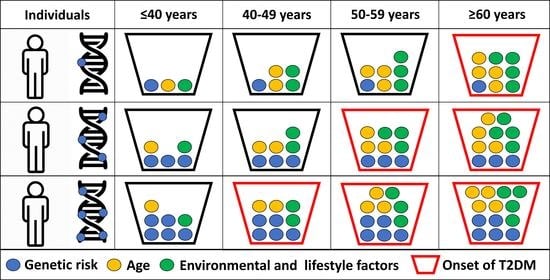
1. Introduction
The global prevalence of diabetes among adults rose from 4.7% to 8.5% between 1980 and 2014 [
1]. The majority of diabetic patients (~90%) have type 2 diabetes mellitus (T2DM) [
2]. Previously, T2DM was considered to be a disease of older adults (formerly known as adult-onset diabetes); however, in recent decades the age of onset has been falling and T2DM has been reported in younger adults and even in children and adolescents as well [
3,
4]. In addition to the growth of the proportion of younger age groups among T2DM patients, the emergence of it is still characteristic of late middle-aged and older people. The highest estimated crude incidence of diagnosed diabetes was among people aged 45–64 years in the United States of America in 2018 [
5]. This number was lower in those under 45 or over 64 years [
6].
Early onset T2DM (EOT2DM), defined as a diagnosis at the age below 45 years of age, has become more prevalent throughout the world since the early 1990s [
7]. Several countries around the world (e.g.,: China [
8], the United Kingdom [
9]), reported an increase in the incidence and consequently the prevalence of EOT2DM. In Japan [
10] and Taiwan [
11], more than 50% of diabetes cases in children and adolescents have type 2 diabetes mellitus.
The early onset of T2DM exerts an extensive impact on individuals, healthcare service planning, and delivery [
7]. It is supported by evidence that individuals with EOT2DM appear to have poorer quality of health outcomes compared with the non-diabetic population or individuals with the usual-onset of T2DM (UOT2DM) [
12,
13]. It was also corroborated that EOT2DM is associated with a higher risk of many types of diabetes-related complications [
12,
14]. The odds of developing macrovascular and microvascular complications are higher among diabetes patients with EOT2DM compared with UOT2DM [
12]. Individuals with EOT2DM have a much higher risk of the development of cardiovascular diseases, and microalbuminuria is also more common among them compared with people with UOT2DM [
15]. In general, individuals with EOT2DM have significantly poorer metabolic profiles than individuals with UOT2DM [
16].
Diabetes was the seventh leading cause of death estimated by the WHO in 2016 (it was 13th in 2000) [
17]. The early identification of T2DM could help reduce the number of deaths caused by them [
18]. To develop effective screening, it is essential to know the key factors involved in the development of the disease. Type 2 diabetes mellitus results from a complex interplay between multiple genetic factors and a wide variety of environmental and lifestyle risk factors, such as physical inactivity, obesity, high sugar intake, and low socio-economic status [
7]. It is a well-known fact that genetic factors also play a major role in the development of diabetes (the estimated heritability of fasting blood sugar is 29%) [
19]. However, until now, only a small number of studies have focused on how these genetic factors contribute to the age of the development of T2DM. The existence of genetic causes underlying EOT2DM is supported by the fact that in one study 81% of children and young people had a family history of T2DM (70% with first-degree relatives: 17% both parents affected, 50% mother alone, 23% father alone, and 10% sibling alone affected, and 11% with second-degree relatives) in the United Kingdom [
20]. Over the last two decades, several single nucleotide polymorphisms (SNPs) have been shown to be associated with the development of EOT2DM [
21,
22,
23,
24,
25,
26,
27,
28,
29,
30]. The majority of these SNPs exert their effect on disease risk through deficient insulin secretion [
31].
In Hungary, according to the 2019 International Diabetic Federation report, the age-adjusted comparative prevalence of diabetes (type 1 and 2) among the adult population (aged between 20–70 years) is 6.9%, more than 90% of which is accounted for by T2DM [
32]. A recent nationwide population-based study showed that the prevalence of T2DM among children and adolescents in Hungary was 22 out of 100,000 [
33] and this data was higher than in most European countries (such as Denmark [
34] and Germany [
35]). The prevalence of obesity among children or adolescents was 6.6% [
36], while among adults (above 18 years) it was 32% [
37]. Hungary has the highest obesity rate in Europe based on OECD data from 2016 [
38]. Although the high incidence of obesity in the Hungarian population can be partly explained by the high incidence of T2DM, the underlying genetic causes cannot be neglected either [
39,
40].
Our previous studies confirmed that in the case of obesity [
39,
41] and T2DM [
40], in addition to environmental and lifestyle factors, genetic susceptibility is also very significant in the Hungarian general population. T2DM is developing earlier for individuals with a higher genetic risk, in comparison with people with the same lifestyle and environmental characteristics but with a lower genetic risk [
42]. By identifying these genetically high-risk individuals, it is possible to develop a cost-effective targeted prevention program for EOT2DM. So far only a limited number of studies have examined the effect of genetic factors (such as SNPs) on the age of development of T2DM, and most of these have been studied in Asian, as Chinese Han, Japanese and Taiwanese populations [
31,
43,
44,
45]. Although in a genome-wide search for type 2 diabetes-related traits in a group of people with French ancestry evidence for a susceptibility locus for diabetes and impaired glucose tolerance with early onset (at age < 46 years) on chromosome 3q27-qter was provided [
46], we have little knowledge (restricted to diabetics in Tayside, Scotland) on European populations [
47].
Our current research aims to determine whether the addition of genetic risk variant (SNPs) data related to the development of T2DM in the form of genetic risk scores to conventional risk factors (sex, BMI, TG/HDL-C ratio) improves risk assessment of the early onset of T2DM or not. Furthermore, based on the relevant SNPs identified in the T2DM case population, a genetic risk score model was created and tested on an independent sample population (representative of the Hungarian general population). The contribution of genetic and non-genetic factors to the development of T2DM was also tested on patients with EOT2DM and UOT2DM.
2. Materials and Methods
2.1. Sample Populations
2.1.1. T2DM Case Population Representative of Hungarian T2DM Patients above 50 Years Old
The study subjects were obtained from a previous survey (in 2008), which was conducted in the framework of General Practitioner’s Morbidity Sentinel Stations Program and included 1168 individuals representative of Hungarian T2DM patients above the age of 50 years [
48]. Physical examinations (weight, height, waist circumference, and blood pressure) were carried out and blood samples (native and EDTA-anticoagulated) for laboratory investigation and DNA isolation were collected by physicians. Information on sociodemographic characteristics and self-assessed health status were obtained using a self-administered questionnaire. More details on sample collection are described by Nagy et al. [
49].
2.1.2. Hungarian General Population
One-thousand seven-hundred and eighty-three samples from the Hungarian general population, obtained from our previous study from 2008 [
50], were included and used as an independent population for validating the genetic risk score model. The Hungarian general sample population is representative for the Hungarian adult (>20 years) population in terms of age, sex, and geographic distribution. As part of the survey, interviewer-assisted questionnaires were used to collect data on sociodemographic factors and self-assessed health status. Medical histories were recorded, and each participant went through physical examination. Native and EDTA-anticoagulated blood samples were taken for genotyping and laboratory examinations. Further details of sample collection and characteristics are described by Szigethy et al. and Kosa et al. [
50,
51].
2.2. DNA Extraction, Selection of SNPs, Genotyping, Testing Hardy-Weinberg Equilibrium and Linkage Disequilibrium
DNA was extracted from EDTA-anticoagulated blood samples using the DNA Isolation Kit–Large Volume on a MagNA Pure LC instrument (Roche Diagnostics, Basel, Switzerland) following the manufacturer’s instructions.
Twenty-three SNPs that affect the development of T2DM were identified through a systematic literature search on online databases (PubMed, HuGE Navigator, and Ensembl). Details on the selection process of SNPs was described by Werissa et al. [
40] and showed also in
Table S1.
Genotyping was performed on the MassARRAY platform (Sequenom Inc., San Diego, CA, USA) with iPLEX Gold chemistry by the service provider (Mutation Analysis Core Facility of the Karolinska University Hospital (MAF), Sweden). Validation, concordance analysis, and quality control were conducted by the MAF, according to their protocols.
A χ
2 test was used to assess whether the agreement of frequencies of genotypes for SNPs with Hardy-Weinberg equilibrium (HWE) expectations (by Plink software [
52]). Linkage disequilibrium (LD) between polymorphisms was tested by Haploview software (version 4.2). In the presence of an LD block, one SNP was selected from each block to avoid multicollinearity.
2.3. Sample Selection for Study Populations
For T2DM Case and Hungarian general populations those samples were selected that have a complete record of diabetes-related pheno- and genotype data. These data include age, sex, body mass index (BMI), triglyceride (TG), and high-density lipoprotein cholesterol (HDL-C) levels in both populations and the age at diagnosis of diabetes in the T2DM case population (the age at diagnosis is not available for subjects of the Hungarian general one). The TG to HDL-C ratio (TG/HDL-C ratio) was defined as a better indicator for T2DM [
53] than TG or HDL-C levels individually.
The samples of the HG population were divided into three subpopulations based on their fasting glucose levels (FG) and/or treatment for diabetes.
The three subpopulations:
Subjects with normal fasting glucose level: FG < 5.6 mmol/L, n = 1197
Prediabetic subjects: FG between 5.6 and 6.9 mmol/L, n = 108
Type 2 diabetic patients: any person who had a FG level of 7 mmol/L or higher and/or was under antidiabetic treatment,
n = 110 [
54]
2.4. Determination of the Best Fitting Genetic Model for SNPs
For each SNP we tested [
55] which of the genetic model of inheritance (codominant, dominant, and recessive) showed the strongest correlation with the outcome (age of onset for T2DM) in the Case population. Adjusted (by age, sex, TG/HDL-C ratio) regression analyses were applied to test the association of SNPs individually with the age of onset for T2DM by SNPStats online tool (
http://bioinfo.iconcologia.net/SNPstats). The Akaike information criterion (AIC), Bayesian information criterion (BIC), and
p-value were used to find the best fitting genetic model of inheritance under the selection process [
56].
2.5. Unweighted Genetic Risk Score Calculation
2.5.1. Coding SNPs by the Best Fitting Genetic Model
The best fitting genetic model of inheritance was determined for each SNP and these SNPs were encoded according to the criteria of the model as follows:
In case of the codominant genetic model:
homozygote genes with two risk alleles were counted as “2′’, while heterozygote genotypes as “1′’ and homozygote non-risk genes as “0′’.
In case of the dominant genetic model:
homo- and heterozygote genes with two or one risk alleles were counted as “2′’, while homozygote non-risk genes as “0′’.
In case of the recessive genetic model:
homozygote genes with two risk alleles were counted as “2′’, while heterozygote genotypes with one risk allele and homozygote without risk allele as “0′’.
2.5.2. Calculation and Optimization of the GRS Model
Subsequently, the number of risk effects (2, 1, or 0) was summed using equation 1, where Gi is the number of risk effects in the respective locus (see more detailed in
Section 2.5.1) for the unweighted genetic risk score.
The effect of SNPs and the strength of their association on the age of onset for T2DM were determined as described in
Section 2.4.
During the optimization of the GRS model, SNPs which do not reinforce the association of the model with the outcome variable were excluded. To avoid the possibility of false-positive association, SNPs were tested in an ascending order of p-value (from the strongest association with the weakest one). Starting with the SNP with the lowest p-value, we inserted them one by one in GRS model, the association of which with the age of onset was tested after each inserted SNP. For each step, the number of risk alleles for the next SNP was added to the GRS. Regression analysis was applied to monitor the changes in the strength of the association. The inserted SNPs were selected for the final GRS model only if they increased the model’s R-squared value and decreased its p-value.
Under the optimization process, all calculation was adjusted by BMI, TG/HDL-C ratio, sex, and duration of T2DM.
2.6. Estimation of the Effect of Genetic (Unweighted Genetic Risk Score) and Non-Genetic (Sex, BMI, and TG/HDL Ratio) Factors on the Age of Onset for T2DM on the Case Population
Linear regression was used to estimate the effect of GRS and non-genetic factors (sex, BMI, and TG/HDL ratio) on the age of onset for T2DM on the Case population. The results of this calculation were used to determine the weighted GRS as well as to construct a risk estimation model for the age of onset for T2DM on the HG population.
2.7. Weighted Genetic Risk Score Calculation
Weighted GRS calculation was performed on the HG population by using the beta values determined on the Case population for weighting (wβ_i). Then the GRS for each person (xi) was multiplied by the weight (wβ_i). Equation (2) describes the calculation of the weighted genetic risk score.
2.8. Calculation of a Score for an Estimated Age of Onset for T2DM
The weight of genetic and non-genetic factors was determined by the Case population. Using these weights, it is possible to calculate a score to estimate the age of onset for T2DM. To investigate the combined effect of non-genetic (sex, BMI, and TG/HDL-C ratio) and genetic (GRSs) factors with a reasonable impact on the development of T2DM, a score was calculated for each sample. The effect of non-genetic and genetic factors on the age of onset for T2DM was estimated on the Case population and it was tested on the HG one.
2.9. Statistical Analysis
The normality of data for quantitative variables was tested using the Shapiro-Wilk test; and when it was necessary, non-normal variables were transformed using Templeton’s two-step approach [
57]. Two-tailed Student’s
t-tests were used to assess the statistical difference of variables among the groups. Multiple linear regression analyses were used to estimate the individual and combined (GRS) effect of SNPs on the early onset of T2DM. The Jonckheere-Terpstra trend test was [
58] used to analyze the statistically significant trend between the ordinal independent variable and continuous or ordinal dependent variables.
All biostatistics calculations were adjusted by relevant covariates (e.g., sex, BMI, and TG/HDL-C ratio). IBM SPSS statistics for Windows (version 26, IBM Company, Armonk, NY, USA) and the SNPStats online tool were employed to carry out regression analyses. The Bonferroni correction was applied when several statistical tests were being performed simultaneously (p < 0.0042).
2.10. Ethical Statement
All subjects gave their informed consent to participate in the study. The study was conducted following the ethical standards of the institutional and national research committees, in harmony with the declaration of Helsinki. This study was commenced after getting ethical permission from the Ethical Committee of the University of Debrecen, Medical Health Sciences Centre (reference No. 2699-2007), and by the Ethical Committee of the Hungarian Scientific Council on Health (reference No. TUKEB 48495-2/2014/EKU).
4. Discussion
Over the past few decades, the prevalence of EOT2DM has been increasing throughout the world and it is becoming the predominant form of diabetes among adolescents and younger adults in some populations [
7]. Since the early onset of T2DM is associated with a higher cardiovascular risk (micro- and macrovascular complications) and a higher frequency of comorbidities among the patients, early identification of EOT2DM risk would be important for the development of effective preventive intervention strategies. Developing a sensitive risk assessment tool is essential to know the weight of genetic and non-genetic factors in the disease manifestation.
The current study aims to evaluate the joint effect of type 2 diabetes mellitus associated SNPs (using genetic risk score modeling) and known non-genetic risk factors (such as sex, BMI, and TG/HDL-C ratio) on the age of onset for T2DM in the Hungarian general population.
When the effect of SNPs on the age of onset for type 2 diabetes mellitus was evaluated, no SNP was identified to have an individually significant association with the age of onset for type 2 diabetes mellitus. Under the optimization process of the genetic risk score model, 12 SNPs have been identified that increase the strength of the association between the GRS and the age of onset for T2DM on the Case population. The GRS (summarizing the individual effect of SNPs) shows a strongly significant association with the age of onset for type 2 diabetes mellitus (beta (95%CI): −0.454 (−0.674–−0.234), p < 0.001) even after adjusting for relevant covariates (sex, BMI, TG/HDL-C and duration of T2DM) in the Case population. The result was similar when this association was evaluated separately for the two sexes (males: beta (95%CI): −0.434 (−0.722–−0.145), p = 0.003; females: beta (95%CI): −0.405 (−0.796–−0.120), p = 0.008). A significant trend could also be observed between age groups (≤49 yrs, 50–59 yrs, and ≥60 yrs) and the mean value of GRS in the Case population (p < 0.001) and separately by sex (males: p = 0.002, females: p = 0.0038).
We tested the optimized GRS model (created on the Case population) on the HG population. The GRS showed no significant association with the presence of T2DM under the adjusted logistic regression model (OR (95%CI): 1.032 (0.945–1.126), p = 0.488). There was no significant association between individual age and GRS in the normal blood glucose (beta (95%CI): 0.104 (−0.158–0.365), p = 0.437) and prediabetic groups (beta (95%CI): −0.245 (−0.972–0.483), p = 0.507). In case of the type 2 diabetic group, GRS had a significant effect on the age of onset for T2DM; one point increase in AORS point of GRS reduced the age of onset by the average of one year (beta (95%CI): −0.999 (−1.660–−0.337), p = 0.003).
A limited number of studies examined and reported the association between genetic risk scores and the age of onset for T2DM. These studies support our findings according to which genetic factors have an effect on the age of onset for T2DM. Iwata M. et al. [
43] constructed GRS using 14 SNPs and evaluated its association with the early onset of T2DM in the Japanese population. The same result was observed by Kong X. et al. [
31] based on 24 SNPs in the Chinese. A study from pooled data of the Framingham Offspring Study which examined GRS constructed from 40 SNPs on individuals below or above 50 years of age witnessed that the risk per risk allele was higher (more than double) in individuals below 50 years than in people above 50 years of age [
60].
The investigators also concluded that the genetic risk score effectively increased the predictive value in addition to clinical risk factors in younger adults (<50 years of age) but not in older people (≥50 years of age). Our findings were also supported by The EPIC InterAct case-cohort study [
61] where the relative genetic risk comprised of 49 SNPs is higher in individuals who developed T2DM at a younger age (<55years) compared to that of individuals who developed it later (55–65 years or ≥65 years of age). Similarly, a more recent study by Mars et al. [
5], which showed that an individual with higher GRS developed the disease at an earlier age than individuals with lower GRS, concluded that a higher GRS has an impact on the age of onset for T2DM.
In our validation study, we could show that the age of onset was significantly associated with GRS and wGRS among individuals with T2DM. We also show that the higher the GRS is, the lower the age of onset for type 2 diabetes mellitus is (GRS: beta = −0.999, p = 0.003; wGRS: beta = −2.011, p = 0.0036). To estimate the age of onset for type 2 diabetes mellitus, we calculated the AORS for HG population’s normal, prediabetic, and T2DM subpopulations and found that the risk score was significantly higher in the T2DM group compared to the normal one. In this analysis, significant differences were observed for all non-genetic factors but not for GRS among the three groups (normal, prediabetic, and T2DM). This proves that genetic determination always remains constant throughout the life-course of an individual. A person’s increased genetic risk of the early onset of T2DM only exists if the effects of non-genetic risk factors are high enough. We further analyzed the effect of AORS and its components only for T2DM individuals from the HG population, and the findings explained the association between the age of onset for T2DM and genetic risk scores, i.e., the earlier the age of onset for T2DM is, the higher the GRS and wGRS are.
We observed a very strong association between TG/HDL-C and the age of onset for type 2 diabetes mellitus in men, and no association was seen in women. The age of onset for T2DM could be influenced by the TG/HDL-C ratio in men. In the Bogalusa Heart Study, the investigators observed that the TG/HDL-C ratio was associated with the age of onset for type 2 diabetes mellitus [
38]. However, the researchers did not assess their association separately for the male and female populations. Indeed, men are generally diagnosed with diabetes at a younger age than women [
62].
The strength of our current study is that we validated our results obtained on a T2DM case group by using an independent sample population (Hungarian general one). It is undeniable that this study has some limitations. Owing to a lack of information on gene-gene interactions, gene-environment interactions, epigenetic factors, and structural variants, we did not consider them in our analysis. In the current study, 12 SNPs that play a role in the age of onset for type 2 diabetes mellitus were considered in the GRS model. Incorporating a larger number of SNPs may further improve the predictive ability of the GRS model. Nonetheless, adding a large number of SNPs into the GRS model does not necessarily lead to a better predictive ability [
63,
64].
The clinical applicability of our present study is limited, further studies are needed to understand the SNP-mediated development of T2DM. It is also necessary to test the results in other populations to gain certainty that the results obtained are similar in non-Hungarian populations. Nevertheless, our results may pave the way for the development of genetic tests that can be used to predict the timing of T2DM development and delay or prevent its manifestation through targeted interventions, which would also reduce the burden on health care systems.
Owing to the emergence of “big data”, and the advancement of analytical tools based on the results of genomic, epigenetic, metabolomic, proteomic and pharmacogenetic studies, personalized type 2 diabetes treatments are creating, and a one-size-fits-all approach is becoming antiquated. Because of polygenic nature of the disease and the influence of both environmental and genetic factors on its development, defining subgroups using molecular testing is difficult in type 2 diabetes mellitus patients. So the best approach for the accurate and most convenient treatment of T2DM is to categorize patients based on their SNPs-based expected response to medicines. Exploring how the SNPs influence drug efficacy may help us uncover new drug targets and personalized treatment.
In summary, this is the first study that investigated the association of the combined effect of T2DM associated SNPs using GRS on the age of onset for T2DM in the Hungarian population. Genetic risk score modeling showed that the cumulative effect of T2DM related SNPs was associated with the age of onset for type 2 diabetes mellitus. Individuals who developed T2DM at a younger age carried greater risk allele load compared with individuals who developed T2DM later in their life. Our results suggest that there is a considerable genetic predisposition for the early onset of T2DM. Therefore, GRS can be used as a tool for stratifying and estimating the risk of the earlier onset of type 2 diabetes mellitus in the Hungarian population.