The Effects of Chloride on the High Temperature Pressure Oxidation of Chalcopyrite: Some Insights from Batch Tests—Part 2: Leach Residue Mineralogy
Abstract
:1. Introduction
2. Experimental
3. Ore Characterisation
4. Results
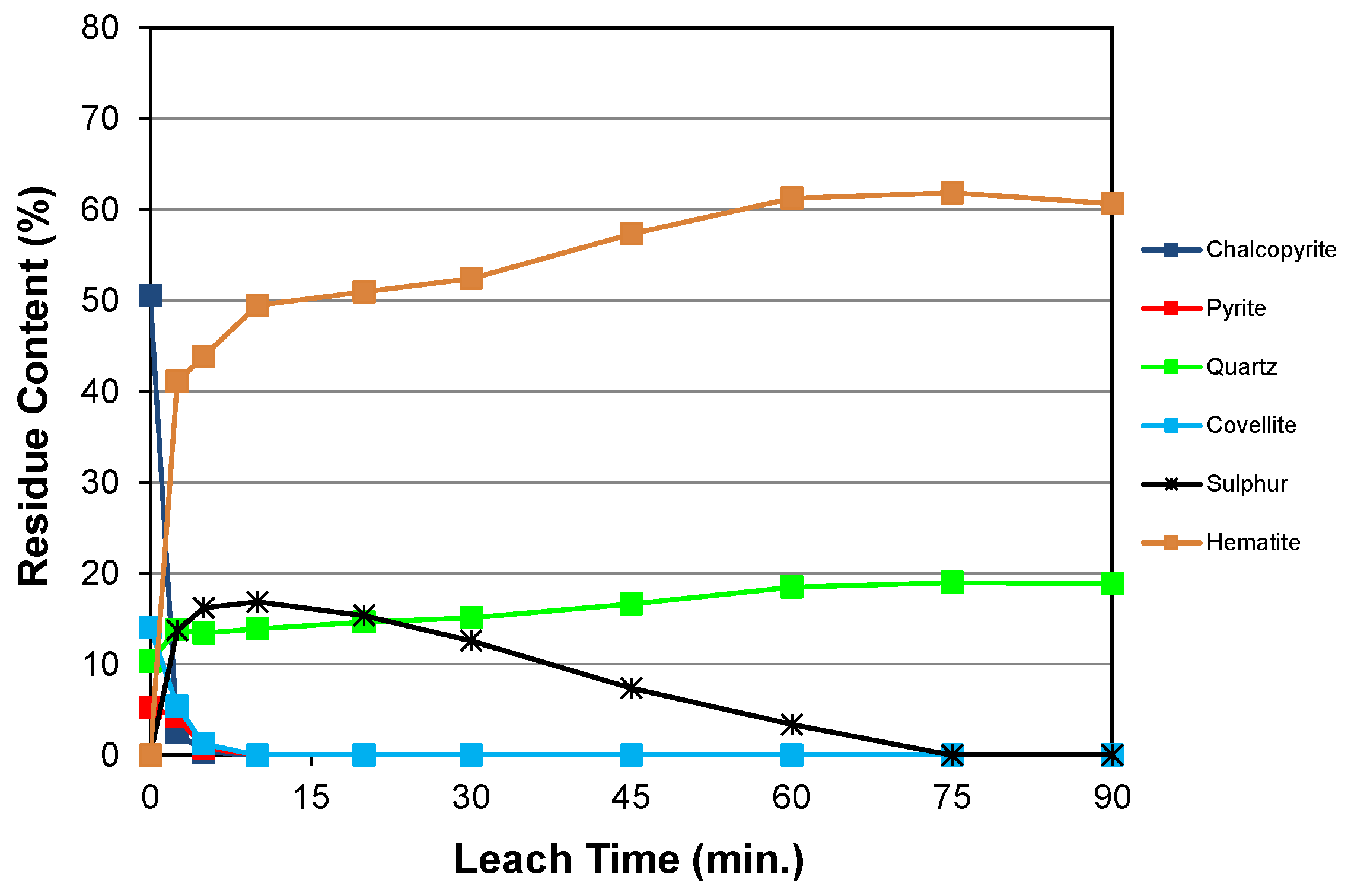
4.1. Summary of Dissolution Behaviour
4.2. Overview of Residue Mineralogy
4.3. Copper Metathesis
4.4. Reaction of Chalcopyrite with Acid
4.5. Impacts of Chloride and Other Additives
4.5.1. Effects of Pulp Density and Added Chloride
4.5.2. Effects of Added Chloride and Oxygen Partial Pressure
4.5.3. Effect of Added Sulphuric Acid
4.5.4. Effects of Temperature
4.5.5. Co-Presence of Pyrite
4.5.6. Hydrohematite Formation
4.5.7. Effects of Additional Aluminium and Hematite Seeding
4.5.8. Formation of Basic Copper Salts
4.5.9. Overview of Mineral Reactions
4.5.10. Chemical Thermodynamics
5. Implications for Industrial Application
6. Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- McDonald, R.G. The effects of chloride on the high temperature pressure oxidation of chalcopyrite: Some insights from batch tests—Part 1. Leach chemistry. Minerals 2023, 13, 1065. [Google Scholar] [CrossRef]
- Marsden, J.O.; Brewer, R.E. Hydrometallurgical processing of copper concentrates by Phelps Dodge at the Bagdad Mine in Arizona. In ALTA 2003 Copper 8 Conference; ALTA Metallurgical Services: Melbourne, Australia, 2003; 17p. [Google Scholar]
- Kuhn, M.C.; Arbiter, N.; Kling, H. Anacondas Arbiter process for copper. Can. Min. Metall. Bull. 1974, 67, 62–71. [Google Scholar]
- Shin, D.; Ahn, J.; Lee, J. Kinetic study of copper leaching from chalcopyrite concentrate in alkaline glycine solution. Hydrometallurgy 2019, 183, 71–78. [Google Scholar] [CrossRef]
- Khezri, M.; Rezai, B.; Abdollahzadeh, A.A.; Wilson, B.P.; Molaeinasab, M.; Lundström, M. Investigation into the effect of mechanical activation on the leaching of chalcopyrite in a glycine medium. Hydrometallurgy 2020, 203, 105492. [Google Scholar] [CrossRef]
- McDonald, R.G.; Muir, D.M. Pressure oxidation leaching of chalcopyrite. Part I. Comparison of high and low temperature reaction kinetics and products. Hydrometallurgy 2007, 86, 191–205. [Google Scholar] [CrossRef]
- Fleuriault, C.M.; Anderson, C.G.; Shuey, S. Iron phase control during pressure oxidation at elevated temperature. Miner. Eng. 2016, 98, 161–168. [Google Scholar] [CrossRef]
- McDonald, R.G.; Muir, D.M. Pressure oxidation leaching of chalcopyrite Part, I.I. Comparison of medium temperature kinetics and products and effect of chloride ion. Hydrometallurgy 2007, 86, 206–220. [Google Scholar] [CrossRef]
- Sahu, S.K.; Asselin, E. Characterization of residue generated during medium temperature leaching of chalcopyrite concentrate under CESL conditions. Hydrometallurgy 2011, 110, 107–114. [Google Scholar] [CrossRef]
- Javed, T.; Abdul, B.; Ryan, D.; Raudsepp, M.; Asselin, E. Amorphous iron phases in medium temperature leach residues and associated metal loss. Int. J. Miner. Process. 2016, 148, 65–71. [Google Scholar] [CrossRef]
- Javed, T.; Xie, M.; Asselin, E. Factors affecting hematite precipitation and characterization of the product from simulated sulphate-chloride solutions at 150 °C. Hydrometallurgy 2018, 179, 8–19. [Google Scholar] [CrossRef]
- Drits, V.A.; Sakharov, B.A.; Manceau, A. Structure of feroxyhite as determined by simulation of X-ray diffraction curves. Clay Miner. 1993, 28, 209–222. [Google Scholar] [CrossRef]
- Leach, D.L.; Braun, R.L. Leaching of Primary Sulfide ores in Sulfuric Acid Solutions at Elevated Temperatures and Pressures; No. UCRL-75899; CONF-750204-1; Lawrence Livermore Laboratory, California University: Livermore, CA, USA, 1974. [Google Scholar]
- Chaiko, D.; Rocks, S.S.; Walters, T.; Asihene, S.; Eyzaguirre, C.; Klepper, R.; Baczek, F.; McMahon, G. The FLSmidth® rapid-oxidative leach (ROL) process Part II: A new chemical activation process for chalcopyrite. In COM 2015, Conference of Metallurgists; Canadian Institute of Mining, Metallurgy and Petroleum: Montreal, QC, Canada, 2015; 15p. [Google Scholar]
- Jiang, L.; Leng, H.; Han, B. Dissolution and passivation mechanism of chalcopyrite during pressurized water leaching. Minerals 2023, 13, 996. [Google Scholar] [CrossRef]
- Bartlett, R.W. Upgrading copper concentrate by hydrothermally converting chalcopyrite to digenite. Metall. Trans. B Process Metall. 1992, 23, 241–248. [Google Scholar] [CrossRef]
- Muszer, A.; Wódka, J.; Chmielewski, T.; Matsuka, S. Covellinisation of copper sulphide minerals under pressure leaching conditions. Hydrometallurgy 2013, 137, 1–7. [Google Scholar] [CrossRef]
- Hawker, W.; Byrne, K.; Vaughan, J. Upgrading copper sulphide concentrates through high temperature metathesis reactions. In COM 2019, Proceedings of the 58th Annual Conference of Metallurgists Hosting the 10th International Copper Conference 2019, Vancouver, BC, Canada, 18–21 August, 2019; MetSoc: Montreal, QC, Canada, 2019; 10p. [Google Scholar]
- Sohn, H.J.; Wadsworth, M.E. Rate processes in the secondary mineralization of copper sulfides. Appl. Mineral. 1984, 485–506. [Google Scholar]
- Bartlett, R.W.; Wilson, D.B.; Savage, B.J.; Wesely, R.J. A process for enriching chalcopyrite concentrates. In Hydrometallurgical Reactor Design and Kinetics; Bautista, R.G., Ed.; TMS: Warrendale, PA, USA, 1986; pp. 227–246. [Google Scholar]
- Jang, J.H.; Wadsworth, M.E. Kinetics of hydrothermal enrichment of chalcopyrite. ACS Symp. Ser. 1993, 550, 45–58. [Google Scholar]
- Peterson, R.D.; Wadsworth, M.E. Solid, solution reactions in the hydrothermal enrichment of chalcopyrite at elevated temperatures. In EPD Congress; Warren, G., Ed.; TMS: Warrendale, PA, USA, 1994; pp. 275–291. [Google Scholar]
- Sequeira, C.A.C.; Santos, D.M.F.; Chen, Y.; Anastassakis, G. Chemical metathesis of chalcopyrite in acidic solutions. Hydrometallurgy 2008, 92, 135–140. [Google Scholar] [CrossRef]
- Kritskii, A.; Naboichenko, S.; Karimov, K.; Agarwal, V.; Lundström, M. Hydrothermal pretreatment of chalcopyrite concentrate with copper sulfate solution. Hydrometallurgy 2020, 195, 105359. [Google Scholar]
- Fomenko, I.V.; Pleshkov, M.A.; Shneerson, Y.M.; Ospanov, E.A.; Shakhalov, A.A.; Naboychenko, S.S. Low-grade copper concentrate purification and enrichment by complex pressure oxidation—Hydrothermal alteration technology. In COM 2019, Proceedings of the 58th Annual Conference of Metallurgists Hosting the 10th International Copper Conference 2019, Vancouver, BC, Canada, 18–21 August, 2019; MetSoc: Montreal, QC, Canada, 2019; 13p. [Google Scholar]
- Viñals, J.; Fuentes, G.; Hernández, M.C.; Herreros, O. Transformation of sphalerite particles into copper sulfide particles by hydrothermal treatment with Cu (II) ions. Hydrometallurgy 2004, 75, 177–187. [Google Scholar] [CrossRef]
- Dunn, G.M. Single Stage Pressure Leach Hydrometallurgical Method for Upgrade of Copper Concentrates. U.S. Patent Application 16/032,726, 17 January 2019. [Google Scholar]
- Van Wyk, A.P.; Akdogan, G.; Bradshaw, S.M. Behaviour of Cu, Fe, Ni, and PGMs during leaching of Ni-Fe-Cu-S converter matte. J. South. Afr. Inst. Min. Metall. 2021, 121, 599–606. [Google Scholar] [CrossRef]
- Dreisinger, D.B. Polymet Mining Corp. Metathetic copper concentrate enrichment. U.S. Patent 8,741,238 B2, 3 June 2014. [Google Scholar]
- Collins, M.J.; Berezowsky, R.M.G.S.; Vardill, W.D.; Ketcham, V.J.; Stojsic, A. The Lihir Gold project: Pressure oxidation process development. In Hydrometallurgy Fundamentals, Technology and Innovations; Hiskey, J.B., Warren, G.W., Eds.; TMS: Littleton, CO, USA, 1993; Chapter 38; pp. 611–628. [Google Scholar]
- Wardell-Johnson, M.; Steiper, G.; Dreisinger, D. Engineering aspects of the Platsol™ process. In ALTA 2009 Nickel/Cobalt 14 Conference; ALTA Metallurgical Services: Melbourne, Australia, 2009; 20p. [Google Scholar]
- Dreisinger, D.; Murray, W.; Hunter, D.; Baxter, K.; Ferron, J.; Fleming, C. The application of the Platsol™ Process to copper-nickel-cobalt-PGE/PGM concentrates from Polymet Mining’s NorthMet Deposit. In ALTA 2005 Nickel/Cobalt 10 Conference; ALTA Metallurgical Services: Melbourne, Australia, 2005; 16p. [Google Scholar]
- King, J.A.; Dreisinger, D.B.; Knight, D.A. The total pressure oxidation of copper concentrates. In The Paul E. Queneau International Symposium Extractive Metallurgy of Copper, Nickel and Cobalt, Volume I: Fundamental Aspects; Reddy, R.G., Weizenbach, R.N., Eds.; TMS: Warrendale, PA, USA, 1993; pp. 735–756. [Google Scholar]
- King, J.; Dreisinger, D.B. Autoclaving of copper concentrates. In Proceedings of Copper 95-Cobre 95, Volume III, Electrorefining and Hydrometallurgy of Copper; Cooper, W.C., Dreisinger, D.B., Dutrizac, J.E., Hein, H., Ugarte, G., Eds.; The Metallurgical Society of CIM: Montreal, QC, Canada, 1995; pp. 511–533. [Google Scholar]
- Defreyne, J.; Grieve, W.; Jones, D.L.; Mayhew, K. The role of iron in the CESL process. In Iron Control Technologies: Proceedings of 3rd International Symposium on Iron Control and Hydrometallurgy; Dutrizac, J.E., Ed.; CIM: Montreal, QC, Canada, 2006; pp. 205–220. [Google Scholar]
- Krebs, D.; Hyvarinen, O.; Dreisinger, D. Processing nickel sulphide ore or concentrates with sodium chloride. CA Patent 2624609C, 31 March 2015. [Google Scholar]
- Lundström, M.; Liipo, J.; Aromaa, J. Dissolution of copper and iron from sulfide concentrates in cupric chloride solution. Int. J. Miner. Process. 2012, 102, 13–18. [Google Scholar] [CrossRef]
- Abdul, B.; Asselin, E. Novel reagents for iron and sulphur control in medium temperature leaching of sulphide concentrates. Can. Metall. Q. 2017, 56, 382–392. [Google Scholar] [CrossRef]
- Yazici, E.; Deveci, H.; Ehsani, A. Recovery of metals from chloride leach solutions of waste of printed circuit boards by adsorption and precipitation. In Proceedings of the XXVII. International Mineral Processing Congress (IMPC 2014); Gecamin/International Mineral Processing Council: Santiago, Chile, 2014; pp. 170–179. [Google Scholar]
- Zhang, R.; Mao, Y.; Liu, C.; Ni, W. Synergistic catalytic effect of chloride ion and ammonium ion on the leaching of chalcopyrite in sulfuric acid solution. Miner. Eng. 2022, 185, 107686. [Google Scholar] [CrossRef]
- FIZ Karlsruhe—Leibniz Institute for Information Infrastructure. Inorganic Crystal Structure Database (ICSD). 2011. Available online: http://icsd.fiz-karlsruhe.de/icsd/ (accessed on 6 March 2020).
- International Centre for Diffraction Data. PDF-4+ 2022 Powder Diffraction File Database. Available online: https://www.icdd.com/ (accessed on 3 May 2022).
- Ventruti, G.; Scordari, F.; Schingaro, E.; Gualtieri, A.F.; Meneghini, C. The order-disorder character of FeOHSO4 obtained from the thermal decomposition of metahohmannite, Fe3+2(H2O)4[O(SO4)2]. Am. Mineral. 2005, 90, 679–686. [Google Scholar] [CrossRef]
- Zies, E.G.; Allen, E.T.; Merwin, H.E. Some reactions involved in secondary copper sulphide enrichment. Econ. Geol. 1916, 11, 407–503. [Google Scholar] [CrossRef]
- Young, S.W.; Moore, N.P. Laboratory studies on secondary sulphide ore enrichment, I. The copper sulphides and hydrogen sulphide. Econ. Geol. 1916, 11, 349–365. [Google Scholar] [CrossRef]
- Young, S.W.; Moore, N.P. Laboratory studies on secondary sulphide ore enrichment, II. The formation of chalcopyrite by artificial replacement. Econ. Geol. 1916, 11, 574–581. [Google Scholar] [CrossRef]
- Viramontes Gamboa, G.; Velásquez Roque, S.; Ibarra Bracamontes, L.A.; Gonzalez Bernal, R. Lixiviación de calcopirita en minutos. In XXI Congreso Internacional de Metalurgia Extractiva; Lapidus Lavine, G.T., Alonso Gómez, A.R., Fuentes Aceituno, J.C., Hernández Cruz, L., Herrera Urbina, R., Lara Castro, R.H., Luna Sánchez, R.M., Pérez Tello, M., Zárate Gutiérrez, R., Eds.; Universidad Autónoma Metropolitana: Mexico City, Mexico, 2012; pp. 336–350. [Google Scholar]
- Byrne, K.; Hawker, W.; Vaughan, J. Deportment of radionuclides during copper concentrate metathesis reactions. In ALTA 2018 23rd Annual Conference Proceedings, Uranium-REE-Lithium Conference; ALTA Metallurgical Services: Melbourne, Australia, 2018; 12p. [Google Scholar]
- Chaudhari, A.; Webster, N.A.; Xia, F.; Frierdich, A.; Ram, R.; Etschmann, B.; Liu, W.; Wykes, J.; Brand, H.E.; Brugger, J. Anatomy of a complex mineral replacement reaction: Role of aqueous redox, mineral nucleation, and ion transport properties revealed by an in-situ study of the replacement of chalcopyrite by copper sulfides. Chem. Geol. 2021, 581, 120390. [Google Scholar] [CrossRef]
- Chaudhari, A.; Brugger, J.; Ram, R.; Chowdhury, P.; Etschmann, B.; Guagliardo, P.; Xia, F.; Pring, A.; Gervinskas, G.; Liu, A.; et al. Synchronous solid-state diffusion, dissolution-reprecipitation, and recrystallization leading to isotopic resetting: Insights from chalcopyrite replacement by copper sulfides. Geochim. Cosmochim. Acta 2022, 331, 48–68. [Google Scholar] [CrossRef]
- Harmer, S.L.; Pratt, A.R.; Nesbitt, W.H.; Fleet, M.E. Sulfur species at chalcopyrite (CuFeS2) fracture surfaces. Am. Mineral. 2004, 89, 1026–1032. [Google Scholar] [CrossRef]
- Senanayake, G. A review of chloride assisted copper sulphide leaching by oxygenated sulphuric acid and mechanistic considerations. Hydrometallurgy 2009, 98, 21–32. [Google Scholar] [CrossRef]
- Cheng, T.C.M. Production of Hematite in Acidic Zinc Sulphate Media. Ph.D. Thesis, McGill University, Montreal, QC, Canada, 2002; 299p. [Google Scholar]
- Reid, M.; Papangelakis, V.G. New data on hematite solubility in sulphuric acid solution from 130 to 270 °C. In Iron Control Technologies; Durizac, J.E., Riveros, P.A., Eds.; MetSoc: Montreal, QC, Canada, 2006; pp. 673–686. [Google Scholar]
- Liu, H.; Papangelakis, V.G.; Alam, M.S.; Singh, G. Solubility of hematite in H2SO4 solutions at 230–270 °C. Can. Metall. Q. 2003, 42, 199–208. [Google Scholar] [CrossRef]
- Kobylin, P.M.; Sippola, H.; Taskinen, P.A. Thermodynamic modelling of aqueous Fe(II) sulphate solutions. CALPHAD Comput. Coupling Phase Diagr. Thermochem. 2011, 35, 499–511. [Google Scholar] [CrossRef]
- Hasegawa, F.; Tozawa, K.; Nishimura, T. Solubility of ferrous sulphate in aqueous solutions at high temperatures. Shigen Sozai 1996, 112, 879–884. [Google Scholar] [CrossRef]
- Cai, Y.; Chen, X.; Ding, J.; Zhou, D. Leaching mechanism for chalcopyrite in hydrochloric acid. Hydrometallurgy 2012, 113–114, 109–118. [Google Scholar] [CrossRef]
- Xia, F.; Brugger, J.; Chen, G.; Ngothai, Y.; O’Neill, B.; Putnis, A.; Pring, A. Mechanism and kinetics of pseudomorphic mineral replacement reactions: A case study of the replacement of pentlandite by violarite. Geochim. Cosmochim. Acta 2009, 73, 1945–1969. [Google Scholar] [CrossRef]
- Harmer, S.L.; Thomas, J.E.; Fornasiero, D.; Gerson, A.R. The evolution of surface layers formed during chalcopyrite leaching. Geochim. Cosmochim. Acta 2006, 70, 4392–4402. [Google Scholar] [CrossRef]
- Senanayake, G. Chloride assisted leaching of chalcocite by oxygenated sulphuric acid via Cu(II)-OH-Cl. Miner. Eng. 2007, 20, 1075–1088. [Google Scholar] [CrossRef]
- Pearce, C.I.; Pattrick, R.A.D.; Vaughan, D.J.; Henderson, C.M.B.; van der Laan, G. Copper oxidation state in chalcopyrite: Mixed Cu d9 and d10 characteristics. Geochim. Cosmochim. Acta 2006, 70, 4635–4642. [Google Scholar] [CrossRef]
- Li, Y.; Liu, Y.; Chen, J.; Zhao, C.; Cui, W. Comparison study of crystal and electronic structures for chalcopyrite (CuFeS2) and pyrite (FeS2). Physicochem. Probl. Miner. Process. 2021, 57, 100–111. [Google Scholar] [CrossRef]
- Conejeros, S.; Alemany, P.; Llunell, M.; Moreira, I.D.P.; Sánchez, V.; Llanos, J. Electronic structure and magnetic properties of CuFeS2. Inorg. Chem. 2015, 54, 4840–4849. [Google Scholar] [CrossRef] [PubMed]
- McDonald, G.W.; Udovic, T.J.; Dumesic, J.A.; Langer, S.H. Equilibria associated with cupric chloride leaching of chalcopyrite concentrate. Hydrometallurgy 1984, 13, 125–135. [Google Scholar] [CrossRef]
- Lundström, M.; Liipo, J.; Taskinen, P.; Aromaa, J. Copper precipitation during leaching of various copper sulfide concentrates with cupric chloride in acidic solutions. Hydrometallurgy 2016, 166, 136–142. [Google Scholar] [CrossRef]
- Fjellvåg, H.; Grønvold, F.; Stølen, S. Low-temperature distortion in CuS. Z. Krist. 1988, 184, 111–121. [Google Scholar] [CrossRef]
- Conejeros, S.; Moreira, I.D.P.; Alemany, P.; Canadell, E. Nature of holes, oxidation states, and hypervalency in covellite (CuS). Inorg. Chem. 2014, 53, 12402–12406. [Google Scholar] [CrossRef] [PubMed]
- Kalanur, S.S.; Seo, H. Synthesis of CuxS thin films with tunable localized surface plasmon resonances. Chem. Sel. 2018, 3, 5920–5926. [Google Scholar]
- Evans, H.T.; Konnert, J.A. Crystal structure refinement of covellite. Am. Mineral. 1976, 61, 996–1000. [Google Scholar]
- Banerjee, N.; Krupanidhi, S.B. Facile hydrothermal synthesis and observation of bubbled growth mechanism in nano-ribbons aggregated microspherical covellite blue-phosphor. Dalton Trans. 2010, 39, 9789–9793. [Google Scholar] [CrossRef]
- Krishnamoorthy, K.; Veerasubramani, G.K.; Rao, A.N.; Kim, S.J. One-pot hydrothermal synthesis, characterization and electrochemical properties of CuS nanoparticles towards supercapacitor applications. Mater. Res. Express 2014, 1, 035006. [Google Scholar] [CrossRef]
- Auyoong, Y.L.; Yap, P.L.; Huang, X.; Abd Hamid, S.B. Optimization of reaction parameters in hydrothermal synthesis: A strategy towards the formation of CuS hexagonal plates. Chem. Cent. J. 2013, 7, 67. [Google Scholar] [CrossRef]
- Patel, T.A.; Balasubramanian, C.; Panda, E. Role of reducing agent and self-sacrificed copper-thiourea complex in the synthesis of precisely controlled Cu2-xS microtubes. J. Cryst. Growth 2019, 505, 26–32. [Google Scholar] [CrossRef]
- Mikhlin, Y.; Nasluzov, V.; Ivaneeva, A.; Vorobyev, S.; Likhatski, M.; Romanchenko, A.; Krylov, A.; Zharkov, S.; Meira, D.M. Formation, evolution and characteristics of copper sulfide nanoparticles in the reactions of aqueous cupric and sulfide ions. Mater. Chem. Phys. 2020, 255, 123600. [Google Scholar] [CrossRef]
- Pejjai, B.; Reddivari, M.; Kotte, T.R.R. Phase controllable synthesis of CuS nanoparticles by chemical co-precipitation method: Effect of copper precursors on the properties of CuS. Mater. Chem. Phys. 2020, 239, 122030. [Google Scholar] [CrossRef]
- Almeida, T.; Garcia, E.M.; da Silva, H.W.A.; Matencio, T.; Lins, V.D.F.C. Electrochemical study of chalcopyrite dissolution in sulfuric, nitric and hydrochloric acid solutions. Int. J. Miner. Process. 2016, 149, 25–33. [Google Scholar] [CrossRef]
- Ghahremaninezhad, A.; Radzinski, R.; Gheorghiu, T.; Dixon, D.G.; Asselin, E. A model for silver ion catalysis of chalcopyrite (CuFeS2) dissolution. Hydrometallurgy 2015, 155, 95–104. [Google Scholar] [CrossRef]
- Recalde Chiluiza, E.L.; Navarro Donoso, P. Chalcopyrite leaching in acidic chloride solution without sulphates. J. Mex. Chem. Soc. 2016, 60, 238–246. [Google Scholar] [CrossRef]
- Prater, J.D.; Queneau, P.B.; Hudson, T.J. The sulfation of copper-iron sulfides with concentrated sulfuric acid. JOM 1970, 22, 23–27. [Google Scholar] [CrossRef]
- Yang, Y.; Liu, W.; Chen, M. XANES and XRD study of the effect of ferrous and ferric ions on chalcopyrite bioleaching at 30 °C and 48 °C. Miner. Eng. 2015, 70, 99–108. [Google Scholar] [CrossRef]
- Yang, C.; Qin, W.; Zhao, H.; Wang, J.; Wang, X. Mixed potential plays a key role in leaching of chalcopyrite: Experimental and theoretical analysis. Ind. Eng. Chem. Res. 2018, 57, 1733–1744. [Google Scholar] [CrossRef]
- Yoo, K.; Kim, S.; Lee, J.; Ito, M.; Tsunekawa, M.; Hiroyoshi, M. Effect of chloride ions on the leaching rate of chalcopyrite. Miner. Eng. 2010, 23, 471–477. [Google Scholar] [CrossRef]
- Gao, X.; Yang, Y.; Pownceby, M.I.; Zhong, S.; Chen, M. A sulfur K-edge XANES and Raman study on the effect of chloride ion on bacterial and chemical leaching of chalcopyrite at 25 °C. Min. Metall. Explor. 2019, 36, 343–352. [Google Scholar] [CrossRef]
- Majuste, D.; Ciminelli, V.S.T.; Eng, P.J.; Osseo-Asare, K. Applications of in situ synchrotron XRD in hydrometallurgy: Literature review and investigation of chalcopyrite dissolution. Hydrometallurgy 2013, 131, 54–66. [Google Scholar] [CrossRef]
- Velásquez-Yévenes, L.; Nicol, M.; Miki, H. The dissolution of chalcopyrite in chloride solutions. Part 1. The effect of solution potential. Hydrometallurgy 2010, 103, 113–118. [Google Scholar] [CrossRef]
- Peters, E.; Loewen, F. Pressure leaching of copper minerals in perchloric acid solutions. Metall. Trans. 1973, 4, 5–14. [Google Scholar] [CrossRef]
- Corriou, J.-P.; Kikindai, T. The aqueous oxidation of elemental sulfur and different chemical properties of the allotropic forms Sλ and Sμ. J. Inorg. Nucl. Chem. 1981, 43, 9–15. [Google Scholar] [CrossRef]
- Whittington, B.I.; Muir, D.M. Pressure acid leaching of nickel laterites. A review. Miner. Process. Extr. Metall. Rev. 2000, 21, 527–599. [Google Scholar] [CrossRef]
- Tozawa, K.; Sasaki, K. Effect of co-existing sulphates on precipitation of ferric oxide from ferric sulphate solutions at elevated temperatures. In Iron Control in Hydrometallurgy; Dutrizac, J.E., Monhemius, A.J., Eds.; Ellis Horwood: Chichester, UK, 1986; pp. 454–476. [Google Scholar]
- Umetsu, V.; Tozawa, K.; Sasaki, K.-I. The hydrolysis of ferric sulphate solutions at elevated temperatures. Can. Metall. Q. 1977, 16, 111–117. [Google Scholar] [CrossRef]
- Cheng, T.C.; Demopoulos, G.P. Hydrolysis of ferric sulfate in the presence of zinc sulfate at 200 °C: Precipitation kinetics and product characterization. Ind. Chem. Eng. Res. 2004, 43, 6299–6308. [Google Scholar] [CrossRef]
- Habashi, F.; Bauer, E.L. Aqueous oxidation of elemental sulfur. Ind. Eng. Chem. Fundam. 1966, 5, 469–471. [Google Scholar] [CrossRef]
- Majuste, D.; Ciminelli, V.S.T.; Osseo-Asare, K.; Dantas, M.S.S.; Magalhães-Paniago, R. Electrochemical dissolution of chalcopyrite: Detection of bornite by synchrotron small angle X-ray diffraction and its correlation with the hindered dissolution process. Hydrometallurgy 2012, 111, 114–123. [Google Scholar] [CrossRef]
- Ruiz, M.C.; Zapata, J.; Padilla, R. Effect of variables on the quality of hematite precipitated from sulfate solutions. Hydrometallurgy 2007, 89, 32–39. [Google Scholar] [CrossRef]
- Antonijević, M.; Bogdanović, G.D. Investigation of the leaching of chalcopyritic ore in acidic solutions. Hydrometallurgy 2004, 73, 245–256. [Google Scholar] [CrossRef]
- Córdoba, E.M.; Muñoz, J.A.; Blázquez, M.L.; González, F.; Ballester, A. Leaching of chalcopyrite with ferric ion. Part I: General aspects. Hydrometallurgy 2008, 93, 81–87. [Google Scholar] [CrossRef]
- Fleming, C. Basic iron sulfate. A potential killer in the processing of refractory gold concentrates by pressure oxidation. Miner. Metall. Process. 2010, 27, 81–88. [Google Scholar] [CrossRef]
- Welham, N.J.; Malatt, K.A.; Vukcevic, S. The stability of iron phases presently used for disposal from metallurgical systems—A review. Miner. Eng. 2000, 13, 911–931. [Google Scholar] [CrossRef]
- Nazari, G.; Dixon, D.G.; Dreisinger, D. Enhancing the kinetic of chalcopyrite leaching in the Galvanox™ process. Hydrometallurgy 2011, 105, 251–258. [Google Scholar] [CrossRef]
- Nazari, G.; Dixon, D.G.; Dreisinger, D.B. The mechanism of chalcopyrite leaching in the presence of silver-enhanced pyrite in the Galvanox™ process. Hydrometallurgy 2012, 113, 122–130. [Google Scholar] [CrossRef]
- Chehreghani, S.; Yari, M.; Zeynali, A.; Akhgar, B.N.; Gharegheshlagh, H.H.; Pishravian, M. Optimization of chalcopyrite galvanic leaching in the presence of pyrite and silver as catalysts by using Response Surface Methodology (RSM). Rud.-Geološko-Naft. Zb. 2021, 36, 37–47. [Google Scholar] [CrossRef]
- Nicol, M.J. Does galvanic coupling with pyrite increase the rate of dissolution of chalcopyrite under ambient conditions? An electrochemical study. Hydrometallurgy 2022, 208, 105824. [Google Scholar] [CrossRef]
- Javed, T.; Asselin, E. Fe (III) Precipitation and Copper Loss from Sulphate-Chloride Solutions at 150 °C: A Statistical Approach. Metals 2020, 10, 669. [Google Scholar] [CrossRef]
- Wolska, E. The structure of hydrohematite. Z. Krist. 1981, 154, 69–75. [Google Scholar]
- Wolska, E.; Schwertmann, U. Selective X-ray line broadening in the goethite-derived hematite phase. Phys. Status Solidi A 1989, 114, K11–K16. [Google Scholar] [CrossRef]
- Stanjek, H.; Schwertmann, U. The influence of aluminum on iron oxides. Part XVI: Hydroxyl and aluminum substitution in synthetic hematites. Clays Clay Miner. 1992, 40, 347–354. [Google Scholar] [CrossRef]
- Whittington, B.I.; Johnson, J.A.; Quan, L.P.; McDonald, R.G.; Muir, D.M. Pressure acid leaching of arid-region nickel laterite ore: Part II. Effect of ore type. Hydrometallurgy 2003, 70, 47–62. [Google Scholar] [CrossRef]
- Whittington, B.I. Characterization of scales obtained during continuous nickel laterite pilot-plant leaching. Metall. Mater. Trans. B 2000, 31, 1175–1186. [Google Scholar] [CrossRef]
- Barr, G.; Defreyne, J.; Jones, D.; Moore, R. The CESL process: Successful refining of a low grade copper concentrate. In ALTA 2000 Copper 6 Forum, Perth; ALTA Metallurgical Services: Melbourne, Australia, 2000; 10p. [Google Scholar]
- Cheng, C.Y.; Lawson, F. The kinetics of leaching covellite in acidic oxygenated sulphate-chloride solutions. Hydrometallurgy 1991, 27, 269–284. [Google Scholar] [CrossRef]
- Pollard, A.M.; Thomas, R.G.; Williams, P.A. The stabilities of antlerite and Cu3SO4(OH)4·2H2O: Their formation and relationships to other copper (II) sulfate minerals. Mineral. Mag. 1992, 56, 359–365. [Google Scholar] [CrossRef]
- Yoder, C.H.; Agee, T.M.; Ginion, K.E.; Hofmann, A.E.; Ewanichak, J.E.; Schaeffer, C.D., Jr.; Carroll, M.J.; Schaeffer, R.W.; McCaffrey, P.F. The relative stabilities of the copper hydroxyl sulphates. Mineral. Mag. 2007, 71, 571–577. [Google Scholar] [CrossRef]
- Zittlau, A.H.; Shi, Q.; Boerio-Goates, J.; Woodfield, B.F.; Majzlan, J. Thermodynamics of the basic copper sulfates antlerite, posnjakite, and brochantite. Chem. Erde-Geochem. 2013, 73, 39–50. [Google Scholar] [CrossRef]
- Woods, T.L.; Garrels, R.M. Phase relations of some cupric hydroxy minerals. Econ. Geol. 1986, 81, 1989–2007. [Google Scholar] [CrossRef]
- Pollard, A.M.; Thomas, R.G.; Williams, P.A. Connellite: Stability relationships with other secondary copper minerals. Mineral. Mag. 1990, 54, 425–430. [Google Scholar] [CrossRef]
- Scott, D.A. A review of copper chlorides and related salts in bronze corrosion and as painting pigments. Stud. Conserv. 2000, 45, 39–53. [Google Scholar]
- Yin, Q.; Vaughan, D.J.; England, K.E.R.; Kelsall, G.H.; Brandon, N.P. Surface oxidation of chalcopyrite (CuFeS2) in alkaline solutions. J. Electrochem. Soc. 2000, 147, 2945–2951. [Google Scholar] [CrossRef]
- Azizkarimi, M.; Tabaian, S.H.; Rezai, B. Electrochemical investigation of chalcopyrite oxidation in alkaline solutions. Sep. Sci. Technol. 2014, 49, 2595–2601. [Google Scholar] [CrossRef]
- Nicol, M.J. The electrochemistry of chalcopyrite in alkaline solutions. Hydrometallurgy 2019, 187, 134–140. [Google Scholar] [CrossRef]
- O’Connor, G.M.; Lepkova, K.; Eksteen, J.J.; Oraby, E.A. Electrochemical behaviour and surface analysis of chalcopyrite in alkaline glycine solutions. Hydrometallurgy 2018, 182, 32–43. [Google Scholar] [CrossRef]
- Liu, X.D.; Meng, D.D.; Zheng, X.G.; Hagihala, M.; Guo, Q.X. Mid-IR and Raman spectral properties of clinoatacamite-structure basic copper chlorides. Adv. Mater. Res. 2011, 146, 1202–1205. [Google Scholar] [CrossRef]
- Zhao, Y.; Cui, H.; Zhang, J.; Ma, Y.; Tian, H.; Wu, L.; Cui, Q.; Ma, Y. Pressure-induced phase transformation of botallackite α-Cu2(OH)3Cl with a two-dimensional layered structure synthesized via a hydrothermal strategy. J. Phys. Chem. C 2020, 124, 9581–9590. [Google Scholar] [CrossRef]
- Krivovichev, S.V.; Hawthorne, F.C.; Williams, P.A. Structural complexity and crystallization: The Ostwald sequence of phases in the Cu2(OH)3Cl system (botallackite–atacamite–clinoatacamite). Struct. Chem. 2017, 28, 153–159. [Google Scholar] [CrossRef]
- Pollard, A.M.; Thomas, R.G.; Williams, P.A. Synthesis and stabilities of the basic copper (II) chlorides atacamite, paratacamite and botallackite. Mineral. Mag. 1989, 53, 557–563. [Google Scholar] [CrossRef]
- Sharkey, J.B.; Lewin, S.Z. Conditions governing the formation of atacamite and paratacamite. Am. Mineral. J. Earth Planet. Mater. 1971, 56, 179–192. [Google Scholar]
- Kampf, A.R.; Sciberras, M.J.; Leverett, P.; Williams, P.A.; Malcherek, T.; Schlüter, J.; Welch, M.D.; Dini, M.; Donoso, A.M. Paratacamite-(Mg), Cu3(Mg,Cu)Cl2(OH)6; a new substituted basic copper chloride mineral from Camerones, Chile. Mineral. Mag. 2013, 77, 3113–3124. [Google Scholar] [CrossRef]
- Sciberras, M.J.; Leverett, P.; Williams, P.A.; Hibbs, D.E.; Downes, P.J.; Welch, M.D.; Kampf, A.R. Paratacamite-(Ni), Cu3(Ni, Cu)Cl2(OH)6, a new mineral from the Carr Boyd Rocks mine, Western Australia. Aust. J. Mineral. 2013, 17, 39–44. [Google Scholar]
- Welch, M.D.; Sciberras, M.J.; Williams, P.A.; Leverett, P.; Schlüter, J.; Malcherek, T. A temperature-induced reversible transformation between paratacamite and herbertsmithite. Phys. Chem. Miner. 2014, 41, 33–48. [Google Scholar] [CrossRef]
- Sciberras, M.J. Substitution in Basic Secondary Cu(II) Chloride Minerals. Ph.D. Thesis, University of Western Sydney, Penrith, Australia, 2013; 132p. [Google Scholar]
- Grice, J.D.; Szymanski, J.T.; Jambor, J.L. The crystal structure of clinoatacamite, a new polymorph of Cu2(OH)3Cl. Can. Mineral. 1996, 34, 73–78. [Google Scholar]
- Oswald, H.R.; Guenter, J.R. Crystal data on paratacamite, γ-Cu2(OH)3Cl. J. Appl. Crystallogr. 1971, 4, 530–531. [Google Scholar] [CrossRef]
- Jambor, J.L.; Dutrizac, J.E.; Roberts, A.C.; Grice, J.D.; Szymanski, J.T. Clinoatacamite, a new polymorph of Cu2(OH)3Cl, and its relationship to paratacamite and “anarakite”. Can. Mineral. 1996, 34, 61–72. [Google Scholar]
- Zheng, X.G.; Kawae, T.; Kashitani, Y.; Li, C.S.; Tateiwa, N.; Takeda, K.; Yamada, H.; Xu, C.N.; Ren, Y. Unconventional magnetic transitions in the mineral clinoatacamite Cu2Cl(OH)3. Phys. Rev. B 2005, 71, 052409. [Google Scholar] [CrossRef]
- Malcherek, T.; Schlüter, J. Structures of the pseudo-trigonal polymorphs of Cu2(OH)3Cl. Acta Crystallogr. Sect. B Struct. Sci. 2009, 65, 334–341. [Google Scholar] [CrossRef]
- Vieillard, P. Proprietes thermochimiques des composes du cuivre. Sci. Geol. Bull. 1988, 41, 289–308. [Google Scholar] [CrossRef]
- Moore, T.P. What’s new in the mineral world? The Mineralogical Record, Report #36, 2013. Available online: https://cdn.mineralogicalrecord.com/wp-content/uploads/2020/10/whats_new/Toms-Online-report-36.pdf (accessed on 24 August 2023).
- Cook, R.B. Ataeamite: Moonta Mine, South Australia, Australia. Rocks Miner. 2006, 81, 374–378. [Google Scholar] [CrossRef]
- Gablina, I.F.; Semkova, T.A.; Stepanova, T.V.; Gor’Kova, N.V. Diagenetic alterations of copper sulfides in modern ore-bearing sediments of the Logatchev-1 hydrothermal field (Mid-Atlantic Ridge 14 45′ N). Lithol. Miner. Resour. 2006, 41, 27–44. [Google Scholar] [CrossRef]
- Chen, J.; Xu, Z.; Chen, Y. Electronic Structure and Surfaces of Sulfide Minerals: Density Functional Theory and Applications; Elsevier: Amsterdam, The Netherlands, 2020; 379p. [Google Scholar]
- Speight, J. Lange’s Handbook of Chemistry, 16th ed.; McGraw-Hill: New York, NY, USA, 2005. [Google Scholar]
- Reed, J.J. Digitizing “The NBS Tables of Chemical Thermodynamic Properties: Selected Values for Inorganic and C1 and C2 Organic Substances in SI Units”. J. Res. Natl. Inst. Stand. Technol. 2020, 125, 125007. [Google Scholar] [CrossRef]
- Robie, R.A.; Hemingway, B.S. Thermodynamic Properties of Minerals and Related Substances at 298.15 K and 1 Bar (105 Pascals) Pressure and at Higher Temperatures; US Government Printing Office: Washington, DC, USA, 1995; Volume 2131.
- Drouet, C.; Navrotsky, A. Synthesis, characterization, and thermochemistry of K-Na-H3O jarosites. Geochim. Cosmochim. Acta 2003, 67, 2063–2076. [Google Scholar] [CrossRef]
- Stoffregen, R.E. Stability relations of jarosite and natrojarosite at 150–250 °C. Geochim. Cosmochim. Acta 1993, 57, 2417–2429. [Google Scholar] [CrossRef]
- Gaboreau, S.; Vieillard, P. Prediction of Gibbs free energies of formation of minerals of the alunite supergroup. Geochim. Cosmochim. Acta 2004, 68, 3307–3316. [Google Scholar] [CrossRef]
- Stoffregen, R.E.; Cygan, G.L. An experimental study of Na-K exchange between alunite and aqueous sulfate solutions. Am. Mineral. 1990, 75, 209–220. [Google Scholar]
- Majzlan, J.; Dachs, E.; Benisek, A.; Plášil, J.; Sejkora, J. Thermodynamics, crystal chemistry and structural complexity of the Fe(SO4)(OH)(H2O)x phases: Fe(SO4)(OH), metahohmannite, butlerite, parabutlerite, amarantite, hohmannite, and fibroferrite. Eur. J. Mineral. 2018, 30, 259–275. [Google Scholar] [CrossRef]
- Bissengaliyeva, M.R.; Ogorodova, L.P.; Mel’Chakova, L.V.; Vigasina, M.F. Thermochemical investigation of natural antlerite. J. Therm. Anal. Calorim. 2012, 109, 467–471. [Google Scholar] [CrossRef]
- Yoder, C.H.; Flora, N.J. Geochemical applications of the simple salt approximation to the lattice energies of complex materials. Am. Mineral. 2005, 90, 488–496. [Google Scholar] [CrossRef]
- Puigdomenech, I.; Taxén, C. Thermodynamic Data for Copper. Implications for the Corrosion of Copper under Repository Conditions. Swedish Nuclear Fuel and Waste Management Company Report, TR-00-13, 2000; 95p. Available online: https://inis.iaea.org/search/search.aspx?orig_q=RN:32010691 (accessed on 16 August 2023).
- Wang, L.; Xue, N.; Zhang, Y.; Hu, P. Controlled hydrothermal precipitation of alunite and natroalunite in high-aluminum vanadium-bearing aqueous system. Minerals 2021, 11, 892. [Google Scholar] [CrossRef]
- Ball, J.W.; Nordstrom, D.K. User’s Manual for Wateq4f, with Revised Thermodynamic Data Base and Test Cases for Calculating Speciation of Major, Trace, and Redox Elements in Natural Waters; Open-File Report 91-183; US Geological Survey: Reston, VA, USA, 1991; 59p.
- Torres, C.M.; Ghorbani, Y.; Hernández, P.C.; Justel, F.J.; Aravena, M.I.; Herreros, O.O. Cupric and chloride ions: Leaching of chalcopyrite concentrate with low chloride concentration media. Minerals 2019, 9, 639. [Google Scholar] [CrossRef]
- Martins, F.L.; Patto, G.B.; Leão, V.A. Chalcopyrite bioleaching in the presence of high chloride concentrations. J. Chem. Technol. Biotechnol. 2019, 94, 2333–2344. [Google Scholar] [CrossRef]
- Peters, E. Direct leaching of sulfides: Chemistry and applications. Metall. Trans. B Process Metall. 1976, 7, 505–517. [Google Scholar] [CrossRef]











| Elemental Content (% w/w) | |||||||
|---|---|---|---|---|---|---|---|
| Cu | Fe | Al | Mg | Ca | Si | Na | S |
| 24.5 | 26.9 | 0.240 | 1.14 | 0.015 | 4.35 | 0.008 | 31.4 |
| Pulp Density (% w/w) | Cu | Fe | Al | Mg | Ca | Si | Na | S |
|---|---|---|---|---|---|---|---|---|
| Elemental concentration range (g/L) | ||||||||
| 10 | 24.4 29.5 | 0.54 1.49 | 0.25 0.57 | 0.55 3.23 | 0.23 0.25 | 0.51 0.54 | 0.01 10.3 | 31.5 34.0 |
| 20 | 58.3 68.5 | 0.21 21.0 | 0.21 1.35 | 1.18 3.72 | 0.03 0.52 | 0.04 0.51 | 0.03 64.9 | 60.1 89.6 |
| Extraction range (%) | ||||||||
| 10 | 98.9 99.3 | 1.8 4.9 | 68.8 87.3 | 50.6 87.9 | 97.9 99.5 | 9.3 9.8 | - | 96.5 97.9 |
| 20 | 97.2 99.8 | 3.5 24.8 | 10.0 80.0 | 38.3 80.7 | 82.6 99.4 | 0.4 4.2 | - | 66.2 98.1 |
| Phase | Max. (% w/w) | Phase | Range (% w/w) * |
|---|---|---|---|
| Hematite | 70.5 | Antlerite | 1.5–9.3 |
| Alunite/jarosite # | 60.0 | Clinoatacamite | 6.1–16.1 |
| Basic ferric sulphate | 59.4 | ||
| Covellite | 36.9 | ||
| Sulphur | 20.2 |
| Unit Cell Parameter | Source | |||||
|---|---|---|---|---|---|---|
| [132] | [133] | [131] | [134] | [135] * | This Study | |
| a (Å) | 6.166 | 6.157 | 6.144 | 6.164 | 6.1223 | 6.161 |
| b (Å) | 6.822 | 6.814 | 6.805 | 6.817 | 6.8346 | 6.823 |
| c (Å) | 9.120 | 9.105 | 9.112 | 9.114 | 9.1841 | 9.122 |
| β (°) | 99.63 | 99.65 | 99.55 | 99.65 | 99.492 | 99.57 |
| Mineral | ΔHo (kJ/mol) | So (J/mol K) | ΔGo (kJ/mol) | References | ||
|---|---|---|---|---|---|---|
| ΔHo | So | ΔGo | ||||
| Chalcopyrite | −194.9 | 124.9 | −195.1 | [143] | [143] | [143] |
| Covellite | −54.6 | 67.4 | −55.3 | |||
| Hematite | −826.2 | 87.4 | −744.4 | |||
| Natrojarosite | −3783.4 | 382.4 | −3270.7 | [144] | [145] | [146] |
| Natroalunite | −5132.0 | 321.1 | −4622.4 | [147] | [147] | [147] |
| Basic ferric sulphate | −1160.2 | 145.9 | −1013.7 | [148] | [148] | [148] |
| Antlerite | −1733.6 | 274.3 | −1453.6 | [114] | [114] | [114] |
| −1750 | 263.5 | −1467 | [149] | [149] | [149] | |
| Atacamite | −1672.7 −1444 | 335.6 | −1339.9 | [150] calc. | [151] | [151] |
| Clinoatacamite | −1341.8 | [115] | ||||
| Reaction | ΔHo (kJ/mol) * | So (J/mol K) | ΔGo (kJ/mol) * |
|---|---|---|---|
| Metathesis (14) | −68.2 | −28.2 | −59.9 |
| Chalcopyrite partial oxidation (15) | −858.1 | −585.1 | −683.7 |
| Covellite dissolution with sulphur formation (16) | −656.7 | −443.6 | −534.0 |
| Covellite dissolution with acid formation (17) | −1280 | −801.0 | −1041 |
| Covellite complete dissolution (18) | −1580 | −1114 | −1248 |
| Aqueous copper oxidation with Fe(III) (19) | −95.0 | 76.1 | −117.5 |
| Aqueous copper oxidation with O2 (20) | −299.6 | −312.9 | −206.1 |
| Aqueous iron oxidation with O2 (21) | −102.3 | −194.5 | −44.3 |
| Liquid sulphur oxidation (22) | −624.9 | −392.5 | −507.8 |
| Hematite formation (23) | 128.4 | 509.4 | −23.7 |
| Natrojarosite formation (24) | 135.9 | 811.9 | −82.6 |
| Basic ferric sulphate formation (25) | 83.4 | 371.8 | −27.3 |
| Hematite to natrojarosite conversion (26) | −37.7 | 31.8 | −31.4 |
| Hematite to basic ferric sulphate conversion (27) | 38.5 | 234.1 | −30.9 |
| Natroalunite formation (28) | 235.8 | 768.2 | 7.9 |
| Clinoatacamite formation (29) | −548.9 | 268.5 | −628.2 |
| Antlerite formation (30) ** | 124.7 108.3 | 273.3 262.5 | 43.2 29.8 |
| Chalcopyrite partial oxidation with hematite formation (31) | −896.2 | −524.9 | −739.8 |
| Chalcopyrite partial oxidation with sulphur and hematite formation (32) | −1063 | −724.5 | −856.2 |
| Complete chalcopyrite complete oxidation with hematite formation (33) | −1686 | −1082 | −1364 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
McDonald, R.G. The Effects of Chloride on the High Temperature Pressure Oxidation of Chalcopyrite: Some Insights from Batch Tests—Part 2: Leach Residue Mineralogy. Minerals 2023, 13, 1162. https://doi.org/10.3390/min13091162
McDonald RG. The Effects of Chloride on the High Temperature Pressure Oxidation of Chalcopyrite: Some Insights from Batch Tests—Part 2: Leach Residue Mineralogy. Minerals. 2023; 13(9):1162. https://doi.org/10.3390/min13091162
Chicago/Turabian StyleMcDonald, Robbie G. 2023. "The Effects of Chloride on the High Temperature Pressure Oxidation of Chalcopyrite: Some Insights from Batch Tests—Part 2: Leach Residue Mineralogy" Minerals 13, no. 9: 1162. https://doi.org/10.3390/min13091162





