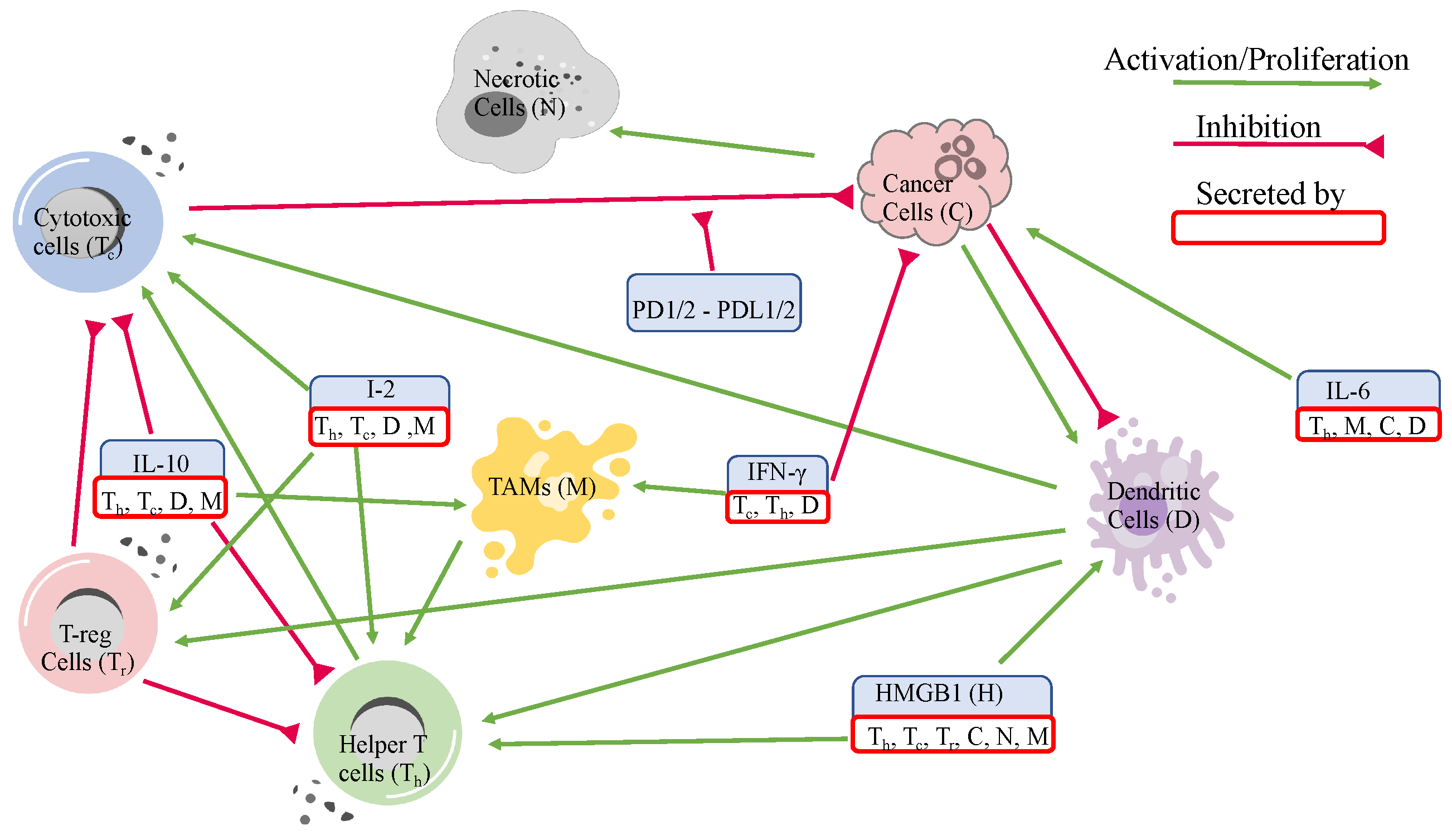
3.1. Dynamics
The dynamics of the cells and molecules over 5000 days are presented in
Figure 3.
The helper T-cell population decreases at the beginning and eventually increases to reach a steady state in clusters 3 and 4. In cluster 2, it increases, and in cluster 1, it decreases to reach a steady state within the first few days. As a result, the helper T-cell populations in clusters 1 and 2 remain constant almost all of the time.
Cytotoxic cells in all clusters decrease in the first few days but eventually increase in population to reach a steady state. Furthermore, cytotoxic cells in cluster 2 start with the highest population and achieve the highest saturation level. This is followed by cluster 4, with the second highest saturation level, where the cells grow faster and reach a steady state in around 1500 days. In cluster 3, cytotoxic cells reach the lowest saturation level, leaving cluster 1 with the second lowest saturation level. Overall, the steady state populations and the time points that each cluster reaches steady states are noticeably different in each cluster.
T-reg cells in clusters 1, 2, and 4 increase quickly from their initial conditions and decrease to reach a steady state. T-reg cells in cluster 4 reach a steady state at around 1500 days and clusters 1 and 2 reach a steady state at around 3000 days. In cluster 3, T-reg cells decrease and reach a steady state faster than in any other cluster. Furthermore, the slow increase in cytotoxic cells in clusters 1 and 2 could be due to regulations by T-reg cells that slowly decrease in these clusters. Similarly, the faster growth in cluster 4 of cytotoxic cells could be related to the fast decrease of T-reg cells in cluster 4.
Naive T-cells in cluster 1 attain the highest steady state population, followed by 3, 2, and 4, respectively. In clusters 1 and 2, naive T-cells increase slowly and reach a steady state of around 2000 days, whereas clusters 3 and 4 stabilize before and after 1000 days.
Macrophages () in clusters 1 and 3 initially increase and then soon start to decrease to reach a steady state, but in clusters 2 and 4, they decrease to reach a steady state. Naive macrophage growth in all clusters does almost the opposite of macrophages, which could be because macrophages are derived from naive macrophages. The overall trend for macrophages is that they decrease to reach a steady state in all clusters, while naive macrophages increase to reach a steady state.
The overall trend for both mature and naive dendritic cells is to decrease to reach a steady state. However, dendritic cells in cluster 3 initially increase quickly and drastically and then suddenly decrease. The same happens to naive dendritic cells in cluster 1. Moreover, cluster 1 stands out in naive dendritic cells by achieving comparatively higher steady state values than the others.
Cancer and necrotic cells exhibit exponential growth until they reach a steady state and have similar curves, as necrotic cells are produced at a rate proportional to cancer cell decay. Cancer cells in cluster 1 attain the highest steady state population. Cluster 3 stands out by growing the fastest and requiring less time to reach a steady state, which could be related to its low level of cytotoxic cells (especially since cytotoxic cells also reach a steady state at around 1000 days in this cluster). Cluster 2 has the slowest growth despite having the highest initial population. Clusters 2, 3, and 4 all achieve a similar steady state population sooner or later within 5000 days. The slow growth in cluster 2 can also be attributed to the significantly high cytotoxic levels. Overall, we can see a clear correlation between cancer progression and cytotoxicity levels.
Interferon- initially increases but starts to decrease to reach a steady state in all clusters. HMGB1 concentration drops at the beginning and then increases to reach a similar steady state in all clusters. Cluster 1, as with cancer and necrotic cells, achieves the highest steady state concentration in HMGB1. It is consistent with our assumptions of parameters, as HMGB1 is mainly secreted by cancer and necrotic cells in the cancer microenvironment. IL-10 concentrations in clusters 1, 3, and 4 increase initially but decrease within a few days. However, cluster 3 concentration of IL-10 increases rapidly to a higher concentration than other clusters and decreases to reach a similar steady state concentration. The cluster 1 concentration of IL-10 reaches a steady state later than other clusters (around 700 days). Cluster 2’s IL-10 concentration decreases to reach the lowest steady state concentration among all clusters. IL-2 and IL-12 concentrations in clusters 1, 3, and 4 also increase at the beginning, then decrease to reach a steady state. As with IL-10, cluster 3 concentration attains its maximum concentration very fast and decreases to reach a steady state concentration. In cluster 1, it follows a similar trend as IL-10 and slowly reaches a steady state at around 2000 days. Finally, the concentration of IL-6 in clusters 1, 3, and 4 increases and then decreases to reach a steady state. However, the concentration of IL-6 in cluster 2 remains constant for the most part.
Since we assume that the concentrations of are proportional to cytotoxic cells and the concentration of is proportional to cancer cells, we refrain from including their dynamics.
3.2. Sensitivity
The sensitivity analysis reveals that the parameters directly involved in the cancer ODE are the most sensitive parameters for cancer cells and total cells. Additionally, macrophage-related parameters, such as
,
, and
, and T-reg cell-related parameters, such as
and
, and an IL-6-related parameter,
, also show significant sensitivity values. The sensitivity plots for the most varying parameters on cancer cells and total cells are shown in
Figure 4 and
Figure 5.
We could say that a quantity is positively sensitive to a parameter when an increase in the parameter causes the quantity to become larger. A quantity is negatively sensitive to a parameter when an increase in the parameter causes a decrease in the quantity. According to the sensitivity plot in
Figure 4, cancer cells in all clusters are negatively sensitive to their decay rates and then positively sensitive to their growth rates. For all clusters, the most sensitive parameters for total cells are the decay/inhibition rates of cancer and macrophages and cancer cell growth/promotion rates.
In addition,
Figure 5 illustrates more sensitive parameters for cancer cells and total cells. For sensitivity to cancer and total cells, we see a significant emphasis on macrophage-related parameters in all clusters, especially clusters 1 and 2. Additionally, regulatory T-cell promotion and decay rates play more significant parts in the cancer sensitivity results for clusters 3 and 4. Finally, we see
as the sixth sensitive parameter for cancer in cluster 1.
The sensitivity analyses of the parameters show that the direct and indirect interactions between the variables impact cancer or total cell growth in the cancer microenvironment. For instance, the inhibition of cancer cells by cytotoxic T-cells, IFN-
, or natural decay, and its promotion by
and natural growth, are direct pathways that increase or decrease the number of cancer cells. On the other hand, we notice that cancer cells are negatively sensitive to the decay rate of macrophages (
Figure 4) and positively sensitive to their promotion rates (
Figure 5). This is because the macrophages secrete IL-6, and then IL-6 helps promote cancer. So, decay in macrophages would lead to less secretion of IL-6, leaving cancer cells with fewer resources and vice versa. Moreover, T-reg cell parameters play an important role in cancer development by negatively impacting cancer with their growth and positively affecting cancer with their decay, especially in clusters 3 and 4. Let us only consider that T-reg cells inhibit helper-T cells and cytotoxic cells. This correlation does not seem reasonable as helper T-cells promote cytotoxic cells and cytotoxic cells kill cancer cells. However, T-reg cells secrete HMGB1, which promotes dendritic cells, and both HMGB1 and dendritic cells promote helper T-cells, which help promote cytotoxic cells that kill cancer cells. Thus, through this pathway, T-reg cells negatively impact cancer cells with their growth and positively impact cancer cells with their decay rate.
3.3. Varying Dynamics of Cancer Cells with Scaled Assumptions
Appendix A.1 shows that most of the parameters (except possibly some decay rates), including the sensitive ones, were derived from restrictive assumptions. To assess the validity of these assumptions, we scaled them using factors of 1, 0.2, and 5. These scalings created a new set of parameters with significantly different values. Then from these sets, we perturbed the most sensitive parameters illustrated in
Figure 4 and
Figure 5 by 5% to create an interval of confidence for the new dynamics. After scaling all the assumptions in
Appendix A.1, only 3 caused significant changes to cancer dynamics. These assumptions are as follows.
These three scalings cause significant changes in the parameters as illustrated in
Figure 6. We can see that assumption (
20) causes a significant deviation from the original values for parameters
,
,
, and
. Moreover, scaling assumptions (
21) and (
22) causes parameters, such as
,
,
, and
to change but not as drastically as the changes imposed by assumption (
20). What is interesting is that none of these parameters (except for
in cluster 1) are among the sensitive parameters. However, the changes caused by the scaling of the assumptions were so large that the effects were tangible. We emphasize that none of the other assumptions left such an impact after scaling.
Figure 7 shows the cancer dynamics with the original parameter values next to the dynamics acquired by scaling the assumptions (
20)–(
22) by 0.2 and 5, respectively. The shaded regions are the regions of confidence acquired from perturbing the most sensitive parameters in
Figure 4 and
Figure 5 by 5%. We see slight changes in clusters 2, 3, and 4, and a more significant change in cluster 1. Moreover, the width of the shaded regions remained the same in all cases. We saw that by changing the assumptions (
20)–(
22), significant deviations occurred to the parameter values mainly involved in the dendritic cells and
production. Cluster 1, which was more impacted, was the only cluster sensitive to the parameter
. Even though the assumptions are mostly modeling artifacts for parameter estimations (and one must be cautious when using them), these results suggest interesting control potentials for
.
Although the parameters
and
were not among the most sensitive parameters, to make sure that the assumptions on these parameters were reasonable, we scaled
with
and 5 with
variations to the most sensitive parameters to see their impacts on cancer cells (
Figure 8). Since
is multiplied by
in the ODE system, we did not scale it as our goal was to keep the scaling of the term that involved these parameters similar to
. Thus, the scaled equation for
becomes,
As a result, the term involving
in the cancer cell equation becomes,
The figure indicates that the dynamic of the cancer cell population remains unchanged after varying the term . Moreover, the perturbation of the sensitive parameters causes similar variations in all three cases.