Review on the Beneficiation of Li, Be, Ta, Nb-Bearing Polymetallic Pegmatite Ores in China
Abstract
:1. Introduction
2. Geological Aspects of LPPO Deposits in China
3. Methods in Recovering Li2O and BeO Commonly Used in China
3.1. Manual and Color Sorting
3.2. X-ray Transmission Sorting
3.3. Gravity Separation
3.4. Dense Medium Separation
3.5. Magnetic Separation
3.6. Flotation Method
3.6.1. Slime Treatment Method before Flotation
| No. | Slime Processing Method | Technological Feature | Reference |
|---|---|---|---|
| 1 | Depression method by autogenous sodium silicate at alkaline pulp |
| [55] [57] |
| 2 | Selective erosion method |
| [58] [51] |
| 3 | Gravity desliming method | Deslime by a countercurrent gravity column. | [59] |
| 4 | Ultrasonic washing method |
| [60] |
| 5 | Ore washing method using surface-cleaning agents |
| [61] |
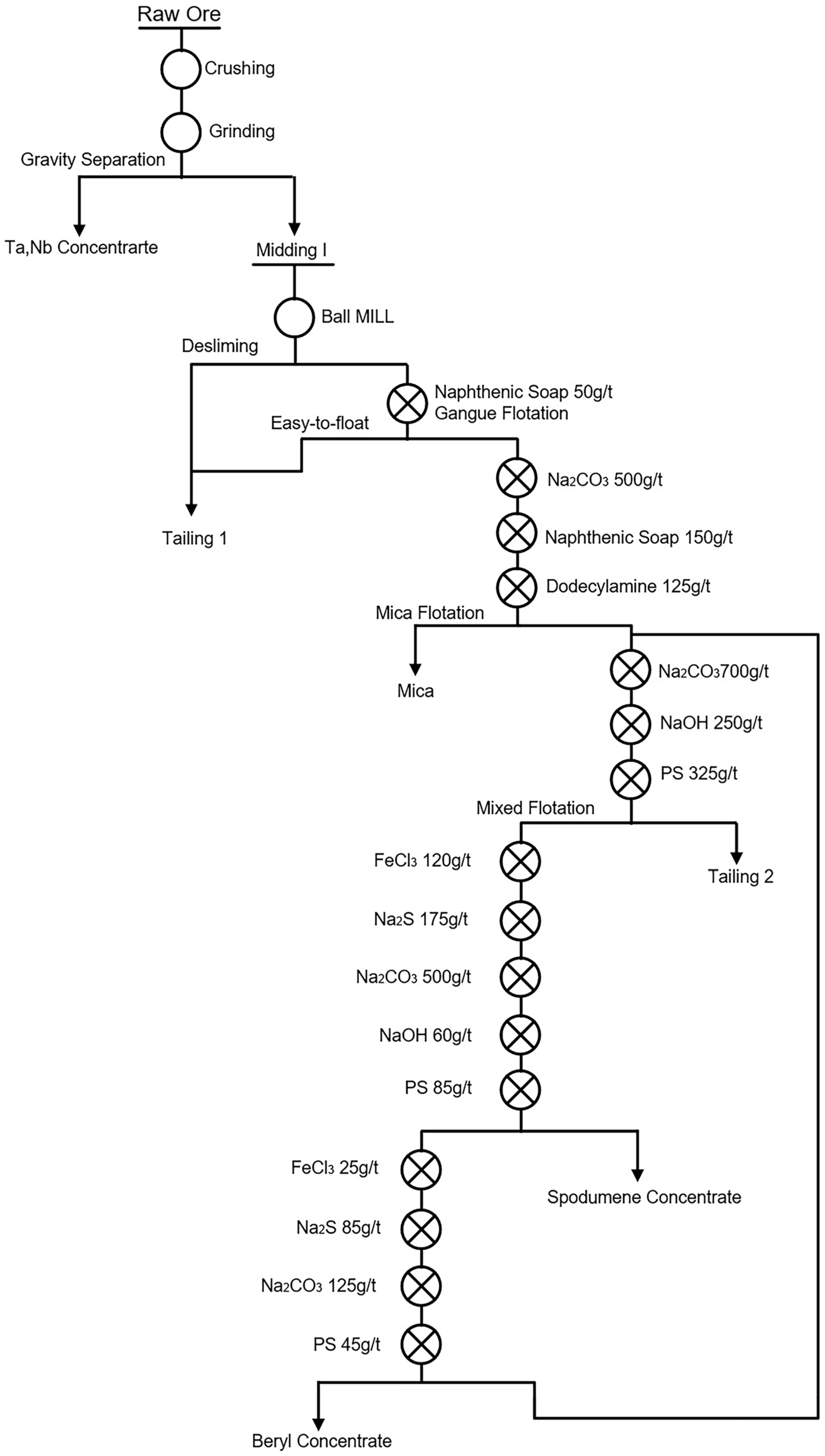
| 6 | Flotation desliming method | The slime minerals were floated by a small amount collector. The foam products with a lower grade of lithium than the original ore were directly mixed into tailings to achieve effective desliming. An example of a flotation desliming method is shown in Figure 3. | [9] |
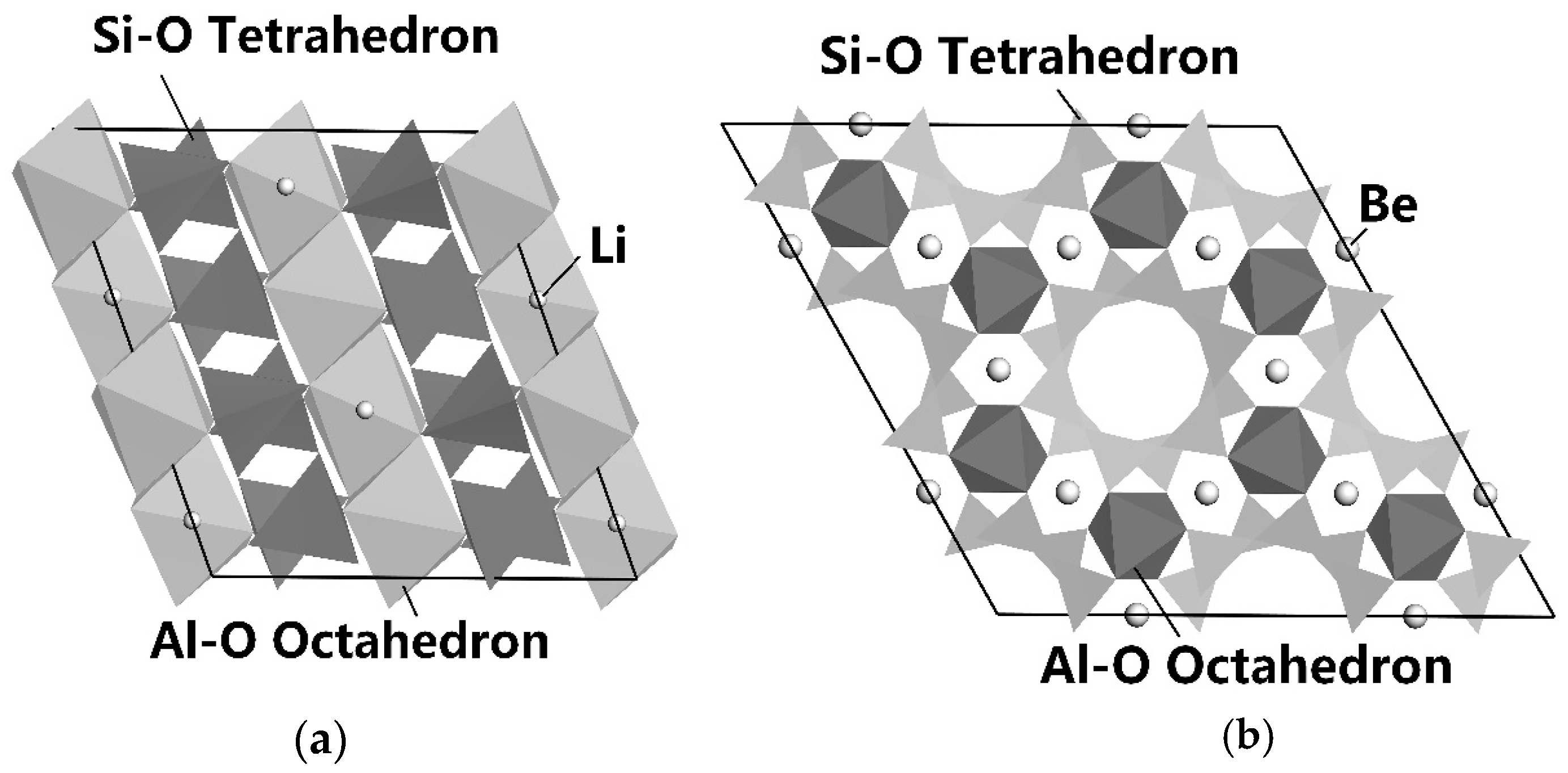
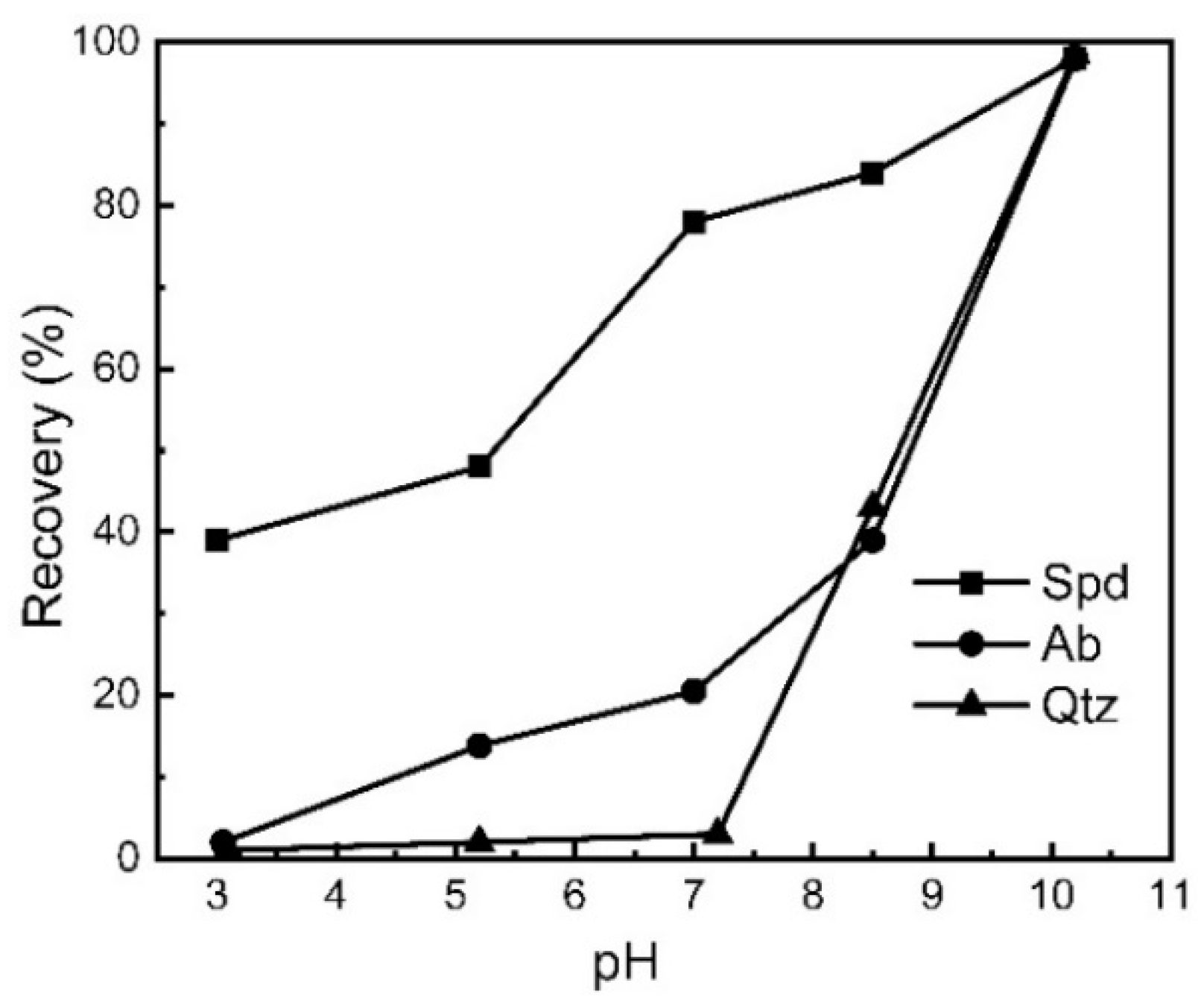
3.6.2. Enhanced Differences in Mineral Surface Properties
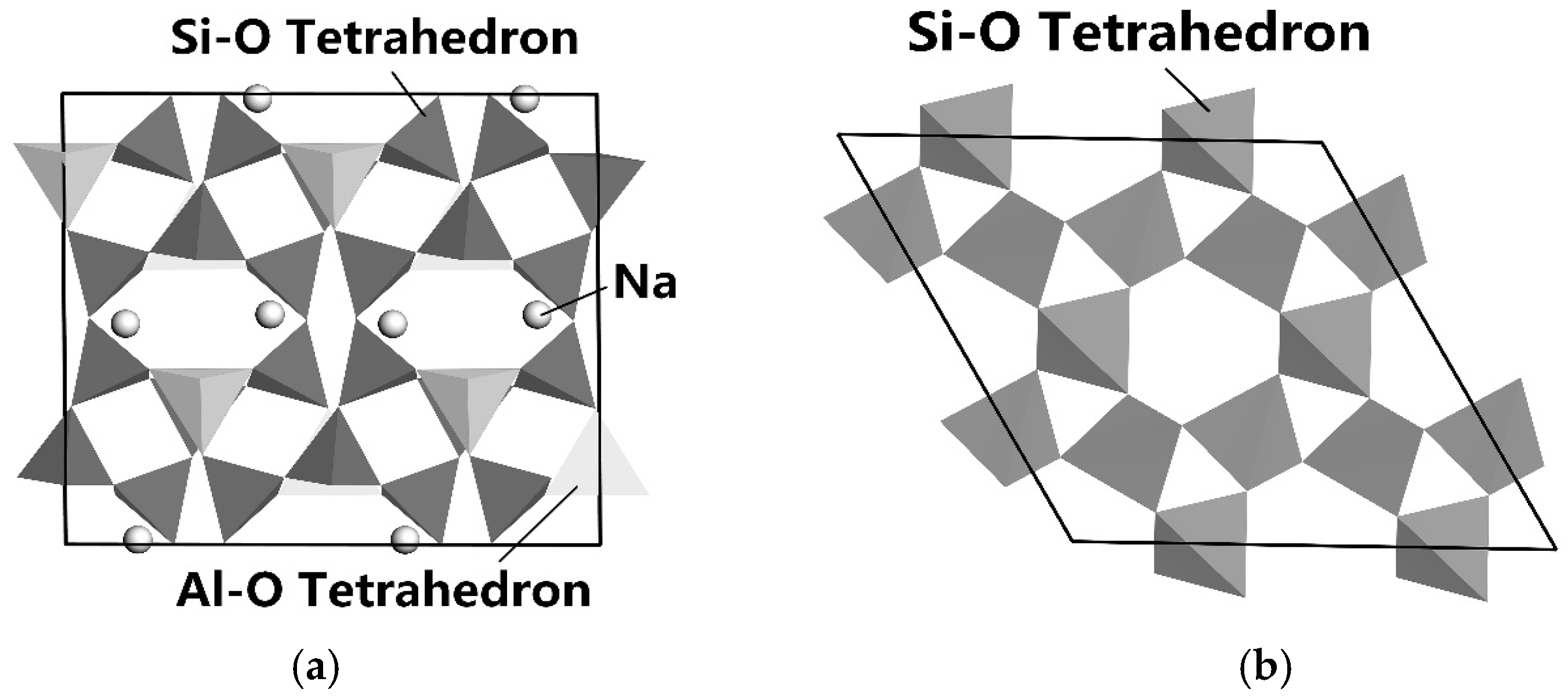
3.6.3. Selective Flotation Reagents for Lithium and Beryllium Mineral Separation
3.6.4. Industrial Application Cases of Lithium and Beryllium Separation in China
3.7. The Leaching Method Used in the Extraction of LPPOs
3.8. Brief Analysis of the Economic Efficiency of the Separation Methods
4. Conclusions
- LPPO, with large reserves and a high lithium grade, is an important lithium extraction ore. The beneficiation technologies of LPPO in China include the following: manual or color sorting, X-ray transmission sorting, dense medium separation, gravity separation, magnetic separation, flotation, and leaching.
- At present, the main industrial practice includes manual selection, dense medium beneficiation, and the flotation method. Color sorting and X-ray transmission sorting are promising alternatives to manual sorting, which are still in the research stage.
- When the ore contains magnetic substances, such as hematite, magnetite, lepidolite, tantalum-niobite, and other minerals, the magnetic separation method is used. Magnetic separation is an auxiliary method for improving the quality of lithium concentrate.
- Flotation is the main method for the separation of fine-grained intercalated lithium beryllium minerals. The key to the success of flotation lies in the treatment of argillaceous minerals and the selection of appropriate flotation reagents to expand the floatation difference between spodumene, beryl, and gangue minerals.
- Since beryllium, tantalum, niobium, etc. are all metals with important uses, beryllium, tantalum, and niobium in LPPO should be recovered. The development of lithium-beryllium separation and beneficiation technology is lagging behind and needs to be further strengthened.
- The future research on lithium-beryllium flotation separation will focus on: (1) the research and development of high-efficiency collectors, depressants, and activators; (2) the improvement of the efficiency of the separation process with the help of modern research methods and under the premise of economic sustainability to develop the green separation process.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Sahoo, S.K.; Tripathy, S.K.; Nayak, A.; Hembrom, K.C.; Dey, S.; Rath, K.R.; Mohanta, M.K. Beneficiation of lithium bearing pegmatite rock: A review. Miner. Process. Extr. Metall. Rev. 2022, 43, 1–27. [Google Scholar] [CrossRef]
- Meng, F.; McNeice, J.; Zadeh, S.S.; Ghahreman, A. Review of Lithium Production and Recovery from Minerals, Brines, and Lithium-Ion Batteries. Miner. Process. Extr. Metall. Rev. 2021, 42, 123–141. [Google Scholar] [CrossRef]
- Kesler, S.E.; Gruber, P.W.; Medina, P.A.; Keoleian, G.A.; Everson, M.P.; Wall, T.J. Global lithium resources: Relative importance of pegmatite, brine and other deposits. Ore Geol. Rev. 2012, 48, 55–69. [Google Scholar] [CrossRef]
- Salakjani, N.K.; Singh, P.; Nikoloski, A.N. Production of Lithium—A Literature Review Part 1: Pretreatment of Spodumene. Miner. Process. Extr. Metall. Rev. 2020, 41, 335–348. [Google Scholar] [CrossRef]
- Zhang, S.; Cui, L.; Kong, L.; Jiang, A.; Li, J. Overview of lithium resources and distribution at home and abroad. Nonferr. Met. Eng. 2020, 10, 95–104. [Google Scholar] [CrossRef]
- Yang, H.; Liu, L.; Ding, G. Status quo and development trend of global lithium resources. Conserv. Util. Miner. Resour. 2019, 39, 26–40. [Google Scholar] [CrossRef]
- Zhou, Y. Analysis on supply and demand situation and external dependence of Lithium resources in China. Resour. Ind. 2019, 21, 46–50. [Google Scholar] [CrossRef]
- Zhang, B.; Qi, F.; Gao, X.; Li, X.; Shang, Y.; Kong, Z.; Jia, L.; Meng, J.; Guo, H.; Fang, F.; et al. Geological characteristics, metallogenic regularity, and research progress of lithium deposits in China. China Geol. 2022, 5, 734–767. [Google Scholar] [CrossRef]
- Sun, C. Mineral Processing Engineer’s Manual; Metallurgical Industry Press: Beijing, China, 2015; Volume 4, pp. 59–129. ISBN 978-7-5024-6797-5. [Google Scholar]
- Rao, N.K.; Sreenivas, T. Beryllium—Geochemistry, Mineralogy and Beneficiation. Miner. Process. Extr. Metall. Rev. 1994, 13, 19–42. [Google Scholar] [CrossRef]
- Gupta, C.K.; Saha, S. Extractive Metallurgy of Beryllium. Miner. Process. Extr. Metall. Rev. 2010, 22, 413–451. [Google Scholar] [CrossRef]
- Babu, R.S.; Gupta, C.K. Beryllium Extraction—A Review. Miner. Process. Extr. Metall. Rev. 2007, 4, 39–94. [Google Scholar] [CrossRef]
- Wang, J.; Yan, Z.; Xu, L.; Li, K.; Li, Y.; Zheng, Y.; Wang, H.; Wang, Y. Resource potential analysis of pegmatite type lithium beryllium in Dahongliutan Xinjiang. Gold Sci. Technol. 2019, 27, 802–815. [Google Scholar] [CrossRef]
- Li, C.; Li, Q.; Zhu, J. Overview of lithium resources at home and abroad and its beneficiation, metallurgy and processing technology. World Nonferr. Met. 2001, 8, 4–8. [Google Scholar]
- Editorial Board of Handbook of Mineral Resources Industry Requirements. Mineral Resources Industry Requirements Manual; 2014 Revised Edition; Geological Publishing House: Beijing, China, 2014. [Google Scholar]
- Che, X.; Wang, R.; Hu, H.; Zhang, W.; Hang, X. Beryllium mineralization in the topaze-lithium mica granite in Yichun, Jiangxi province: Beryllium phosphate mineral assemblages. Acta Petrol. Sin. 2007, 6, 1552–1560. [Google Scholar] [CrossRef]
- Krishnamurthy, N.; Gupta, C.K. Rare Earth Metals and Alloys by Electrolytic Methods. Miner. Process. Extr. Metall. Rev. 2002, 22, 477–507. [Google Scholar] [CrossRef]
- Xu, X.; Jiao, Z.; Hai, G.; Yang, Y.; Li, J.; Teng, Y. Development status of beryllium industry. Xinjiang Non-Ferr. Met. 2021, 44, 4–8. [Google Scholar] [CrossRef]
- Liu, J.; Gao, L. Analysis on supply and demand pattern and industrial policy of American beryllium industry. China Min. Mag. 2022, 31, 31–36. [Google Scholar] [CrossRef]
- Butterman, W.C. Current Status of the Specialty Metals. Miner. Process. Extr. Metall. Rev. 1988, 3, 69–86. [Google Scholar] [CrossRef]
- Bulatovic, S.M. Handbook of Flotation Reagents (Volume III), Chemistry, Theory and Practice, Flotation of Gold PGM and Oxide Minerals; Elsevier B.V.: Amsterdam, The Netherlands, 2010; pp. 21–56. ISBN 978-7-122-19692-7. [Google Scholar]
- Foley, N.K.; Jaskula, B.W.; Piatak, N.M.; Schulte, R.F. Beryllium, Chap E of Critical Mineral Resources of the United States—Economic and Environmental Geology and Prospects for Future Supply; Schulz, K.J., DeYoung, J.H., Jr., Seal, R.R., II, Bradley, D.C., Eds.; U.S. Geological Survey: Reston, VA, USA, 2017; pp. E1–E32. [CrossRef]
- Li, H.; Hong, T.; Yang, Z.; Chen, J.; Ke, Q.; Wang, X.; Niu, L.; Xu, X. Zircon dating of rare metal granitic pegmatite, Cassiterite and tantalite U-Pb dating and Muscovite -(40) Ar/-(39) Ar dating: A case study of the Tugman North Li-Be deposit in the middle Altun. Acta Petrol. Sin. 2020, 36, 2869–2892. [Google Scholar] [CrossRef]
- Yao, W. Process progress and Industrial practice of caustic flotation of beryl. Xinjiang Min. Metall. 1985, 2, 1–4. [Google Scholar]
- Zheng, Y.; Wang, G. Experimental study on flotation of Bertrandit from Yangzhuang, Xinjiang. Xinjiang Nonferr. Met. 2012, 35, 66–68. [Google Scholar]
- Li, H.; Tan, X.; Zhang, X.; Zhang, L.; Yi, Y.; Wang, W. The present situation of beryllium resources and its beneficiation technology. Nonferr. Met. Sci. Eng. 2022, 13, 44–53. [Google Scholar]
- Rui, H. Study on Extraction Technology of Beryllium from Chrysoberyl Type Beryllium Ore. Ph.D. Thesis, Xiangtan University, Xiangtan, China, 2017. [Google Scholar]
- Deng, C. A Technology of Extracting Lithium Beryllium from Li-Be-Bearing Chrysolite. Ph.D. Thesis, Xiangtan University, Xiangtan, China, 2018; pp. 48–56. [Google Scholar]
- Bordbar-Khiabani, A.; Bahrampour, S.; Mozafari, M.; Gasik, M. Surface functionalization of anodized tantalum with Mn3O4 nanoparticles for effective corrosion protection in simulated inflammatory condition. Ceram. Int. 2022, 48, 3148–3156. [Google Scholar] [CrossRef]
- Nzeh, N.S.; Adeosun, S.; Popoola, A.P.; Adeleke, A.; Okanigbe, D. Process Applications and Challenges in Mineral Beneficiation and Recovery of Niobium from Ore Deposits—A Review. Miner. Process. Extr. Metall. Rev. 2022, 43, 833–864. [Google Scholar] [CrossRef]
- Lee, H.; Mishra, B. Recovery of Copper and Precious Metals and Separation of Lead from Flue Dust of Electronic Waste Processing. Miner. Process. Extr. Metall. Rev. 2020, 41, 153–161. [Google Scholar] [CrossRef]
- Zhang, A.; Wang, R.; Hu, H.; Zhang, H.; Zhu, J. The structure and petrological significance of niobite family mineral ring in Keketuohai No.3 Pegmatite dike, Altai. Acta Geol. Sin. 2004, 2, 181–189. [Google Scholar] [CrossRef]
- Liu, S.; Yang, Y.; Wang, D.; Dai, H.; Ma, S.; Liu, L.; Wang, C. Discovery and significance of granite type lithium ore body in Jiajika ore field, Sichuan province. Acta Geol. Sin. 2019, 93, 1309–1320. [Google Scholar] [CrossRef]
- Fei, G.; Fang, B. Ore fabric characteristics of Lijiagou spodumene deposit in Keeryin ore field, western Sichuan. Acta Mineral. Sin. 2015, 35, 1000. [Google Scholar]
- Pei, Y. Research status of keketuohai pegmatite deposit in Altay. Xinjiang West Leather 2020, 42, 1. [Google Scholar]
- Wang, H.; Xu, Y.; Yan, Q.; Zhang, X. Research progress of pegmatite type lithium deposit in Bailongshan, Xinjiang. Acta Geol. Sin. 2021, 95, 3085–3098. [Google Scholar] [CrossRef]
- Wang, T.; Hu, Z.; Wang, W. Research status and prospect of mineral processing and comprehensive utilization of rare lithium beryllium metals. Nonferr. Met. (Miner. Process. Sect.) 2020, 6, 24–29. [Google Scholar] [CrossRef]
- Xu, G. Enrichment of spodumene minerals in Keketuohai granitic pegmatite deposit. Xinjiang Non-Ferr. Met. 2010, 33, 7–9. [Google Scholar]
- Liu, L.; Wang, D.; Liu, X.; Li, J.; Dai, H.; Yan, W. The main types, distribution features and present situation of exploration and development for domestic and foreign lithium mine. Geol. China 2017, 44, 263–278. [Google Scholar] [CrossRef]
- Gao, Y.; Bagas, L.; Li, K.; Jin, M.; Liu, Y.; Teng, J. Newly Discovered Triassic Lithium Deposits in the Dahongliutan Area, Northwest China: A Case Study for the Detection of Lithium-Bearing Pegmatite Deposits in Rugged Terrains Using Remote Sensing Data and Images. Front. Earth Sci. 2020, 8, 591966. [Google Scholar] [CrossRef]
- Wang, T.; Li, P.; Li, H.; Zou, J.; Wang, W.; Yang, K.; Wang, C. Recovery of associated elements from a pegmatite type lithium polymetallic ore in Xinjiang. Met. Mine 2021, 11, 81–85. [Google Scholar] [CrossRef]
- Song, X. Application of hand separation in a spodumene ore. Xinjiang Non-Ferr. Met. 2012, 35, 50–51. [Google Scholar]
- Gülcan, E.; Gülsoy, Y.Ö. Evaluation of complex copper ore sorting: Effect of optical filtering on particle recognition. Miner. Eng. 2018, 127, 208–223. [Google Scholar] [CrossRef]
- Zhang, S.; Guo, Y. Measurement of Gem Colour Using a Computer Vision System: A Case Study with Jadeite-Jade. Minerals 2021, 11, 791. [Google Scholar] [CrossRef]
- Liu, G.; Peng, T.; Liu, L.; Li, S. Recovery of lithium from a granite-type spodumene by mineral processing combined with gravity separation, color separation and flotation. Met. Mine 2021, 3, 124–129. [Google Scholar] [CrossRef]
- Ge, D.; Liang, D. Study on ore grade detection method based on X-ray transmission. Nonferr. Met. (Miner. Process. Sect.) 2019, 4, 87–93. [Google Scholar]
- Wu, Z. Key points of ore pre-concentration and waste disposal technology and selection of intelligent photoelectric beneficiation equipment. World Nonferr. Met. 2020, 16, 202–205. [Google Scholar] [CrossRef]
- Tao, J. Industrial test and research on dense medium beneficiation of spodumene ore. Non-Ferr. Met. (Benef. Part) 2002, 2, 13–16. [Google Scholar]
- Xian, H. Dense medium beneficiation technology and its application in spodumene beneficiation. Xinjiang Non-Ferr. Met. 2018, 41, 71–73. [Google Scholar]
- Liang, X.; Huang, J.; Wu, G.; Hang, G.; Zhang, Y.; Li, M. Extended continuous beneficiation test of dense medium in a spodumene ore. Mod. Min. 2017, 33, 132–134. [Google Scholar] [CrossRef]
- Dai, Y.; Wang, Q.; Zhao, J.; Zhou, H.; Wei, D.; Le, Y.; Liu, W. High efficiency recovery of lithium from a pegmatite spodumene ore. Met. Mine 2021, 9, 107–112. [Google Scholar] [CrossRef]
- Han, M.; Li, Z.; Sun, Z.; Sun, Y.; Liu, Y. Experimental study on comprehensive recovery of a Lithium polymetallic ore. Conserv. Util. Miner. Resour. 2012, 2, 27–31. [Google Scholar] [CrossRef]
- Xie, R.; Zhu, Y.; Liu, J.; Li, Y.; Wang, X.; Zhang, S. Research Status of Spodumene Flotation: A Review. Miner. Process. Extr. Metall. Rev. 2020, 42, 321–334. [Google Scholar] [CrossRef]
- Wang, Y.; Sun, N.; Chu, H. Surface dissolution behavior and its influences on the flotation separation of spodumene from silicates. Sep. Sci. Technol. 2021, 56, 1407–1417. [Google Scholar] [CrossRef]
- Yu, F.; Jiang, M.; Wang, J.; Li, J.; An, J.; Pan, D. Study on Pre-desliming flotation tests of spodumene ore in Australia. Non-Ferr. Met. (Miner. Process. Part) 2019, 6, 69–73. [Google Scholar]
- Lv, Y.; Xing, W.; Li, J. Generalization on flotation theory and practice of spodumene and beryl. Nonferr. Met. 1965, 6, 14–19. [Google Scholar]
- Lv, Y. A new flotation separation method of spodumene and beryl—Selective desorption separation of contaminated ion Ca2+. Comprehensive utilization of mineral resources. Miner. Resour. 1980, 1, 11–19. [Google Scholar]
- Wang, Y.; Zhu, G.; Zhang, L.; Lu, D.; Wang, L.; Zhao, Y.; Zheng, H. Surface dissolution of spodumene and its role in the flotation concentration of a spodumene ore. Miner. Eng. 2018, 125, 120–125. [Google Scholar] [CrossRef]
- Gao, D.; Wang, Y.; Zheng, H.; Chu, H.; Lu, D.; Zheng, X. Effect of surface pretreatment on flotation of spodumene and silicate minerals. J. Chin. Univ. Min. Technol. 2020, 49, 991–997. [Google Scholar]
- Chu, H.; Chen, L.; Lu, D.; Wang, Y.; Zheng, X. Ultrasonic pretreatment of spodumene with different size fractions and its influence on flotation. Ultrason. Sonochem. 2022, 82, 105889. [Google Scholar] [CrossRef]
- Ji, G.; Wang, Y. Application of non-dissolution cleaning technology for Lithium beryllium minerals. Xinjiang Non-Ferr. Met. 2020, 1, 73–76. [Google Scholar]
- Xie, R.; Zhu, Y.; Li, Y.; Han, Y. Flotation behavior and mechanism of a new mixed collector on separation of spodumene from feldspar. Colloids Surf. A 2020, 599, 124932. [Google Scholar] [CrossRef]
- Xie, R.; Zhu, Y.; Han, X.; Zhang, M.; Li, Y. Study on flotation performance and action mechanism of a new spodumene collector DRQ-3. Met. Mine 2020, 6, 68–74. [Google Scholar]
- Xie, R.; Zhu, Y.; Liu, J.; Li, Y. Effects of metal ions on the flotation separation of spodumene from feldspar and quartz. Miner. Eng. 2021, 168, 106931. [Google Scholar] [CrossRef]
- Wang, Y.; Zhu, Y.; Xie, R.; Cheng, Z. Crystal chemical gene characteristics and floatability prediction of albite. Met. Mine 2020, 6, 81–86. [Google Scholar]
- Xu, L.; Peng, T.; Tian, J.; Lu, Z.; Hu, Y.; Sun, W. Anisotropic surface physicochemical properties of spodumene and albite crystals: Implications for flotation separation. Appl. Surf. Sci. 2017, 426, 1005–1022. [Google Scholar] [CrossRef]
- Wang, Y.; Yu, F.; Chen, X. Selective flotation between spodumene and beryl. Rare Met. 2005, 3, 320–324. [Google Scholar] [CrossRef]
- Xiang, H.; He, R.; Zhang, H. Flotation research on spodumene pure mineral. China Sci. Technol. Inf. 2015, 5, 64–66. [Google Scholar] [CrossRef]
- Liu, N. Experimental study on flotation of spodumene in V26 and V38 ore bodies of Keketuohai Rare Mine. Xinjiang Non-Ferr. Met. 2008, 5, 48–49. [Google Scholar]
- Tian, J.; Xu, L.; Wu, H.; Fang, S.; Deng, W.; Peng, T.; Sun, W.; Hu, Y. A novel approach for flotation recovery of spodumene, mica and feldspar from a lithium pegmatite ore. J. Clean. Prod. 2018, 174, 625–633. [Google Scholar] [CrossRef]
- Zhao, Y. Exploration of flotation recovery of high grade spodumene. Xinjiang Non-Ferr. Met. (S1) 2005, 37–38+41. [Google Scholar]
- He, Y.; Xie, Z. Discussion on beneficiation test of a spodumene ore in western Sichuan. Chem. Miner. Process. 2017, 46, 13–15+51. [Google Scholar]
- Zhang, J.; Wang, W.; Dong, F. Experimental study on flotation of spodumene ore. Acta Mineral. Sin. 2013, 33, 423–426. [Google Scholar]
- Wang, Y.; Yu, F. Flotation of spodumene and beryl with a new collector. J. Cent. South Univ. Sci. Technol. 2005, 5, 93–97. [Google Scholar] [CrossRef]
- He, J. Application of new collector in lithium beryllium flotation. Xinjiang Non-Ferr. Met. 2009, 32, 37–38. [Google Scholar]
- Weng, C.; Wen, S. Desliming and flotation recovery test of a low-grade spodumene in Southern Jiangxi Province. Mod. Min. 2017, 33, 126–131. [Google Scholar] [CrossRef]
- Xu, L.; Hu, Y.; Tian, J.; Wu, H.; Yang, Y.; Zeng, X.; Wang, Z.; Wang, J. Selective flotation separation of spodumene from feldspar using new mixed anionic/cationic collectors. Miner. Eng. 2016, 89, 84–92. [Google Scholar] [CrossRef]
- Wu, H.; Tian, J.; Xu, L.; Fang, S.; Zhang, Z.; Chi, R. Flotation and adsorption of a new mixed anionic/cationic collector in the spodumene-feldspar system. Miner. Eng. 2018, 127, 42–47. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, Y.; Sun, J.; Dong, Y. Study on the action rule and mechanism of sodium carbonate, sodium fluoride and sodium sulfide on Ca2+ and Fe3+ activated beryl and spodumene. Rare Met. 1983, 4, 2–7. [Google Scholar]
- Liu, F.; Sun, C. Effect of adding sequence of metal cation and dodecylamine on flotation of silicate minerals. Non-Ferr. Met. Miner. Process. Sect. 2011, 4, 58–62. [Google Scholar] [CrossRef]
- Jiao, Y. Preparation and application of peroxide fatty acid soap. Chem. World 1988, 8, 337–341. [Google Scholar] [CrossRef]
- Luo, X.; Lv, L.; Chen, X.; Zhou, H. Direct flotation process for a low-grade refractory spodumene ore in Jiangxi Province. Nonferr. Met. Eng. 2012, 2, 36–39. [Google Scholar] [CrossRef]
- Zhu, J.; Yu, M. Mechanism of activation and flotation separation of beryl and spodumene by grinding iron medium. Hunan Nonferr. Met. 1993, 9, 332–336. [Google Scholar]
- Piao, Z.; Wei, D.; Liu, Z. Effects of Small Molecule Organic Depressants on the Flotation Behavior of Chalcopyrite and Galena. J. Northeast. Univ. (Nat. Sci.) 2013, 34, 884–888. [Google Scholar] [CrossRef]
- Liu, F.; Sun, C. Influence of regulator and the adding order of sodium oleate to the flotation of silicate minerals. Met. Mine 2011, 3, 90–94. [Google Scholar]
- Hu, Y.; Wu, G.; Chu, H.; Wang, Y.; Sun, N.; Feng, H.; Lu, D.; Zheng, X. New Development in the flotation Theory and reagents of Spodumene Ore. Nonferr. Met. Eng. 2021, 11, 10–19. [Google Scholar] [CrossRef]
- Zhang, C. Study on flotation of Lithium beryllium from Sichuan Methyl card rare metal Ore. Sichuan Nonferr. Met. 1994, 1, 22–26. [Google Scholar]
- Ren, W. Separation of lithium beryllium from Keketuohai beryllium ore. Xinjiang Non-Ferr. Met. 2012, 5, 67–69. [Google Scholar]
- Wu, Y.; Zhang, X.; Tian, X.; Yao, Z.; Luo, Y. Recovery of Lithium from Acidic Leaching Solution of Lithium-containing Ore. Hydrometall. China 2020, 39, 182–185. [Google Scholar] [CrossRef]
- Xu, Z.; Liang, J.; Li, H.; Guo, J. Research Status and Prospects of Lithium Extraction from Lithium Containing Resources. Multipurp. Util. Miner. Resour. 2021, 5, 32–36. [Google Scholar] [CrossRef]
- Mao, S.; Li, G.; Zhong, L.; Wen, W.; Yu, X.; He, T.; Zhang, C. Present Situation and Prospect of Mineral Processing of Beryllium. Non-Ferr. Met. (Benef. Part) 2022, 6, 17–24. [Google Scholar] [CrossRef]
- Zhang, M.; Tian, H.; Wei, H.; Tian, Y.; Yu, J.; Zhao, Z. Present Situation and Development Trend of Lithium Resource Extraction Process. Rare Met. Cem. Carbides 2018, 46, 11–19. [Google Scholar]
- Liu, Y.; Liu, M.; Liu, Z. Experimental Study on new metallurgical treatment process of low-grade beryllium concentrate. Rare Met. Cem. Carbides 2014, 2, 13–15. [Google Scholar]
- Hu, Z. Analysis of Typical Lithium Extraction from Mines and its Economic Benefit. J. Salt Sci. Chem. Ind. 2019, 48, 5–8. [Google Scholar] [CrossRef]
- Tian, J.; Li, T.; Wang, M.; Zhao, H.; Qi, T. Research progress in lithium extraction technology for typical lithium ores. J. Hubei Univ. (Nat. Sci.) 2020, 42, 56–60. [Google Scholar]
- Su, H.; Zhu, Z.; Wang, L.; Qi, T. Research progress in the extraction and recovery of lithium from ore resources. CIESC J. 2019, 70, 10–23. [Google Scholar] [CrossRef]
- Xiao, M. Study on the process of extracting lithium salts from spodumene using sulfuric acid. Xinjiang Nonferr. Met. 1982, 2, 97–102. [Google Scholar] [CrossRef]
- Tian, Q.; Chen, B.; Chen, Y.; Ma, L.; Shi, X. Roasting and Leaching Behavior of Spodumene in Sulphuric Acid Process. Chin. J. Rare Met. 2011, 35, 118–123. [Google Scholar] [CrossRef]
- Guo, H.; Yu, H.; Zhou, A.; Lv, M.; Wang, Q.; Kuang, G.; Wang, H. Kinetics of leaching lithium from α-spodumene in enhanced acid treatment using HF/H2SO4 as medium. Trans. Nonferr. Met. Soc. China 2019, 29, 407–415. [Google Scholar] [CrossRef]
- Tu, T.; Guo, H.; Cheng, H.; Qiu, J.; Wang, X.; Liu, Q. Phase reconstruction and kinetics of lithium extraction by spodumene calcium oxide sintering method. Chem. Ind. Eng. Prog. 2020, 39, 3478–3486. [Google Scholar] [CrossRef]
- Chen, Y.; Liao, T.; Chen, B.; Tian, Q. Study on the extraction of lithium from spodumene by soda pressure boiling. Nonferr. Met. (Extr. Metall.) 2011, 9, 21–23+32. [Google Scholar] [CrossRef]
- Du, Y.; Zhang, J.; Li, S.; Ma, H. Separation performance and value analysis of typical pegmatite lithium ore. Min. Metall. 2020, 29, 49–52+97. [Google Scholar]
- Peng, J. Domestic lithium carbonate production process and benefit analysis. J. Salt Sci. Chem. Ind. 2019, 48, 18–21. [Google Scholar] [CrossRef]



| Mine Region | Deposit or Mining Area | Reserves (10,000 tons) | Average Grade (%) | ||
|---|---|---|---|---|---|
| Li2O | BeO | Li2O | BeO | ||
| Sichuan [31] | Jiajika #134 lithium deposit in Ganzi Prefecture | 41.2 | 6.5 | 1.20 | 0.04 |
| Sichuan [32] | Lijiagou deposit in Jinchuan County | 51.2 | 0.2~1.0 | 1.20~1.27 | 0.04~0.045 |
| Xinjiang [33] | Keketuohai #3 lithium deposit in Fuyun County | 92.0 | 5.8 | 0.98 | 0.043 |
| Xinjiang [12,37] | Dahongliutan lithium deposit in Hetian County | 506.0 | 16.0 | 0.93~3.44 | 0.54~0.63 |
| Minerals | Mineral Composition | Li2O Content (%) | Density (g/cm3) | Hardness (MoOS) | |
|---|---|---|---|---|---|
| Theoretical | Actual | ||||
| Spodumene | LiAl [Si2O6] | 8.03 | 4.5–8.0 | 3.20 | 6.5–7.0 |
| Lepidolite | KLi1.5Al1.5[Si3AlO10] [FOH]2 | 5.90 | 1.2–5.9 | 2.8–2.90 | 2.0–3.0 |
| Amblygonite | LiAl (PO4) (FOH) Li2O3·2LiFP2O5·Li2O | 10.10 | 8.0–9.5 | 3.00–3.20 | 6.0 |
| Zinnwaldite | KLiFeAl [Si3AlO10] [FOH]2 | 4.13 | 2.0–3.0 | 2.90–3.20 | 2.0–3.0 |
| Petalite | (LiNa) [AlSi4O11] | 4.89 | 2.0–4.0 | 2.39–2.46 | 6.0–6.5 |
| Eucryptite | LiAlSiO4 | 11.90 | 11.9 | 2.67 | 6.5 |
| Minerals | Mineral Composition | BeO Content (%) Theoretical Value | Density (g/cm3) | Hardness (MoOS) |
|---|---|---|---|---|
| Beryl | Be3Al2(Si6O18) | 5.03 | 2.60–2.90 | 7.5–8.0 |
| Bertrandite | Be4(Si2O7) (OH)2 | 15.13 | 2.60 | 6.0 |
| Xrizoberyl | Al2BeO4 | 7.09 | 3.50–3.80 | 8.5 |
| Phenacite | BeO2(SiO4) | 6.77 | 3.00 | 7.5 |
| Gelvin | Mn8(BeSiO4)6S2 | 4.86 | 3.30 | 6.0–6.6 |
| Getgelvin | Zn8 (BeSiO4)6S2 | 4.54 | 3.66 | - |
| Danolit | Fe8(BeSiO4) S2 | 1.47 | 3.40 | 5.5–6.0 |
| Minerals | Mineral Composition | Density (g/cm3) | Hardness (MoOS) |
|---|---|---|---|
| Tantalite | (Fe, Mn) Ta2O6 | 6.25–7.90 | 6.5 |
| Columbite | (Fe, Mn) Nb2O6 | 5.00 | 6.0 |
| Ferrotapiolite | (Fe, Mn) (Ta, Nb)2O6 | 5.00–7.90 | 6.0–6.5 |
| Quartz | SiO2 | 2.65 | 7.0 |
| Plagioclase | Na [AlSi3O8]-Ca [Al2Si2O8] | 2.61–2.76 | 6.0–6.5 |
| Albite | Na2O·Al2O3·6SiO2 | 2.62 | 6.0–6.5 |
| Muscovite | KAl2[AlSi3O10] [OH]2 | 2.77–2.88 | 2.5–4.0 |
| No. | Beneficiation Methods | Main Technological Features and Application Cases |
|---|---|---|
| 1 | Manual sorting | Suitable particle size of 10~25 mm. In Keketohai Mine of Xinjiang, manual sorting was used, as for raw ore, Li2O 1.5%~1.8%, a concentrate with an Li2O grade of 5%~6% and a recovery rate of 20%~30%, was obtained. |
| 2 | Color sorting | Suitable particle size of 2~10 mm. A sample was separated by a color selector. The color difference was obvious between the color sorting concentrate and color sorting tailing. Most of the dark gangue minerals were found in the color sorting tailing. A concentrate with an Li2O grade of 6.18% was obtained, and the Li2O recovery was 89.23%. |
| 3 | X-ray transmission sorting | Suitable particle size of −60 + 10 mm. The processing capacity is 50–70 tons per machine per hour. It is one of the most promising methods for replacing manual sorting. |
| 4 | Gravity separation | LPPO contains tantalite, columbite, or ferrotapiolite; thus, gravity beneficiation can separate the denser tantalite, columbite, or ferrotapiolite from spodumene and gangue. |
| 5 | Dense media separation | Suitable particle size of −10 mm + 0.5 mm. A dense medium with a density of 2.95~3.0 g/cm3 was used for an LPPO sample from Sichuan, obtaining a concentrate with an Li2O grade of 7.06% by a two-stage process of “one coarse and one fine” dense medium separation. Spodumene concentrate has a total recovery of 87.47%. |
| 6 | Magnetic separation | Magnetic separation is mainly used for the purification of spodumene and beryl concentrate. The high-quality concentrate product was obtained by magnetic separation to remove the impurity minerals with iron. |
| 7 | Flotation | Suitable particle size of −0.074 + 0.038 mm. LPPO flotation can be divided into direct flotation and reverse flotation. For example, a raw ore with Li2O grade of 0.895% and BeO grade of 0.054% from Keketohai Mine of Xinjiang was crushed and ground; and then gravity separation was conducted, followed by direct flotation. A lithium concentrate with Li2O grade of 6.01% and a recovery of 84.6%, and a beryllium concentrate with BeO grade 8.82% and a recovery of 60.2% were finally obtained. |
| 8 | Leaching | Leaching pretreatment: stable α-spodumene is heated into β-spodumene with higher activity at 1050 °C. A sulfuric acid leaching process under a leaching temperature of 250–280 °C and a leaching time of 15–60 min is the most commonly used process in industry. For example, Tian Qianqiu, in 2021, calcined a spodumene sample at 1050 °C for 30 min and then roasted it with sulfuric acid at 250 °C for 30 min. Finally, the leaching rate of Li2O reached 96.93% at room temperature, with a liquid–solid ratio of 1.85 for 15 min. |
| Code or Name of the Flotation Reagent | Polar Group or Molecular Structure | Application | Reference |
|---|---|---|---|
| OA (Oleic acid) | CH3(CH2)7CH=CH(CH2)7 COOH | Collectors for spodumene | [65,68] |
| OPS (Oxidized Paraffin Soap) | C=O, C–OH Mixed fatty acids | Collectors for spodumene or beryl | [69,70] |
| TO (Tal oil) | C=O, C–OH Fatty Acids and Rosin Acids | Collectors for spodumene or beryl | [71] |
| NAS (Naphthenic acid soap) | C=O, C–OH Naphthenic acid with five carbons | Collectors for spodumene or beryl | [72,73] |
| Name or Code of the Flotation Reagent | Polar Group or Molecular Formula | Application | Reference |
|---|---|---|---|
| YOA-15 | –COOH, –NH2 | New type of mixed collector for spodumene and beryl. | [74] |
| YZB-17 | –CONHOH, –C–OH | Mixed collector for spodumene or beryl flotation. | [75] |
| HL-1 | -COOH | A mixture of OA, OPS, and NAS; mixed collector for the separation of spodumene from albite. | [76] |
| NaOL/DTAC | -COOH and C15H34ClN | Dodecyl trimethyl ammonium chloride (DTAC); when the mole ratio of NaOL:DTAC is 9:1, spodumene and albite could be separated effectively. | [77] |
| NaOL/TTPC | -COOH and C26H56ClP | Tributyl tetradecyl phosphonium chloride (TTPC); a mixed collector for the separation of spodumene from albite. | [78] |
| HPA/DIDA | (HPA)–C–O–C, –NH2 (DIDA)–COOH | New mixed collector of spodumene under natural pH conditions. | [63] |
| Name or Code of the Flotation Reagent | Polar Group or Molecular Structure | Application | Reference |
|---|---|---|---|
| Soda | Na2CO3 | Depressant of spodumene and beryl activated by Ca2+. | [79] |
| Sodium sulfide | Na2S | Depressant of spodumene activated by Fe3+. When oleic acid was used as the collector, Na2S was added before the collector, which had a strong inhibitory effect on spodumene. | [80] |
| “Three bases” regulators | NaOH-Na2CO3-CaCl2 | Extensive combination of regulators: NaOH or Na2CO3 as pH regulators or inhibitors and CaCl2 as an activator of spodumene. | [81,82] |
| Lignosulfonate | C20H24Na2O10S2 | Special inhibitor for spodumene; used to separate spodumene and beryl. | [83] |
| Disodium EDTA | C10H14N2Na2O8·2H2O | Depressant for spodumene; used to separate spodumene and beryl. | [67] |
| Hexametaphosphate | (NaPO3)6 | Depressant for spodumene; used to separate spodumene and beryl. | [74] |
| No. | Flotation Process | Ore Dressing Plant | Reference |
|---|---|---|---|
| 1 | Preferential flotation of part of a spodumene—mixed flotation of spodumene and beryl—flotation separation between spodumene and beryl. | Keketuohai, Xinjiang | [87] |
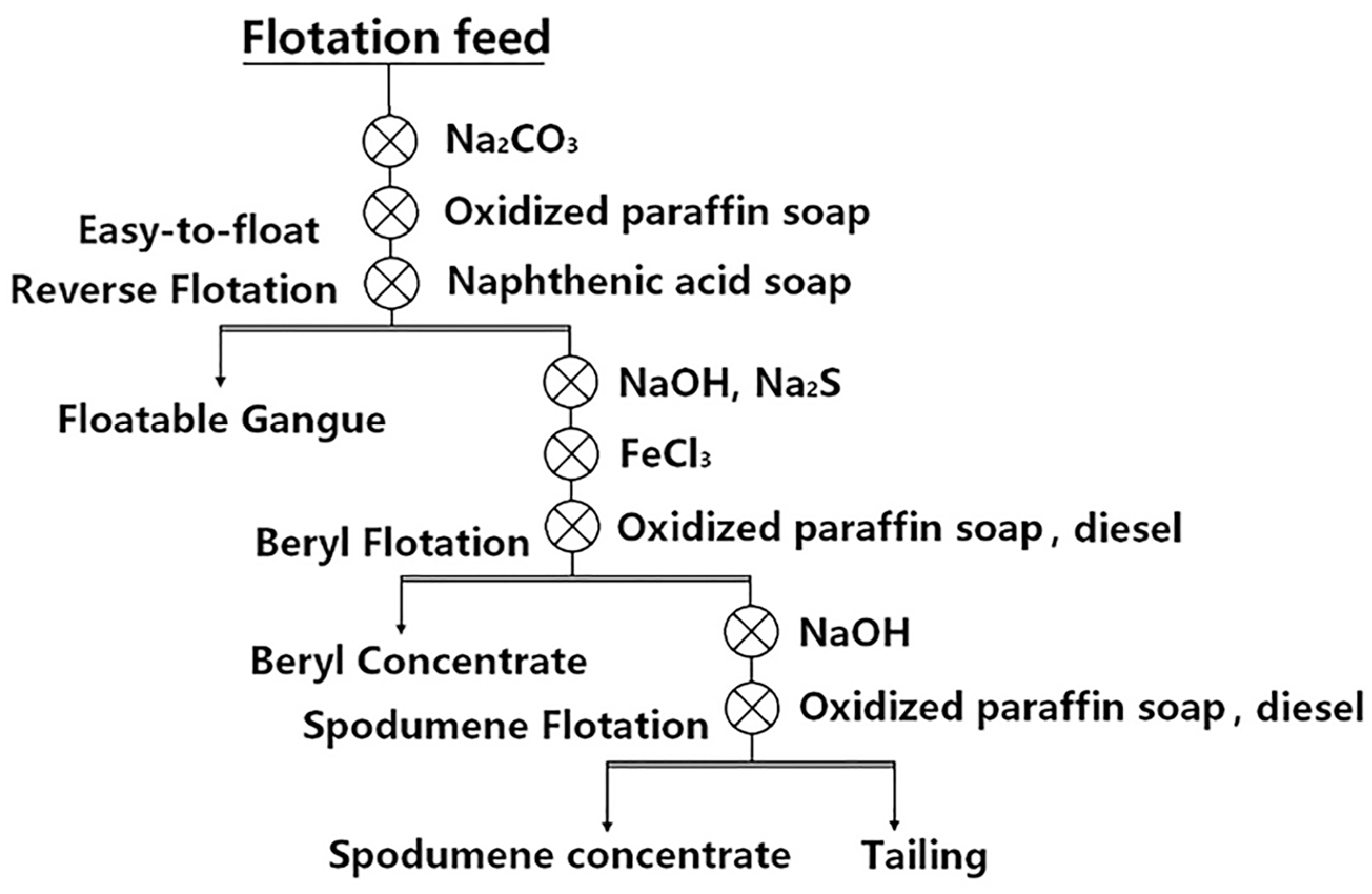
| 2 | Preferential flotation of beryl—flotation of spodumene. (Figure 8) | Keketuohai, Xinjiang | [9] |
| 3 | Preferential flotation of spodumene—flotation of beryl. | Keketuohai, Xinjiang | [56,88] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, S.; Liu, J.; Han, Y.; Zhang, S. Review on the Beneficiation of Li, Be, Ta, Nb-Bearing Polymetallic Pegmatite Ores in China. Minerals 2023, 13, 865. https://doi.org/10.3390/min13070865
Li S, Liu J, Han Y, Zhang S. Review on the Beneficiation of Li, Be, Ta, Nb-Bearing Polymetallic Pegmatite Ores in China. Minerals. 2023; 13(7):865. https://doi.org/10.3390/min13070865
Chicago/Turabian StyleLi, Siyang, Jie Liu, Yuexin Han, and Shumin Zhang. 2023. "Review on the Beneficiation of Li, Be, Ta, Nb-Bearing Polymetallic Pegmatite Ores in China" Minerals 13, no. 7: 865. https://doi.org/10.3390/min13070865





