Equivocal PI-RADS Three Lesions on Prostate Magnetic Resonance Imaging: Risk Stratification Strategies to Avoid MRI-Targeted Biopsies
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
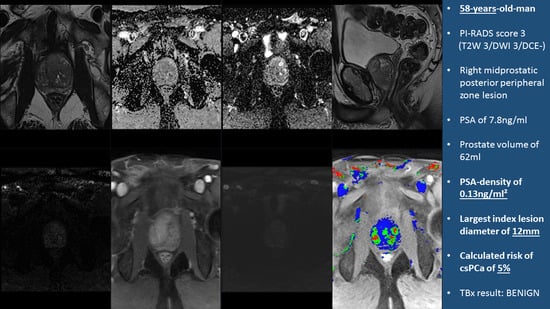
2.2. Multiparametric MRI
2.3. Prostate Biopsy
2.4. Pathological Review of Biopsy Specimens
2.5. Study Endpoints
- I:
- ISUP grade ≥2 PCa (currently the most used, = primary outcome);
- II:
- ISUP grade ≥2 with CR and/or IDC PCa;
- III:
- ISUP grade ≥3 PCa.
- -
- patient level = a complete TBx session avoided per patient;
- -
- lesion level = a TBx procedure avoided per lesion (= Supplementary Material).
- -
- PCa detection in TBx specimens of men with equivocal MRI results (PI-RADS score 3);
- -
- results in terms of avoiding TBx sessions and the detection of low-risk PCa of a risk stratification strategy based on the largest index lesion diameter accepting missing ≤5% of csPCa diagnoses (definition I).
- -
- results in terms of avoiding TBx sessions and the detection of low-risk PCa of a risk stratification strategy based on PSA-D accepting missing ≤5% of csPCa diagnoses (definition I);
- -
- performance and clinical utility of a multivariable prediction model including age, largest index lesion diameter, and PSA-D for TBx decision management in men with equivocal MRI results (PI-RADS score 3).
2.6. Statistical Analysis
3. Results
3.1. Cohort Characteristics and Prostate Cancer Detection
3.2. Risk stratification for TBx Decision Based on Largest Index Lesion Diameter
3.3. Risk Stratification for TBx Decision Based on PSA-Density
3.4. Multivariable Risk Prediction for TBx Decision
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ADC | apparent diffusion coefficient |
| AUC | area under the receiver operation curve |
| AS | active surveillance |
| CR | cribriform growth pattern |
| csPCa | clinically significant prostate cancer |
| DCE | dynamic contrast enhanced |
| DRE | digital rectal examination |
| DWI | diffusion weighted imaging |
| G | ISUP grade |
| GS | Gleason score |
| IDC | intraductal carcinoma |
| IQR | interquartile range |
| ISUP | International Society of Urological Pathology |
| mpMRI | multiparametric MRI |
| MRI | magnetic resonance imaging |
| MRI ± TBx | MRI with or without TBx |
| PCa | prostate cancer |
| PI-RADS | Prostate Imaging—Reporting and Data System |
| PSA | prostate-specific antigen |
| PSA-D | PSA-density |
| SBx | systematic prostate biopsy |
| T2W | T2-weighted |
| TBx | targeted biopsy |
| TRUS | transrectal ultrasound |
References
- Weinreb, J.; Barentsz, J.O.; Choyke, P.L.; Cornud, F.; Haider, M.A.; Macura, K.J.; A Margolis, D.J.; Schnall, M.D.; Shtern, F.; Tempany, C.M.; et al. PI-RADS Prostate Imaging–Reporting and Data System: 2015, Version 2. Eur. Urol. 2016, 69, 16–40. [Google Scholar] [CrossRef] [PubMed]
- Padhani, A.R.; Barentsz, J.; Villeirs, G.; Rosenkrantz, A.B.; Margolis, D.J.; Turkbey, B.; Thoeny, H.C.; Cornud, F.; Haider, M.A.; Macura, K.J.; et al. PI-RADS Steering Committee: The PI-RADS Multiparametric MRI and MRI-directed Biopsy Pathway. Radiology 2019, 292, 464–474. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.T.; Mehta, K.A.; Turkbey, B.; Verma, S. PI-RADS: Past, present, and future. J. Magn. Reson. Imaging 2019, 52, 33–53. [Google Scholar] [CrossRef] [PubMed]
- Barentsz, J.O.; Richenberg, J.; Clements, R.; Choyke, P.; Verma, S.; Villeirs, G.; Rouviere, O.; Logager, V.; Fütterer, J.J. ESUR prostate MR guidelines 2012. Eur. Radiol. 2012, 22, 746–757. [Google Scholar] [CrossRef]
- Remmers, S.; Roobol, M.J. Personalized strategies in population screening for prostate cancer. Int. J. Cancer 2020, 147, 2977–2987. [Google Scholar] [CrossRef]
- Schoots, I.G. MRI in early prostate cancer detection: How to manage indeterminate or equivocal PI-RADS 3 lesions? Transl. Androl. Urol. 2018, 7, 70–82. [Google Scholar] [CrossRef]
- Maggi, M.; Panebianco, V.; Mosca, A.; Salciccia, S.; Gentilucci, A.; Di Pierro, G.; Busetto, G.M.; Barchetti, G.; Campa, R.; Sperduti, I.; et al. Prostate Imaging Reporting and Data System 3 Category Cases at Multiparametric Magnetic Resonance for Prostate Cancer: A Systematic Review and Meta-analysis. Eur. Urol. Focus 2020, 6, 463–478. [Google Scholar] [CrossRef]
- Quint, L.E.; Van Erp, J.S.; Bland, P.H.; A Del Buono, E.; Mandell, S.H.; Grossman, H.B.; Gikas, P.W. Prostate cancer: Correlation of MR images with tissue optical density at pathologic examination. Radiology 1991, 179, 837–842. [Google Scholar] [CrossRef]
- Shukla-Dave, A.; Hricak, H.; Eberhardt, S.C.; Olgac, S.; Muruganandham, M.; Scardino, P.T.; Reuter, V.E.; Koutcher, J.A.; Zakian, K.L. Chronic Prostatitis: MR Imaging and1H MR Spectroscopic Imaging Findings—Initial Observations. Radiology 2004, 231, 717–724. [Google Scholar] [CrossRef]
- Langer, D.L.; Van Der Kwast, T.H.; Evans, A.J.; Sun, L.; Yaffe, M.J.; Trachtenberg, J.; Haider, M.A. Intermixed Normal Tissue within Prostate Cancer: Effect on MR Imaging Measurements of Apparent Diffusion Coefficient and T2—Sparse versus Dense Cancers. Radiology 2008, 249, 900–908. [Google Scholar] [CrossRef]
- Ullrich, T.; Quentin, M.; Arsov, C.; Schmaltz, A.K.; Tschischka, A.; Laqua, N.; Hiester, A.; Blondin, D.; Rabenalt, R.; Albers, P.; et al. Risk Stratification of Equivocal Lesions on Multiparametric Magnetic Resonance Imaging of the Prostate. J. Urol. 2018, 199, 691–698. [Google Scholar] [CrossRef]
- Felker, E.R.; Raman, S.S.; Margolis, D.J.; Lu, D.S.K.; Shaheen, N.; Natarajan, S.; Sharma, D.; Huang, J.; Dorey, F.; Marks, L.S. Risk Stratification Among Men With Prostate Imaging Reporting and Data System version 2 Category 3 Transition Zone Lesions: Is Biopsy Always Necessary? Am. J. Roentgenol. 2017, 209, 1272–1277. [Google Scholar] [CrossRef]
- Washino, S.; Okochi, T.; Saito, K.; Konishi, T.; Hirai, M.; Kobayashi, Y.; Miyagawa, T. Combination of prostate imaging reporting and data system (PI-RADS) score and prostate-specific antigen (PSA) density predicts biopsy outcome in prostate biopsy naïve patients. BJU Int. 2017, 119, 225–233. [Google Scholar] [CrossRef]
- Zalesky, M.; Stejskal, J.; Adamcova, V.; Hrbáček, J.; Minarik, I.; Pavlicko, A.; Votrubova, J.; Babjuk, M.; Zachoval, R. Use of Prostate Specific Antigen Density Combined with Multiparametric Magnetic Resonance Imaging Improves Triage for Prostate Biopsy. Urol. Int. 2019, 103, 33–40. [Google Scholar] [CrossRef]
- Schoots, I.G.; Osses, D.F.; Drost, F.-J.H.; Verbeek, J.F.M.; Remmers, S.; Van Leenders, G.J.L.H.; Bangma, C.H.; Roobol, M.J. Reduction of MRI-targeted biopsies in men with low-risk prostate cancer on active surveillance by stratifying to PI-RADS and PSA-density, with different thresholds for significant disease. Transl. Androl. Urol. 2018, 7, 132–144. [Google Scholar] [CrossRef]
- Hansen, N.L.; Kesch, C.; Barrett, T.; Koo, B.; Radtke, J.P.; Bonekamp, D.; Schlemmer, H.-P.; Warren, A.Y.; Wieczorek, K.; Hohenfellner, M.; et al. Multicentre evaluation of targeted and systematic biopsies using magnetic resonance and ultrasound image-fusion guided transperineal prostate biopsy in patients with a previous negative biopsy. BJU Int. 2017, 120, 631–638. [Google Scholar] [CrossRef]
- Görtz, M.; Radtke, J.P.; Hatiboglu, G.; Schütz, V.; Tosev, G.; Güttlein, M.; Leichsenring, J.; Stenzinger, A.; Bonekamp, D.; Schlemmer, H.-P.; et al. The Value of Prostate-specific Antigen Density for Prostate Imaging-Reporting and Data System 3 Lesions on Multiparametric Magnetic Resonance Imaging: A Strategy to Avoid Unnecessary Prostate Biopsies. Eur. Urol. Focus 2019. [Google Scholar] [CrossRef] [PubMed]
- Rais-Bahrami, S.; Turkbey, B.; Rastinehad, A.R.; Walton-Diaz, A.; Hoang, A.N.; Siddiqui, M.M.; Stamatakis, L.; Truong, H.; Nix, J.W.; Vourganti, S.; et al. Natural history of small index lesions suspicious for prostate cancer on multiparametric MRI: Recommendations for interval imaging follow-up. Diagn. Interv. Radiol. 2014, 20, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Scialpi, M.; Martorana, E.; Aisa, M.C.; Rondoni, V.; D’Andrea, A.; Bianchi, G. Score 3 prostate lesions: A gray zone for PI-RADS v2. Türk Üroloji Derg./Turk. J. Urol. 2017, 43, 237–240. [Google Scholar] [CrossRef]
- Rosenkrantz, A.B.; Babb, J.S.; Taneja, S.S.; Ream, J.M. Proposed Adjustments to PI-RADS Version 2 Decision Rules: Impact on Prostate Cancer Detection. Radiology 2017, 283, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Martorana, E.; Pirola, G.M.; Scialpi, M.; Micali, S.; Iseppi, A.; Reggiani-Bonetti, L.; Kaleci, S.; Torricelli, P.; Bianchi, G. Lesion volume predicts prostate cancer risk and aggressiveness: Validation of its value alone and matched with prostate imaging reporting and data system score. BJU Int. 2017, 120, 92–103. [Google Scholar] [CrossRef]
- Epstein, J.I.; Egevad, L.; Amin, M.B.; Delahunt, B.; Srigley, J.R.; Humphrey, P.A. The 2014 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason Grading of Prostatic Carcinoma. Am. J. Surg. Pathol. 2016, 40, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Alberts, A.R.; Roobol, M.J.; Verbeek, J.F.; Schoots, I.G.; Chiu, P.K.-F.; Osses, D.F.; Tijsterman, J.D.; Beerlage, H.P.; Mannaerts, C.K.; Schimmöller, L.; et al. Prediction of High-grade Prostate Cancer Following Multiparametric Magnetic Resonance Imaging: Improving the Rotterdam European Randomized Study of Screening for Prostate Cancer Risk Calculators. Eur. Urol. 2019, 75, 310–318. [Google Scholar] [CrossRef] [PubMed]
- Van Hemelrijck, M.; Ji, X.; Helleman, J.; Roobol, M.J.; Van Der Linden, W.; Nieboer, D.; Bangma, C.H.; Frydenberg, M.; Rannikko, A.; Lee, L.S.; et al. Reasons for Discontinuing Active Surveillance: Assessment of 21 Centres in 12 Countries in the Movember GAP3 Consortium. Eur. Urol. 2019, 75, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Vickers, A.; Sjoberg, D.D. Guidelines for Reporting of Statistics in European Urology. Eur. Urol. 2015, 67, 181–187. [Google Scholar] [CrossRef]
- Park, S.Y.; Park, B.K. Necessity of differentiating small (<10 mm) and large (≥10 mm) PI-RADS 4. World J. Urol. 2020, 38, 1473–1479. [Google Scholar] [PubMed]
- Vargas, H.A.; Hotker, A.M.; A Goldman, D.; Moskowitz, C.S.; Gondo, T.; Matsumoto, K.; Ehdaie, B.; Woo, S.; Fine, S.W.; E Reuter, V.; et al. Updated prostate imaging reporting and data system (PIRADS v2) recommendations for the detection of clinically significant prostate cancer using multiparametric MRI: Critical evaluation using whole-mount pathology as standard of reference. Eur. Radiol. 2016, 26, 1606–1612. [Google Scholar] [CrossRef]
- Venderink, W.; Van Luijtelaar, A.; Bomers, J.G.; Van Der Leest, M.; De Kaa, C.H.-V.; Barentsz, J.O.; Sedelaar, J.M.; Fütterer, J.J. Results of Targeted Biopsy in Men with Magnetic Resonance Imaging Lesions Classified Equivocal, Likely or Highly Likely to Be Clinically Significant Prostate Cancer. Eur. Urol. 2018, 73, 353–360. [Google Scholar] [CrossRef]
- Di Trapani, E.; Musi, G.; Ferro, M.; Cordima, G.; Mistretta, F.A.; Luzzago, S.; Bianchi, R.; Cozzi, G.; Alessi, S.; Catellani, M.; et al. Clinical evaluation and disease management of PI-RADS 3 lesions. Analysis from a single tertiary high-volume center. Scand. J. Urol. 2020, 54, 1–5. [Google Scholar] [CrossRef]
- Zlotta, A.R.; Egawa, S.; Pushkar, D.; Govorov, A.; Kimura, T.; Kido, M.; Takahashi, H.; Kuk, C.; Kovylina, M.; Aldaoud, N.; et al. Prevalence of Prostate Cancer on Autopsy: Cross-Sectional Study on Unscreened Caucasian and Asian Men. J. Natl. Cancer Inst. 2013, 105, 1050–1058. [Google Scholar] [CrossRef]
- Kweldam, C.F.; Kümmerlin, I.P.; Nieboer, D.; I Verhoef, E.; Steyerberg, E.W.; Van Der Kwast, T.H.; Roobol, M.J.; Van Leenders, G.J. Disease-specific survival of patients with invasive cribriform and intraductal prostate cancer at diagnostic biopsy. Mod. Pathol. 2016, 29, 630–636. [Google Scholar] [CrossRef] [PubMed]
- Wilt, T.J.; Jones, K.M.; Barry, M.J.; Andriole, G.L.; Culkin, D.; Wheeler, T. Faculty Opinions recommendation of Follow-up of Prostatectomy versus Observation for Early Prostate Cancer. Fac. Opin. Post-Publ. Peer Rev. Biomed. Lit. 2017, 377, 132–142. [Google Scholar]
- Boesen, L.; Nørgaard, N.; Løgager, V.B.; Balslev, I.; Bisbjerg, R.; Thestrup, K.-C.; Jakobsen, H.; Thomsen, H.S. Prebiopsy Biparametric Magnetic Resonance Imaging Combined with Prostate-specific Antigen Density in Detecting and Ruling out Gleason 7–10 Prostate Cancer in Biopsy-naïve Men. Eur. Urol. Oncol. 2019, 2, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Schaudinn, A.; Gawlitza, J.; Mucha, S.; Linder, N.; Franz, T.; Horn, L.-C.; Kahn, T.; Busse, H. Comparison of PI-RADS v1 and v2 for multiparametric MRI detection of prostate cancer with whole-mount histological workup as reference standard. Eur. J. Radiol. 2019, 116, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Becker, A.; Cornelius, A.; Reiner, C.S.; Stocker, D.; Ulbrich, E.J.; Barth, B.K.; Mortezavi, A.; Eberli, D.; Donati, O.F. Direct comparison of PI-RADS version 2 and version 1 regarding interreader agreement and diagnostic accuracy for the detection of clinically significant prostate cancer. Eur. J. Radiol. 2017, 94, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Polanec, S.; Helbich, T.H.; Bickel, H.; Pinker-Domenig, K.; Georg, D.; Shariat, S.F.; Aulitzky, W.; Susani, M.; Baltzer, P.A.T. Head-to-head comparison of PI-RADS v2 and PI-RADS v1. Eur. J. Radiol. 2016, 85, 1125–1131. [Google Scholar] [CrossRef] [PubMed]
- Krishna, S.; Lim, C.S.; McInnes, M.D.F.; Flood, T.A.; Shabana, W.; Lim, R.S.; Schieda, N.; Bmbs, C.S.L.; Mbbs, R.S.L. Evaluation of MRI for diagnosis of extraprostatic extension in prostate cancer. J. Magn. Reson. Imaging 2018, 47, 176–185. [Google Scholar] [CrossRef]
- Kan, Y.; Zhang, Q.; Hao, J.; Wang, W.; Zhuang, J.; Gao, J. Clinico-radiological characteristic-based machine learning in reducing unnecessary prostate biopsies of PI-RADS 3 lesions with dual validation. Eur Radiol. 2020, 30, 6274–6284. [Google Scholar] [CrossRef]


| Total Cohort (n = 292) | Düsseldorf (n = 154) | Rotterdam (n = 138) | p Value | |
|---|---|---|---|---|
| Age (yr), median (IQR) | 64 (58–69) | 61 (53–67) | 67 (61–72) | <0.001 |
| Follow-up time (yr), median (IQR) | 2 (1–3) | 3 (2–4) | 2 (1–3) | 0.083 |
| PSA level (ng/mL), median (IQR) | 8.1 (6–12.2) | 7.3 (5.5–11) | 9.5 (6.6–13.1) | 0.001 |
| Prostate volume on MRI (mL), median (IQR) | 52.3 (36–78.8) | 58 (44.8–83.3) | 46 (31–68.5) | <0.001 |
| PSA-density (ng/mL/mL), median (IQR) | 0.16 (0.11–0.25) | 0.13 (0.1–0.18) | 0.23 (0.13–0.31) | <0.001 |
| Indication of prostate MRI, no. (%) | ||||
| Initial PCa diagnosis | 69 (24) | 64 (42) | 5 (4) | <0.001 |
| Previous negative biopsy | 144 (49) | 82 (53) | 62 (45) | |
| Active Surveillance | 79 (27) | 8 (5) | 71 (51) | |
| DRE findings, no. (%) | ||||
| Benign | 191 (65) | 75 (49) | 116 (84) | <0.001 |
| Suspected | 60 (21) | 38 (25) | 22 (16) | |
| Unknown | 41 (14) | 41 (27) | 0 (0) | |
| PI-RADS 3 lesions on MRI, no. (%) | ||||
| 1 | 132 (45) | 16 (10) | 116 (84) | <0.001 |
| 2 | 89 (31) | 69 (45) | 20 (15) | |
| 3 | 69 (24) | 67 (44) | 2 (1) | |
| 4 | 2 (1) | 2 (1) | 0 (0) | |
| Highest grade at TBx, no. (%) | ||||
| no PCa | 200 (69) | 141 (92) | 59 (43) | <0.001 |
| G 1 | 53 (18) | 9 (6) | 44 (32) | |
| G 2 | 19 (7) | 2 (1) | 17 (12) | |
| G 2 with CR and/or IDC | 10 (3) | 0 (0) | 10 (7) | |
| G 3 | 9 (3) | 2 (1) | 7 (5) | |
| G 4–5 | 1 (1) | 0 (0) | 1 (1) |
| Total PI-RADS 3 Lesions (n = 525) | PI-RADS 3 Lesions Düsseldorf (n = 363) | PI-RADS 3 Lesions Rotterdam (n = 162) | p Value | |
|---|---|---|---|---|
| Lesion size (largest diameter, mm), median (IQR) | 11 (9–13) | 12 (10–13) | 10 (7–12) | <0.001 |
| Lesion zone, no. (%) | ||||
| Peripheral zone | 186 (35) | 107 (30) | 79 (49) | <0.001 |
| Transition zone | 324 (62) | 246 (68) | 78 (48) | |
| Central zone | 2 (1) | 0 (0) | 2 (1) | |
| >1 zone | 13 (3) | 10 (3) | 3 (2) | |
| Lesion location, no. (%) | ||||
| Anterior | 340 (65) | 258 (71) | 82 (51) | <0.001 |
| Posterior | 178 (34) | 103 (28) | 75 (46) | |
| Both | 7 (1) | 2 (1) | 5 (3) | |
| Grade at TBx, no. (%) | ||||
| no PCa | 417 (79) | 346 (95) | 71 (44) | <0.001 |
| G 1 | 65 (12) | 10 (3) | 55 (34) | |
| G 2 | 23 (4) | 5 (1) | 18 (11) | |
| G 2 with CR and/or IDC | 10 (2) | 0 (0) | 10 (6) | |
| G 3 | 9 (2) | 2 (1) | 7 (4) | |
| G 4–5 | 1 (1) | 0 (0) | 1 (1) |
| Univariable Analysis | Multivariable Analysis (Model) | |||||
|---|---|---|---|---|---|---|
| Variable | Odds Ratio | 95% CI | p Value | Odds Ratio | 95% CI | p Value |
| Age (per 10 years) | 2.62 | 1.62–4.25 | <0.001 | 2.31 | 1.43–3.92 | 0.001 |
| Largest index lesion diameter | 1.01 | 0.96–1.01 | 0.70 | 1.02 | 0.96–1.08 | 0.51 |
| PSA-density (multiplied by 10) | 1.65 | 1.31–2.10 | <0.001 | 1.53 | 1.21–1.97 | <0.001 |
| Thresholds | TBx Sessions | ISUP Grade 1 PCa | ISUP Grade ≥2 PCa | ISUP grade ≥2 with CR and/or IDC PCa | ISUP Grade ≥3 PCa | |
|---|---|---|---|---|---|---|
| Largest Index Lesion Diameter | PSA-Density | Avoided (n, %) | Not Detected (n, %) | Missed Diagnosis (n, %) | Missed Diagnosis (n, %) | Missed Diagnosis (n, %) |
| Monitor all patients | 292 (100%) | 53 (100%) | 39 (100%) | 20 (100%) | 10 (100%) | |
| Biopsy all patients | 0 (%) | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | |
| ≥4 mm | - | 3 (1%) | 3 (6%) | 0 (0%) | 0 (0%) | 0 (0%) |
| ≥5 mm | - | 11 (4%) | 5 (9%) | 1 (3%) | 1 (5%) | 0 (0%) |
| ≥6 mm | - | 19 (7%) | 10 (19%) | 1 (3%) | 1 (5%) | 0 (0%) |
| ≥7 mm | - | 29 (10%) | 14 (26%) | 2 (5%) | 2 (10%) | 1 (10%) |
| ≥8 mm | - | 41 (14%) | 16 (30%) | 5 (13%) | 3 (15%) | 2 (20%) |
| - | ≥0.05 ng/mL2 | 5 (2%) | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) |
| - | ≥0.10 ng/mL2 | 55 (20%) | 4 (1%) | 2 (5%) | 1 (5%) | 1 (10%) |
| - | ≥0.11 ng/mL2 | 73 (25%) | 6 (11%) | 2 (5%) | 1 (5%) | 1 (10%) |
| - | ≥0.12 ng/mL2 | 91 (31%) | 9 (17%) | 3 (8%) | 1 (5%) | 1 (10%) |
| - | ≥0.15 ng/mL2 | 141 (48%) | 14 (26%) | 5 (13%) | 1 (5%) | 1 (10%) |
| - | ≥0.20 ng/mL2 | 183 (63%) | 19 (36%) | 10 (26%) | 2 (10%) | 2 (20%) |
| Threshold | TBx Sessions | ISUP Grade 1 PCa | ISUP Grade ≥2 PCa |
|---|---|---|---|
| Risk of csPCa | Avoided (n, %) | Not Detected (n, %) | Missed Diagnosis (n, %) |
| Monitor all patients | 292 (100%) | 53 (100%) | 39 (100%) |
| Biopsy all patients | 0 (0%) | 0 (0%) | 0 (0%) |
| ≥2% | 10 (3%) | 1 (2%) | 0 (0%) |
| ≥3% | 30 (10%) | 2 (4%) | 0 (0%) |
| ≥4% | 47 (16%) | 4 (8%) | 1 (3%) |
| ≥5% | 65 (23%) | 7 (13%) | 1 (3%) |
| ≥6% | 84 (30%) | 9 (17%) | 2 (5%) |
| ≥7% | 97 (34%) | 12 (23%) | 2 (5%) |
| ≥8% | 119 (42%) | 16 (30%) | 3 (8%) |
| ≥9% | 137 (48%) | 20 (38%) | 3 (8%) |
| ≥10% | 150 (53%) | 22 (42%) | 4 (10%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Osses, D.F.; Arsov, C.; Schimmöller, L.; Schoots, I.G.; van Leenders, G.J.L.H.; Esposito, I.; Remmers, S.; Albers, P.; Roobol, M.J. Equivocal PI-RADS Three Lesions on Prostate Magnetic Resonance Imaging: Risk Stratification Strategies to Avoid MRI-Targeted Biopsies. J. Pers. Med. 2020, 10, 270. https://doi.org/10.3390/jpm10040270
Osses DF, Arsov C, Schimmöller L, Schoots IG, van Leenders GJLH, Esposito I, Remmers S, Albers P, Roobol MJ. Equivocal PI-RADS Three Lesions on Prostate Magnetic Resonance Imaging: Risk Stratification Strategies to Avoid MRI-Targeted Biopsies. Journal of Personalized Medicine. 2020; 10(4):270. https://doi.org/10.3390/jpm10040270
Chicago/Turabian StyleOsses, Daniël F., Christian Arsov, Lars Schimmöller, Ivo G. Schoots, Geert J.L.H. van Leenders, Irene Esposito, Sebastiaan Remmers, Peter Albers, and Monique J. Roobol. 2020. "Equivocal PI-RADS Three Lesions on Prostate Magnetic Resonance Imaging: Risk Stratification Strategies to Avoid MRI-Targeted Biopsies" Journal of Personalized Medicine 10, no. 4: 270. https://doi.org/10.3390/jpm10040270
APA StyleOsses, D. F., Arsov, C., Schimmöller, L., Schoots, I. G., van Leenders, G. J. L. H., Esposito, I., Remmers, S., Albers, P., & Roobol, M. J. (2020). Equivocal PI-RADS Three Lesions on Prostate Magnetic Resonance Imaging: Risk Stratification Strategies to Avoid MRI-Targeted Biopsies. Journal of Personalized Medicine, 10(4), 270. https://doi.org/10.3390/jpm10040270