A Gel-Based Proteomic Analysis Reveals Synovial α-Enolase and Fibrinogen β-Chain Dysregulation in Knee Osteoarthritis: A Controlled Trial
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients Selection
2.2. Synovial Fluid Sample Collection and Processing
2.3. Two-Dimensional Gel Electrophoresis (2DE)
2.4. Mass Spectrometry Analysis and Bioinformatic Analysis
2.5. Statistical Analysis
3. Results and Discussion
3.1. Results
| OA-Group | Control-Group | p | |
|---|---|---|---|
| Patients (n) | 24 | 19 | |
| Age | |||
| Mean ± SD | 68.19 ± 6.18 | 24.65 ± 4.73 | 0.001 * |
| Range | 54–80 | 19–34 | - |
| Gender | |||
| Male, n (%) | 11 (45.83%) | 9 (47.37%) | 0.221 |
| Female, n (%) | 13 (54.17%) | 10 (52.63%) | 0.124 |
| BMI (kg/m2) | |||
| Mean ± SD | 27.75 ± 4.88 | 22.16 ± 1.7 | 0.002 * |
| Smoking status | |||
| Number of smokers, n (%) | 6 (20.69%) | 2 (12.5%) | 0.03 * |
| Kellgren Lawrence classification | |||
| Grade 3 | 11 (37–93%) | // | // |
| Grade 4 | 18 (62.07%) | // | // |
| CRP (mg/dL) | |||
| Mean ± SD | 1.73 ± 1.25 | 0.21 ± 0.43 | 0.001 * |
| Serum Fibrinogen (mg/dL) | |||
| Mean ± SD | 302.4 ± 78.5 | 245.6 ± 57.3 | 0.003 * |
| KSS | |||
| Mean ± SD | 42.09 ± 7.24 | 94.45 ± 4.35 | <0.001 * |
| IKDC | |||
| Mean ± SD | 0.346 ± 0.0825 | 0.964 ± 0.021 | <0.001 * |
| KOOS main outcome | |||
| Mean ± SD | 38.45 ± 7.6 | 95.7 ± 3.5 | <0.001 * |
| VAS | |||
| Mean ± SD | 5.78 ± 1.45 | 4.53 ± 1.65 | 0.07 |
3.2. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
References
- Mobasheri, A.; Bay-Jensen, A.-C.; van Spil, W.E.; Larkin, J.; Levesque, M.C. Osteoarthritis Year in Review 2016: Biomarkers (Biochemical Markers). Osteoarthr. Cartil. 2017, 25, 199–208. [Google Scholar] [CrossRef]
- Hunter, D.J.; Nevitt, M.; Losina, E.; Kraus, V. Biomarkers for Osteoarthritis: Current Position and Steps towards Further Validation. Best Pract. Res. Clin. Rheumatol. 2014, 28, 61–71. [Google Scholar] [CrossRef]
- Goldring, M.B.; Goldring, S.R. Osteoarthritis. J. Cell. Physiol. 2007, 213, 626–634. [Google Scholar] [CrossRef] [PubMed]
- Blanco, F.J.; Ruiz-Romero, C. Metabolomic Characterization of Metabolic Phenotypes in OA. Nat. Rev. Rheumatol. 2012, 8, 130–132. [Google Scholar] [CrossRef] [PubMed]
- Cross, M.; Smith, E.; Hoy, D.; Nolte, S.; Ackerman, I.; Fransen, M.; Bridgett, L.; Williams, S.; Guillemin, F.; Hill, C.L.; et al. The global burden of hip and knee osteoarthritis: Estimates from the global burden of disease 2010 study. Ann. Rheum. Dis. 2014, 73, 1323–1330. [Google Scholar] [CrossRef]
- Bizzoca, D.; Moretti, L.; Gnoni, A.; Moretti, F.L.; Scacco, S.; Banfi, G.; Piazzolla, A.; Solarino, G.; Moretti, B. The Usefulness of Synovial Fluid Proteome Analysis in Orthopaedics: Focus on Osteoarthritis and Periprosthetic Joint Infections. J. Funct. Morphol. Kinesiol. 2022, 7, 97. [Google Scholar] [CrossRef]
- Moretti, L.; Bizzoca, D.; Geronimo, A.; Moretti, F.L.; Monaco, E.; Solarino, G.; Moretti, B. Towards Precision Medicine for Osteoarthritis: Focus on the Synovial Fluid Proteome. Int. J. Mol. Sci. 2022, 23, 9731. [Google Scholar] [CrossRef]
- Balakrishnan, L.; Nirujogi, R.; Ahmad, S.; Bhattacharjee, M.; Manda, S.S.; Renuse, S.; Kelkar, D.S.; Subbannayya, Y.; Raju, R.; Goel, R.; et al. Proteomic Analysis of Human Osteoarthritis Synovial Fluid. Clin. Proteom. 2014, 11, 6. [Google Scholar] [CrossRef] [PubMed]
- Hepinstall, M.S.; Rutledge, J.R.; Bornstein, L.J.; Mazumdar, M.; Westrich, G.H. Factors that impact expectations before total knee arthroplasty. J. Arthroplasty. 2011, 26, 870–876. [Google Scholar] [CrossRef]
- Corigliano, A.; Preianò, M.; Terracciano, R.; Savino, R.; De Gori, M.; Galasso, O.; Gasparini, G. C3f Is a Potential Tool for the Staging of Osteoarthritis. J. Biol. Regul. Homeost. Agents 2017, 31, 29–35. [Google Scholar]
- Mahendran, S.M.; Oikonomopoulou, K.; Diamandis, E.P.; Chandran, V. Synovial Fluid Proteomics in the Pursuit of Arthritis Mediators: An Evolving Field of Novel Biomarker Discovery. Crit. Rev. Clin. Lab. Sci. 2017, 54, 495–505. [Google Scholar] [CrossRef] [PubMed]
- Wanner, J.P.; Subbaiah, R.; Skomorovska-Prokvolit, Y.; Shishani, Y.; Boilard, E.; Mohan, S.; Gillespie, R.; Miyagi, M.; Gobezie, R. Proteomic Profiling and Functional Characterization of Early and Late Shoulder Osteoarthritis. Arthritis Res. Ther. 2013, 15, R180. [Google Scholar] [CrossRef] [PubMed]
- Tsuchida, A.I.; Beekhuizen, M.; Rutgers, M.; van Osch, G.J.; Bekkers, J.E.; Bot, A.G.; Geurts, B.; Dhert, W.J.; Saris, D.B.; Creemers, L.B. Interleukin-6 Is Elevated in Synovial Fluid of Patients with Focal Cartilage Defects and Stimulates Cartilage Matrix Production in an In Vitro Regeneration Model. Arthritis Res. Ther. 2012, 14, R262. [Google Scholar] [CrossRef] [PubMed]
- Kokebie, R.; Aggarwal, R.; Lidder, S.; Hakimiyan, A.A.; Rueger, D.C.; Block, J.A.; Chubinskaya, S. The Role of Synovial Fluid Markers of Catabolism and Anabolism in Osteoarthritis, Rheumatoid Arthritis and Asymptomatic Organ Donors. Arthritis Res. Ther. 2011, 13, R50. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, S.; Satoh, T.; Chiba, J.; Ju, C.; Inoue, K.; Kagawa, J. Interleukin-6 and Interleukin-8 Levels in Serum and Synovial Fluid of Patients with Osteoarthritis. Cytokines. Cell. Mol. Ther. 2000, 6, 71–79. [Google Scholar] [CrossRef]
- Liao, W.; Li, Z.; Li, T.; Zhang, Q.; Zhang, H.; Wang, X. Proteomic Analysis of Synovial Fluid in Osteoarthritis Using SWATH-mass Spectrometry. Mol. Med. Rep. 2018, 17, 2827–2836. [Google Scholar] [CrossRef]
- Insall, J.N.; Dorr, L.D.; Scott, R.D.; Scott, W.N. Rationale of the Knee Society clinical rating system. Clin. Orthop. Relat. Res. 1989, 248, 13–14. [Google Scholar] [CrossRef]
- Higgins, L.D.; Taylor, M.K.; Park, D.; Ghodadra, N.; Marchant, M.; Pietrobon, R.; Cook, C. Reliability and validity of the International Knee Documentation Committee (IKDC) Subjective Knee Form. Jt. Bone Spine 2007, 74, 594–599. [Google Scholar] [CrossRef]
- Lyman, S.; Lee, Y.Y.; McLawhorn, A.S.; Islam, W.; MacLean, C.H. What Are the Minimal and Substantial Improvements in the HOOS and KOOS and JR Versions after Total Joint Replacement? Clin. Orthop. Relat. Res. 2018, 476, 2432–2441. [Google Scholar] [CrossRef]
- Bizzoca, D.; Rocchetti, M.T.; Scacco, S.; Taurino, F.; Vicenti, G.; Spadaccino, F.; Moretti, L.; Ranieri, E.; Gesualdo, L.; Moretti, F.; et al. Beyond pre-analytical and analytical concerns in the study of synovial fluid proteome: Description of an optimized Gel-based protocol. J. Biol. Regul. Homeost. Agents 2021, 35, 827–832. [Google Scholar] [CrossRef]
- Rocchetti, M.T.; Papale, M.; d’Apollo, A.M.; Suriano, I.V.; Di Palma, A.M.; Vocino, G.; Montemurno, E.; Varraso, L.; Grandaliano, G.; Di Paolo, S.; et al. Association of urinary laminin G-like 3 and free K light chains with disease activity and histological injury in IgA nephropathy. Clin. J. Am. Soc. Nephrol. 2013, 8, 1115–1125. [Google Scholar] [CrossRef] [PubMed]
- He, P.; Zhang, Z.; Liao, W.; Xu, D.; Fu, M.; Kang, Y. Screening of gene signatures for rheumatoid arthritis and osteoarthritis based on bioinformatics analysis. Mol. Med. Rep. 2016, 14, 1587–1593. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Dong, Z.; Cheng, L.; Guo, Z.; Lv, Z. Identifying the gender-specific genes and pathways in osteoarthritis by bioinformatics. Osteoarthr. Cartil. 2020, 28, S207–S208. [Google Scholar] [CrossRef]
- Shorter, E.; Avelar, R.; Zachariou, M.; Spyrou, G.M.; Raina, P.; Smagul, A.; Ashraf Kharaz, Y.; Peffers, M.; Goljanek-Whysall, K.; de Magalhães, J.P.; et al. Identifying Novel Osteoarthritis-Associated Genes in Human Cartilage Using a Systematic Meta-Analysis and a Multi-Source Integrated Network. Int. J. Mol. Sci. 2022, 23, 4395. [Google Scholar] [CrossRef] [PubMed]
- Gedeon, A.K.; Colley, A.; Jamieson, R.; Thompson, E.M.; Rogers, J.; Sillence, D.; Tiller, G.E.; Mulley, J.C.; Gécz, J. Identification of the gene (SEDL) causing X-linked spondyloepiphyseal dysplasia tarda. Nat. Genet. 1999, 22, 400–404. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, H.; Ishii, H.; Takahashi, K.; Takai, S.; Ozawa, H. Suitable reference gene selection for gene expression studies in knee osteoarthritis synovium using quantitative PCR analysis. Connect. Tissue Res. 2018, 59, 356–368. [Google Scholar] [CrossRef]
- Pereira, D.; Ramos, E.; Branco, J. Osteoarthritis. Acta Med. Port. 2015, 28, 99–106. [Google Scholar] [CrossRef]
- Marshall, D.A.; Vanderby, S.; Barnabe, C.; MacDonald, K.V.; Maxwell, C.; Mosher, D.; Wasylak, T.; Lix, L.; Enns, E.; Frank, C.; et al. Estimating the Burden of Osteoarthritis to Plan for the Future. Arthritis Care Res. 2015, 67, 1379–1386. [Google Scholar] [CrossRef]
- Bae, S.; Kim, H.; Lee, N.; Won, C.; Kim, H.R.; Hwang, Y.I.; Song, Y.W.; Kang, J.S.; Lee, W.J. α-Enolase expressed on the surfaces of monocytes and macrophages induces robust synovial inflammation in rheumatoid arthritis. J. Immunol. 2012, 189, 365–372. [Google Scholar] [CrossRef]
- Capello, M.; Ferri-Borgogno, S.; Cappello, P.; Novelli, F. α-Enolase: A promising therapeutic and diagnostic tumour target. FEBS J. 2011, 278, 1064–1074. [Google Scholar] [CrossRef]
- Pancholi, V. Multifunctional alpha-enolase: Its role in diseases. Cell. Mol. Life Sci. 2001, 58, 902–920. [Google Scholar] [CrossRef] [PubMed]
- Oughtred, R.; Rust, J.; Chang, C.; Breitkreutz, B.J.; Stark, C.; Willems, A.; Boucher, L.; Leung, G.; Kolas, N.; Zhang, F.; et al. The BioGRID database: A comprehensive biomedical resource of curated protein, genetic, and chemical interactions. Protein Sci. 2021, 30, 187–200. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Luo, L.; Gui, T.; Yu, F.; Yan, L.; Yao, L.; Zhong, L.; Yu, W.; Han, B.; Patel, J.M.; et al. Targeting cartilage EGFR pathway for osteoarthritis treatment. Sci. Transl. Med. 2021, 13, eabb3946. [Google Scholar] [CrossRef] [PubMed]
- Gui, T.; Wei, Y.; Luo, L.; Li, J.; Zhong, L.; Yao, L.; Beier, F.; Nelson, C.L.; Tsourkas, A.; Liu, X.S.; et al. Activating EGFR Signaling Attenuates Osteoarthritis Development Following Loading Injury in Mice. J. Bone Min. Res. 2022, 37, 2498–2511. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Ma, X.; Sun, H.; Gui, T.; Li, J.; Yao, L.; Zhong, L.; Yu, W.; Han, B.; Nelson, C.L.; et al. EGFR Signaling Is Required for Maintaining Adult Cartilage Homeostasis and Attenuating Osteoarthritis Progression. J. Bone Min. Res. 2022, 37, 1012–1023. [Google Scholar] [CrossRef]
- Lu, Q.; Liu, P.; Miao, Z.; Luo, D.; Li, S.; Lu, M. SIRT1 restoration enhances chondrocyte autophagy in osteoarthritis through PTEN-mediated EGFR ubiquitination. Cell. Death Discov. 2022, 8, 203. [Google Scholar] [CrossRef]
- Jiang, L.; Zhou, X.; Xu, K.; Hu, P.; Bao, J.; Li, J.; Zhu, J.; Wu, L. miR-7/EGFR/MEGF9 axis regulates cartilage degradation in osteoarthritis via PI3K/AKT/mTOR signalling pathway. Bioengineered 2021, 12, 8622–8634. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Zhang, A.; Wang, J.; Han, K.; Gao, H. Estrogen alleviates post-traumatic osteoarthritis progression and decreases p-EGFR levels in female mouse cartilage. BMC Musculoskelet. Disord. 2022, 23, 685. [Google Scholar] [CrossRef]
- Qin, L.; Beier, F. EGFR Signaling: Friend or Foe for Cartilage? JBMR Plus. 2019, 3, e10177. [Google Scholar] [CrossRef]
- Damgaard, D.; Bawadekar, M.; Senolt, L.; Stensballe, A.; Shelef, M.A.; Nielsen, C.H. Relative efficiencies of peptidyl arginine deiminase 2 and 4 in generating target sites for anti-citrullinated protein antibodies in fibrinogen, alpha-enolase and histone H3. PLoS ONE 2018, 13, e0203214. [Google Scholar] [CrossRef]
- Beydoun, H.A.; Archer, D.F.; Zonderman, A.B.; Beydoun, M.A. Interrelationships of Sex, Physician-Diagnosed Arthritis, Chronic Inflammation, and Physical Functioning in the Third National Health and Nutrition Examination Surveys. Gerontol. Geriatr. Med. 2015, 1. [Google Scholar] [CrossRef]
- Antony, B.; Venn, A.; Cicuttini, F.; March, L.; Blizzard, L.; Dwyer, T.; Cross, M.; Jones, G.; Ding, C. Association of Body Composition and Hormonal and Inflammatory Factors with Tibial Cartilage Volume and Sex Difference in Cartilage Volume in Young Adults. Arthritis Care Res. 2016, 68, 517–525. [Google Scholar] [CrossRef]
- Skriner, K.; Adolph, K.; Jungblut, P.R.; Burmester, G.R. Association of citrullinated proteins with synovial exosomes. Arthritis Rheum. 2006, 54, 3809–3814. [Google Scholar] [CrossRef]
- Ebrahimi-Rad, M.; Khatami, S.; Akhbari, H.; Mahmoudzadeh-Niknam, H.; Valadbeigi, S.; Mahmoudi, M.; Jamshidi, A.; Riazi-Rad, F.; Saghiri, R. Evaluation of autoantibodies against vimentin and α-enolase in rheumatoid arthritis patients. Rheumatology 2020, 58, 350–356. [Google Scholar] [CrossRef]
- Schadler, P.; Lohberger, B.; Thauerer, B.; Faschingbauer, M.; Kullich, W.; Stradner, M.H.; Husic, R.; Leithner, A.; Steinecker-Frohnwieser, B. Fatty Acid-Binding Protein 4 (FABP4) Is Associated with Cartilage Thickness in End-Stage Knee Osteoarthritis. Cartilage 2021, 13 (Suppl. S2), 1165S–1173S. [Google Scholar] [CrossRef]
- Belcaro, G.; Cesarone, M.R.; Errichi, S.; Zulli, C.; Errichi, B.M.; Vinciguerra, G.; Ledda, A.; Di Renzo, A.; Stuard, S.; Dugall, M.; et al. Variations in C-reactive protein, plasma free radicals and fibrinogen values in patients with osteoarthritis treated with Pycnogenol. Redox Rep. 2008, 13, 271–276. [Google Scholar] [CrossRef]
- Masson-Bessieère, C.; Sebbag, M.; Girbal-Neuhauser, E.; Nogueira, L.; Vincent, C.; Senshu, T.; Serre, G. The Major Synovial Targets of the Rheumatoid Arthritis-Specific Antifilaggrin Autoantibodies Are Deiminated Forms of the α- and β-Chains of Fibrin1. J. Immunol. 2001, 166, 4177–4184. [Google Scholar] [CrossRef]
- Kandel, L.; Agar, G.; Elkayam, O.; Sharipov, A.; Slevin, O.; Rivkin, G.; Dahan, M.; Aloush, V.; Pyeser, A.B.; Brin, Y.; et al. A novel approach for knee osteoarthritis using high molecular weight hyaluronic acid conjugated to plasma fibrinogen—Interim findings of a double-blind clinical study. Heliyon 2020, 6, e04475. [Google Scholar] [CrossRef]
- Vicenti, G.; Bizzoca, D.; Nappi, V.S.; Moretti, F.; Carrozzo, M.; Belviso, V.; Moretti, B. Biophysical stimulation of the knee with PEMFs: From bench to bedside. J. Biol. Regul. Homeost. Agents 2018, 32 (Suppl. S1), 23–28. [Google Scholar]
- Moretti, L.; Bizzoca, D.; Giancaspro, G.A.; Cassano, G.D.; Moretti, F.; Setti, S.; Moretti, B. Biophysical Stimulation in Athletes’ Joint Degeneration: A Narrative Review. Medicina 2021, 57, 1206. [Google Scholar] [CrossRef]
- Scanzello, C.R.; McKeon, B.; Swaim, B.H.; DiCarlo, E.; Asomugha, E.U.; Kanda, V.; Nair, A.; Lee, D.M.; Richmond, J.C.; Katz, J.N.; et al. Synovial inflammation in patients undergoing arthroscopic meniscectomy: Molecular characterization and relationship to symptoms. Arthritis Rheum. 2011, 63, 391–400. [Google Scholar] [CrossRef] [PubMed]
- Ding, C.; Martel-Pelletier, J.; Pelletier, J.P.; Abram, F.; Raynauld, J.P.; Cicuttini, F.; Jones, G. Tear as an osteoarthritis risk factor in a largely non-osteoarthritic cohort: A cross-sectional study. J. Rheumatol. 2007, 34, 776–784. [Google Scholar] [PubMed]
- Vicenti, G.; Bizzoca, D.; Solarino, G.; Moretti, F.; Ottaviani, G.; Simone, F.; Zavattini, G.; Maccagnano, G.; Noia, G.; Moretti, B. The role of biophysical stimulation with pemfs in fracture healing: From bench to bedside. J. Biol. Regul. Homeost. Agents 2020, 34 (Suppl. S1), 131–135. [Google Scholar] [PubMed]
- Vicenti, G.; Bizzoca, D.; Cotugno, D.; Carrozzo, M.; Riefoli, F.; Rifino, F.; Belviso, V.; Elia, R.; Solarino, G.; Moretti, B. The use of a gentamicin-coated titanium nail, combined with RIA system, in the management of non-unions of open tibial fractures: A single centre prospective study. Injury 2020, 51 (Suppl. S3), S86–S91. [Google Scholar] [CrossRef] [PubMed]
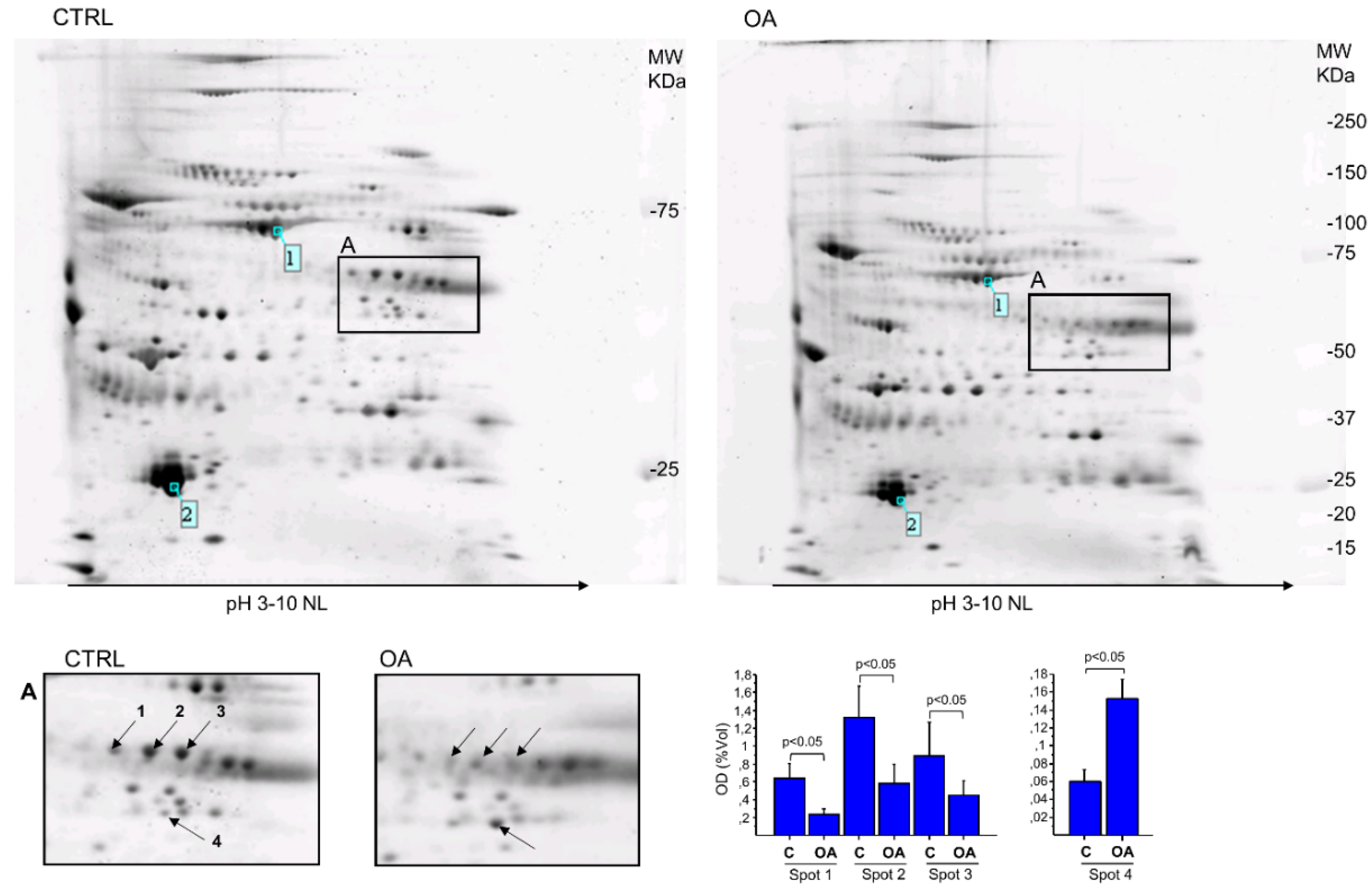


| Spot N° | Protein Name | Gene Name | Accession Number * | Theoretical MW (kDa)/pI | Experimental MW (kDa)/pI | Mascot Score | SC % | Unique Peptide | Fold Change (%) |
|---|---|---|---|---|---|---|---|---|---|
| OA/CTRL | |||||||||
| 1 | Fibrinogen beta chain | FGB | P02675 | 56.7/8.54 | ~57/8.8 | 106 | 25 | 13 | 0.37 (−63%) |
| 2 | Fibrinogen beta chain | FGB | P02675 | 56.7/8.54 | ~57/9.0 | 105 | 34 | 15 | 0.30 (−70%) |
| 3 | Fibrinogen beta chain | FGB | P02675 | 56.7/8.54 | ~57/9.2 | 67 | 25 | 12 | 0.30 (−70%) |
| 4 | Alpha-enolase | ENO1 | P06733 | 47.4/7.01 | ~48/8.8 | 74 | 31 | 11 | 2.5 (+153%) |
| FGB | ENO1 | |||
|---|---|---|---|---|
| R | p | R | p | |
| Clinical scales | ||||
| KSS | −0.61 | 0.02 * | 0.33 | 0.108 |
| IKDC | −0.32 | 0.12 | 0.43 | 0.09 |
| KOOS main outcome | −0.44 | 0.08 | 0.68 | 0.01 * |
| VAS | −0.76 | 0.001 * | 0.72 | 0.005 * |
| Serum biomarkers | ||||
| CRP | −0.23 | 0.45 | 0.27 | 0.32 |
| Fibrinogen | −0.77 | 0.001 * | 0.756 | 0.001 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rocchetti, M.T.; Bizzoca, D.; Moretti, L.; Ragni, E.; Moretti, F.L.; Vicenti, G.; Solarino, G.; Rizzello, A.; Petruzzella, V.; Palese, L.L.; et al. A Gel-Based Proteomic Analysis Reveals Synovial α-Enolase and Fibrinogen β-Chain Dysregulation in Knee Osteoarthritis: A Controlled Trial. J. Pers. Med. 2023, 13, 916. https://doi.org/10.3390/jpm13060916
Rocchetti MT, Bizzoca D, Moretti L, Ragni E, Moretti FL, Vicenti G, Solarino G, Rizzello A, Petruzzella V, Palese LL, et al. A Gel-Based Proteomic Analysis Reveals Synovial α-Enolase and Fibrinogen β-Chain Dysregulation in Knee Osteoarthritis: A Controlled Trial. Journal of Personalized Medicine. 2023; 13(6):916. https://doi.org/10.3390/jpm13060916
Chicago/Turabian StyleRocchetti, Maria Teresa, Davide Bizzoca, Lorenzo Moretti, Enrico Ragni, Francesco Luca Moretti, Giovanni Vicenti, Giuseppe Solarino, Alessandro Rizzello, Vittoria Petruzzella, Luigi Leonardo Palese, and et al. 2023. "A Gel-Based Proteomic Analysis Reveals Synovial α-Enolase and Fibrinogen β-Chain Dysregulation in Knee Osteoarthritis: A Controlled Trial" Journal of Personalized Medicine 13, no. 6: 916. https://doi.org/10.3390/jpm13060916








