1. Introduction
In the metallurgical industry, bauxite ores are the main source of alumina, which is the primary feedstock for aluminum production. The majority of the world’s bauxite is used to produce alumina via the Bayer process, which is then converted into aluminum via the Hall-Heroult process [
1]. Bauxite is a mixture of three different aluminum hydroxides in varying amounts, with some oxide impurities. As shown in
Table 1, the three aluminum hydroxides commonly found in bauxite ores are gibbsite (γ-Al(OH)
3), boehmite (γ-AlOOH) and diaspore (α-AlOOH). It can be noted that boehmite and diaspore are dimorphous. The most frequently occurring oxide impurity minerals are: goethite (FeO(OH)), aluminum-substituted goethite ((Al
x,Fe
1−x)OOH), hematite (Fe
2O
3), anatase (TiO
2), kaolinite (Al
4Si
4O
10(OH)
8) and quartz (SiO
2). Consequently, the major oxide impurities are Fe
2O
3, TiO
2 and SiO
2. Based on the amounts of the different aluminum hydroxides that are present, bauxite can be classified into three different types: gibbsite-type bauxite, boehmite-type bauxite or diaspore-type bauxite. The thermal dewatering of bauxite ores mainly involves the removal of the loosely bound water at low temperatures, and this is followed by the more strongly bonded water as the temperature increases. Loosely bound water or free water would be expected to be removed by about 100 °C, and this would be followed by the more strongly bonded water and the dewatering of the major minerals, such as gibbsite (Al(OH)
3), followed by boehmite (γ-AlO(OH)) or diaspore (α-AlO(OH)). In some cases, other hydroxylated minor minerals, such as goethite (FeO(OH)) and kaolinite (Al
2Si
2O
5(OH)
4), may also be present in the bauxite ores.
Table 1.
Major mineralogical components in a typical bauxite ore (adapted from Ref [
2]).
Table 1.
Major mineralogical components in a typical bauxite ore (adapted from Ref [
2]).
| Minerals | Formula |
|---|
| Gibbsite | γ-Al (OH)3 |
| Boehmite | γ-AlOOH |
| Diaspore | α-AlOOH |
| Goethite | FeOOH |
| Aluminum Goethite | (Alx,Fe1−x)OOH |
| Anatase/Rutile | TiO2 |
| Kaolinite/Halloysite | Al4Si4O10(OH)8 |
| Quartz | SiO2 |
Numerous experimental studies have been conducted on the thermal decomposition processes of the various bauxite ores [
3,
4,
5,
6,
7,
8,
9,
10,
11,
12]. Typically, these studies have utilized thermogravimetric analysis (TGA) and/or differential thermal analysis (DTA) techniques. These results exhibit considerable variability, as they are dependent on both sample and equipment factors, such as: impurity content, particle size, degree of crystallinity, heating rate, atmospheric conditions, gas flowrate, ambient pressure and water vapor pressure. It is generally accepted that the thermal decomposition of gibbsite proceeds via two stages as the temperature increases. Firstly, at relatively low temperatures of up to about 270 °C, the gibbsite is converted into boehmite as follows:
Based on this stoichiometry, the theoretical mass loss for this process is 23.1%. Secondly, at somewhat higher temperatures of up to about 500 °C, the boehmite is converted into alumina (Al
2O
3) as follows:
Here, the stoichiometric mass loss would be 11.5%, and thus, the overall mass loss would be 34.6%. For finer and/or less crystalline bauxite ores, it has been found that these two processes can occur simultaneously to some degree at about 270 °C, and any remaining more stable boehmite may decompose at higher temperatures [
13,
14,
15,
16]. On the other hand, for coarser and/or more crystalline ores, the two dewatering stages can be more distinct [
13,
14,
15,
16].
Gibbsite contains hydroxyl groups, and the basic layered structure consists of stacked sheets of aluminum hydroxide octahedrons (Al(OH)
6). Each octahedron consists of six hydroxyl anions (OH
−) that are octahedrally shared with two aluminum cations (Al
3+), and one octahedral site is vacant. On the other hand, boehmite is composed of sheets of edge-sharing Al-(O,OH) octahedrons. Oxygen is at the corners of four of the octahedra, and the hydroxyl ion is at the corners of two of the octahedra. During dehydroxylation, the smaller protons diffuse and combine with the larger hydroxyl ions to form water vapor according to the following reaction [
17]:
As a result of the distinct bonding mechanisms in gibbsite and boehmite, the interactions of the electromagnetic field during microwave dewatering of these minerals would be expected to be different.
1.1. Microwave Fundamentals
Microwave radiation is the part of the electromagnetic spectrum where frequencies are relatively low, ranging from 300 MHz to 300 GHz, and wavelengths are relatively large, ranging from about 1 mm to 1 m. For domestic applications, 2.45 GHz is used and 915 MHz for industrial applications. The interaction of a microwave field with a given material is dependent on both the magnetic permeabilities and the electrical permittivities of the material. For most ores and minerals, the contribution of the magnetic permeability is not significant, and therefore, the interaction can be adequately described by the complex permittivity (ε) as follows:
In Equation (4), ε′ is the real permittivity or the dielectric constant; ε″ is the imaginary permittivity or the dielectric loss; j is the imaginary component in the + j-axis direction
. Equation (4) can be converted into Equation (5), where ε
o is the permittivity of free space or in vacuum with a value of 8.86 × 10
–12 F/m; is the relative real permittivity or relative dielectric constant; and
is the relative imaginary permittivity or relative dielectric loss. The real permittivity is a measure of the capacitance and is related to the polarizability of the material and hence its ability to store electrical energy from the external field. The imaginary permittivity is a measure of the material’s ability to dissipate the electrical energy from the electromagnetic field, typically as heat. Usually, the imaginary permittivity is much smaller than the real permittivity. Additionally, since the imaginary permittivity is greater than zero, most materials will heat to some extent. The imaginary permittivity is a function of electrical conductivity (σ) and frequency (f) as follows:
Thus, the imaginary permittivity increases with conductivity and decreases with frequency. Additionally, since the conductivities of minerals increase with temperature, particularly at higher temperatures, then the imaginary permittivity would be expected to exhibit a similar behavior.
An important intrinsic material property in microwave processing is the ratio of the imaginary permittivity to the real permittivity, and this is referred to as the loss tangent or dissipation factor as follows:
The loss tangent is a measure of the energy transferred from the field into the material, which is converted into heat. For efficient microwave heating, the imaginary permittivity should be high, and the real permittivity should have an intermediate value.
1.2. Microwaves and Minerals
Microwaves have the ability to heat materials by various mechanisms, such as dipolar polarization, ionic polarization, atomic polarization and surface polarization. A number of studies have been performed on the microwave heating behaviors of materials of interest, in particular compounds and minerals. Ford and Pei observed that darker colored materials heated more readily than lighter ones [
18]. Chen et al. concluded that the heating behaviors of minerals in a microwave field are composition dependent [
19]. A review by the Atomic Energy of Canada Limited in 1990 divided materials into four groups based on the heating rate in a microwave field [
20]. In comparison to conventional heating involving radiation, conduction and convection, microwave heating has several unique characteristics. Being a form of electromagnetic radiation, microwaves can potentially penetrate into the interior of a material, and thus, any limitations due to low thermal conductivity can be overcome to some extent. Another unique characteristic of microwave processing of multiphase materials is the potential for selective heating. Since the microwave absorption characteristics are material dependent, then some degree of selective heating can be achieved in heterogeneous materials, where one phase heats faster than another phase. Of particular interest for the microwave processing of ores or concentrates is the effect of the field on the individual heterogeneous particles. Minerals exhibit different microwave absorption characteristics, and therefore, they can have dissimilar heating behaviors. This phenomenon of anisothermal conditions or localized heating has been confirmed by measurement of the temperatures of the individual phases in multiphase materials [
21]. The degree of this selective heating would be expected to depend on a number of material properties, such as the size and the composition of the phases and their thermal conductivities.
A number of attempts have been made to utilize selective heating for various applications in mineral processing [
22,
23,
24,
25,
26]. Perhaps the most researched application is microwave pretreatment in comminution, which has been widely reported in the literature as a method to reduce the overall energy consumed [
27,
28,
29,
30,
31]. Usually, ores are highly heterogeneous and are composed of at least two mineral components, and one of these phases will heat more readily than the others, and this leads to thermal stress fracturing along the grain boundaries. This is particularly relevant for ores where a sulphide phase, which is a good microwave absorber, is in an oxide matrix, which is a poor absorber. One method to investigate the effect of selective heating in heterogeneous ores is to determine the change in the strength of the material after microwave processing. This weakening of the structure can potentially reduce the energy requirements in subsequent crushing and grinding operations. The reported benefits of microwave treatment for comminution in the literature include reduced ore competency, improved liberation and reduced mill wear and maintenance [
32,
33,
34].
On the other hand, there is a paucity of information regarding the effect of microwave heating on the strength reduction of ores or minerals as a result of dewatering. Water in the bulk form is a very good microwave absorber, and the interaction depends on the strength of the hydrogen bonded network and any ions that are present. Therefore, it would be expected that some degree of selective heating could be achieved when water is present in an ore or concentrate. However, this water can be more strongly bonded than bulk water, and therefore, its behavior in these materials can be quite different. However, there is a dearth of information regarding the interaction of microwaves with the different types of water that are present and also the effects of temperature and frequency. The interaction depends on the ability of the microwaves to polarize the water molecules, and this, in turn, depends on the bond strength between the water molecules and the substrate. Water trapped in pores or cracks can be relatively free and thus would be expected to absorb microwaves. On the other hand, adsorbed water, which is strongly bonded, would have less of an interaction, with water multilayers being more absorbent than a monolayer. This type of water is removed at about 100 °C, while hydrated water (·nH2O) requires a few hundred degrees to remove. The hydroxyl ion (OH−), which is bonded to a cation in the crystal structure, will require even higher temperatures to be converted into water. Thus, in general, for minerals and ores, the type of water can be ranked in terms of increasing interaction with the microwaves as follows: hydroxylated water < hydrated water < adsorbed monolayer < adsorbed multilayers < bulk water.
There is very little information in the literature regarding the effects of microwave dewatering of ores or minerals on their mechanical properties. Peinsitt et al. have investigated the properties of both dry and water saturated granite, basalt and sandstone using uniaxial compressive strength and P-wave velocity testing [
35]. After microwave irradiation, the water saturated samples were weaker, and this was more significant for sandstone than for granite, and there was no effect for basalt. Large cracks and bursting were observed to occur in the sandstone. These results would indicate that the weakening effect was more pronounced for the porous rocks. In some materials such as coal, it has been shown that microwaves can facilitate fracturing and hence lower the mechanical strength of the material [
36,
37,
38]. Huang et al. have reported that water saturation levels between 25% and 50% can provide optimum microwave fracturing in coal [
39]. In general, this phenomenon can occur by at least three mechanisms. Firstly, selective heating of different phases in the coal can result in thermal stress fracturing along grain boundaries. Secondly, the microwaves can cause microdamage of the pore structure. This damage can accumulate and become macrodamage, which can ultimately result in crack formation. This method of crack formation is different than in conventional heating, and also, the crack formation can be more extensive. Thirdly, water trapped in pores and cracks below the surface can be heated above its boiling point, and the resulting internal pressures can induce fracturing.
In the present research, the major objective was to perform a comparative study of the dewatering and weakening of gibbsite-type bauxite ores by both conventional and microwave heating processes. In order to understand the interaction of the microwaves with the ores, permittivity studies were conducted, and also, the heating behavior was determined. The changes in the properties of the ore as a result of dewatering were evaluated using the compressive strength index. Roughly spherical bauxite ore pisoids were utilized to study the effects of conventional and microwave processing on the compressive strength indices. There is considerable controversy regarding the effect of particle size on the microwave heating behaviors of materials. Consequently, single particle compressive strength index studies were performed to investigate the effect of particle size. Finally, based on the compressive strength reductions for both conventional and microwave processing, a laboratory-scale energy comparison for the two processes is given.
2. Experimental Section
2.1. Raw Materials
Chemical and Mineralogical Compositions
Three different bauxite ores (1,2,3) were obtained from Alcan International Limited operations for this study. Bauxite (1) was used in the TGA, permittivity and the microwave heating behavior studies, while bauxite (2) and bauxite (3) were utilized to investigate the effect of microwaves on the ore strength. The chemical compositions of the three bauxite ore samples were determined by X-ray fluorescence (XRF), and the results are shown in
Table 2. It can be seen that the alumina content was in the range of 49% to 55%, and the loss on ignition (LOI) varied from about 25% to 28%. The iron oxide content varied quite widely from about 5% to 19%. With bauxite (1) and bauxite (2), the major impurities were iron oxide, followed by silica and then titanium oxide. For the bauxite (3) sample, the amount of iron oxide was much lower than that of the other bauxite samples. For all the ores, the major impurities, in order of decreasing weight percentage, were iron oxide, silica and titanium oxide.
Table 3 shows the major minerals in the bauxite ore samples, as determined by X-ray diffraction (XRD). The results indicated that all the samples were the gibbsite (γ-Al(OH)
3)-type bauxite with a small amount of kaolinite (Al
4Si
4O
10(OH)
8). In addition, some boehmite (γ-AlOOH) was found in bauxite (2) and bauxite (3). The iron oxide in all of the bauxite samples was present as hematite. The titanium oxide was present as anatase (TiO
2) in all the bauxites.
Figure 1 shows a scanning electron microscope (SEM) image of the surface of a fractured as-received bauxite (2) sample. The highlighted area was analyzed by energy dispersive X-ray spectroscopy (EDS) and demonstrated that it contained a large amount of aluminum, as well as some iron. This would indicate that these crystals are gibbsite.
2.2. Physical Characteristics
Since the chemical compositions and the mineralogies of the ores were similar, then their utilization for the various aspects of the experimental work was mainly determined by their physical characteristics. Bauxite (1) was composed of relatively fine irregular particles with a wide particle size distribution and was used in the TGA, permittivity and heating behavior studies. The particle size distribution of this bauxite was analyzed by dry sieving using Tyler screens in a vibrating sieve shaker, and the results are shown in
Figure 2. The particle size was below 2.362 mm, and the sample mass in each size fraction was relatively uniform. The pisolitic bauxite ore (2) was employed for the compressive strength index studies, as it was approximately spherical. The particle size range of −6.680 + 4.699 mm was utilized in the tests, and this size range was obtained by dry sieving. The bauxite (3) sample was relatively coarse and was used to investigate the effect of sample size on the compressive strength. It was crushed and sized into three coarse size ranges of −4.5 + 3.5 cm, −3.5 + 3.0 cm and −3.0 + 2.5 cm.
2.3. Thermogravimetric Analysis (TGA)
Thermogravimetric analysis (TGA) was employed to study the dewatering of bauxite ore (1). The particle size of the samples was reduced to less than 0.038 mm by pulverizing. Samples of 10 g were heated to 1000 °C in air at 25 °C intervals at a heating rate of 0.02 °C/min. At each temperature, the samples were held for 24 h in order to ensure that equilibrium was achieved. Then, the samples were weighed using an electronic balance (Model SC 4020). The results were plotted as mass loss percentage versus time, and also, derivative thermogravimetric analysis (DTGA) curves were generated.
2.4. Real and Imaginary Permittivities/
The real and the imaginary permittivities of bauxite ore (1) were determined at different temperatures and frequencies using the cavity perturbation method. An iron-rich fraction (1-Fe) of bauxite ore (1) was separately measured. This ore (1-Fe) contained 27.6% Fe, and the LOI was 20.6%. The cavity perturbation technique has been described previously in the literature [
40,
41], and only a summary of the important features is given here. The measurement is based on the determination of the quality factor Q and also the resonant frequency in a high electric field cavity, both with and without the sample. For a sample with a small mass, the shifts of the Q factor and the resonant frequency are related to the complex susceptibility (χ) of the sample, which is also a function of the complex permittivities (χ = ε − 1). Using these complex permittivities, both the real and the imaginary permittivities were calculated. By using the multimode (n = 6) cavity, the real and the imaginary permittivities of the sample can be measured at the various frequencies. In addition, the sample temperature could be varied from room temperature up to 1000 °C.
For the measurements, the samples in powdered form were first compressed in a tungsten carbide lined die in a uniaxial press to produce briquettes. Subsequently, the sample briquette was placed in a high-purity amorphous silica tube sample holder, which was cleaned, dried and heated to 500 °C before each test. The sample in the holder at room temperature was inserted into the thick-walled, well-cooled multimode cavity. Typically, about two seconds was required for the measurements at a given frequency. Measurements could be performed at 912 MHz, 1499 MHz, 1977 MHz, 2466 MHz and 2986 MHz. The resonance frequency and the loaded quality factor Q of the cavity with the sample in the holder were recorded by a Hewlett-Packard 8753 network analyzer, and data analysis was performed offline. This included subtracting the resonant frequency and the quality factor of the sample in the cavity from the values for the sample holder alone in the cavity.
For the measurements at temperatures higher than 24 °C, the holder with sample was heated in a conventional furnace in an air atmosphere at the desired temperature for at least 5 min. The heating rate of bauxite ore (1) was set as 2.9 °C/min, and the studied temperatures ranged from 25 °C to 977 °C. For the iron-rich fraction (1-Fe), the heating rate was 4.0 °C/min, and the temperature varied from 24 °C to 397 °C. The hot sample in the holder was then rapidly removed from the furnace and returned to the cavity for the next series of measurements. As a result, both the real and the imaginary permittivitites of bauxite (1) and the iron-rich fraction (1-Fe) were determined at different temperatures and frequencies, and also, the loss tangents could be calculated using Equation (7).
2.5. Microwave Heating Behavior Studies
In the microwave heating behavior studies, the sample temperature was measured by inserting a type-K thermocouple into the powdered sample along the vertical centerline and at the bottom of the sample. This position corresponded with the location where the maximum temperature was attained. A thermocouple wire diameter of 0.5 mm was employed because this wire size has both a relatively fast response time and a long lifetime. In order to eliminate any interference from the microwaves, the power was turned off during the measurement.
Sintering can affect the compressive strength of the particles, particularly in microwave processing where thermal runaway can occur. Therefore, the microwave heating behavior of bauxite ore (1) was studied using a single replicate 2
4 factorial experiment. The four factors were heating time, microwave power, sample mass and sample bulk density, and the factor levels used in the design are shown in
Table 4. The three- and four-factor interactions were utilized as an estimate of error. The design experiments were performed randomly. The test results were used to estimate the factor effects and to provide an initial linear model of the sample temperature as a function of the significant factors and/or the two-factor interactions. From the obtained test conditions, a steep ascent method was employed to reach the desired operating temperature.
2.6. Conventional and Microwave Heating Systems
Prior to the compressive strength studies, the bauxite ores (2 or 3) were preheated either in a conventional laboratory resistance furnace or in a microwave cavity. The maximum power of the conventional furnace was 1200 W (120 V, 10 A).
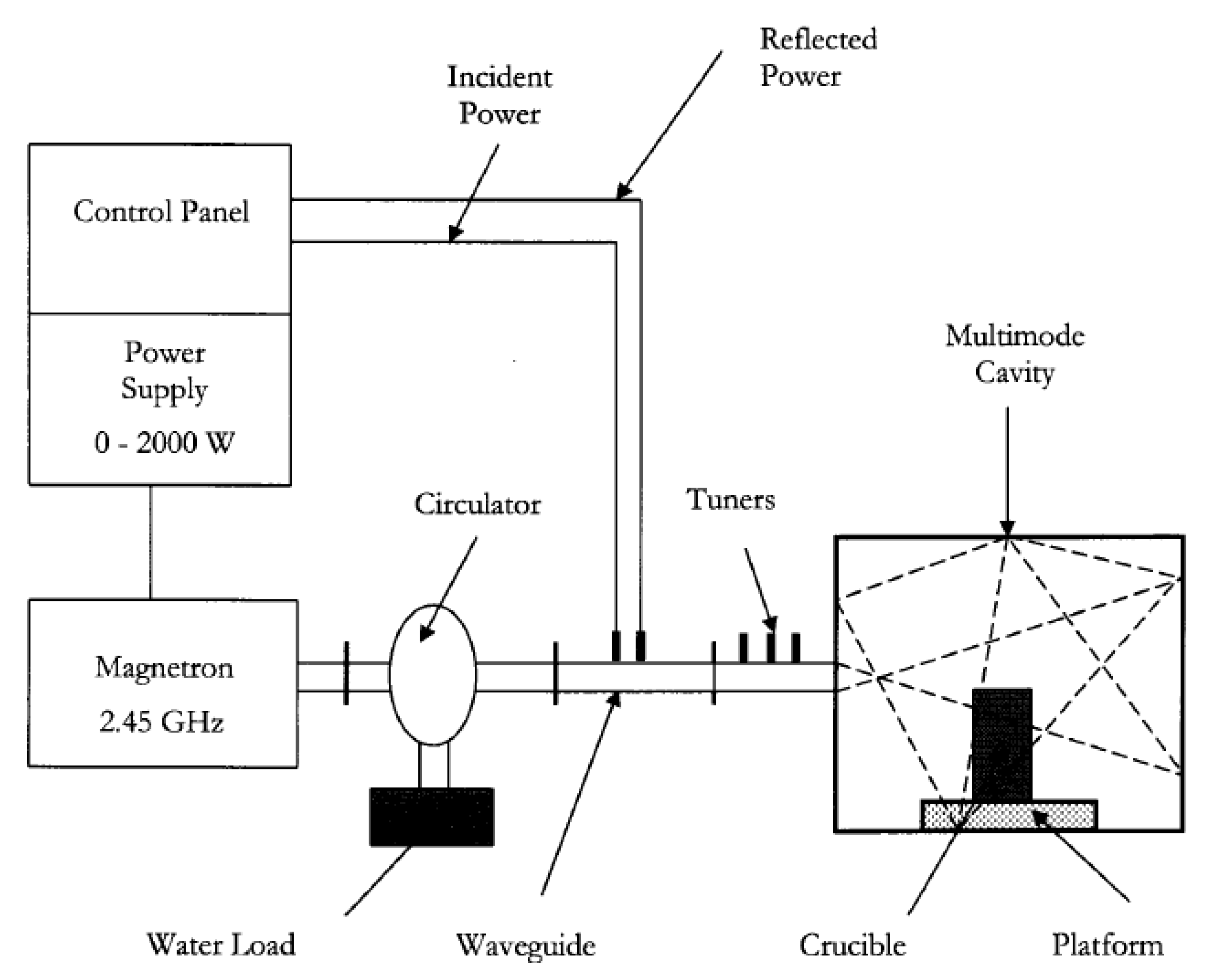
Figure 2 shows a schematic diagram of the microwave system employed in this work. The major features were a power supply, a control panel, a 2.45 GHz air-cooled magnetron, a rectangular aluminum waveguide, a circulator and a multimode cavity containing the sample to be processed. The Model P2000L power supply was manufactured by Conversion Technology Corporation (CTC, Southlake, TX, USA). The incident power could be varied continuously from 0 to 2000 W, and the microwaves traveled along the rectangular waveguide to the cavity. The cavity dimensions were 26 cm in height, 40 cm in width and 40 cm in length. The samples to be processed were placed in a cylindrical quartz crucible on a platform, at the center of the base. The cylindrical quartz crucible had the following dimensions: 37 mm in height, 28 mm in diameter and 1.5 mm in thickness. The microwave transparent platform was made from 80% Al
2O
3 and 20% SiO
2. As illustrated by the dashed lines in the cavity, the microwaves can be absorbed, reflected or transmitted by the sample but only reflected by the walls. Any microwaves not absorbed by the sample will eventually return back down the waveguide and will be absorbed by the water load. Additionally, both the incident and the reflected microwave power were monitored using two detectors installed in the waveguide. The three stub tuners in the waveguide were utilized to manually adjust the incident microwave power in order to minimize the reflected power by matching the incident power with the load.
2.7. Conventional and Microwave Heating of Spherical Pisoids
For these tests, bauxite (2) was utilized due to its roughly spherical shape, and the particle size range was relatively small at −6.680 mm +4.699 mm. Ten-gram samples were heated for 20 min at 200 °C, 500 °C, 700 °C and 900 °C. Additionally, a fifty-gram sample was heated for 2 min at 900 °C. During the heating process, the sample temperature was measured by inserting the tip of the Type-K thermocouple into the middle of the sample. The temperatures were recorded as a function of heating time. After heating, the mass loss was calculated, and this was related to the moisture content. For the microwave heating tests, fifty-gram samples were microwaved for 2 min at 825 W, 1000 W and 1200 W. Again, the mass loss was determined.
2.8. Compressive Strengths of Spherical Pisoids and Single Particle Studies
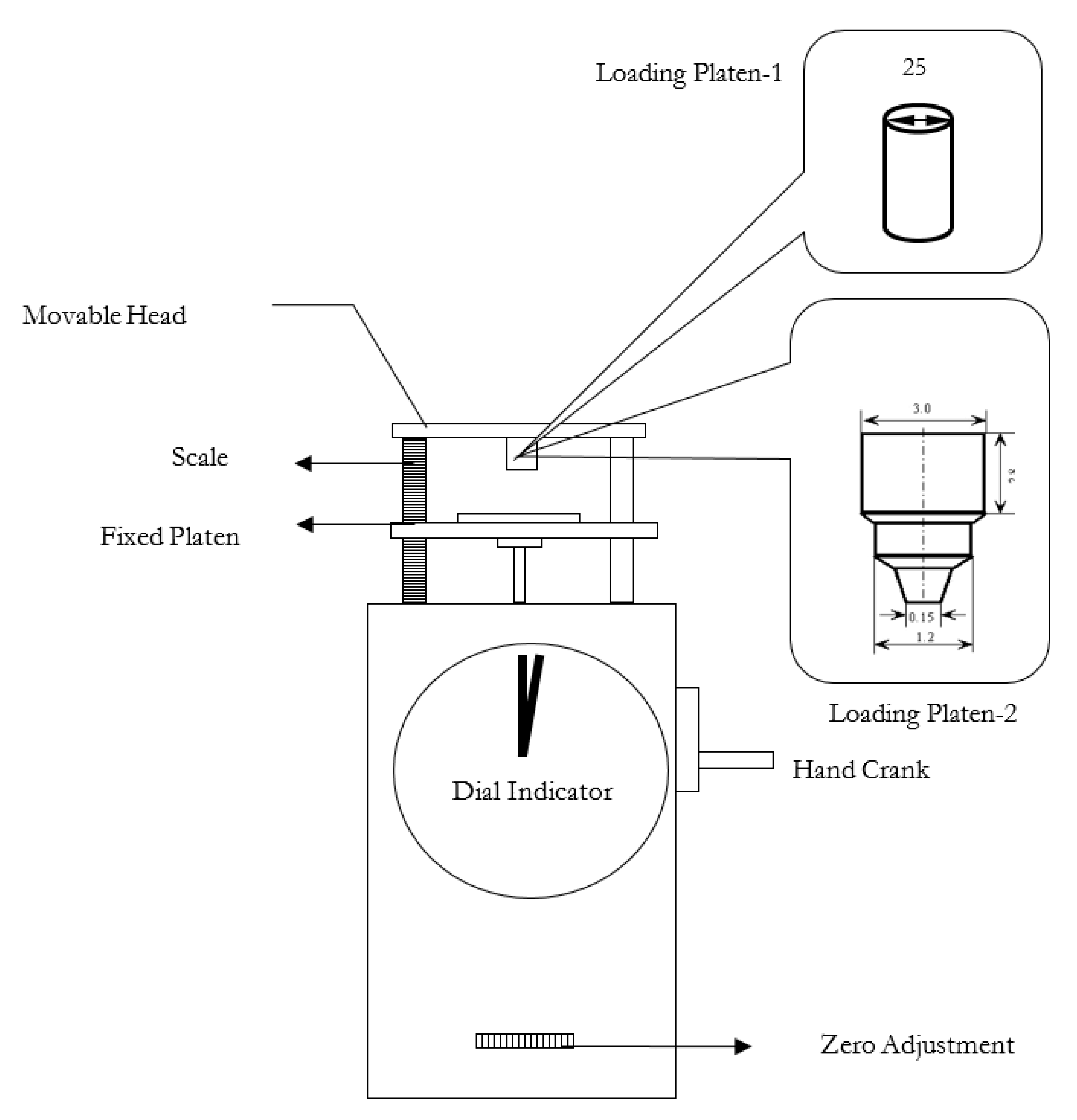
The compressive strengths of the bauxite ore (2) pisoids after both conventional and microwave heating were measured. Additionally, for comparison purposes, the strength index of the as-received bauxite ore was determined. The compressive strengths were measured using a Chatillon compression pellet tester, as shown in
Figure 3. The tester consisted of a movable head, a loading platen, a fixed platen and a dial indicator to show the load at failure. The sample was placed between the loading and the fixed platen and was compressed uniaxially. The cylindrical loading platen had a diameter of 25 mm, and the sample height could be measured using a scale. The zero adjustment was utilized to set the load to zero before the sample was compressed.
For both conventional and microwave processing, eighty bauxite particles within the particle size range of −6.68 mm + 4.70 mm were selected randomly for each strength index test. Since the particles were roughly spherical, then the contact area between the sample and the platens was a small constant value, and therefore, the compression could be considered as a point load. For a sample with a regular geometry, the failure load is only related to the square of the sample height [
42]. Consequently, the ratio of the failure load to the square of the sample height was utilized to evaluate the strength of the bauxite particles using the following equation:
where P is the failure load (kN), h is the sample height (m), and I is the strength index (kN/m
2).
There is considerable controversy regarding the effect of particle size on the microwave heating behaviors of materials. Consequently, compressive strength index measurements were carried out on individual bauxite particles after conventional and microwave treatment as a function of processing time, and hence, moisture content. The conventional and microwave experiments were performed for various times at 900 °C and 1200 W, respectively. For these studies, relatively large individual particles of bauxite (3) were randomly selected from within the three size ranges of −4.5 cm + 3.5 cm, −3.5 cm + 3.0 cm and −3.0 cm + 2.5 cm. For each size range, ten microwave experiments were performed over a range of times and also for ten conventional microwave times. For the strength indices, since the bauxite (3) particles were irregular, then loading platen-2 was employed to reduce the influence of surface area on the measurements. As shown in
Figure 4, loading platen-2 had a very small contact area with a diameter of 1.5 mm. In these tests, each sample was placed on the fixed platen in a manner such that the sample was most stable. The sample heights between the loading platen and the fixed platen were set at 3.4 cm, 3.5 cm and 3.7 cm for samples of −3.0 + 2.5 cm, −3.5 + 3.0 cm and −4.5 + 3.5 cm, respectively. The sample failure strength was measured, and the sample strength index was calculated using Equation (8). Additionally, the sample mass was measured both before and after the experiment, and thus, the moisture loss could be determined for a particular processing time.
4. Conclusions
(1) The decomposition processes of a gibbsite-type bauxite ore were investigated using thermogravimetric analysis (TGA), and the results showed that the decomposition occurred in two stages. Firstly, a small amount of about 1% loosely bonded water was removed up to about 100 °C. From about 175 °C to 300 °C, the mass loss was about 22%, which was greater than that for the decomposition of gibbsite to boehmite. Thus, gibbsite decomposed to boehmite and also to alumina. From about 300 °C up to 400 °C, the remaining more stable boehmite was converted into alumina. At higher temperatures, there was no mass change, indicating that all the water had been removed.
(2) The permitivities of the gibbsite-type bauxite ore were determined. The removal of the free water resulted in a slight increase in the permittivities, and this was followed by a slight decrease up to about 175 °C. Subsequently, the permitivities increased slowly and then more rapidly, reaching a maximum at 290 °C for the real permittivity and 330 °C for the imaginary permittivity. Thereafter, the permitivities dropped rapidly and then more slowly to 500 °C for the real permittivity and 400 °C for the imaginary permittivity. Then, the permitivities increased slowly and then more rapidly with increasing temperature, before leveling off at about 1000 °C. Here, the imaginary permittivity increased more rapidly with temperature due to its dependency on conductivity. The frequency dependency for both permitivities was relatively weak.
(3) Microwave heating behavior studies were performed for the bauxite ore using the single replicate 24 factorial technique in an attempt to obtain a stable microwave dewatering temperature. The major factors considered were: heating time, sample mass, microwave power and sample bulk density. These variables could be ranked in terms of increasing positive influence on the sample temperature as follows: microwave power < sample mass < heating time. In the coded form, bulk density had a negative effect. An attempt was made to utilize the model to predict the conditions for thermal processing of the bauxite at 500 °C. However, the model predicted that thermal runaway would occur, resulting in excessively high temperatures and sintering. Consequently, compressive strength indices were measured below the sintering temperature, unless noted.
(4) In order to delineate the potential decreases in the strength indices with microwave processing, tests were performed on samples of a pisolitic bauxite ore, which were heated in a conventional furnace to various temperatures. From room temperature to 250 °C, there was only a slight decrease in the strength index, with the major decrease occurring over the temperature range from 200 °C to 400 °C. This temperature range corresponded to that observed in the TGA studies for the conversion of gibbsite to boehmite and alumina. At higher temperatures up to 1000 °C, there were only modest decreases in the strength index. Thus, the major decrease in strength could be attributed to the decomposition of gibbsite to alumina.
(5) With microwave processing, strength index decreases approaching those obtainable in conventional heating could be attained at relatively short processing times and high microwave powers. In comparison to conventional heating, the microwave treatment was not uniform, and although some particles were affected by the microwave radiation, others were not. Correspondingly, significant decreases in the strength indices of some particles could be achieved at very low bulk sample moisture removals, indicating that only a fraction of the particles were being selectively microwave treated. The strength index decreased with increasing microwave power, but at high powers, some sintering occurred, which resulted in an increased strength index.
(6) To further understand the effects of microwave radiation, single particle studies were performed for both conventional and microwave heating. For all sizes, the strength indices of the microwaved samples were lower than the conventionally heated samples. These lower strength indices occurred at lower water removals than for the conventionally heated samples. This would indicate that water removal process with microwave heating resulted in high internal water vapor pressures, which resulted in additional weakening. For microwave processing, the strength index decreased with increasing particle size, reflecting the increased microwave absorption. Additionally, some particles failed as a result of thermal processing only, and this effect was more significant for the microwaved samples.
(7) A laboratory-scale energy comparison was made between conventional heating and microwave heating for the same input power, sample mass and strength index reduction. With conventional heating, there was a significant thermal load associated with the requirement to heat the furnace up to the operating temperature. Consequently, heating of the surroundings is more significant than heating of the sample for conventional heating. On the other hand, for microwave processing only, the sample is heated, and very little heat is lost to the surroundings. Consequently, for the laboratory tests reported in this research, the energy requirements for microwave processing were significantly lower than those for conventional heating.