Correlations between Electrophysiological Parameters, Lymphocyte Distribution and Cytokine Levels in Patients with Chronic Demyelinating Inflammatory Polyneuropathy
Abstract
:1. Introduction
2. Materials and Methods
2.1. Blood Collection and Staining for Markers of Leukocyte Subpopulations
2.2. Serum Collection, Storage and Cytokine Analysis
2.3. Electroneurography
3. Statistical Analysis
Data Availability
4. Results
5. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dimachkie, M.; Barohn, R.; Katz, J. Multifocal Motor Neuropathy, Multifocal Acquired Demyelinating Sensory and Motor Neuropathy and Other Chronic Acquired Demyelinating Polyneuropathy Variants. Neurol. Clin. 2013, 31, 533–555. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schafflick, D.; Kieseier, B.C.; Wiendl, H.; Meyer zu Horste, G. Novel pathomechanisms in inflammatory neuropathies. J. Neuroinflammation 2017, 14, 232. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Finsterer, J.; Löscher, W.N.; Wanschitz, J.; Iglseder, S. Orphan Peripheral Neuropathies. J. Neuromuscul. Dis. 2021, 8, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Lawson, V.H.; Arnold, W.D. Multifocal motor neuropathy: A review of pathogenesis, diagnosis, and treatment. Neuropsychiatr. Dis. Treat. 2014, 5, 567–576. [Google Scholar] [CrossRef] [Green Version]
- Olney, R.K.; Lewis, R.A.; Putnam, T.D.; Campellone, J.V. Consensus criteria for the diagnosis or multifocal motor neuropathy. Muscle Nerve 2003, 27, 117–121. [Google Scholar] [CrossRef]
- Jann, S.; Beretta, S.; Bramerio, M.A. Different types of chronic inflammatory demyelinating polyneuropathy have a different clinical course and response to treatment. Muscle Nerve 2005, 32, 351–356. [Google Scholar] [CrossRef]
- Sharma, K.R.; Cross, J.; Farronay, O.; Ayyar, D.R.; Shebert, R.T.; Bradley, W.G. Demyelinating neuropathy in diabetes mellitus. Arch. Neurol. 2002, 59, 758–765. [Google Scholar] [CrossRef] [Green Version]
- Sun, Q.L.; Sun, A.P.; Fu, Y.; Fan, D.S. The clinical analysis of diabetic patients with chronic inflammatory demyelinating polyradiculoneuropathy. Zhonghua Nei Ke Za Zhi 2018, 57, 749–752. [Google Scholar] [CrossRef]
- Fisse, A.; Motte, J.; Grüter, T.; Sgodzai, M.; Pitarokoili, K.; Gold, R. Comprehensive approaches for diagnosis, monitoring and treatment of chronic inflammatory demyelinating polyneuropathy. Neurol. Res. Pract. 2020, 2, 42. [Google Scholar] [CrossRef] [PubMed]
- Ubogu, E.E. Inflammatory Neuropathies: Pathology, molecular markers and targets for specific therapeutic intervention. Acta Neuropathol. 2015, 130, 445–468. [Google Scholar] [CrossRef] [Green Version]
- Mathey, E.K.; Park, S.B.; Hughes, R.A.; Pollard, J.D.; Armati, P.J.; Barnett, M.H.; Taylor, B.V.; Dyck, P.J.B.; Kiernan, M.C.; Lin, C.S.-Y. Chronic inflammatory demyelinating polyradiculoneuropathy: From pathology to phenotype. J. Neurol. Neurosurg. Psychiatry 2015, 86, 973–985. [Google Scholar] [CrossRef] [Green Version]
- Joshi, A.R.; Holtmann, L.; Bobylev, I.; Schneider, C.; Ritter, C.; Weis, J.; Lehmann, H.C. Loss of Schwann cell plasticity in chronic inflammatory demyelinating polyneuropathy (CIDP). J. Neuroinflammation 2016, 13, 255. [Google Scholar] [CrossRef] [Green Version]
- Yang, X.; Jin, T.; Press, R.; Quezada, H.C.; Fredrikson, S.; Zhu, J. The expression of TNF-alpha receptors 1 and 2 on peripheral blood mononuclear cells in chronic inflammatory demyelinating polyneuropathy. J. Neuroimmunol. 2008, 30, 129–132. [Google Scholar] [CrossRef]
- Madia, F.; Frisullo, G.; Nociti, V.; Conte, A.; Luigetti, M.; Del Grande, A.; Patanella, A.K.; Iorio, R.; Tonali, P.A.; Batocchi, A.P.; et al. pSTAT1, pSTAT3, and T-bet as markers of disease activity in chronic inflammatory demyelinating polyradiculoneuropathy. J. Peripher. Nerv. Syst. 2009, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Athanasopoulos, D.; Motte, J.; Grüter, T.; Köse, N.; Yoon, M.-Y.; Otto, S.; Schneider-Gold, C.; Gold, R.; Fisse, A.L.; Pitarokoiliet, K. Evaluation of the EFNS/PNS diagnostic criteria in a cohort of CIDP patients. Ann. Clin. Transl. Neurol. 2021, 8, 1110–1121. [Google Scholar] [CrossRef]
- Van den Bergh, P.Y.; Hadden, R.D.; Bouche, P. European Federation of Neurological Societies/Peripheral Nerve Society guideline on management of chronic inflammatory demyelinating polyradiculoneuropathy: Report of a joint task force of the European Federation of Neurological Societies and the Peripheral Nerve Society—First revision. Eur. J. Neurol. 2010, 17, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Research Criteria for Diagnosis of Chronic Inflammatory Demyelinating Polyneuropathy (CIDP). Report from an ad hoc subcommittee of the American Academy of Neurology AIDS task force. Neurology 1991, 41, 617–618. [Google Scholar]
- Mukaka, M.M. Statistics corner: A guide to appropriate use of correlation coefficient in medical research. Malawi Med. J. 2012, 24, 69–71. [Google Scholar] [PubMed]
- Quan, S.; Sheng, J.R.; Abraham, P.M.; Soliven, B. Regulatory T and B lymphocytes in a spontaneous autoimmune polyneuropathy. Clin. Exp. Immunol. 2016, 184, 50–61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alberts, B.; Johnson, A.; Lewis, J. T Cells and MHC Proteins. In Molecular Biology of the Cell, 4th ed.; Garland Science: New York, NY, USA, 2002. Available online: https://www.ncbi.nlm.nih.gov/books/NBK26926/ (accessed on 1 May 2021).
- Yang, M.; Peyret, C.; Shi, X.Q.; Siron, N.; Jang, J.H.; Wu, S.; Fournier, S.; Zhang, J. Evidence from Human and Animal Studies: Pathological Roles of CD8+ T Cells in Autoimmune Peripheral Neuropathies. Front. Immunol. 2015, 6, 532. [Google Scholar] [CrossRef] [Green Version]
- Tanaka, T.; Narazaki, M.; Kishimoto, T. IL-6 in Inflammation, Immunity, and Disease. Cold Spring Harb. Perspect. Biol. 2014, 6, a016295. [Google Scholar] [CrossRef]
- Mei, F.-J.; Ishizu, T.; Murai, H.; Osoegawa, M.; Minohara, M.; Zhang, K.-N.; Kira, J. Th1 shift in CIDP versus Th2 shift in vasculitic neuropathy in CSF. J. Neurol. Sci. 2005, 228, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Podwinska, J.; Lusiak, M.; Zaba, R.; Bowszyc, J. The pattern and level of cytokines secreted by Th1 and Th2 lymphocytes of syphilitic patients correlate to the progression of the disease. FEMS Immunol. Med. Microbiol. 2000, 28, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Huehnchen, P.; Muenzfeld, H.; Boehmerle, W.; Endres, M. Blockade of IL-6 signaling prevents paclitaxel-induced neuropathy in C57Bl/6 mice. Cell Death Dis. 2020, 11, 45. [Google Scholar] [CrossRef] [PubMed]
- Hirano, T. IL-6 in inflammation, autoimmunity and cancer. Int. Immunol. 2021, 33, 127–148. [Google Scholar] [CrossRef] [PubMed]
- Staudt, M.; Diederich, J.M.; Meisel, C.; Meisel, A.; Klehmet, J. Differences in peripheral myelin antigen-specific T cell responses and T memory subsets in atypical versus typical CIDP. BMC Neurol. 2017, 17, 81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klehmet, J.; Goehler, J.; Ulm, L.; Kohler, S.; Meisel, C.; Meisel, A.; Harms, H. Effective treatment with intravenous immunoglobulins reduces autoreactive T-cell response in patients with CIDP. J. Neurol. Neurosurg. Psychiatry 2015, 6, 686–691. [Google Scholar] [CrossRef]
- Klehmet, J.; Staudt, M.; Ulm, L.; Unterwalder, N.; Meisel, A.; Meisel, C. Circulating lymphocyte and T memory subsets in glucocorticosteroid versus IVIG treated patients with CIDP. J. Neuroimmunol. 2015, 283, 17–22. [Google Scholar] [CrossRef]
- Sanvito, L.; Makowska, A.; Gregson, N.; Nemni, R.; Hughes, R.A.C. Circulating subsets and CD4(+)CD25(+) regulatory T cell function in chronic inflammatory demyelinating polyradiculoneuropathy. Autoimmunity 2009, 42, 667–677. [Google Scholar] [CrossRef]
- Yang, M.; Shi, X.Q.; Peyret, C.; Oladiran, O.; Wu, S.; Chambon, J.; Fournier, S.; Zhang, J. Effector/memory CD8+ T cells synergize with co-stimulation competent macrophages to trigger autoimmune peripheral neuropathy. Brain Behav. Immun. 2018, 71, 142–157. [Google Scholar] [CrossRef]
- Hagen, K.M.; Ousman, S.S. The immune response and aging in chronic inflammatory demyelinating polyradiculoneuropathy. J. Neuroinflammation 2021, 18, 78. [Google Scholar] [CrossRef]
- Liu, M.; Saredy, J.; Zhang, R.; Shao, Y.; Sun, Y.; Yang, W.Y.; Wang, J.; Liu, L.; Drummer, C.; Johnson, C.; et al. Approaching Inflammation Paradoxes—Proinflammatory Cytokine Blockages Induce Inflammatory Regulators. Front. Immunol. 2020, 11, 554301. [Google Scholar] [CrossRef] [PubMed]
- Hartung, H.P.; Reiners, K.; Schmidt, B.; Stoll, G.; Toyka, K.V. Serum interleukin-2 concentrations in Guillain-Barre syndrome and chronic idiopathic demyelinating polyradiculoneuropathy: Comparison with other neurological diseases of presumed immunopathogenesis. Ann. Neurol. 1991, 30, 48–53. [Google Scholar] [CrossRef]
- Schneider-Hohendorf, T.; Schwab, N.; Uçeyler, N.; Göbel, K.; Sommer, C.; Wiendl, H. CD8+ T-cell immunity in chronic inflammatory demyelinating polyradiculoneuropathy. Neurology 2012, 78, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Matsumuro, K.; Izumo, S.; Umehara, F.; Osame, M. Chronic inflammatory demyelinating polyneuropathy: Histological and immunopathological studies on biopsied sural nerves. J. Neurol. Sci. 1994, 127, 170–178. [Google Scholar] [CrossRef]
- Cifuentes-Diaz, C.; Dubourg, O.; Irinopoulou, T.; Vigny, M.; Lachkar, S.; Decker, L.; Charnay, P.; Denisenko, N.; Maisonobe, T.; Léger, J.M.; et al. Nodes of Ranvier and paranodes in chronic acquired neuropathies. PLoS ONE 2011, 6, e14533. [Google Scholar] [CrossRef] [Green Version]
- Doppler, K.; Werner, C.; Sommer, C. Disruption of nodal architecture in skin biopsies of patients with demyelinating neuropathies. J. Peripher. Nerv. Syst. 2013, 18, 168–176. [Google Scholar] [CrossRef]
- Wolbert, J.; Cheng, M.; Meyer zu Horste, G.; Su, M.A. Deciphering immune mechanisms in chronic inflammatory demyelinating polyneuropathies. JCI Insight 2020, 5, e132411. [Google Scholar] [CrossRef]
- Gorson, K.C.; Ropper, A.H.; Adelman, L.S.; Weinberg, D.H. Influence of diabetes mellitus on chronic inflammatory demyelinating polyneuropathy. Muscle Nerve 2000, 23, 37–43. [Google Scholar] [CrossRef]
- Zier, K.S.; Leo, M.M.; Spielman, R.S.; Baker, L. Decreased synthesis of interleukin-2 (IL-2) in inusulin-dependent diabetes mellitus. Diabetes 1984, 33, 552. [Google Scholar] [CrossRef] [PubMed]
- Kobessho, H.; Oishi, K.; Hamaguchi, H.; Kanda, F. Elevation of cerebrospinal fluid protein in patients with diabetes mellitus is associated with duration of diabetes. Eur. Neurol. 2008, 60, 132–136. [Google Scholar] [CrossRef] [PubMed]

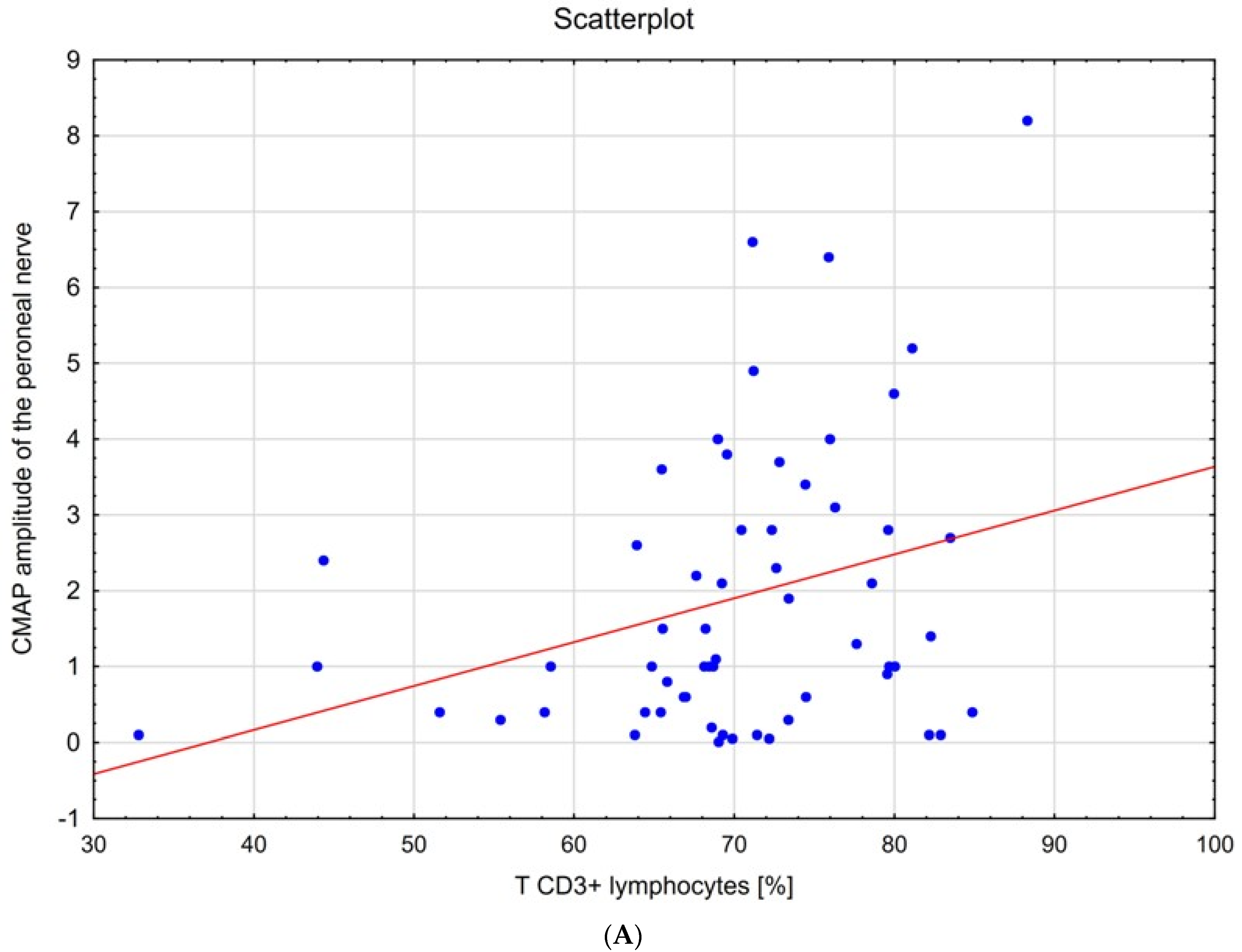

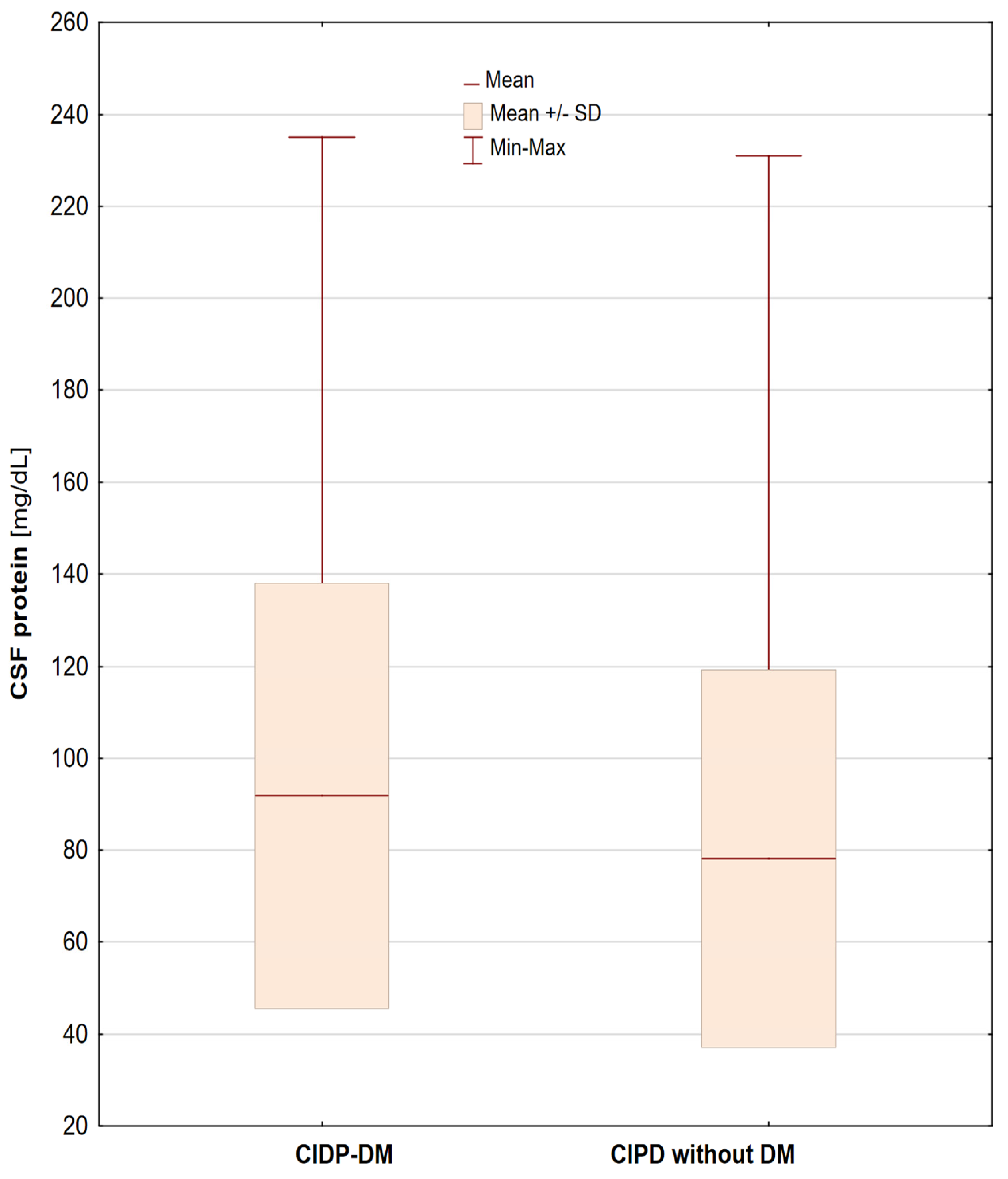
| Distal Latency ± SD [ms] | Conduction Velocity ± SD [m/s] | Amplitude ± SD [mV] | F-Wave [ms] | ||
|---|---|---|---|---|---|
| CMAP | median | 5.57 ± 3.17 | 46.84 ± 9.87 | 5.34 ± 3.42 | 36.24 ± 9.87 |
| ulnar | 3.49 ± 1.48 | 48.07 ± 9.91 | 7.16 ± 2.87 | 35.15 ± 7.94 | |
| peroneal | 6.14 ± 1.83 | 34.43 ± 12.27 | 1.98 ± 1.95 | 76.33 ± 13.71 | |
| tibial | 6.37 ± 1.76 | 34.46 ± 11.37 | 2.96 ± 2.52 | 70.45 ± 9.29 | |
| SNAP | median | 3.82 ± 0.94 | 36.90 ± 17.51 | 11.42 ± 8.97 | - |
| ulnar | 3.54 ± 0.94 | 33.72 ± 17.71 | 11.08 ± 9.34 | - | |
| sural | 4.31 ± 1.24 | 25.06 ± 21.55 | 4.29 ± 3.33 | - | |
| Leukocytes | All Patients n = 60 | Normal Results | Incorrect Results | |||
|---|---|---|---|---|---|---|
| Mean ± SD | n | Mean ± SD | n | Mean ± SD | ||
| WBC [Cells/µL] | 6937.45 ± 2064.83 | 51 | 6645.29 ± 1422.33 | 9 | 8593.00 ± 3895.14 | |
| CD45+ Leukocytes | Monocytes [%] | 8.01 ± 2.46 | 46 | 7.04 ± 1.87 | 14 | 11.21 ± 1.08 |
| Monocytes cells/µL | 553.55 ± 235.37 | 54 | 513.39 ± 170.19 | 6 | 914.97 ± 415.31 | |
| Lymphocytes [%] | 39.17 ± 13.72 | 36 | 33.08 ± 6.64 | 24 | 48.30 ± 16.45 | |
| Lymphocytes cells/µL | 2702.10 ± 1208.41 | 45 | 2671.27 ± 723.27 | 15 | 2794.59 ± 2120.75 | |
| Lymphocytes | B CD19+ Lymphocytes [%] | 11.54 ± 5.55 | 19 | 13.01 ± 1.54 | 41 | 10.86 ± 6.55 |
| B CD19+ Lymphocytes cells/µL | 325.66 ± 262.29 | 50 | 290.66 ± 148.05 | 10 | 500.66 ± 539.29 | |
| T CD3+ Lymphocytes [%] | 69.98 ± 10.03 | 16 | 73.98 ± 2.31 | 44 | 68.53 ± 11.32 | |
| T CD3+ Lymphocytes cells/µL | 1894.47 ± 869.09 | 45 | 1670.97 ± 438.67 | 15 | 2564.97 ± 1391.45 | |
| T Lymphocytes | T CD3+ CD4+ Lymphocytes [%] | 62.97 ± 12.05 | 9 | 48.69 ± 3.58 | 51 | 65.50 ± 11.23 |
| T CD3+ CD4+ Lymphocytes cells/µL | 1223.41 ± 660.56 | 38 | 1026.65 ± 295.22 | 22 | 1563.28 ± 938.76 | |
| T CD3+ CD8+ Lymphocytes [%] | 25.33 ± 10.09 | 10 | 32.19 ± 2.78 | 50 | 23.96 ± 10.47 | |
| T CD3+ CD8+ Lymphocytes cells/µL | 461.82 ± 299.29 | 42 | 468.15 ± 153.12 | 18 | 447.06 ± 488.45 | |
| CD4/CD8 | 3.16 ± 2.18 | 6 | 2.02 ± 0.12 | 54 | 3.28 ± 2.27 | |
| Cytokines | CIDP Patients n = 60 | Control Group n = 18 | p-Value |
|---|---|---|---|
| Mean ± SD [pg/mL] | Mean ± SD [pg/mL] | ||
| IFN-y | 7.72 ± 1.31 | 7.29 ± 1.96 | 0.590 |
| TNF-α | 10.04 ± 11.12 | 9.99 ± 2.92 | 0.009 |
| IL-10 | 8.15 ± 1.12 | 7.77 ± 1.75 | 0.524 |
| IL-6 | 10.18 ± 4.44 | 8.33 ± 2.65 | 0.019 |
| IL-4 | 7.98 ± 0.98 | 7.40 ± 1.33 | 0.047 |
| IL-2 | 7.35 ± 0.83 | 6.54 ± 0.85 | 0.0006 |
| CMAP Amplitude | SNAP Amplitude | F-Wave | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Median | Ulnar | Peroneal | Tibial | Median | Ulnar | Sural | Median | Ulnar | Peroneal | Tibial | |||
| Lymphocytes | Monocytes | [%] | −0.17 | ||||||||||
| cells/µL | −0.11 | −0.23 | −0.19 | 0.12 | |||||||||
| Lymphocytes | [%] | 0.32 | −0.18 | −0.28 | −0.14 | −0.12 | |||||||
| cells/µL | −0.13 | −0.17 | −0.19 | −0.14 | |||||||||
| B CD19+ Lymphocytes | [%] | 0.24 | −0.14 | −0.24 | −0.19 | 0.14 | 0.14 | 0.13 | |||||
| cells/µL | 0.26 | −0.14 | −0.21 | −0.12 | |||||||||
| T CD3+ Lymphocytes | [%] | 0.16 | 0.22 | 0.28 | 0.27 | 0.11 | −0.20 | −0.17 | −0.20 | −0.31 | |||
| cells/µL | −0.15 | −0.22 | −0.17 | −0.13 | −0.16 | ||||||||
| TCD3+ CD4+ Lymphocytes | [%] | 0.16 | −0.14 | −0.14 | −0.14 | ||||||||
| cells/µL | 0.11 | −0.11 | −0.16 | −0.19 | −0.11 | −0.17 | |||||||
| TCD3+ CD8+ Lymphocytes | [%] | 0.12 | |||||||||||
| cells/µL | 0.13 | 0.14 | −0.15 | −0.26 | |||||||||
| Cytokines | IFN-y | −0.24 | −0.19 | −0.30 | 0.25 | −0.13 | −0.24 | ||||||
| TNF-α | −0.20 | −0.25 | −0.16 | −0.18 | |||||||||
| IL-10 | −0.13 | −0.32 | −0.28 | 0.13 | −0.12 | ||||||||
| IL-6 | −0.25 | −0.17 | −0.24 | −0.15 | −0.13 | −0.20 | |||||||
| IL-4 | −0.30 | −0.21 | 0.19 | −0.24 | |||||||||
| IL-2 | −0.24 | 0.14 | 0.27 | −0.37 | −0.22 | −0.12 | |||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dziadkowiak, E.; Moreira, H.; Wieczorek, M.; Budrewicz, S.; Barg, E.; Koszewicz, M. Correlations between Electrophysiological Parameters, Lymphocyte Distribution and Cytokine Levels in Patients with Chronic Demyelinating Inflammatory Polyneuropathy. J. Pers. Med. 2021, 11, 766. https://doi.org/10.3390/jpm11080766
Dziadkowiak E, Moreira H, Wieczorek M, Budrewicz S, Barg E, Koszewicz M. Correlations between Electrophysiological Parameters, Lymphocyte Distribution and Cytokine Levels in Patients with Chronic Demyelinating Inflammatory Polyneuropathy. Journal of Personalized Medicine. 2021; 11(8):766. https://doi.org/10.3390/jpm11080766
Chicago/Turabian StyleDziadkowiak, Edyta, Helena Moreira, Malgorzata Wieczorek, Slawomir Budrewicz, Ewa Barg, and Magdalena Koszewicz. 2021. "Correlations between Electrophysiological Parameters, Lymphocyte Distribution and Cytokine Levels in Patients with Chronic Demyelinating Inflammatory Polyneuropathy" Journal of Personalized Medicine 11, no. 8: 766. https://doi.org/10.3390/jpm11080766
APA StyleDziadkowiak, E., Moreira, H., Wieczorek, M., Budrewicz, S., Barg, E., & Koszewicz, M. (2021). Correlations between Electrophysiological Parameters, Lymphocyte Distribution and Cytokine Levels in Patients with Chronic Demyelinating Inflammatory Polyneuropathy. Journal of Personalized Medicine, 11(8), 766. https://doi.org/10.3390/jpm11080766






