Rituximab as a Treatment Option after Autologous Hematopoietic Stem Cell Transplantation in a Patient with Systemic Sclerosis
Abstract
:1. Introduction
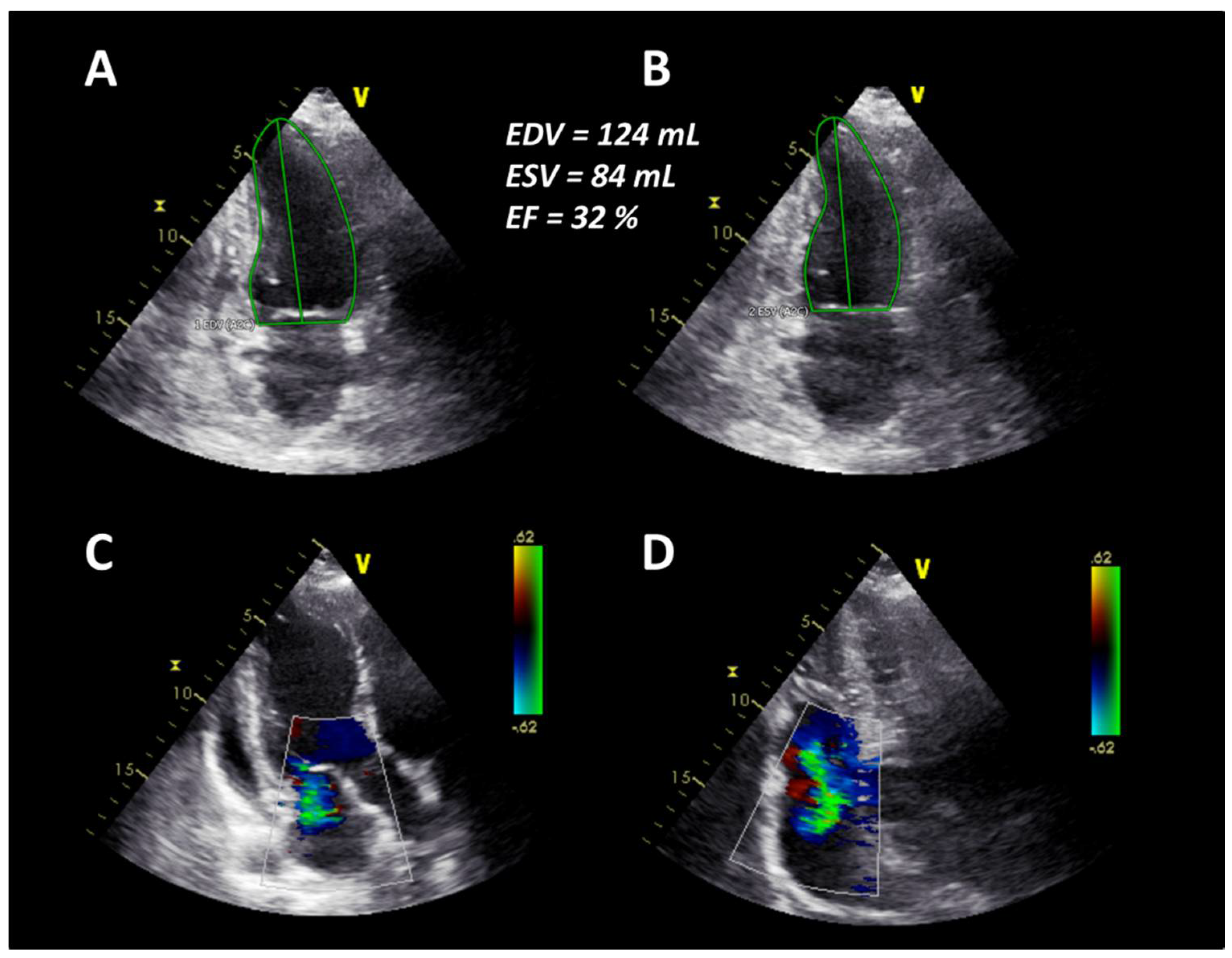
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hughes, M.; Denton, C.P.; Khanna, D. Rituximab for the treatment of systemic sclerosis-interstitial lung disease. Rheumatology 2021, 60, 489–491. [Google Scholar] [CrossRef] [PubMed]
- Misra, D.P.; Ahmed, S.; Agarwal, V. Is biological therapy in systemic sclerosis the answer? Rheumatol. Int. 2020, 40, 679–694. [Google Scholar] [CrossRef]
- Del Papa, N.; Pignataro, F.; Zaccara, E.; Maglione, W.; Minniti, A. Autologous Hematopoietic Stem Cell Transplantation for Treatment of Systemic Sclerosis. Front. Immunol. 2018, 9, 2390. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sullivan, K.M.; Goldmuntz, E.A.; Keyes-Elstein, L.; McSweeney, P.A.; Pinckney, A.; Welch, B.; Mayes, M.D.; Nash, R.A.; Crofford, L.J.; Eggleston, B.; et al. Myeloablative Autologous Stem-Cell Transplantation for Severe Scleroderma. N. Engl. J. Med. 2018, 378, 35–47. [Google Scholar] [CrossRef] [PubMed]
- van den Hoogen, F.; Khanna, D.; Fransen, J.; Johnson, S.R.; Baron, M.; Tyndall, A.; Matucci-Cerinic, M.; Naden, R.P.; Medsger, T.A., Jr.; Carreira, P.E.; et al. Classification Criteria for Systemic Sclerosis: An ACR-EULAR Collaborative Initiative. Arthritis Rheum. 2013, 65, 2737–2747. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moazedi-Fuerst, F.C.; Kielhauser, S.M.; Brickmann, K.; Hermann, J.; Lutfi, A.; Meilinger, M.; Brezinschek, H.; Graninger, W. Rituximab for systemic sclerosis: Arrest of pulmonary disease progression in five cases. Results of a lower dosage and shorter interval regimen. Scand. J. Rheumatol. 2014, 43, 257–258. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tyndall, A.J.; Bannert, B.; Vonk, M.; Al, E.; Distler, O.; Chizzolini, C. Causes and risk factors for death in systemic sclerosis: A study from the EULAR Scleroderma Trials and Research (EUSTAR) database. Ann. Rheum. Dis. 2010, 69, 1809–1815. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kowal-Bielecka, O.; Fransen, J.; Avouac, J.; Becker, M.; Kulak, A.; Allanore, Y.; Distler, O.; Clements, P.J.; Cutolo, M.; Czirjak, L.; et al. Update of EULAR recommendations for the treatment of systemic sclerosis. Ann. Rheum. Dis. 2017, 76, 1327–1339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Henes, J.; Oliveira, M.C.; Labopin, M.; Badoglio, M.; Scherer, H.U.; Del Papa, N.; Daikeler, T.; Schmalzing, M.; Schroers, R.; Martin, T.; et al. Autologous stem cell transplantation for progressive systemic sclerosis: A prospective non-interventional study from the European Society for Blood and Marrow Transplantation Autoimmune Disease Working Party. Haematologica 2020. Epub ahead of print. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Bijnen, S.; de Vries-Bouwstra, J.; van den Ende, C.H.; Boonstra, M.; Kroft, L.; Geurts, B.; Snoeren, M.; Schouffoer, A.; Spierings, J.; van Laar, J.M.; et al. Predictive factors for treatment-related mortality and major adverse events after autologous haematopoietic stem cell transplantation for systemic sclerosis: Results of a long-term follow-up multicentre study. Ann. Rheum. Dis. 2020, 79, 1084–1089. [Google Scholar] [CrossRef] [PubMed]
- Elhai, M.; Boubaya, M.; Distler, O.; Smith, V.; Matucci-Cerinic, M.; Sancho, J.J.A.; Truchetet, M.-E.; Braun-Moscovici, Y.; Iannone, F.; Novikov, P.I.; et al. Outcomes of patients with systemic sclerosis treated with rituximab in contemporary practice: A prospective cohort study. Ann. Rheum. Dis. 2019, 78, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Distler, O.; Highland, K.B.; Gahlemann, M.; Azuma, A.; Fischer, A.; Mayes, M.D.; Raghu, G.; Sauter, W.; Girard, M.; Alves, M.; et al. Nintedanib for systemic sclerosis-asso-ciated interstitial lung disease. N. Engl. J. Med. 2019, 380, 2518–2528. [Google Scholar] [CrossRef] [PubMed]
- Swart, J.F.; Delemarre, E.M.; van Wijk, F.; Boelens, J.-J.; Kuball, J.; Van Laar, J.M.; Wulffraat, N.M. Haematopoietic stem cell transplantation for autoimmune diseases. Nat. Rev. Rheumatol. 2017, 13, 244–256. [Google Scholar] [CrossRef] [PubMed]
- Farge, D.; Arruda, L.C.M.; Brigant, F.; Clave, E.; Douay, C.; Marjanovic, Z.; Deligny, C.; Maki, G.; Gluckman, E.; Toubert, A.; et al. Long-term immune reconstitution and T cell repertoire analysis after autologous hematopoietic stem cell transplantation in systemic sclerosis patients. J. Hematol. Oncol. 2017, 10, 21. [Google Scholar] [CrossRef] [PubMed]
- Arruda, L.C.M.; Malmegrim, K.C.R.; Lima-Júnior, J.R.; Clave, E.; Dias, J.B.E.; Moraes, D.A.; Douay, C.; Fournier, I.; Moins-Teisserenc, H.; Alberdi, A.J.; et al. Immune rebound associates with a favorable clinical response to autologous HSCT in systemic sclerosis patients. Blood Adv. 2018, 2, 126–141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ebata, S.; Yoshizaki, A.; Oba, K.; Kashiwabara, K.; Ueda, K.; Uemura, Y.; Watadani, T.; Fukasawa, T.; Miura, S.; Yoshizaki-Ogawa, A.; et al. Safety and efficacy of rituximab in systemic sclerosis (DESIRES): A double-blind, investigator-initiated, randomised, placebo-controlled trial. Lancet Rheumatol. 2021. Epub ahead of print. [Google Scholar] [CrossRef]
- Daoussis, D.; Liossis, S.N.; Tsamandas, A.C.; Kalogeropoulou, C.; Paliogianni, F.; Sirinian, C.; Yiannopoulos, G.; Andonopoulos, A.P. Effect of long-term treatment with rituximab on pulmonary function and skin fibrosis in patients with diffuse systemic sclerosis. Clin. Exp. Rheumatol. 2012, 30, 17–22. [Google Scholar]
- Moazedi-Fuerst, F.C.; Kielhauser, S.M.; Bodo, K.; Graninger, W. Dosage of rituximab in systemic sclerosis: 2-year results of five cases. Clin. Exp. Dermatol. 2015, 40, 211–212. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gressenberger, P.; Jud, P.; Kovacs, G.; Kreuzer, S.; Brezinsek, H.-P.; Guetl, K.; Muster, V.; Kolesnik, E.; Schmidt, A.; Odler, B.; et al. Rituximab as a Treatment Option after Autologous Hematopoietic Stem Cell Transplantation in a Patient with Systemic Sclerosis. J. Pers. Med. 2021, 11, 600. https://doi.org/10.3390/jpm11070600
Gressenberger P, Jud P, Kovacs G, Kreuzer S, Brezinsek H-P, Guetl K, Muster V, Kolesnik E, Schmidt A, Odler B, et al. Rituximab as a Treatment Option after Autologous Hematopoietic Stem Cell Transplantation in a Patient with Systemic Sclerosis. Journal of Personalized Medicine. 2021; 11(7):600. https://doi.org/10.3390/jpm11070600
Chicago/Turabian StyleGressenberger, Paul, Philipp Jud, Gabor Kovacs, Sonja Kreuzer, Hans-Peter Brezinsek, Katharina Guetl, Viktoria Muster, Ewald Kolesnik, Albrecht Schmidt, Balazs Odler, and et al. 2021. "Rituximab as a Treatment Option after Autologous Hematopoietic Stem Cell Transplantation in a Patient with Systemic Sclerosis" Journal of Personalized Medicine 11, no. 7: 600. https://doi.org/10.3390/jpm11070600






