Thermal Transport Properties of Olivine, Wadsleyite, and Ringwoodite—A Review
Abstract
1. Introduction
2. Basic Concepts and Experimental Techniques
2.1. D and κ
2.2. Heat Transfer Mechanism
2.3. Influencing Factors of D and κ
2.3.1. Temperature
2.3.2. Pressure
2.3.3. Porosity
2.3.4. Crystal Structure
2.3.5. Water
2.4. Measurement Technology
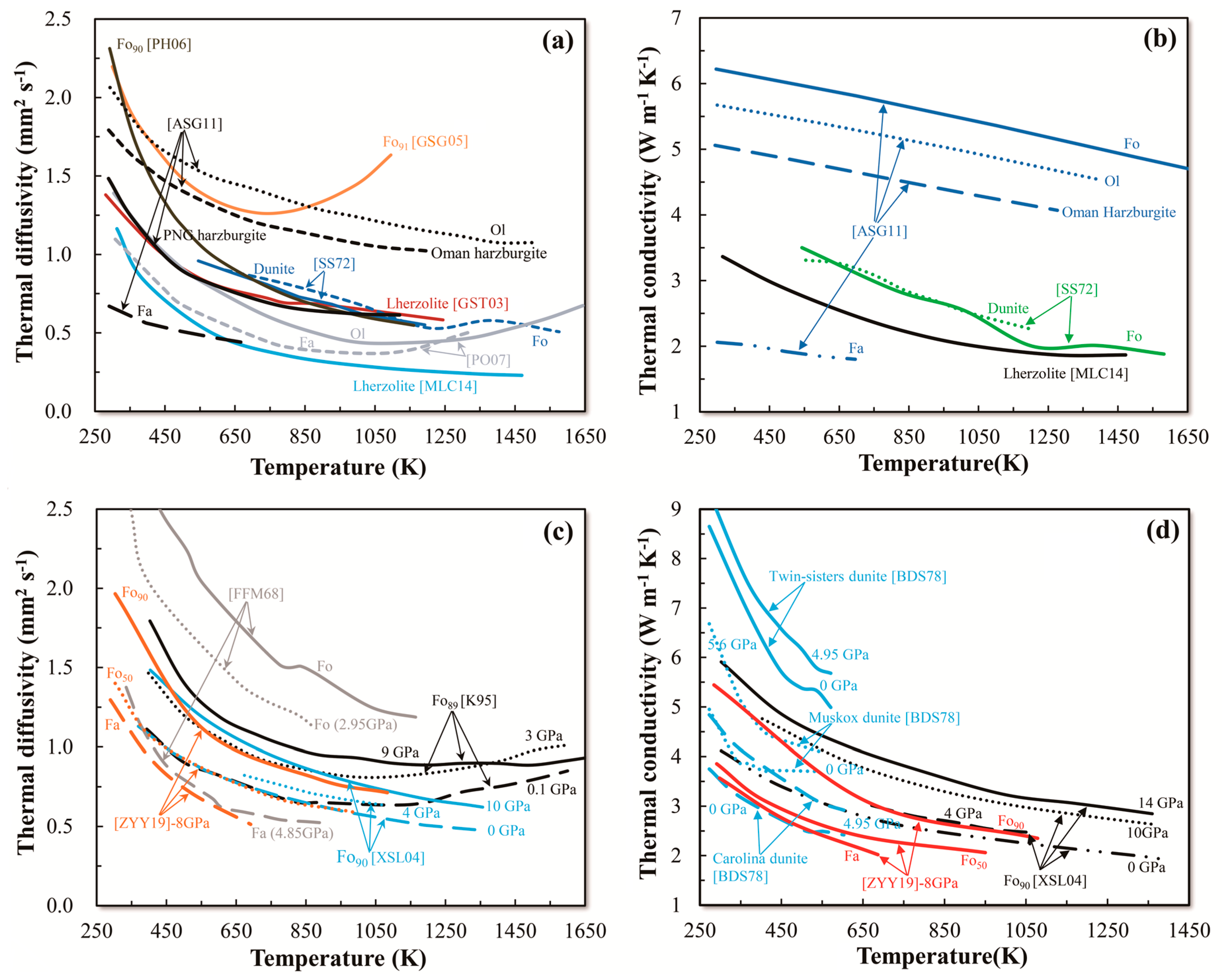
3. Summary of D and κ Data
3.1. Olivine
3.1.1. Single-Crystal Olivine
3.1.2. Polycrystalline Olivine
3.1.3. Pressure Dependence of D and κ in Olivine
3.2. Wadsleyite and Ringwoodite
4. Effect of Fe Content on the D and κ of Olivine
5. Geophysical Applications
6. Concluding Remarks and Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Hofmeister, A.M. Mantle values of thermal conductivity and the geotherm from phonon lifetimes. Science 1999, 283, 1699–1706. [Google Scholar] [CrossRef] [PubMed]
- Dubuffet, F.; Yuen, D.A.; Rainey, E.S.G. Controlling thermal chaos in the mantle by positive feedback from radiative thermal conductivity. Nonlinear Process. Geophys. 2002, 9, 311–323. [Google Scholar] [CrossRef]
- Van Den Berg, A.P.; Yuen, D.A.; Allwardt, J.R. Non-linear effects from variable thermal conductivity and mantle internal heating: Implications for massive melting and secular cooling of the mantle. Phys. Earth Planet. Inter. 2002, 129, 359–375. [Google Scholar] [CrossRef]
- Hofmeister, A.M.; Yuen, D.A. Critical phenomena in thermal conductivity: Implications for lower mantle dynamics. J. Geodyn. 2007, 44, 186–199. [Google Scholar] [CrossRef]
- Michaut, C.; Jaupart, C.; Bell, D.R. Transient geotherms in Archean continental lithosphere: New constraints on thickness and heat production of the subcontinental lithospheric mantle. J. Geophys. Res. 2007, 112, B04408. [Google Scholar] [CrossRef]
- Ringwood, A.E.; Major, A. The system Mg2SiO4-Fe2SiO4 at high pressures and temperatures. Phys. Earth Planet. Inter. 1970, 3, 89–108. [Google Scholar] [CrossRef]
- Schmeling, H.; Monz, R.; Rubie, D.C. The influence of olivine metastability on the dynamics of subduction. Earth Planet. Sci. Lett. 1999, 165, 55–66. [Google Scholar] [CrossRef]
- Emmerson, B.; McKenzie, D. Thermal structure and seismicity of subducting lithosphere. Phys. Earth Planet. Inter. 2007, 163, 191–208. [Google Scholar] [CrossRef]
- Maierová, P.; Chust, T.; Steinle-Neumann, G.; Čadek, O.; Čížková, H. The effect of variable thermal diffusivity on kinematic models of subduction. J. Geophys. Res. 2012, 117, B07202. [Google Scholar] [CrossRef]
- McKenzie, D.; Jackson, J.; Priestley, K. Thermal structure of oceanic and continental lithosphere. Earth Planet. Sci. Lett. 2005, 233, 337–349. [Google Scholar] [CrossRef]
- Hofmeister, A.M.; Branlund, J.M.; Pertermann, M. Properties of rocks and minerals-Thermal conductivity of the Earth. In Treatise on Geophysics; Elsevier: Amsterdam, The Netherlands, 2007; Volume 2, pp. 543–577. [Google Scholar]
- Furlong, K.P.; Chapman, D.S. Heat flow, heat generation, and the thermal state of the lithosphere. Annu. Rev. Earth Planet. Sci. 2013, 41, 385–410. [Google Scholar] [CrossRef]
- Bridgman, P.W. The thermal conductivity and compressibility of several rocks under high pressures. Am. J. Sci. 1924, 38, 81–102. [Google Scholar] [CrossRef]
- Parker, J.W.; Jenkins, J.R.; Butler, P.C.; Abbott, G.I. Flash method of determining thermal diffusivity, heat capacity, and thermal conductivity. J. Appl. Phys. 1961, 32, 1679–1684. [Google Scholar] [CrossRef]
- Cowan, D.R. Pulse method of measuring thermal diffusivity at high temperatures. J. Appl. Phys. 1963, 34, 926–927. [Google Scholar] [CrossRef]
- Heckman, R.C. Finite pulse-time and heat loss effects in pulse thermal diffusivity measurements. J. Appl. Phys. 1973, 44, 1455–1460. [Google Scholar] [CrossRef]
- Azumi, T.; Takahashi, Y. Novel finite pulse-width correction in flash thermal diffusivity measurement. Rev. Sci. Instrum. 1981, 52, 1411–1413. [Google Scholar] [CrossRef]
- Josell, D.; Warren, K.; Czairliyan, A. Correcting an error in Cape and Lehman’s analysis for determining thermal diffusivity from thermal pulse experiments. J. Appl. Phys. 1995, 78, 6867–6869. [Google Scholar] [CrossRef]
- Blumm, J.; Opfermann, J. Improvement of the mathematical modeling of flash measurements. High Temp. High Press. 2002, 34, 515–521. [Google Scholar] [CrossRef]
- Fujisawa, H.; Fujii, N.; Mizutani, H.; Kanamori, H.; Akimoto, S. Thermal diffusivity of Mg2SiO4, Fe2SiO4, and NaCl at high pressures and temperatures. J. Geophys. Res. 1968, 73, 4727–4733. [Google Scholar] [CrossRef]
- Katsura, T. Measurement of thermal diffusivity at high pressures and high temperatures. Proc. Jpn. Acad. B Phys. 1992, 68, 81–86. [Google Scholar] [CrossRef][Green Version]
- Xu, Y.; Shankland, T.J.; Linhardt, S.; Rubie, D.C.; Langenhorst, F.; Klasinski, K. Thermal diffusivity and conductivity of olivine, wadsleyite and ringwoodite to 20 GPa and 1373 K. Phys. Earth Planet. Inter. 2004, 143, 321–336. [Google Scholar] [CrossRef]
- Dzhavadov, L.N. Measurement of thermophysical properties of dielectrics under pressure. High Temp. High Press. 1975, 7, 49–54. [Google Scholar]
- Osako, M.; Ito, E.; Yoneda, A. Simultaneous measurements of thermal conductivity and thermal diffusivity for garnet and olivine under high pressure. Phys. Earth Planet. Inter. 2004, 143, 311–320. [Google Scholar] [CrossRef]
- Yoneda, A.; Yonehara, M.; Osako, M. Anisotropic thermal properties of talc under high temperature and pressure. Phys. Earth Planet. Inter. 2012, 190–191, 10–14. [Google Scholar] [CrossRef]
- Yagi, T.; Ohta, K.; Kobayashi, K.; Taketoshi, N.; Hirose, K.; Baba, T. Thermal diffusivity measurement in a diamond anvil cell using a light pulse thermoreflectance technique. Meas. Sci. Technol. 2011, 22, 24011. [Google Scholar] [CrossRef]
- Hofmeister, A.M.; Pertermann, M.; Branlund, J.M.; Whittington, A.G. Geophysical implications of reduction in thermal conductivity due to hydration. Geophys. Res. Lett. 2006, 33, L11310. [Google Scholar] [CrossRef]
- Hofmeister, A.M.; Dong, J.; Branlund, J.M. Thermal diffusivity of electrical insulators at high temperatures: Evidence for diffusion of bulk phonon-polaritons at infrared frequencies augmenting phonon heat conduction. J. Appl. Phys. 2014, 115, 163517. [Google Scholar] [CrossRef]
- Chang, Y.Y.; Hsieh, W.P.; Tan, E.; Chen, J. Hydration-reduced lattice thermal conductivity of olivine in Earth’s upper mantle. Proc. Natl. Acad. Sci. USA. 2017, 114, 4078–4081. [Google Scholar] [CrossRef]
- Zhang, Y.Y.; Yoshino, T.; Yoneda, A.; Osako, M. Effect of iron content on thermal conductivity of olivine with implications for cooling history of rocky planets. Earth Planet. Sci. Lett. 2019, 519, 109–119. [Google Scholar] [CrossRef]
- Grigull, U.; Sandner, H. Wärmeleitung; Springer: Berlin, Germany, 1979. [Google Scholar]
- Kittel, C. Introduction to Solid State Physics, 8th ed.; Wiley: New York, NY, USA, 2004; p. 704. [Google Scholar]
- Hunt, S.A.; Walker, A.M.; McCormack, R.J.; Dobson, D.P.; Wills, A.S.; Li, L. The effect of pressure on thermal diffusivity in pyroxenes. Mineral. Mag. 2011, 75, 2597–2610. [Google Scholar] [CrossRef]
- Eucken, A. Über die Temperaturabhä ngigkeit der Wä rmeleitfä higkeit fester Nichtmetalle. Ann. Phys. (Leipzig) 1911, 34, 186–221. [Google Scholar]
- Shankland, T.J. Pressure shift of infrared absorption bands in minerals and the effect on radiative heat transport. J. Geophys. Res. 1970, 75, 409–413. [Google Scholar] [CrossRef]
- Reiss, H. Radiative Transfer in Nontransparent, Dispersed Media; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 1988. [Google Scholar]
- Gibert, B.; Schilling, F.R.; Gratz, K.; Tommasi, A. Thermal diffusivity of olivine single crystals and a dunite at high temperature: Evidence for heat transfer by radiation in the upper mantle. Phys. Earth Planet. Inter. 2005, 151, 129–141. [Google Scholar] [CrossRef]
- Katsura, T. Thermal diffusivity of olivine under upper mantle conditions. Geophys. J. Int. 1995, 122, 63–69. [Google Scholar] [CrossRef]
- Clauser, C. Geothermal energy. In Landolt-Börnstein, Group VIII: Advanced Materials and Technologies, Vol. 3: Energy Technologies, Subvol. C: Renewable Energies; Heinloth, K., Ed.; Springer: Heidelberg/Berlin, Germany, 2006; pp. 480–595. [Google Scholar]
- Kobayashi, Y. Anisotropy of thermal diffusivity in olivine, pyroxene and dunite. J. Phys. Earth 1974, 22, 359–373. [Google Scholar] [CrossRef]
- Hofmeister, A.M.; Schilling, F.R. Heat transfer in quartz, orthoclase, and sanidine at elevated temperature. Phys. Chem. Miner. 2002, 29, 571–584. [Google Scholar] [CrossRef]
- Pertermann, M.; Hofmeister, A.M. Thermal diffusivity of olivine-group minerals at high temperature. Am. Mineral. 2006, 91, 1747–1760. [Google Scholar] [CrossRef]
- Gibert, B.; Mainprice, D. Effect of crystal preferred orientations on the thermal diffusivity of quartz polycrystalline aggregates at high temperature. Tectonophysics 2009, 465, 150–163. [Google Scholar] [CrossRef]
- Branlund, J.M.; Hofmeister, A.M. Heat transfer in plagioclase feldspars. Am. Mineral. 2012, 97, 1145–1154. [Google Scholar] [CrossRef]
- Tommasi, A.; Gibert, B.; Seipold, U.; Mainprice, D. Anisotropy of thermal diffusivity in the upper mantle. Nature 2001, 411, 783–786. [Google Scholar] [CrossRef]
- Beck, A.E.; Darbha, D.M.; Schloessin, H.H. Lattice conductivities of single-crystal and polycrystalline materials at mantle pressures and temperatures. Phys. Earth Planet. Inter. 1978, 17, 35–53. [Google Scholar] [CrossRef]
- Yukutake, H.; Shimada, M. Thermal conductivity of NaCl, MgO, coesite and stishovite up to 40 kbar. Phys. Earth Planet. Inter. 1978, 17, 193–200. [Google Scholar] [CrossRef]
- Abdulagatov, I.M.; Emirov, S.N.; Abdulagatova, Z.Z. Effect of pressure and temperature on the thermal conductivity of rocks. J. Chem. Eng. Data. 2006, 51, 22–33. [Google Scholar] [CrossRef]
- Chai, M.; Brown, J.M.; Slutsky, L.J. Thermal diffusivity of mantle minerals. Phys. Chem. Miner. 1996, 23, 470–475. [Google Scholar] [CrossRef]
- Zaug, J.; Abramson, E.H.; Brown, J.M.; Slutsky, L.J. Elastic constants, equations of state and thermal diffusivity at high pressure. In High Pressure Research: Applications to Earth and Planetary Science; Syono, Y., Manghnani, M., Eds.; American Geophysical Union: Washington, DC, USA, 1992; pp. 157–166. [Google Scholar]
- Schilling, F.R. A transient technique to measure thermal diffusivity at elevated temperatures. Eur. J. Mineral. 1999, 11, 1115–1124. [Google Scholar] [CrossRef]
- Katsura, T. Thermal diffusivity of silica glass at pressures up to 9 GPa. Phys. Chem. Miner. 1993, 20, 201–208. [Google Scholar] [CrossRef]
- Katsura, T. Thermal diffusivity of periclase at high temperatures and high pressures. Phys. Earth Planet. Inter. 1997, 101, 73–77. [Google Scholar] [CrossRef]
- Hofmeister, A.M. Thermal diffusivity of garnets at high temperature. Phys. Chem. Miner. 2006, 33, 45–62. [Google Scholar] [CrossRef]
- Braeuer, H.; Dusza, L.; Schulz, B. New laser flash equipment LFA 427. Interceram. 1992, 41, 489–492. [Google Scholar]
- Gibert, B.; Seipold, U.; Tommasi, A.; Mainprice, D. Thermal diffusivity of upper mantle rocks: Influence of temperature, pressure, and the deformation fabric. J. Geophys. Res. 2003, 108, 2359. [Google Scholar] [CrossRef]
- Seipold, U. Simultaneous measurements of thermal diffusivity and thermal conductivity under high pressure using thermal pulse of finite length. High Temp. High Press. 1988, 20, 609–613. [Google Scholar]
- Schatz, J.F.; Simmons, G. Method of simultaneous measurement of radiative and lattice thermal conductivity. J. Appl. Phys. 1972, 43, 2586–2594. [Google Scholar] [CrossRef]
- Abramson, E.H.; Brown, J.M.; Slutsky, L.J. The thermal diffusivity of water at high pressures and temperatures. J. Chem. Phys. 2001, 115, 10461–10463. [Google Scholar] [CrossRef]
- Cahill, D.; Watson, S.K.; Pohl, R.O. Lower limit of thermal conductivity of disordered solids. Phys. Rev. B 1992, 46, 6131–6140. [Google Scholar] [CrossRef] [PubMed]
- Kanamori, H.; Fujii, N.; Mizutani, H. Thermal diffusivity measurement of rock-forming minerals from 300 to 1100 K. J. Geophys. Res. 1968, 73, 595–605. [Google Scholar] [CrossRef]
- Schatz, J.F.; Simmons, G. Thermal conductivity of Earth materials at high temperatures. J. Geophys. Res. 1972, 77, 6966–6983. [Google Scholar] [CrossRef]
- Kanamori, H.; Mizutani, H.; Fujii, N. Method of thermal diffusivity measurement. J. Phys. Earth. 1969, 17, 43–53. [Google Scholar] [CrossRef]
- Schärmeli, G.H. Anisotropy of olivine thermal conductivity at 2.5 GPa up to 1500 K measured on optically non-thick sample. In High-Pressure Researches in Geoscience; Schreyer, W., Ed.; Schweizerbart: Stuttgart, Germany, 1982; pp. 349–373. [Google Scholar]
- Petrunin, G.I.; Orlik, E.V. Thermal diffusivity of mantle (pyrolite) minerals at temperatures between room temperature and melting point (300–1700 K). Mosc. Univ. Phys. Bull. 2007, 62, 388–392. [Google Scholar] [CrossRef]
- Miao, S.; Li, H.; Chen, G. The temperature dependence of thermal conductivity for lherzolites from the North China Craton and the associated constraints on the thermodynamic thickness of the lithosphere. Geophys. J. Int. 2014, 197, 900–909. [Google Scholar] [CrossRef]
- Arafin, S.; Singh, R.N.; George, A.K. Thermal Diffusivity and Heat of Formation of Harzburgite and Its Major Constituent Minerals. Int. J. Thermophys. 2011, 32, 2139–2147. [Google Scholar] [CrossRef]
- Horai, K.; Simmons, G. Thermal conductivity of rock-forming minerals. Earth Planet. Sci. Lett. 1969, 6, 359–368. [Google Scholar] [CrossRef]
- Padture, N.; Klemens, P. Low thermal conductivity in garnets. J. Am. Ceram. Soc. 1997, 80, 1018–1020. [Google Scholar] [CrossRef]
- Green, H.W., II; Houston, H. The mechanics of deep earthquakes. Ann. Rev. Earth Planet. Sci. 1995, 23, 169–213. [Google Scholar] [CrossRef]
- Wiens, D.A. Seismological constraints on the mechanism of deep earthquakes: Temperature dependence of deep earthquake source properties. Phys. Earth Planet. Inter. 2001, 127, 145–163. [Google Scholar] [CrossRef]
- Marton, F.C.; Shankland, T.J.; Rubie, D.C.; Xu, Y. Effects of variable thermal conductivity on the mineralogy of subducting slabs and implications for mechanisms of deep earthquakes. Phys. Earth Planet. Inter. 2005, 149, 53–64. [Google Scholar] [CrossRef]
- Chapman, D.S. Thermal gradients in the continental crust. Geol. Soc. Spec. Publ. 1986, 24, 63–70. [Google Scholar] [CrossRef]
- Grant, K.; Brooker, R.A.; Kohn, S.C.; Wood, B. The effect of oxygen fugacity on hydroxyl concentrations and speciation in olivine: Implications for water solubility in the upper mantle. Earth Planet. Sci. Lett. 2007, 261, 217–229. [Google Scholar] [CrossRef]
- Kolzenburg, S.; Genova, D.D.; Giordano, D.; Hess, K.U.; Dingwell, D.B. The effect of oxygen fugacity on the rheological evolution of crystallizing basaltic melts. Earth Planet. Sci. Lett. 2018, 487, 21–32. [Google Scholar] [CrossRef]




| Method | The Maximum T and P | Advantages | Disadvantages | References |
|---|---|---|---|---|
| An Ångström method | 1700 K, 20 GPa | Simultaneously obtains D or κ. | Contact thermal resistance; photon radiation may exist at high temperatures. | [20,22,52] |
| A pulse heating method | 1100 K, 8.3 GPa | Simultaneously obtains D or κ. | Contact thermal resistance; photon radiation may exist at high temperatures. | [23,24] |
| A transient method | 1200 K | Allows the separation of non-conductive radiative processes and purely diffusive mechanisms. | Only get the D; large sample sizes; thermal cracking; includes spurious radiative transfer and contact resistance effects. | [56,57] |
| A laser-wave method | 1900 K | Simultaneous measurement of κradiative and κlattice; contact-free for measurement. | Has not been applied to high pressure; thermal cracking. | [58] |
| Picosecond transient grating spectroscopy method | 40 GPa | Contact-free for measurement; small experimental error; separation of lattice and radiative contributions to transport. | Only get the D; higher technical requirement; application in DAC is not mature enough. | [49,59] |
| The laser-flash method | 2200 K | Avoids photon radiation; the most accurate method so far. | Only get the D; has not yet been applied to high pressure; thermal cracking. | [55] |
| Steady-state radial heat flow method | 650 K, 5.6 GPa | Simultaneously obtains D or κ. | Large contact thermal resistance; change in spacing between two thermocouples. | [46] |
| 3ω method | 30–300 K; 0.8 GPa | Insensitive to errors from black-body radiation. | Involves a single contact; Earth materials are less measured. | [60] |
| The light pulse thermoreflectance method | 144 GPa | Accurately obtains D at high pressure. | Only get D; has not yet been applied to high temperature. | [26] |
| Sample | T (K) | P (GPa) | Test Method | Fitting Formula | Reference |
|---|---|---|---|---|---|
| Olivine | |||||
| Fo and Fa | 400–1300 | 2.4–5.0 | An Ångström method | D or κ = a + b/T + c/T2 + d/T3 | [20] |
| Single-crystal olivine | 300–1100 | 1 atm | An Ångström method | D or κ = a + b/T + c/T2 + d/T3 | [61] |
| Single-crystal olivine | 500–1900 | 1 atm | An laser-wave method | D or κ = a + b/T + c/T2 + d/T3 | [62] |
| Single-crystal olivine | 1250 | 1 atm | A modified Ångström method | D or κ = a + b/T + c/T2 + d/T3 | [40] |
| Single-crystal olivine | 350–650 | 0–5.6 | Steady-state radial heat flow method | κ = a + b/T + c/T2 + d/T3 | [46] |
| Dunite | 350–650 | 0–5.6 | Steady-state radial heat flow method | κ = a + b/T + c/T2 + d/T3 | [46] |
| Fo89 | 1700 | 9.0 | An Ångström method | D or κ = a + b/T + c/T2 + d/T3 | [38] |
| Peridotite | 1250 | 1.0 | A transient method | D = a + b/T + c/T2 + d/T3 | [52] |
| Single-crystal olivine | 1100 | 8.3 | A pulse heating method | D or κ = a + b/T | [24] |
| Fo90 | 1373 | 20 | An Ångström method | D = D298 (298/T)n × (1 + a × P) κ = κ298 (298/T)n × (1 + a × P) | [22] |
| Single-crystal olivine | 1123 | 1 atm | A transient method | D = a + b/T + c/T2 | [37] |
| Fo91 | 1123 | 1 atm | A transient method | D = a + b/T + c/T2 | [37] |
| Single-crystal olivine | 1500 | 1 atm | The laser-flash method | D = a + b/T + c/T2 | [42] |
| Fo90 | 1500 | 1 atm | The laser-flash method | D = a + b/T + c/T2 | [42] |
| Single-crystal olivine | 1100 | 1 atm | The temperature wave method | D–1 = a + b × T | [65] |
| Fo and Fa | 1700 | 1 atm | The temperature wave method | D–1 = a + b × T | [65] |
| Olivine | 1700 | 1 atm | The temperature wave method | D–1 = a + b × T | [65] |
| Lherzolite | 1473 | 1 atm | The laser-flash method | D or κ = a + b/T + c/T2 | [66] |
| Olivine | 1500 | 1 atm | A theoretical formulation | D = D0 exp (–ΔHD/R × T) κ = 0.0164 × ӨD – 6.292 | [67] |
| Harzburgite | 1300 | 1 atm | A theoretical formulation | D = D0 exp (–ΔHD/R × T) κ = 0.0164 × ӨD – 6.292 | [67] |
| Fo | 1700 | 1 atm | A theoretical formulation | D = D0 exp (–ΔHD/R × T) κ = 0.0164 × ӨD – 6.292 | [67] |
| Fa | 700 | 1 atm | A theoretical formulation | D = D0 exp (–ΔHD/R × T) κ = 0.0164 × ӨD – 6.292 | [67] |
| Fo, Fo90, Fo70, Fo31, Fa | 1100 | 10 | A pulse heating method | D = D298 (298/T)n × (1 + a × P) κ = κ298 (298/T)n × (1 + a × P) | [30] |
| Wadsleyite | |||||
| (Mg0.9Fe0.1)2SiO4 | 1373 | 14 | An Ångström method | D = D298 (298/T)n × (1 + a × P) κ = κ298 (298/T)n × (1 + a × P) | [22] |
| Ringwoodite | |||||
| (Mg0.9Fe0.1)2SiO4 | 1373 | 20 | An Ångström method | D = D298 (298/T)n × (1 + a × P) κ = κ298 (298/T)n × (1 + a × P) | [22] |
| Sample | T (K) | P (GPa) | Fitting Formula | Pressure Coefficient dlnD/dP or dlnκ/dP (GPa−1) | Reference |
|---|---|---|---|---|---|
| dlnD/dP | |||||
| Fo93 [100] | 293 | 0–8.3 | D = 2.5 × exp (0.033 × P) | 0.033 | [24] |
| Fo93 [010] | 293 | 0–8.3 | D = 1.53 × exp (0.04 × P) | 0.040 | [24] |
| Fo93 [001] | 293 | 0–8.3 | D = 2.16 × exp (0.035 × P) | 0.035 | [24] |
| Fo68 | 700 | 2.4–5.0 | D = 0.1563 × P + 0.966 | 0.162 | [20] |
| Fo68 | 1100 | 2.4–5.0 | D = 0.087 × P + 0.6742 | 0.129 | [20] |
| Fo89 | 400 | 0–9.0 | D = 0.052 × P + 1.1473 | 0.045 | [38] |
| Fo89 | 600 | 0–9.0 | D = 0.0315 × P + 0.8406 | 0.037 | [38] |
| Fo90 | 298–1300 | 0–20 | D = D298 (298/T)n (1 + a × P) | 0.036 | [22] |
| Fo | 298 | 0–10 | D = D298 (298/T)n (1 + a × P) | 0.040 | [30] |
| Fo90 | 298 | 0–10 | D = D298 (298/T)n (1 + a × P) | 0.024 | [30] |
| Fo50 | 298 | 0–10 | D = D298 (298/T)n (1 + a × P) | 0.016 | [30] |
| Fa | 298 | 0–10 | D = D298 (298/T)n (1 + a × P) | 0.025 | [30] |
| dlnκ/dP | |||||
| Fo93 [100] | 293 | 0–8.3 | κ = 6.61 × exp (0.038 × P) | 0.038 | [24] |
| Fo93 [010] | 293 | 0–8.3 | κ = 3.98 × exp (0.042 × P) | 0.042 | [24] |
| Fo93 [001] | 293 | 0–8.3 | κ = 5.91 × exp (0.034 × P) | 0.034 | [24] |
| Single-crystal olivine | 273 | 0–4.95 | κ = 0.7512 × P + 9.4932 | 0.079 | [46] |
| Single-crystal olivine | 538 | 0–4.95 | κ = 0.3209 × P + 6.6862 | 0.048 | [46] |
| Twin Sisters dunite | 273 | 0–4.95 | κ = 1.0529 × P + 6.2453 | 0.168 | [46] |
| Twin Sisters dunite | 515 | 0–4.95 | κ = 0.3022 × P + 4.3789 | 0.069 | [46] |
| Carolina dunite | 273 | 0–4.95 | κ = 0.1635 × P + 3.6785 | 0.044 | [46] |
| Carolina dunite | 572 | 0–4.95 | κ = 0.092 × P + 2.4294 | 0.037 | [46] |
| Muskox dunite | 273 | 0–5.6 | κ = 0.2765 × P + 4.8771 | 0.056 | [46] |
| Muskox dunite | 544 | 0–5.6 | κ = 0.0455 × P + 3.7220 | 0.012 | [46] |
| Fo90 | 298–1300 | 0–20 | κ = κ298 (298/T)n (1 + a × P) | 0.032 | [22] |
| Fo | 298 | 0–10 | κ = κ298 (298/T)n (1 + a × P) | 0.036 | [30] |
| Fo90 | 298 | 0–10 | κ = κ298 (298/T)n (1 + a × P) | 0.021 | [30] |
| Fo50 | 298 | 0–10 | κ = κ298 (298/T)n (1 + a × P) | 0.026 | [30] |
| Fa | 298 | 0–10 | κ = κ298 (298/T)n (1 + a × P) | 0.018 | [30] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xiong, Z.; Zhang, B. Thermal Transport Properties of Olivine, Wadsleyite, and Ringwoodite—A Review. Minerals 2019, 9, 519. https://doi.org/10.3390/min9090519
Xiong Z, Zhang B. Thermal Transport Properties of Olivine, Wadsleyite, and Ringwoodite—A Review. Minerals. 2019; 9(9):519. https://doi.org/10.3390/min9090519
Chicago/Turabian StyleXiong, Zili, and Baohua Zhang. 2019. "Thermal Transport Properties of Olivine, Wadsleyite, and Ringwoodite—A Review" Minerals 9, no. 9: 519. https://doi.org/10.3390/min9090519
APA StyleXiong, Z., & Zhang, B. (2019). Thermal Transport Properties of Olivine, Wadsleyite, and Ringwoodite—A Review. Minerals, 9(9), 519. https://doi.org/10.3390/min9090519




