Retinal Vascular Signs as Screening and Prognostic Factors for Chronic Kidney Disease: A Systematic Review and Meta-Analysis of Current Evidence
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy and Study Selection
2.2. Data Extraction
2.3. Retinal Microvascular Signs
- Fundus photography-derived signs:
- Central retinal arteriolar equivalent (CRAE) and central retinal venular equivalent (CRVE), measured as the average of the diameter of the largest 6 arterioles and largest 6 venules, respectively [9].
- Arterio-venous ratio (AVR), i.e., the ratio of CRAE to CRVE [12].
- Simple tortuosity, derived from the ratio of actual path length to straight-line length of a retinal vessel segment; or curvature tortuosity, derived from the integral of the curvature squared along the path of the vessel, and normalized by the total path length.
- Focal narrowing over a constricted area of ≤⅔ of the widths of proximal and distal vessel segments [2].
- Fractal dimension (Df), used to quantify the branching architecture of the retinal vasculature using the box-counting method [6].
- Optimality ratio, a measure of blood-flow loss in bifurcations related to endothelial dysfunction; optimality deviation measures the extent to which the optimality ratio deviates from the theoretically predicted optimum.
- Branching angle, the first angle between two daughter vessels at each vascular bifurcation [6].
- Retinopathy, considered to be present if any characteristic lesion (microaneurysms, hemorrhages, cotton-wool spots, intraretinal microvascular abnormalities, hard exudates, venous beading or new vessels) was present [6]. A retinopathy severity score was assigned according to the modified Airlie House classification system [8] or the Early Treatment Diabetic Retinopathy Study (ETDRS) score [9].
- OCTA-derived signs:
2.4. Evaluation of Renal Disease
- Albuminuria was calculated from spot urine specimens and was defined as:
- ACR > 2.5 mg/mmol for men and ACR > 3.5 mg/mmol for women were considered as albuminuria [21].
- Incident CKD
- Or, defined as eGFR < 60 mL/min/1.73 m2 accompanied by a decrease in eGFR of at least 25% over the follow-up period among subjects free of CKD at baseline, or as eGFR < 45 mL/min/1.73 m2 accompanied by a decrease in eGFR of at least 25% over the follow-up period among subjects free of CKD at baseline, or as a composite of incident CKD and/or rapid decline in eGFR (annual eGFR rate reduction of > 3 mL/min/1.73 m2/year) [6].
- Incident ESRD was defined as the initiation of chronic dialysis therapy or kidney transplantation [12].
- Prevalent ESRD was defined as eGFR of < 15 mL/min, or a serum creatinine level ≤ 5.7 mg/dL, or a new requirement of kidney failure treatment [8].
2.5. Statistical Analysis
2.6. Risk of Bias
3. Results
3.1. Risk of Bias
3.2. Retinal Microvascular Signs and CKD
3.2.1. Retinal Microvascular Signs and Incident CKD
3.2.2. Retinal Microvascular Signs and Deterioration of CKD
3.2.3. Retinal Microvascular Signs and Concurrent CKD
3.2.4. Retinal Microvascular Signs and Concurrent ESRD
3.3. Retinal Microvascular Signs and Concurrent eGFR
3.4. Retinal Microvascular Signs and Concurrent Albuminuria
3.5. Retinal Microvascular Signs and Renal Dysfunction in Diabetes
3.5.1. Retinal Microvascular Signs and Incident CKD
3.5.2. Retinal Microvascular Signs and Deterioration of CKD
3.5.3. Retinal Microvascular Signs and Concurrent CKD
3.5.4. Retinal Microvascular Signs and Diabetic Nephropathy
3.5.5. Retinal Microvascular Signs and Concurrent eGFR in Diabetes
3.5.6. Retinal Microvascular Signs and Albuminuria in Diabetes
4. Discussion
5. Conclusions and Further Study
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Appendix A

References
- Anothaisintawee, T.; Rattanasiri, S.; Ingsathit, A.; Attia, J.; Thakkinstian, A. Prevalence of chronic kidney disease: A systematic review and meta-analysis. Clin. Nephrol. 2009, 71, 244–254. [Google Scholar] [CrossRef]
- Wong, T.Y.; Coresh, J.; Klein, R.; Muntner, P.; Couper, D.J.; Sharrett, A.R.; Klein, B.E.K.; Heiss, G.; Hubbard, L.D.; Duncan, B.B. Retinal microvascular abnormalities and renal dysfunction: The Atherosclerosis Risk in Communities Study. J. Am. Soc. Nephrol. 2004, 15, 2469–2476. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bao, S.; Huang, W.; Liang, Y.; Jiang, L.; Wang, F.; Peng, Y.; Zhang, G.; Wang, N. Retinal vessel diameter and chronic kidney disease in rural China: A cross-sectional study. Medicine 2015, 94, 1–7. [Google Scholar] [CrossRef]
- Zheng, Y.; Ley, S.H.; Hu, F.B. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat. Rev. Endocrinol. 2018, 14, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Cankurtaran, V.; Inanc, M.; Tekin, K.; Turgut, F. Retinal Microcirculation in Predicting Diabetic Nephropathy in Type 2 Diabetic Patients without Retinopathy. Ophthalmologica 2019. [Google Scholar] [CrossRef]
- Yip, W.; Ong, P.G.; Teo, B.W.; Cheung, C.Y.L.; Tai, E.S.; Cheng, C.Y.; Lamoureux, E.; Wong, T.Y.; Sabanayagam, C. Retinal vascular imaging markers and incident chronic kidney disease: A prospective cohort study. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Treatment, E.; Retinopathy, D. Grading Diabetic Retinopathy from Stereoscopic Color Fundus Photographs—An Extension of the Modified Airlie House Classification: ETDRS Report Number 10. Ophthalmology 1991, 98, 786–806. [Google Scholar] [CrossRef]
- Yip, W.F.; Sabanayagam, C.; Teo, B.W.; Tay, W.T.; Ikram, M.K.; Tai, E.S.; Chow, K.Y.; Wong, T.Y.; Cheung, C.Y. Retinal Microvascular Abnormalities and Risk of Renal Failure in Asian Populations. PLoS ONE 2015, 33, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yau, J.W.Y.; Xie, J.; Kawasaki, R.; Kramer, H.; Shlipak, M.; Klein, R.; Klein, B.; Cotch, M.F.; Wong, T.Y. Retinal arteriolar narrowing and subsequent development of CKD stage 3: The multi-ethnic study of atherosclerosis (MESA). Am. J. Kidney Dis. 2011, 58, 39–46. [Google Scholar] [CrossRef] [Green Version]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef] [PubMed]
- Summary of Recommendation Statements. Kidney Int. Suppl. 2013, 3, 5–14. [CrossRef] [PubMed] [Green Version]
- Grunwald, J.E.; Pistilli, M.; Ying, G.; Daniel, E.; Maguire, M.G.; Xie, D.; Whittock-martin, R.; Ostroff, C.P.; Lo, J.C.; Townsend, R.R.; et al. Article Retinopathy and Progression of CKD: The CRIC Study. Epidemiol. Outcomes 2014, 9, 1217–1224. [Google Scholar] [CrossRef] [Green Version]
- Vadalà, M.; Castellucci, M.; Guarrasi, G.; Terrasi, M.; La Blasca, T.; Mulè, G.; Blasca, T.L.; Mulè, G. Retinal and choroidal vasculature changes associated with chronic kidney disease. Graefe’s Arch. Clin. Exp. Ophthalmol. 2019, 257, 1687–1698. [Google Scholar] [CrossRef]
- Sabanayagam, C.; Tai, E.S.; Shankar, A.; Lee, J.; Sun, C.; Wong, T.Y. Retinal arteriolar narrowing increases the likelihood of chronic kidney disease in hypertension. J. Hypertens. 2009, 27, 2209–2217. [Google Scholar] [CrossRef] [PubMed]
- Sabanayagam, C.; Shankar, A.; Koh, D.; Chia, K.S.; Saw, S.M.; Lim, S.C.; Tai, E.S.; Wong, T.Y. Retinal microvascular caliber and chronic kidney disease in an Asian population. Am. J. Epidemiol. 2009, 169, 625–632. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lim, L.S.; Cheung, C.Y.L.; Sabanayagam, C.; Lim, S.C.; Tai, E.S.; Huang, L.; Wong, T.Y. Structural changes in the retinal microvasculature and renal function. Investig. Ophthalmol. Vis. Sci. 2013, 54, 2970–2976. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Wang, J.; Ying, G.S.; Shen, L.; Zhang, Z. Diabetic retinopathy and renal function in Chinese type 2 diabetic patients. Int. Urol. Nephrol. 2014, 46, 1375–1381. [Google Scholar] [CrossRef] [PubMed]
- Broe, R.; Rasmussen, M.L.; Frydkjaer-olsen, U.; Olsen, B.S.; Mortensen, H.B.; Hodgson, L.; Wong, T.Y.; Peto, T.; Grauslund, J. Retinal vessel calibers predict long-term microvascular complications in type 1 diabetes: The Danish cohort of pediatric diabetes 1987 (DCPD1987). Diabetes 2014, 63, 3906–3914. [Google Scholar] [CrossRef]
- Mottl, A.K.; Kwon, K.S.; Garg, S.; Mayer-Davis, E.J.; Klein, R.; Kshirsagar, A.V. The association of retinopathy and low GFR in type 2 diabetes. Diabetes Res. Clin. Pract. 2012, 98, 487–493. [Google Scholar] [CrossRef] [Green Version]
- Awua-larbi, S.; Wong, T.Y.; Cotch, M.F.; Durazo-Arvizu, R.; Jr, D.R.J.; Klein, B.E.K.K.; Klein, R.; Lima, J.; Liu, K.; Kramer, H.; et al. Retinal arteriolar caliber and urine albumin excretion: The Multi-Ethnic Study of Atherosclerosis. Nephrol. Dial. Transplant. 2011, 26, 3523–3528. [Google Scholar] [CrossRef] [Green Version]
- Keel, S.; Itsiopoulos, C.; Koklanis, K.; Vukicevic, M.; Cameron, F.; Brazionis, L. Retinal Vascular Caliber and Kidney Function in Children and Adolescents with Type 1 Diabetes Retinal Vascular Caliber and Kidney Function in Children and Adolescents with. Ophthalmic Epidemiol. 2017, 24, 204–208. [Google Scholar] [CrossRef]
- Inker, L.A.; Astor, B.C.; Fox, C.H.; Isakova, T.; Lash, J.P.; Peralta, C.A.; Kurella Tamura, M.; Feldman, H.I. KDOQI US commentary on the 2012 KDIGO clinical practice guideline for the evaluation and management of CKD. Am. J. Kidney Dis. 2014, 63, 713–735. [Google Scholar] [CrossRef] [Green Version]
- Hayden, J.A.; van der Windt, D.A.; Cartwright, J.L.; Co, P. Research and Reporting Methods Annals of Internal Medicine Assessing Bias in Studies of Prognostic Factors. Ann. Intern. Med. 2013, 158, 280–286. [Google Scholar] [CrossRef]
- Lazzerini, M.; Sonego, M.; Pellegrin, M.C. Hypoxaemia as a mortality risk factor in acute lower respiratory infections in children in low and middle-income countries: Systematic review and meta-analysis. PLoS ONE 2015, 10, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Sabanayagam, C.; Shankar, A.; Klein, B.E.K.; Lee, K.E.; Muntner, P.; Nieto, F.J.; Tsai, M.Y.; Cruickshanks, K.J.; Schubert, C.R.; Brazy, P.C.; et al. Bidirectional Association of Retinal Vessel Diameters and Estimated GFR Decline : The Beaver Dam CKD Study. YAJKD 2011, 57, 682–691. [Google Scholar] [CrossRef] [Green Version]
- Baumann, M.; Burkhardt, K.; Heemann, U. Microcirculatory marker for the prediction of renal end points: A prospective cohort study in patients with chronic kidney disease stage 2 to 4. Hypertension 2014, 64, 338–346. [Google Scholar] [CrossRef] [Green Version]
- Liew, G.; Mitchell, P.; Wong, T.Y.; Wang, J.J. Retinal Microvascular Signs Are Associated with Chronic Kidney Disease in Persons with and without Diabetes. Kidney Blood Press. Res. 2012, 35, 589–594. [Google Scholar] [CrossRef]
- Phan, K.; Au, C.; Mitchell, P.; Liew, G.; Plant, A.J.H.H.; Wang, S.B.; Chiha, J.; Thiagalingam, A.; Burlutsky, G.; Gopinath, B. Chronic kidney disease and the severity of coronary artery disease and retinal microvasculature changes: A cross-sectional study. J. Thorac. Dis. 2016, 8, 2111–2114. [Google Scholar] [CrossRef] [Green Version]
- Gu, Y.M.; Petit, T.; Wei, F.F.; Thijs, L.; Jacobs, L.; Zhang, Z.Y.; Yang, W.Y.; Cauwenberghs, N.; Knez, J.; Struijker-Boudier, H.A.J.J.; et al. Renal glomerular dysfunction in relation to retinal arteriolar narrowing and high pulse pressure in seniors. Hypertens. Res. 2016, 39, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Ooi, Q.L.; Newk-Fon Hey Tow, F.K.; Deva, R.; Alias, M.A.; Kawasaki, R.; Wong, T.Y.; Mohamad, N.; Colville, D.; Hutchinson, A.; Savige, J. The microvasculature in chronic kidney disease. Clin. J. Am. Soc. Nephrol. 2011, 6, 1872–1878. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sng, C.C.A.; Sabanayagam, C.; Lamoureux, E.L.; Liu, E.; Lim, S.C.; Hamzah, H.; Lee, J.; Tai, E.S.; Wong, T.Y. Fractal analysis of the retinal vasculature and chronic kidney disease. Nephrol. Dial. Transpl. 2010, 25, 2252–2258. [Google Scholar] [CrossRef] [Green Version]
- Grunwald, J.E.; Pistilli, M.; Ying, G.S.; Daniel, E.; Maguire, M.; Xie, D.; Roy, J.; Whittock-Martin, R.; Parker Ostroff, C.; Lo, J.C.; et al. Association between Progression of Retinopathy and Concurrent Progression of Kidney Disease: Findings from the Chronic Renal Insufficiency Cohort (CRIC) Study. JAMA Ophthalmol. 2019, 137, 767–774. [Google Scholar] [CrossRef] [PubMed]
- Baumann, M.; Schwarz, S.; Kotliar, K.; Von Eynatten, M.; Schmidt Trucksaess, A.; Burkhardt, K.; Lutz, J.; Heemann, U.; Lanzl, I. Non-diabetic chronic kidney disease influences retinal microvasculature. Kidney Blood Press. Res. 2010, 32, 428–433. [Google Scholar] [CrossRef] [Green Version]
- Edwards, M.S.; Wilson, D.B.; Craven, T.E.; Stafford, J.; Fried, L.F.; Wong, T.Y.; Klein, R.; Burke, G.L.; Hansen, K.J. Associations between retinal microvascular abnormalities and declining renal function in the elderly population: The cardiovascular health study. Am. J. Kidney Dis. 2005, 46, 214–224. [Google Scholar] [CrossRef]
- Hwang, H.S.; Kim, S.Y.; Hong, Y.A.; Cho, W.K.; Chang, Y.K.; Shin, S.J.; Yang, C.W.; Kim, S.Y.; Yoon, H.E. Clinical impact of coexisting retinopathy and vascular calcification on chronic kidney disease progression and cardiovascular events. Nutr. Metab. Cardiovasc. Dis. 2016, 26, 590–596. [Google Scholar] [CrossRef]
- García-Ortiz, L.; Recio-Rodríguez, J.I.; Parra-sanchez, J.; Elena, L.J.G.; Patino-Alonso, M.C.; Agudo-Conde, C.; Rodríguez-Sánchez, E.; Gómez-Marcos, M.A.; Garcı, L.; Parra-sanchez, J.; et al. A new tool to assess retinal vessel caliber. Reliability and validity of measures and their relationship with cardiovascular risk. J. Hypertens. 2012, 30, 770–777. [Google Scholar] [CrossRef]
- Wong, T.Y.; Shankar, A.; Klein, R.; Klein, B.E.K. Retinal Vessel Diameters and the Incidence of Gross Proteinuria and Renal Insufficiency in People with Type 1 Diabetes. Diabetes 2004, 53, 179–184. [Google Scholar] [CrossRef] [Green Version]
- Lee, W.J.; Sobrin, L.; Kang, M.H.; Seong, M.; Kim, Y.J.; Yi, J.H.; Miller, J.W.; Cho, H.Y. Ischemic diabetic retinopathy as a possible prognostic factor for chronic kidney disease progression. Eye 2014, 28, 1119–1125. [Google Scholar] [CrossRef] [Green Version]
- Mckay, G.J.; Paterson, E.N.; Maxwell, A.P.; Cardwell, C.C.; Wang, R.; Hogg, S.; Macgillivray, T.J.; Trucco, E.; Doney, A.S. Retinal microvascular parameters are not associated with reduced renal function in a study of individuals with type 2 diabetes. Sci. Rep. 2018, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Broe, R.; Rasmussen, M.L.; Frydkjaer-Olsen, U.; Olsen, B.S.; Mortensen, H.B.; Peto, T.; Grauslund, J. Retinal vascular fractals predict long-term microvascular complications in type 1 diabetes mellitus: The Danish Cohort of Pediatric Diabetes 1987 (DCPD1987). Diabetologia 2014, 57, 2215–2221. [Google Scholar] [CrossRef] [Green Version]
- Rasmussen, M.L.; Broe, R.; Frydkjaer-Olsen, U.; Olsen, B.S.; Mortensen, H.B.; Peto, T.; Grauslund, J. Retinal vascular geometry and its association to microvascular complications in patients with type 1 diabetes: The Danish Cohort of Pediatric Diabetes 1987 (DCPD1987). Graefe’s Arch. Clin. Exp. Ophthalmol. 2017, 255, 293–299. [Google Scholar] [CrossRef]
- Benitez-Aguirre, P.Z.; Wong, T.Y.; Craig, M.E.; Davis, E.A.; Cotterill, A.; Couper, J.J.; Cameron, F.J.; Mahmud, F.H.; Jones, T.W.; Hodgson, L.A.B.B.; et al. The Adolescent Cardio-Renal Intervention Trial (AdDIT): Retinal vascular geometry and renal function in adolescents with type 1 diabetes. Diabetologia 2018, 61, 968–976. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Benitez-Aguirre, P.Z.; Sasongko, M.B.; Craig, M.E.; Jenkins, A.J.; Cusumano, J.; Cheung, N.; Wong, T.Y.; Donaghue, K.C. Retinal vascular geometry predicts incident renal dysfunction in young people with type 1 diabetes. Diabetes Care 2012, 35, 599–604. [Google Scholar] [CrossRef] [Green Version]
- Grauslund, J.; Hodgson, L.; Kawasaki, R.; Green, A.; Sjølie, A.K.; Wong, T.Y. Retinal vessel calibre and micro- and macrovascular complications in type 1 diabetes. Diabetologia 2009, 52, 2213–2217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ha, M.; Choi, S.Y.; Kim, M.; Na, J.K.; Park, Y.-H. Diabetic Nephropathy in Type 2 Diabetic Retinopathy Requiring Panretinal Photocoagulation. Korean J. Ophthalmol. 2019, 33, 46. [Google Scholar] [CrossRef]
- Grauslund, J.; Green, A.; Kawasaki, R.; Hodgson, L.; Sjolie, A.K.; Wong, T.Y. Retinal vascular fractals and microvascular and macrovascular complications in type 1 diabetes. Ophthalmology 2010, 117, 1400–1405. [Google Scholar] [CrossRef] [PubMed]
- Sasongko, M.B.; Wong, T.Y.; Donaghue, K.C.; Cheung, N.; Jenkins, A.J.; Benitez-Aguirre, P.; Wang, J.J. Retinal arteriolar tortuosity is associated with retinopathy and early kidney dysfunction in type 1 diabetes. Am. J. Ophthalmol. 2012, 153, 176–183.e1. [Google Scholar] [CrossRef]
- Garrido-hermosilla, A.M.; Méndez-Muros, M.; Gutiérrez-Sánchez, E.; Morales-Portillo, C.; Díaz-Granda, M.J.; Esteban-González, E.; Relimpio-López, I.; Martínez-Brocca, M.A.; Rodríguez-De-la-rúa-franch, E. Renal function and choroidal thickness using swept-source optical coherence tomography in diabetic patients. Int. J. Ophthalmol. 2019, 12, 985–989. [Google Scholar] [CrossRef]
- Disease, K.; Public, M.; Problem, H.; Treated, E.R.D. 2018 USRDS Annual Data Report: Executive Summary. Am. J. Kidney Dis. 2019, 73, A9–A22. [Google Scholar] [CrossRef]
- Ritz, E.; Orth, S.R. Nephropathy in patients with type 2 diabetes mellitus. N. Engl. J. Med. 1999, 341, 1127–1133. [Google Scholar] [CrossRef]
- Perneger, T.V.; Nieto, F.J.; Whelton, P.K.; Klag, M.J.; Comstock, G.W.; Szklo, M. A Prospective Study of Blood Pressure and Serum Creatinine: Results From the ‘Clue’ Study and the ARIC Study. JAMA J. Am. Med. Assoc. 1993, 269, 488–493. [Google Scholar] [CrossRef]
- Kawasaki, R.; Cheung, N.; Wang, J.J.; Klein, R.; Klein, B.E.; Cotch, M.F.; Sharrett, A.R.; Shea, S.; Islam, F.A.; Wong, T.Y. Retinal vessel diameters and risk of hypertension: The Multiethnic Study of Atherosclerosis. J. Hypertens. 2009, 27, 2386–2393. [Google Scholar] [CrossRef] [Green Version]
- Wong, T.Y.; Klein, R.; Richey Sharrett, A.; Schmidt, M.I.; Pankow, J.S.; Couper, D.J.; Klein, B.E.K.; Hubbard, L.D.; Duncan, B.B. Retinal arteriolar narrowing and risk of diabetes mellitus in middle-aged persons. J. Am. Med. Assoc. 2002, 287, 2528–2533. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, W.; Lo, A.C.Y. Diabetic retinopathy: Pathophysiology and treatments. Int. J. Mol. Sci. 2018, 19, 1816. [Google Scholar] [CrossRef] [Green Version]
- Agapitov, A.V.; Haynes, W.G. Role of endothelin in cardiovascular disease. JRAAS J. Renin-Angiotensin-Aldosterone Syst. 2002, 3, 1–15. [Google Scholar] [CrossRef]
- Amann, K.; Münter, K.; Wessels, S.; Wagner, J.; Balajew, V.; Hergenröder, S.; Mall, G.; Ritz, E. Endothelin a receptor blockade prevents capillary/myocyte mismatch in the heart of uremic animals. J. Am. Soc. Nephrol. 2000, 11, 1702–1711. [Google Scholar] [CrossRef] [PubMed]
- Törnig, J.; Amann, K.; Ritz, E.; Nichols, C.; Zeier, M.; Mall, G. Arteriolar wall thickening, capillary rarefaction and interstitial fibrosis in the heart of rats with renal failure: The effects of ramipril, nifedipine and moxonidine. J. Am. Soc. Nephrol. 1996, 7, 667–675. [Google Scholar] [CrossRef] [PubMed]
- Klein, R.; Knudtson, M.D.; Klein, B.E.K.; Zinman, B.; Gardiner, R.; Suissa, S.; Sinaiko, A.R.; Donnelly, S.M.; Goodyer, P.; Strand, T.; et al. The relationship of retinal vessel diameter to changes in diabetic nephropathy structural variables in patients with type 1 diabetes. Diabetologia 2010, 53, 1638–1646. [Google Scholar] [CrossRef] [Green Version]
- Ellis, E.N.; Steffes, M.W.; Goetz, F.C.; Sutherland, D.E.; Mauer, S.M. Glomerular filtration surface in type I diabetes mellitus. Kidney Int. 1986, 29, 889–894. [Google Scholar] [CrossRef] [Green Version]
- Sabanayagam, C.; Lye, W.K.; Klein, R.; Klein, B.E.K.; Cotch, M.F.; Wang, J.J.; Mitchell, P.; Shaw, J.E.; Selvin, E.; Sharrett, A.R.; et al. Retinal microvascular calibre and risk of diabetes mellitus: A systematic review and participant-level meta-analysis. Diabetologia 2015, 58, 2476–2485. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patel, V.; Rassam, S.; Newsom, R.; Wiek, J.; Kohner, E. Retinal blood flow in diabetic retinopathy. Br. Med. J. 1992, 305, 678–683. [Google Scholar] [CrossRef] [Green Version]
- Grunwald, J.E.; DuPont, J.; Riva, C.E. Retinal haemodynamics in patients with early diabetes mellitus. Br. J. Ophthalmol. 1996, 80, 327–331. [Google Scholar] [CrossRef] [Green Version]
- Falck, A.; Laatikainen, L. Retinal vasodilation and hyperglycaemia in diabetic children and adolescents. Acta Ophthalmol. Scand. 1995, 73, 119–124. [Google Scholar] [CrossRef]
- Feke, G.T.; Buzney, S.M.; Ogasawara, H.; Fujio, N.; Goger, D.G.; Spack, N.P.; Gabbay, K.H. Retinal circulatory abnormalities in type 1 diabetes. Investig. Ophthalmol. Vis. Sci. 1994, 35, 2968–2975. [Google Scholar]
- Bursell, S.E.; Clermont, A.C.; Kinsley, B.T.; Simonson, D.C.; Aiello, L.M.; Wolpert, H.A. Retinal blood flow changes in patients with insulin-dependent diabetes mellitus and no diabetic retinopathy: A video fluorescein angiography study. Investig. Ophthalmol. Vis. Sci. 1996, 37, 886–897. [Google Scholar]
- Ikram, M.K.; Cheung, C.Y.; Lorenzi, M.; Klein, R.; Jones, T.L.Z.; Wong, T.Y. Retinal vascular caliber as a biomarker for diabetes microvascular complications. Diabetes Care 2013, 36, 750–759. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Charles, J.J.; Olson, J.L.; Silva, F.G.; D’Agati, V.D. Heptinstall’s pathology of the kidney; Amazon.com Services LLC: Seattle, WA, USA, 2014. [Google Scholar]
- Allon, R.; Aronov, M.; Belkin, M.; Maor, E.; Shechter, M.; Fabian, I.D. Retinal Microvascular Signs as Screening and Prognostic Factors for Cardiac Disease: A Systematic Review of Current Evidence. Am. J. Med. 2021, 134, 36–47.e7. [Google Scholar] [CrossRef]
- Luo, T.; Gast, T.J.; Vermeer, T.J.; Burns, S.A. Retinal vascular branching in healthy and diabetic subjects. Investig. Ophthalmol. Vis. Sci. 2017, 58, 2685–2694. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ding, J.; Ikram, M.K.; Cheung, C.Y.; Wong, T.Y. Retinal vascular calibre as a predictor of incidence and progression of diabetic retinopathy. Clin. Exp. Optom. 2012, 95, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Bek, T. Diameter Changes of Retinal Vessels in Diabetic Retinopathy. Curr. Diab. Rep. 2017, 17, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Gerendas, B.S.; Prager, S.; Deak, G.; Simader, C.; Lammer, J.; Waldstein, S.M.; Guerin, T.; Kundi, M.; Schmidt-Erfurth, U.M. Predictive imaging biomarkers relevant for functional and anatomical outcomes during ranibizumab therapy of diabetic macular oedema. Br. J. Ophthalmol. 2018, 102, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Spaide, R.F. Improving the age-related macular degeneration construct: A New Classification System. RETINA 2018, 38, 891–899. [Google Scholar] [CrossRef] [PubMed]

| S/N | Author | Sample Size | Demographics | Main Results | HR (95% CI) | Adjustment Parameters | Follow-Up (Years) | |
|---|---|---|---|---|---|---|---|---|
| Age (Years) | % Males | |||||||
| Retinal vascular diameter | ||||||||
| 1 | Yip (2017) [6] | n= 1256 | 53.6–62.4 ** | 36.9–57.7 | CRAE and CRVE were associated with incident CKD | 1.34 (1.0–1.78) for CRAE (per SD ↓) and 2.35 (1.12–5.94) for CRVE (per SD ↑) | Age, sex, education level, eGFR, glucose levels, SBP, hypertension, smoking, anti-hypertensive medications, hsCRP, total cholesterol, HDL, fellow retinal vessel caliber (CRAE in models including CRVE and vice versa) | 6 * |
| 2 | Yau (2011) [9] | n = 4594 | 59–69 ** | 40.1–49.1 | CRAE was associated with incident CKD in whites | 1.78 (1.01–3.15) for CRAE in tertile 1 | Age, sex, study center, venular caliber, SBP, DM, medication for diabetes, antihypertensive medications, BMI, triglycerides, smoking, fasting glucose level, CRP, HbA1c level, logACR, any retinopathy | 4.8 * |
| 3 | Sabanayagam (2011) [25] | N = 3302 | 54.1–71.7 ** | 26–48.8 | Retinal vascular diameter was not associated with incident CKD | - | Age, sex, diabetes, hypertension, education, smoking, alcohol intake, BMI, diabetes, glycated hemoglobin, CRP, total cholesterol and HDL, cholesterol | 15 * |
| Retinopathy | ||||||||
| 1 | Yip (2017) [6] | n = 1256 | 53.6–62.4 ** | 36.9–57.7 | Retinopathy was associated with incident CKD | 2.54 (1.48–4.36) | Age, sex, education level, baseline eGFR, glucose levels, SBP, hypertension, smoking, anti-hypertensive medications, hsCRP, total cholesterol, HDL cholesterol | 6 * |
| S/N | Author | Sample Size | Demographics | Main Results | HR (95% CI) | Adjustment Parameters | Follow-Up (Years) | |
|---|---|---|---|---|---|---|---|---|
| Age (Years) | % Males | |||||||
| Retinal vascular diameter | ||||||||
| 1 | Baumann (2014) [26] | n = 164 | 60.8 * | 57 | CRAE and CRAE + albuminuria were associated with incident ESRD | 3.0 (1.2–7.5) for CRAE (in tertile 1) and 10.0 (2.6–38.7) for CRAE + albuminuria | Age, eGFR, DM, SBP, RAAS inhibition | 3.8 * |
| 2 | Grunwald (2014) [12] | n = 1852 | 62 * | 54.1 | AVR was associated with incident ESRD | 3.11 (1.51–6.4) for AVR in quartile 4. 1.32 (1.03–1.7) for AVR Per SD ↑ | Age, SBP, race, DM, BMI, smoking, clinical site, 24 h urine protein, eGFR | 2.3 * |
| 3 | Yip (2015) [8] | n = 5763 | 55.1 * | 48.7 | Retinal vascular diameter was not associated with incident ESRD | - | Age, gender, race, hypertension, DM, eGFR | 4.3 * |
| Retinopathy | ||||||||
| 1 | Grunwald (2014) [12] | n = 1852 | 62 * | 54.1 | Retinopathy was not associated with incident ESRD in the fully adjusted model | 1.26 (0.76–2.11) | Age, SBP, race, DM, BMI, smoking, clinical site, 24 h urine protein, eGFR | 2.3 * |
| 2 | Yip (2015) [8] | n = 5763 | 55.1 * | 48.7 | Retinopathy was associated with incident ESRD | 2.51 (1.14–5.54) | Age, gender, race, hypertension, DM, eGFR | 4.3 * |
| S/N | Author | Sample Size | Demographics | Main Results | OR (95% CI) | Adjustment Parameters | Follow-Up (Years) | |
|---|---|---|---|---|---|---|---|---|
| Age (Years) | % Males | |||||||
| Retinal vascular diameter | ||||||||
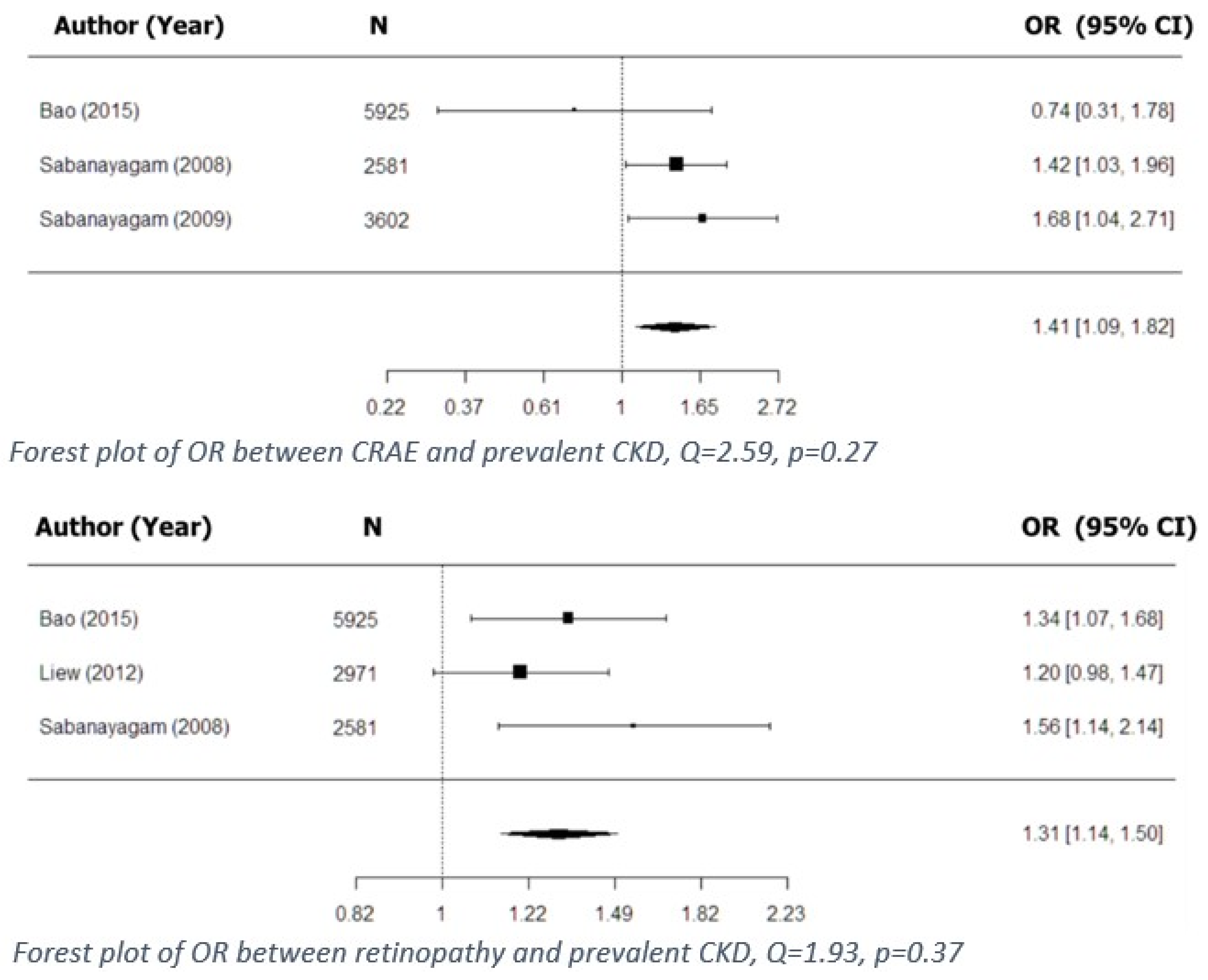
| 1 | Bao (2015) [3] | n = 5925 | 59.1 * | 45.3 | AVR was associated with prevalent CKD | 1.24 (1.0–1.53) for AVR in quartile 1 | Age, sex, smoking, alcohol consumption, BMI, education, total cholesterol, triglycerides, LDL and HDL levels | 1–2 * |
| 2 | Sabanayagam (2008) [15] | n = 3280 | 56.4–58.5 ** | 39.6–57.7 | CRAE was associated with prevalent CKD | 1.42 (1.03–1.96) for CRAE in quartile 1; 1.11 (1.0–1.24) for CRAE per SD ↓ | Age, gender, smoking, DM, hypertension, BMI, total cholesterol, HDL levels | 1.8 * |
| 3 | Sabanayagam (2009) [14] | n = 3602 | 45.2–54.7 ** | 44.4–51.7 | CRAE was associated with prevalent CKD | 1.68 (1.04–2.71) for CRAE in quartile 1; 1.2 (1.02–1.4) for CRAE per SD ↓; 3.61 (1.86–6.93) for CRAE + hypertension | Age, sex, ethnicity, education, smoking, alcohol consumption, DM, mean arterial blood pressure, BMI, total cholesterol, HDL cholesterol | 4 * |
| 4 | Liew (2012) [27] | n = 2971 | 59.1–71.2 ** | - | CRVE was associated with prevalent CKD | 1.2 (1.0–1.5) for CRVE in quintile 5 | Age, gender, fasting plasma glucose, SBP | – |
| 5 | Lim (2013) [16] | n = 3280 | 56.8–66.1 ** | - | CRAE and CRVE were not associated with prevalent CKD | - | Age, sex, hypertension, DM, smoking, history of stroke, BMI, lipids, education | 1–3 * |
| 6 | Phan (2016) [28] | n = 1512 | 60–69.4 ** | 65.1–77.3 | CRAE and CRVE were not associated with prevalent CKD | - | Age, sex, BMI, ethnicity, hypertension, DM, cholesterol level | 3.5 * |
| 7 | Gu (2015) [29] | n = 292 | 65.7–72 ** | 45–53.7 | CRAE < 150 μm discriminated CKD stage 1 from stage 2 and beyond | 2.81 (1.68–4.69) | Sex, age, DBP, smoking | 4 * |
| 8 | Ooi (2011) [30] | n = 252 | 60.5–61 ** | 65.1 | CRAE and CRVE discriminated CKD stage 1–2 from stage 3–5 | 2.84 (1.25–6.46) for CRAE in quartile 1; 4.75 (2.00–11.3) for CRVE in quartile 1 | Age, gender, hypertension, DM, dyslipidemia, smoking history | |
| Retinopathy | ||||||||
| 1 | Bao (2015) [3] | n = 5925 | 59.1 ** | 45.3 | Retinopathy was associated with prevalent CKD | 1.34 (1.07–1.68) | Age, sex, smoking, alcohol consumption, BMI, education, total cholesterol, triglycerides, LDL and HDL levels | 1–2 * |
| 2 | Liew (2012) [27] | n = 2971 | 59.1–71.2 ** | - | Retinopathy was associated with prevalent CKD | 1.2 (1.0–1.5) | Age, gender, fasting plasma glucose, SBP | - |
| 3 | Sabanayagam (2008) [15] | n = 3280 | 56.4–58.5 ** | 39.6–57.7 | Retinopathy was associated with prevalent CKD | 1.56 (1.14–2.14) | Age, gender, smoking, DM, hypertension, BMI, total cholesterol, HDL levels | 1.8 * |
| 4 | Wong (2004) [2] | n = 10,056 | 59.7–61.8 ** | 43.9–58.5 | Any retinopathy, microaneurysms, retinal hemorrhages, and soft exudates were associated with renal dysfunction ** | 2.0 (1.4–2.8) for any retinopathy; 2.0 (1.3–3.1) for microaneurysms; 2.6 (1.6–4.0) for retinal hemorrhages; 2.7 (1.6–4.8) for soft exudates | Age, gender, race, field center, DM, fasting glucose, antihypertensive medication, MABP, fasting HDL cholesterol and triglyceride, BMI, smoking, alcohol consumption | 6 * |
| Other retinal microvascular signs | ||||||||
| 1 | Liew (2012) [27] | n = 2971 | 59.1–71.2 ** | - | Retinal microvascular signs were not associated with prevalent CKD | - | Age, gender, fasting plasma glucose, SBP | - |
| 2 | Lim (2013) [16] | n = 3280 | 56.8–66.1 ** | - | Retinal microvascular signs were not associated with prevalent CKD | - | Age, sex, hypertension, DM, smoking, history of stroke, BMI, lipids, education | 1–3 * |
| 3 | Sng (2010) [31] | n = 884 | 60.4 * | 55.3 | Df was associated with prevalent CKD | 2.1 (1.15–3.83) for Df in quintile 1 | Age, gender, ethnicity, DM, SBP, BMI, alcohol consumption, smoking status, total cholesterol, HDL cholesterol, DR, DR treatment | - |
| 4 | Wong (2004) [2] | n = 10,056 | 59.7–61.8 ** | 43.9–58.5 | AVN was associated with renal dysfunction *** | 1.4 (1.0–1.9) | Age, gender, race, field center, DM, fasting glucose, antihypertensive medication, MABP, fasting HDL cholesterol and triglyceride, BMI, smoking, alcohol consumption | 6 * |
| 5 | Vadala (2018) [13] | n = 120 | 50.5 * | 68.3 | Decreased retinal and choroidal thickness, superficial and deep parafoveal vascular density, were associated with prevalent CKD | p < 0.05 for each of the parameters examined ** | - | 1 * |
| S/N | Author | Sample Size | Demographics | Main Results | OR (95% CI) | Adjustment Parameters | Follow-Up (Years) | |
|---|---|---|---|---|---|---|---|---|
| Age (Years) | % Males | |||||||
| 1 | Yip (2015) [8] | n = 5763 | 55.1 * | 48.7 | Retinopathy was associated with prevalent ESRD | 3.21 (1.28, 8.05) | Age, gender, race | 4.3 * |
| 2 | Grunwald (2019) [32] | n = 1583 | 60 * | 54.1 | Changes in CRAE or CRVE, and retinopathy worsening, were not associated with prevalent ESRD or 50% decline in eGFR between initial and follow-up photography | - | eGFR, level of retinopathy, age, race, BMI, smoking, DM, SBP, clinical site (adjustment for retinopathy); eGFR, retinal vascular diameter, age, race, BMI, smoking, DM, SBP, clinical site (adjustment for retinal vascular diameter) | 3.5 * |
| S/N | Author | Sample Size | Demographics | Main Results | OR (95% CI)/β (95% CI/±SE) | Adjustment Parameters | Follow-Up (Years) | |
|---|---|---|---|---|---|---|---|---|
| Age (Years) | % Males | |||||||
| Retinal vascular diameter | ||||||||
| 1 | Lim (2013) [16] | n = 3280 | 56.8–66.1 ** | - | CRAE was associated with lower eGFR | β 0.77 (0.20–1.35) for CRAE (per SD ↑) | Age, sex, hypertension, DM, smoking, history of stroke, BMI, lipids, education | 1–3 * |
| 2 | Baumann (2009) [33] | n = 67 | 61.5–62.3 ** | 50–61.5 | CRAE was associated with eGFR decline | CRAE independently predicted eGFR (β = 0.33) | - | - |
| 3 | Gu (2015) [29] | n = 292 | 65.7–72 ** | 45–53.7 | CRAE was associated with eGFR decline | β 2.27 (0.53–4.01) for CRAE per SD ↓ | Sex, age, DBP, smoking | 4 * |
| Retinopathy | ||||||||
| 1 | Edwards (2005) [34] | n = 1394 | 78 * | 38.9 | Any retinopathy, retinal hemorrhages, microaneurysms and hard/soft exudates were associated with 20% or more decline in eGFR | OR 2.84 (1.56–5.16) for any retinopathy; OR 2.18 (1.01–4.71) for retinal hemorrhages; OR 2.25 (1.0–5.09) for microaneurysms; OR 6.63 (2.7–16.3) for hard/soft exudates | Age, race, sex, weight, DM, hypertension, ACE inhibitor use, proteinuria | 5 * |
| 2 | Grunwald (2019) [32] | n = 1583 | 60 * | 54.1 | Retinopathy worsening was not associated with change in eGFR slope | - | eGFR, retinopathy level, age, race/ethnicity, BMI, smoking | 3.5 * |
| 3 | Hwang (2016) [35] | n = 523 | 63.9–73.2 ** | 48.5–57.6 | Patients with retinopathy showed a faster eGFR decline than those without retinopathy | −7.2 ± 10.2 vs. −3.1 ± 10.1 | Age, BMI, DM, smoking, previous CV events, serum albumin level | 2–2.7 * |
| Other retinal microvascular signs | ||||||||
| 1 | Edwards (2005) [34] | n = 1394 | 78 * | 38.9 | AVN and FAN were not associated with change in eGFR | - | Age, race, sex, weight, DM, hypertension, ACE inhibitor use, proteinuria | 5 * |
| 2 | Lim (2013) [16] | n = 3280 | 56.8–66.1 ** | - | Df and AVN were associated with lower eGFR | β 0.83 (0.17–1.48) for Df; β −3.09 (−5.25–(−0.94)) for AVN | Age, sex, hypertension, DM, smoking, history of stroke, BMI, lipids, education | 1–3 * |
| S/N | Author | Sample Size | Demographics | Main Results | OR (95% CI)/β (95% CI) | Adjustment Parameters | Follow-Up (Years) | |
|---|---|---|---|---|---|---|---|---|
| Age (Years) | % Males | |||||||
| Retinal vascular diameter | ||||||||
| 1 | Bao (2015) [3] | n = 5925 | 59.1 * | 45.3 | AVR was associated with albuminuria | OR 1.26 (1.01–1.56) for AVR in quartile 1 | Age, sex, smoking, alcohol consumption, BMI, education, total cholesterol, triglycerides, LDL and HDL levels | 1–2 * |
| 2 | Awua-larbi (2011) [20] | n = 5897 | 63.2 * | 47.9 | CRAE was associated with albuminuria | OR 1.55 (1.17–2.04) for CRAE in quintile 1; OR 1.44 (1.07–1.93) for CRAE in quintile 5 | Age, sex, race, SBP, antihypertensive drugs, use of RAAS inhibitor, DM, smoking, waist circumference, education, access to healthcare | - |
| 3 | Garcia-Ortiz (2012) [36] | n = 205 | 55.6 * | 57.1 | CRVE and AVR were associated with albuminuria | β −27.350 (−53.66–(− 1.039)) for AVR; β 0.18 (0.04–0.32) for CRVE | Sex, age | 2 * |
| 4 | Lim (2013) [16] | n = 3280 | 56.8–66.1 ** | − | Retinal vascular diameter was not associated with albuminuria | - | Age, sex, hypertension, DM, smoking, history of stroke, BMI, lipids, education | 3 * |
| 5 | Sabanayagam (2008) [15] | n = 3280 | 56.4–58.5 ** | 39.6–57.7 | CRAE and CRVE were associated with albuminuria | OR 1.80 (1.11–2.91) for CRAE in quartile 1 and OR 1.22 (1.03–1.44) for CRAE per SD ↓; OR 1.63 (1.02–2.60) for CRVE in quartile 2 | Age, gender, smoking, DM, hypertension, BMI, total cholesterol, HDL levels | 1.8 * |
| Retinopathy | ||||||||
| 1 | Bao (2015) [3] | n = 5925 | 59.1 * | 45.3 | Retinopathy was associated with albuminuria | OR 1.34 (1.06–1.68) | Age, sex, smoking, alcohol consumption, BMI, education, total cholesterol, triglycerides, LDL and HDL levels | 1–2 * |
| 2 | Sabanayagam (2008) [15] | n = 3280 | 56.4–58.5 ** | 39.6–57.7 | Retinopathy was associated with micro/macroalbuminuria | OR 1.88 (1.13–3.15) | Age, gender, smoking, DM, hypertension, BMI, total cholesterol, HDL levels | 1.8 * |
| Other retinal microvascular signs | ||||||||
| 1 | Lim (2013) [16] | n = 3280 | 56.8–66.1 ** | - | Df, FAN, and AVN were associated with albuminuria | β −0.21 (−0.32–(−0.10)) for Df (per SD ↑); β 0.60 (0.28–0.92) for FAN; β 0.47 (0.12–0.83) for AVN | Age, sex, hypertension, DM, smoking, history of stroke, BMI, lipids, education | 3 * |
| S/N | Author | Sample Size | Demographics | Main Results | HR (95% CI) | Adjustment Parameters | Follow-Up (Years) | |
|---|---|---|---|---|---|---|---|---|
| Age (Years) | % Males | |||||||
| Retinal vascular diameter | ||||||||
| 1 | Yip (2015) [8] | n = 5763 | 55.1 * | 48.7 | CRAE and CRVE were not associated with incident ESRD | - | Age, gender, race, hypertension, eGFR, HbA1c | 4.3 * |
| Retinopathy | ||||||||
| 1 | Yip (2015) [8] | n = 5763 | 55.1 * | 48.7 | Retinopathy was associated with incident ESRD | 2.6 (1.01–6.66) | Age, gender, race, hypertension, eGFR, HbA1c | 4.3 * |
| 2 | Lee (2014) [38] | n = 51 | 58.2 * | 60.8 | Nonperfusion area ≥ 10 disc areas (on FA) was associated with incident ESRD | 6.64 (1.96–22.52) | - | 2 * |
| Other retinal microvascular signs | ||||||||
| 1 | Yip (2015) [8] | n = 5763 | 55.1 * | 48.7 | Df was not associated with incident ESRD | - | Age, gender, race, hypertension, eGFR, HbA1c | 4.3 * |
| S/N | Author | Sample Size | Demographics | Main Results | OR (95% CI) | Adjustment Parameters | Follow-Up (Years) | |
|---|---|---|---|---|---|---|---|---|
| Age (Years) | % Males | |||||||
| Retinal vascular diameter | ||||||||
| 1 | Bao (2015) [3] | n = 5925 | 59.1 * | 45.3 | CRAE, CRVE, and AVR were not associated with prevalent CKD | - | Age, sex, smoking, alcohol consumption, BMI, education, total cholesterol, triglycerides, LDL and HDL levels | 1–2 * |
| 2 | Liew (2012) [27] | n = 2971 | 59.1–71.2 ** | - | CRVE was not associated with prevalent CKD | 1.2 (0.6–2.4) for CRVE in quintile 5 | Age, gender, fasting plasma glucose, SBP | - |
| 3 | Mckay (2018) [39] | n = 1072 | 63 * | 51 | CRAE, CRVE, and AVR were not associated with reduced renal function *** | - | Age, gender, SBP, HbA1c | 3 * |
| Retinopathy | ||||||||
| 1 | Sabanayagam (2008) [8] | n = 3280 | 56.4–58.5 ** | 39.6–57.7 | Retinopathy was associated with prevalent CKD | 1.50 (1.00–2.25) | Age, gender, smoking, DM, hypertension, BMI, total cholesterol, HDL cholesterol | 1.8 * |
| 2 | Liew (2011) [27] | n = 2971 | 59.1–71.2 ** | Retinopathy was associated with prevalent CKD | 1.3 (0.7–2.5) | Age, gender, fasting plasma glucose, SBP | - | |
| 3 | Bao (2015) [3] | n = 5925 | 59.1 * | 45.3 | Retinopathy was associated with prevalent CKD | 1.63 (0.35–7.58) | Age, sex, smoking, alcohol consumption, BMI, education, total cholesterol, triglycerides, LDL and HDL levels | 1–2 * |
| 4 | Zhang (2014) [17] | n = 523 | 36–59.7 ** | 54–61 | NPDR and PDR were associated with prevalent CKD | 2.22 (1.01–4.86) for NPDR; 3.52 (1.3–9.55) for PDR | Age, gender, SBP, hypertension, HbA1c, duration of diabetes | - |
| 5 | Mottl (2020) [19] | n = 1292 | 60.4–72 ** | 40–51 | Retinopathy was associated with prevalent CKD in subgroups of the study population | 2.7 (1.2–6.1) for non-Hispanic blacks; 2.6 (1.3–5.5) for obesity; 2.5 (1.1–5.7) for patients not using RAAS blockers | Age, gender, HbA1c, SBP, DBP | - |
| 6 | Wong (2004) [37] | n = 10,056 | 59.7–61.8 ** | 43.9–58.5 | Retinopathy was associated with renal dysfunction † | 2.6 (1.6–4.3) for diabetic patients; 2.1 (1.2–3.8) for diabetes + hypertension | Age, gender, race, field center, DM, fasting glucose, antihypertensive medication, MABP, fasting HDL cholesterol, triglyceride, BMI, smoking, alcohol consumption | 6 * |
| Other retinal microvascular signs | ||||||||
| 1 | Mckay (2018) [39] | n = 1072 | 63 * | 51 | Df and tortuosity were not associated with reduced renal function | - | Age, gender, SBP, HbA1c | 3 * |
| S/N | Author | Sample Size | Demographics | Main Results | OR (95% CI) | Adjustment Parameters | Follow-Up (Years) | |
|---|---|---|---|---|---|---|---|---|
| Age (Years) | % Males | |||||||
| Retinal vascular diameter | ||||||||
| 1 | Broe (2014) [18] | n = 185 | 21 * | - | CRAE and CRVE were associated with DN | 2.63 (1.09–6.36) per 10 µm ↓ for CRAE; 1.76 (1.05–2.94) per 10 µm ↑ for CRVE | - | 16 * |
| Other retinal microvascular signs | ||||||||
| 1 | Broe (2014) [40] | n = 180 | - | - | Df was associated with DN | 1.40 (1.10–1.79) per 0.01 ↓ in Df | Sex, age, duration of diabetes, HbA1c, SBP, DBP, BMI, retinopathy, VPT, albuminuria, CRAE, CRVE | 16 * |
| 2 | Rasmussen (2017) [41] | n = 181 | 37 * | 50.8 | Arteriolar BC was associated with DN | 3.1 (1.01–9.54) | Sex, age, duration of diabetes, SBP, DBP, HbA1c, retinopathy, VPT | - |
| S/N | Author | Sample Size | Demographics | Main Results | OR (95% CI)/β (95% CI) | Adjustment Parameters | Follow-Up (Years) | |
|---|---|---|---|---|---|---|---|---|
| Age (Years) | % Males | |||||||
| Retinal vascular diameter | ||||||||
| 1 | Edwards (2005) [34] | n = 1394 | 78 * | 38.9 | AVR was not associated with 20% decline in eGFR | - | Cr, age, sex, race, weight, BP, ACE-inhibitor use, albuminuria/proteinuria | 9 * |
| 2 | Mckay (2018) [39] | n = 1072 | 63 * | 51 | CRAE, CRVE, and AVR were not associated with eGFR decline at follow-up | - | Age, gender, SBP, HbA1c | 3 * |
| Retinopathy | ||||||||
| 1 | Edwards (2005) [34] | n = 1394 | 78 * | 38.9 | Microaneurysms and hard/soft exudates were associated with 20% or more decline in eGFR | OR 4.1 (1.04–16.3) for microaneurysms; OR 7.09 (1.1–45.6) for hard/soft exudates | Cr, age, sex, race, weight, BP, ACE-inhibitor use, albuminuria/proteinuria | 9 * |
| 2 | Grunwald (2019) [32] | n = 1583 | 60 * | 54.1 | eGFR slope between patients with retinopathy worsening did not differ from eGFR slope in patients without retinopathy worsening | - | eGFR, retinopathy, age, race, BMI, smoking | 3.5 * |
| Other retinal microvascular signs | ||||||||
| 1 | Edwards (2005) [34] | n = 1394 | 78 * | 38.9 | AVN and FAN were not associated with 20% decline in eGFR | - | Cr, age, sex, race, weight, BP, ACE-inhibitor use, albuminuria/proteinuria | 9 * |
| 2 | Mckay (2018) [39] | n = 1072 | 63 * | 51 | Df and tortuosity were not associated with eGFR decline at follow-up | - | Age, gender, SBP, HbA1c | 3 * |
| S/N | Author | Sample Size | Demographics | Main Results | OR (95% CI) | Adjustment Parameters | Follow-Up (Years) | |
|---|---|---|---|---|---|---|---|---|
| Age (Years) | % Males | |||||||
| Retinal vascular diameter | ||||||||
| 1 | Benitez (2018) [42] | n = 963 | 14.4 * | 53 | exMWa was associated with high risk for albuminuria | 1.67 (1.17–2.38) for exMWa; 1.39 (0.98–1.99) for exMWv | BMI SDS, duration, and SBP SDS | - |
| 2 | Grauslund (2009) [44] | n = 208 | 57.9 * | - | CRAE and AVR were associated with albuminuria | 2.17 (1.29–3.68) for CRAE per SD ↓; 1.48 (1.01–2.16) for AVR per SD ↓ | Age, sex, duration of diabetes, HbA1c, SBP, pack years | - |
| 3 | Awua-larbi (2011) [20] | n = 5897 | 63.2 * | 47.9 | CRAE was associated with albuminuria | 2.26 (1.34–3.81) for CRAE in quintile 1 | Age, sex, race, SBP, antihypertensive medication, RAAS inhibitors, smoking, waist circumference, education, access to healthcare | - |
| 4 | Keel (2017) [21] | n = 483 | 14.5 * | 53 | CRAE, CRVE, and AVR were not associated with albuminuria | - | Age, sex, ethnicity, HbA1c, total and LDL cholesterol, BMI, duration of diabetes, SBP | - |
| 5 | Bao (2015) [3] | n = 5925 | 59.1 * | 45.3 | CRAE was associated with albuminuria | 2.17 (1.03–4.56) for CRAE in quartile 2 | Age, sex, BMI, education, hypertension, diabetes, smoking, drinking, total cholesterol, triglyceride, LDL, HDL | 1–2 * |
| Retinopathy | ||||||||
| 1 | Mottl (2012) [19] | n = 1292 | 60.4–72 ** | 40–51 | Any retinopathy and moderate-to-severe retinopathy were associated with micro/macro-albuminuria | 1.8 (1.1–2.8) for any retinopathy; 2.7 (1.4–5.5) for moderate-severe retinopathy | - | - |
| 2 | Sabanayagam (2008) [15] | n = 3280 | 56.4–58.5 ** | 39.6–57.7 | Retinopathy was not associated with albuminuria | - | Age, gender, smoking, diabetes, hypertension, BMI, total cholesterol, HDL cholesterol | 1.8 * |
| 3 | Bao (2015) [3] | n = 5925 | 59.1 * | 45.3 | Retinopathy was not associated with albuminuria | - | Age, sex, BMI, education, hypertension, diabetes, smoking, drinking, total cholesterol, triglyceride, LDL, HDL | 1–2 * |
| 4 | Ha M (2019) [45] | n = 103 | 61–67.3 ** | 20–31 | Vitreous hemorrhage showed a higher incidence in the microalbuminuria group and in the advanced nephropathy group than in the no nephropathy group | p = 0.017 (this study was retrospective) | - | - |
| Other retinal microvascular signs | ||||||||
| 1 | Grauslund (2010) [46] | n = 208 | 57.8 * | 46.7–62.4 | Df was not associated with albuminuria | - | Age, gender, duration of diabetes, SBP, smoking | - |
| 2 | Benitez (2012) [43] | n = 666 | 13.5 * | 47–54 | LDRv and STv were associated with increased AER | 1.69 (1.17–2.44) for LDRv in quartile 4; 1.55 (1.08–2.22) for STv in quartile 1 | Age, diabetes duration, HbA1c, BP, BMI, cholesterol | 3.7 ** |
| 3 | Sasongko (2012) [47] | n = 944 | 13.6–14 ** | 44.1–49.8 | Arteriolar tortuosity index was associated with increased AER | 1.56 (1.06–2.28) for arteriolar tortuosity index per SD ↑ | Age, sex, duration of diabetes, HbA1c, SBP, cholesterol, BMI, retinal vessel diameter | - |
| 4 | Cankurtaran (2019) [5] | n = 137 | 54.8–56.7 ** | 45.4–54.7 | SCP, DCP vessel densities, whole disc, and peripapillary areas in microalbuminuric patients differed significantly from those in normoalbuminuric or control patients | p < 0.05 in all comparisons (multilinear regression analysis was not performed) | - | - |
| 5 | Garrido (2019) [48] | n = 21 | 49.76 * | - | CMT and CT were associated with albuminuria | p < 0.05 in all comparisons (only bivariate analysis was performed) | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aronov, M.; Allon, R.; Stave, D.; Belkin, M.; Margalit, E.; Fabian, I.D.; Rosenzweig, B. Retinal Vascular Signs as Screening and Prognostic Factors for Chronic Kidney Disease: A Systematic Review and Meta-Analysis of Current Evidence. J. Pers. Med. 2021, 11, 665. https://doi.org/10.3390/jpm11070665
Aronov M, Allon R, Stave D, Belkin M, Margalit E, Fabian ID, Rosenzweig B. Retinal Vascular Signs as Screening and Prognostic Factors for Chronic Kidney Disease: A Systematic Review and Meta-Analysis of Current Evidence. Journal of Personalized Medicine. 2021; 11(7):665. https://doi.org/10.3390/jpm11070665
Chicago/Turabian StyleAronov, Michael, Raviv Allon, Danielle Stave, Michael Belkin, Eyal Margalit, Ido Didi Fabian, and Barak Rosenzweig. 2021. "Retinal Vascular Signs as Screening and Prognostic Factors for Chronic Kidney Disease: A Systematic Review and Meta-Analysis of Current Evidence" Journal of Personalized Medicine 11, no. 7: 665. https://doi.org/10.3390/jpm11070665
APA StyleAronov, M., Allon, R., Stave, D., Belkin, M., Margalit, E., Fabian, I. D., & Rosenzweig, B. (2021). Retinal Vascular Signs as Screening and Prognostic Factors for Chronic Kidney Disease: A Systematic Review and Meta-Analysis of Current Evidence. Journal of Personalized Medicine, 11(7), 665. https://doi.org/10.3390/jpm11070665






