Theoretical Investigation on Rare Earth Elements of Y, Nd and La Atoms’ Adsorption on the Kaolinite (001) and
Abstract
:1. Introduction
2. Method of Calculations
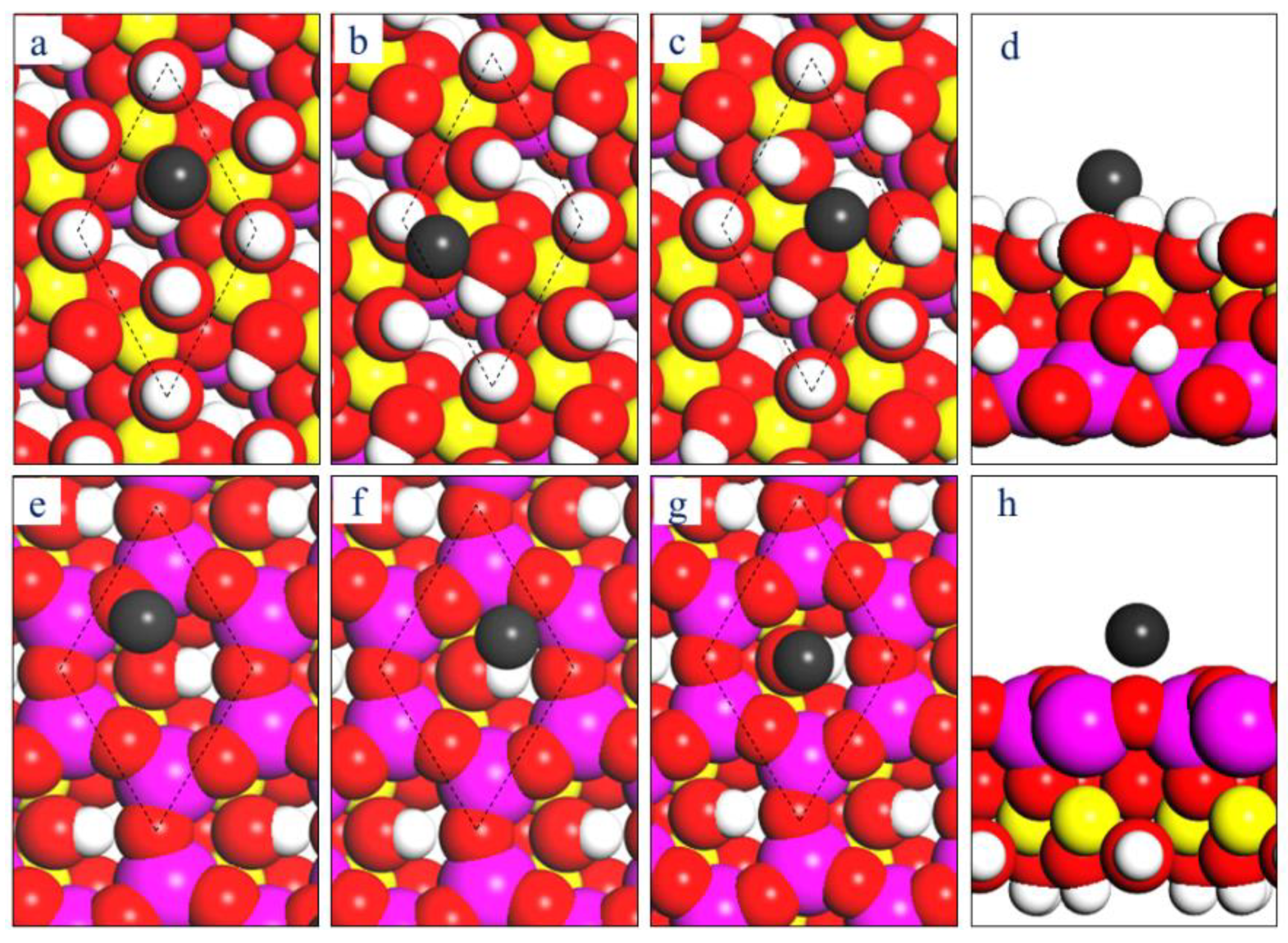
3. Results and Discussions
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ogata, T.; Narita, H.; Tanaka, M. Adsorption behavior of rare earth elements on silica gel modified with diglycol amic acid. Hydrometallurgy 2015, 152, 178–182. [Google Scholar] [CrossRef]
- Greaves, M.; Elderfield, H.; Sholkovitz, E. Aeolian sources of rare earth elements to the Western Pacific Ocean. Mar. Chem. 1999, 68, 31–38. [Google Scholar] [CrossRef]
- Voßenkaul, D.; Stoltz, N.; Meyer, F.M.; Friedrich, B. Extraction of Rare Earth Elements from non-Chinese Ion Adsorption Clays. In Proceedings of the European Metallurgical Conference (EMC), Düsseldorf, Germany, 14–17 June 2015. [Google Scholar] [CrossRef]
- Anastopoulos, I.; Bhatnagar, A.; Lima, E.C. Adsorption of rare earth metals: A review of recent literature. J. Mol. Liq. 2016, 221, 954–962. [Google Scholar] [CrossRef]
- Kondo, K.; Kamio, E. Separation of rare earth metals with a polymeric micro-capsule membrane. Desalination 2002, 144, 249–254. [Google Scholar] [CrossRef]
- Araki, K.; Yoshida, M.; Uezu, K.; Goto, M.; Furusaki, S. Lanthanide-Imprinted Resins Prepared by Surface Template Polymerization. J. Chem. Eng. Jpn. 2000, 33, 665–668. [Google Scholar] [CrossRef]
- Alakhras, F.; Abu Dari, K.; Mubarak, M.S. Synthesis and chelating properties of some poly(amidoxime-hydroxamic acid) resins toward some trivalent lanthanide metal ions. J. Appl. Polym. Sci. 2005, 97, 691–696. [Google Scholar] [CrossRef]
- Choi, S.-H.; Lee, K.-P.; Sohn, S.-H. Graft copolymer-lanthanide complexes obtained by radiation grafting on polyethylene film. J. Appl. Polym. Sci. 2002, 87, 328–336. [Google Scholar] [CrossRef]
- Chen, Y.G.; Zhu, B.H.; Wu, D.B.; Wang, Q.G.; Yang, Y.H.; Ye, W.M.; Guo, J.F. Eu(III) adsorption using di(2-ethylhexyl) phosphoric acid-immobilized magnetic GMZ bentonite. Chem. Eng. J 2012, 181, 387–396. [Google Scholar] [CrossRef]
- Iannicelli-Zubiani, E.M.; Cristiani, C.; Dotelli, G.; Stampino, P.G.; Pelosato, R.; Mesto, E.; Schingaro, E.; Lacalamita, M. Use of natural clays as sorbent materials for rare earth ions: Materials characterization and set up of the operative parameters. Waste Manag. 2015, 46, 546–556. [Google Scholar] [CrossRef]
- Moldoveanu, G.A.; Papangelakis, V.G. Recovery of rare earth elements adsorbed on clay minerals: I. Desorption mechanism. Hydrometallurgy 2012, 117, 71–78. [Google Scholar] [CrossRef]
- Moldoveanu, G.A.; Papangelakis, V.G. Recovery of rare earth elements adsorbed on clay minerals: II. Leaching with ammonium sulfate. Hydrometallurgy 2013, 131, 158–166. [Google Scholar] [CrossRef]
- Chegrouche, S.; Mellah, A.; Telmoune, S. Removal of lanthanum from aqueous solutions by natural bentonite. Water Res. 1997, 31, 1733–1737. [Google Scholar] [CrossRef]
- Chen, Y.; Zhu, C.; Sun, Y.; Duan, H.; Ye, W.; Wu, D. Adsorption of La(III) onto GMZ bentonite: Effect of contact time, bentonite content, pH value and ionic strength. J. Radioanal. Nucl. Chem. 2012, 292, 1339–1347. [Google Scholar] [CrossRef]
- Chen, Y.H.; Lu, D.L. H2 capture by kaolinite and its adsorption mechanism. Appl. Clay Sci. 2015, 104, 221–228. [Google Scholar] [CrossRef]
- Brigatti, M.F.; Galan, E.; Theng, B.K.G. Handbook of Clay Science. In General Introduction: Clays Clay Minerals, and Clay Science; Bergaya, F., Theng, B.K.G., Lagaly, G., Eds.; Elsevier Ltd: Amsterdam, The Netherlands, 2006; pp. 27–30. [Google Scholar]
- Laufer, F.; Yariv, S.; Steinberg, M. The adsorption of quadrivalent cerium by kaolinite. Clay Miner. 1984, 19, 137–149. [Google Scholar] [CrossRef]
- Aja, S.U. The Sorption of the Rare Earth Element, Nd, onto Kaolinite at 25 °C. Clays Clay Miner. 1998, 46, 103–109. [Google Scholar] [CrossRef]
- Coppin, F.; Berger, G.; Bauer, A.; Castet, S.; Loubet, M. Sorption of lanthanides on smectite and kaolinite. Chem. Geol. 2002, 182, 57–68. [Google Scholar] [CrossRef]
- Wan, Y.; Liu, C. The effect of humic acid on the adsorption of REEs on kaolin. Colloids Surf. A Physicochem. Eng. Asp. 2006, 290, 112–117. [Google Scholar] [CrossRef]
- Xiao, Y.; Huang, L.; Long, Z.; Feng, Z.; Wang, L. Adsorption ability of rare earth elements on clay minerals and its practical performance. J. Rare Earths 2016, 34, 543–548. [Google Scholar] [CrossRef]
- He, M.-C.; Zhao, J. Effects of Mg, Ca, and Fe(II) Doping on the Kaolinite (001) Surface with H2O Adsorption. Clays Clay Miner. 2012, 60, 330–337. [Google Scholar] [CrossRef]
- Zhao, J.; Wang, J.; Qin, X.; Cao, Y. An ab-initio study of H2O adsorption on the calcite (104) surface with different coverages. Solid State Commun. 2020, 313, 113892. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhou, Q.; Zhuang, L.; Zhao, Z. Adsorption of Ca(II) and K(I) on the kaolinite surface: A DFT study with an experimental verification. Mol. Phys. 2021, 119, e1896047. [Google Scholar] [CrossRef]
- Hou, J.L.; Chen, M.; Zhou, Y.F.; Bian, L.; Dong, F.Q.; Tang, Y.H.; Ni, Y.X.; Zhang, H.P. Regulating the effect of element doping on the CO2 capture performance of kaolinite: A density functional theory study. Appl. Surf. Sci. 2020, 512. [Google Scholar] [CrossRef]
- Kuriki, K.; Koike, Y.; Okamoto, Y. Plastic Optical Fiber Lasers and Amplifiers Containing Lanthanide Complexes. Chem. Rev. 2002, 102, 2347–2356. [Google Scholar] [CrossRef] [PubMed]
- Hull, R.; Parisi, J.; Osgood, R.M. Spectroscopic Properties of Rare Earths in Optical Materials Volume 83: Rare Earth Ions in Advanced X-ray Imaging Materials; Springer Series in Materials Science; Springer: Berlin/Heidelberg, Germany, 2005; Chapter 10; pp. 500–529. [Google Scholar]
- Zou, J.H.; Liu, D.; Tian, H.M.; Liu, F.; Li, T.; Yang, H.Y. Geochemical characteristics of trace elements and rare earth elements of Late Paleozoic Coal in Adaohai mine, Inner Mongolia. Acta Coalae Sin. 2013, 38, 12–1018. (In Chinese) [Google Scholar]
- Kresse, G.; Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 1996, 54, 11169–111173. [Google Scholar] [CrossRef]
- Blóchl, P.E. Projector augmented-wave method. Phys. Rev. B 1994, 50, 17953–17979. [Google Scholar] [CrossRef] [Green Version]
- Adams, J.M. Hydrogen Atom Positions in Kaolinite by Neutron Profile Refinement. Clays Clay Miner. 1983, 31, 352–356. [Google Scholar] [CrossRef]
- Bish, D.L. Rietveld Refinement of the Kaolinite Structure at 1.5 K. Clays Clay Miner. 1993, 41, 738–744. [Google Scholar] [CrossRef]
- Benco, L.; Tunega, D.; Hafner, J.; Lischka, H. Orientation of OH groups in kaolinite and dickite: Ab initio molecular dynamics study. Am. Miner. 2001, 86, 1057–1065. [Google Scholar] [CrossRef]
- Bailey, S.W. Structure of layer silicates. In Crystal Structures of Clay Minerals and Their X-Ray Identification; Brindley, G.W., Brown, G., Eds.; Mineralogical Society: London, UK, 1980; pp. 36–123. [Google Scholar]
- Hayashi, S. NMR Study of Dynamics and Evolution of Guest Molecules in Kaolinite/Dimethyl Sulfoxide Intercalation Compound. Clays Clay Miner. 1997, 45, 724–732. [Google Scholar] [CrossRef]
- Hess, A.C.; Saunders, V.R. Periodic ab initio Hartree-Fock calculations of the low-symmetry mineral kaolinite. J. Phys. Chem. 1992, 96, 4367–4374. [Google Scholar] [CrossRef]
- Hobbs, J.D.; Schultz, P.A.; Sears, M.P.; Cygan, R.T.; Nagy, K.L. All-atom ab initio energy minimization of the kaolinite crystal structure. Am. Miner. 1997, 82, 657–662. [Google Scholar] [CrossRef]
- Hu, X.L.; Angelos, M. Water on the hydroxylated (001) surface of kaolinite: From monomer adsorption to a flat 2D wetting layer. Surf. Sci. 2008, 602, 960–974. [Google Scholar] [CrossRef]
- Plancon, A.; GieseJr, R.F.; Snyder, R.; Drits, V.A.; Bookin, A.S. Stacking faults in the kaolinite-group minerals: Defect structures of kaolinite. Clays Clay Miner. 1997, 37, 195–198. [Google Scholar] [CrossRef]
- Teppen, B.J.; Rasmussen, K.; Bertsch, P.; Miller, D.M.; Schäfer, L. Molecular Dynamics Modeling of Clay Minerals. 1. Gibbsite, Kaolinite, Pyrophyllite, and Beidellite. J. Phys. Chem. B 1997, 101, 1579–1587. [Google Scholar] [CrossRef]
- Schwieger, W.; Lagaly, G.; Dutta, P.; Auerbach, S.; Carrad, K. Alkali Silicates and Crystalline Silicic Acids. In Handbook of Layered Materials; Informa UK Limited: London, UK, 2004; pp. 541–629. [Google Scholar]
- Sato, H.; Ono, K.; Johnston, C.T.; Yamagishi, A. First-principles studies on the elastic constants of a 1:1 layered kaolinite mineral. Am. Miner. 2005, 90, 1824–1826. [Google Scholar] [CrossRef]
- Kresse, G.; Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 1999, 59, 1758–1775. [Google Scholar] [CrossRef]
- Zhao, J.; He, M.-C. Theoretical study of heavy metal Cd, Cu, Hg, and Ni(II) adsorption on the kaolinite (001) surface. Appl. Surf. Sci. 2014, 317, 718–723. [Google Scholar] [CrossRef]
- Chi, R.A.; Wang, D.Z. Quantum chemical calculation of adsorption properties of clay minerals and enrichment of rare earths. Chin. J. Rare Earths 1993, 4, 199–203. (In Chinese) [Google Scholar]
- Zhao, J.; Gao, W.; Qin, X.Z. First-principles study on adsorption behavior of as on the kaolinite (001) and (001) surfaces Adsorption. Adsorption 2020, 26, 443–452. [Google Scholar] [CrossRef]



| Site | Y | Nd | La | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Top | Bridge | Hollow | Top | Bridge | Hollow | Top | Bridge | Hollow | |
| ML 0.11 | 109.6 | 137.5 | 108.6 | 94.2 | 126.9 | 132.7 | 166.3 | 151.9 | 168.2 |
| ML 0.25 | 128.8 | 141.3 | 128.8 | 103.8 | 140.4 | 141.3 | 169.2 | 154.8 | 172.1 |
| ML 0.33 | 129.8 | 147.1 | 132.7 | 146.1 | 149.0 | 151.9 | 170.2 | 159.6 | 176.0 |
| ML 0.5 | 146.1 | 153.8 | 137.5 | 152.9 | 148.1 | 155.8 | 221.1 | 180.8 | 226.9 |
| ML 0.75 | 152.9 | 155.8 | 146.1 | 156.7 | 159.6 | 176.9 | 224.0 | 215.4 | 226.0 |
| ML 1.0 | 156.7 | 165.4 | 149.0 | 166.3 | 164.4 | 189.4 | 227.8 | 221.1 | 232.7 |
| Site | Y | Nd | La | |||
|---|---|---|---|---|---|---|
| Bridge | Hollow | Top | Hollow | Bridge | Hollow | |
| Eads | 41.3 | 42.3 | 36.5 | 43.2 | 43.2 | 48.1 |
| Phase | Θ | Y | Nd | La | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Top | Bridge | Hollow | Top | Bridge | Hollow | Top | Bridge | Hollow | ||
| hREEs-Al (Å) | 0.25 ML | 3.42 | 3.32 | 3.44 | 3.43 | 3.24 | 3.15 | 3.33 | 3.34 | 3.10 |
| 0.5 ML | 3.42 | 3.29 | 3.43 | 3.42 | 3.17 | 3.09 | 3.32 | 3.34 | 3.08 | |
| 0.75 ML | 3.39 | 3.15 | 3.42 | 3.42 | 3.13 | 3.07 | 3.1 | 3.26 | 2.98 | |
| 1.00 ML | 3.38 | 3.10 | 3.42 | 3.40 | 3.10 | 2.95 | 2.92 | 3.26 | 2.93 | |
| Ra (Å) | 0.25 ML | 2.57 | 2.49 | 2.52 | 2.49 | 2.45 | 2.44 | 2.45 | 2.49 | 2.45 |
| 0.5 ML | 2.53 | 2.47 | 2.53 | 2.49 | 2.45 | 2.44 | 2.45 | 2.48 | 2.43 | |
| 0.75 ML | 2.50 | 2.43 | 2.42 | 2.48 | 2.44 | 2.42 | 2.42 | 2.46 | 2.42 | |
| 1.00 ML | 2.57 | 2.45 | 2.59 | 2.48 | 2.43 | 2.42 | 2.45 | 2.52 | 2.42 | |
| Δd12 (%) | 0.25 ML | 1.95 | 2.11 | 1.83 | 2.10 | 2.23 | 2.23 | 1.83 | 1.81 | 1.95 |
| 0.5 ML | 3.45 | 3.21 | 2.53 | 2.13 | 2.10 | 2.11 | 2.59 | 2.34 | 1.86 | |
| 0.75 ML | 2.23 | 4.10 | 2.48 | 3.84 | 3.85 | 2.35 | 2.70 | 3.18 | 3.10 | |
| 1.00 ML | 4.82 | 3.70 | 2.68 | 4.05 | 2.11 | 3.26 | 3.70 | 2.52 | 2.88 | |
| Δd23 (%) | 0.25 ML | 3.77 | 1.73 | 3.87 | 2.12 | 0.21 | 1.98 | 3.36 | 3.22 | 2.25 |
| 0.5 ML | 1.52 | 1.93 | 5.16 | 2.15 | 0.71 | 2.02 | 2.04 | −6.72 | 1.25 | |
| 0.75 ML | 10.2 | 7.14 | −4.18 | −0.32 | −5.32 | 2.25 | −3.82 | 1.01 | 0.81 | |
| 1.00 ML | 12.6 | 12.34 | 1.63 | −0.55 | −7.14 | 1.62 | 2.90 | 1.25 | 0.81 | |
| Site | Y | Nd | La | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Before | After | Dif. | Before | After | Dif. | Before | After | Dif. | ||
| Top | Y (Nd or La) | 11 | 10.68 | −0.32 | 14 | 13.68 | −0.32 | 11 | 10.46 | −0.54 |
| O1 | 1.43 | 1.48 | 0.05 | 1.43 | 1.47 | 0.04 | 1.43 | 1.46 | 0.03 | |
| Bridge | Y (Nd or La) | 11 | 10.54 | −0.46 | 14 | 13.51 | −0.49 | 11 | 10.58 | −0.42 |
| O2 | 1.43 | 1.45 | 0.02 | 1.52 | 1.53 | 0.01 | 1.46 | 1.53 | 0.07 | |
| O3 | 1.52 | 1.56 | 0.04 | 1.47 | 1.55 | 0.08 | 1.50 | 1.51 | 0.01 | |
| Y (Nd or La) | 11 | 10.55 | −0.45 | 14 | 13.30 | −0.70 | 11 | 10.29 | −0.71 | |
| Hollow | O4 | 1.52 | 1.54 | 0.02 | 1.46 | 1.52 | 0.06 | 1.52 | 1.59 | 0.07 |
| O5 | 1.44 | 1.46 | 0.02 | 1.48 | 1.50 | 0.02 | 1.45 | 1.48 | 0.03 | |
| O6 | 1.43 | 1.44 | 0.01 | 1.45 | 1.47 | 0.02 | 1.46 | 1.48 | 0.02 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, J.; Wang, Z.; Gao, W.; Wang, Y.-F.; Huang, B.-W.
Theoretical Investigation on Rare Earth Elements of Y, Nd and La Atoms’ Adsorption on the Kaolinite (001) and
Zhao J, Wang Z, Gao W, Wang Y-F, Huang B-W.
Theoretical Investigation on Rare Earth Elements of Y, Nd and La Atoms’ Adsorption on the Kaolinite (001) and
Zhao, Jian, Zheng Wang, Wei Gao, Yi-Fei Wang, and Bo-Wen Huang.
2021. "Theoretical Investigation on Rare Earth Elements of Y, Nd and La Atoms’ Adsorption on the Kaolinite (001) and





