Propensity Matched Outcomes of Minimally Invasive Mitral Surgery: Does a Heart-Team Approach Eliminate Female Gender as an Independent Risk Factor?
Abstract
1. Introduction
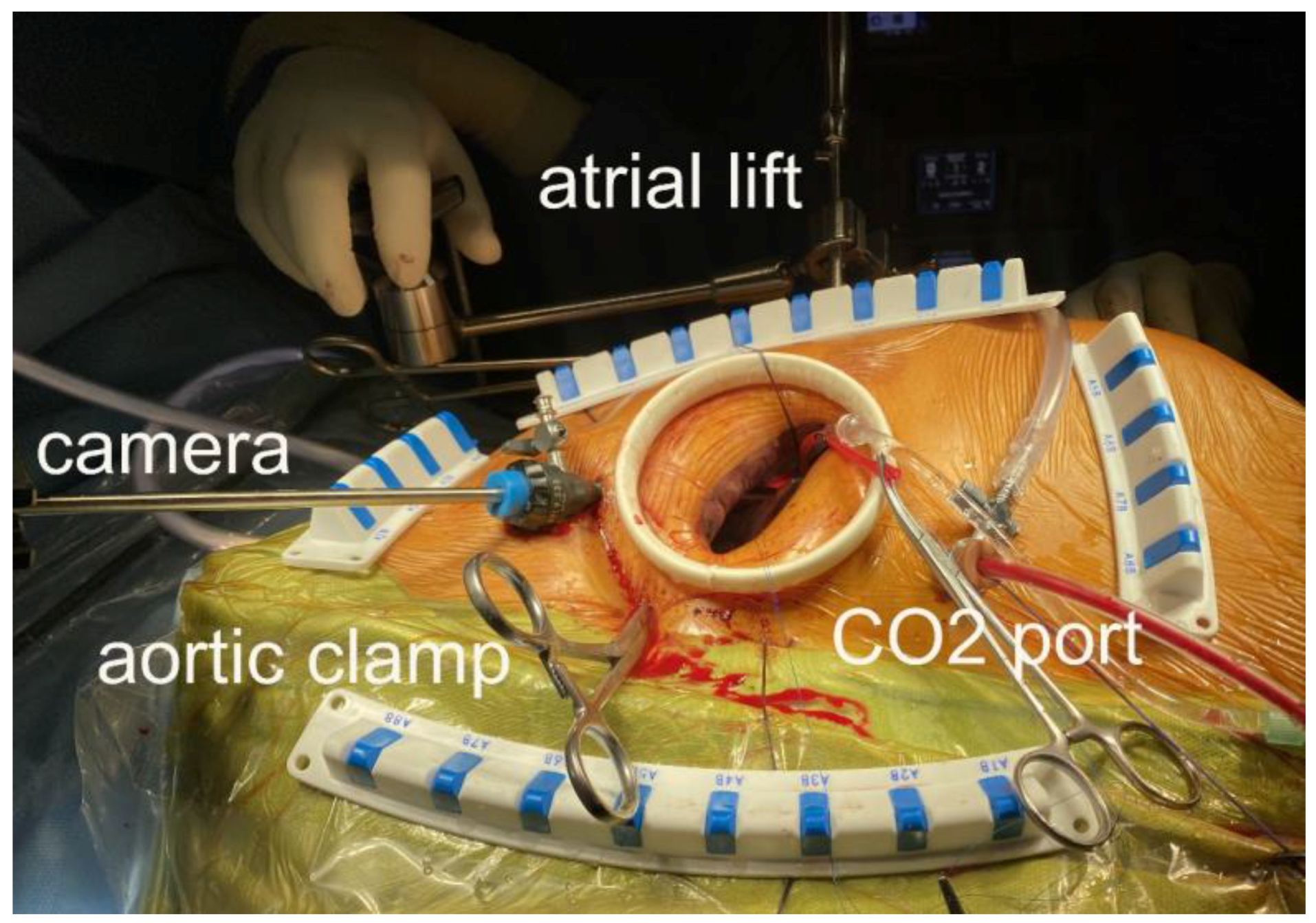
2. Materials and Methods
3. Results
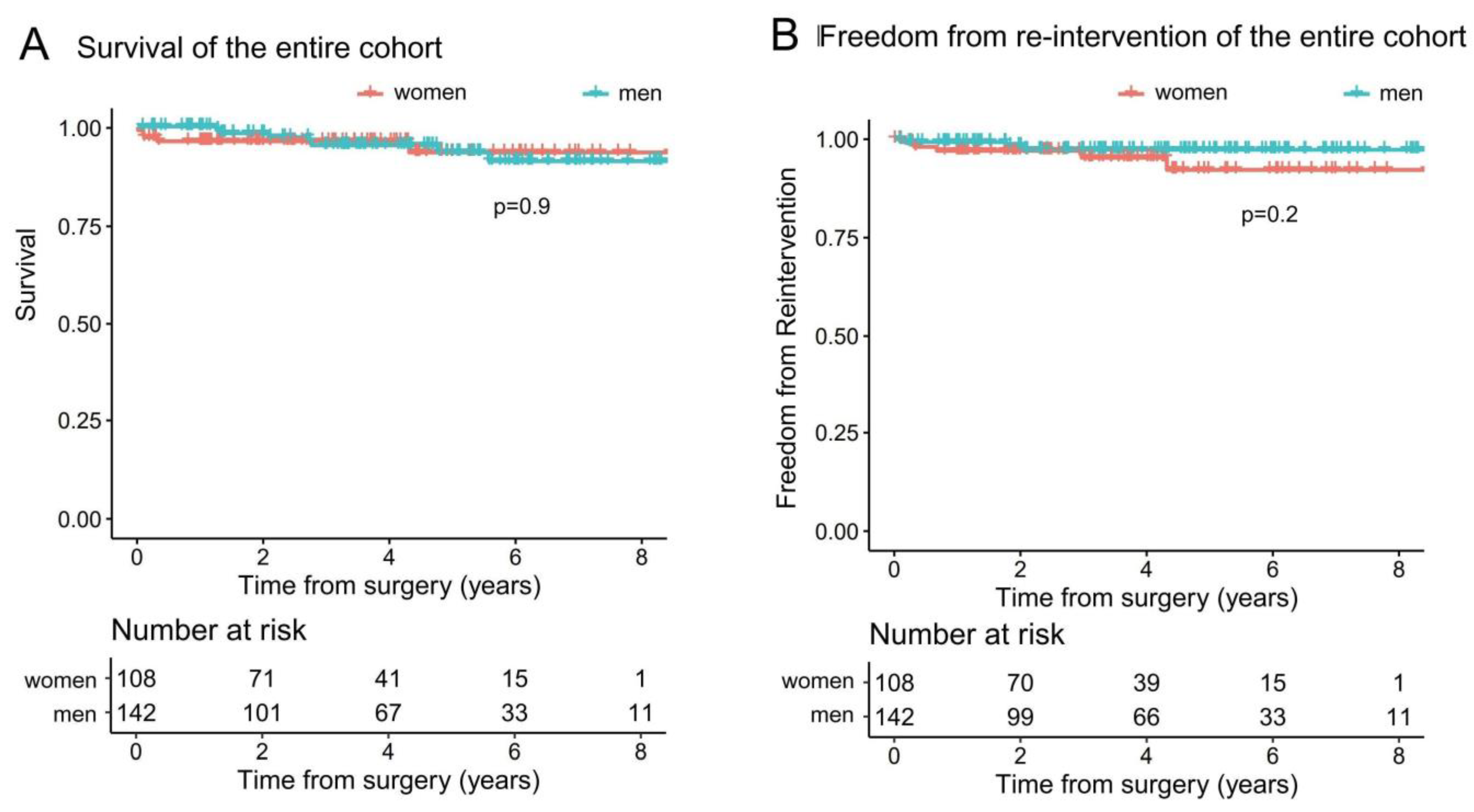
3.1. Total Unmatched Cohort
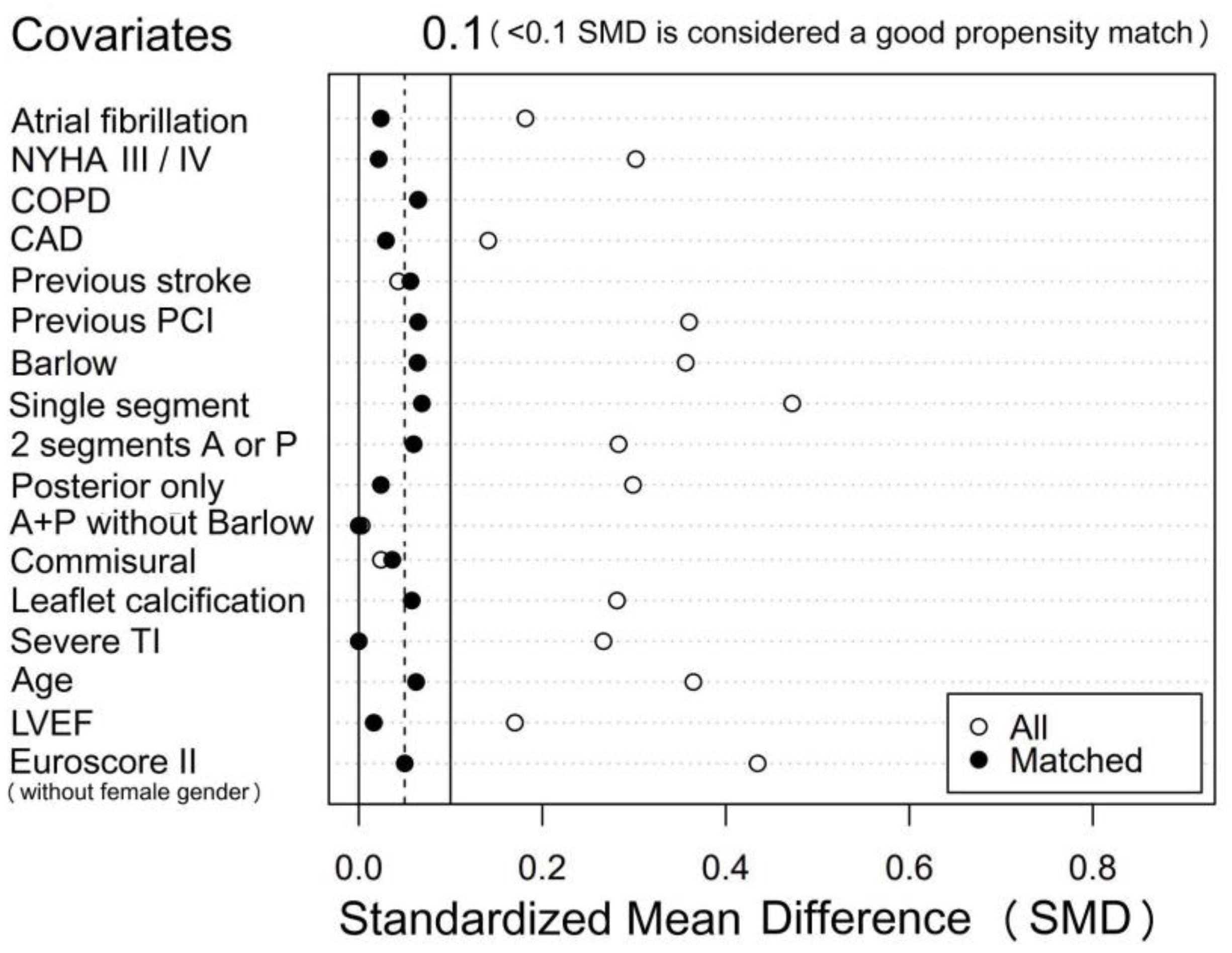
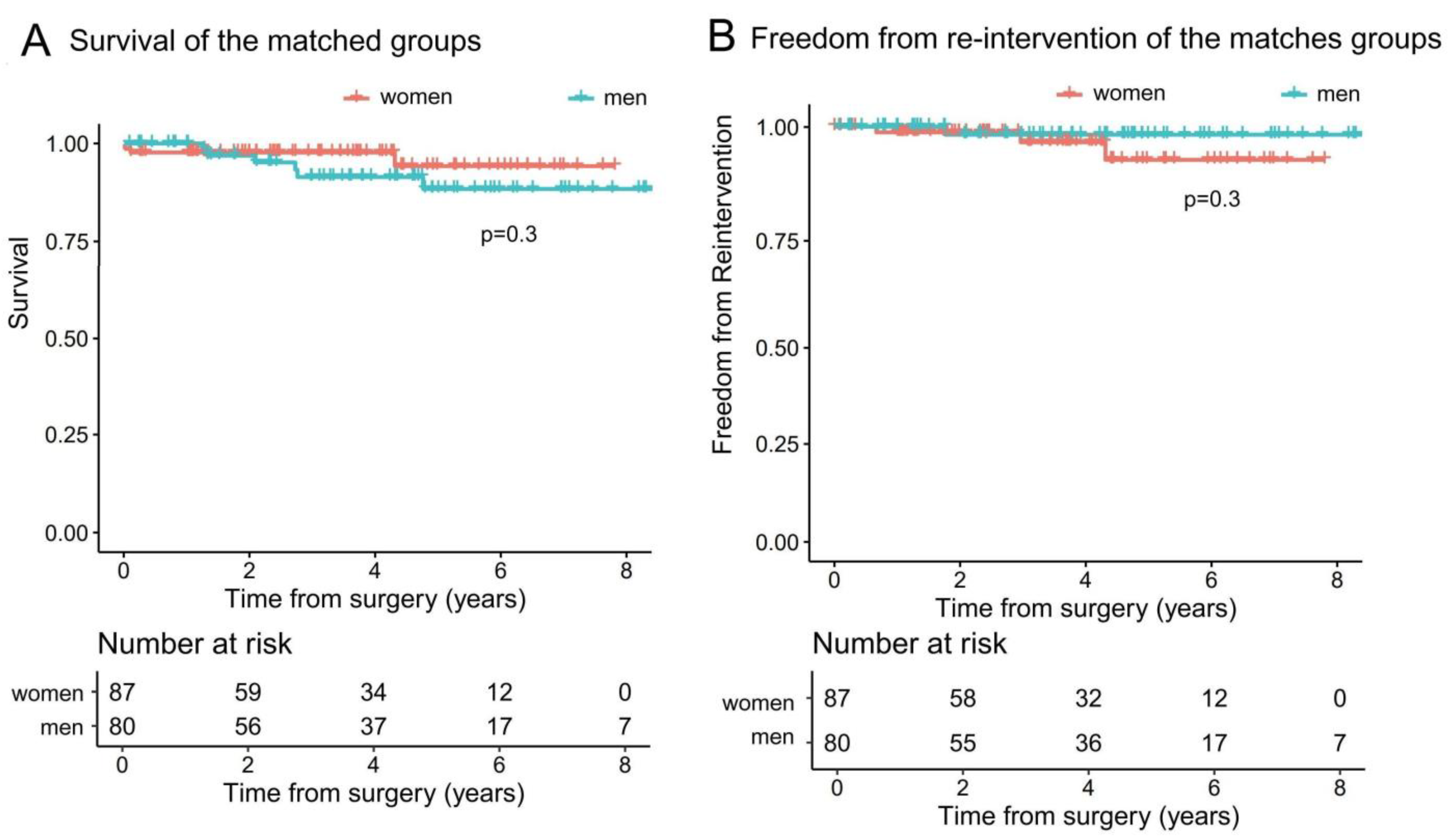
3.2. Propensity Score Matched Groups
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rankin, J.S.; Hammill, B.G.; Ferguson, T.B., Jr.; Glower, D.D.; O’Brien, S.M.; DeLong, E.R.; Peterson, E.D.; Edwards, F.H. Determinants of operative mortality in valvular heart surgery. J. Thorac. Cardiovasc. Surg. 2006, 131, 547–557. [Google Scholar] [CrossRef] [PubMed]
- Dodson, J.A.; Wang, Y.; Desai, M.M.; Barreto-Filho, J.A.; Sugeng, L.; Hashim, S.W.; Krumholz, H.M. Outcomes for mitral valve surgery among Medicare fee-for-service beneficiaries, 1999 to 2008. Circ. Cardiovasc. Qual. Outcomes 2012, 5, 298–307. [Google Scholar] [CrossRef]
- Johnston, A.; Mesana, T.G.; Lee, D.S.; Eddeen, A.B.; Sun, L.Y. Sex differences in long-term survival after major cardiac surgery: A population-based cohort study. J. Am. Heart Assoc. 2019, 8, e013260. [Google Scholar] [CrossRef]
- Song, H.K.; Grab, J.D.; O'Brien, S.M.; Welke, K.F.; Edwards, F.; Ungerleider, R.M. Gender differences in mortality after mitral valve operation: Evidence for higher mortality in perimenopausal women. Ann. Thorac. Surg. 2008, 85, 2040–2044. [Google Scholar] [CrossRef] [PubMed]
- Kislitsina, O.N.; Thomas, J.D.; Crawford, E.; Michel, E.; Kruse, J.; Liu, M.; Andrei, A.-C.; Cox, J.L.; McCarthy, P.M. Predictors of left ventricular dysfunction after surgery for degenerative mitral regurgitation. Ann. Thorac. Surg. 2020, 109, 669–677. [Google Scholar] [CrossRef] [PubMed]
- Nkomo, V.T.; Gardin, J.M.; Skelton, T.N.; Gottdiener, J.S.; Scott, C.G.; Enriquez-Sarano, M. Burden of valvular heart diseases: A population-based study. Lancet 2006, 368, 1005–1011. [Google Scholar] [CrossRef]
- Avierinos, J.F.; Gersh, B.J.; Melton, L.J., III; Bailey, K.R.; Shub, C.; Nishimura, R.A.; Tajik, A.J.; Enriquez-Sarano, M. Natural History of Mitral Valve Prolapse in the Community. Circulation 2002, 106, 1355–1361. [Google Scholar] [CrossRef]
- Vassileva, C.M.; McNeely, C.; Mishkel, G.; Boley, T.; Markwell, S.; Hazelrigg, S. Gender differences in long-term survival of Medicare beneficiaries undergoing mitral valve operations. Ann. Thorac. Surg. 2013, 96, 1367–1373. [Google Scholar] [CrossRef]
- O'Brien, S.M.; Shahian, D.M.; Filardo, G.; Ferraris, V.A.; Haan, C.K.; Rich, J.B.; Normand, S.-L.T.; DeLong, E.R.; Shewan, C.M.; Dokholyan, R.S.; et al. Society of Thoracic Surgeons Quality Measurement Task Force. The Society of Thoracic Surgeons 2008 cardiac surgery risk models: Part 2–isolated valve surgery. Ann. Thorac. Surg. 2009, 88, S23–S42. [Google Scholar] [CrossRef]
- Nashef, S.A.; Roques, F.; Sharples, L.D.; Nilsson, J.; Smith, C.; Goldstone, A.R.; Lockowandt, U. EuroSCORE II. Eur. J. Cardiothorac. Surg. 2012, 41, 734–744; discussion 44–45. [Google Scholar] [CrossRef]
- Cheng, D.C.; Martin, J.; Lal, A.; Diegeler, A.; Folliguet, T.A.; Nifong, L.W.; Perier, P.; Raanani, E.; Smith, J.M.; Seeburger, J.; et al. Minimally invasive versus conventional open mitral valve surgery: A meta-analysis and systematic review. Innovations 2011, 6, 84–103. [Google Scholar] [CrossRef]
- Davierwala, P.M.; Seeburger, J.; Pfannmueller, B.; Garbade, J.; Misfeld, M.; Borger, M.A.; Mohr, F.W. Minimally invasive mitral valve surgery: The Leipzig experience. Ann. Cardiothorac. Surg. 2013, 6, 744–750. [Google Scholar]
- Goldstone, A.B.; Atluri, P.; Szeto, W.Y.; Trubelja, A.; Howard, J.L.; MacArthur Jr, J.W.; Newcomb, C.; Donnelly, J.P.; Kobrin, D.M.; Sheridan, M.A.; et al. Minimally invasive approach provides at least equivalent results for surgical correction of mitral regurgitation: A propensity-matched comparison. J. Thorac. Cardiovasc. Surg. 2013, 145, 748–756. [Google Scholar] [CrossRef] [PubMed]
- Sündermann, S.H.; Sromicki, J.; Biefer, H.R.C.; Seifert, B.; Holubec, T.; Falk, V.; Jacobs, S. Mitral valve surgery: Right lateral minithoracotomy or sternotomy? A systematic review and meta-analysis. J. Thorac. Cardiovasc. Surg. 2014, 148, 1989–1995.e4. [Google Scholar] [CrossRef]
- Baumgartner, H.; Falk, V.; Bax, J.J.; De Bonis, M.; Hamm, C.; Holm, P.J.; Lung, B.; Lancellotti, P.; Lansac, L.; Munoz, D.B.; et al. 2017 ESC/EACTS Guidelines for the management of valvular heart disease. Eur. Heart J. 2017, 38, 2739–2791. [Google Scholar] [CrossRef]
- Külling, M.; Corti, R.; Noll, G.; Küest, S.; Hürlimann, D.; Wyss, C.; Reho, I.; Tanner, F.C.; Külling, J.; Meinshausen, N.; et al. Heart team approach in treatment of mitral regurgitation: Patient selection and outcome. Open Heart 2020, 7, e001280. [Google Scholar] [CrossRef] [PubMed]
- Reser, D.; van Hemelrijck, M.; Pavicevic, J.; Platzmann, A.; Caliskan, E.; Falk, V.; Jacobs, S. Repair rate and durability of video assisted minimally invasive mitral valve surgery. J. Card. Surg. 2014, 29, 766–771. [Google Scholar] [CrossRef] [PubMed]
- Benedetto, U.; Head, S.J.; Angelini, G.D.; Blackstone, E.H. Statistical primer: Propensity score matching and its alternatives. Eur. J. Cardio-Thorac. Surg. 2018, 53, 1112–1117. [Google Scholar] [CrossRef]
- Seeburger, J.; Eifert, S.; Pfannmüller, B.; Garbade, J.; Vollroth, M.; Misfeld, M.; Borger, M.; Mohr, F.W. Gender differences in mitral valve surgery. Thorac. Cardiovasc. Surg. 2013, 61, 42–46. [Google Scholar]
- Falk, V.; Seeburger, J.; Czesla, M.; Borger, M.A.; Willige, J.; Kuntze, T.; Doll, N.; Borger, F.; Perrior, P.; Mohr, F.W. How does the use of polytetrafluoroethylene neochordae for posterior mitral valve prolapse (loop technique) compare with leaflet resection? A prospective randomized trial. J. Thorac. Cardiovasc. Surg. 2008, 136, 1205–1206. [Google Scholar] [CrossRef]
- Mohty, D.; Orszulak, T.A.; Schaff, H.V.; Avierinos, J.-F.; Tajik, J.A.; Enriquez-Sarano, M. Very Long-Term Survival and Durability of Mitral Valve Repair for Mitral Valve Prolapse. Circulation 2001, 104, 11–17. [Google Scholar] [CrossRef]
- Pfannmueller, B.; Misfeld, M.; Verevkin, A.; Garbade, J.; Holzhey, D.M.; Davierwala, P.; Seeburger, J.; Noack, T.; Borger, M.A. Loop neochord versus leaflet resection techniques for minimally invasive mitral valve repair: Long-term results. Eur. J. Cardio-Thoracic Surg. 2020, 59, 180–186. [Google Scholar] [CrossRef]
- David, T.E.; David, C.M.; Lafreniere-Roula, M.; Manlhiot, C. Long-term outcomes of chordal replacement with expanded polytetrafluo-roethylene sutures to repair mitral leaflet prolapse. J. Thorac. Cardiovasc. Surg. 2020, 160, 385–394. [Google Scholar] [CrossRef]
- Flameng, W.; Herijgers, P.; Bogaerts, K. Recurrence of Mitral Valve Regurgitation After Mitral Valve Repair in Degenerative Valve Disease. Circulation 2003, 107, 1609–1613. [Google Scholar] [CrossRef]
- Moscarelli, M.; Lorusso, R.; Angelini, G.D.; Di Bari, N.; Paparella, D.; Fattouch, K.; Albertini, A.; Nasso, G.; Fiorentino, F.; Speziale, G. Sex-specific differences and postoperative outcomes of minimally invasive and sternotomy valve surgery. Eur. J. Cardio-Thoracic Surg. 2021, 61, 695–702. [Google Scholar] [CrossRef]
- Dębski, M.; Taylor, R.; Abdelrahman, A.; Dębska, K.; Assaf, O.; Qadri, S.; Wong, K.Y.-K.; Vassiliou, V.; Zacharias, J. Effect of sex and surgical incision on survival after isolated primary mitral valve operations. Eur. J. Cardio-Thoracic Surg. 2022, 62, ezac273. [Google Scholar] [CrossRef]
- Buzzatti, N.; Van Hemelrijck, M.; Denti, P.; Ruggeri, S.; Schiavi, D.; Scarfò, I.S.; Reser, D.; Taramasso, M.; Weber, A.; La Canna, G.; et al. Transcatheter or surgical repair for degenerative mitral regurgitation in elderly patients: A propensity-weighted analysis. J. Thorac. Cardiovasc. Surg. 2019, 158, 86–94. [Google Scholar] [CrossRef]
- Malik, A.H.; Zaid, S.; Yandrapalli, S.; Shetty, S.; Aronow, W.S.; Ahmad, H.; Tang, G.H. Trends and Outcomes with Transcatheter Versus Surgical Mitral Valve Repair in Patients ≥80 Years of Age. Am. J. Cardiol. 2020, 125, 1083–1087. [Google Scholar] [CrossRef] [PubMed]
- Chan, V.; Chen, L.; Elmistekawy, E.; Ruel, M.; Mesana, T.G. Determinants of late outcomes in women undergoing mitral repair of myxomatous degeneration. Interact. Cardiovasc. Thorac. Surg. 2016, 23, 779–783. [Google Scholar] [CrossRef] [PubMed]
- El-Andari, R.; Bozso, S.J.; Fialka, N.M.; Kang, J.J.; Nagendran, J. Does sex impact outcomes after mitral valve surgery? A systematic review and me-ta-analysis. Scand. J. Surg. 2022, 111, 99–109. [Google Scholar] [CrossRef]
- Cho, L.; Kibbe, M.R.; Bakaeen, F.; Aggarwal, N.R.; Davis, M.B.; Karmalou, T.; Lawton, J.S.; Ouzounian, M.; Preventza, O.; Russo, A.M.; et al. Cardiac Surgery in Women in the Current Era: What Are the Gaps in Care? Circulation 2021, 144, 1172–1185. [Google Scholar] [CrossRef]
- Avierinos, J.-F.; Inamo, J.; Grigioni, F.; Gersh, B.; Shub, C.; Enriquez-Sarano, M. Sex Differences in Morphology and Outcomes of Mitral Valve Prolapse. Ann. Intern. Med. 2008, 149, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Kandula, V.; Kislitsina, O.N.; Rigolin, V.H.; Thomas, J.D.; Malaisrie, S.C.; Andrei, A.C.; Ramesh, A.; Kruse, J.; Cox, J.L.; McCarthy, P.M. Does gender bias affect outcomes in mitral valve surgery for degenerative mitral regurgitation? Interact Cardiovasc. Thorac. Surg. 2021, 33, 325–332. [Google Scholar] [CrossRef]
- Holzhey, D.M.; Shi, W.; Borger, M.; Seeburger, J.; Garbade, J.; Pfannmüller, B.; Mohr, F.W. Minimally Invasive Versus Sternotomy Approach for Mitral Valve Surgery in Patients Greater Than 70 Years Old: A Propensity-Matched Comparison. Ann. Thorac. Surg. 2011, 91, 401–405. [Google Scholar] [CrossRef] [PubMed]
- Kudo, M.; Yozu, R.; Kokaji, K.; Iwanaga, S. Feasibility of mitral valve repair using the loop technique. Ann. Thorac. Cardiovasc. Surg. 2007, 13, 21–26. [Google Scholar] [PubMed]
- Dreyfus, G.D.; Dulguerov, F.; Marcacci, C.; Haley, S.R.; Gkouma, A.; Dommerc, C.; Albert, A. Respect when you can, resect when you should”: A realistic approach to pos-terior leaflet mitral valve repair. J. Thorac. Cardiovasc. Surg. 2018, 156, 1856–1866.e3. [Google Scholar] [CrossRef]
- Ma, J.; Liu, J.; Wei, P.; Yao, X.; Zhang, Y.; Fang, L.; Chen, Z.; Liu, Y.; Tan, T.; Wu, H.; et al. Quadrangular resection versus chordal replacement for degenerative posterior mitral leaflet prolapse. Ann. Transl. Med. 2021, 9, 60. [Google Scholar] [CrossRef]
| Male | Female | p | |
|---|---|---|---|
| Number of patients | 177 (58.6) | 125 (41.4) | - |
| EuroSCORE II (%) | 1.23 ± 0.98 | 1.98 ± 1.35 | <0.001 |
| Age (years) | 62.6 ± 11.9 | 66.8 ± 11.6 | <0.001 |
| LVEF (%) | 62.9 ± 7.41 | 63.9 ± 6.52 | 0.38 |
| NYHA Class (III/IV) | 28 (15.8) | 37 (29.6) | 0.005 |
| Atrial fibrillation | 25 (14.1) | 27 (21.6) | 0.12 |
| Previous stroke | 7 (4) | 4 (3.2) | 1 |
| Coronary heart disease | 31 (17.5) | 16 (12.8) | 0.33 |
| Previous PCI | 14 (7.9) | 3 (2.4) | 0.045 |
| COPD | 3 (1.7) | 6 (4.8) | 0.74 |
| Hypertension | 51 (28.8) | 38 (30.4) | 0.8 |
| Peripheral vascular disease | 3 (1.7) | 2 (1.6) | 1 |
| Previous cardiac surgery | 2 (1.1) | 1 (0.8) | 1 |
| Mitral regurgitation | 175 (98.9) | 119 (95.2) | 0.07 |
| Mitral stenosis | 2 (1.1) | 6 (4.8) | 0.07 |
| Degenerative mitral disease | 174 (98.3) | 118 (94.4) | 0.1 |
| Functional mitral disease | 1 (0.6) | 6 (4.8) | 0.02 |
| Barlow’s disease | 26 (14.7) | 39 (31.2) | <0.001 |
| Single-segment pathology | 80 (45.2) | 31 (24.8) | <0.001 |
| Double-segment pathology | 53 (30) | 55 (55) | 0.015 |
| Posterior leaflet pathology | 60 (33.9) | 27 (21.6) | 0.02 |
| Antero-posterior pathology | 38 (21.5) | 27 (21.6) | 1 |
| Commissural pathology | 13 (7.3) | 10 (8) | 0.83 |
| Flial leaflet | 98 (55.4) | 34 (27.2) | <0.001 |
| Leaflet calcification | 7 (4) | 17 (13.6) | 0.004 |
| Annular calcification | 18 (10.2) | 26 (20.8) | 0.02 |
| Tricuspid valve regurgitation | |||
| Moderate | 10 (5.6) | 16 (12.8) | 0.038 |
| Severe | 4 (2.3) | 13 (10.4) | 0.004 |
| Male | Female | p | |
|---|---|---|---|
| Number of patients | 177 (58.6) | 125 (41.4) | - |
| Bypass time (minutes) | 164 ± 47 | 160 ± 46 | 0.34 |
| Cross-clamp time (minutes) | 107 ± 36 | 103 ± 32 | 0.28 |
| Conversion (to clamshell) | 0 | 1 (0.8) | 0.41 |
| Successful valve repair | 164 (92.6) | 97 (77.6) | <0.001 |
| 34.8 ± 2.2 | 33.6 ± 2.8 | <0.001 |
| Leaflet resection | 81 (45.7) | 26 (20.8) | <0.001 |
| Neochordae | 60 (33.9) | 30 (24) | 0.07 |
| Sliding plasty | 11 (6.2) | 3 (2.4) | 0.16 |
| Cleft closure | 82 (46.3) | 55 (44) | 0.72 |
| Commissuroplasty | 14 (7.9) | 15 (12) | 0.24 |
| Baleout replacement | 6 (3.4) | 7 (5.6) | 0.39 |
| Valve replacement | 13 (7.4) | 28 (22.4) | <0.001 |
| 0 | 3 (2.4) | 0.07 |
| 19 (10.7) | 33 (26.4) | <0.001 |
| 30.6 ± 1.8 | 29.2 ± 1.6 | 0.02 |
| Tricuspid valve repair | 8 (4.5) | 28 (22.4) | <0.001 |
| Cryoablation | 5 (2.8) | 7 (5.6) | 0.24 |
| Left atrial appendage closure | |||
| 9 (5.1) | 12 (9.6) | 0.16 |
| 11 (6.2) | 14 (11.2) | 0.14 |
| Foramen ovale closure | 16 (9) | 16 (12.8) | 0.34 |
| In-hospital mortality | 0 | 3 (2.4) | 0.06 |
| Permanent stroke | 4 (2.3) | 1 (0.8) | 0.4 |
| Tamponade | 0 | 1 (0.8) | 0.4 |
| Rethoracotomy | 1 (0.6) | 3 (2.4) | 0.3 |
| Intensive care stay (days) | 1.8 ± 2.8 | 2.2 ± 3.6 | 0.05 |
| Hospital stay (days) | 10.3 ± 4.4 | 11.2 ± 4.7 | 0.01 |
| Pacemaker implantation | 12 (6.8) | 9 (7.2) | 1 |
| Post-op ECMO | 1 (0.6) | 2 (1.6) | 0.5 |
| Need for re-operation | 1 (0.6) | 0 | 0.5 |
| LVEF postop (%) | 56.3 ± 7.6 | 58.2 ± 8.1 | 0.12 |
| Incidence of atrial fibrillation | 18 (10.2) | 29 23.3) | 0.003 |
| Mitral regurgitation > 2 | 6 (3.4) | 2 (1.6) | 0.48 |
| Mitral gradient (mmHg) | 3 ± 1.9 | 3.2 ± 1.5 | 0.29 |
| Tricuspid regurgitation > 2 | 1 (0.6) | 3 (2.4) | 0.42 |
| Male | Female | p | |
|---|---|---|---|
| Survivors | 177 (100) | 122 (97.5) | 0.07 |
| Patients followed up | 142 (80.2) | 105 (86.1) | 0.17 |
| Patients abroad (lost to FU) | 4 (2.3) | 0 | 0.14 |
| Patients did not consent | 5 (2.8) | 0 | 0.08 |
| Waiting for follow up | 26 (14.7) | 17 (13.9) | 0.87 |
| Stroke | 0 | 0 | - |
| Mortality | 7 | 2 | 0.9 |
| Reintervention | 3 | 5 | 0.2 |
| LVEF | |||
| 57.6 ± 7.2 | 59.3 ± 7.3 | 0.06 |
| 57.7 ± 7.8 | 60.8 ± 6.3 | 0.01 |
| 57.9 ± 8.2 | 61.8 ± 6.2 | 0.005 |
| NYHA III/IV | |||
| 3 (2.1) | 1 (0.8) | 0.63 |
| 3 (2.1) | 1 (0.8) | 0.26 |
| 0 | 2 (1.6) | 0.15 |
| Atrial fibrillation | |||
| 15 (10.6) | 24 (19.7) | 0.01 |
| 7 (4.9) | 14 (11.5) | 0.03 |
| 7 (4.9) | 11 (9.0) | 0.07 |
| Tricuspid regurtigation > grade 2 | |||
| 2 (1.4) | 1 (0.8) | 1 |
| 2 (1.4) | 3 (2.5) | 0.65 |
| 0 | 4 (3.3) | 0.04 |
| Mitral regurgitation > grade 2 | |||
| 6 (4.2) | 4 (3.3) | 1 |
| 6 (4.2) | 4 (3.3) | 1 |
| 7 (4.9) | 4 (3.3) | 0.82 |
| Mitral gradient (mmHg) | |||
| 2.7 ± 1.4 | 2.9 ± 1.3 | 0.06 |
| 2.7 ± 1.4 | 2.9 ± 1.6 | 0.36 |
| 2.5 ± 1.2 | 3.2 ± 1.5 | 0.03 |
| Male | Female | p | |
|---|---|---|---|
| Number of patients | 101 | 101 | - |
| EuroSCORE II (%) | 1.4 ± 1.2 | 1.4 ± 0.9 | 0.46 |
| Age (years) | 65.1 ± 11.5 | 65.8 ± 12.1 | 0.5 |
| LVEF (%) | 64.1 ± 7.3 | 64.2 ± 5.8 | 0.6 |
| NYHA Class (III/IV) | 21 (20.8) | 20 (19.8) | 1 |
| Atrial fibrillation | 17 (16.8) | 18 (17.8) | 1 |
| Previous stroke | 5 (4.95) | 4 (3.96) | 1 |
| Coronary heart disease | 14 (13.9) | 15 (14.9) | 1 |
| Previous PCI | 1 (0.99) | 2 (1.98) | 1 |
| COPD | 4 (3.96) | 3 (2.97) | 1 |
| Hypertension | 30 (29.7) | 32 (31.7) | 0.9 |
| Peripheral vascular disease | 2 (1.98) | 1 (0.99) | 1 |
| Previous cardiac surgery | 2 (1.98) | 1 (0.99) | 1 |
| Mitral regurgitation | 99 (98) | 98 (97) | 1 |
| Mitral stenosis | 2 (1.98) | 3 (2.97) | 1 |
| Degenerative mitral disease | 99 (98) | 98 (97) | 1 |
| Functional mitral disease | 1 (0.99) | 2 (1.98) | 1 |
| Barlow’s disease | 24 (23.8) | 27 (26.7) | 0.7 |
| Single-segment pathology | 32 (31.7) | 29 (28.7) | 0.7 |
| Double-segment pathology | 38 (37.6) | 41 (40.6) | 0.8 |
| Posterior leaflet pathology | 27 (26.7) | 26 (25.8) | 1 |
| Antero-posterior pathology | 22 (21.8) | 22 (21.8) | 1 |
| Commissural pathology | 9 (8.9) | 8 (7.9) | 1 |
| Leaflet calcification | 6 (5.9) | 8 (7.9) | 0.8 |
| Annular calcification | 13 (12.9) | 21 (20.8) | 0.3 |
| Tricuspid valve regurgitation | |||
| Moderate | 6 (5.9) | 10 (9.9) | 0.4 |
| Severe | 4 (3.96) | 4 (3.96) | 1 |
| Male | Female | p | |
|---|---|---|---|
| Number of patients | 101 | 101 | - |
| Bypass time (minutes) | 167 ± 51 | 162 ± 47 | 0.35 |
| Cross-clamp time (minutes) | 108 ± 39 | 104 ± 34 | 0.35 |
| Conversion (to clamshell) | 0 | 1 (0.99) | 1 |
| Successful valve repair | 91 (90.1) | 84 (81.2) | 0.21 |
| 34.7 ± 2.1 | 33.6 ± 2.8 | 0.007 |
| Leaflet resection | 47 (46.5) | 24 (23.8) | 0.001 |
| Neochordae | 30 (29.7) | 30 (29.7) | 1 |
| Sliding plasty | 8 (7.9) | 3 (2.97) | 0.21 |
| Cleft closure | 45 (44.6) | 49 (48.5) | 0.67 |
| Commissuroplasty | 5 (4.95) | 15 (14.9) | 0.03 |
| Baleout replacement | 4 (3.96) | 7 (6.93) | 0.53 |
| Valve replacement | 10 (9.9) | 17 (16.8) | 0.21 |
| 0 | 2 (1.98) | 0.49 |
| 14 (13.9) | 22 (21.8) | 0.19 |
| 30.9 ± 1.9 | 29 ± 1.6 | 0.009 |
| Tricuspid valve repair | 6 (5.94) | 15 (14.9) | 0.06 |
| Cryoablation | 3 (2.97) | 2 (1.98) | 1 |
| Left atrial appendage closure | |||
| 7 (6.93) | 9 (8.9) | 0.79 |
| 4 (3.96) | 8 (7.9) | 0.37 |
| Foramen ovale closure | 7 (6.93) | 10 (9.9) | 0.61 |
| In-hospital mortality | 0 | 2 (1.98) | 0.49 |
| Permanent stroke | 3 (2.97) | 1 (0.99) | 0.62 |
| Tamponade | 0 | 1 (0.99) | 1 |
| Rethoracotomy | 1 (0.99) | 1 (0.99) | 1 |
| Intensive care stay (days) | 2 ± 3.6 | 1.8 ± 2 | 0.39 |
| Hospital stay (days) | 10.9 ± 5.5 | 10.6 ± 3.3 | 0.5 |
| Pacemaker implantation | 8 (7.92) | 5 (4.95) | 0.56 |
| Post-op ECMO | 1 (0.99) | 2 (1.98) | 1 |
| Need for re-operation | 0 | 0 | - |
| LVEF postop (%) | 57.5 ± 7.5 | 57.3 ± 7.9 | 0.67 |
| Incidence of atrial fibrillation | 10 (9.9) | 21 (20.8) | 0.04 |
| Mitral regurgitation > 2 | 3 (2.97) | 1 (0.99) | 0.49 |
| Mitral gradient (mmHg) | 3 ± 1.4 | 3.3 ± 1.5 | 0.23 |
| Tricuspid regurgitation > 2 | 0 | 3 (2.97) | 0.18 |
| Male | Female | p | |
|---|---|---|---|
| Survivors | 101 | 99 | 0.5 |
| Patients followed up | 80 (79.2) | 87 (87.9) | 0.26 |
| Patients abroad (no FU) | 1 (0.99) | 0 | 1 |
| Patients did not consent | 3 (2.97) | 0 | 0.25 |
| Waiting for follow up | 17 (16.8) | 14 (14.14) | 0.67 |
| Stroke | 0 | 0 | - |
| Mortality | 6 (5.9) | 2 (2.1) | 0.3 |
| Reintervention | 1 (0.99) | 3 (3.45) | 0.3 |
| LVEF | |||
| 57.9 ± 7.3 | 59.8 ± 7.0 | 0.1 |
| 57.8 ± 6.8 | 60.8 ± 6.0 | 0.04 |
| 58.1 ± 7.9 | 62.6 ± 5.8 | 0.004 |
| NYHA III/IV | |||
| 1 (1.25) | 0 | 0.48 |
| 0 | 2 (2.3) | 0.22 |
| 0 | 2 (2.3) | 0.49 |
| Atrial fibrillation | |||
| 10 (12.5) | 13 (15) | 0.66 |
| 6 (7.5) | 11 (12.6) | 0.43 |
| 5 (4.9) | 9 (9.1) | 0.38 |
| Tricuspid regurtigation > grade 2 | |||
| 1 (1.25) | 1 (1.15) | 0.74 |
| 2 (2.5) | 3 (3.45) | 0.87 |
| 0 | 3 (3.45) | 0.08 |
| Mitral regurgitation > grade 2 | |||
| 2 (2.5) | 2 (2.3) | 0.7 |
| 3 (3.75) | 4 (4.6) | 1 |
| 2 (2.5) | 4 (4.6) | 0.51 |
| Mitral gradient (mmHg) | |||
| 2.8 ± 1.4 | 2.9 ± 1.4 | 0.44 |
| 2.9 ± 1.6 | 2.9 ± 1.7 | 0.95 |
| 2.5 ± 1.2 | 3.2 ± 1.6 | 0.05 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Passos, L.; Lavanchy, I.; Aymard, T.; Morjan, M.; Kapos, I.; Corti, R.; Gruenenfelder, J.; Biaggi, P.; Reser, D. Propensity Matched Outcomes of Minimally Invasive Mitral Surgery: Does a Heart-Team Approach Eliminate Female Gender as an Independent Risk Factor? J. Pers. Med. 2023, 13, 949. https://doi.org/10.3390/jpm13060949
Passos L, Lavanchy I, Aymard T, Morjan M, Kapos I, Corti R, Gruenenfelder J, Biaggi P, Reser D. Propensity Matched Outcomes of Minimally Invasive Mitral Surgery: Does a Heart-Team Approach Eliminate Female Gender as an Independent Risk Factor? Journal of Personalized Medicine. 2023; 13(6):949. https://doi.org/10.3390/jpm13060949
Chicago/Turabian StylePassos, Laina, Isabel Lavanchy, Thierry Aymard, Mohammed Morjan, Ioannis Kapos, Roberto Corti, Juerg Gruenenfelder, Patric Biaggi, and Diana Reser. 2023. "Propensity Matched Outcomes of Minimally Invasive Mitral Surgery: Does a Heart-Team Approach Eliminate Female Gender as an Independent Risk Factor?" Journal of Personalized Medicine 13, no. 6: 949. https://doi.org/10.3390/jpm13060949
APA StylePassos, L., Lavanchy, I., Aymard, T., Morjan, M., Kapos, I., Corti, R., Gruenenfelder, J., Biaggi, P., & Reser, D. (2023). Propensity Matched Outcomes of Minimally Invasive Mitral Surgery: Does a Heart-Team Approach Eliminate Female Gender as an Independent Risk Factor? Journal of Personalized Medicine, 13(6), 949. https://doi.org/10.3390/jpm13060949






