1. Introduction
Cobalt and cobalt oxides are extensively used in a wide range of industrial and military applications such as superalloys, catalysts, cemented carbide, and lithium-ion batteries [
1]. With the rapid growth in electric vehicle use in recent years, the growth of lithium battery use, driven by electric vehicles, promotes the demand for cobalt. According to the demand data of cobalt in all the applied fields, consumption of cobalt in lithium-ion batteries accounted for 49% of total cobalt consumption in 2015; it is estimated that the consumption of cobalt in lithium-ion batteries will increase to 80% in 2030. It is estimated by the IEA 2DS (International Energy Agency 2-Degree Scenario) and IEA B2DS (International Energy Agency Below 2-Degree Scenario) that the annual cobalt demand will reach 300,000–400,000 tons in 2030 [
2]. According to survey data published by the USGS (United States Geological Survey) in 2021, the world cobalt mine reserves are about 7.1 million tons. However, more than 120 million tons of cobalt resources have been identified in manganese nodules and crusts on the floor of the Atlantic, Indian, and Pacific Oceans and the cobalt resources in the ocean are 4.8 times more than that on land [
3]. Due to the increase in demand for cobalt coupled with the depletion of land-based high-grade ores, ocean resources containing cobalt are being viewed as a future potential resource [
4].
In the 1980s, some researchers drew attention to the cobalt-rich crusts and pointed out that the areas of cobalt-rich crust were centered mainly in the western and southern Pacific [
5,
6,
7]. In the same era, the cooperative sea-floor research organization of Germany and the U.S discovered cobalt-rich crust at sea mounts of the Central Pacific Ocean [
8]. Due to higher cobalt content in the cobalt-rich crust than in ferromanganese nodules, research on cobalt-rich crusts as a potential and strategic substitute for cobalt mines on land gained prominence in the early 1980s [
9,
10]. Until recent years, the cobalt-rich crust is still seen as an additional source of raw materials besides land-based sources [
11]. Up to now, several studies have been investigated to concentrate and recover Co and Ni from cobalt-rich crust including mineral processing [
12], pyrometallurgy [
13,
14,
15,
16,
17,
18], and hydrometallurgy [
19,
20,
21,
22,
23]. Because of the high-water content in cobalt-rich crust, hydrometallurgy may be a good choice for the recovery of Co and Ni. Due to the high content of Mn and Fe in the cobalt-rich crust, the reductive ammonia leaching process is preferred to recover Co and Ni selectively. However, the ammonia-nitrogen wastewater discharged from the reductive ammonia leaching technique limits the industrial application of this technique [
24,
25,
26]. Due to the inevitable environmental problem caused by the ammonia leaching process, the reductive acid leaching process of the cobalt-rich crust could be a good choice for the recovery of Co and Ni. The hydrochloric acid and nitric acid leaching techniques are not suitable for practical application for the cobalt-rich crust because of the high price of hydrochloric acid and nitric acid. The reductive sulfuric acid leaching technique is an appropriate approach for treating cobalt-rich crust. To eliminate the effects of impurities brought by the reduction agent in the sulfuric acid leaching technique, SO
2 and H
2O
2 were used as reduction agents in the sulfuric acid leaching process [
27,
28]. However, the high-cost problem of SO
2 and H
2O
2 always ailed the industrial application of the reductive sulfuric acid leaching technique.
The aim of this study was to solve the high-cost problem of reduction agents in traditional sulfuric acid leaching techniques and use carbon as the reduction agent to reduce the high valence Mn in the cobalt-rich crust acid baking process. Then, the Co and Ni which were associated with Mn-oxides in the crust are released to the aqueous phase concurrent with the dissolution of low valence Mn reduced. Finally, the Co, Ni, and a part of Mn could be recovered from the leaching solution. Compared with other sulfuric acid leaching technologies, our proposed sulfuric acid baking technique could make full use of the heat of reactions between sulfuric acid and oxides in the cobalt-rich crust. In addition, the cheaper anthracite used as the reducing agent in acid baking replaced the more expensive H2O2 and SO2. Therefore, the cost of our technology based on such advantages might be lower than other sulfuric acid leaching techniques. Ultimately, this research provided a Co and Ni recovery process from the cobalt-rich crust by using carbon as the reduction agent in the acid baking process, which might not only solve the reproduction of the reduction agent but also be industrialized easily.
2. Materials and Methods
2.1. Materials
The cobalt-rich crust samples were obtained from the China Ocean Sample Repository (COSR), Qingdao, China. The fine crust samples were crushed and ground finely (more than 80% particle size < 75 μm), and the raw materials were dried at 80 °C in a vacuum dryer for 12 h before subsequent experiments.
2.2. Experiments
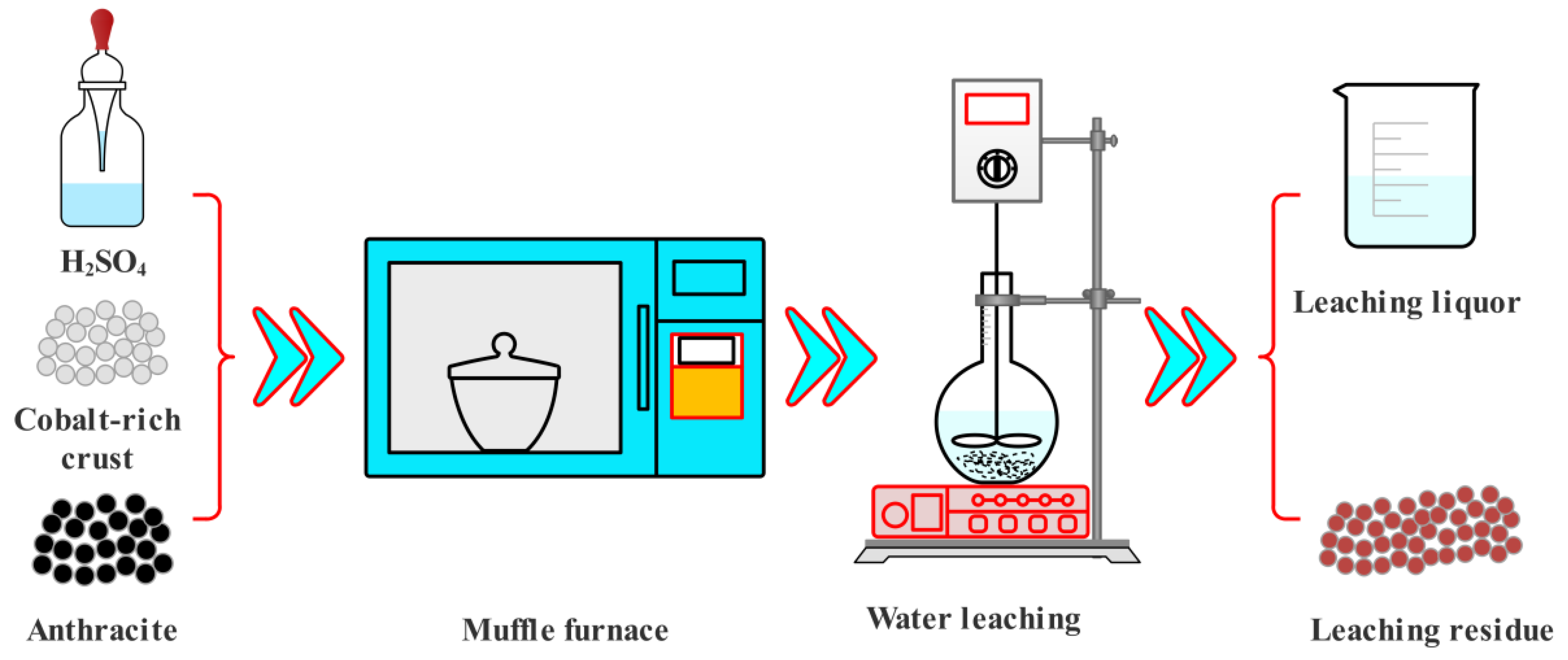
The details of the acid baking process are presented as follows: The 30 g fine cobalt-rich crusts and sulfuric acid (98 wt%) with different acid/crust mass ratios (A/C ratio) were homogeneously mixed. The mixtures were roasted for 240 min at different temperatures by using a muffle furnace (KSXQ-16-12, Yixing Sunnorn Furnace Co., Ltd., Yixing, China) with a temperature control accuracy of ±2 °C. After naturally cooling to room temperature, the baking product was used as the experimental material for the subsequent leaching process.
The details of the leaching process are presented as follows: The baking products were leached under the conditions of leaching temperature of 90 °C, leaching time of 60 min, and solid/liquid (g/mL) of 1/4. The schematic of the experimental process is shown in
Figure 1. At the end of each leaching test, the residues were separated from the leachate by vacuum filtration and were later washed using hot deionized water (80–90 °C) until the pH of the last filtrate was approximate 7. After drying at 100 °C for 24 h, the leaching residues were analyzed by ICP-OES for chemical composition and by XRD, and SEM for phase composition. The metals extraction efficiency of cobalt-rich crusts was calculated using Equation (1):
where
is the leaching ratio of metals (Ni, Co, Cu, Mn, and Fe);
or
is the weight percentage of metals in the cobalt-rich crust or leaching residue;
or
is the weight of the cobalt-rich crust or leaching residue.
2.3. Analysis Methods
The chemical composition of the cobalt-rich crusts, acid baking products, and leaching residues was determined by an inductively coupled plasma optical emission spectrometer (ICP-OES, Agilent 700, Agilent Technologies, Inc., Santa Clara, CA, USA). The high frequency-infrared carbon-sulfur spectrometry (HF-100, CS-344, LECO Corporation, St Joseph, MI, USA) was used to determine the contents of sulfur and carbon. The phase compositions of the cobalt-rich crusts and leaching residues were analyzed by an X-ray diffractometer (XRD, PANalytical Empyrean, Malvern Panalytical, Malvern, Worcs, UK) which was performed using Cu-Kα X-radiation at a scan rate of 10° 2θ/min. Morphological and chemical analyses of samples were performed via scanning electron microscopy (SEM, QUANTA 600, FEI Company, Hillsboro, OR, USA) with energy dispersive spectroscopy (EDS, EDAX genesis 7000, EDAX Inc., Mahwah, NJ, USA). The gas components of the gas exhausted during the acid baking process were analyzed by a gas analyzer (Gasmet Portable FT-IR Gas Analyzer; GASMET DX4000, Gasmet Technologies, Helsinki, Finland). The gas flow of N2 as the carrier gas during the gas analysis process was 3L/min, and the gas components of gas were analyzed every 5 s.
4. Discussion
Figure 11 displays the scanning images of the cobalt-rich crust leaching residues during the acid baking process, with and without carbon, under optimal conditions in this technique. We found the leaching residue particles during the acid baking process with carbon are more complete than that without carbon. As shown in
Figure 11a–c, a large part of leaching residues after the acid baking process with carbon are completed particles. The completed particles indicate that the cobalt-rich crust particles are not destroyed completely by sulfuric acid when carbon is added in the acid baking process. However, we easily found that a large number of leaching residues after the acid baking process without carbon have become the porous particles in
Figure 11d–f, which may explain why the sulfuric acid corrodes the FeO(OH) more easily in the cobalt-rich crust during the acid baking process without carbon. The thermodynamic analysis results in
Figure 4 demonstrate thatH
2SO
4 reacts with FeO(OH)more easily than that with MnO(OH) when there is no carbon added in the acid baking process (as can be seen in
Figure 4, the ΔG of Equation (7) is lower than that of Equation (2)). However, the ΔG of Equation (1) is lower than that of Equation (7), demonstrating that MnO(OH) reacts with H
2SO
4more easily than FeO(OH) when there is carbon in the acid baking process. Combined with the higher content of Fe in the EDS results of
Figure 12 (
Table 4) than that of
Figure 13 (
Table 5), we could confirm completely that carbon significantly promotes the reactions between MnO(OH) and sulfuric acid during the acid baking process, with the same amount of sulfuric acid.
Figure 12 and
Figure 13 show the SEM of leaching residues during the acid baking process, with and without carbon, under optimal conditions and the EDS results show the elemental compositions of the points corresponding to the SEM. As can be seen, the main compositions of leaching residues are iron and manganese oxides and silicon dioxide. Combined with the experimental result in
Figure 5, the higher content of Fe and lower content of Mn in
Figure 12 than in
Figure 13 confirms that the carbon obviously promotes the reactions between sulfuric acid and MnO(OH) in the cobalt-rich crust.
Figure 14 presents the gas compositions during the acid baking process, with and without carbon. It was found that the gas component of exhausted gas is CO
2 during acid baking and SO
2 was not detected during acid baking. The concentration of CO
2 increased from30 ppm to 120 ppm as the time increased from 0 to 10 min at the temperature of 200 °C during the acid baking without carbon (
Figure 14b). The concentration of CO
2 decreased further over time. It can be inferred that the CO
2 was generated from the reactions between H
2SO
4 and a small amount of C which exist in the cobalt-rich crust during acid baking without carbon.
As can be seen in
Figure 14, the concentration of CO
2 increased significantly when the anthracite was added to the acid baking process and the concentration of CO
2 reached 400 ppm (
Figure 14a). Meanwhile, the CO was still detected in spite of lower content. A large amount of CO
2 and a small amount of CO in the exhausted gas illustrates that the carbon directly participated in the reactions between H
2SO
4 and MnO(OH). Furthermore, the undetected SO
2 in exhausted gas confirms well that the carbon participated in the reactions directly rather than indirectly. These things considered, the reactions between H
2SO
4 and C as well MnO(OH) during the acid baking process maybe comprised of the Reactions (8) and (9) (
Table 6).
5. Conclusions
The valuable metal recovery process from a cobalt-rich crust by carbon as a reduction agent was a facile and environment-friendly alternative technology. The thermodynamic results demonstrated that carbon could promote the reactions of manganese minerals with sulfuric acid directly or indirectly. The diffraction peak of pyrolusite disappeared in the leaching residue indicating that the Mn minerals were decomposed during the acid baking process at an A/C ratio of 0.733 when the carbon existed during the acid baking process. The experimental results indicate that the average leaching ratios of Ni, Co, and Cu can reach 98.59%, 91.62%, and 93.81%, respectively, and the average leaching ratios of Mn and Fe are just 41.27% and 26.94%, respectively, under the conditions of a temperature of 200 °C, a time of 240 min, an A/C ratio of 0.567, and mass ratio of carbon and crust of 0.1.