The Effects of Chloride on the High-Temperature Pressure Oxidation of Chalcopyrite: Some Insights from Batch Tests—Part 1: Leach Chemistry
Abstract
:1. Introduction
- The rate of leaching is initially high (e.g., [66]).
2. Experimental
3. Ore Characterization
4. Background
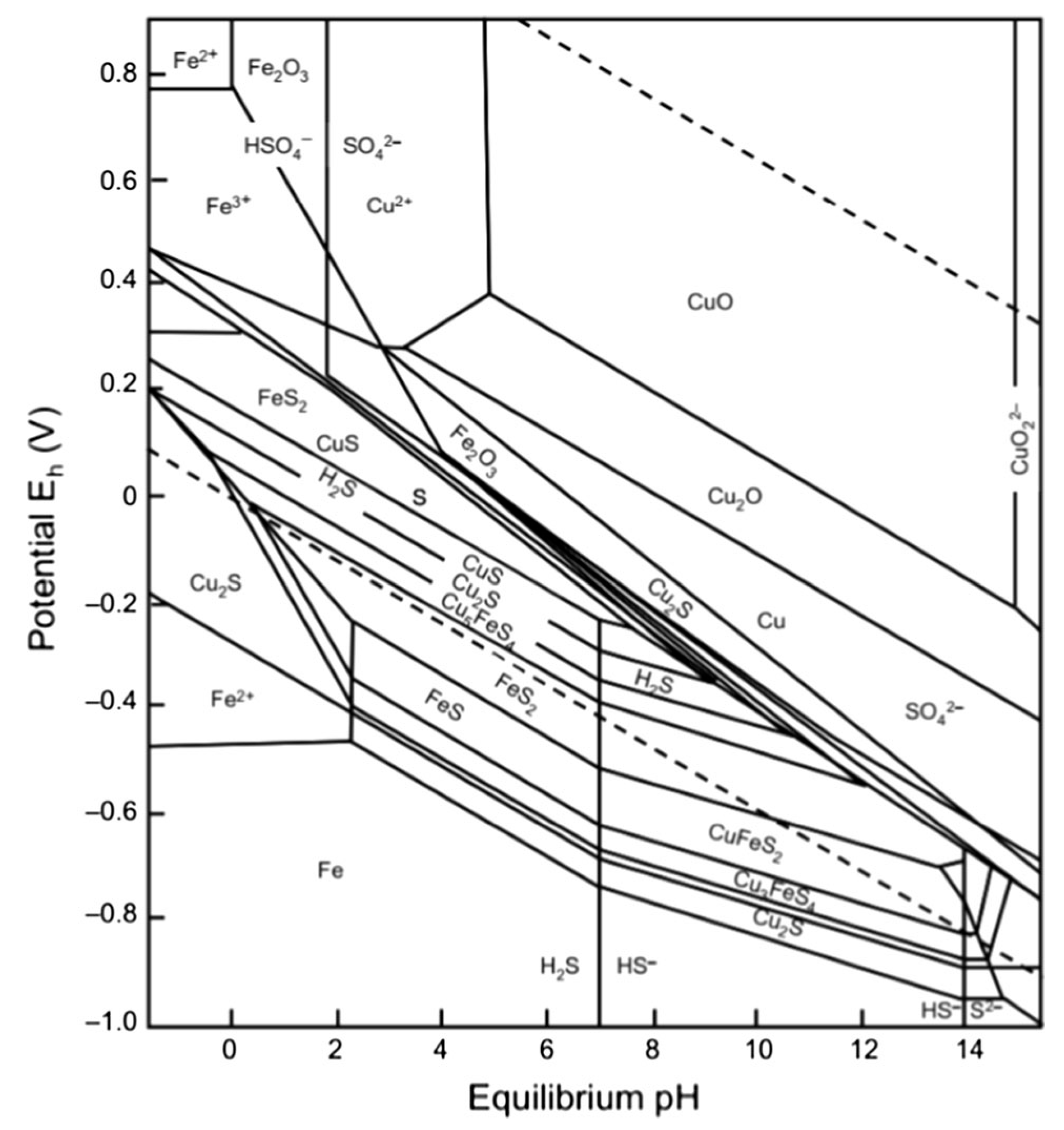
4.1. Eh-pH and Eh-log[Cl−] Diagrams
4.2. Copper Sulphides’ Extraction and the Effect of Eh
4.3. Surface Passivation of Chalcopyrite
5. Results
5.1. Copper Extraction during Batch Processing
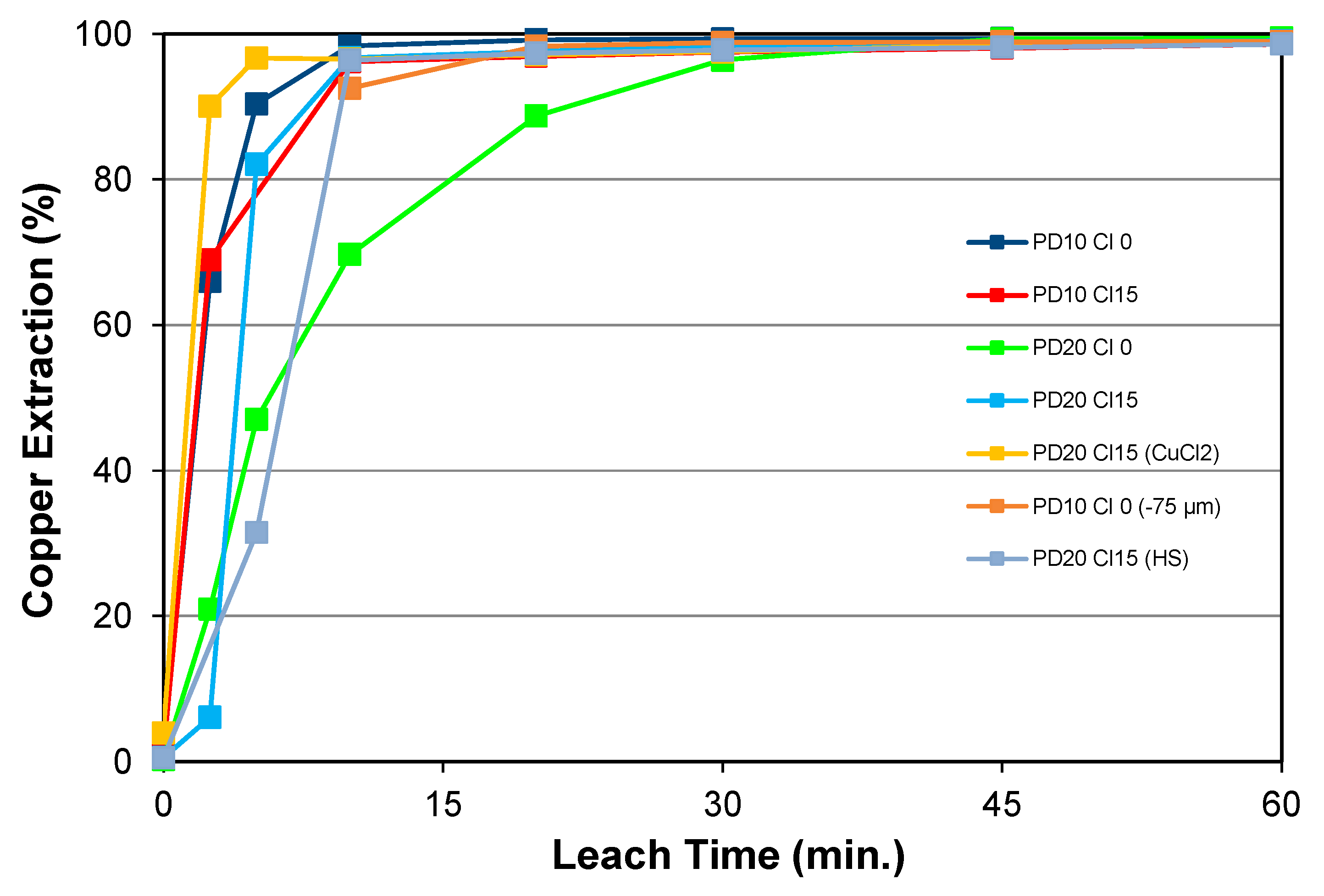
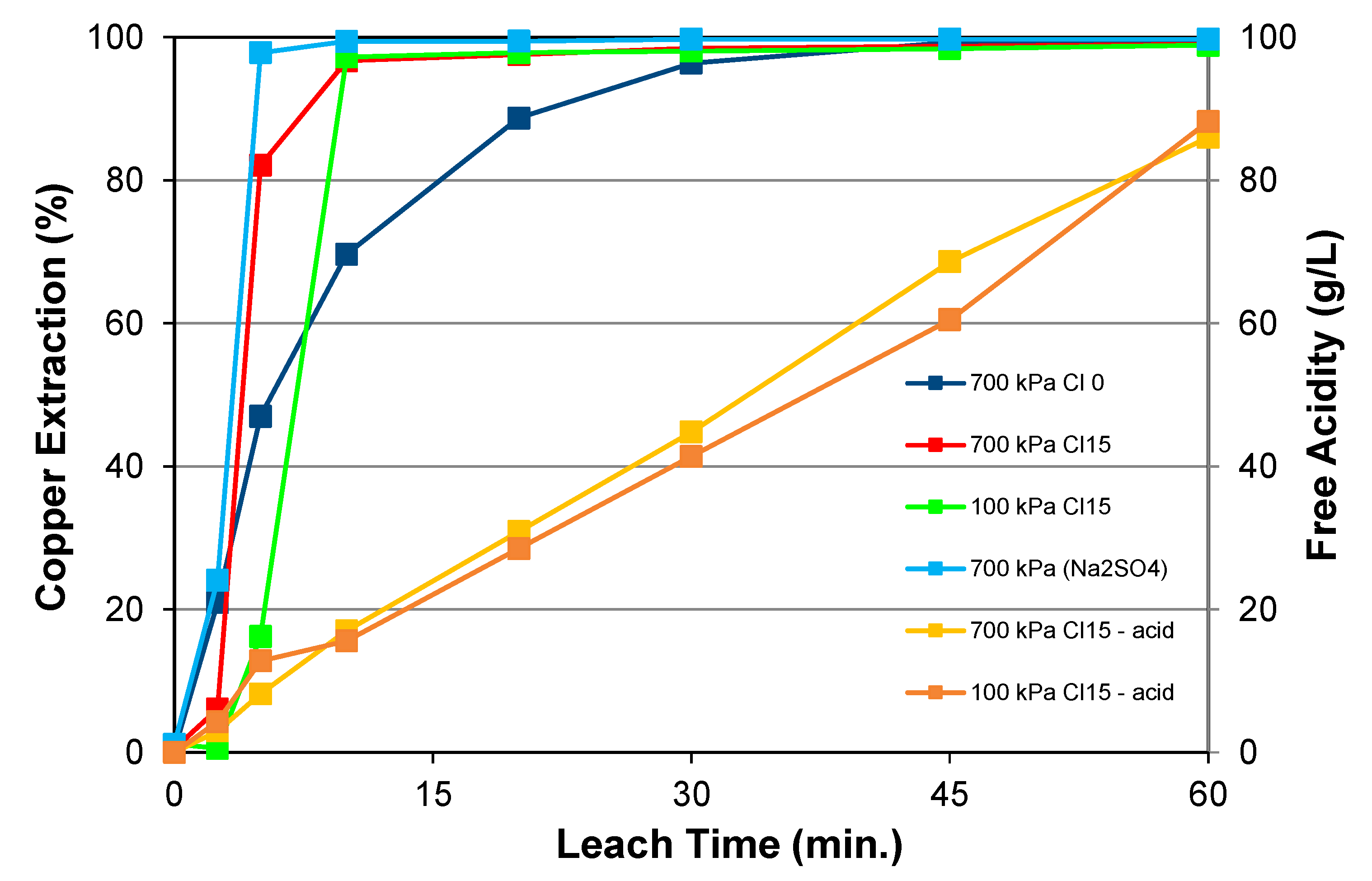
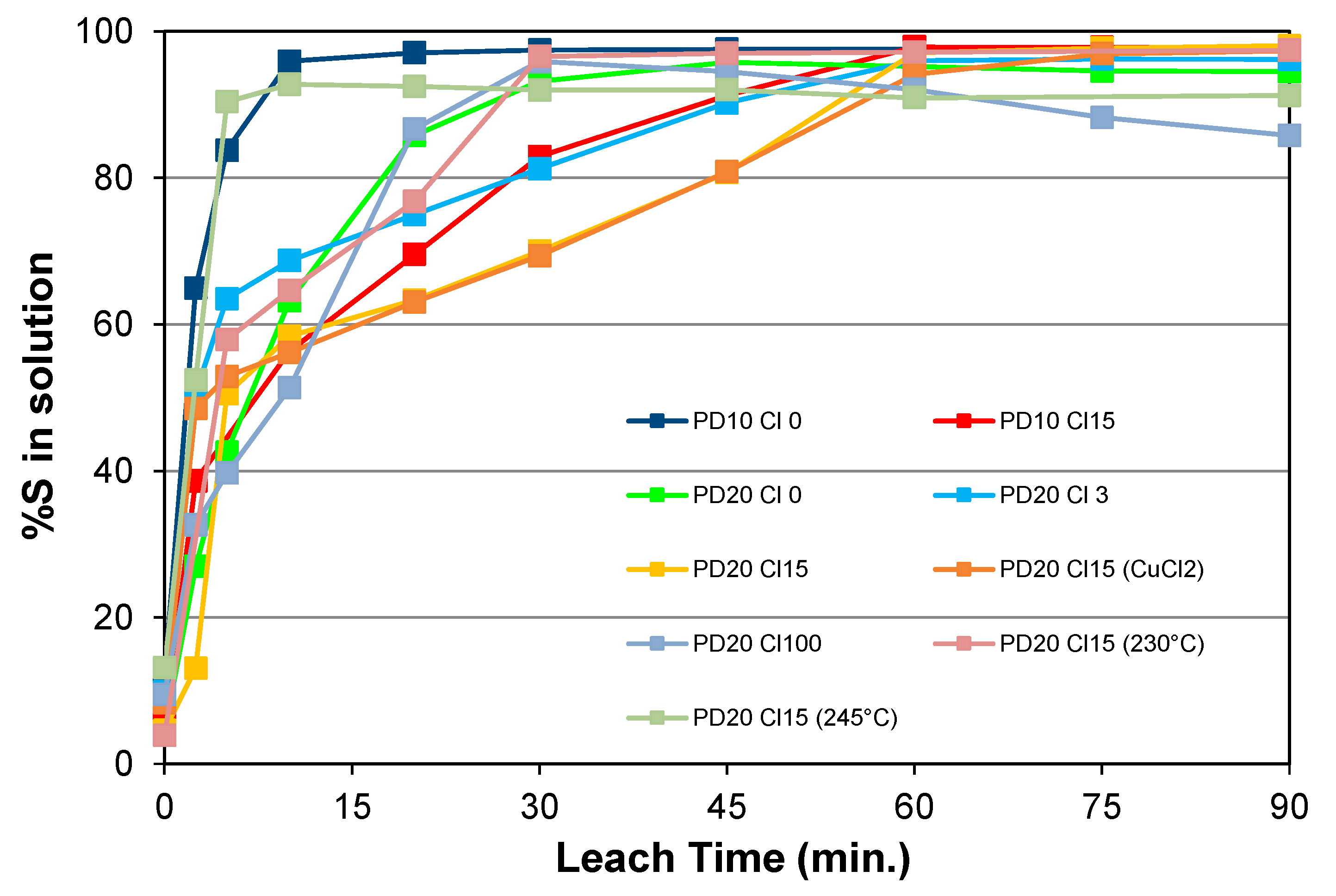
5.1.1. Effects of Chloride Addition, Pulp Density, and Particle Size
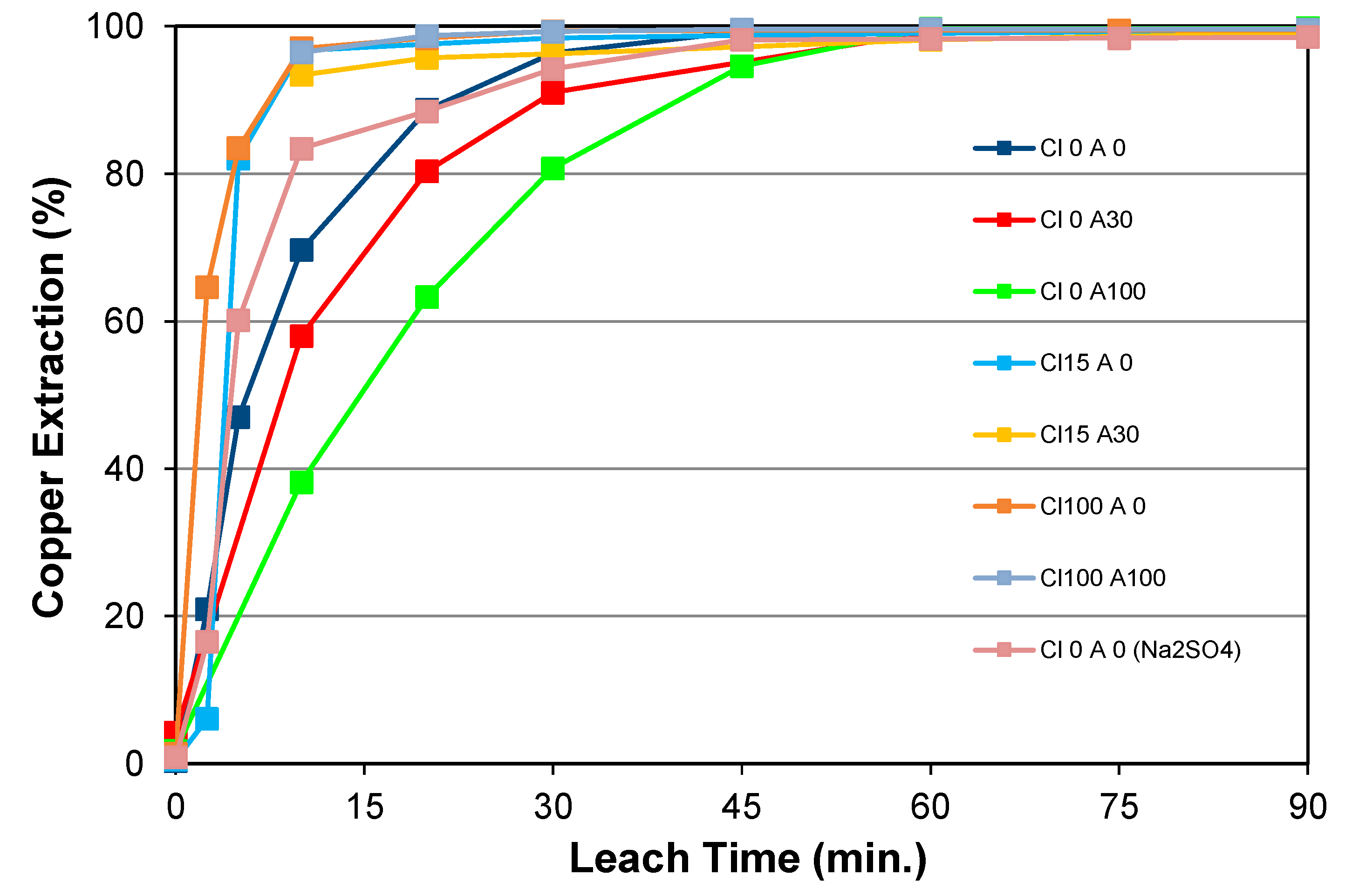
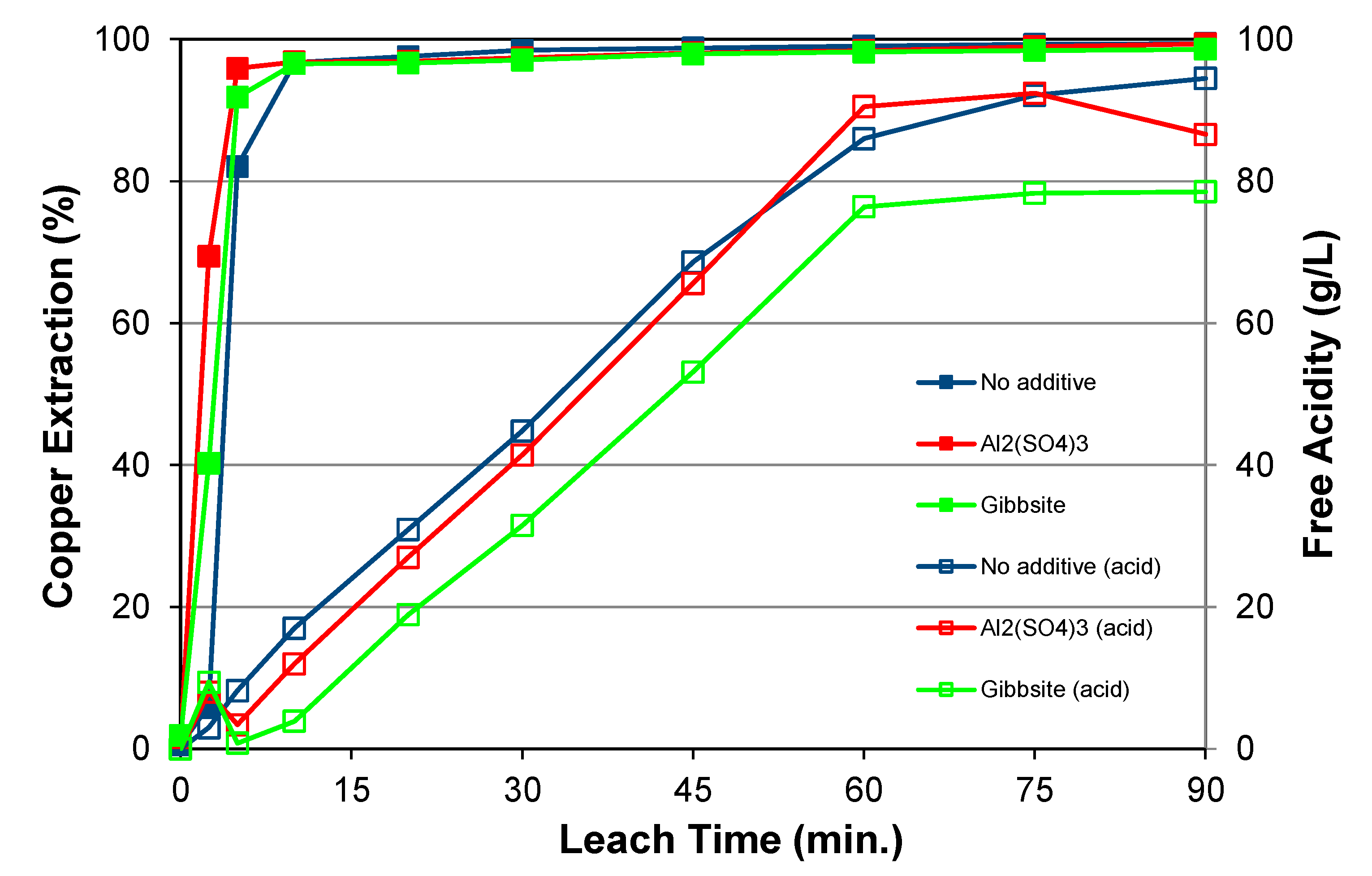
5.1.2. Effects of Acid Addition and Chloride
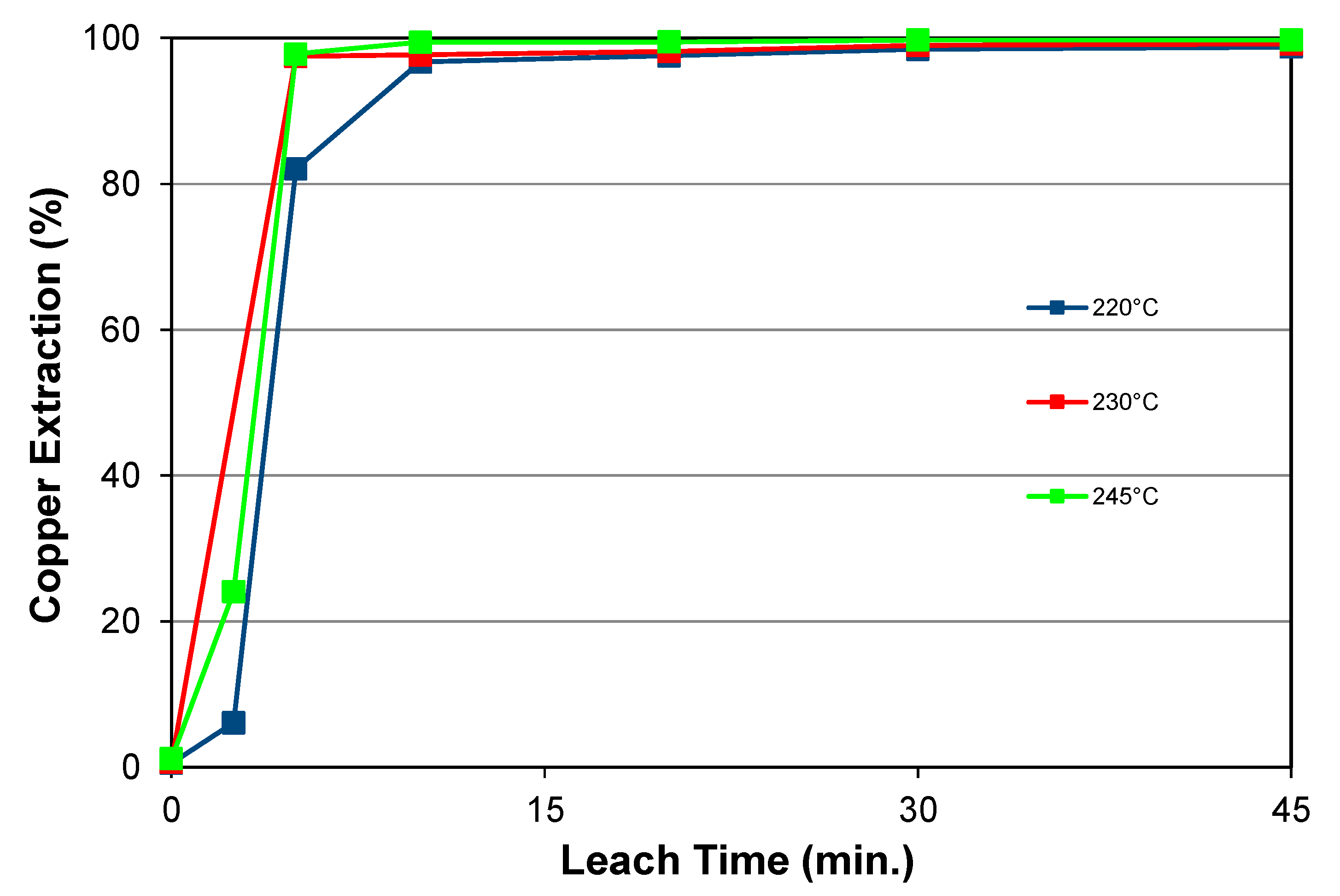
5.1.3. Effect of Temperature with Chloride Addition
5.1.4. Effect of Oxygen Pressure
5.2. Sulphuric Acid Addition and Formation
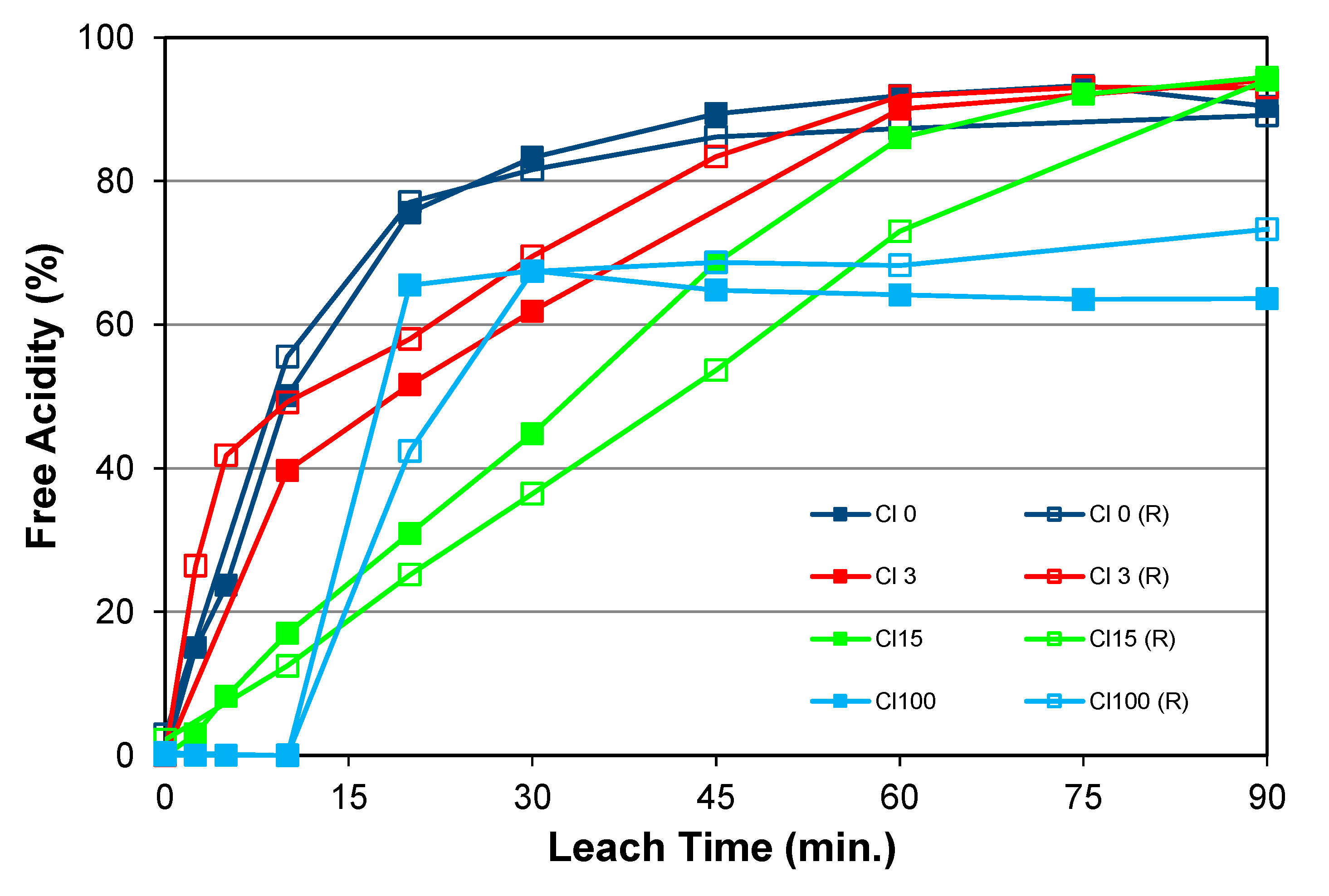
5.2.1. Effects of Pulp Density, Chloride Addition, and Temperature
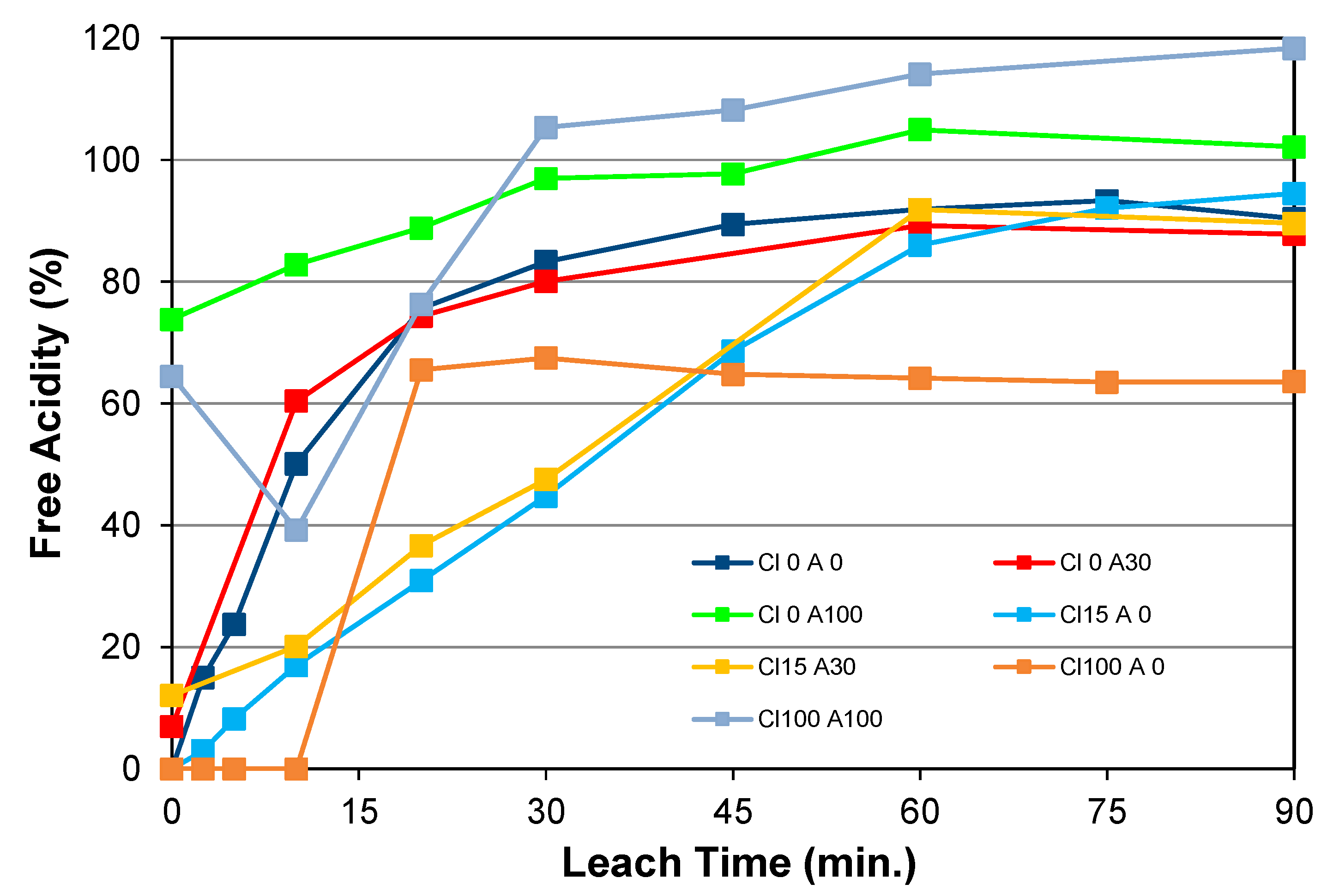
5.2.2. Effect of Initial Acid Addition
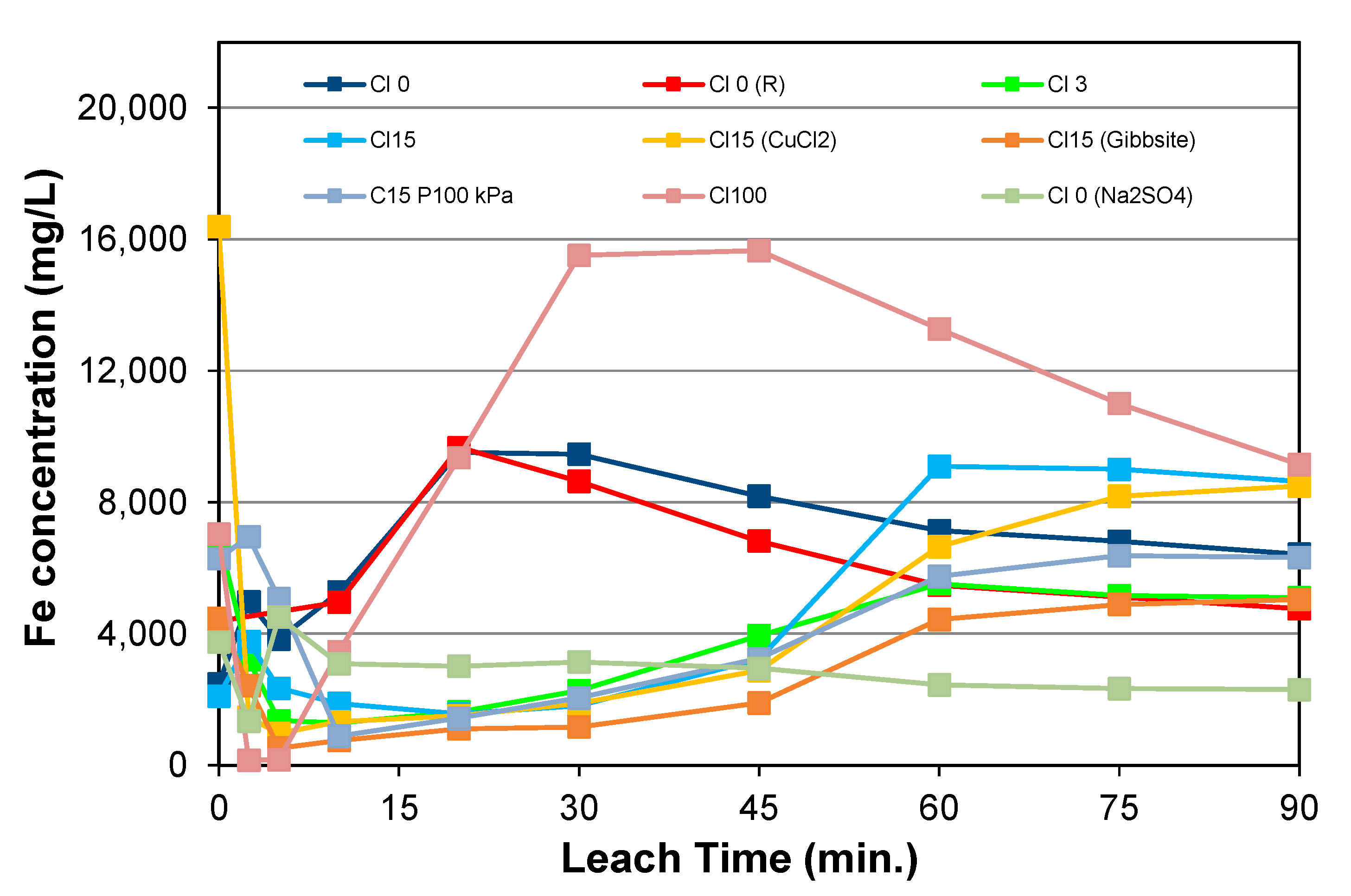
5.3. Behaviour of Dissolved Iron
- The iron concentrations during early leaching were somewhat higher when a lower oxygen partial pressure was employed, reflecting slower rates of sulphuric acid generation and Fe(II) oxidation.
- As the dissolution of added gibbsite was net acid-consuming, the final free acidity was lower, and this corresponds to a lower final iron concentration.
- The variation in final iron concentrations for the various datasets suggests that the equilibrium between the solids and liquor compositions was not reached after 90 min.
5.4. Behaviour of Other Elements
5.5. Additives Other Than Chloride Salts
5.5.1. Effect of Sodium Sulphate Addition
5.5.2. Effects of Aluminium Addition
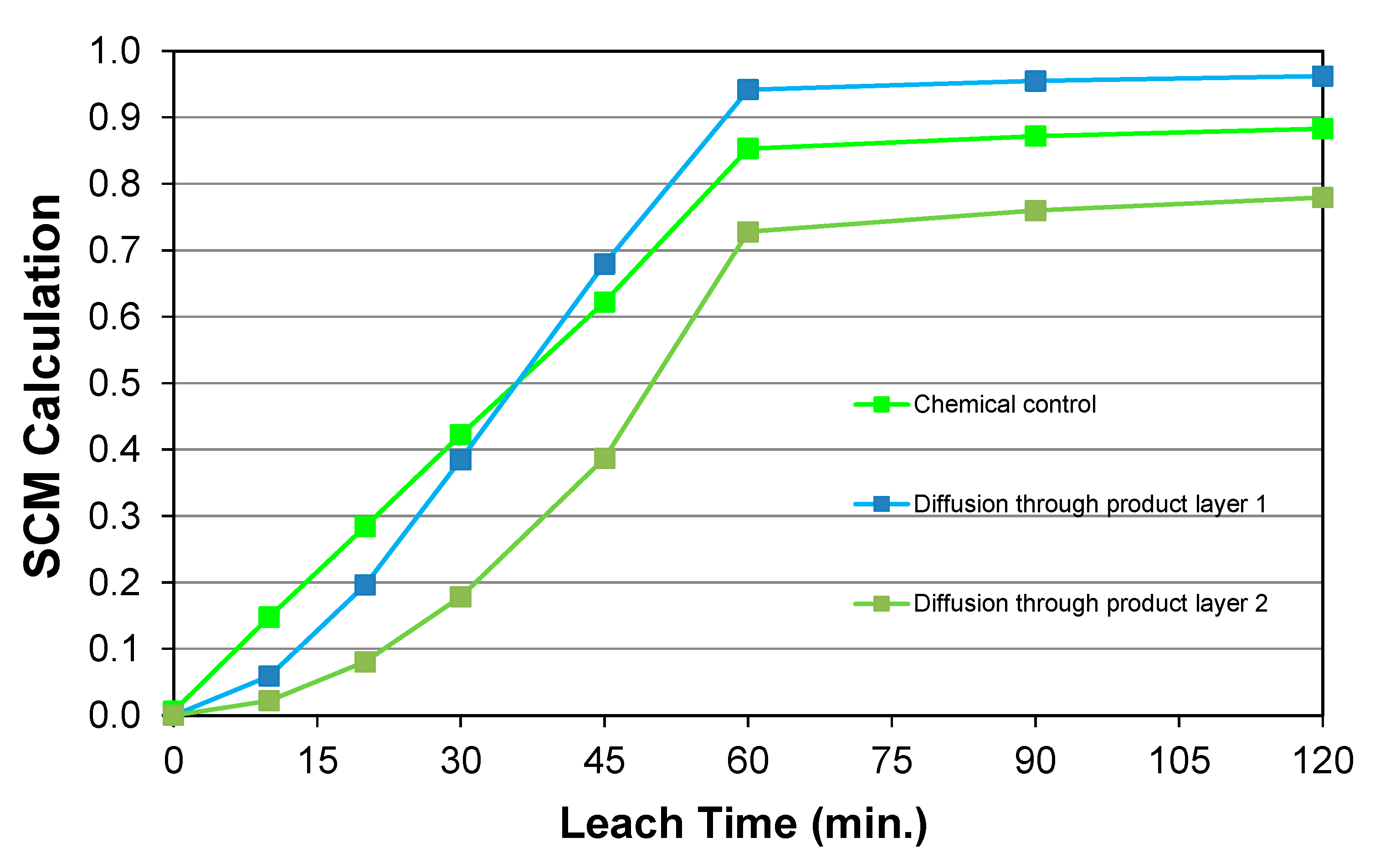
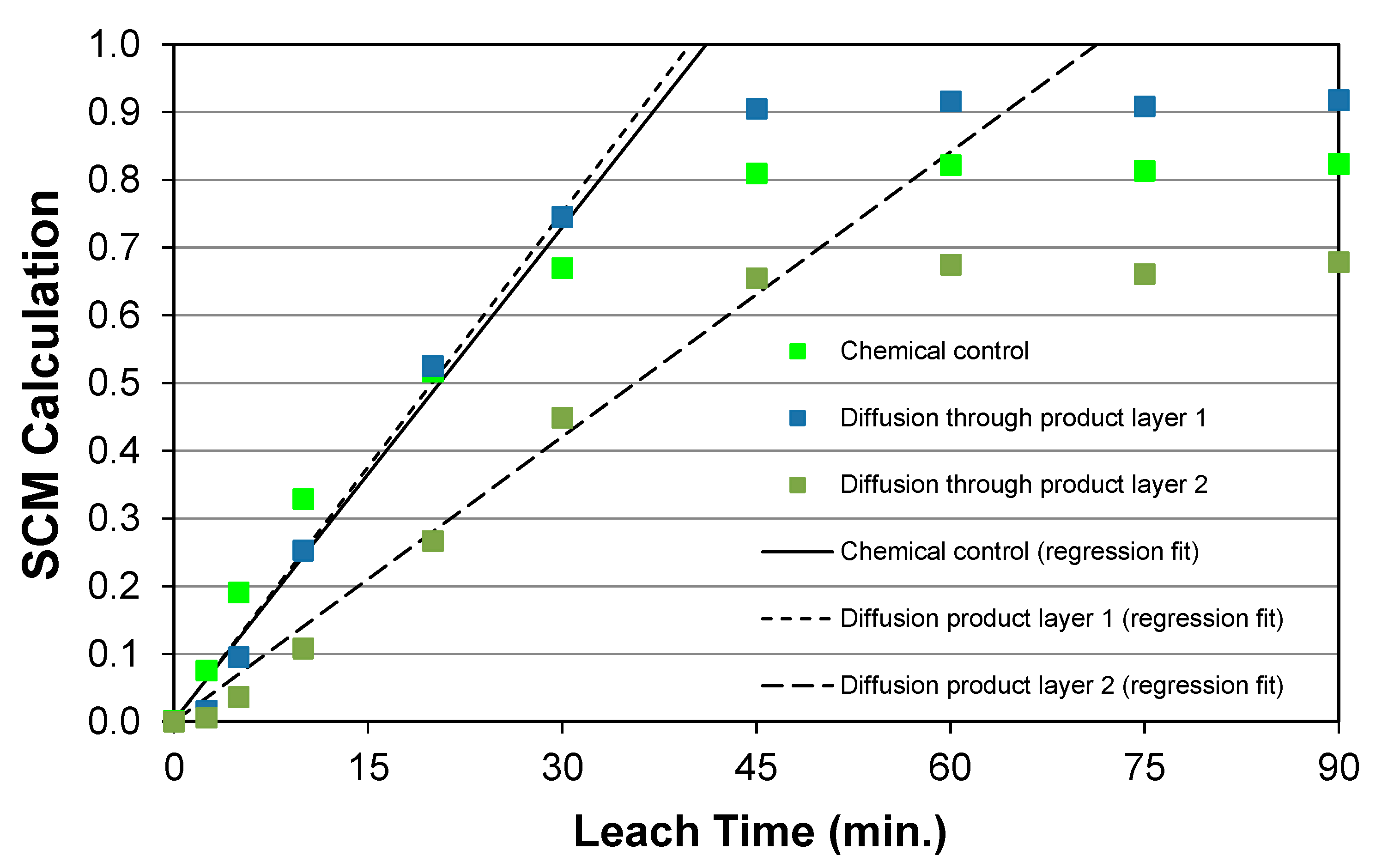
5.6. Reaction Kinetics
6. Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kruesi, P.R.; Allen, E.S.; Lake, J.L. Cymet Process—Hydrometallurgical conversion of base- metal sulphides to pure metals. CIM Bulletin 1973, 66, 81–87. [Google Scholar]
- Dalton, R.F.; Diaz, G.; Price, R.; Zunkel, A.D. The Cuprex metal extraction process: Recovering copper from sulfide ores. JOM 1991, 43, 51–56. [Google Scholar] [CrossRef]
- Moyes, J.; Sammut, D.; Houllis, F. The Intec Copper Process: Superior and Sustainable Metals Production. In Proceedings of the ALTA 2002 Copper 7 Conference, Perth, Australia, 23–24 May 2002; ALTA Metallurgical Services: Melbourne, Australia, 2002. 22p. [Google Scholar]
- Hourn, M.M.; Turner, D.W.; Holzberger, I.R. Atmospheric mineral leaching process. U.S. Patent 5,993,635A1, 30 November 1999. [Google Scholar]
- Lundström, M.; Aromaa, J.; Forsén, O.; Hyvärinen, O.; Barker, M.H. Leaching of chalcopyrite in cupric chloride solution. Hydrometallurgy 2005, 77, 89–95. [Google Scholar] [CrossRef]
- Nazari, G.; Dixon, D.G.; Dreisinger, D. Enhancing the kinetic of chalcopyrite leaching in the GalvanoxTM process. Hydrometallurgy 2011, 105, 251–258. [Google Scholar] [CrossRef]
- Chaiko, D.; Baczek, F.; Rocks, S.S.; Walters, T.; Klepper, R. The FLSmidth® rapid-oxidative leach (ROL) process Part I: Mechanochemical process for treating chalcopyrite. Proceeding of the COM2015, Conference of Metallurgists, Montreal, QC, Canada; Canadian Institute of Mining, Metallurgy and Petroleum: Montreal, QC, Canada, 2015. 11p. [Google Scholar]
- Chaiko, D.; Rocks, S.S.; Walters, T.; Asihene, S.; Eyzaguirre, C.; Klepper, R.; Baczek, F.; McMahon, G. The FLSmidth® rapid-oxidative leach (ROL) process Part II: A new chemical activation process for chalcopyrite. In COM2015, Conference of Metallurgists; Canadian Institute of Mining, Metallurgy and Petroleum: Montreal, QC, Canada, 2015; 15p. [Google Scholar]
- Mulligan, M.; Chaiko, D.; Baczek, F.; Rocks, S.; Eyzaguirre, C.; Dickinson, C.; Klepper, R. The FLSmidth® Rapid Oxidative Leach (ROL) process: A mechano-chemical approach and industrial applications for rapid metal sulphide dissolution. J. South. Afr. Inst. Min. Metall. 2017, 117, 741–747. [Google Scholar] [CrossRef] [Green Version]
- Oraby, E.A.; Eksteen, J.J. The selective leaching of copper from a gold–copper concentrate in glycine solutions. Hydrometallurgy 2014, 150, 14–19. [Google Scholar] [CrossRef]
- Evans, H.A.; Johnson, G.D. Activox® Technology for treatment of copper sulfide concentrates. In Proceedings of the International Conference of Randol, Copper Hydromet Roundtable ’99, Phoenix, AZ, USA, 10–13 October 1999; Randol International: Golden, CO, USA, 1999. 3p. [Google Scholar]
- Subramanian, K.N.; Jennings, P.H. Review of the hydrometallurgy of chalcopyrite concentrates. Can. Metall. Q. 1972, 11, 387–400. [Google Scholar] [CrossRef]
- Dutrizac, J.E. The leaching of sulphide minerals in chloride media. Hydrometallurgy 1992, 27, 1–45. [Google Scholar] [CrossRef]
- Venkatachalam, S. Treatment of chalcopyrite concentrates by hydrometallurgical techniques. Miner. Eng. 1991, 4, 1115–1126. [Google Scholar] [CrossRef]
- Prasad, S.; Pandey, B.D. Alternative processes for treatment of chalcopyrite. Miner. Eng. 1998, 11, 763–781. [Google Scholar] [CrossRef]
- Berezowsky, R.; Trytten, L. Commercialization of the acid pressure leaching of chalcopyrite. In Proceedings of the ALTA 2002 Copper 7 Conference, Perth, Australia, 23–24 May 2002; ALTA Metallurgical Services: Melbourne, Australia, 2002. 40p. [Google Scholar]
- Dreisinger, D. Copper leaching from primary sulfides: Options for biological and chemical extraction of copper. Hydrometallurgy 2006, 83, 10–20. [Google Scholar] [CrossRef]
- Dreisinger, D.B. Case study flowsheets: Copper–gold concentrate treatment. In Gold Ore Processing, 2nd ed.; Adams, M.D., Ed.; Elsevier B.V.: Amsterdam, The Netherlands, 2016; pp. 803–820. [Google Scholar]
- Baba, A.A.; Ayinla, K.I.; Adekola, F.A.; Ghosh, M.K.; Ayanda, O.S.; Bale, R.B.; Sheik, A.R.; Pradhan, S.R. A review on novel techniques for chalcopyrite ore processing. Int. J. Min. Eng. Miner. Process. 2012, 1, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Klauber, C. A critical review of the surface chemistry of acidic ferric sulphate dissolution of chalcopyrite with regards to hindered dissolution. Int. J. Miner. Process. 2008, 86, 1–17. [Google Scholar] [CrossRef]
- Córdoba, E.M.; Muñoz, J.A.; Blázquez, M.L.; González, F.; Ballester, A. Leaching of chalcopyrite with ferric ion. Part I: General aspects. Hydrometallurgy 2008, 93, 81–87. [Google Scholar] [CrossRef]
- Córdoba, E.M.; Muñoz, J.A.; Blázquez, M.L.; González, F.; Ballester, A. Leaching of chalcopyrite with ferric ion. Part II: Effect of redox potential. Hydrometallurgy 2008, 93, 88–96. [Google Scholar] [CrossRef]
- Li, Y.; Kawashima, N.; Li, J.; Chandra, A.P.; Gerson, A.R. A review of the structure, and fundamental mechanisms and kinetics of the leaching of chalcopyrite. Adv. Colloid Interface Sci. 2013, 197, 1–32. [Google Scholar] [CrossRef]
- Watling, H.R. Chalcopyrite hydrometallurgy at atmospheric pressure: 1. Review of acidic sulfate, sulfate–chloride and sulfate–nitrate process options. Hydrometallurgy 2013, 140, 163–180. [Google Scholar] [CrossRef]
- Watling, H.R. Chalcopyrite hydrometallurgy at atmospheric pressure: 2. Review of acidic chloride process options. Hydrometallurgy 2014, 146, 96–110. [Google Scholar] [CrossRef]
- Barton, I.F.; Hiskey, J.B. Chalcopyrite leaching in novel lixiviants. Hydrometallurgy 2022, 207, 105775. [Google Scholar] [CrossRef]
- Marsden, J.O.; Brewer, R.E.; Hazen, N. Copper concentrate leaching developments by Phelps Dodge Corporation. In Proceedings of the Hydrometallurgy 2003—Fifth International Conference in Honour of Professor Ian Ritchie, Volume 2: Electrometallurgy and Environmental Hydrometallurgy, Vancouver, BC, Canada, 24–27 August 2003; Young, C.A., Alfantazi, A.M., Anderson, C.G., Dreisinger, D.B., Harris, B., James, A., Eds.; TMS: Warrendale, PA, USA; pp. 1429–1446. [Google Scholar]
- Marsden, J.O.; Wilmot, J.C.; Mathern, D.R. Medium-temperature pressure leaching of copper concentrates—Part III. Commercial demonstration plant at Bagdad Arizona. Miner. Metall. Process. 2007, 24, 218–225. [Google Scholar]
- Green, C.; Robertson, J.; Marsden, J.O. Pressure leaching of copper concentrates at Morenci, Arizona—10 years of experience. Miner. Metall. Process. 2018, 35, 109–116. [Google Scholar] [CrossRef]
- Voigt, P.; Stieper, G.; Hourn, M. First commercialization of the Albion Process™ for copper. In Proceedings of the 58th Annual Conference of Metallurgists Hosting the 10th International Copper Conference 2019, COM 2019, Vancouver, BC, Canada, 18–21 August 2019; MetSoc: Montreal, QC, Canada, 2019. 13p. [Google Scholar]
- Jones, D.L.; Mayhew, K.; O’Connor, L. Nickel and cobalt recovery from a bulk copper-nickel concentrate using the CESL process. In Hydrometallurgy of Nickel and Cobalt, Proceedings of the 48th Annual Conference of Metallurgists of CIM, Sudbury, ON, Canada, 23–26 August 2009; Budac, J.J., Fraser, R., Mihaylov, I., Papangelakis, V.G., Robinson, D.J., Eds.; MetSoc: Montreal, QC, Canada, 2009; pp. 45–57. [Google Scholar]
- Wardell-Johnson, M.; Steiper, G.; Dreisinger, D. Engineering aspects of the Platsol™ process. In Proceedings of the ALTA 2009 Nickel/Cobalt 14 Conference, Perth, Australia, 25–27 May 2009; ALTA Metallurgical Services: Melbourne, Australia, 2009. 20p. [Google Scholar]
- Dreisinger, D.; Molnar, R.; Mezei, A.; Baxter, K.; Wardell-Johnson, M. The recovery of nickel and cobalt from the NorthMet deposit using the Platsol™ process with production of mixed or separate nickel and cobalt hydroxides. In Hydrometallurgy of Nickel and Cobalt, Proceedings of the 48th Annual Conference of Metallurgists of CIM, Sudbury, ON, Canada, 23–26 August 2009; Budac, J.J., Fraser, R., Mihaylov, I., Papangelakis, V.G., Robinson, D.J., Eds.; MetSoc: Montreal, QC, Canada, 2009; pp. 123–138. [Google Scholar]
- Karcz, A.P.; Damø, A.J.; Illerup, J.B.; Rocks, S.; Dam-Johansen, K.; Chaiko, D. Electron microscope investigations of activated chalcopyrite particles via the FLSmidth® ROL process. J. Mater. Sci. 2017, 52, 12044–12053. [Google Scholar] [CrossRef] [Green Version]
- McDonald, R.G. The effects of chloride on the high temperature pressure oxidation of chalcopyrite: Some insights from batch tests—Part 2. Leach residue mineralogy. Miner. Eng. 2023, in press. [Google Scholar]
- Weidenbach, M.; Dunn, G.; Teo, Y.Y. Removal of impurities from copper sulphide mineral concentrates. In Proceedings of the ALTA 2016 Copper 7 Forum, Perth, Australia, 21–28 May 2016; ALTA Metallurgical Services: Melbourne, Australia, 2016; pp. 335–351. [Google Scholar]
- Fomenko, I.V.; Pleshkov, M.A.; Shneerson, Y.M.; Ospanov, E.A.; Shakhalov, A.A.; Naboychenko, S.S. Low-grade copper concentrate purification and enrichment by complex pressure oxidation—Hydrothermal alteration technology. In Proceedings of the 58th Annual Conference of Metallurgists Hosting the 10th International Copper Conference 2019, COM 2019, Vancouver, BC, Canada, 18–21 August 2019; MetSoc: Montreal, QC, Canada, 2019. 13p. [Google Scholar]
- Chaudhari, A.; Webster, N.A.; Xia, F.; Frierdich, A.; Ram, R.; Etschmann, B.; Liu, W.; Wykes, J.; Brand, H.E.; Brugger, J. Anatomy of a complex mineral replacement reaction: Role of aqueous redox, mineral nucleation, and ion transport properties revealed by an in-situ study of the replacement of chalcopyrite by copper sulfides. Chem. Geol. 2021, 581, 120390. [Google Scholar] [CrossRef]
- McDonald, R.G.; Muir, D.M. Pressure oxidation leaching of chalcopyrite. Part I. Comparison of high and low temperature reaction kinetics and products. Hydrometallurgy 2007, 86, 191–205. [Google Scholar] [CrossRef]
- McDonald, R.G.; Muir, D.M. Pressure oxidation leaching of chalcopyrite. Part II. Comparison of medium temperature kinetics and products and effect of chloride ion. Hydrometallurgy 2007, 86, 206–220. [Google Scholar] [CrossRef]
- Hackl, R.P.; Dreisinger, D.B.; Peters, E.; King, J.A. Passivation of chalcopyrite during oxidative leaching in sulphate media. Hydrometallurgy 1995, 39, 25–48. [Google Scholar] [CrossRef]
- Petersen, A.E. Oxidation leaching. CA Patent 1263540A, 12 May 1989. [Google Scholar]
- Viramontes-Gamboa, G.; Rivera-Vasquez, B.F.; Dixon, D.G. The active-passive behaviour of chalcopyrite: Comparative study between electrochemical and leaching responses. J. Electrochem. Soc. 2007, 154, C299–C304. [Google Scholar] [CrossRef]
- Lundström, M.; Aromaa, J.; Forsén, O. The rate-controlling step of chalcopyrite dissolution in concentrated cupric chloride solution. In Copper 2010; Harre, J., Ed.; GDMB: Clausthal-Zellerfeld, Germany, 2010; Volume 5, pp. 1959–1972. [Google Scholar]
- Velásquez-Yévenes, L.; Nicol, M.; Miki, H. The dissolution of chalcopyrite in chloride solutions. Part 1. The effect of solution potential. Hydrometallurgy 2010, 103, 113–118. [Google Scholar] [CrossRef]
- Nicol, M.; Miki, H.; Zhang, S. The anodic behaviour of chalcopyrite in chloride solutions: Voltammetry. Hydrometallurgy 2017, 171, 198–205. [Google Scholar] [CrossRef] [Green Version]
- Debernardi, G.; Gentina, J.C.; Albistur, P.; Slanzi, G. Evaluation of processing options to avoid the passivation of chalcopyrite. Int. J. Miner. Process. 2013, 125, 1–4. [Google Scholar] [CrossRef]
- Yin, Q.; Kelsall, G.H.; Vaughan, D.J.; England, K.E.R. Atmospheric and electrochemical oxidation of the surface of chalcopyrite (CuFeS2). Geochim. Cosmochim. Acta 1995, 59, 1091–1100. [Google Scholar] [CrossRef]
- Liu, Q.; Chen, M.; Yang, Y. The effect of chloride ions on the electrochemical dissolution of chalcopyrite in sulfuric acid solutions. Electrochim. Acta 2017, 253, 257–267. [Google Scholar] [CrossRef]
- Ren, Z.; Chao, C.; Krishnamoorthy, P.; Asselin, E.; Dixon, D.G.; Mora, N. The overlooked mechanism of chalcopyrite passivation. Acta Mater. 2022, 236, 118111. [Google Scholar] [CrossRef]
- Winand, R. Chloride hydrometallurgy. Hydrometallurgy 1991, 27, 285–316. [Google Scholar] [CrossRef]
- Wang, S. Copper leaching from chalcopyrite concentrates. JOM 2005, 57, 48–51. [Google Scholar] [CrossRef]
- Lundström, M.; Aromaa, J.; Forsén, O.; Haavanlammi, L. Concentrated cupric chloride solutions: Possibilities offered in copper production. Acta Metall. Slov. 2007, 13, 447–459. [Google Scholar]
- Senanayake, G. A review of chloride assisted copper sulphide leaching by oxygenated sulphuric acid and mechanistic considerations. Hydrometallurgy 2009, 98, 21–32. [Google Scholar] [CrossRef] [Green Version]
- Torres, D.; Ayala, L.; Jeldres, R.I.; Cerecedo-Sáenz, E.; Salinas-Rodríguez, E.; Robles, P.; Toro, N. Leaching chalcopyrite with high MnO2 and chloride concentrations. Metals 2020, 10, 107. [Google Scholar] [CrossRef] [Green Version]
- Kowalczuk, P.B.; Manaig, D.O.; Drivenes, K.; Snook, B.; Aasly, K.; Kleiv, R.A. Galvanic leaching of seafloor massive sulphides using MnO2 in H2SO4-NaCl media. Minerals 2018, 8, 235. [Google Scholar] [CrossRef] [Green Version]
- Muñoz-Ribadeneira, F.J.; Gomberg, H.J. Leaching of chalcopyrite (CuFeS2) with sodium chloride sulfuric acid solutions. Nucl. Technol. 1971, 11, 367–371. [Google Scholar] [CrossRef]
- Subramanian, K.N.; Ferrajuolo, R. Oxygen pressure leaching of Fe-Ni-Cu sulphide concentrates at 110 °C—Effect of chloride addition. Hydrometallurgy 1976, 2, 117–125. [Google Scholar] [CrossRef]
- Sullivan, J.D. Chemical and physical features of copper leaching. Trans. Am. Inst. Min. Metall. Eng. 1933, 106, 515–546. [Google Scholar]
- Habashi, F.; Toor, T. Aqueous oxidation of chalcopyrite by hydrochloric acid. Metall. Trans. B Process Metall. 1979, 10B, 49–56. [Google Scholar] [CrossRef]
- Dutrizac, J.E. The dissolution of chalcopyrite in ferric sulphate and ferric chloride media. Metall. Trans. B Process Metall. 1981, 12B, 49–56. [Google Scholar]
- Majima, H.; Awakura, Y.; Hirato, T.; Tanaka, T. The leaching of chalcopyrite in ferric chloride and ferric sulfate solutions. Can. Metall. Q. 1985, 24, 283–291. [Google Scholar] [CrossRef]
- Hirato, T.; Kinoshita, M.; Awakura, Y.; Majima, H. The leaching of chalcopyrite with ferric chloride. Metall. Trans. B Process Metall. 1986, 17B, 19–28. [Google Scholar] [CrossRef]
- Lu, Z.Y.; Jeffrey, M.I.; Lawson, F. The effect of chloride ions on the dissolution of chalcopyrite in acidic solutions. Hydrometallurgy 2000, 56, 189–202. [Google Scholar] [CrossRef]
- Lu, Z.Y.; Jeffrey, M.I.; Lawson, F. An electrochemical study of the effect of chloride ion on the dissolution of chalcopyrite in acid solutions. Hydrometallurgy 2000, 56, 145–155. [Google Scholar] [CrossRef]
- Skrobian, M.; Havlik, T.; Ukasik, M. Effect of NaCl concentration and particle size on chalcopyrite leaching in cupric chloride solution. Hydrometallurgy 2005, 77, 109–114. [Google Scholar] [CrossRef]
- Palmer, B.R.; Nebo, C.O.; Rau, M.F.; Fursteneau, M.C. Rate phenomena involved in the dissolution of chalcopyrite in chloride-bearing lixiviants. Metall. Trans. B Process Metall. 1981, 12, 595–601. [Google Scholar] [CrossRef]
- Carneiro, M.F.C.; Leão, V.A. The role of sodium chloride on surface properties of chalcopyrite leached with ferric sulphate. Hydrometallurgy 2007, 87, 73–82. [Google Scholar] [CrossRef] [Green Version]
- Yoo, K.; Kim, S.; Lee, J.; Ito, M.; Tsunekawa, M.; Hiroyoshi, M. Effect of chloride ions on the leaching rate of chalcopyrite. Miner. Eng. 2010, 23, 471–477. [Google Scholar] [CrossRef]
- Whittington, B.I.; McDonald, R.G.; Johnson, J.A.; Muir, D.M. Pressure acid leaching of arid-region nickel laterite ore: Part I: Effect of water quality. Hydrometallurgy 2003, 70, 31–46. [Google Scholar] [CrossRef]
- Bruce, R.; Seaman, T. Reducing Freshwater Use in the Production of Metals. Teck Resources Limited. 2014. Available online: https://www.teck.com/media/CESL-Publication-Copper-reducing-fresh-water-use-in-the-production-of-metals.pdf (accessed on 4 July 2022).
- Toro, N.; Gálvez, E.; Robles, P.; Castillo, J.; Villca, G.; Salinas-Rodríguez, E. Use of alternative water resources in copper leaching processes in Chilean mining industry—A Review. Metals 2022, 12, 445. [Google Scholar] [CrossRef]
- Padilla, R.; Rodríguez, G.; Ruiz, M.C. Copper and arsenic dissolution from chalcopyrite-enargite concentrate by sulfidation and pressure leaching in H2SO4-O2. Hydrometallurgy 2010, 100, 152–156. [Google Scholar] [CrossRef]
- Ruiz, M.C.; Montes, K.; Padilla, R. Pressure leaching of a chalcopyrite concentrate. In Copper 2010; Harre, J., Ed.; GDMB: Clausthal-Zellerfeld, Germany, 2010; Volume 5, pp. 2017–2027. [Google Scholar]
- Peters, E. Direct leaching of sulfides: Chemistry and applications. Metall. Trans. B Process Metall. 1976, 7, 505–517. [Google Scholar] [CrossRef]
- Peters, E. The electrochemistry of sulphide minerals. In Trends in Electrochemistry; Bockris, J.O.M., Rand, D.A.F., Welch, R.J., Eds.; Plenum Press: New York, NY, USA, 1977; pp. 267–290. [Google Scholar]
- Huang, H.H. The Eh-pH diagram and its advances. Metals 2016, 6, 23. [Google Scholar] [CrossRef] [Green Version]
- Hawker, W.; Byrne, K.; Vaughan, J. Upgrading copper sulphide concentrates through high temperature metathesis reactions. In Proceedings of the 58th Annual Conference of Metallurgists Hosting the 10th International Copper Conference 2019, COM 2019, Vancouver, BC, Canada, 18–21 August 2019; MetSoc: Montreal, QC, Canada, 2019. 10p. [Google Scholar]
- Velásquez-Yévenes, L.; Miki, H.; Nicol, M. The dissolution of chalcopyrite in chloride solutions: Part 2: Effect of various parameters on the rate. Hydrometallurgy 2010, 103, 80–85. [Google Scholar] [CrossRef]
- Jafari, M.; Karimi, G.; Ahmadi, R. Improvement of chalcopyrite atmospheric leaching using controlled slurry potential and additive treatments. Physicochem. Probl. Miner. Process. 2017, 53, 1228–1240. [Google Scholar]
- Koleini, S.J.; Aghazadeh, V.; Sandström, Å. Acidic sulphate leaching of chalcopyrite concentrates in presence of pyrite. Miner. Eng. 2011, 24, 381–386. [Google Scholar] [CrossRef]
- Hiroyoshi, N.; Kitagawa, H.; Tsunekawa, M. Effect of solution composition on the optimum redox potential for chalcopyrite leaching in sulphuric acid solutions. Hydrometallurgy 2008, 91, 144–149. [Google Scholar] [CrossRef]
- Miki, H.; Nicol, M.; Velásquez-Yévenes, L. The kinetics of dissolution of synthetic covellite, chalcocite and digenite in dilute chloride solutions at ambient temperatures. Hydrometallurgy 2011, 105, 321–327. [Google Scholar] [CrossRef] [Green Version]
- Cheng, C.Y.; Lawson, F. The kinetics of leaching covellite in acidic oxygenated sulphate-chloride solutions. Hydrometallurgy 1991, 27, 269–284. [Google Scholar] [CrossRef]
- Kametani, H.; Aoki, A. Effect of suspension potential on the oxidation rate of copper concentrate in a sulfuric acid solution. Metall. Trans. B Process Metall. 1985, 16, 695–705. [Google Scholar] [CrossRef]
- Harmer, S.L.; Thomas, J.E.; Fornasiero, D.; Gerson, A.R. The evolution of surface layers formed during chalcopyrite leaching. Geochim. Cosmochim. Acta 2006, 70, 4392–4402. [Google Scholar] [CrossRef]
- Nava, D.; González, I. Electrochemical characterization of chemical species formed during the electrochemical treatment of chalcopyrite in sulphuric acid. Electrochim. Acta 2006, 51, 5295–5303. [Google Scholar] [CrossRef]
- Ghahremaninezhad, A.; Asselin, E.; Dixon, D.G. Electrochemical evaluation of the surface of chalcopyrite during dissolution in sulfuric acid solution. Electrochim. Acta 2010, 55, 5041–5056. [Google Scholar] [CrossRef]
- Ruiz, M.C.; Montes, K.S.; Padilla, R. Chalcopyrite leaching in sulfate–chloride media at ambient pressure. Hydrometallurgy 2011, 109, 37–42. [Google Scholar] [CrossRef]
- Qiu, T.-S.; Nie, G.-H.; Wang, J.-F.; Ciu, L.-F. Kinetic process of oxidative leaching of chalcopyrite under low oxygen pressure and low temperature. Trans. Nonferrous Metall. Soc. China 2007, 17, 418–422. [Google Scholar] [CrossRef]
- Hirato, T.; Majima, H.; Awakura, Y. The leaching of chalcopyrite with cupric chloride. Metall. Trans. B Process Metall. 1987, 18, 31–39. [Google Scholar] [CrossRef]
- Parkman, R.H.; Charnock, J.M.; Bryan, N.D.; Livens, F.R.; Vaughan, D.J. Reactions of copper and cadmium ions in aqueous solution with goethite, lepidocrocite, mackinawite, and pyrite. Am. Mineral. 1999, 84, 407–419. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, W.; Cai, Y.; Qu, Y.; Pan, Y.; Zhang, W.; Zhao, K. Experimental investigation of the reactions between pyrite and aqueous Cu(I) chloride solution at 100–250 °C. Geochim. Cosmochim. Acta 2021, 298, 1–20. [Google Scholar] [CrossRef]
- Ruiz, M.C.; Honores, S.; Padilla, R. Leaching kinetics of digenite concentrate in oxygenated chloride media at ambient pressure. Metall. Trans. B Process Metall. 1998, 29, 961–969. [Google Scholar] [CrossRef]
- Peters, E. Oxygen utilization in hydrometallurgy: Fundamentals and basic principles. In Proceedings of the International Symposium, Impact of Oxygen on the Productivity of Non-Ferrous Metallurgical Processes, CIM, Winnipeg, MB, Canada, 23–26 August 1987; Kachaniwsky, G., Newman, C., Eds.; Pergamon Press: New York, NY, USA, 1987; pp. 151–164. [Google Scholar]
- Heidel, C.; Tichomirowa, M. The role of dissolved molecular oxygen in abiotic pyrite oxidation under acid pH conditions–experiments with 18O-enriched molecular oxygen. Appl. Geochem. 2010, 25, 1664–1675. [Google Scholar] [CrossRef]
- Tichomirowa, M.; Junghans, M. Oxygen isotope evidence for sorption of molecular oxygen to pyrite surface sites and incorporation into sulfate in oxidation experiments. Appl. Geochem. 2009, 24, 2072–2092. [Google Scholar] [CrossRef]
- Sit, P.H.L.; Cohen, M.H.; Selloni, A. Interaction of oxygen and water with the (100) surface of pyrite: Mechanism of sulfur oxidation. J. Phys. Chem. Lett. 2012, 3, 2409–2414. [Google Scholar] [CrossRef]
- Al-Harahsheh, M.; Kingman, S.; Al-Harahsheh, A. Ferric chloride leaching of chalcopyrite: Synergistic effect of CuCl2. Hydrometallurgy 2008, 91, 89–97. [Google Scholar] [CrossRef]
- Ferron, C.J.; Fleming, C.; O’Kane, P.T.; Dreisinger, D.B. Pilot plant demonstration of the Platsol process for the treatment of the NorthMet copper-nickel-PGM deposit. Min. Eng. 2002, 54, 33–39. [Google Scholar]
- Dreisinger, D. Case study flowsheets: Copper–gold concentrate treatment. In Developments in Mineral Processing: Advances in Gold Processing, 1st ed.; Adams, M.D., Wills, B.A., Eds.; Elsevier B.V.: Amsterdam, The Netherlands, 2005; pp. 825–848. [Google Scholar]
- Harris, G.B.; White, C.W.; Demopoulos, G.P.; Ballantyne, B. Recovery of copper from a massive polymetallic sulphide by high concentration chloride leaching. Can. Metall. Q. 2008, 47, 347–356. [Google Scholar] [CrossRef]
- King, J.A.; Dreisinger, D.B.; Knight, D.A. The total pressure oxidation of copper concentrates. In The Paul E. Queneau International Symposium Extractive Metallurgy of Copper, Nickel and Cobalt, Volume I: Fundamental Aspects; Reddy, R.G., Weizenbach, R.N., Eds.; TMS: Warrendale, PA, USA, 1993; pp. 735–756. [Google Scholar]
- Dreisinger, D.; Murray, W.; Hunter, D.; Baxter, K.; Ferron, J.; Fleming, C. The application of the Platsol™ Process to copper-nickel-cobalt-PGE/PGM concentrates from Polymet Mining’s NorthMet Deposit. In Proceedings of the ALTA 2005 Nickel/Cobalt 10 Conference, Perth, Australia, 16–18 May 2005; ALTA Metallurgical Services: Melbourne, Australia, 2005. 16p. [Google Scholar]
- Defreyne, J.; Brace, T.; Miller, C.; Omena, A.; Matos, M.; Cobral, T. Commissioning UHC: A Vale Copper Refinery based on CESL technology. In Hydrometallurgy 2008, Proceedings of the Sixth International Symposium; Young, C.A., Taylor, P.R., Anderson, C.G., Eds.; SME: Littleton, CO, USA, 2008; pp. 357–366. [Google Scholar]
- Antonijević, M.; Bogdanović, G.D. Investigation of the leaching of chalcopyritic ore in acidic solutions. Hydrometallurgy 2004, 73, 245–256. [Google Scholar] [CrossRef]
- Abraitis, P.K.; Pattrick, R.A.D.; Kelsall, G.H.; Vaughan, D.J. Acid leaching and dissolution of major sulphide ore minerals: Processes and galvanic effects in complex systems. Mineral. Mag. 2004, 68, 343–351. [Google Scholar] [CrossRef]
- Fu, K.B.; Lin, H.; Mo, X.L.; Wang, H.; Wen, H.W.; Wen, Z.L. Comparative study on the passivation layers of copper sulphide minerals during bioleaching. Int. J. Miner. Metall. Mater. 2012, 19, 886–892. [Google Scholar] [CrossRef]
- Nicol, M.; Basson, P. The anodic behaviour of covellite in chloride solutions. Hydrometallurgy 2017, 172, 60–68. [Google Scholar] [CrossRef]
- Recalde Chiluiza, E.L.; Navarro Donoso, P. Chalcopyrite leaching in acidic chloride solution without sulphates. J. Mex. Chem. Soc. 2016, 60, 238–246. [Google Scholar] [CrossRef]
- Papangelakis, V.G.; Demopoulos, G.P. Acid pressure oxidation of pyrite: Reaction kinetics. Hydrometallurgy 1991, 26, 309–325. [Google Scholar] [CrossRef]
- Yu, P.H.; Hansen, C.K.; Wadsworth, M.E. A kinetic study of the leaching of chalcopyrite at elevated temperatures. Metall. Trans. 1973, 4, 2137–2144. [Google Scholar] [CrossRef]
- Narita, E.; Lawson, F.; Han, K.N. Solubility of oxygen in aqueous electrolyte solutions. Hydrometallurgy 1983, 10, 21–37. [Google Scholar] [CrossRef]
- Tromans, D. Oxygen solubility modeling in inorganic solutions: Concentration, temperature and pressure effects. Hydrometallurgy 1998, 50, 279–296. [Google Scholar] [CrossRef]
- Tromans, D. Modeling oxygen solubility in water and electrolyte solutions. Ind. Eng. Chem. Res. 2000, 39, 805–812. [Google Scholar] [CrossRef]
- Corriou, J.-P.; Kikindai, T. The aqueous oxidation of elemental sulfur and different chemical properties of the allotropic forms Sλ and Sμ. J. Inorg. Nucl. Chem. 1981, 43, 9–15. [Google Scholar] [CrossRef]
- Collins, M.J.; Berezowsky, R.M.G.S.; Vardill, W.D.; Ketcham, V.J.; Stojsic, A. The Lihir Gold project: Pressure oxidation process development. In Hydrometallurgy Fundamentals, Technology and Innovations; Hiskey, J.B., Warren, G.W., Eds.; TMS: Littleton, CO, USA, 1993; Chapter 38; pp. 611–628. [Google Scholar]
- Bartlett, P.D.; Davis, R.E. Reactions of elemental sulfur. II. The reaction of alkali cyanide with sulfur, and some single-sulfur transfer reactions. J. Am. Chem. Soc. 1958, 80, 2513–2516. [Google Scholar] [CrossRef]
- Habashi, F.; Bauer, E.L. Aqueous oxidation of elemental sulfur. Ind. Eng. Chem. Fundam. 1966, 5, 469–471. [Google Scholar] [CrossRef]
- Ghali, E.; Danapani, B.; Lewenstam, A. Electrodissolution of synthetic covellite in hydrochloric acid. J. Appl. Electrochem. 1982, 12, 369–376. [Google Scholar] [CrossRef]
- Robinson, B.W. Sulphur isotope equilibrium during sulphur hydrolysis at high temperatures. Earth Planet. Sci. Lett. 1973, 18, 443–450. [Google Scholar] [CrossRef]
- Smith, J.W. Isotopic fractionations accompanying sulfur hydrolysis. Geochem. J. 2000, 34, 95–99. [Google Scholar] [CrossRef] [Green Version]
- Rimstidt, J.D.; Vaughan, D.J. Pyrite oxidation: A state-of-the-art assessment of the reaction mechanism. Geochim. Cosmochim. Acta 2003, 67, 873–880. [Google Scholar] [CrossRef]
- Reedy, B.J.; Beattie, J.K.; Lowson, R.T. A vibrational spectroscopic 18O tracer study of pyrite oxidation. Geochim. Cosmochim. Acta 1991, 55, 1609–1614. [Google Scholar] [CrossRef]
- Usher, C.R.; Cleveland, C.A., Jr.; Strongin, D.R.; Schoonen, M.A. Origin of oxygen in sulfate during pyrite oxidation with water and dissolved oxygen: An in situ horizontal attenuated total reflectance spectroscopy isotope study. Environ. Sci. Technol. 2004, 38, 5404–5406. [Google Scholar] [CrossRef]
- Gil-Lozano, C.; Davila, A.F.; Losa-Adams, E.; Fairén, A.G.; Gago-Duport, L. Quantifying Fenton reaction pathways driven by self-generated H2O2 on pyrite surfaces. Sci. Rep. 2017, 7, 43703. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Chen, J.; Chen, Y.; Zhao, C.; Zhang, Y.; Ke, B. Interactions of oxygen and water molecules with pyrite surface: A new insight. Langmuir 2018, 34, 1941–1952. [Google Scholar] [CrossRef]
- Li, Y.; Chandra, A.P.; Gerson, A.R. Scanning photoelectron microscopy studies of freshly fractured chalcopyrite exposed to O2 and H2O. Geochim. Cosmochim. Acta 2014, 133, 372–386. [Google Scholar] [CrossRef]
- Wei, Z.; Li, Y.; Gao, H.; Zhu, Y.; Qian, G.; Yao, J. New insights into the surface relaxation and oxidation of chalcopyrite exposed to O2 and H2O: A first-principles DFT study. Appl. Surf. Sci. 2019, 492, 89–98. [Google Scholar] [CrossRef]
- Nicol, M.; Miki, H.; Velásquez-Yévenes, L. The dissolution of chalcopyrite in chloride solutions: Part 3. Mechanisms. Hydrometallurgy 2010, 103, 86–95. [Google Scholar] [CrossRef]
- Corriou, J.-P.; Gely, R.; Viers, P. Thermodynamic and kinetic study of the pressure leaching of zinc sulfide in aqueous sulfuric acid. Hydrometallurgy 1988, 21, 85–102. [Google Scholar] [CrossRef]
- Zhang, Q.; Dalla Lana, I.G.; Chuang, K.T.; Wang, H. Reactions between hydrogen sulfide and sulfuric acid: A novel process for sulfur removal and recovery. Ind. Eng. Chem. Res. 2000, 39, 2505–2509. [Google Scholar] [CrossRef]
- Reid, M.; Papangelakis, V.G. New data on hematite solubility in sulphuric acid solution from 130 to 270 °C. In Iron Control Technologies; Durizac, J.E., Riveros, P.A., Eds.; MetSoc: Montreal, QC, Canada, 2006; pp. 673–686. [Google Scholar]
- Liu, H.; Papangelakis, V.G.; Alam, M.S.; Singh, G. Solubility of hematite in H2SO4 solutions at 230–270 °C. Can. Metall. Q. 2003, 42, 199–208. [Google Scholar] [CrossRef]
- Meyer, B. Sulfur, Energy and Environment. Chapter 3 Properties; Elsevier Scientific Publishing Company: Amsterdam, The Netherlands, 1977; pp. 38–79. [Google Scholar]
- Tozawa, K.; Sasaki, K. Effect of co-existing sulphates on precipitation of ferric oxide from ferric sulphate solutions at elevated temperatures. In Iron Control in Hydrometallurgy; Dutrizac, J.E., Monhemius, A.J., Eds.; Ellis Horwood: Chichester, UK, 1986; pp. 454–476. [Google Scholar]
- Munoz, P.B.; Miller, J.B.; Wadsworth, M.E. Reaction mechanism for the acid ferric sulphate leaching of chalcopyrite. Metall. Trans. B Process Metall. 1979, 10B, 149–158. [Google Scholar] [CrossRef]
- Li, Y.; Wei, Z.; Qian, G.; Li, J.; Gerson, A.R. Kinetics and mechanisms of chalcopyrite dissolution at controlled redox potential of 750 mV in sulfuric acid solution. Minerals 2016, 6, 83. [Google Scholar] [CrossRef]
- Yue, G.; Asselin, E. Kinetics of ferric ion reduction on chalcopyrite and its influence on leaching up to 150 C. Electrochim. Acta 2014, 146, 307–321. [Google Scholar] [CrossRef]
- Hidalgo, T.; Kuhar, L.; Beinlich, A.; Putnis, A. Kinetic study of chalcopyrite dissolution with iron (III) chloride in methanesulfonic acid. Miner. Eng. 2018, 125, 66–74. [Google Scholar] [CrossRef]
- Baba, A.A.; Ayinla, K.I.; Adekola, F.A.; Bale, R.B.; Ghosh, M.K.; Alabi, A.G.; Sheik, A.R.; Folorunso, I.O. Hydrometallurgical application for treating a Nigerian chalcopyrite ore in chloride medium: Part I. Dissolution kinetics assessment. Int. J. Miner. Metall. Mater. 2013, 20, 1021–1028. [Google Scholar] [CrossRef]
- Baba, A.A.; Ayinla, K.I.; Bale, R.B.; Adekola, F.A. Quantitative leaching of a Nigerian chalcopyrite ore by nitric acid. Bayero J. Pure Appl. Sci. 2014, 7, 115–121. [Google Scholar]
- Sokić, M.D.; Marković, B.; Živković, D. Kinetics of chalcopyrite leaching by sodium nitrate in sulphuric acid. Hydrometallurgy 2009, 95, 273–279. [Google Scholar] [CrossRef]
- Bobeck, G.; Su, H. The kinetics of dissolution of sphalerite in ferric chloride solution. Metall. Trans. B 1985, 16, 413–424. [Google Scholar] [CrossRef]
- Qian, G.; Li, J.; Li, Y.; Gerson, A.R. Probing the effect of aqueous impurities on the leaching of chalcopyrite under controlled conditions. Hydrometallurgy 2014, 149, 195–209. [Google Scholar] [CrossRef]
- Fuentes, G.; Viñals, J. Hydrothermal purification and enrichment of Chilean copper concentrates. The behavior of bornite, covellite, pyrite and chalcopyrite. In Proceedings of the Pressure Hydrometallurgy 2012, 42nd Annual Hydrometallurgy Meeting, Niagara Falls, ON, Canada, 30 September–3 October 2012; Collins, M.J., Filippou, D., Harlamovs, J., Peek, E., Eds.; MetSoc on behalf of the Canadian Institute of Mining, Metallurgy and Petroleum: Montreal, QC, Canada, 2012; pp. 257–267. [Google Scholar]
- Petrović, S.J.; Bogdanović, G.D.; Antonijević, M.M. Leaching of chalcopyrite with hydrogen peroxide in hydrochloric acid solution. Trans. Nonferrous. Met. Soc. China 2018, 28, 1444–1455. [Google Scholar] [CrossRef]
- He, G.; Zhao, Z.; Wang, X.; Li, J.; Chen, X.; He, L.; Liu, X. Leaching kinetics of scheelite in hydrochloric acid solution containing hydrogen peroxide as complexing agent. Hydrometallurgy 2014, 144, 140–147. [Google Scholar] [CrossRef]
- Prosser, A.P. Review of uncertainty in the collection and interpretation of leaching data. Hydrometallurgy 1996, 41, 119–153. [Google Scholar] [CrossRef]





| Pulp Density (% w/w) | Acid (g/L) | Chloride (g/L) | Additive(s) | T( °C) | P(O2) (kPa) |
|---|---|---|---|---|---|
| 10 | Nil | Nil | None | 220 | 700 |
| 10 * | Nil | Nil | None | 220 | 700 |
| 10 | Nil | 15 | NaCl | 220 | 700 |
| 20 | Nil | Nil | None | 220 | 700 |
| 20 | 30 | Nil | None | 220 | 700 |
| 20 | 100 | Nil | None | 220 | 700 |
| 20 | Nil | 3 | NaCl | 220 | 700 |
| 20 | Nil | 15 | NaCl | 220 | 700 |
| 20 | Nil | 15 | NaCl, hematite seed | 220 | 700 |
| 20 | 30 | 15 | NaCl | 220 | 700 |
| 20 | Nil | 15 | NaCl | 230 | 700 |
| 20 | Nil | 15 | NaCl | 245 | 700 |
| 20 | Nil | 15 | NaCl | 220 | 100 |
| 20 | Nil | 15 | CuCl2.2H2O | 220 | 700 |
| 20 | Nil | 15 | NaCl, Al2(SO4)3.18H2O # | 220 | 700 |
| 20 | Nil | 15 | NaCl, Gibbsite # | 220 | 700 |
| 20 | Nil | 100 | NaCl | 220 | 700 |
| 20 | 100 | 100 | NaCl | 220 | 700 |
| 20 | Nil | Nil | Na2SO4 ** | 220 | 700 |
| Elemental Content (% w/w) | |||||||
|---|---|---|---|---|---|---|---|
| Cu | Fe | Al | Mg | Ca | Si | Na | S |
| 24.5 | 26.9 | 0.240 | 1.14 | 0.015 | 4.35 | 0.008 | 31.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
McDonald, R.G. The Effects of Chloride on the High-Temperature Pressure Oxidation of Chalcopyrite: Some Insights from Batch Tests—Part 1: Leach Chemistry. Minerals 2023, 13, 1065. https://doi.org/10.3390/min13081065
McDonald RG. The Effects of Chloride on the High-Temperature Pressure Oxidation of Chalcopyrite: Some Insights from Batch Tests—Part 1: Leach Chemistry. Minerals. 2023; 13(8):1065. https://doi.org/10.3390/min13081065
Chicago/Turabian StyleMcDonald, Robbie G. 2023. "The Effects of Chloride on the High-Temperature Pressure Oxidation of Chalcopyrite: Some Insights from Batch Tests—Part 1: Leach Chemistry" Minerals 13, no. 8: 1065. https://doi.org/10.3390/min13081065






