genes under dark and light conditions. NRT: Nitrate transporter; NR: Nitrate reductase; NiR/SiR: Nitrite reductase/Sulfite reductase.
5. Conclusions
This study suggests that light is a significant regulatory factor controlling genome-wide gene expression through an alternative splicing mechanism in rice. All spliced genes did not necessarily produce novel isoforms, which indicates the coupling of AS and nonsense-mediated decay (NMD). NMD prevents the translation of mutant mRNAs harboring potential premature stop codons by targeting them for degradation. In Arabidopsis, 77.2% of light-regulated AS events exhibit NMD features within a splicing isoform [111]. We conclude that light induces a significant number of splicing events in rice protein-coding and non-coding lncRNA genes. This photomorphogenesis transcriptome study is a valuable resource for lncRNA research in rice and provides insights into the portion of the genome regulated at the level of alternative splicing in response to light.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/genes15081072/s1, Figure S1: Expression pattern of differentially expressed genes (14,766) with target FDR controlled at 5%. Figure S2: Gene Ontology enrichment analysis of differentially expressed genes. Figure S3: Bar plots of most significantly enriched GO terms for DES genes with -log10-transformed FDR values. Light-upregulated (green), light-downregulated (grey). Figure S4: Splicing events and expression pattern of circadian clock genes under dark and light conditions. Figure S5: Transcript graphs showing splicing of Casein kinase beta subunit:CK2β-1 (Os07g0495100), Gigantea:GI (Os01g0182600) genes during photomorphogenesis. Table S1: Summary of the RNA-Seq read mapping to the reference rice genome. Table S2: Differentially expressed genes. Table S3: Plant Reactome Pathway enrichment analysis for light-regulated genes. Table S4: Transcription factor genes regulated in light. Table S5: Transcription factors regulating MVA pathway genes. Table S6: lncRNA annotation using BLAST.
Author Contributions
Conceptualization, P.J.; Methodology, P.J. and P.G.; Data analysis, P.G.; Writing, review, and editing, P.G. and P.J. All authors have read and agreed to the published version of the manuscript.
Funding
This work is supported by startup funds provided to P.J. by the Department of Botany and Plant Pathology in the College of Agricultural Sciences at Oregon State University. P.G. and P.J. are also supported by the National Science Foundation of USA awards (IOS-1127112, IOS-1340112, and DBI-2029854). This material is based upon the work supported by (while serving at) the National Science Foundation.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The sequence reads and metadata are accessible from EMBL-EBI ArrayExpress (accession number E-MTAB-5689). The analyzed dataset is available at Gupta and Jaiswal, 2024 [112].
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Kami, C.; Lorrain, S.; Hornitschek, P.; Fankhauser, C. Chapter Two—Light-Regulated Plant Growth and Development. In Current Topics in Developmental Biology; Timmermans, M.C.P., Ed.; Plant Development; Academic Press: Cambridge, MA, USA, 2010; Volume 91, pp. 29–66. [Google Scholar]
- Poolman, M.G.; Kundu, S.; Shaw, R.; Fell, D.A. Responses to Light Intensity in a Genome-Scale Model of Rice Metabolism. Plant Physiol. 2013, 162, 1060–1072. [Google Scholar] [CrossRef]
- Naithani, S.; Nonogaki, H.; Jaiswal, P. Exploring Crossroads Between Seed Development and Stress Response. In Mechanism of Plant Hormone Signaling under Stress; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2017; pp. 415–454. ISBN 978-1-118-88902-2. [Google Scholar]
- Li, H.; Liang, Z.; Ding, G.; Shi, L.; Xu, F.; Cai, H. A Natural Light/Dark Cycle Regulation of Carbon-Nitrogen Metabolism and Gene Expression in Rice Shoots. Front. Plant Sci. 2016, 7, 1318. [Google Scholar] [CrossRef] [PubMed]
- Tobin, E.M.; Silverthorne, J. Light Regulation of Gene Expression in Higher Plants. Annu. Rev. Plant Physiol. 1985, 36, 569–593. [Google Scholar] [CrossRef]
- Ma, L.; Li, J.; Qu, L.; Hager, J.; Chen, Z.; Zhao, H.; Deng, X.W. Light Control of Arabidopsis Development Entails Coordinated Regulation of Genome Expression and Cellular Pathways. Plant Cell 2001, 13, 2589–2608. [Google Scholar] [CrossRef] [PubMed]
- Casal, J.J.; Yanovsky, M.J. Regulation of Gene Expression by Light. Int. J. Dev. Biol. 2004, 49, 501–511. [Google Scholar] [CrossRef]
- Jiao, Y.; Ma, L.; Strickland, E.; Deng, X.W. Conservation and Divergence of Light-Regulated Genome Expression Patterns during Seedling Development in Rice and Arabidopsis. Plant Cell 2005, 17, 3239–3256. [Google Scholar] [CrossRef]
- Majláth, I.; Szalai, G.; Soós, V.; Sebestyén, E.; Balázs, E.; Vanková, R.; Dobrev, P.I.; Tari, I.; Tandori, J.; Janda, T. Effect of Light on the Gene Expression and Hormonal Status of Winter and Spring Wheat Plants during Cold Hardening. Physiol. Plant. 2012, 145, 296–314. [Google Scholar] [CrossRef]
- Petrillo, E.; Herz, M.A.G.; Barta, A.; Kalyna, M.; Kornblihtt, A.R. Let There Be Light: Regulation of Gene Expression in Plants. RNA Biol. 2014, 11, 1215–1220. [Google Scholar] [CrossRef] [PubMed]
- Godoy Herz, M.A.; Kubaczka, M.G.; Brzyżek, G.; Servi, L.; Krzyszton, M.; Simpson, C.; Brown, J.; Swiezewski, S.; Petrillo, E.; Kornblihtt, A.R. Light Regulates Plant Alternative Splicing through the Control of Transcriptional Elongation. Mol. Cell 2019, 73, 1066–1074.e3. [Google Scholar] [CrossRef]
- Bai, S.; Tao, R.; Tang, Y.; Yin, L.; Ma, Y.; Ni, J.; Yan, X.; Yang, Q.; Wu, Z.; Zeng, Y.; et al. BBX16, a B-Box Protein, Positively Regulates Light-Induced Anthocyanin Accumulation by Activating MYB10 in Red Pear. Plant Biotechnol. J. 2019, 17, 1985–1997. [Google Scholar] [CrossRef] [PubMed]
- Quail, P.H. Phytochrome Photosensory Signalling Networks. Nat. Rev. Mol. Cell Biol. 2002, 3, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Shalitin, D. Cryptochrome Structure and Signal Transduction. Annu. Rev. Plant Biol. 2003, 54, 469–496. [Google Scholar] [CrossRef]
- Huang, X.; Ouyang, X.; Deng, X.W. Beyond Repression of Photomorphogenesis: Role Switching of COP/DET/FUS in Light Signaling. Curr. Opin. Plant Biol. 2014, 21, 96–103. [Google Scholar] [CrossRef]
- Sheerin, D.J.; Menon, C.; zur Oven-Krockhaus, S.; Enderle, B.; Zhu, L.; Johnen, P.; Schleifenbaum, F.; Stierhof, Y.-D.; Huq, E.; Hiltbrunner, A. Light-Activated Phytochrome A and B Interact with Members of the SPA Family to Promote Photomorphogenesis in Arabidopsis by Reorganizing the COP1/SPA Complex. Plant Cell 2015, 27, 189–201. [Google Scholar] [CrossRef] [PubMed]
- Leivar, P.; Monte, E.; Oka, Y.; Liu, T.; Carle, C.; Castillon, A.; Huq, E.; Quail, P.H. Multiple Phytochrome-Interacting bHLH Transcription Factors Repress Premature Photomorphogenesis during Early Seedling Development in Darkness. Curr. Biol. 2008, 18, 1815–1823. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.; Kim, K.; Kang, H.; Zulfugarov, I.S.; Bae, G.; Lee, C.-H.; Lee, D.; Choi, G. Phytochromes Promote Seedling Light Responses by Inhibiting Four Negatively-Acting Phytochrome-Interacting Factors. Proc. Natl. Acad. Sci. USA 2009, 106, 7660–7665. [Google Scholar] [CrossRef]
- Xie, C.; Zhang, G.; An, L.; Chen, X.; Fang, R. Phytochrome-Interacting Factor-like Protein OsPIL15 Integrates Light and Gravitropism to Regulate Tiller Angle in Rice. Planta 2019, 250, 105–114. [Google Scholar] [CrossRef]
- Liu, M.-J.; Wu, S.-H.; Chen, H.-M.; Wu, S.-H. Widespread Translational Control Contributes to the Regulation of Arabidopsis Photomorphogenesis. Mol. Syst. Biol. 2012, 8, 566. [Google Scholar] [CrossRef]
- Kianianmomeni, A. More Light behind Gene Expression. Trends Plant Sci. 2014, 19, 488–490. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.-P.; Su, Y.; Chen, H.-C.; Chen, Y.-R.; Wu, C.-C.; Lin, W.-D.; Tu, S.-L. Genome-Wide Analysis of Light-Regulated Alternative Splicing Mediated by Photoreceptors in Physcomitrella Patens. Genome Biol. 2014, 15, R10. [Google Scholar] [CrossRef]
- Shikata, H.; Hanada, K.; Ushijima, T.; Nakashima, M.; Suzuki, Y.; Matsushita, T. Phytochrome Controls Alternative Splicing to Mediate Light Responses in Arabidopsis. Proc. Natl. Acad. Sci. USA 2014, 111, 18781–18786. [Google Scholar] [CrossRef] [PubMed]
- Mancini, E.; Sanchez, S.E.; Romanowski, A.; Schlaen, R.G.; Sanchez-Lamas, M.; Cerdán, P.D.; Yanovsky, M.J. Acute Effects of Light on Alternative Splicing in Light-Grown Plants. Photochem. Photobiol. 2016, 92, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.-B.; Brendel, V. Genomewide Comparative Analysis of Alternative Splicing in Plants. Proc. Natl. Acad. Sci. USA 2006, 103, 7175–7180. [Google Scholar] [CrossRef] [PubMed]
- Ner-Gaon, H.; Halachmi, R.; Savaldi-Goldstein, S.; Rubin, E.; Ophir, R.; Fluhr, R. Intron Retention Is a Major Phenomenon in Alternative Splicing in Arabidopsis. Plant J. 2004, 39, 877–885. [Google Scholar] [CrossRef]
- Filichkin, S.A.; Hamilton, M.; Dharmawardhana, P.D.; Singh, S.K.; Sullivan, C.; Ben-Hur, A.; Reddy, A.S.N.; Jaiswal, P. Abiotic Stresses Modulate Landscape of Poplar Transcriptome via Alternative Splicing, Differential Intron Retention, and Isoform Ratio Switching. Front. Plant Sci. 2018, 9, 5. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.; Geniza, M.; Elser, J.; Al-Bader, N.; Baschieri, R.; Phillips, J.L.; Haq, E.; Preece, J.; Naithani, S.; Jaiswal, P. Reference Genome of the Nutrition-Rich Orphan Crop Chia (Salvia Hispanica) and Its Implications for Future Breeding. Front. Plant Sci. 2023, 14, 1272966. [Google Scholar] [CrossRef]
- Chaudhary, S.; Khokhar, W.; Jabre, I.; Reddy, A.S.N.; Byrne, L.J.; Wilson, C.M.; Syed, N.H. Alternative Splicing and Protein Diversity: Plants Versus Animals. Front. Plant Sci. 2019, 10, 708. [Google Scholar] [CrossRef]
- Ganie, S.A.; Reddy, A.S.N. Stress-Induced Changes in Alternative Splicing Landscape in Rice: Functional Significance of Splice Isoforms in Stress Tolerance. Biology 2021, 10, 309. [Google Scholar] [CrossRef]
- Gao, P.; Quilichini, T.D.; Zhai, C.; Qin, L.; Nilsen, K.T.; Li, Q.; Sharpe, A.G.; Kochian, L.V.; Zou, J.; Reddy, A.S.N.; et al. Alternative Splicing Dynamics and Evolutionary Divergence during Embryogenesis in Wheat Species. Plant Biotechnol. J. 2021, 19, 1624–1643. [Google Scholar] [CrossRef]
- Mercer, T.R.; Dinger, M.E.; Mattick, J.S. Long Non-Coding RNAs: Insights into Functions. Nat. Rev. Genet. 2009, 10, 155–159. [Google Scholar] [CrossRef]
- Liu, J.; Wang, H.; Chua, N.-H. Long Noncoding RNA Transcriptome of Plants. Plant Biotechnol. J. 2015, 13, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Palos, K.; Yu, L.; Railey, C.E.; Nelson Dittrich, A.C.; Nelson, A.D.L. Linking Discoveries, Mechanisms, and Technologies to Develop a Clearer Perspective on Plant Long Noncoding RNAs. Plant Cell 2023, 35, 1762–1786. [Google Scholar] [CrossRef]
- Wang, X.; Fan, H.; Wang, B.; Yuan, F. Research Progress on the Roles of lncRNAs in Plant Development and Stress Responses. Front. Plant Sci. 2023, 14, 1138901. [Google Scholar] [CrossRef]
- Liu, H.; Wang, R.; Mao, B.; Zhao, B.; Wang, J. Identification of lncRNAs Involved in Rice Ovule Development and Female Gametophyte Abortion by Genome-Wide Screening and Functional Analysis. BMC Genom. 2019, 20, 90. [Google Scholar] [CrossRef]
- Zhang, Y.-C.; Liao, J.-Y.; Li, Z.-Y.; Yu, Y.; Zhang, J.-P.; Li, Q.-F.; Qu, L.-H.; Shu, W.-S.; Chen, Y.-Q. Genome-Wide Screening and Functional Analysis Identify a Large Number of Long Noncoding RNAs Involved in the Sexual Reproduction of Rice. Genome Biol. 2014, 15, 512. [Google Scholar] [CrossRef]
- Chen, L.; Shi, S.; Jiang, N.; Khanzada, H.; Wassan, G.M.; Zhu, C.; Peng, X.; Xu, J.; Chen, Y.; Yu, Q.; et al. Genome-Wide Analysis of Long Non-Coding RNAs Affecting Roots Development at an Early Stage in the Rice Response to Cadmium Stress. BMC Genom. 2018, 19, 460. [Google Scholar] [CrossRef] [PubMed]
- Li, W.Q.; Jia, Y.L.; Liu, F.Q.; Wang, F.Q.; Fan, F.J.; Wang, J.; Zhu, J.Y.; Xu, Y.; Zhong, W.G.; Yang, J. Genome-Wide Identification and Characterization of Long Non-Coding RNAs Responsive to Dickeya zeae in Rice. RSC Adv. 2018, 8, 34408–34417. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Shahid, M.Q.; Wen, M.; Chen, S.; Yu, H.; Jiao, Y.; Lu, Z.; Li, Y.; Liu, X. Global Identification and Analysis Revealed Differentially Expressed lncRNAs Associated with Meiosis and Low Fertility in Autotetraploid Rice. BMC Plant Biol. 2020, 20, 82. [Google Scholar] [CrossRef]
- Wang, Y.; Fan, X.; Lin, F.; He, G.; Terzaghi, W.; Zhu, D.; Deng, X.W. Arabidopsis Noncoding RNA Mediates Control of Photomorphogenesis by Red Light. Proc. Natl. Acad. Sci. USA 2014, 111, 10359–10364. [Google Scholar] [CrossRef]
- Martín, G.; Márquez, Y.; Mantica, F.; Duque, P.; Irimia, M. Alternative Splicing Landscapes in Arabidopsis Thaliana across Tissues and Stress Conditions Highlight Major Functional Differences with Animals. Genome Biol. 2021, 22, 35. [Google Scholar] [CrossRef]
- Gupta, P.; Geniza, M.; Naithani, S.; Phillips, J.L.; Haq, E.; Jaiswal, P. Chia (Salvia hispanica) Gene Expression Atlas Elucidates Dynamic Spatio-Temporal Changes Associated with Plant Growth and Development. Front. Plant Sci. 2021, 12, 667678. [Google Scholar] [CrossRef] [PubMed]
- Reddy, A.S.N.; Marquez, Y.; Kalyna, M.; Barta, A. Complexity of the Alternative Splicing Landscape in Plants[C][W][OPEN]. Plant Cell 2013, 25, 3657–3683. [Google Scholar] [CrossRef]
- Fox, S.E.; Geniza, M.; Hanumappa, M.; Naithani, S.; Sullivan, C.; Preece, J.; Tiwari, V.K.; Elser, J.; Leonard, J.M.; Sage, A.; et al. De Novo Transcriptome Assembly and Analyses of Gene Expression during Photomorphogenesis in Diploid Wheat Triticum Monococcum. PLoS ONE 2014, 9, e96855. [Google Scholar] [CrossRef]
- Najoshi Najoshi/Sickle. 2020. Available online: https://github.com/najoshi/sickle (accessed on 16 August 2023).
- Trapnell, C.; Pachter, L.; Salzberg, S.L. TopHat: Discovering Splice Junctions with RNA-Seq. Bioinformatics 2009, 25, 1105–1111. [Google Scholar] [CrossRef] [PubMed]
- Trapnell, C.; Roberts, A.; Goff, L.; Pertea, G.; Kim, D.; Kelley, D.R.; Pimentel, H.; Salzberg, S.L.; Rinn, J.L.; Pachter, L. Differential Gene and Transcript Expression Analysis of RNA-Seq Experiments with TopHat and Cufflinks. Nat. Protoc. 2012, 7, 562–578. [Google Scholar] [CrossRef]
- Li, B.; Dewey, C.N. RSEM: Accurate Transcript Quantification from RNA-Seq Data with or without a Reference Genome. BMC Bioinform. 2011, 12, 323. [Google Scholar] [CrossRef] [PubMed]
- Leng, N.; Dawson, J.A.; Thomson, J.A.; Ruotti, V.; Rissman, A.I.; Smits, B.M.G.; Haag, J.D.; Gould, M.N.; Stewart, R.M.; Kendziorski, C. EBSeq: An Empirical Bayes Hierarchical Model for Inference in RNA-Seq Experiments. Bioinformatics 2013, 29, 1035–1043. [Google Scholar] [CrossRef]
- The Gene Ontology Consortium. The Gene Ontology Resource: 20 Years and Still GOing Strong. Nucleic Acids Res. 2019, 47, D330–D338. [Google Scholar] [CrossRef]
- Naithani, S.; Preece, J.; D’Eustachio, P.; Gupta, P.; Amarasinghe, V.; Dharmawardhana, P.D.; Wu, G.; Fabregat, A.; Elser, J.L.; Weiser, J.; et al. Plant Reactome: A Resource for Plant Pathways and Comparative Analysis. Nucleic Acids Res. 2017, 45, D1029–D1039. [Google Scholar] [CrossRef]
- Gupta, P.; Naithani, S.; Preece, J.; Kim, S.; Cheng, T.; D’Eustachio, P.; Elser, J.; Bolton, E.E.; Jaiswal, P. Plant Reactome and PubChem: The Plant Pathway and (Bio)Chemical Entity Knowledgebases. Methods Mol. Biol. 2022, 2443, 511–525. [Google Scholar] [CrossRef]
- Tello-Ruiz, M.K.; Naithani, S.; Gupta, P.; Olson, A.; Wei, S.; Preece, J.; Jiao, Y.; Wang, B.; Chougule, K.; Garg, P.; et al. Gramene 2021: Harnessing the Power of Comparative Genomics and Pathways for Plant Research. Nucleic Acids Res 2021, 49, D1452–D1463. [Google Scholar] [CrossRef] [PubMed]
- Rogers, M.F.; Thomas, J.; Reddy, A.S.; Ben-Hur, A. SpliceGrapher: Detecting Patterns of Alternative Splicing from RNA-Seq Data in the Context of Gene Models and EST Data. Genome Biol. 2012, 13, R4. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Feng, J.; Jiang, T. IsoLasso: A LASSO Regression Approach to RNA-Seq Based Transcriptome Assembly. J. Comput. Biol. 2011, 18, 1693–1707. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.-J.; Yang, D.-C.; Kong, L.; Hou, M.; Meng, Y.-Q.; Wei, L.; Gao, G. CPC2: A Fast and Accurate Coding Potential Calculator Based on Sequence Intrinsic Features. Nucleic Acids Res. 2017, 45, W12–W16. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.; Binns, D.; Chang, H.-Y.; Fraser, M.; Li, W.; McAnulla, C.; McWilliam, H.; Maslen, J.; Mitchell, A.; Nuka, G.; et al. InterProScan 5: Genome-Scale Protein Function Classification. Bioinformatics 2014, 30, 1236–1240. [Google Scholar] [CrossRef]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic Local Alignment Search Tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Yi, X.; Zhang, Z.; Ling, Y.; Xu, W.; Su, Z. PNRD: A Plant Non-Coding RNA Database. Nucleic Acids Res. 2015, 43, D982–D989. [Google Scholar] [CrossRef] [PubMed]
- Paytuví Gallart, A.; Hermoso Pulido, A.; Anzar Martínez de Lagrán, I.; Sanseverino, W.; Aiese Cigliano, R. GREENC: A Wiki-Based Database of Plant lncRNAs. Nucleic Acids Res. 2016, 44, D1161–D1166. [Google Scholar] [CrossRef]
- Zhang, Z.; Xu, Y.; Yang, F.; Xiao, B.; Li, G. RiceLncPedia: A Comprehensive Database of Rice Long Non-coding RNAs. Plant Biotechnol. J. 2021, 19, 1492–1494. [Google Scholar] [CrossRef]
- Hofacker, I.L. Vienna RNA Secondary Structure Server. Nucleic Acids Res. 2003, 31, 3429–3431. [Google Scholar] [CrossRef]
- Gruber, A.R.; Lorenz, R.; Bernhart, S.H.; Neuböck, R.; Hofacker, I.L. The Vienna RNA Websuite. Nucleic Acids Res. 2008, 36, W70–W74. [Google Scholar] [CrossRef] [PubMed]
- Naithani, S.; Gupta, P.; Preece, J.; D’Eustachio, P.; Elser, J.L.; Garg, P.; Dikeman, D.A.; Kiff, J.; Cook, J.; Olson, A.; et al. Plant Reactome: A Knowledgebase and Resource for Comparative Pathway Analysis. Nucleic Acids Res. 2020, 48, D1093–D1103. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.; Elser, J.; Hooks, E.; D’Eustachio, P.; Jaiswal, P.; Naithani, S. Plant Reactome Knowledgebase: Empowering Plant Pathway Exploration and OMICS Data Analysis. Nucleic Acids Res. 2023, 52, D1538–D1547. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Tian, F.; Yang, D.-C.; Meng, Y.-Q.; Kong, L.; Luo, J.; Gao, G. PlantTFDB 4.0: Toward a Central Hub for Transcription Factors and Regulatory Interactions in Plants. Nucleic Acids Res. 2017, 45, D1040–D1045. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Li, X.; Guo, L.; Lu, F.; Feng, X.; He, K.; Wei, L.; Chen, Z.; Qu, L.-J.; Gu, H. A Subgroup of MYB Transcription Factor Genes Undergoes Highly Conserved Alternative Splicing in Arabidopsis and Rice. J. Exp. Bot. 2006, 57, 1263–1273. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, N.; Izawa, T.; Oikawa, T.; Shimamoto, K. Light Regulation of Circadian Clock-Controlled Gene Expression in Rice. Plant J. 2001, 26, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Matsuzaki, J.; Kawahara, Y.; Izawa, T. Punctual Transcriptional Regulation by the Rice Circadian Clock under Fluctuating Field Conditions[OPEN]. Plant Cell 2015, 27, 633–648. [Google Scholar] [CrossRef] [PubMed]
- Cordoba, E.; Salmi, M.; León, P. Unravelling the Regulatory Mechanisms That Modulate the MEP Pathway in Higher Plants. J. Exp. Bot. 2009, 60, 2933–2943. [Google Scholar] [CrossRef] [PubMed]
- Vranová, E.; Coman, D.; Gruissem, W. Network Analysis of the MVA and MEP Pathways for Isoprenoid Synthesis. Annu. Rev. Plant Biol. 2013, 64, 665–700. [Google Scholar] [CrossRef]
- Vranová, E.; Coman, D.; Gruissem, W. Structure and Dynamics of the Isoprenoid Pathway Network. Mol. Plant 2012, 5, 318–333. [Google Scholar] [CrossRef]
- Liu, X.; Hao, L.; Li, D.; Zhu, L.; Hu, S. Long Non-Coding RNAs and Their Biological Roles in Plants. Genom. Proteom. Bioinform. 2015, 13, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Sanz-Jimenez, P.; Zhu, X.-T.; Feng, J.-W.; Shao, L.; Song, J.-M.; Chen, L.-L. Analysis of Rice Transcriptome Reveals the LncRNA/CircRNA Regulation in Tissue Development. Rice 2021, 14, 14. [Google Scholar] [CrossRef] [PubMed]
- Serra, T.S.; Figueiredo, D.D.; Cordeiro, A.M.; Almeida, D.M.; Lourenço, T.; Abreu, I.A.; Sebastián, A.; Fernandes, L.; Contreras-Moreira, B.; Oliveira, M.M.; et al. OsRMC, a Negative Regulator of Salt Stress Response in Rice, Is Regulated by Two AP2/ERF Transcription Factors. Plant Mol. Biol. 2013, 82, 439–455. [Google Scholar] [CrossRef] [PubMed]
- Argueso, C.T.; Ferreira, F.J.; Kieber, J.J. Environmental Perception Avenues: The Interaction of Cytokinin and Environmental Response Pathways. Plant Cell Environ. 2009, 32, 1147–1160. [Google Scholar] [CrossRef]
- Worthen, J.M.; Yamburenko, M.V.; Lim, J.; Nimchuk, Z.L.; Kieber, J.J.; Schaller, G.E. Type-B Response Regulators of Rice Play Key Roles in Growth, Development and Cytokinin Signaling. Development 2019, 146, dev174870. [Google Scholar] [CrossRef]
- Cordeiro, A.M.; Figueiredo, D.D.; Tepperman, J.; Borba, A.R.; Lourenço, T.; Abreu, I.A.; Ouwerkerk, P.B.F.; Quail, P.H.; Oliveira, M.M.; Saibo, N.J.M. Rice Phytochrome-Interacting Factor Protein OsPIF14 Represses OsDREB1B Gene Expression through an Extended N-Box and Interacts Preferentially with the Active Form of Phytochrome B. Biochim. Biophys. Acta 2016, 1859, 393–404. [Google Scholar] [CrossRef]
- Liang, C.; Cheng, S.; Zhang, Y.; Sun, Y.; Fernie, A.R.; Kang, K.; Panagiotou, G.; Lo, C.; Lim, B.L. Transcriptomic, Proteomic and Metabolic Changes in Arabidopsis Thaliana Leaves after the Onset of Illumination. BMC Plant Biol. 2016, 16, 43. [Google Scholar] [CrossRef]
- Ding, J.; Zhao, J.; Pan, T.; Xi, L.; Zhang, J.; Zou, Z. Comparative Transcriptome Analysis of Gene Expression Patterns in Tomato Under Dynamic Light Conditions. Genes 2019, 10, 662. [Google Scholar] [CrossRef]
- Jin, X.; Baysal, C.; Drapal, M.; Sheng, Y.; Huang, X.; He, W.; Shi, L.; Capell, T.; Fraser, P.D.; Christou, P.; et al. The Coordinated Upregulated Expression of Genes Involved in MEP, Chlorophyll, Carotenoid and Tocopherol Pathways, Mirrored the Corresponding Metabolite Contents in Rice Leaves during De-Etiolation. Plants 2021, 10, 1456. [Google Scholar] [CrossRef]
- Bian, Z.; Wang, Y.; Zhang, X.; Grundy, S.; Hardy, K.; Yang, Q.; Lu, C. A Transcriptome Analysis Revealing the New Insight of Green Light on Tomato Plant Growth and Drought Stress Tolerance. Front. Plant Sci. 2021, 12, 649283. [Google Scholar] [CrossRef]
- Jiao, Y.; Lau, O.S.; Deng, X.W. Light-Regulated Transcriptional Networks in Higher Plants. Nat. Rev. Genet. 2007, 8, 217–230. [Google Scholar] [CrossRef] [PubMed]
- de Wit, M.; Galvão, V.C.; Fankhauser, C. Light-Mediated Hormonal Regulation of Plant Growth and Development. Annu. Rev. Plant Biol. 2016, 67, 513–537. [Google Scholar] [CrossRef] [PubMed]
- Hunter, W.N. The Non-Mevalonate Pathway of Isoprenoid Precursor Biosynthesis. J. Biol. Chem. 2007, 282, 21573–21577. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.; Agarwal, A.V.; Akhtar, N.; Sangwan, R.S.; Singh, S.P.; Trivedi, P.K. Cloning and Characterization of 2-C-Methyl-d-Erythritol-4-Phosphate Pathway Genes for Isoprenoid Biosynthesis from Indian Ginseng, Withania somnifera. Protoplasma 2013, 250, 285–295. [Google Scholar] [CrossRef] [PubMed]
- Pulido, P.; Perello, C.; Rodriguez-Concepcion, M. New Insights into Plant Isoprenoid Metabolism. Mol. Plant 2012, 5, 964–967. [Google Scholar] [CrossRef]
- Nagata, N.; Suzuki, M.; Yoshida, S.; Muranaka, T. Mevalonic Acid Partially Restores Chloroplast and Etioplast Development in Arabidopsis Lacking the Non-Mevalonate Pathway. Planta 2002, 216, 345–350. [Google Scholar] [CrossRef]
- Rodríguez-Concepción, M. Early Steps in Isoprenoid Biosynthesis: Multilevel Regulation of the Supply of Common Precursors in Plant Cells. Phytochem. Rev. 2006, 5, 1–15. [Google Scholar] [CrossRef]
- Ghassemian, M.; Lutes, J.; Tepperman, J.M.; Chang, H.-S.; Zhu, T.; Wang, X.; Quail, P.H.; Markus Lange, B. Integrative Analysis of Transcript and Metabolite Profiling Data Sets to Evaluate the Regulation of Biochemical Pathways during Photomorphogenesis. Arch. Biochem. Biophys. 2006, 448, 45–59. [Google Scholar] [CrossRef]
- Filichkin, S.A.; Priest, H.D.; Givan, S.A.; Shen, R.; Bryant, D.W.; Fox, S.E.; Wong, W.-K.; Mockler, T.C. Genome-Wide Mapping of Alternative Splicing in Arabidopsis Thaliana. Genome Res. 2010, 20, 45–58. [Google Scholar] [CrossRef]
- Fung-Uceda, J.; Lee, K.; Seo, P.J.; Polyn, S.; Veylder, L.D.; Mas, P. The Circadian Clock Sets the Time of DNA Replication Licensing to Regulate Growth in Arabidopsis. Dev. Cell 2018, 45, 101–113.e4. [Google Scholar] [CrossRef]
- Li, P.; Ponnala, L.; Gandotra, N.; Wang, L.; Si, Y.; Tausta, S.L.; Kebrom, T.H.; Provart, N.; Patel, R.; Myers, C.R.; et al. The Developmental Dynamics of the Maize Leaf Transcriptome. Nat. Genet. 2010, 42, 1060–1067. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Zhou, Z.; Wang, Z.; Li, W.; Fang, C.; Wu, M.; Ma, Y.; Liu, T.; Kong, L.-A.; Peng, D.-L.; et al. Global Dissection of Alternative Splicing in Paleopolyploid Soybean. Plant Cell 2014, 26, 996–1008. [Google Scholar] [CrossRef]
- Campbell, M.A.; Haas, B.J.; Hamilton, J.P.; Mount, S.M.; Buell, C.R. Comprehensive Analysis of Alternative Splicing in Rice and Comparative Analyses with Arabidopsis. BMC Genom. 2006, 7, 327. [Google Scholar] [CrossRef] [PubMed]
- Csorba, T.; Questa, J.I.; Sun, Q.; Dean, C. Antisense COOLAIR Mediates the Coordinated Switching of Chromatin States at FLC during Vernalization. Proc. Natl. Acad. Sci. USA 2014, 111, 16160–16165. [Google Scholar] [CrossRef]
- Deng, P.; Liu, S.; Nie, X.; Weining, S.; Wu, L. Conservation Analysis of Long Non-Coding RNAs in Plants. Sci. China Life Sci. 2018, 61, 190–198. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Jung, C.; Xu, J.; Wang, H.; Deng, S.; Bernad, L.; Arenas-Huertero, C.; Chua, N.-H. Genome-Wide Analysis Uncovers Regulation of Long Intergenic Noncoding RNAs in Arabidopsis[C][W]. Plant Cell 2012, 24, 4333–4345. [Google Scholar] [CrossRef] [PubMed]
- Corona-Gomez, J.A.; Garcia-Lopez, I.J.; Stadler, P.F.; Fernandez-Valverde, S.L. Splicing Conservation Signals in Plant Long Non-Coding RNAs. RNA 2020, 26, 784–793. [Google Scholar] [CrossRef] [PubMed]
- Komiya, R.; Ohyanagi, H.; Niihama, M.; Watanabe, T.; Nakano, M.; Kurata, N.; Nonomura, K.-I. Rice Germline-Specific Argonaute MEL1 Protein Binds to phasiRNAs Generated from More than 700 lincRNAs. Plant J. 2014, 78, 385–397. [Google Scholar] [CrossRef]
- Lavarenne, J.; Gonin, M.; Champion, A.; Javelle, M.; Adam, H.; Rouster, J.; Conejéro, G.; Lartaud, M.; Verdeil, J.-L.; Laplaze, L.; et al. Transcriptome Profiling of Laser-Captured Crown Root Primordia Reveals New Pathways Activated during Early Stages of Crown Root Formation in Rice. PLoS ONE 2020, 15, e0238736. [Google Scholar] [CrossRef]
- Yan, Y.; Wang, H.; Hamera, S.; Chen, X.; Fang, R. miR444a Has Multiple Functions in the Rice Nitrate-Signaling Pathway. Plant J. 2014, 78, 44–55. [Google Scholar] [CrossRef]
- Chen, H.; Xu, N.; Wu, Q.; Yu, B.; Chu, Y.; Li, X.; Huang, J.; Jin, L. OsMADS27 Regulates the Root Development in a NO3−—Dependent Manner and Modulates the Salt Tolerance in Rice (Oryza sativa L.). Plant Sci. 2018, 277, 20–32. [Google Scholar] [CrossRef] [PubMed]
- Pachamuthu, K.; Hari Sundar, V.; Narjala, A.; Singh, R.R.; Das, S.; Avik Pal, H.C.Y.; Shivaprasad, P.V. Nitrate-Dependent Regulation of miR444-OsMADS27 Signalling Cascade Controls Root Development in Rice. J. Exp. Bot. 2022, 73, 3511–3530. [Google Scholar] [CrossRef] [PubMed]
- Pathak, R.R.; Jangam, A.P.; Malik, A.; Sharma, N.; Jaiswal, D.K.; Raghuram, N. Transcriptomic and Network Analyses Reveal Distinct Nitrate Responses in Light and Dark in Rice Leaves (Oryza sativa Indica Var. Panvel1). Sci. Rep. 2020, 10, 12228. [Google Scholar] [CrossRef]
- Yoneyama, T.; Suzuki, A. Light-Independent Nitrogen Assimilation in Plant Leaves: Nitrate Incorporation into Glutamine, Glutamate, Aspartate, and Asparagine Traced by 15N. Plants 2020, 9, 1303. [Google Scholar] [CrossRef] [PubMed]
- Geniza, M.; Fox, S.E.; Sage, A.; Ansariola, M.; Megraw, M.; Jaiswal, P. Comparative Analysis of Salinity Response Transcriptomes in Salt-Tolerant Pokkali and Susceptible IR29 Rice. bioRxiv 2023. 2023.08.20.551368. [Google Scholar] [CrossRef]
- Al-Bader, N.; Meier, A.; Geniza, M.; Gongora, Y.S.; Oard, J.; Jaiswal, P. Loss of a Premature Stop Codon in the Rice Wall-Associated Kinase 91 (WAK91) Gene Is a Candidate for Improving Leaf Sheath Blight Disease Resistance. Genes 2023, 14, 1673. [Google Scholar] [CrossRef] [PubMed]
- Naithani, S.; Deng, C.H.; Sahu, S.K.; Jaiswal, P. Exploring Pan-Genomes of Crops: Genomic Resources and Tools for Unraveling Gene Evolution, Function, and Adaptation. Biomolecules 2023, 13, 1403. [Google Scholar] [CrossRef]
- Hartmann, L.; Drewe-Boß, P.; Wießner, T.; Wagner, G.; Geue, S.; Lee, H.-C.; Obermüller, D.M.; Kahles, A.; Behr, J.; Sinz, F.H.; et al. Alternative Splicing Substantially Diversifies the Transcriptome during Early Photomorphogenesis and Correlates with the Energy Availability in Arabidopsis. Plant Cell 2016, 28, 2715–2734. [Google Scholar] [CrossRef]
- Gupta, P.; Jaiswal, P. Light-Regulated Gene Expression and Alternative Splicing Data from Rice Seedlings. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).