1. Introduction
With the increased use of the rare earth elements (REE) in modern technologies across numerous sectors including energy, automobile, and defense, the criticality of securing reliable sources has moved to the forefront of many nations’ economic outlooks [
1,
2]. The rare earth metals market was valued at
$9 billion in 2019 and is expected to surpass
$20 billion by 2024 [
3]. A large driver for that market is the end-users of the REEs, namely electronics, magnets, lasers, etc., that are worth an estimated
$7 trillion in a global economy worth
$75 trillion [
4]. The 14 naturally occurring lanthanide elements plus yttrium (Y) and scandium (Sc) that comprise the REE are relatively abundant in the Earth’s crust but generally not found in concentrations economical to mine. Presently, conventional mining efforts have been focused primarily on the ‘hard mineral’ occurrences of the REE, in the form of minerals such as bastnaesite, monazite, xenotime, and carbonatites (e.g., [
1,
5]). These mineral deposits often must be first subjected to significant processing of the ore to include crushing and grinding, calcining, roasting, flotation separation, and other beneficiation techniques [
6]. Following beneficiation, the REE are extracted using strong inorganic acids such as sulfuric acid, H
2SO
4. Such a process is often heavily capital- and energy-intensive and poses additional handling considerations for waste management and environmental protections.
The weathering of REE-rich host rocks can lead to the accumulation of REE in adjacent aluminosilicate-rich clay deposits. During weathering, REE ions can potentially be re-mobilized and redeposited onto active surfaces of clay-rich soil or sediments. These clay-rich deposits can contain elevated concentrations of ion-adsorbed REE as well as REE bearing colloids (e.g., Fe, Mn-oxides) and crystalline minerals (e.g., REE-phosphates). In the geologic past, the weathering of REE-rich igneous rocks led to the formation of aluminosilicate-rich clays in soils and laterites. These clay-rich deposits can contain up to 80–90% of their REEs in various adsorbed states (exchangeable, colloid, mineral). China has the largest REE reserves of an estimated 44 million tons, and produced approximately 63% of the world total mine production of REE in 2019 [
7]. As recently as 2008, alternative deposits located in southern China were found to contain a significant portion of ion-adsorbed REEs [
8]. These weathered crust deposits are readily amenable to ion exchange reactions using salts such as ammonium sulfate, (NH
4)
2SO
4, and exchanging REE ions from the surfaces of clay minerals [
8]. While lower grades (0.05 to 0.2 wt%) than conventional hard mineral deposits (>7 wt% REE as oxides), ion-adsorbed clay deposits host a significant enrichment of heavier mass REE [
5,
9].
Easily recoverable ion-adsorbed REE were also noted for certain coal underclays and roof rock material in the U.S. [
10]. Rozelle et al. (2016) demonstrated that, in some cases, >90% of the REE could be recovered from coal underclays and roof rock using (NH
4)
2SO
4 as an ion exchange lixiviant [
10]. Additional studies on REE occurrences in underclays, however, have identified significantly lower amounts (e.g., <10%) of ion-adsorbed REE [
11,
12]. The amount of ion-adsorbed REE present is likely to vary from sample to sample. However, the historical extent and large volume of reject waste from U.S. coal production presents a large source of easily accessible material with the potential to provide additional streams of domestic source of REEs and other critical elements. According to the Appalachian Region Independent Power Producers Association, the Pennsylvania Department of Environmental Protection states that there are 840 such coal refuse piles overlying nearly 10,000 acres of abandoned mine lands and scattered throughout PA’s anthracite and bituminous coalfields [
13]. The amount of coal refuse in Pennsylvania is estimated to be 2 billion cubic yards [
13]. The amount of REE present in these refuse piles and associated waste rock has been measured between 200 and 500 ppm (this work). Comparatively, world averages for coal and coal ash contain approximately 68 and 404 ppm of REE, respectively, with US averages at 62 and 517 ppm, respectively [
4].
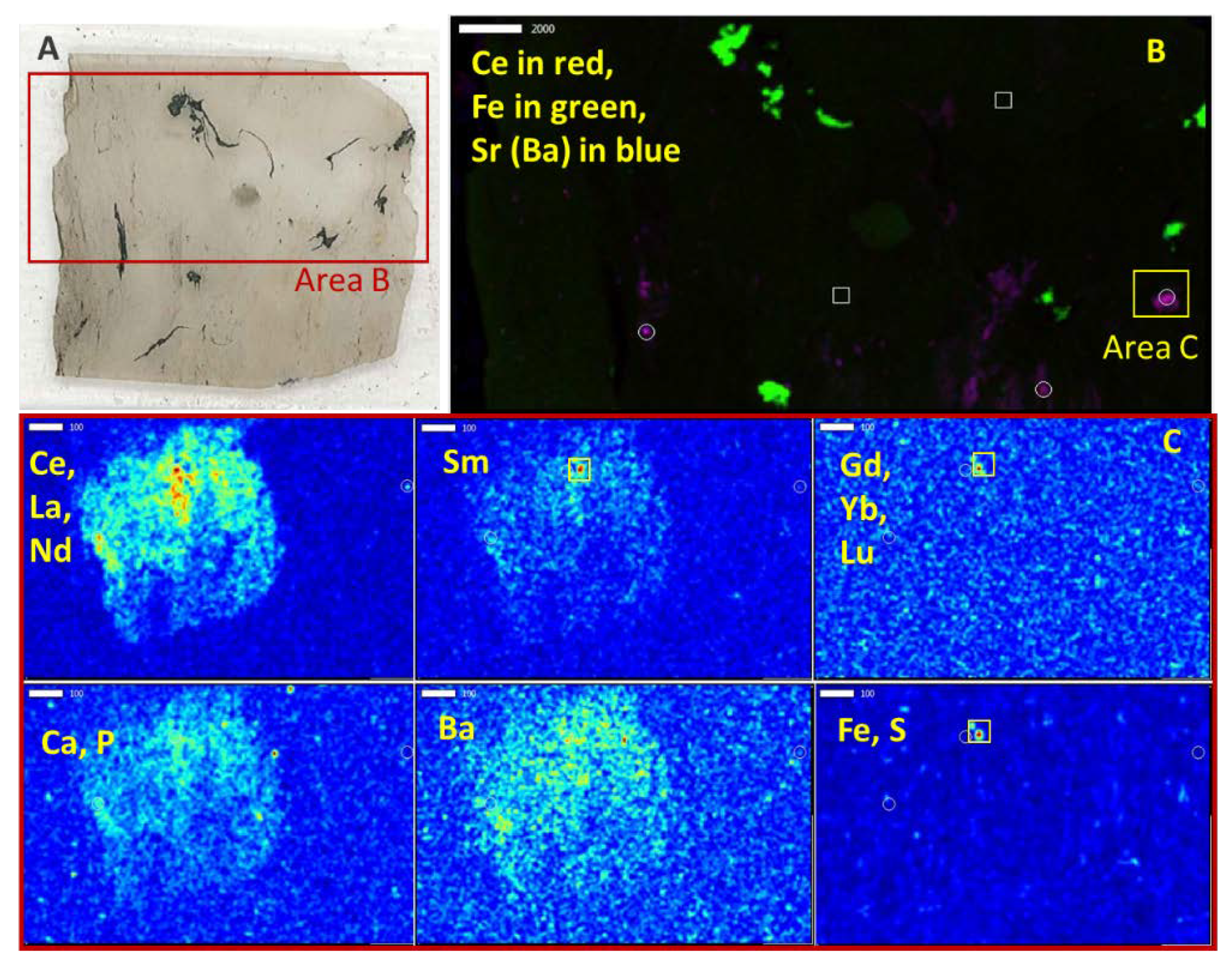
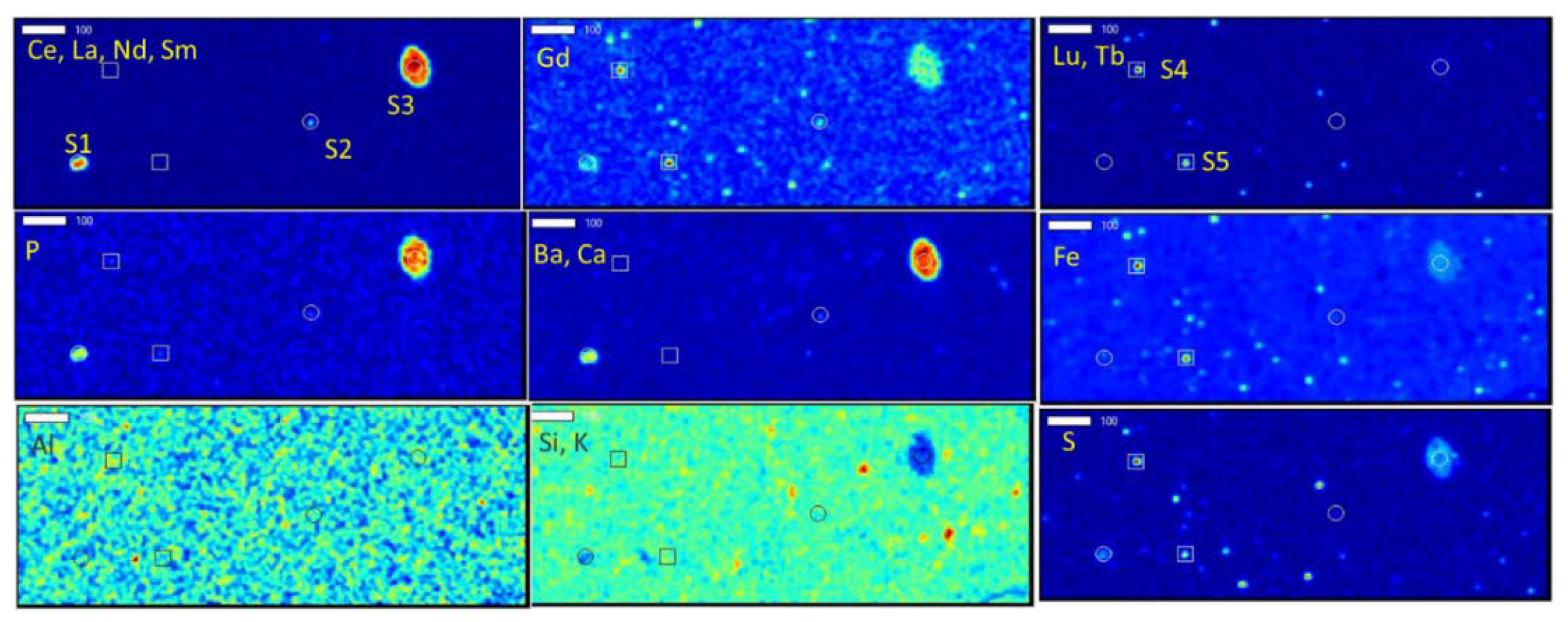
In this study, the composition of several underclays from the Appalachian Basin are analyzed from a microscopic workflow to visually document and characterize the modes of occurrence of REEs. Distributed occurrences of REE-bearing minerals (~100–200 µm), such as monazite and xenotime embedded in the clay matrix, are interpreted as representing the detrital loading of REE materials from the time of deposition. In addition, occurrences of REE-bearing minerals (presumedly rhabdophane and churchite) in altered material of pore-filling spaces is also documented. This textural relationship is interpreted to signify the formation of in situ authigenic minerals during diagenesis or later alterations or weathering and provides evidence for the mobility of the REEs, including potentially onto the abundant surfaces of the clay matrix. Such interpretations and observations are vital to designing an extraction process targeting REE phases and maximizing extraction efficiencies.
1.1. Appalachian Basin
The Appalachian Basin formed ~480 million years ago (Ma) at a time when the North American continent straddled the equator on a passive plate margin and the inland sea harbored warm, tropical waters. Initial basin formation at the onset of the Taconic Orogeny during the Middle Ordovician Period (458–470 Ma) was triggered when the Iapetus oceanic plate collided with and subducted under the North American plate [
14]. Clastic deposition during this period was derived from a sediment supply from the eastern highlands [
15]. Paleosols deposited during this time would serve as the underlying strata (i.e., underclays) to large peat mires deposited during the Pennsylvanian Period (360–300 Ma) that would later form the major coal beds of the Appalachian Basin in the present day.
1.2. Study Units
Study units were selected from four widely dispersed areas within the Appalachian basin focusing on target coals with known elevated REE values based on personal communication, previous work, and legacy coal chemistry data stored at the West Virginia Geological and Economic Survey (WVGES). Final selection of West Virginia samples was limited to cores available at the WVGES core facility. Scope was further limited to zones from areas suspected to have high REE concentrations and focused on paleo-laterite deposits, seat earths, and underclays where, based on modern production practices, the REE are known to accumulate. Samples from Pennsylvania obtained from the Pittsburgh and Brookville coal seams, were selected to include a regional survey of underclays in the Appalachian Basin (
Table 1).
Six coals horizons were selected to be sampled: the Pittsburgh, Lower Freeport, Middle Kittanning, Brooksville, No 5 Block coal and the lower split of the No. 5 Block. A total of 8 samples were collected from West Virginia and Pennsylvania. Six samples were collected from 3 cores stored at the West Virginia Geological and Economic Survey. Three samples were from south-western Ritchie County in western West Virginia targeting the Allegheny Formation Lower Freeport, Middle Kittanning, and Monongahela Formation Pittsburgh coal paleosols. Coal has never been mined in this portion of the state due in part to depth, accessibility issues and market considerations.
Two samples were from north central Kanawha County in southern West Virginia in the Allegheny Formation No. 5 Block and No. 5 Block Lower split underclays, while the sixth sample was located in Barbour County in north central West Virginia in seat earth below the Allegheny Lower Freeport horizon (
Figure 1). Both areas are highly developed coal fields. The final two samples were collected in Pennsylvania from the Pittsburgh and Brookville coal seams.