Natural Iron Silicides: A Systematic Review
Abstract
:1. Introduction
2. Artificial Production of Iron Silicides
3. Irons Silicides as Minerals
4. FexSiy as Components of Fulgurites
5. Iron Silicides within Planetary Mantles and Cores
6. Iron Silicides in the Core-Mantle Boundaries of Mercury, the Moon, and Super Earths
7. Iron Silicide in Interplanetary Dust
8. Iron Silicides in Meteorites
8.1. Iron Silicides on the Moon
8.2. Iron Silicides and an Ureilite Parent Body (UPB)
8.3. Iron Silicides in other Types of Meteorites
9. Iron Silicides Deriving from Extraterrestrial Dust
10. Iron Silicides as Recondensation of Ejecta Vapour
11. Iron Silicides Associated with Craters
12. Other Iron Silicide Findings
13. Iron Silicides as a Component of Circumstellar Envelops (CSE) and in Interstellar Matter (ISM)
14. Iron Silicides Related to Novae and Supernovae
15. Discussion
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kaess, F. Die technische Herstellung des Ferrosiliziums. In Elektrothermie: Die Elektrische Erzeugung und Technische Anwendung hoher Temperaturen, Zweite völlig Neubearbeitete und Erweiterte Auflage; Breil, G., Dawihl, W., Hänlein, W., Kaess, F., Koenig, P., Pirani, M., Ragoss, A., Reidt, E., Rummel, T., Schaidhauf, M., et al., Eds.; Springer: Berlin/Heidelberg, Germany, 1960; pp. 205–215. ISBN 978-3-642-92779-9. [Google Scholar]
- Lyon, D.A.; Keeney, R.M.; Cullen, J.F. The Electric Furnace in Metallurgical Work; Department of the Interior Bureau of Mines: Washington, DC, USA, 1914; pp. 1–216.
- Hahn, H. Chemische Untersuchung der beim Lösen des Roheisens entstehenden Producte. Eur. J. Org. Chem. 1864, 129, 57–77. [Google Scholar] [CrossRef]
- Hanson, D.; West, E.G. The Constitution of Copper-Iron-Silicon Alloys. Mon. J. Inst. Met. Metall. Abstr. 1934, 1, 95–116. [Google Scholar]
- Guertler, W.; Tammann, G. Über die Verbindungen des Eisens mit Silicium. XVII. Z. Anorg. Chem. 1905, 47, 163–179. [Google Scholar] [CrossRef] [Green Version]
- Yap, C.-P. A Critical Study of Some Iron-rich Iron-Silicon Alloys. J. Phys. Chem. 1933, 37, 951–967. [Google Scholar] [CrossRef]
- Weill, A.R. Structure of the Eta Phase of the Iron-Silicon System. Nature 1943, 152, 413. [Google Scholar] [CrossRef]
- Dodero, M. Sur l’électrolyse de mélanges fondus de fluosilicates alcalist et de fluorures ou d’oxydes. C. R. Acad. Sci. Paris 1939, 208, 799–801. [Google Scholar]
- Khalaff, K.; Schubert, K. Kristallstruktur von Fe2Si(h). J. Less Common Met. 1974, 35, 341–345. [Google Scholar] [CrossRef]
- Heinz, K. Die Kristallstruktur von Fe2Si, ihre Verwandtschaft zu den Ordnungsstrukturen des α-(Fe, Si)-Mischkristalls und zur Fe5Si3-Struktur. Z. Krist.-Cryst. Mater. 1977, 145, 177–189. [Google Scholar]
- Rix, W. Über Eisensilizide: Züchtung von ß-FeSi2-Einkristallen durch Chemischen Transport, Strukturelle und Physikalische Charakterisierung. Ph.D. Thesis, Albert-Ludwigs-Universität, Freiburg im Breisgau, Germany, 2001. [Google Scholar]
- Tang, C.P.; Tam, K.V.; Xiong, S.J.; Cao, J.; Zhang, X. The structure and electronic properties of hexagonal Fe2Si. AIP Adv. 2016, 6, 65317. [Google Scholar] [CrossRef] [Green Version]
- Tang, C.P.; Tam, K.V. The Study of Hexagonal Fe2Si: In Terms of Its Structure and Electronic Properties. In 21st Century Nanoscience—A Handbook; Sattler, K.D., Ed.; CRC Press: Boca Raton, FL, USA, 2020; pp. 20–21. [Google Scholar] [CrossRef]
- Ye, Z.; Gupta, S.; Kerkkonen, O.; Kanniala, R.; Sahajwalla, V. SiC and Ferro-silicides Formation in Tuyere Cokes. ISIJ Int. 2013, 53, 181–183. [Google Scholar] [CrossRef] [Green Version]
- Gornostayev, S.S.; Heikkinen, E.-P.; Heino, J.J.; Fabritius, T.M.J. Fe-Si particles on the surface of blast furnace coke. Int. J. Miner. Met. Mater. 2015, 22, 697–703. [Google Scholar] [CrossRef]
- Gornostayev, S.S.; Fabritius, T.M.J.; Kerkkonen, O.; Härkki, J.J. Fe-Si droplets associated with graphite on blast furnace coke. Int. J. Miner. Met. Mater. 2012, 19, 478–482. [Google Scholar] [CrossRef]
- Li, K.; Zhang, J.; Liu, Z.; Barati, M.; Zhong, J.; Wei, M.; Wang, G.; Jiao, K.; Yang, T. Interfaces Between Coke, Slag, and Metal in the Tuyere Level of a Blast Furnace. Met. Mater. Trans. A 2015, 46, 1104–1111. [Google Scholar] [CrossRef]
- Qian, W.; Huang, X. Invention of cast iron smelting in early China: Archaeological survey and numerical simulation. Adv. Archaeomater. 2021, 2, 4–14. [Google Scholar] [CrossRef]
- Schmidt, P.R. Science in Africa: A History of Ingenuity and Invention in African Iron Technology. In A Companion to African History, 1st ed.; Worger, W.H., Ambler, C.H., Achebe, N., Eds.; Wiley Blackwell: Hoboken, NJ, USA, 2018; pp. 267–288. ISBN 9781119063575. [Google Scholar]
- Schmidt, P.; Avery, D.H. Complex Iron Smelting and Prehistoric Culture in Tanzania. Science 1978, 201, 1085–1089. [Google Scholar] [CrossRef]
- Schmidt, P.R. Resisting homogenization and recovering variation and innovation in African iron smelting. Mediterr. Archaeol. 2001, 14, 219–227. [Google Scholar]
- Mineralogy Database. Minerals Arranged by the New Dana Classification: 01.01.23 Suessite Group Silicides. Available online: http://webmineral.com/dana/dana.php?class=01&subclass=01&group=23 (accessed on 19 October 2021).
- Anthony, J.W.; Bideaux, R.A.; Bladh, K.W.; Nichols, M.C. Handbook of Mineralogy. Available online: http://www.handbookofmineralogy.org/ (accessed on 23 November 2021).
- Mineralogy Database. Minerals Arranged by Nickel-Strunz (Version 10) Classification: 01.BB Silicides. Available online: http://webmineral.com/strunz/strunz.php?class=01&subclass=01.B&group=01.BB (accessed on 31 October 2021).
- mindat.org. Xifengite. Available online: https://www.mindat.org/min-4335.html (accessed on 31 October 2021).
- mindat.org. Gupeiite. Available online: https://www.mindat.org/min-1780.html (accessed on 31 October 2021).
- mindat.org. Hapkeite. Available online: https://www.mindat.org/min-26419.html (accessed on 31 October 2021).
- mindat.org. Suessite. Available online: https://www.mindat.org/min-3821.html (accessed on 31 October 2021).
- mindat.org. Luobusaite. Available online: https://www.mindat.org/min-7677.html (accessed on 31 October 2021).
- Wasson, J.; Wai, C.M. Composition of the metal, schreibersite and perryite of enstatite achondrites and the origin of enstatite chondrites and achondrites. Geochim. Cosmochim. Acta 1970, 34, 169–184. [Google Scholar] [CrossRef]
- Mineralogy Database. Gupeiite Mineral Data. Available online: http://webmineral.com/data/Gupeiite.shtml (accessed on 31 October 2021).
- Mineralogy Database. Hapkeite Mineral Data. Available online: http://webmineral.com/data/Hapkeite.shtml (accessed on 31 October 2021).
- Mineralogy Database. Luobusaite Mineral Data. Available online: http://webmineral.com/data/Luobusaite.shtml (accessed on 31 October 2021).
- Mineralogy Database. Suessite Mineral Data. Available online: http://webmineral.com/data/Suessite.shtml (accessed on 31 October 2021).
- Mineralogy Database. Xifengite Mineral Data. Available online: http://webmineral.com/data/Xifengite.shtml (accessed on 31 October 2021).
- Gopon, P.; Fournelle, J.; Sobol, P.E.; Llovet, X. Low-Voltage Electron-Probe Microanalysis of Fe–Si Compounds Using Soft X-Rays. Microsc. Microanal. 2013, 19, 1698–1708. [Google Scholar] [CrossRef] [Green Version]
- International Mineralogical Association. Commission on New Minerals, Nomeclature and Classification: The official IMA-CNMNC List of Mineral Names. Updated List of IMA-Approved Minerals. September 2021. Available online: http://cnmnc.main.jp/IMA_Master_List_%282021-09%29.pdf (accessed on 31 October 2021).
- mindat.org. Fersilicite Mineral Data: Naquite. Available online: https://www.mindat.org/min-40148.html (accessed on 31 October 2021).
- Mineralogy Database. Fersilicite Mineral Data. Available online: http://webmineral.com/data/Fersilicite.shtml (accessed on 31 October 2021).
- Mineralogy Database. Ferdisilicite Mineral Data. Available online: http://webmineral.com/data/Ferdisilicite.shtml (accessed on 31 October 2021).
- mindat.org. Linzhiite. Available online: https://www.mindat.org/min-40150.html (accessed on 31 October 2021).
- Yang, Z.; Wu, S.; Zhao, X.; Nguyen, M.C.; Yu, S.; Wen, T.; Tang, L.; Li, F.; Ho, K.-M.; Wang, C.-Z. Structures and magnetic properties of iron silicide from adaptive genetic algorithm and first-principles calculations. J. Appl. Phys. 2018, 124, 073901. [Google Scholar] [CrossRef]
- Wang, G.-M.; Zeng, W.; Xu, X.; Liu, W.-H.; Tang, B.; Fan, D.-H.; Liu, Q.-J.; Chang, X.-H.; Zhong, M. Effects of Pressure on Structural, Mechanical, and Electronic Properties and Stability of Fe x Si y Compounds. Phys. Status Solidi B 2020, 257, 2000083. [Google Scholar] [CrossRef]
- Macías Hemer, P.A. Estudio de Aleaciones de Fe1-xSx y Fe1-xSix a Condiciones del Núcleo Interno Terrestre. Tesis o trabajo de Investigación Presentada(o) como Requisito Parcial para optar al título de; Universidad del Norte: Barranquilla, Colombia, 2020. [Google Scholar]
- Nikolaychuk, P.A.; Tyurin, A.G. The estimation of Fe-Si system oxidation at 298 K in air and water environments. In Proceedings of the 11th International Conference on Fundamental and Applied Aspects of Physical Chemistry, Belgrade, Serbia, 24–28 September 2012; Anić, S., Draganić, I.G., Eds.; Society of Physical Chemists of Serbia: Belgrade, Serbia, 2012; pp. 37–39. ISBN 9788682475279. [Google Scholar]
- Atkinson, A. A theoretical analysis of the oxidation of FeSi alloys. Corros. Sci. 1982, 22, 87–102. [Google Scholar] [CrossRef]
- Moss, R.J.; Seymour, H.J. A Supposed New Mineral. Nature 1909, 81, 518. [Google Scholar] [CrossRef] [Green Version]
- Sutton, J.R. A new mineral? Nature 1911, 87, 314. [Google Scholar] [CrossRef]
- Spencer, L.J. Fictitious occurrences of iron silicide (ferrosilicon). Miner. Mag. J. Miner. Soc. 1935, 24, 160–164. [Google Scholar] [CrossRef]
- Conder, H. Geological pitfalls. Ind. Aust. Min. Stand. 1926, 76, 6. [Google Scholar]
- Minyuk, P.S.; Savva, N.E.; Subbotnikova, T.V. Magnetic Property of Exotic Iron Silicides. In Proceedings of the International Conference on Paleomagnetism and Rock Magnetism, Kazan, Russia, 2–7 October 2017; Fattakhova, L.A., Kuzina, D.M., Eds.; KFU Publishing House: Kazan, Russia, 2017; p. 64. [Google Scholar]
- Stępniewski, M.; Borucki, J. Pseudometeorite from Łapino (Pomerania, North Poland) Marian. Geol. Q. 2001, 45, 343–345. [Google Scholar]
- Kieu, N.; Gordillo-Vázquez, F.J.; Passas, M.; Sánchez, J.; Pérez-Invernón, F.J.; Luque, A.; Montanyá, J.; Christian, H. Submicrosecond Spectroscopy of Lightning-Like Discharges: Exploring New Time Regimes. Geophys. Res. Lett. 2020, 47, e2020GL088755. [Google Scholar] [CrossRef]
- Walker, T.D.; Christian, H.J. Triggered Lightning Spectroscopy: 2. A Quantitative Analysis. J. Geophys. Res. Atmos. 2019, 124, 3930–3942. [Google Scholar] [CrossRef]
- Kenny, G.G.; Pasek, M.A. The response of zircon to the extreme pressures and temperatures of a lightning strike. Sci. Rep. 2021, 11, 1560. [Google Scholar] [CrossRef]
- Chen, J.; Elmi, C.; Goldsby, D.; Gieré, R. Generation of shock lamellae and melting in rocks by lightning-induced shock waves and electrical heating. Geophys. Res. Lett. 2017, 44, 8757–8768. [Google Scholar] [CrossRef] [Green Version]
- Collins, S.; Melosh, H.J.; Pasek, M.A. Can Lightning Strikes Produce Shocked Quarz? In Proceedings of the 43rd Lunar and Planetary Science Conference, The Woodlands, TX, USA, 19–23 March 2012. [Google Scholar]
- Carter, E.A.; Hargreaves, M.D.; Kee, T.P.; Pasek, M.; Edwards, H.G.M. A Raman spectroscopic study of a fulgurite. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2010, 368, 3087–3097. [Google Scholar] [CrossRef] [Green Version]
- Walker, T.D.; Christian, H.J. Triggered lightning spectroscopy: Part 1. A qualitative analysis. J. Geophys. Res. Atmos. 2017, 122, 8000–8011. [Google Scholar] [CrossRef]
- Gilbert, L. Noch einiges von den Blitzröhren. Ann. Phys. 1819, 61, 249–262. [Google Scholar] [CrossRef] [Green Version]
- Merrill, G.P. On Fulgurites; United States National Museum: New York, NY, USA, 1886; pp. 83–91. [Google Scholar]
- García-Guinea, J.; Furió, M.; Fernandez-Hernan, M.; Bustillo, M.; Crespo-Feo, E.; Correcher, V.; Sánchez-Muñoz, L.; Matesanz, E. The Quartzofeldspathic Fulgurite of Bustarviejo (Madrid): Glassy Matrix and Silicon Phases; Universidad Complutense de Madrid: Madrid, Spain, 2009. [Google Scholar]
- Withering, W. On Fulgurites. Trans. Philos. Soc. Lond. 1790, 29, 3. [Google Scholar]
- Julien, A.A. A Study of the Structure of Fulgurites. J. Geol. 1901, 9, 673–693. [Google Scholar] [CrossRef]
- Fiedler, K.G. Ueber die Blitzröhren und ihre Entstehung. Ann. Phys. 1817, 55, 121–164. [Google Scholar] [CrossRef] [Green Version]
- Ende, M.; Schorr, S.; Kloess, G.; Franz, A.; Tovar, M. Shocked quartz in Sahara fulgurite. Eur. J. Miner. 2012, 24, 499–507. [Google Scholar] [CrossRef]
- Pye, K. Sem Observations on Some Sand Fulgurites from Northern Australia. J. Sediment. Res. 1982, 52, 991–998. [Google Scholar] [CrossRef]
- Feng, T.; Abbatiello, J.; Omran, A.; Mehta, C.; Pasek, M.A. Iron Silicides in Fulgurites. Minerals 2021, 11, 1394. [Google Scholar] [CrossRef]
- Harland, W.B.; Hacker, J. ‘Fossil’ lightning strikes 250 million years ago. Adv. Sci. 1966, 22, 663–671. [Google Scholar]
- Sponholz, B.; Baumhauer, R.; Felix-Hennignsen, P. Fulgurites in the southern central Sahara, Republic of Niger and their palaeoenvironmental significance. Holcene 1993, 3, 97–104. [Google Scholar] [CrossRef] [Green Version]
- Navarro-González, R.; Mahan, S.A.; Singhvi, A.K.; Navarro-Aceves, R.; Rajot, J.-L.; McKay, C.P.; Coll, P.; Raulin, F. Paleoecology reconstruction from trapped gases in a fulgurite from the late Pleistocene of the Libyan Desert. Geology 2007, 35, 171. [Google Scholar] [CrossRef]
- Thalheim, K. Die Bltzröhre im Staatlichen Museum für Mineralogie und Geologie zu Dresden. Lapis 1992, 17, 66–68. [Google Scholar]
- Wimmenauer, W. Vorkommen und Strukturen von Fulguriten im Schwarzwald. Aufschluss 2006, 57, 325–328. [Google Scholar]
- Rutley, F. On Fulgurite from Mont Blanc; with a Note on the Bouteillenstein, or Pseudo-chrysolite of Moldauthein, in Bohemia. Q. J. Geol. Soc. 1885, 41, 152–156. [Google Scholar] [CrossRef]
- Rutley, F. On Fulgurites from Monte Viso. Q. J. Geol. Soc. 1889, 45, 60–66. [Google Scholar] [CrossRef]
- Aston, E.; Bonney, T.G. On an Alpine Nickel-bearing Serpentine, with Fulgurites. Q. J. Geol. Soc. 1896, 52, 452–460. [Google Scholar] [CrossRef]
- Wimmenauer, W.; Himstedt, F. Dokumentation zum Thema: Fulgurite auf Felsen und Mauerwerk in Südwestdeutschland und Weiteren Fundgebieten: Geländebefunde und Lichtmikroskopische Untersuchungen: Further Findings of Fulgurites in the Black Forest and Other Regions; Universität Freiburg: Freiburg, Germany, 2012. [Google Scholar]
- Pasek, M.A.; Block, K.; Pasek, V. Fulgurite morphology: A classification scheme and clues to formation. Contrib. Miner. Pet. 2012, 164, 477–492. [Google Scholar] [CrossRef]
- Pasek, M.A.; Pasek, V.D. The forensics of fulgurite formation. Miner. Pet. 2017, 112, 185–198. [Google Scholar] [CrossRef]
- Block, K.M. Fulgurite Classification, Petrology, and Implications for Planetary Processes. Master’s Thesis, The University of Arizona, Tucson, AZ, USA, 2011. [Google Scholar]
- Roberts, S.; Sheffer, A.; McCanta, M.; Dyar, M.; Sklute, E. Oxidation state of iron in fulgurites and Trinitite: Implications for redox changes during abrupt high-temperature and pressure events. Geochim. Cosmochim. Acta 2019, 266, 332–350. [Google Scholar] [CrossRef]
- Ballhaus, C.; Wirth, R.; Fonseca, R.O.C.; Blanchard, H.; Pröll, W.; Bragagni, A.; Nagel, T.; Schreiber, A.; Dittrich, S.; Thome, V.; et al. Ultra-high pressure and ultra-reduced minerals in ophiolites may form by lightning strikes. Geochem. Perspect. Lett. 2017, 5, 42–46. [Google Scholar] [CrossRef]
- Ballhaus, C.; Helmy, H.M.; Fonseca, R.O.; Wirth, R.; Schreiber, A.; Jöns, N. Ultra-reduced phases in ophiolites cannot come from Earth’s mantle. Am. Miner. 2021, 106, 1053–1063. [Google Scholar] [CrossRef]
- Gieré, R.; Wimmenauer, W.; Müller-Sigmund, H.; Wirth, R.; Lumpkin, G.R.; Smith, K.L. Lightning-induced shock lamellae in quartz. Am. Miner. 2015, 100, 1645–1648. [Google Scholar] [CrossRef]
- Pasek, M.A.; Collins, G.S.; Carter, E.A.; Melosh, H.J.; Atlas, Z. Shocked Quartz in a Fulgurite. In Proceedings of the 73rd Annual Meteoritical Society Meeting, New York, NY, USA, 26–30 July 2010. [Google Scholar]
- Maki, D. Lightning strikes and prehistoric ovens: Determining the source of magnetic anomalies using techniques of environmental magnetism. Geoarchaeology 2005, 20, 449–459. [Google Scholar] [CrossRef] [Green Version]
- Jones, G.; Maki, D.L. Lightning-induced magnetic anomalies on archaeological sites. Archaeol. Prospect. 2005, 12, 191–197. [Google Scholar] [CrossRef]
- Burks, J.; Viberg, A.; Bevan, B. Lightning strikes in archaeological magnetometry data. A case study from the High Bank Works site, Ohio, USA. Archaeol. Pol. 2015, 53, 256–260. [Google Scholar]
- Sheffer, A.A.; Melosh, H.; Jarnot, B.; Lauretta, D. Reduction of Silicates at High Temperature: Fulgurites and Thermodynamic Modeling; Northern Trust: Chicago, IL, USA, 2003. [Google Scholar]
- Parnell, J.; Thackrey, S.; Muirhead, D.; Wright, A. Transient high-temperature processing of silicates in fulgurites as analogues for meteorite and impact melts. In Proceedings of the 39th Lunar and Planetary Science Conference, League City, TX, USA, 10–14 March 2008. Abstract #1286. [Google Scholar]
- Stefano, C.J.; Hackney, S.A.; Kampf, A.R. The occurrence of iron silicides in a fulgurite: Implications for fulgurite genesis. Can. Miner. 2020, 58, 115–123. [Google Scholar] [CrossRef]
- Ramírez Cardona, M.; Castro, K.F.; Cortès Garcia, P.P. Mineralogical study of binary iron silicides (Fe-Si systeM) in a fulgurite from Hidalgo, Mexico. Boletín Mineral. 2006, 17, 69–76. [Google Scholar]
- Sheffer, A.A. Chemical Reduction of Silicates by Meteorite Impacts and Lightning Strikes. Ph.D. Thesis, The University of Arizona, Tucson, AZ, USA, 2007. [Google Scholar]
- Garcia-Guinea, J.; Furio, M.; Fernandez-Hernan, M.; Bustillo, M.A.; Crespo-Feo, E.; Correcher, V.; Sanchez-Muñoz, L.; Matesanz, E.; Gucsik, A. The Quartzofeldspathic Fulgurite of Bustaviejo (Madrid): Cathodoluminescence and Raman Emission. In AIP Conference Proceedings: Micro-Raman Spectroscopy and Luminiscence Studies in the Earth and Planetary Sciences Micro-Raman Spectroscopy and Luminescence Studies in the Earth and Planetary Sciences: Proceedings of the International Conference Spectroscopy 2009, Mainz, Germany, 2–4 April 2009; AIP: University Park, ML, USA, 2009; pp. 128–134. [Google Scholar]
- Ballhaus, C.; Fonseca, R.; Bragagni, A. Reply to Comment on “Ultra-high pressure and ultra-reduced minerals in ophiolites may form by lightning strikes” by Griffin et al., 2018: No evidence for transition zone metamorphism in the Luobusa ophiolite. Geochem. Perspect. Lett. 2018, 7, 3–4. [Google Scholar] [CrossRef]
- Essene, E.J.; Fisher, D.C. Lightning Strike Fusion: Extreme Reduction and Metal-Silicate Liquid Immiscibility. Science 1986, 234, 189–193. [Google Scholar] [CrossRef]
- Miyahara, M.; Tomioka, N.; Bindi, L. Natural and experimental high-pressure, shock-produced terrestrial and extraterrestrial materials. Prog. Earth Planet. Sci. 2021, 8, 59. [Google Scholar] [CrossRef]
- MacDonald, F.A.; Mitchel, K.; Cina, S.E. Evidence for a Lightning-Strike Origin if the Edeowie Glass. In Proceedings of the Lunar and Planetary Science XXXV, Houston, TX, USA, 15–19 March 2004. [Google Scholar]
- Sheffer, A.A.; Dyar, M.D.; Sklute, E.C. Lightning Strike Glas as an Analog for Impact Glasses: 57Fe Mössbauer Spectroscopy of Fulgurites. In Proceedings of the Lunar and Planetary Science XXXVII, Houston, TX, USA, 13–17 March 2006. [Google Scholar]
- French, B.M. Traces of Catastrophe: A Handbook of Shock-Metamorphic Effects in Terrestrial Meteorite Impact Structures; Lunar and Planetary Institute: Houston, TX, USA, 1998. [Google Scholar]
- Eby, G.N.; Charnley, N.; Pirrie, D.; Hermes, R.; Smoliga, J.; Rollinson, G. Trinitite redux: Mineralogy and petrology. Am. Miner. 2015, 100, 427–441. [Google Scholar] [CrossRef]
- Foley, S.F. A Reappraisal of Redox Melting in the Earth’s Mantle as a Function of Tectonic Setting and Time. J. Pet. 2010, 52, 1363–1391. [Google Scholar] [CrossRef]
- Trønnes, R.; Baron, M.; Eigenmann, K.; Guren, M.; Heyn, B.; Løken, A.; Mohn, C. Core formation, mantle differentiation and core-mantle interaction within Earth and the terrestrial planets. Tectonophysics 2018, 760, 165–198. [Google Scholar] [CrossRef]
- Lin, Y.; van Westrenen, W. Oxygen as a catalyst in the Earth’s interior? Natl. Sci. Rev. 2021, 8, nwab009. [Google Scholar] [CrossRef]
- Mao, H.-K.; Hu, Q.; Yang, L.; Liu, J.; Kim, D.Y.; Meng, Y.; Zhang, L.; Prakapenka, V.B.; Yang, W.; Mao, W.L. When water meets iron at Earth’s core–mantle boundary. Natl. Sci. Rev. 2017, 4, 870–878. [Google Scholar] [CrossRef]
- Rohrbach, A.; Ballhaus, C.; Golla–Schindler, U.; Ulmer, P.; Kamenetsky, V.S.; Kuzmin, D.V. Metal saturation in the upper mantle. Nature 2007, 449, 456–458. [Google Scholar] [CrossRef]
- Ballhaus, C.; Berry, R.; Green, D.H. Oxygen fugacity controls in the Earth’s upper mantle. Nature 1990, 348, 437–440. [Google Scholar] [CrossRef]
- Armstrong, K. Redox Evolution of the Early Earth’s Mantle; University of Bayreuth: Bayreuth, Germany, 2018. [Google Scholar]
- Williams, H.M.; McCammon, C.A.; Peslier, A.H.; Halliday, A.N.; Teutsch, N.; Levasseur, S.; Burg, J.-P. Iron Isotope Fractionation and the Oxygen Fugacity of the Mantle. Science 2004, 304, 1656–1659. [Google Scholar] [CrossRef]
- Lukin, A.; Shestopalov, V. Ferrosilicide as indicator of mineral composition of the Earth mantle? Geofizicheskiy Zhurnal 2020, 42, 3–15. [Google Scholar] [CrossRef]
- Mergner, V.; Kupenko, I.; Spiekermann, G.; Petitgirard, S.; Libon, L.; Chariton, S.; Krug, M.; Steinbrügge, R.; Sergueev, I.; Sanchez-Valle, C. Sound Velocities in FeSi at Lower Mantle Conditions and the Origin of Ultralow-Velocity Zones. Geophys. Res. Lett. 2021, 48, e092257. [Google Scholar] [CrossRef]
- Dobrzhinetskaya, L.; Mukhin, P.; Wang, Q.; Wirth, R.; O’Bannon, E.; Zhao, W.; Eppelbaum, L.; Sokhonchuk, T. Moissanite (SiC) with metal-silicide and silicon inclusions from tuff of Israel: Raman spectroscopy and electron microscope studies. Lithos 2018, 310–311, 355–368. [Google Scholar] [CrossRef]
- Doglioni, C.; Tonarini, S.; Innocenti, F. Mantle wedge asymmetries and geochemical signatures along W- and E–NE-directed subduction zones. Lithos 2009, 113, 179–189. [Google Scholar] [CrossRef]
- Xiong, Q.; Griffin, W.L.; Huang, J.; Gain, S.; Toledo, V.; Pearson, N.J.; O’Reilly, S.Y. Super-reduced mineral assemblages in “ophiolitic” chromitites and peridotites: The view from Mount Carmel. Eur. J. Miner. 2017, 29, 557–570. [Google Scholar] [CrossRef]
- Griffin, W.L.; Gain, S.E.M.; Saunders, M.J.; Huang, J.-X.; Alard, O.; Toledo, V.; O’Reilly, S.Y. Immiscible metallic melts in the upper mantle beneath Mount Carmel, Israel: Silicides, phosphides and carbides. Am. Miner. 2021, 2021, 1–67. [Google Scholar] [CrossRef]
- Griffin, W.; Gain, S.; Adams, D.; Huang, J.-X.; Saunders, M.; Toledo, V.; Pearson, N.; O’Reilly, S. First terrestrial occurrence of tistarite (Ti2O3): Ultra-low oxygen fugacity in the upper mantle beneath Mount Carmel, Israel. Geology 2016, 44, 815–818. [Google Scholar] [CrossRef] [Green Version]
- Mathez, E.; Fogel, R.; Hutcheon, I.; Marshintsev, V. Carbon isotopic composition and origin of SiC from kimberlites of Yakutia, Russia. Geochim. Cosmochim. Acta 1995, 59, 781–791. [Google Scholar] [CrossRef]
- Nazzareni, S.; Nestola, F.; Zanon, V.; Bindi, L.; Scricciolo, E.; Petrelli, M.; Zanatta, M.; Mariotto, G.; Giuli, G. Discovery of moissanite in a peralkaline syenite from the Azores Islands. Lithos 2018, 324–325, 68–73. [Google Scholar] [CrossRef]
- Di Pierro, S.; Grobety, B.H.; Bernasconi, S.M.; Gnos, E.; Armbruster, T.; Ulmer, P. Rock-forming moissanite (natural α-silicon carbide). Am. Miner. 2003, 88, 1817–1821. [Google Scholar] [CrossRef]
- Shiryaev, A.; Griffin, W.L.; Stoyanov, E. Moissanite (SiC) from kimberlites: Polytypes, trace elements, inclusions and speculations on origin. Lithos 2011, 122, 152–164. [Google Scholar] [CrossRef]
- Gnoevaja, N.; Grozdanov, L. Moissanite from Triassic rocks, NW Bulgaria. Proc. Bulg. Geol. Soc. 1965, 26, 89–95. [Google Scholar]
- Pankov, V.Y.; Spetsius, Z.V. Inclusions of iron silicides and native silicon in moissanite from the Sytykan kimberlite pipe. Doklady Akademii Nauk SSSR 1989, 305, 704–708. [Google Scholar]
- Marshintsev, V.K. Nature of silicon carbide in kimberlite rocks of Yakutia. Mineralogicheskiy Zhurnal 1990, 12, 17–26. (In Russian) [Google Scholar]
- Jambor, J.L.; Grew, E.S. New mineral names. Am. Mineral. 1992, 77, 207–2013. [Google Scholar]
- Ashchepkov, I.; Logvinova, A.; Reimers, L.; Ntaflos, T.; Spetsius, Z.; Vladykin, N.; Downes, H.; Yudin, D.; Travin, A.; Makovchuk, I.; et al. The Sytykanskaya kimberlite pipe: Evidence from deep-seated xenoliths and xenocrysts for the evolution of the mantle beneath Alakit, Yakutia, Russia. Geosci. Front. 2015, 6, 687–714. [Google Scholar] [CrossRef] [Green Version]
- Ishimaru, S.; Arai, S.; Shukuno, H. Metal-saturated peridotite in the mantle wedge inferred from metal-bearing peridotite xenoliths from Avacha volcano, Kamchatka. Earth Planet. Sci. Lett. 2009, 284, 352–360. [Google Scholar] [CrossRef]
- Griffin, W.L.; Gain, S.; Huang, J.; Saunders, M.; Shaw, J.; Toledo, V.; O’Reilly, S.Y. A terrestrial magmatic hibonite-grossite-vanadium assemblage: Desilication and extreme reduction in a volcanic plumbing system, Mount Carmel, Israel. Am. Miner. 2019, 104, 207–219. [Google Scholar] [CrossRef]
- Griffin, W.L.; Gain, S.E.; Saunders, M.; Bindi, L.; Alard, O.; Toledo, V.; O’Reilly, S.Y. Parageneses of TiB2 in corundum xenoliths from Mt. Carmel, Israel: Siderophile behavior of boron under reducing conditions. Am. Miner. 2020, 105, 1609–1621. [Google Scholar] [CrossRef]
- Litasov, K.D.; Kagi, H.; Bekker, T.B. Enigmatic super-reduced phases in corundum from natural rocks: Possible contamination from artificial abrasive materials or metallurgical slags. Lithos 2019, 340–341, 181–190. [Google Scholar] [CrossRef]
- Litasov, K.; Bekker, T.; Kagi, H. Reply to the discussion of “Enigmatic super-reduced phases in corundum from natural rocks: Possible contamination from artificial abrasive materials or metallurgical slags” by Litasov et al. (Lithos, 340–341, pp. 181–190) by W.L. Griffin, V. Toledo and S.Y. O’Reilly. Lithos 2019, 348–349, 105170. [Google Scholar] [CrossRef]
- Griffin, W.L.; Toledo, V.; O’Reilly, S.Y. Discussion of “Enigmatic super-reduced phases in corundum from natural rocks: Possible contamination from artificial abrasive materials or metallurgical slags” by Litasov et al. (Lithos, 340–341, pp. 181–190). Lithos 2019, 348–349, 105122. [Google Scholar] [CrossRef]
- Xiong, F.; Xu, X.; Mugnaioli, E.; Gemmi, M.; Wirth, R.; Grew, E.S.; Robinson, P.T.; Yang, J. Two new minerals, badengzhuite, TiP, and zhiqinite, TiSi2, from the Cr-11 chromitite orebody, Luobusa ophiolite, Tibet, China: Is this evidence for super-reduced mantle-derived fluids? Eur. J. Miner. 2020, 32, 557–574. [Google Scholar] [CrossRef]
- Griffin, W.L.; Gain, S.E.M.; Bindi, L.; Toledo, V.; Cámara, F.; Saunders, M.; O’Reilly, S.Y. Carmeltazite, ZrAl2Ti4O11, a New Mineral Trapped in Corundum from Volcanic Rocks of Mt Carmel, Northern Israel. Minerals 2018, 8, 601. [Google Scholar] [CrossRef] [Green Version]
- Generalov, M.E.; Naumov, V.; Mokhov, A.V.; Trubkin, N.V. Isovite (Cr,Fe)23C6—A new mineral from the gold-platinum—Bearing placiers of the Urals. Zap. Vserossiskogo Mineral. Obs. 1998, 127, 26–37. (In Russian) [Google Scholar]
- Yang, J.; Wu, W.; Lian, D.; Rui, H. Peridotites, chromitites and diamonds in ophiolites. Nat. Rev. Earth Environ. 2021, 2, 198–212. [Google Scholar] [CrossRef]
- Zhou, M.-F.; Robinson, P.T.; Malpas, J.; Li, Z. Podiform Chromitites in the Luobusa Ophiolite (Southern Tibet): Implications for Melt-Rock Interaction and Chromite Segregation in the Upper Mantle. J. Pet. 1996, 37, 3–21. [Google Scholar] [CrossRef] [Green Version]
- Trumbull, R.B.; Yang, J.-S.; Robinson, P.T.; Di Pierro, S.; Vennemann, T.; Wiedenbeck, M. The carbon isotope composition of natural SiC (moissanite) from the Earth’s mantle: New discoveries from ophiolites. Lithos 2009, 113, 612–620. [Google Scholar] [CrossRef] [Green Version]
- Shi, N.; Bai, W.; Li, G.; Siong, M.; Yang, J.; Ma, Z.; Rong, H. Naquite, FeSi, a New Mineral Species from Luobusha, Tibet, Western China. Actsa Geol. Sin. 2012, 86, 533–538. [Google Scholar]
- Li, G.; Shi, N.; Xiong, M.; Ma, Z.; Bai, W.; Fang, Q. X-ray diffraction investigation of native Si-Fe alloy minerals from Luobusha, Tibet. Front. Earth Sci. China 2007, 1, 21–25. [Google Scholar] [CrossRef]
- Li, G.; Bai, W.; Shi, N.; Fang, Q.; Xiong, M.; Yang, J.; Ma, Z.; Rong, H. Linzhiite, FeSi2, a redefined and revalidated new mineral species from Luobusha, Tibet, China. Eur. J. Miner. 2012, 24, 1047–1052. [Google Scholar] [CrossRef]
- Tishchenko, A.I.; Kasatkin, A.V.; Shkoda, R. Silicides (naquite, linzhiite, luobusaite and zangboite) in Sarmatian limestones of Crimea. Noviye Danniye o Mineralakh 2016, 51, 30–37. (In Russian) [Google Scholar]
- Bai, W.; Robinson, P.T.; Fang, Q.; Yang, J.; Yan, B.; Zhang, Z.; Hu, X.-F.; Zhou, M.-F.; Malpas, J. The pge and base-metal alloys in the podiform chromitites of the luobusa ophiolite, Southern Tibet. Can. Miner. 2000, 38, 585–598. [Google Scholar] [CrossRef] [Green Version]
- Bai, W.J.; Yang, J.S.; Tao Shi, L.C.; Fang, Q.S.; Ma, Z.S.; Yan, B.G.; Xiong, M.; Dai, M.Q. Si-Fe alloy assemblage in ophiolite of Tibet and their genesis. Acta Petrol. Mineral. 2003, 22, 279–284, (In Chinese with English abstract). [Google Scholar]
- Bai, W.; Shi, N.; Fang, Q.; Li, G.; Xiong, M.; Yang, J.; Rong, H. Luobusaite: A new mineral. Acta Geol. Sin. 2006, 80, 656–659. [Google Scholar]
- Li Guowu; Fang Qingsong; Shi Nicheng; Bai Wenji; Yang Jingsui; Xiong Ming; Ma Zhesheng; Rong He. Zangboite, TiFeSi2, a new mineral species from Luobusha, Tibet, China, and its crystal structure. Can. Mineral. 2009, 47, 1265–1274. [CrossRef]
- Miyawaki, R.; Hatert, F.; Pasero, M.; Mills, S.J. New minerals and nomenclature modifications approved in 2019. Mineral. Mag. 2019, 83, 887–893. [Google Scholar] [CrossRef] [Green Version]
- Xu, X.; Yang, J.; Robinson, P.T.; Xiong, F.; Ba, D.; Guo, G. Origin of ultrahigh pressure and highly reduced minerals in podiform chromitites and associated mantle peridotites of the Luobusa ophiolite, Tibet. Gondwana Res. 2015, 27, 686–700. [Google Scholar] [CrossRef]
- Dobrzhinetskaya, L.F.; Wirth, R.; Yang, J.; Hutcheon, I.D.; Weber, P.K.; Green, H.W. High-pressure highly reduced nitrides and oxides from chromitite of a Tibetan ophiolite. Proc. Natl. Acad. Sci. USA 2009, 106, 19233–19238. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.-S.; Robinson, P.T.; Dilek, Y. Diamonds in Ophiolites. Elements 2014, 10, 127–130. [Google Scholar] [CrossRef]
- Griffin, W.L.; Howell, D.; Gonzalez-Jimenez, J.M.; Xiong, Q.; O’Reilly, S.Y. Comment on “Ultra-high pressure and ultra-reduced minerals in ophiolites may form by lightning strikes” by Ballhaus et al., 2017: Ultra-high pressure and super-reduced minerals in ophiolites do not form by lightning strikes. Geochem. Perspect. Lett. 2018, 8, 6–7. [Google Scholar] [CrossRef] [Green Version]
- Hu, X. Native fersilicite of China. Acta Petrol. Mineral. 1995, 14, 71–77. (In Chinese) [Google Scholar]
- He, D.; Gao, C.; Chen, C.; Liu, Y.; Hu, Z. SiC-dominated ultra-reduced mineral assemblage in carbonatitic xenoliths from the Dalihu basalt, Inner Mongolia, China. Am. Miner. 2017, 102, 312–320. [Google Scholar] [CrossRef]
- Liu, Y.; He, D.; Gao, C.; Foley, S.; Gao, S.; Hu, Z.; Zong, K.; Chen, H. First direct evidence of sedimentary carbonate recycling in subduction-related xenoliths. Sci. Rep. 2015, 5, 11547. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Meng, F.; Xu, X.; Robinson, P.T.; Dilek, Y.; Makeyev, A.B.; Wirth, R.; Wiedenbeck, M.; Cliff, J. Diamonds, native elements and metal alloys from chromitites of the Ray-Iz ophiolite of the Polar Urals. Gondwana Res. 2014, 27, 459–485. [Google Scholar] [CrossRef]
- Nikulova, N.Y.; Kozyreva, I.V. Moissanite, Native Si and Iron Silicide from the Lower Paleozoic sandstones of the Polar Urals. Vestn. Inst. Geol. Komi 2015, 193, 17–18. [Google Scholar]
- Batovrin, S.; Lipovsky, B.; Gulbin, Y.; Pushkarev, Y.; Shukolyukov, Y.A.; Brownlee, D. Constraints on the origins of iron silicide spherules in ultrahigh-temperature distal impact ejecta. Meteorit. Planet. Sci. 2021, 56, 1369–1405. [Google Scholar] [CrossRef]
- Lei, Z. Ti-rich Minerals and a New Variety—Chromian Kennedyite from Kimberlites in Eastern China. Geol. Sci. Technol. Inf. 1991, 10, 71–76. [Google Scholar]
- Hu, X. Natural silica-iron ore in China. Journal of Rock Mineralogy. J. Rock Mineral. 1995, 14, 71–78. [Google Scholar]
- Wei, J.; Zheng, Z.; Chen, S. A study on ferdisilicite and its geological implications. Acta Mineral. Sin. 1985, 2, 184–186. [Google Scholar]
- Zhang, R.; Wang, Z.; Wang, Y. The discovery of ferdisilicite in Anhui, China. J. Chengdu Coll. Geol. 1985, 4, 48–52. [Google Scholar]
- Liu, Q.; Tan, Q.; Jiang, Y.; Han, C.; Sun, Y.; Zhu, C. First discovery of ferdisilicite-silicon interlocking mineral gneiss of Jiaodong group of China. Mar. Geol. Quart. Geol. 1995, 4, 107–112, (In Chinese with English abstract). [Google Scholar]
- Hu, X. A preliminary study on ferrosilicium from the Proterozoic, southwestern Zhejiang Province. Acta Mineral. Sin. 1991, 11, 285–289, (In Chinese with English abstract). [Google Scholar]
- Marchi, S.; Drabon, N.; Schulz, T.; Schaefer, L.; Nesvorny, D.; Bottke, W.F.; Koeberl, C.; Lyons, T. Delayed and variable late Archaean atmospheric oxidation due to high collision rates on Earth. Nat. Geosci. 2021, 14, 827–831. [Google Scholar] [CrossRef]
- Bottke, W.F.; Vokrouhlický, D.; Minton, D.A.; Nesvorný, D.; Morbidelli, A.; Brasser, R.; Simonson, B.; Levison, H.F. An Archaean heavy bombardment from a destabilized extension of the asteroid belt. Nature 2012, 485, 78–81. [Google Scholar] [CrossRef] [PubMed]
- Lehmer, O.R.; Catling, D.C.; Buick, R.; Brownlee, D.E.; Newport, S. Atmospheric CO2 levels from 2.7 billion years ago inferred from micrometeorite oxidation. Sci. Adv. 2020, 6, eaay4644. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yakymchuk, C.; Kirkland, C.L.; Cavosie, A.J.; Szilas, K.; Hollis, J.; Gardiner, N.J.; Waterton, P.; Steenfelt, A.; Martin, L. Stirred not shaken; critical evaluation of a proposed Archean meteorite impact in West Greenland. Earth Planet. Sci. Lett. 2021, 557, 116730. [Google Scholar] [CrossRef]
- Novgorodova, M.I.; Yusupov, R.G.; Dmitrieva, M.T.; Tsepin, A.I.; Sivtsov, A.V.; Gorshkov, A.I. Khamrabaevite, (Ti,V,Fe)C, a new mineral. Zapiski Vsesoyuznogo Mineralogicheskogo Obshchestva 1984, 133, 697–703. [Google Scholar]
- Novgorodova, M.I.; Yusupov, R.G.; Dmitrieva, M.T.; Tsepin, A.I.; Sivtsov, A.V.; Gorshkov, A.I.; Korovushkin, V.V.; Yakubovskaya, N.Y. First Occurrence of Suessite on the Earth. Int. Geol. Rev. 1984, 26, 98–101. [Google Scholar] [CrossRef]
- Yusupov, R.G.; Dzhenchuraev, D.D.; Radzhabov, F.F. Accessory native chromium and a natural compound of the series Fe-Cr-Si in rocks of the Gavasai ore field. Izvest. Akad. Nauk Kirgiz SSR 1982, 5, 25–26. (In Russian) [Google Scholar]
- Dunn, P.L.; Grice, J.D.; Fleischer, M.; Pabst, A. New Mineral Names. Am. Mineral. 1984, 69, 210–215. [Google Scholar]
- Novoseleva, L.N. Iron silicides in Lower Cambrian limestones on the Bazaikha River (tributary of the Yenisei), Krasnoyarsk region. Zap. Vsesoyuznogo Mineral.-Kogo Obs. 1975, 104, 228–234. [Google Scholar]
- Novoselova, L.N.; Bagdasarov, E.A. New data on iron silicides. Int. Geol. Rev. 1980, 22, 691–696. [Google Scholar] [CrossRef]
- Novoselova, L.N.; Lyul’, A.Y. Geochemical characteristics of iron silicides from lower cambrian reefal limestones, krasnoyarsk. Int. Geol. Rev. 1986, 28, 114–118. [Google Scholar] [CrossRef]
- Novoselova, L.N.; Bagdasarov, E.A. Novye dannye o silitsidakh zheleza: New data on iron silicides. Zap. Vsesoyuznogo Mineral. Obs. 1979, 108, 326–333. [Google Scholar]
- Novoselova, L.N.; Sokhor, M.I. Moissanite and iron silicides from Lower Cambrian carbonate rocks in the Altai-Sayan region. Zap. Vses. Min. Obshch. 1983, 5, 582–588. [Google Scholar]
- Eremenko, G.K.; Polkanov, Y.A.; Gevork’yan, V.K. Cosmogenic minerals in the Poltava deposits of the Konka–Yalynsk depression in northern Azov Region. Mineral. Osadochnih Obraz. 1974, 1, 66–75. (In Russian) [Google Scholar]
- Tishchenko, A.I.; Kasatkin, A.V.; Shkoda, R. Silicides (naguite, linjia, lobusaite, and cangpoite in Crimean Sarmatian limestone. Novye Dannye O Miner. 2016, 51, 30–37. [Google Scholar]
- Muszer, A. Silicide spherules from Permian sediments of the Fore-Sudetic Monocline (SW Poland). Physicochem. Probl. Miner. Process. 2014, 50, 107–118. [Google Scholar] [CrossRef]
- Khabibullaeva, G.; Dunin-Barkovskaya, E. Rare Accessory Mineral Seussite—Fe3Si From Tien Shan and Conditions of its Formation. Int. J. Geol. Earth Environ. Sci. 2017, 7, 42–44. [Google Scholar]
- Makarova, N.N. O nakhodke redkogo minerala ferdisilitsita v Karpatakh.: The finding of the mineral ferdisilicite in the Carpathians. Zap. Vsesoyuznogo Mineral. Obs. 1977, 2, 236–237. [Google Scholar]
- Jakabská, K. Sférolity z ryolitu z oblasti Banská Štiavnica. Acta Montan. Slovaca 1998, 3, 71–74. [Google Scholar]
- Demko, R.; Kubiš, M.; Bazarnik, J. Petrológia a geochémia acidného aplitu z hodrušsko-štiavnického intruzívneho komplexu z okolia Rumplovskej: Petrology and geochemistry of acid aplites from the Hodruša-Štiavnica intrusive complex in the Rumplovská area. Miner. Slovaca 2011, 43, 215–226. [Google Scholar]
- Elekes, Z. Ion Beam Based Nuclear Microanalysis of Geological and Archaeological Objects. Ph.D. Thesis, University of Debrecen, Debrecen, Hungary, 2001. [Google Scholar]
- Jakabská, K.; Rozložník, L. Spherical accessories “spherules” in Gemeric Granites (West Carpathians—Czechoslovakia). Geol. Zborník—Geol. Carpathica 1989, 40, 305–322. [Google Scholar]
- Tchaikovsky, I.; Korotchenkova, O.V. Explosive mineral phases diamondiferous visherites of the Western Urals. Lithosphäre 2012, 2012, 125–140. [Google Scholar]
- Glavatskikh, S.F. Metal formation in the exhalation products of the Great Fissure Tolbachik Eruption, Kamchatka. Vulkanol. Seismol. 1995, 4–5, 193–214. [Google Scholar]
- Karpov, G.A.; Silaev, V.I.; Anikin, L.P.; Rakin, V.I.; Vasilev, E.; Filatov, S.K.; Petrovskii, V.A.; Flerov, G.B. Diamonds and accessory minerals in products of the 2012–2013 Tolbachik Fissure Eruption. J. Volcanol. Seism. 2014, 8, 323–339. [Google Scholar] [CrossRef]
- Litasov, K.D.; Kagi, H.; Bekker, T.B.; Makino, Y.; Hirata, T.; Brazhkin, V.V. Why Tolbachik Diamonds Cannot be Natural. Am. Miner. 2021, 106, 44–53. [Google Scholar] [CrossRef]
- Howell, D.; Griffin, W.L.; Yang, J.; Gain, S.; Stern, R.; Huang, J.; Jacob, D.; Xu, X.; Stokes, A.; O’Reilly, S.Y.; et al. Diamonds in ophiolites: Contamination or a new diamond growth environment? Earth Planet. Sci. Lett. 2015, 430, 284–295. [Google Scholar] [CrossRef]
- Levitskiy, V.I.; Solodilova, V.V.; Zavadich, N.S.; Pavlova, L.A.; Spetsius, Z.V.; Levitskiy, I.V. Genetic Nature of Mineralization with Native and Intermetallic Compounds in the Bobruisk Ring Structure (Republic of Belarus). Doklady Akademii Nauk SSSR 2018, 481, 857–861. [Google Scholar] [CrossRef]
- Lukin, A.E.; Novgorodova, M.I. On finds of ferro-silicide of extraterrestrial origin. Doklady Akademii Nauk SSSR 1994, 334, 73–76. (In Russian) [Google Scholar]
- Lukin, A.E. On the origin of the Shungite. Geologicheskiy Zhurnal 2005, 4, 28–47. (In Russian) [Google Scholar]
- Safonov, Y.; Belov, A.N.; Galyamov, A.L.; Genkin, A.D.; Podlesski, K.V. Native Metals, Carbides and Nitrides in Magmatic Breccias of the Voronezh Massif, Their Nature and Metallogenic Significance; Informations Bulletin RFBR; Russian Foundation for Basic Information: Moscow, Russia, 1995. (In Russian)
- Nakamura-Messenger, K.; Keller, L.P.; Clemett, S.J.; Messenger, S.; Jones, J.H.; Palma, R.L.; Pepin, R.O.; Klöck, W.; Zolensky, M.; Tatsuoka, H. Brownleeite: A new manganese silicide mineral in an interplanetary dust particle. Am. Miner. 2010, 95, 221–228. [Google Scholar] [CrossRef]
- mindat.org. Brownleeite Mineral Data: Brownleeite. Available online: https://www.mindat.org/min-36014.html (accessed on 11 May 2021).
- Tatarintsev, V.I.; Tsymbal, S.N.; Sandamirskaya, S.M.; Egorova, L.N.; Vashchenko, A.N.; Khnyazkov, A.P. Iron-bearing manganese silicides from the Priazovye (USSR). Mineral Zhurnal 1990, 12, 35–43. (In Russian) [Google Scholar]
- Mineralogy Database. Brownleeite Mineral Data. Available online: http://webmineral.com/data/Brownleeite.shtml (accessed on 11 May 2021).
- Yusupov, R.G.; Stanley, C.J.; Welch, M.D.; Spratt, J.; Cressey, G.; Rumsey, M.S.; Seltmann, R.; Igamberdiev, E. Mavlyanovite, Mn5Si3: A new mineral species from a lamproite diatreme, Chatkal Ridge, Uzbekistan. Miner. Mag. 2009, 73, 43–50. [Google Scholar] [CrossRef]
- mindat.org. UM1990-56-Si:FeMn. Available online: https://www.mindat.org/min-51742.html (accessed on 28 November 2021).
- mindat.org. Perryite. Available online: https://www.mindat.org/min-3170.html (accessed on 28 November 2021).
- Buchwald, V. Handbook of Iron Meteorites; Center for Meteorite Studies: Tempe, AZ, USA, 1975. [Google Scholar]
- Cabri, L.J.; McDonald, A.M.; Stanley, C.J.; Rudashevsky, N.S.; Poirier, G.; Wilhelmij, H.R.; Zhe, W.; Rudashevsky, V.N. Palladosilicide, Pd2Si, a new mineral from the Kapalagulu Intrusion, Western Tanzania and the Bushveld Complex, South Africa. Miner. Mag. 2015, 79, 295–307. [Google Scholar] [CrossRef] [Green Version]
- Fischer, R.A.; Campbell, A.J.; Caracas, R. Equations of state in the Fe-FeSi system at high pressures and temperatures. J. Geophys. Res. Solid Earth 2014, 119, 2810–2827. [Google Scholar] [CrossRef]
- Shahar, A.; Ziegler, K.; Young, E.; Ricolleau, A.; Schauble, E.A.; Fei, Y. Experimentally determined Si isotope fractionation between silicate and Fe metal and implications for Earth’s core formation. Earth Planet. Sci. Lett. 2009, 288, 228–234. [Google Scholar] [CrossRef]
- Fischer, R.A.; Campbell, A.J.; Caracas, R.; Reaman, D.M.; Dera, P.; Prakapenka, V.B. Equation of state and phase diagram of Fe–16Si alloy as a candidate component of Earth’s core. Earth Planet. Sci. Lett. 2012, 357–358, 268–276. [Google Scholar] [CrossRef]
- Morard, G.; Andrault, D.; Guignot, N.; Siebert, J.; Garbarino, G.; Antonangeli, D. Melting of Fe–Ni–Si and Fe–Ni–S alloys at megabar pressures: Implications for the core–mantle boundary temperature. Phys. Chem. Miner. 2011, 38, 767–776. [Google Scholar] [CrossRef]
- Tagawa, S.; Ohta, K.; Hirose, K.; Kato, C.; Ohishi, Y. Compression of Fe-Si-H alloys to core pressures. Geophys. Res. Lett. 2016, 43, 3686–3692. [Google Scholar] [CrossRef] [Green Version]
- Komabayashi, T.; Pesce, G.; Sinmyo, R.; Kawazoe, T.; Breton, H.; Shimoyama, Y.; Glazyrin, K.; Konôpková, Z.; Mezouar, M. Phase relations in the system Fe–Ni–Si to 200 GPa and 3900 K and implications for Earth’s core. Earth Planet. Sci. Lett. 2019, 512, 83–88. [Google Scholar] [CrossRef]
- Morard, G.; Katsura, T. Pressure–temperature cartography of Fe–S–Si immiscible system. Geochim. Cosmochim. Acta 2010, 74, 3659–3667. [Google Scholar] [CrossRef]
- Ozawa, H.; Hirose, K.; Yonemitsu, K.; Ohishi, Y. High-pressure melting experiments on Fe–Si alloys and implications for silicon as a light element in the core. Earth Planet. Sci. Lett. 2016, 456, 47–54. [Google Scholar] [CrossRef]
- Brosh, E.; Makov, G.; Shneck, R.Z. Thermodynamic analysis of high-pressure phase equilibria in Fe–Si alloys, implications for the inner-core. Phys. Earth Planet. Inter. 2009, 172, 289–298. [Google Scholar] [CrossRef]
- Lin, J.-F.; Scott, H.P.; Fischer, R.A.; Chang, Y.-Y.; Kantor, I.; Prakapenka, V.B. Phase relations of Fe-Si alloy in Earth’s core. Geophys. Res. Lett. 2009, 36, e036990. [Google Scholar] [CrossRef] [Green Version]
- Santamaria-Perez, D.; Errandonea, D.; Vegas, A.; Nuss, J.; Jansen, M.; Rodríguez-Hernández, P.; Munoz, A.; Boehler, R. Phase diagram studies on iron and nickel silicides: High-pressure experiments andab initiocalculations. J. Phys. Conf. Ser. 2008, 121, 022013. [Google Scholar] [CrossRef]
- Ikuta, D.; Ohtani, E.; Hirao, N. Two-phase mixture of iron–nickel–silicon alloys in the Earth’s inner core. Commun. Earth Environ. 2021, 126, 225. [Google Scholar] [CrossRef]
- Brennan, M.C.; Fischer, R.A.; Couper, S.; Miyagi, L.; Antonangeli, D.; Morard, G. High-Pressure Deformation of Iron–Nickel–Silicon Alloys and Implications for Earth’s Inner Core. J. Geophys. Res. Solid Earth 2021, 126, e2020JB021077. [Google Scholar] [CrossRef]
- Berrada, M.; Secco, R.A. Review of Electrical Resistivity Measurements and Calculations of Fe and Fe-Alloys Relating to Planetary Cores. Front. Earth Sci. 2021, 9, e732289. [Google Scholar] [CrossRef]
- Errandonea, D.; Santamaría-Perez, D.; Vegas, A.; Nuss, J.; Jansen, M.; Rodríguez-Hernandez, P.; Muñoz, A. Structural stability of Fe5Si3 and Ni2Si studied by high-pressure x-ray diffraction andab initiototal-energy calculations. Phys. Rev. B 2008, 77, 094113. [Google Scholar] [CrossRef] [Green Version]
- Caracas, R.; Wentzcovitch, R. Equation of state and elasticity of FeSi. Geophys. Res. Lett. 2004, 31, L20603. [Google Scholar] [CrossRef] [Green Version]
- Lord, O.T.; Walter, M.J.; Dobson, D.; Armstrong, L.; Clark, S.; Kleppe, A. The FeSi phase diagram to 150 GPa. J. Geophys. Res. Earth Surf. 2010, 115, e006528. [Google Scholar] [CrossRef] [Green Version]
- Spiekermann, G.; Kupenko, I.; Petitgirard, S.; Harder, M.; Nyrow, A.; Weis, C.; Albers, C.; Biedermann, N.; Libon, L.; Sahle, C.J.; et al. A portable on-axis laser-heating system for near-90° X-ray spectroscopy: Application to ferropericlase and iron silicide. J. Synchrotron Radiat. 2020, 27, 414–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Edmund, E. The Elasticity of Iron-Alloys at Extreme Conditions. Ph.D. Thesis, Sorbonne Université, Paris, France, 2018. [Google Scholar]
- Dobson, D.P.; Vočadlo, L.; Wood, I.G. A new high-pressure phase of FeSi. Am. Miner. 2002, 87, 784–787. [Google Scholar] [CrossRef]
- Zhang, F.; Oganov, A.R. Iron silicides at pressures of the Earth’s inner core. Geophys. Res. Lett. 2010, 37, e041224. [Google Scholar] [CrossRef] [Green Version]
- McGuire, C.P.; Kavner, A.; Santamaria, D. High-pressure high-temperature behavior of iron silicide (Fe5Si3) to 58 GPa and 2400 K. In Proceedings of the AGU Fall Meeting, San Francisco, CA, USA, 14–18 December 2015. [Google Scholar]
- Badro, J.; Côté, A.; Brodholt, J. A seismologically consistent compositional model of Earth’s core. Proc. Natl. Acad. Sci. USA 2014, 111, 7542–7545. [Google Scholar] [CrossRef] [Green Version]
- Tateno, S.; Kuwayama, Y.; Hirose, K.; Ohishi, Y. The structure of Fe–Si alloy in Earth’s inner core. Earth Planet. Sci. Lett. 2015, 418, 11–19. [Google Scholar] [CrossRef]
- Guyot, F.; Zhang, J.; Martinez, I.; Matas, J.; Ricard, Y.; Javoy, M. P-V-T measurements of iron suicide (ε-FeSi) Implications for silicate-metal interactions in the early Earth. Eur. J. Miner. 1997, 9, 277–286. [Google Scholar] [CrossRef]
- Wann, E.T.H.; Vočadlo, L.; Wood, I.G. High-temperature ab initio calculations on FeSi and NiSi at conditions relevant to small planetary cores. Phys. Chem. Miner. 2017, 44, 477–484. [Google Scholar] [CrossRef] [Green Version]
- Wann, E.T.H. The Core Composition of Terrestrial Planets: A Study of the Ternary Fe-Ni-Si System. Ph.D. Thesis, University College London, London, UK, 2015. [Google Scholar]
- Terasaki, H.; Shibazaki, Y.; Sakamaki, T.; Tateyama, R.; Ohtani, E.; Funakoshi, K.-I.; Higo, Y. Hydrogenation of FeSi under high pressure. Am. Miner. 2010, 96, 93–99. [Google Scholar] [CrossRef]
- Dobson, D.; Hunt, S.; Ahmed, J.; Lord, O.; Wann, E.T.; Santangeli, J.; Wood, I.G.; Vočadlo, L.; Walker, A.M.; Thomson, A.; et al. The phase diagram of NiSi under the conditions of small planetary interiors. Phys. Earth Planet. Inter. 2016, 261, 196–206. [Google Scholar] [CrossRef] [Green Version]
- Deng, L.; Kono, Y.; Shen, G. Sound wave velocities of Fe5Si at high-pressure and high-temperature conditions: Implications to lunar and planetary cores. Am. Miner. 2019, 104, 291–299. [Google Scholar] [CrossRef]
- Berrada, M.; Secco, R.A.; Yong, W. Adiabatic heat flow in Mercury’s core from electrical resistivity measurements of liquid Fe-8.5 wt%Si to 24 GPa. Earth Planet. Sci. Lett. 2021, 568, 117053. [Google Scholar] [CrossRef]
- Genova, A.; Goossens, S.; Mazarico, E.; Lemoine, F.G.; Neumann, G.A.; Kuang, W.; Sabaka, T.J.; Hauck, I.S.A.; Smith, D.E.; Solomon, S.C.; et al. Geodetic Evidence That Mercury Has A Solid Inner Core. Geophys. Res. Lett. 2019, 46, 3625–3633. [Google Scholar] [CrossRef]
- Tao, R.; Fei, Y. High-pressure experimental constraints of partitioning behavior of Si and S at the Mercury’s inner core boundary. Earth Planet. Sci. Lett. 2021, 562, 116849. [Google Scholar] [CrossRef]
- Brennan, M.C.; Fischer, R.A.; Irving, J.C. Core formation and geophysical properties of Mars. Earth Planet. Sci. Lett. 2019, 530, 115923. [Google Scholar] [CrossRef] [Green Version]
- Dumoulin, C.; Tobie, G.; Verhoeven, O.; Rosenblatt, P.; Rambaux, N. Tidal constraints on the interior of Venus. J. Geophys. Res. Planets 2017, 122, 1338–1352. [Google Scholar] [CrossRef]
- Berrada, M.; Secco, R.A.; Yong, W.; Littleton, J.A.H. Electrical Resistivity Measurements of Fe-Si With Implications for the Early Lunar Dynamo. J. Geophys. Res. Planets 2020, 125, e2020JE006380. [Google Scholar] [CrossRef]
- Plotnykov, M.; Valencia, D. Chemical fingerprints of formation in rocky super-Earths’ data. Mon. Not. R. Astron. Soc. 2020, 499, 932–947. [Google Scholar] [CrossRef]
- Fulton, B.J.; Petigura, E.A.; Howard, A.W.; Isaacson, H.; Marcy, G.W.; Cargile, P.A.; Hebb, L.; Weiss, L.M.; Johnson, J.A.; Morton, T.D.; et al. The California-KeplerSurvey. III. A Gap in the Radius Distribution of Small Planets. Astron. J. 2017, 154, 109. [Google Scholar] [CrossRef] [Green Version]
- Dorn, C.; Hinkel, N.R.; Venturini, J. Bayesian analysis of interiors of HD 219134b, Kepler-10b, Kepler-93b, CoRoT-7b, 55 Cnc e, and HD 97658b using stellar abundance proxies. Astron. Astrophys. 2016, 597, A38. [Google Scholar] [CrossRef] [Green Version]
- Nakamura-Messenger, K.; Zolensky, M.E.; Keller, L.P. New Manganese Silicide Mineral Phase in an Interplanetary Dust Particle. In Proceedings of the 39th Lunar and Planetary Science Conference, League City, TX, USA, 10–14 March 2008; Lunar and Planetary Institute: Houston, TX, USA, 2008. Abstract #2013. [Google Scholar]
- Rietmeijer, F.J.M.; Nakamura, T.; Tsuchiyama, A.; Uesugi, K.; Nakano, T.; Leroux, H. Origin and formation of iron silicide phases in the aerogel of the Stardust mission. Meteorit. Planet. Sci. 2008, 43, 121–134. [Google Scholar] [CrossRef]
- Nakamura, T.; Tsuchiyama, A.; Akaki, T.; Uesugi, K.; Nakano, T.; Takeuchi, A.; Suzuki, Y.; Noguchi, T. Bulk mineralogy and three-dimensional structures of individual Stardust particles deduced from synchrotron X-ray diffraction and microtomography analysis. Meteorit. Planet. Sci. 2008, 43, 247–259. [Google Scholar] [CrossRef]
- Morello, S.O.; Anesa, J. Hallazgo de siliciuros de Fe en el meteorito El Aybal, Salta. 5°. In Proceedings of the Congreso de Mineralogía y Metalogenia, Actas, La Plata, Argentina, 23–25 October 2000; pp. 495–496. [Google Scholar]
- Acevedo, R.D. Catalogue of Meteorites from South America, 1st ed.; Springer: Cham, Switzerland, 2014; ISBN 3319019252. [Google Scholar]
- Hazen, R.M. Paleomineralogy of the Hadean Eon: A preliminary species list. Am. J. Sci. 2013, 313, 807–843. [Google Scholar] [CrossRef]
- Morrison, S.M.; Hazen, R.M. An evolutionary system of mineralogy, Part IV: Planetesimal differentiation and impact mineralization (4566 to 4560 Ma). Am. Miner. 2021, 106, 730–761. [Google Scholar] [CrossRef]
- Matsuno, J.; Tsuchiyama, A.; Watanabe, T.; Tanaka, M.; Takigawa, A.; Enju, S.; Koike, C.; Chihara, H.; Miyake, A. Condensation of Glass with Multimetal Nanoparticles: Implications for the Formation Process of GEMS Grains. Astrophys. J. Lett. 2021, 911, 47. [Google Scholar] [CrossRef]
- Shepard, C.U. Examination of a supposed meteoric iron, found near Rutherfordton, North Carolina. Am. J. Sci. Arts 1859, XXVIII, 259–270. [Google Scholar]
- Rammelsberg, C.F. XII. Über einige nordamerikanische Meteoriten. J. Prakt. Chem. 1862, 85, 83–88. [Google Scholar] [CrossRef] [Green Version]
- Pieters, C.M.; Taylor, L.A.; Noble, S.K.; Keller, L.P.; Hapke, B.; Morris, R.V.; Allen, C.C.; McKAY, D.S.; Wentworth, S. Space weathering on airless bodies: Resolving a mystery with lunar samples. Meteorit. Planet. Sci. 2000, 35, 1101–1107. [Google Scholar] [CrossRef]
- Hapke, B. Space weathering from Mercury to the asteroid belt. J. Geophys. Res. Earth Surf. 2001, 106, 10039–10073. [Google Scholar] [CrossRef]
- Lucey, P.; Korotev, R.L.; Gillis, J.J.; Taylor, L.A.; Lawrence, D.; Campbell, B.A.; Elphic, R.; Feldman, B.; Hood, L.L.; Hunten, D.; et al. Understanding the Lunar Surface and Space-Moon Interactions. Rev. Miner. Geochem. 2006, 60, 83–219. [Google Scholar] [CrossRef]
- Chapman, C.R. Space weathering of asteroid surfaces. Annu. Rev. Earth Planet. Sci. 2004, 32, 539–567. [Google Scholar] [CrossRef] [Green Version]
- Noble, S.K.; Keller, L.P.; Pieters, C.M. Evidence of space weathering in regolith breccias II: Asteroidal regolith breccias. Meteorit. Planet. Sci. 2010, 45, 2007–2015. [Google Scholar] [CrossRef] [Green Version]
- Noguchi, T.; Nakamura, T.; Kimura, M.; Zolensky, M.E.; Tanaka, M.; Hashimoto, T.; Konno, M.; Nakato, A.; Ogami, T.; Fujimura, A.; et al. Incipient Space Weathering Observed on the Surface of Itokawa Dust Particles. Science 2011, 333, 1121–1125. [Google Scholar] [CrossRef] [PubMed]
- Pieters, C.M.; Ammannito, E.; Blewett, D.T.; Denevi, B.W.; De Sanctis, M.C.; Gaffey, M.J.; Le Corre, L.; Li, J.-Y.; Marchi, S.; McCord, T.B.; et al. Distinctive space weathering on Vesta from regolith mixing processes. Nature 2012, 491, 79–82. [Google Scholar] [CrossRef]
- Thompson, M.S.; Christoffersen, R.; Zega, T.J.; Keller, L.P. Microchemical and structural evidence for space weathering in soils from asteroid Itokawa. Earth Planets Space 2014, 66, 89. [Google Scholar] [CrossRef] [Green Version]
- Hoffmann, H. Space Weathering. In Encyclopedia of Astrobiology; Gargaud, M., Amils, R., Quintanilla, J.C., Cleaves, H.J., Irvine, W.M., Pinti, D.L., Viso, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 1543–1544. ISBN 978-3-642-11271-3. [Google Scholar]
- Thompson, M.S.; Zega, T.J.; Becerra, P.; Keane, J.T.; Byrne, S. The oxidation state of nanophase Fe particles in lunar soil: Implications for space weathering. Meteorit. Planet. Sci. 2016, 51, 1082–1095. [Google Scholar] [CrossRef]
- Wu, Y.-Z.; Wang, Z.-C.; Lu, Y. Space weathering of the Moon from in situ detection. Res. Astron. Astrophys. 2019, 19, 51. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.-Z.; Zhang, A.-C.; Pang, R.-L.; Li, Y.; Chen, J.-N. Possible records of space weathering on Vesta: Case study in a brecciated eucrite Northwest Africa 1109. Meteorit. Planet. Sci. 2019, 54, 836–849. [Google Scholar] [CrossRef]
- Liu, Y.; Keller, L.P.; Fraeman, A.A.; Christoffersen, R.; Rahman, Z.; Ehlmann, B.L.; Noble, S.K.; Barrat, J.A. Agglutinates in howardite 1769: Space weathering on Vesta (Abstract #1706). In Proceedings of the 46th Lunar and Planetary Science Conference, CD-ROM, The Woodlands, TX, USA, 21–25 March 2015. [Google Scholar]
- Gopon, P.; Fournelle, J.; Llovet, X. Soft X-Ray EPMA of Submicron Phase Lunar Fe-Si Compounds; Cambridge University Press: New York, NY, USA, 2012. [Google Scholar]
- Nazarov, M.A.; Shornikov, S.I.; Demidova, S.I. Origin of native silicon and iron silicides in the Dhofar 280 lunar meteorite. Petrology 2015, 23, 168–175. [Google Scholar] [CrossRef]
- Nazarov, M.A.; Demidova, S.; Anosova, M.O.; Kostitsyn, Y.; Ntaflos, T.; Brandstaetter, F. Native silicon and iron silicides in the Dhofar 280 lunar meteorite. Petrology 2012, 20, 506–519. [Google Scholar] [CrossRef]
- Anand, M.; Taylor, L.A.; Patchen, A.; Cahill, J.; Nazarov, M.A. New Minerals from a New Lunar Meteorite, Dhofar 280 (abstract #1653). In Proceedings of the Lunar and Planetary Science XXXIII, League City, TX, USA, 11–15 March 2002. [Google Scholar]
- Anand, M.; Taylor, L.A.; Nazarov, M.A.; Shu, J.; Mao, H.-K.; Hemley, R.J. New lunar mineral HAPKEITE*: Product of impact-induced vapor-phase deposition in the regolith? In Proceedings of the 34th Annual Lunar and Planetary Science Conference, League City, TX, USA, 17–21 March 2003. [Google Scholar]
- Anand, M.; Taylor, L.A.; Nazarov, M.A.; Shu, J.; Mao, H.-K.; Hemley, R.J. Space weathering on airless planetary bodies: Clues from the lunar mineral hapkeite. Proc. Natl. Acad. Sci. USA 2004, 101, 6847–6851. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Korochantseva, E.V.; Buikin, A.I.; Hopp, J.; Lorenz, C.A.; Korochantsev, A.V.; Ott, U.; Trieloff, M. Thermal and irradiation history of lunar meteorite Dhofar 280. Meteorit. Planet. Sci. 2016, 51, 2334–2346. [Google Scholar] [CrossRef]
- Wieczorek, M.A.; Weiss, B.P.; Stewart, S.T. An Impactor Origin for Lunar Magnetic Anomalies. Science 2012, 335, 1212–1215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jambor, J.L.; Grew, E.S.; Roberts, A.C. New mineral names: Hapkeite. Am. Mineral. 2005, 90, 518–522. [Google Scholar] [CrossRef]
- mindat.org. Samples returned by the Russian Luna-24 Mission from Crater Ejecta Blanket. Available online: https://www.mindat.org/loc-7798.html (accessed on 31 October 2021).
- Ashikmina, N.A.; Bogatikov, O.A.; Gorshkov, A. Aktsesornye minerali steklovatikh fragmentov Luni 24. Doklady Akademii Nauk SSSR Earth Sci. Sect. 1979, 248, 953–955. [Google Scholar]
- Spicuzza, M.J.; Valley, J.W.; Fournelle, J.; Huberty, J.M.; Treiman, A. Native silicon and Fe-silicides from the Apollo 16 lunar regolith: Extreme reduction, metal-silicate immiscibility, and shock melting (Abstract #2231). In Proceedings of the 42nd Lunar and Planetary Science Conference, The Woodlands, TX, USA, 7–11 March 2011. [Google Scholar]
- Gopon, P.; Spicuzza, M.J.; Kelly, T.F.; Reinhard, D.; Prosa, T.J.; Fournelle, J. Ultra-reduced phases in Apollo 16 regolith: Combined field emission electron probe microanalysis and atom probe tomography of submicron Fe-Si grains in Apollo 16 sample 61500. Meteorit. Planet. Sci. 2017, 52, 1941–1962. [Google Scholar] [CrossRef]
- Gopon, P.; Fournelle, J.; Spicuzza, M.; Valley, J. Survey for Fe-Si in Apollo 16 Regolith Sample 61501,22. Microsc. Microanal. 2015, 21, 2095–2096. [Google Scholar] [CrossRef] [Green Version]
- Gopon, P.; Spicuzza, M.; Kelly, T.; Reinhard; Prosa, T.; Larson, D.; Fournelle, J. Atom Probe Tomography of Reduced Phases in Apollo 16 Regolith Sample 61501,22. Microsc. Microanal. 2017, 23, 720–721. [Google Scholar] [CrossRef] [Green Version]
- Gopon, P.; Fournelle, J.; Valley, J.; Horn, W.; Pinard, P.; Sobol, P.; Spicuzza, M.; Llovet, X. Soft X-ray EPMA analyses of nanophase lunar Fe-Si compounds. In Proceedings of the Wisconsin Space Conference, Marquette University, Milwaukee, WI, USA, 15–16 August 2013. [Google Scholar] [CrossRef] [Green Version]
- Ross, A.J.; Downes, H.; Smith, C.L.; Jones, A.P. Highly reduced metals and sulfides in ureilites: Remnants of the UPB core? In Proceedings of the 72nd Annual Meteoritical Society Meeting Meteoritics & Planetary Science, Nancy, France, 13–18 July 2009; p. 44. [Google Scholar]
- Smith, C.L. Iron silicide in polymict ureilites—Recording the complex history of the ureilite parent body. In Proceedings of the 73rd Meeting of Meteoritical Society, New York City, NY, USA, 26–30 July 2010. Abstract #5221. [Google Scholar]
- Downes, H.; Mittlefehldt, D.W.; Kita, N.T.; Valley, J.W. Evidence from polymict ureilite meteorites for a disrupted and re-accreted single ureilite parent asteroid gardened by several distinct impactors. Geochim. Cosmochim. Acta 2008, 72, 4825–4844. [Google Scholar] [CrossRef]
- Broadley, M.W.; Bekaert, D.V.; Marty, B.; Yamaguchi, A.; Barrat, J.A. Noble gas variations in ureilites and their implications for ureilite parent body formation. Geochim. Cosmochim. Acta 2019, 270, 325–337. [Google Scholar] [CrossRef]
- Cohen, B.A.; Goodrich, C.A.; Keil, K. Feldspathic clast populations in polymict ureilites: Stalking the missing basalts from the ureilite parent body. Geochim. Cosmochim. Acta 2004, 68, 4249–4266. [Google Scholar] [CrossRef]
- Goodrich, C.A.; Van Orman, J.; Wilson, L. Fractional melting and smelting on the ureilite parent body. Geochim. Cosmochim. Acta 2007, 71, 2876–2895. [Google Scholar] [CrossRef]
- Goodrich, C.A.; Wilson, L.; Van Orman, J.; Michel, P. Comment on “Parent body depth-pressure-temperature relationships and the style of the ureilite anatexis” by P. H. Warren (MAPS 47:209-227). Meteorit. Planet. Sci. 2013, 48, 1096–1106. [Google Scholar] [CrossRef] [Green Version]
- Hoffmann, V.H.; Hochleitner, R.; Torii, M.; Funaki, M.; Mikouchi, T.; Kaliwoda, M.; Jenniskens, P.; Shaddad, M.H. Magnetism and mineralogy of Almahata Sitta polymict ureilite (= asteroid 2008 TC3): Implications for the ureilite parent body magnetic field. Meteorit. Planet. Sci. 2011, 46, 1551–1564. [Google Scholar] [CrossRef] [Green Version]
- Nabiei, F.; Badro, J.; Dennenwaldt, T.; Oveisi, E.; Cantoni, M.; Hébert, C.; El Goresy, A.; Barrat, J.A.; Gillet, P. A large planetary body inferred from diamond inclusions in a ureilite meteorite. Nat. Commun. 2018, 9, 1327. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singletary, S.J.; Grove, T.L. Early petrologic processes on the ureilite parent body. Meteorit. Planet. Sci. 2003, 38, 95–108. [Google Scholar] [CrossRef]
- Smith, C.L. Metal and sulphide phases in interstitial veins in “dimict” ureilites—Insights into the history and petrogenesis of the ureilite parent body. In Proceedings of the 39th LPSC, League City, TX, USA, 10–14 March 2008. Abstract #1669. [Google Scholar]
- Van Kooten, E.M.; Schiller, M.; Bizzarro, M. Magnesium and chromium isotope evidence for initial melting by radioactive decay of 26Al and late stage impact-melting of the ureilite parent body. Geochim. Cosmochim. Acta 2017, 208, 1–23. [Google Scholar] [CrossRef]
- Warren, P.H. Parent body depth-pressure-temperature relationships and the style of the ureilite anatexis. Meteorit. Planet. Sci. 2012, 47, 209–227. [Google Scholar] [CrossRef]
- Gabriel, A.D. Origin and Evolution of Ureilite vein Metal—Fe, Ni, Co and Ni Isotope Systematics of Ureilite Vein Metal and Ureilite Silicates. Ph.D. Thesis, Georg-August-Universität, Göttngen, Germany, 2009. [Google Scholar]
- Young, E.D.; Yin, Q.-Z.; Sanborn, M.E.; Shaddad, M.H. Carbonaceous Chondrite-Like Xenoliths in Polymict Ureilites: A Large Variety of Uniue Outer Solar System Materials, LPI Contrib. No. 2132. In Proceedings of the 50th Lunar and Planetary Science Conference, The Woodlands, Texas, USA, 18–22 March 2019. [Google Scholar]
- Russell, S.S.; Zipfel, J.; Folco, L.; Jones, R.; Grady, M.M.; McCoy, T.; Grossman, J.N. The Meteoritical Bulletin, No. 87, 2003 July. Meteorit. Planet. Sci. 2003, 38, A189–A248. [Google Scholar] [CrossRef]
- Herrin, J.S.; Mittlefehldt, D.W.; Downes, H.; Humayum, M. Diverse metals and sulfides in polymict ureilites EET 83309 and EET 87720. In Proceedings of the 38th LPSC, League City, TX, USA, 12–16 March 2007. [Google Scholar]
- Ross, A.J. DaG 1047: A polymict ureilite containing exotic clasts including a chondrite. In Proceedings of the 41st LPSC, The Woodlands, TX, USA, 1–5 March 2010. Abstract #2361. [Google Scholar]
- Grossman, J.N. The Meteoritical Bulletin, No. 82, 1998 July. Meteorit. Planet. Sci. 1998, 33, A221–A239. [Google Scholar] [CrossRef]
- Ikeda, Y. Lithic and mineral clasts in the Dar al Gani (DaG) 319 polymict ureilite. Antarct. Meteor. Res. 2000, 13, 177–221. [Google Scholar]
- Garvie, L.A.J. The Meteoritical Bulletin, No. 99, April 2012. Meteorit. Planet. Sci. 2012, 99, E1–E52. [Google Scholar] [CrossRef]
- The Meteoritical Society. Dar al Gani 1066: Meteoritical Bulletin Database. Available online: https://www.lpi.usra.edu/meteor/metbull.php?code=61777 (accessed on 31 October 2021).
- Bouvier, A.; Gattacceca, J.; Agee, C.; Grossman, J.; Metzler, K. The Meteoritical Bulletin, No. 104. Meteorit. Planet. Sci. 2017, 52, 2284. [Google Scholar] [CrossRef] [Green Version]
- Moggi Cecchi, V.; Caporali, S.; Pratesi, G. DaG 1066: A newfound anomalous ureilite with chondritic inclusions. In Proceedings of the 78th Annual Meeting of the Meteoritical Society, Berkeley, CA, USA, 27–31 July 2015. Abstract. #5252. [Google Scholar]
- Prinz, M.; Weisberg, M.K.; Nehru, C.E.; Delaney, J.S. EET 83309, a polymict ureilite: Recognition of a new group. Lunar Planet. Sci. 1987, 18, 802. [Google Scholar]
- Downes, H.; Beard, A.D.; Howard, K. Petrology of a Granitic Clast in Polymict Ureilite EET 83309. In Proceedings of the 72nd Annual Meteoritical Society Meeting, Nancy, France, 13–18 July 2009; p. 72. [Google Scholar]
- Beard, A.D.; Downes, H.; Howard, K.T. Significance of Opal in Ureilites—Delivery of H2O to the Inner Solar System? In Proceedings of the 74th Annual Meteoritical Society Meeting, London, UK, 8–12 August 2011; p. 74. [Google Scholar]
- Prinz, M.; Weisberg, M.K.; Nehru, C.E.; Delaney, J.S. North Haig and Nilpena: Paired Polymict Ureilites with Angra DOS Reis-Related and other Clasts. In Proceedings of the Lunar and Planar Science XVII, Houston, TX, USA, 17–21 March 1986; pp. 681–682. [Google Scholar]
- Keil, K.; Berkley, J.L. Suessite, Fe3Si: A new mineral in the North Haig ureilite. Am. Mineral. 1982, 67, 126–131. [Google Scholar]
- Jaques, A.; Fitzgerald, M. The Nilpena ureilite, an unusual polymict breccia: Implications for origin. Geochim. Cosmochim. Acta 1982, 46, 893–900. [Google Scholar] [CrossRef]
- Graham, A.L. The Meteoritical Bulletin No. 59. Meteoritics 1981, 16, 193–199. [Google Scholar] [CrossRef]
- Wlotzka, F. The Meteoritical Bulletin, No. 77, 1994 November. Meteorit. Planet. Sci. 1994, 29, 891–897. [Google Scholar] [CrossRef]
- Wlotzka, F. The Meteoritical Bulletin, No. 72. Meteorit. Planet. Sci. 1992, 27, 109–117. [Google Scholar] [CrossRef]
- The Meteoritical Society. Frontier Mountain 90036: Meteoritical Bulletin Database. Available online: https://www.lpi.usra.edu/meteor/metbull.php?sea=Frontier+Mountain+90036&sfor=names&ants=&nwas=&falls=&valids=&stype=contains&lrec=50&map=ge&browse=&country=All&srt=name&categ=All&mblist=All&rect=&phot=&strewn=&snew=0&pnt=Normal%20table&code=10434 (accessed on 31 October 2021).
- Wlotzka, F. The Meteoritical Bulletin, No. 73. Meteorit. Planet. Sci. 1992, 27, 477–483. [Google Scholar] [CrossRef]
- The Meteoritical Society. Frontier Mountain 90054: Meteoritical Bulletin Database. Available online: https://www.lpi.usra.edu/meteor/metbull.php?sea=Frontier+Mountain+90054&sfor=names&ants=&nwas=&falls=&valids=&stype=contains&lrec=50&map=ge&browse=&country=All&srt=name&categ=All&mblist=All&rect=&phot=&strewn=&snew=0&pnt=Normal%20table&code=10452 (accessed on 31 October 2021).
- The Meteoritical Society. Frontier Mountain 93008: Meteoritical Bulletin Database. Available online: https://www.lpi.usra.edu/meteor/metbull.php?sea=Frontier+Mountain+93008&sfor=names&ants=&nwas=&falls=&valids=&stype=contains&lrec=50&map=ge&browse=&country=All&srt=name&categ=All&mblist=All&rect=&phot=&strewn=&snew=0&pnt=Normal%20table&code=10639 (accessed on 31 October 2021).
- Ruzicka, A.; Grossman, J.; Bouvier, A.; Agee, C.B. The Meteoritical Bulletin, No. 103. Meteorit. Planet. Sci. 2017, 52, 1014. [Google Scholar] [CrossRef] [Green Version]
- The Meteoritical Society. Frontier Mountain 90233: Meteoritical Bulletin Database. Available online: https://www.lpi.usra.edu/meteor/metbull.php?sea=Frontier+Mountain+90233&sfor=names&ants=&nwas=&falls=&valids=&stype=contains&lrec=50&map=ge&browse=&country=All&srt=name&categ=All&mblist=All&rect=&phot=&strewn=&snew=0&pnt=Normal%20table&code=10621 (accessed on 31 October 2021).
- The Meteoritical Society. Mountain 90228: Meteoritical Bulletin Database. Available online: https://www.lpi.usra.edu/meteor/metbull.php?sea=Frontier+Mountain+90228&sfor=names&ants=&nwas=&falls=&valids=&stype=contains&lrec=50&map=ge&browse=&country=All&srt=name&categ=All&mblist=All&rect=&phot=&strewn=&snew=0&pnt=Normal%20table&code=10617 (accessed on 31 October 2021).
- Ikeda, Y. Petrology of an unusual monomict ureilite, NWA1241. Polar Sci. 2007, 1, 45–53. [Google Scholar] [CrossRef] [Green Version]
- Funaki, M.; Mikouchi, T.; Almaha Sitta Consortium. Magnetism and Mineralogy of Almaha Sitta. In Proceedings of the 41st Lunar and Planetary Science Conference, The Woodlands, TX, USA, 1–5 March 2010; p. 41. [Google Scholar]
- Meteoritical Bulletin Database. The Meteoritical Bulletin, No. 110, 2021; in preparation.
- Meteoritical Bulletin Database. Northeast Africa 027. Available online: https://www.lpi.usra.edu/meteor/metbull.php?sea=NEA+027&sfor=names&ants=&nwas=&falls=&valids=&stype=contains&lrec=50&map=ge&browse=&country=All&srt=name&categ=All&mblist=All&rect=&phot=&strewn=&snew=0&pnt=Normal%20table&code=74844 (accessed on 6 November 2021).
- Jenniskens, P.; Shaddad, M.H.; Numan, D.; Elsir, S.; Kudoda, A.M.; Zolensky, M.; Le, L.; Robinson, G.A.; Friedrich, J.M.; Rumble, D.; et al. The impact and recovery of asteroid 2008 TC3. Nature 2009, 458, 485–488. [Google Scholar] [CrossRef] [PubMed]
- Gayon-Markt, J.; Delbo, M.; Morbidelli, A.; Marchi, S. On the origin of the Almahata Sitta meteorite and 2008 TC3 asteroid. Mon. Not. R. Astron. Soc. 2012, 424, 508–518. [Google Scholar] [CrossRef] [Green Version]
- Hamilton, V.E.; Goodrich, C.A.; Treiman, A.H.; Connolly, H.C.; Zolensky, M.E.; Shaddad, M.H. Meteoritic evidence for a Ceres-sized water-rich carbonaceous chondrite parent asteroid. Nat. Astron. 2020, 5, 350–355. [Google Scholar] [CrossRef]
- Kaliwoda, M.; Hochleitner, R.; Hoffmann, V.H.; Mikouchi, T.; Gigler, A.M.; Schmahl, W.W. New Raman Spectroscopic Data of the Almahata Sitta Meteorite. Spectrosc. Lett. 2013, 46, 141–146. [Google Scholar] [CrossRef]
- Horstmann, M.; Humayun, M.; Fischer-Gödde, M.; Bischoff, A.; Weyrauch, M. Si-bearing metal and niningerite in Almahata Sitta fine-grained ureilites and insights into the diversity of metal on the ureilite parent body. Meteorit. Planet. Sci. 2014, 49, 1948–1977. [Google Scholar] [CrossRef]
- Grady, M.M. Catalogue of Meteorites: With Special Reference to Those Represented in the Collection of the Natural History Museum, London, 5th ed.; Cambridge University Press: Cambridge, UK, 2000; ISBN 0521663032. [Google Scholar]
- Grossman, J.N.; Zipfel, J. The Meteoritical Bulletin, No. 85, 2001 September. Meteorit. Planet. Sci. 2001, 36, A293–A322. [Google Scholar] [CrossRef]
- Hoffmann, V.H.; Mikouchi, T.; Torii, M.; Funaki, M.; Kaliwoda, M.; Hochleitner, R.; Horstmann, M.; Bischoff, A.; Gnos, E.; Hofmann, B.; et al. Almahata Sitta Magnetism—A Compilation. Asteroids, Comets, Meteors. In Proceedings of the Asteroids, Comets, Meteors 2012, Niigata, Japan, 16–20 May 2012. [Google Scholar]
- Kuehner, S.M.; Irving, A.J.; Sipiera, P.P. Diversity Among EH Chondrites: Anomallous EH3 Chondrite Northwest Africa 8789 and Rare EH Melt Rocks Northwest Africa 7324 and Northwest Africa 10237. In Proceedings of the 79th Annual Meeting of the Meteoritical Society, Berlin, Germany, 7–12 August 2016; p. 79. [Google Scholar]
- Bouvier, A.; Gattacceca, J.; Grossman, J.; Metzler, K. The Meteoritical Bulletin, No. 105. Meteorit. Planet. Sci. 2017. [Google Scholar] [CrossRef] [Green Version]
- Lin, Y.; El Goresy, A. A comparative study of opaque phases in Qingzhen (EH3) and MacAlpine Hills 88136 (EL3): Representatives of EH and EL parent bodies. Meteorit. Planet. Sci. 2002, 37, 577–599. [Google Scholar] [CrossRef]
- Gattacceca, J.; McCubbin, F.M.; Grossman, J.; Bouvier, A.; Bullock, E.; Aoudjehane, H.C.; Debaille, V.; D’Orazio, M.; Komatsu, M.; Miao, B.; et al. The Meteoritical Bulletin, No. 109. Meteorit. Planet. Sci. 2021, 56, 1626–1630. [Google Scholar] [CrossRef]
- Vernazza, P.; Brunetto, R.; Binzel, R.; Perron, C.; Fulvio, D.; Strazzulla, G.; Fulchignoni, M. Plausible parent bodies for enstatite chondrites and mesosiderites: Implications for Lutetia’s fly-by. Icarus 2009, 202, 477–486. [Google Scholar] [CrossRef] [Green Version]
- Sierks, H.; Lamy, P.; Barbieri, C.; Koschny, D.; Rickman, H.; Rodrigo, R.; A’Hearn, M.F.; Angrilli, F.; Barucci, M.A.; Bertaux, J.L.; et al. Images of Asteroid 21 Lutetia: A Remnant Planetesimal from the Early Solar System. Science 2011, 334, 487–490. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coradini, A.; Capaccioni, F.; Erard, S.; Arnold, G.; De Sanctis, M.C.; Filacchione, G.; Tosi, F.; Barucci, M.A.; Capria, M.T.; Ammannito, E.; et al. The Surface Composition and Temperature of Asteroid 21 Lutetia As Observed by Rosetta/VIRTIS. Science 2011, 334, 492–494. [Google Scholar] [CrossRef] [Green Version]
- Connolly, H.C.; Smith, C.; Benedix, G.; Folco, L.; Righter, K.; Zipfel, J.; Yamaguchi, A.; Aoudjehane, H.C. The Meteoritical Bulletin, No. 92, 2007 September. Meteorit. Planet. Sci. 2007, 42, 1647–1694. [Google Scholar] [CrossRef]
- Kimura, M.; Karube, T.; Weisberg, M.K.; Mikouchi, T.; Noguchi, T. Opaque MInerals in Chi Chondrites: Indicators of Formation Conditions. In Proceedings of the 74th Annual Meteoritical Society Meeting, London, UK, 8–12 August 2011; p. 74. [Google Scholar]
- Moyano-Cambero, C.E.; Trigo-Rodrίguez, J.M.; Llorca, J.; Fornasier, S.; Barucci, M.A.; Rimola, A. A plausible link between the asteroid 21 Lutetia and CH carbonaceous chondrites. Meteorit. Planet. Sci. 2016, 51, 1795–1812. [Google Scholar] [CrossRef] [Green Version]
- Lin, C.; Hollister, L.S.; MacPherson, G.J.; Bindi, L.; Ma, C.; Andronicos, C.L.; Steinhardt, P. Evidence of cross-cutting and redox reaction in Khatyrka meteorite reveals metallic-Al minerals formed in outer space. Sci. Rep. 2017, 7, 1637. [Google Scholar] [CrossRef] [Green Version]
- Ruzicka, A.; Grossman, J.N.; Garvie, L. The Meteoritical Bulletin, No. 100, 2014 June. Meteorit. Planet. Sci. 2014, 49, E1–E101. [Google Scholar] [CrossRef] [Green Version]
- Ma, C.; Lin, C.; Bindi, L.; Steinhardt, P.J. Discovery of New Al-Cu-Fe Minerals in the Khatyrka CV3 Meteorite. In Proceedings of the 79th Annual Meeting of the Meteoritical Society, Berlin, Germany, 7–12 August 2016; p. 79. [Google Scholar]
- MacPherson, G.J.; Andronicos, C.L.; Bindi, L.; Distler, V.V.; Eddy, M.P.; Eiler, J.M.; Guan, Y.; Hollister, L.S.; Kostin, A.; Kryachko, V.; et al. Khatyrka, a new CV3 find from the Koryak Mountains, Eastern Russia. Meteorit. Planet. Sci. 2013, 48, 1499–1514. [Google Scholar] [CrossRef]
- Ma, C.; Lin, C.; Bindi, L.; Steinhardt, P. Hollisterite (Al3Fe), kryachkoite (Al,Cu)6(Fe,Cu), and stolperite (AlCu): Three new minerals from the Khatyrka CV3 carbonaceous chondrite. Am. Miner. 2017, 102, 690–693. [Google Scholar] [CrossRef]
- Khatyrka Meteorite, Iomrautvaam massif, Anadyrsky District, Chukotka Autonomous Okrug, Russia. Available online: https://www.mindat.org/loc-2717.html (accessed on 15 February 2021).
- Bindi, L.; Yao, N.; Lin, C.; Hollister, L.S.; MacPherson, G.J.; Poirier, G.R.; Andronicos, C.L.; Distler, V.V.; Eddy, M.P.; Kostin, A.; et al. Steinhardtite, a new body-centered-cubic allotropic form of aluminum from the Khatyrka CV3 carbonaceous chondrite. Am. Miner. 2014, 99, 2433–2436. [Google Scholar] [CrossRef]
- Steinhardt, P.J. Quasicrystals: A brief history of the impossible. Rend. Lince 2012, 24, 85–91. [Google Scholar] [CrossRef]
- Bindi, L.; Steinhardt, P.; Yao, N.; Lu, P. Icosahedrite, Al63Cu24Fe13, the first natural quasicrystal. Am. Miner. 2011, 96, 928–931. [Google Scholar] [CrossRef]
- Bindi, L.; Yao, N.; Lin, C.; Hollister, L.S.; Andronicos, C.L.; Distler, V.V.; Eddy, M.P.; Kostin, A.; Kryachko, V.; MacPherson, G.J.; et al. Natural quasicrystal with decagonal symmetry. Sci. Rep. 2015, 5, srep09111. [Google Scholar] [CrossRef] [Green Version]
- Bindi, L.; Dmitrienko, V.E.; Steinhardt, P.J. Are quasicrystals really so rare in the Universe? Am. Miner. 2020, 105, 1121–1125. [Google Scholar] [CrossRef]
- mindat.org. Icosahedrite. Available online: https://www.mindat.org/min-40647.html (accessed on 31 October 2021).
- mindat.org. Decagonite. Available online: https://www.mindat.org/min-46637.html (accessed on 31 October 2021).
- Asimow, P.D.; Lin, C.; Bindi, L.; Ma, C.; Tschauner, O.; Hollister, L.S.; Steinhardt, P.J. Shock synthesis of quasicrystals with implications for their origin in asteroid collisions. Proc. Natl. Acad. Sci. USA 2016, 113, 7077–7081. [Google Scholar] [CrossRef] [Green Version]
- Hollister, L.S.; Bindi, L.; Yao, N.; Poirier, G.R.; Andronicos, C.L.; MacPherson, G.J.; Lin, C.; Distler, V.V.; Eddy, M.P.; Kostin, A.; et al. Impact-induced shock and the formation of natural quasicrystals in the early solar system. Nat. Commun. 2014, 5, 4040. [Google Scholar] [CrossRef] [Green Version]
- McGeoch, M.W.; Dikler, S.; McGeoch, J.E.M. Hemolithin: A Meteoritic Protein containing Iron and Lithium. arXiv 2020, arXiv:2002.11688. [Google Scholar]
- Ma, C.; Beckett, J.R. Kaitianite, Ti3+2Ti4+O5, a new titanium oxide mineral from Allende. Meteorit. Planet. Sci. 2020, 56, 96–107. [Google Scholar] [CrossRef]
- Savva, N.E.; Minyuk, P.S.; Subbotnikova, T.V. An exotic find of iron silicide in the zone of passage of the Tunguska meteorite and Vitim car. In Collection of Scientific Papers on Materials International Scientific Conference: Scientific Achievements of the Third Millennium; Scientific Achievements of the Third Millennium, Ed.; Science Publishing Corporation: New York, NY, USA, 2019; pp. 56–62. [Google Scholar]
- Bilyk, N.T.; Makovskyi, J.S.; Poberezhskaya, I.V.; Stepanov, V.B.; Tymoschuk, V.R.; Shevchenko, T.G.; Yatsenko, I.G. New Occurence of Iron Silicide in the Ukraine. Cosmogenic or Telluric Nature? Geologia 2014, 17, 101–111. [Google Scholar]
- Kapišinský, I.; Iždinský, J.; Ivan, K.; Pánek, Z.; Zemánková, M. Reanalysis of the cosmic dust L 2011 S2 and L 2009 I14 NASA samples. Contrib. Geophys. Geod. 2006, 36, 63–71. [Google Scholar]
- Nikolayeva, E.P.; Shabanin, M.A. Muassonit iz osadochnikh otlozheniy Fergani. Zap. Vsesoyuznogo Mineral. Obs. 1971, 100, 291–296. [Google Scholar]
- Gevork’yan, V. The occurrence of natural ferrosilicon in the northern Azov region. Doklady Akademii Nauk SSSR 1969, 185, 416–418. (In Russian) [Google Scholar]
- Gevork’yan, V.; Litvin, A.L.; Povarennykh, A.S. Occurrence of the new minerals fersilicite and ferdisilicite. Geologičeskij Zurnal Akademii Nauk Ukraine SSR 1969, 29, 62–71. (In Russian) [Google Scholar]
- Fleischer, M. Fersilicite, Ferdisilicite. Am. Mineral. 1969, 54, 1737–1742. [Google Scholar]
- Gevork’yan, V. On Natural Ferrosilicon in the Sand Mass of the Poltava Series of the Konsko-Yalynskaya Depression (North Cisasov Area). Doklady Akademii Nauk URSR 1968, 6, 513–517. (In Ukrainian) [Google Scholar]
- Yeremenko, G.K.; Polkanov, Y.A.; Vitrichenko, E.A. Cosmogenic Material in Placers: Dverniye i Pogrebennyye Rossypi SSSR; Naukova Dumka Press: Kiev, Russia, 1977; pp. 68–75. [Google Scholar]
- mindat.org. Barringerite. Available online: https://www.mindat.org/min-537.html (accessed on 17 November 2021).
- Yu, Z. Two new minerals gupeiite and xifengite in cosmic dusts from Yanshan. Acta Petrol. Mineral. Anal. 1984, 3, 231–238, (In Chinese with an English abstract). [Google Scholar]
- Dunn, P.J.; Chao, G.Y.; Fitzpatrick, J.J.; Langley, R.H.; Fleischer, M.; Zilczer, J.A. New Mineral Names. Am. Mineral. 1986, 71, 227–232. [Google Scholar]
- Zuxiang, Y. Some new minerals from platinum-bearing rocks in Yanshan and Tibet regions, China. Bull. Inst. Geol. Chin. Acad. Geol. Sci. 1986, 2, 49–57. (In Chinese) [Google Scholar]
- Szöőr, G.; Elekes, Z.; Rózsa, P.; Uzonyi, I.; Simulák, J.; Kiss, Á.Z. Magnetic spherules: Cosmic dust or markers of a meteoritic impact? Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interactions Mater. Atoms 2001, 181, 557–562. [Google Scholar] [CrossRef]
- Drake, S.M.; Beard, A.D.; Jones, A.P.; Brown, D.J.; Fortes, A.; Millar, I.L.; Carter, A.; Baca, J.; Downes, H. Discovery of a meteoritic ejecta layer containing unmelted impactor fragments at the base of Paleocene lavas, Isle of Skye, Scotland. Geology 2017, 46, 171–174. [Google Scholar] [CrossRef] [Green Version]
- Romano, A. Mineralogy of an unusual type of rock of possible meteoritic origin Symposia: MPN-01 General contributions to mineralogy. In Proceedings of the 33rd IGC, International Geological Congress, Oslo, Norway, 6–14 August 2008; p. 33. [Google Scholar]
- Romano, A. Mineralogy of an Unusual Type of Rock of Possible Meteoritic Origin. In Proceedings of the 33rd International Geological Congress 2008, Oslo, Norway, 6–14 August 2008; Norwegian Academy of Science and Letters, Ed.; Curran Associates Inc.: Red Hook, NY, USA, 2013; p. 4475. ISBN 978-1-62748-373-5. [Google Scholar]
- Wittke, J.H.; Bunch, T.E.; West, A.; Harris, R.S. Melt Spherules from Peter’s Pond, A Carolina Bay, South Carolina. In Proceedings of the 70th Annual Meteoritical Society Meeting, Tucson, AZ, USA, 13–17 August 2007. [Google Scholar]
- Suttle, M.D.; Genge, M.J. Diagenetically altered fossil micrometeorites suggest cosmic dust is common in the geological record. Earth Planet. Sci. Lett. 2017, 476, 132–142. [Google Scholar] [CrossRef]
- Yanev, Y.; Benderev, A.; Zotov, N.; Ilieva, I.; Iliev, T.; Georgiev, S. Tektite or meteorite from Koshava gypsum mine, NW Bulgaria. In Proceedings of the Jubilee Scientific Conference “Geosciences 2015”, Sofia, Bulgaria, 10–11 December 2015. [Google Scholar] [CrossRef]
- Yanev, Y.; Iliev, T.; Zotov, N.; Benderev, A.; Nihtianova, D.; Ilieva, I. Silicides and alloys from Koshava tektite (or meteorite), NW Bulgaria. In Proceedings of the GEOSCIENCES 2016: National Conference with International Participation, Sofia, Bulgaria, 7–8 December 2016; Bulgarian Geological Society, Ed.; Bulgarian Geological Society (BGS): Sofia, Bulgaria, 2016; pp. 43–44. [Google Scholar]
- Yanev, Y.; Benderev, A.; Zotov, N.; Dubinina, E.; Iliev, T.; Georgiev, S.; Ilieva, I.; Sergeeva, I. Exotic rock block from the Koshava gypsum mine, Northwest Bulgaria: Petrography, geochemistry, mineralogy and melting phenomena. Geol. Balc. 2021, 50, 45–65. [Google Scholar] [CrossRef]
- Buchner, E.; Schwarz, W.H.; Schmieder, M.; Trieloff, M. Establishing a 14.6 ± 0.2 Ma age for the Nördlinger Ries impact (Germany)-A prime example for concordant isotopic ages from various dating materials. Meteorit. Planet. Sci. 2010, 45, 662–674. [Google Scholar] [CrossRef]
- Bunch, T.E.; Hermes, R.E.; Moore, A.M.; Kennett, D.J.; Weaver, J.C.; Wittke, J.H.; DeCarli, P.S.; Bischoff, J.L.; Hillman, G.C.; Howard, G.A.; et al. Very high-temperature impact melt products as evidence for cosmic airbursts and impacts 12,900 years ago. Proc. Natl. Acad. Sci. USA 2012, 109, E1903–E1912. [Google Scholar] [CrossRef] [Green Version]
- Wu, Y.; Sharma, M.; LeCompte, M.A.; Demitroff, M.N.; Landis, J.D. Origin and provenance of spherules and magnetic grains at the Younger Dryas boundary. Proc. Natl. Acad. Sci. USA 2013, 110, E3557–E3566. [Google Scholar] [CrossRef] [Green Version]
- Schrand, C.; Deutsch, A. Formation of Lechatelierite and Impact Melt Glasses in Experimentally Shocked Rocks. Lunar Planet. Sci. 1998, 29, 1671. [Google Scholar]
- Kennett, J.P.; Kennett, D.J.; Culleton, B.J.; Tortosa, J.E.A.; Bischoff, J.L.; Bunch, T.E.; Daniel, I.R., Jr.; Erlandson, J.M.; Ferraro, D.; Firestone, R.B.; et al. Bayesian chronological analyses consistent with synchronous age of 12,835–12,735 Cal B.P. for Younger Dryas boundary on four continents. Proc. Natl. Acad. Sci. USA 2015, 112, E4344–E4353. [Google Scholar] [CrossRef] [Green Version]
- Muschitiello, F.; Wohlfarth, B. Time-transgressive environmental shifts across Northern Europe at the onset of the Younger Dryas. Quat. Sci. Rev. 2015, 109, 49–56. [Google Scholar] [CrossRef]
- Nakagawa, T.; Kitagawa, H.; Yasuda, Y.; Tarasov, P.E.; Nishida, K.; Gotanda, K.; Sawai, Y. Yangtze River Civilization Program Members. Asynchronous Climate Changes in the North Atlantic and Japan During the Last Termination. Science 2003, 299, 688–691. [Google Scholar] [CrossRef] [PubMed]
- Partin, J.; Quinn, T.; Shen, C.-C.; Okumura, Y.; Cardenas, M.; Siringan, F.; Banner, J.; Lin, K.; Hu, H.-M.; Taylor, F. Gradual onset and recovery of the Younger Dryas abrupt climate event in the tropics. Nat. Commun. 2015, 6, 8061. [Google Scholar] [CrossRef] [PubMed]
- Sun, N.; Brandon, A.D.; Forman, S.L.; Waters, M.R.; Befus, K.S. Volcanic origin for Younger Dryas geochemical anomalies ca. 12,900 cal B.P. Sci. Adv. 2020, 6, eaax8587. [Google Scholar] [CrossRef] [PubMed]
- Mithen, S. After the Ice: A Global Human History, 20,000–5,000 BC, 1st ed.; Harvard University Press: Cambridge, MA, USA, 2006; ISBN 0-674-01570-3. [Google Scholar]
- Moore, A.M.T.; Kennett, J.P.; Napier, W.M.; Bunch, T.E.; Weaver, J.C.; Lecompte, M.; Adedeji, A.V.; Hackley, P.; Kletetschka, G.; Hermes, R.E.; et al. Evidence of Cosmic Impact at Abu Hureyra, Syria at the Younger Dryas Onset (~12.8 ka): High-temperature melting at >2200 °C. Sci. Rep. 2020, 10, 4185. [Google Scholar] [CrossRef]
- Brakenridge, G.R. Core-collapse supernovae and the Younger Dryas/terminal Rancholabrean extinctions. Icarus 2011, 215, 101–106. [Google Scholar] [CrossRef]
- Baldini, J.U.L.; Brown, R.J.; Mawdsley, N. Evaluating the link between the sulfur-rich Laacher See volcanic eruption and the Younger Dryas climate anomaly. Clim. Past 2018, 14, 969–990. [Google Scholar] [CrossRef] [Green Version]
- Broecker, W.S. Was the Younger Dryas Triggered by a Flood? Science 2006, 312, 1146–1148. [Google Scholar] [CrossRef] [Green Version]
- Murton, J.B.; Bateman, M.; Dallimore, S.R.; Teller, J.T.; Yang, Z. Identification of Younger Dryas outburst flood path from Lake Agassiz to the Arctic Ocean. Nature 2010, 464, 740–743. [Google Scholar] [CrossRef]
- Wang, L.; Jiang, W.Y.; Jiang, D.B.; Zou, Y.F.; Liu, Y.Y.; Zhang, E.L.; Hao, Q.Z.; Zhang, D.G.; Zhang, D.T.; Peng, Z.Y.; et al. Prolonged Heavy Snowfall During the Younger Dryas. J. Geophys. Res. Atmos. 2018, 123, 13748–13762. [Google Scholar] [CrossRef]
- Keigwin, L.D.; Klotsko, S.; Zhao, N.; Reilly, B.; Giosan, L.; Driscoll, N.W. Deglacial floods in the Beaufort Sea preceded Younger Dryas cooling. Nat. Geosci. 2018, 11, 599–604. [Google Scholar] [CrossRef]
- LaViolette, P.A. Evidence for a Solar Flare Cause of the Pleistocene Mass Extinction. Radiocarbon 2011, 53, 303–323. [Google Scholar] [CrossRef] [Green Version]
- Basu, S.; Murty, S.; Shukla, P.N.; Shukla, A.D. Origin of Silicides (Unknown Meteorites). Meteorit. Planet. Sci. 2000, 35, A22–A23. [Google Scholar]
- Batovrin, S.; Lipovsky, B. Previously Unknown Class of Meteorites; Batovrin-Lipovsky: Holmdel, NJ, USA, 1998. [Google Scholar]
- Newman, J.D. Impact-Generated Dykes and Shocked Carbonates from the Tunnunik and Haughton Impact Structures, Canadian High Arctic. Ph.D. Thesis, The University of Western Ontario, London, ON, Canada, 2020. [Google Scholar]
- Glass, B.J.; Domville, S.; Sanjanwala, R.; Lee, P. Constrained model interpreation from Haughton crater geophysical datasets. In Proceedings of the 43rd Lunar and Planetary Science Conference, The Woodlands, TX, USA, 19–23 March 2012; p. 2021. [Google Scholar]
- Rappenglück, M.; Rappenglück, B.; Ernstson, K. Kosmische Kollision in der Frühgeschichte: Der Chiemgau-Impakt: Die Erforschung eines bayerischen Meteoritenkrater-Streufelds. Z. Anomalistik 2017, 17, 235–260. [Google Scholar]
- Ernstson, K.; Mayer, W.; Neumair, A.; Rappenglück, B.; Rappenglück, M.A.; Sudhaus, D.; Zeller, K.W. The Chiemgau Crater Strewn Field: Evidence of a Holocene Large Impact Event in Southeast Bavaria, Germany. J. Sib. Fed. Univ. Eng. Technol. 2010, 1, 72–103. [Google Scholar]
- Hoffmann, V.; Rösler, W.; Partzelt, A.; Raeymaekers, B.; van Espen, P. Characterization of a small crater-like strcuture in SE Bavaria, Germany. In Proceedings of the 68th Annual Meteoritical Society Meeting, Gatlinburg, TN, USA, 12–16 September 2005. [Google Scholar]
- Rappenglück, B.; Hiltl, M.; Rappenglück, M.; Ernstson, K. The Chiemgau Impact—A meteorite impact in the Bronze¬/Iron Age and its extraordinary appearance in the archaeological record. In Himmelswelten und Kosmovisionen—Imaginationen, Modelle, Weltanschauungen: Proceedings der Tagung der Gesellschaft für Archäoastronomie in Gilching, 29–31 März 2019; Wolfschmidt, G., Ed.; Tredion: Hamburg, Germany, 2020; pp. 330–349. [Google Scholar]
- Rappenglück, B.; Hiltl, M.; Ernstson, K. The Chiemgau Impact: Evidence of a Latest Bronze Age/Early Iron Age meteorite impact in the archaeological record, and resulting critical considerations of catastrophism. In Beyond Paradigms in Cultural Astronomy; González-García, C., Frank, R.M., Sims, L.D., Rappenglück, M.A., Zotti, G., Belmonte, J.A., Šprajc, I., Eds.; BAR: Oxford, UK, 2021; pp. 57–64. [Google Scholar]
- Fehr, K.T.; Pohl, J.; Mayer, W.; Hochleitner, R.; Fassbinder, J.; Geiss, E.; Kerscher, H. A meteorite impact crater field in eastern Bavaria? A preliminary report. Meteorit. Planet. Sci. 2005, 40, 187–194. [Google Scholar] [CrossRef]
- Fehr, K.T.; Hochleitner, R.; Hölzl, S.; Geiss, E.; Pohl, J.; Fassbinder, J. Ferrosilizium-Pseudometeorite aus dem Raum Burghausen, Bayern. Aufschluss 2004, 55, 297–303. [Google Scholar]
- Doppler, G.; Geiss, E.; Kroemer, E.; Traidl, R. Response to ‘The fall of Phaethon: A Greco-Roman geomyth preserves the memory of a meteorite impact in Bavaria (south-east Germany)’ by Rappenglück et al. (Antiquity 84). Antiquity 2011, 85, 274–277. [Google Scholar] [CrossRef]
- Huber, R.; Darga, R.; Lauterbach, H. Der späteiszeitliche Tüttensee-Komplex als Ergebnis der Abschmelzgeschichte am Ostrand des Chiemsee-Gletschers und sein Bezug zum “Chiemgau Impakt” (Landkreis Traunstein, Oberbayern). E G Quat. Sci. J. 2020, 69, 93–120. [Google Scholar] [CrossRef]
- Völkel, J.; Andrew, M.; Matthias, L.; Hürkamp, K. Colluvial filling of a glacial bypass channel near the Chiemsee (Stöttham) and its function as geoarchive. Z. Geomorphol. 2012, 56, 371–386. [Google Scholar] [CrossRef]
- Hoffmann, V.; Rösler, W.; Schibler, L. Anomalous magnetic signature of top soils in Burghausen area, SE Germany. Geophys. Res. Abstr. 2004, 6, 5041. [Google Scholar]
- Hoffmann, V.; Rösler, W.; Patzelt, A.; Raeymaekers, B.; van Espen, P. Characterisation of a small crater-like structure in SE Bavaria, Germany. Meteor. Planet. Sci 2005, 40, A129. [Google Scholar]
- Rösler, W.; Hoffmann, V.; Raeymaekers, B.; Schryvers, D.; Popp, J. Diamonds in carbon spherules—Evidence for a cosmic impact? Meteorit. Soc. 2005, 40, 5114. [Google Scholar]
- Schryvers, D.; Raeymakers, B. EM characterisation of a potential meteorite sample. #MS16.P13. In Proceedings of the 13th European Microscopy Congress, Antwerp, Belgium, 22–27 August 2004: Materials Sciences; Van Tendeloo, G., Ed.; Belgian Society for Microscopy: Liege, Belgium, 2004; pp. 859–860. [Google Scholar]
- Yang, Z.; Verbeeck, J.; Schryvers, D.; Tarcea, N.; Popp, J.; Rösler, W. TEM and Raman characterisation of diamond micro- and nanostructures in carbon spherules from upper soils. Diam. Relat. Mater. 2008, 17, 937–943. [Google Scholar] [CrossRef]
- Rappenglück, M.A.; Bauer, F.; Ernstson, K.; Hiltl, M. Meteorite impact on a micrometer scale: Iron silicide, carbide and CAI minerals from the Chiemgau impact event (Germany). In Problems and Perspectives of Modern Mineralogy (Yushkin Memorial Seminar–2014), Proceedings, Syktyvkar, Komi Republic, Russia 19–22 May 2014, Syktyvkar; IG Komi SC UB RAS, Ed.; Geoprint: Syktyvar, Russia, 2014. [Google Scholar]
- Raeymaekers, B. A Prospective Biomonitoring Campaign with Honey Bees in a District of Upper-Bavaria (Germany). Environ. Monit. Assess. 2006, 116, 233–243. [Google Scholar] [CrossRef]
- Rappenglück, B.; Rappenglück, M.A.; Ernstson, K.; Mayer, W.; Neumair, A.; Sudhaus, D.; Liritzis, I. Reply to Doppler et al. ‘Response to “The fall of Phaethon: A Greco-Roman geomyth preserves the memory of a meteorite impact in Bavaria (south-east Germany) (Antiquity 84)”’. Antiquity 2011, 85, 278–280. [Google Scholar] [CrossRef]
- Rappenglück, B.; Rappenglück, M.A.; Ernstson, K.; Mayer, W.; Neumair, A.; Sudhaus, D.; Liritzis, I. The fall of Phaethon: A Greco-Roman geomyth preserves the memory of a meteorite impact in Bavaria (south-east Germany). Antiquity 2010, 84, 428–439. [Google Scholar] [CrossRef]
- Hoffmann, V.; Torii, M.; Funaki, M. Peculiar magnetic signature of Fe-Silicide phases and diamond/Fullerence containing carbon spherules. Travaux Géophysiques 2006, XXVII, 52–53. [Google Scholar]
- Bauer, F.; Hiltl, M.; Rappenglück, M.A.; Ernstson, K. Trigonal and cubic Fe2Si polymorphs (hapkeite) in the eight kilograms find of natural iron silicide from Grabenstätt (Chiemgau, Southeast Germany). In Proceedings of the 50th Lunar and Planetary Science Conference, The Woodlands, Texas, USA, 18–22 March 2019. LPI Contrib. No. 2132. [Google Scholar]
- Bauer, F.; Hiltl, M.; Rappenglück, M.A.; Neumaier, A.; Ernstson, K. Fe2Si (hapkeite) from the subsoil in the Alpine forland (Southeast Germany): Is it associated with an impact? In Proceedings of the 76th Annual Meteoritical Society Meeting, Edmonton, AB, Canada, 29 July–2 August 2013. [Google Scholar]
- Rappenglück, M.A.; Bauer, F.; Hiltl, M.; Neumair, A.; Ernstson, K. Calcium-aluminium-rich inclusions (CAIs) In Iron silicide (xifengite, gupeiite, hapkeite) matter: Evidence of a cosmic origin. In Proceedings of the 76th Annual Meteoritical Society Meeting, Edmonton, AB, Canada, 29 July–2 August 2013. [Google Scholar]
- Hiltl, M.; Bauer, F.; Ernstson, K. SEM and TEM analyses of minerals Xifengite, Gupeiite, Fe2Si (Hapkeite?), titanium carbide (TiC) and cubic moissanite (SiC) from the subsoil in the alpine foreland: Are they cosmochemical? In Proceedings of the 42nd Lunar and Planetary Science Conference, The Woodlands, TX, USA, 7–11 March 2011. [Google Scholar]
- Ma, C.; Kampf, A.R.; Connolly, H.C.; Beckett, J.R.; Rossman, G.R.; Smith, S.A.S.; Schrader, D.L. Krotite, CaAl2O4, a new refractory mineral from the NWA 1934 meteorite. Am. Miner. 2011, 96, 709–715. [Google Scholar] [CrossRef]
- Cámara, F.; Bindi, L.; Pagano, A.; Pagano, R.; Gain, S.E.M.; Griffin, W.L. Dellagiustaite: A Novel Natural Spinel Containing V2+. Minerals 2018, 9, 4. [Google Scholar] [CrossRef] [Green Version]
- Smith, D.G.W.; Nickel, E.H. A system of codification for unnamed minerals: Report of the subcommittee for unnamed minerals of the ima commission on new minerals, nomenclature and classification. Can. Miner. 2007, 45, 983–990. [Google Scholar] [CrossRef]
- Mitchell, R.H.; Welch, M.D.; Chakhmouradian, A.R. Nomenclature of the perovskite supergroup: A hierarchical system of classification based on crystal structure and composition. Miner. Mag. 2017, 81, 411–461. [Google Scholar] [CrossRef] [Green Version]
- Mineralogy Database. Hatrurim Formation, Middle East; Mineralogy Database, Emerald Group Publishing: Bingley, UK, 2004; Available online: https://www.mindat.org/loc-2063.html (accessed on 31 October 2021).
- Gross, S. The mineralogy of the Hatrurim Formation, Israel. Geol. Surv. Isr. Bull. 1977, 70, 1–80. [Google Scholar]
- Mineralogy Database. Bellerberg Volcano, Vordereifel, Mayen-Koblenz District, Rhineland-Palatinate, Germany; Mineralogy Database, Emerald Group Publishing: Bingley, UK, 2004; Available online: https://www.mindat.org/loc-204826.html (accessed on 31 October 2021).
- Rösler, W.; Patzelt, A.; Hoffmann, V.; Raeymaekers, B. Characterisation of a small crater-like structure in SE Bavaria, Germany: Abstract, European Space Agency. In Proceedings of the First International Conference on Impact Cratering in the Solar System, ESTEC, Noordwijk, The Netherlands, 8–12 May 2006. [Google Scholar]
- De Lange, G.; Rispens, F. Indication of a diagenetically induced precipitate of an Fe-Si mineral in sediment from the Nares Abyssal Plain, western North Atlantic. Mar. Geol. 1986, 73, 85–97. [Google Scholar] [CrossRef]
- Rudashevskii, N.S.; Kretser, Y.; Anikeeva, L.I.; Andreev, S.I.; Torokhov, M.P.; Kazakova, V.E. Platinum minerals in oceanic ferromanganese crusts. Dokl. Earth Sci. 2001, 378, 464–467. [Google Scholar]
- Jambor, J.L.; Grew, E.S.; Roberts, A.C. New mineral names. Am. Mineral. 2002, 87, 181–184. [Google Scholar]
- Kruszewski, Ł.; Ciesielczuk, J. The Behaviour of Siderite Rocks in an Experimental Imitation of Pyrometamorphic Processes in Coal-Waste Fires: Upper and Lower Silesian Case, Poland. Minerals 2020, 10, 586. [Google Scholar] [CrossRef]
- Kruszewski, Ł.; Ciesielczuk, J.; Misz-Kennan, M.; Fabiańska, M. Chemical composition of glasses and associating mineral species in various pyrometamorphic rocks from coal-mining dumps of the Lower Silesia. Mineral. Spec. Pap. 2014, 42, 70–71. [Google Scholar]
- mindat.org. Oldhamite. Available online: https://www.mindat.org/min-2970.html (accessed on 18 November 2021).
- Kruszewski, Ł.; Ciesielczuk, J.; Misz-Kennan, M. What have meteorites to do with coal fires? A case of Upper and Lower Silesian Basins. Mineral. Spec. Pap. 2012, 40, 28–30. [Google Scholar]
- Goderis, S.; Chakrabarti, R.; Debaille, V.; Kodolányi, J. Isotopes in cosmochemistry: Recipe for a Solar System. J. Anal. At. Spectrom. 2016, 31, 841–862. [Google Scholar] [CrossRef] [Green Version]
- Ferrarotti, A.; Gail, H.-P.; Degiorgi, L.; Ott, H.R. FeSi as a possible new circumstellar dust component. Astron. Astrophys. 2000, 357, L13–L16. [Google Scholar]
- Ferrarotti, A.S.; Gail, H.-P. Mineral formation in stellar winds. II. Effects of Mg/Si abundance variations on dust composition in AGB stars. Astron. Astrophys. 2001, 371, 133–151. [Google Scholar] [CrossRef] [Green Version]
- Ferrarotti, A.S.; Gail, H.-P. Mineral formation in stellar winds. III. Dust formation in S-stars. Astron. Astrophys. 2002, 382, 256–281. [Google Scholar] [CrossRef]
- Ferrarotti, A.S. Staubbildung in Sternwinden: Inaugural-Dissertation zur Erlangung der Doktorwürde der Naturwissenschaftlich-Mathematischen Gesamtfakultät der Ruprecht-Karls-Universität Heidelberg. Ph.D. Thesis, Ruprecht-Karls-Universität Heidelberg, Heidelberg, Germany, 2003. [Google Scholar]
- Humphreys, R.M.; Stanek, K.Z. (Eds.) The Fate of the Most Massive Stars; Astronomical Society of the Pacific: San Francisco, CA, USA, 2005. [Google Scholar]
- Keenan, P.C.; Boeshaar, P.C. Spectral types of S and SC stars on the revised MK system. Astrophys. J. Suppl. Ser. 1980, 43, 379–391. [Google Scholar] [CrossRef]
- Ramstedt, S.; Schoeier, F.L.; Olofsson, H. Circumstellar molecular line emission from S-type AGB stars: Mass-loss rates and SiO abundances. Astron. Astrophys. 2009, 499, 515–527. [Google Scholar] [CrossRef] [Green Version]
- Vink, J.S. Eta Carinae and the Luminous Blue Variables. In Eta Carinae and the Supernova Impostors; Davidson, K., Humphreys, R.M., Eds.; Springer: Boston, MA, USA, 2012; pp. 221–247. ISBN 978-1-4614-2274-7. [Google Scholar]
- Weis, K.; Bomans, D.J. Luminous Blue Variables. Galaxies 2020, 8, 20. [Google Scholar] [CrossRef] [Green Version]
- Smith, T.L.; Gordon, K.D.; Clayton, G.C. The Newly Discovered NIR Features in NGC 7023 and Other Reflection Nebulae. In Proceedings of the 199th American Astronomical Society Meeting, Washington DC, WA, USA, 7–10 January 2002. [Google Scholar]
- Gordon, K.D.; Witt, A.N.; Rudy, R.J.; Puetter, R.C.; Lynch, D.K.; Mazuk, S.; Misselt, K.A.; Clayton, G.C.; Smith, T.L. Dust Emission Features in NGC 7023 between 0.35 and 2.5 Microns: Extended Red Emission (0.7 Microns) and Two New Emission Features (1.15 and 1.5 Microns). Astrophys. J. Lett. 2000, 544, 859–866. [Google Scholar] [CrossRef]
- Bobrowsky, M.; Ueta, T.; Meixner, M. NICMOS Imaging of HD 179821 and AFGL 4106. Proc. Int. Astron. Union 2006, 2, 371–372. [Google Scholar] [CrossRef] [Green Version]
- Molster, F.J.; Waters, N.R.; van Trams, H.; Decin, L.; van Loon, J.T.; Jager, C.; Henning, T.; Kaufl, H.-U.; de Koetr, A.; Bouwman, J. The composition and nature of the dust shell surrounding the binary AFGL 4106. Astron. Astrophys. 1999, 350, 163–180. [Google Scholar]
- Van Loon, J.T.; Molster, F.J.; van Winckel, H.; Waters, L. The circumstellar envelope of AFGL 4106. Astron. Astrophys. 1999, 350, 120–128. [Google Scholar]
- Prialnik, D. Novae. In Encyclopedia of Astronomy and Astrophysics; Murdin, P., Ed.; IOP Publishing: Bristol, UK, 2001; pp. 1846–1856. ISBN 978-1-56159-268-5. [Google Scholar]
- Martin, P.; Dubus, G.; Jean, P.; Tatischeff, V.; Dosne, C. Gamma-ray emission from internal shocks in novae. Astron. Astrophys. 2018, 612, A38. [Google Scholar] [CrossRef] [Green Version]
- Lodders, K. They Came from the Deep in the Supernova: The Origin of TiC and Metal Subgrains in Presolar Graphite Grains. Astrophys. J. Lett. 2006, 647, L37–L40. [Google Scholar] [CrossRef]
- Fedkin, A.; Meyer, B.; Grossman, L. Condensation and mixing in supernova ejecta. Geochim. Cosmochim. Acta 2010, 74, 3642–3658. [Google Scholar] [CrossRef]
- Ebel, D.; Grossman, L. Condensation from supernova gas made of free atoms. Geochim. Cosmochim. Acta 2001, 65, 469–477. [Google Scholar] [CrossRef]
- Meyer, B.S.; Weaver, T.A.; Woosley, S.E. Isotope source table for a 25 M⊙ supernova. Meteoritics 1995, 30, 325–334. [Google Scholar] [CrossRef]
- Singerling, S.A.; Liu, N.; Nittler, L.R.; Alexander, C.M.O.; Stroud, R.M. TEM Analyses of Unusual Presolar Silicon Carbide: Insights into the Range of Circumstellar Dust Condensation Conditions. Astrophys. J. Lett. 2021, 913, 90. [Google Scholar] [CrossRef]
- Hynes, K.M.; Croat, T.K.; Amari, S.; Mertz, A.F.; Bernatowicz, T.J. Structural and isotopic microanalysis of presolar SiC from supernovae. Meteorit. Planet. Sci. 2010, 45, 596–614. [Google Scholar] [CrossRef]
- Hynes, K. Microanalytical Investigations of Presolar SiC Grains as Probes of Condensation Conditions in Astrophysical Environments. Ph.D. Thesis, Washington University in St. Louis, St. Louis, MO, USA, 2010. [Google Scholar] [CrossRef]
- Hazen, R.M.; Morrison, S.M. An evolutionary system of mineralogy. Part I: Stellar mineralogy (>13 to 4.6 Ga). Am. Miner. 2020, 105, 627–651. [Google Scholar] [CrossRef]
- Heck, P.R.; Greer, J.; Kööp, L.; Trappitsch, R.; Gyngard, F.; Busemann, H.; Maden, C.; Avila, J.N.; Davis, A.M.; Wieler, R. Lifetimes of interstellar dust from cosmic ray exposure ages of presolar silicon carbide. Proc. Natl. Acad. Sci. USA 2020, 117, 1884–1889. [Google Scholar] [CrossRef] [Green Version]
- Croat, T.K.; Jadhav, M.; Lebsack, E.; Bernatowic, T.J. A Unique Supernova Graphite: Contemporaneous Condensation of All Things Carbonaceous. In Proceedings of the 42nd Lunar and Planetary Science Conference, The Woodlands, TX, USA, 7–11 March 2011. Abstract. #1533. [Google Scholar]
- Rowan, L.R. Equation of State of Molten Mid-Ocean Ridge Basalt. Structure of Kilauea Volcano. Ph.D. Thesis, California Institute of Technology, Pasadena, CA, USA, 1993. [Google Scholar]
- Suttle, M.D. The Parent Bodies of Fine-grained Micrometeorites: A Petrologic & Spectroscopic Perspective. Ph.D. Thesis, Imperial College London, London, UK, 2018. [Google Scholar]
- Ross, A.J.; Downes, H.; Herrin, J.S.; Mittlefehldt, D.W.; Humayun, M.; Smith, C. The origin of iron silicides in ureilite meteorites. Geochemistry 2019, 79, 125539. [Google Scholar] [CrossRef]
- Schmidt, M.W.; Gao, C.; Golubkova, A.; Rohrbach, A.; Connolly, J.A. Natural moissanite (SiC)—A low temperature mineral formed from highly fractionated ultra-reducing COH-fluids. Prog. Earth Planet. Sci. 2014, 1, 27. [Google Scholar] [CrossRef] [Green Version]

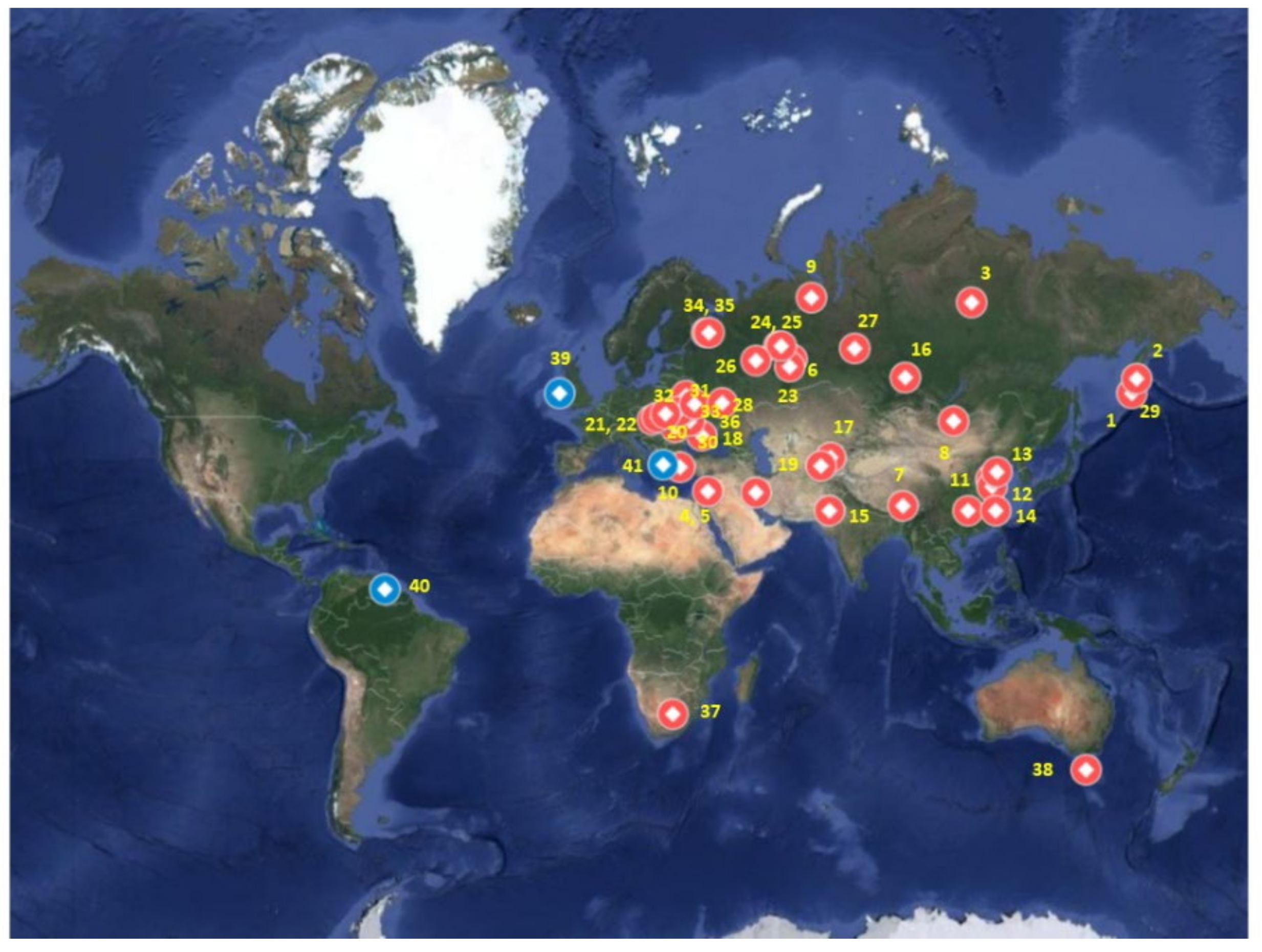
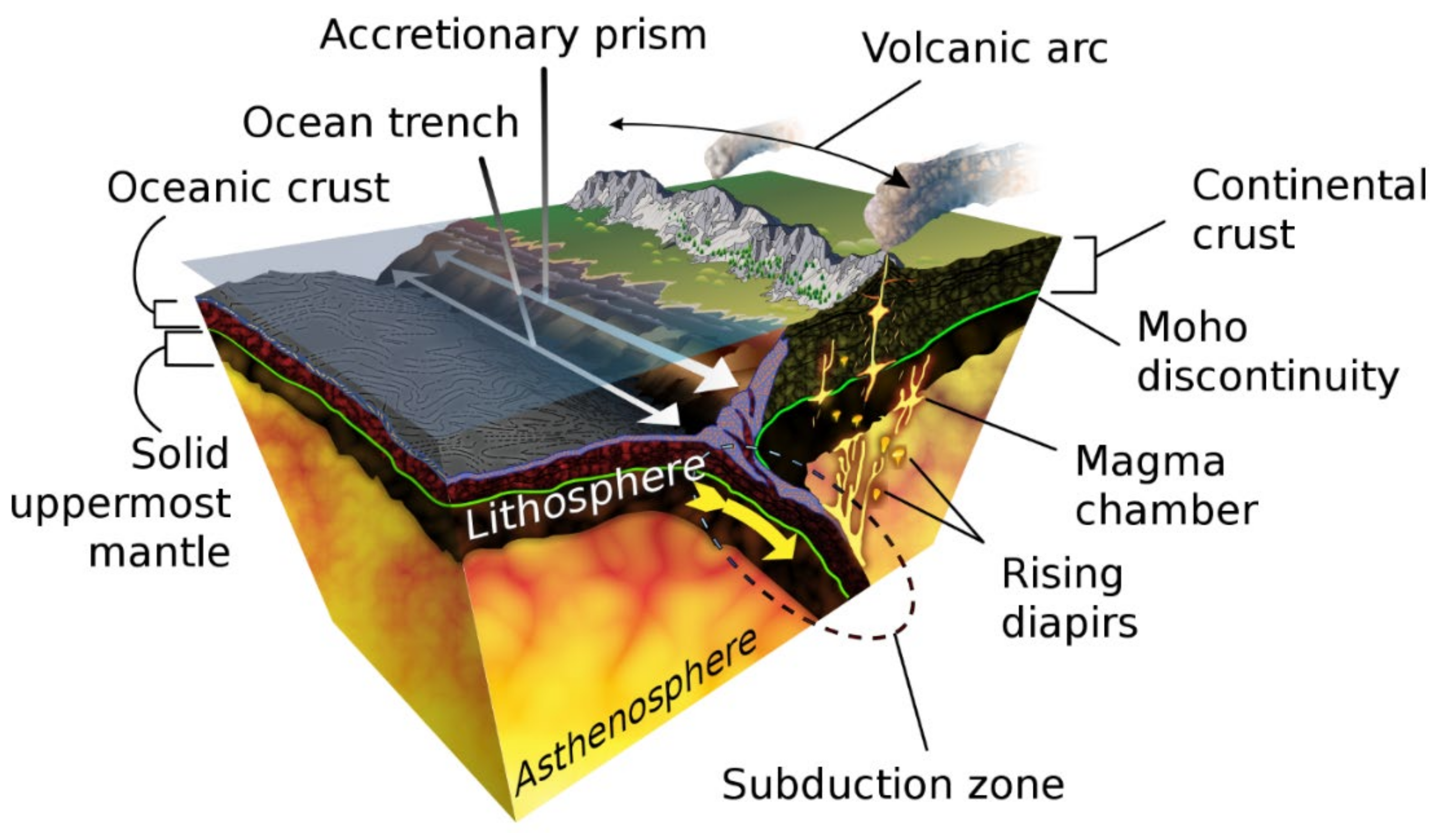





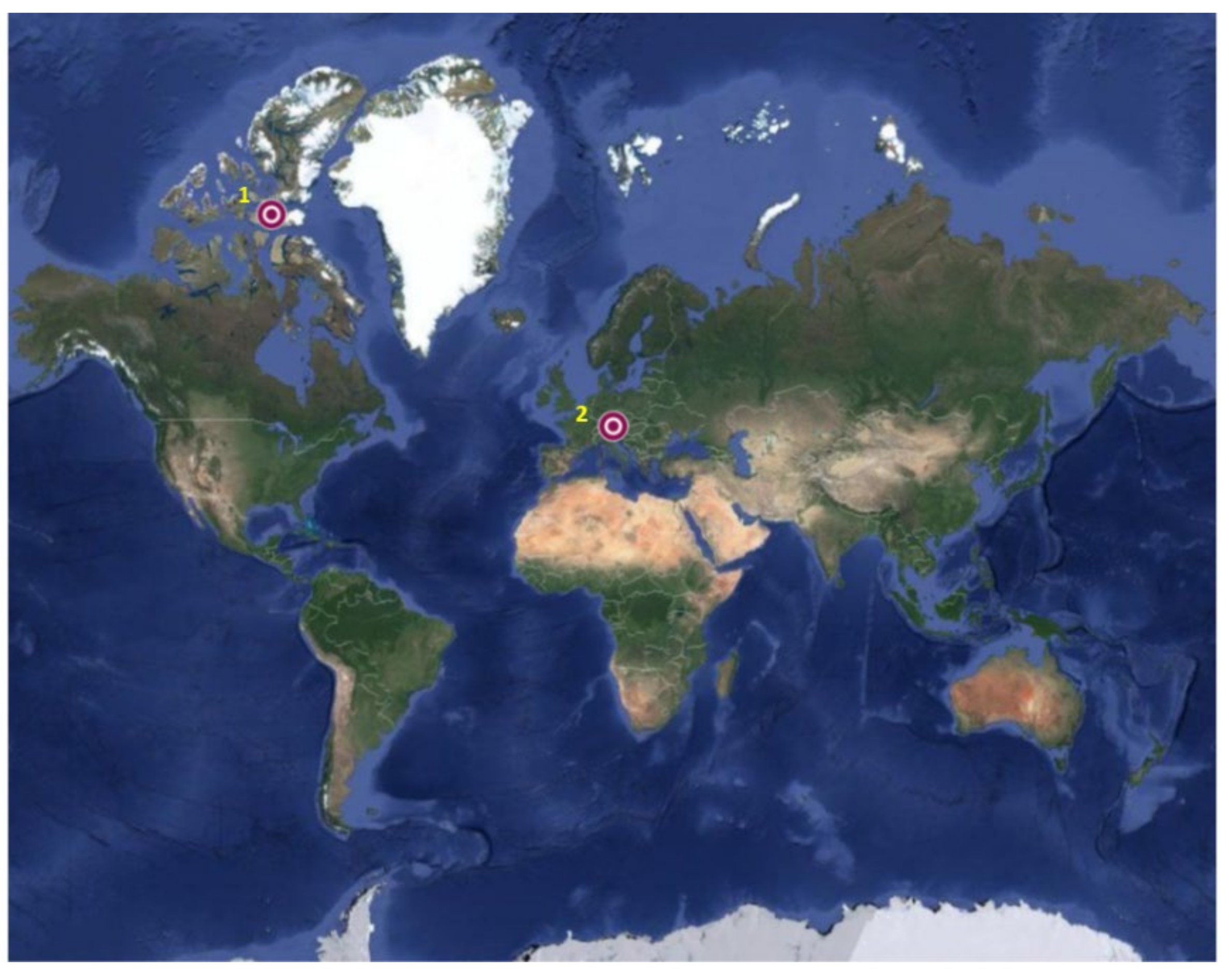




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rappenglück, M.A. Natural Iron Silicides: A Systematic Review. Minerals 2022, 12, 188. https://doi.org/10.3390/min12020188
Rappenglück MA. Natural Iron Silicides: A Systematic Review. Minerals. 2022; 12(2):188. https://doi.org/10.3390/min12020188
Chicago/Turabian StyleRappenglück, Michael A. 2022. "Natural Iron Silicides: A Systematic Review" Minerals 12, no. 2: 188. https://doi.org/10.3390/min12020188
APA StyleRappenglück, M. A. (2022). Natural Iron Silicides: A Systematic Review. Minerals, 12(2), 188. https://doi.org/10.3390/min12020188




