The Relative Apical Sparing Strain Pattern in Severe Aortic Valve Stenosis: A Marker of Adverse Cardiac Remodeling
Abstract
1. Introduction
2. Materials and Methods
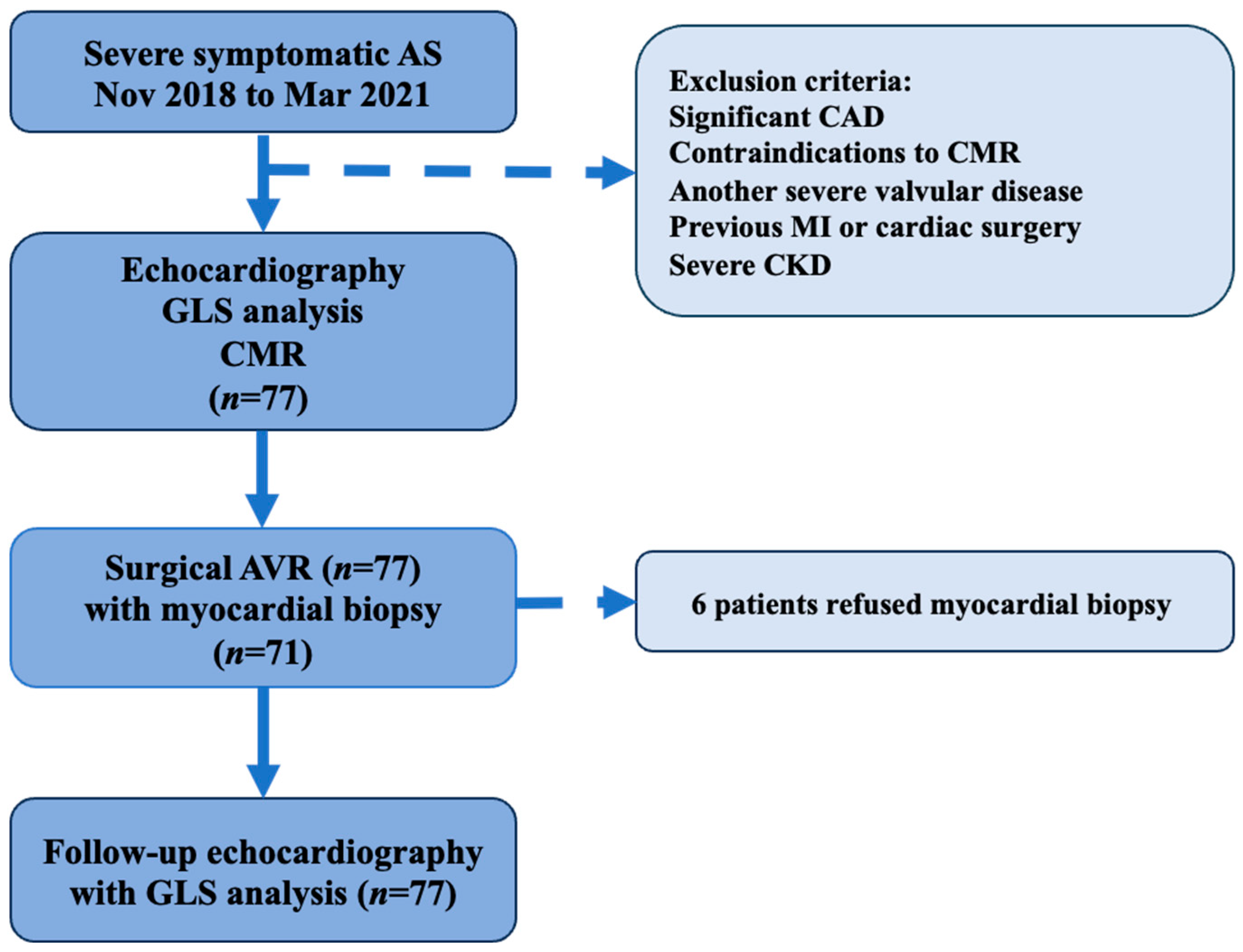
2.1. Study Design and Population
2.2. Inclusion/Exclusion Criteria
2.3. Cardiac Imaging
2.3.1. Echocardiography
2.3.2. CMR Protocol
2.4. Histological Analysis
2.5. Statistical Analysis
2.6. Intra-Observer and Inter-Observer Variability Analysis
3. Results
3.1. Study Cohort Data
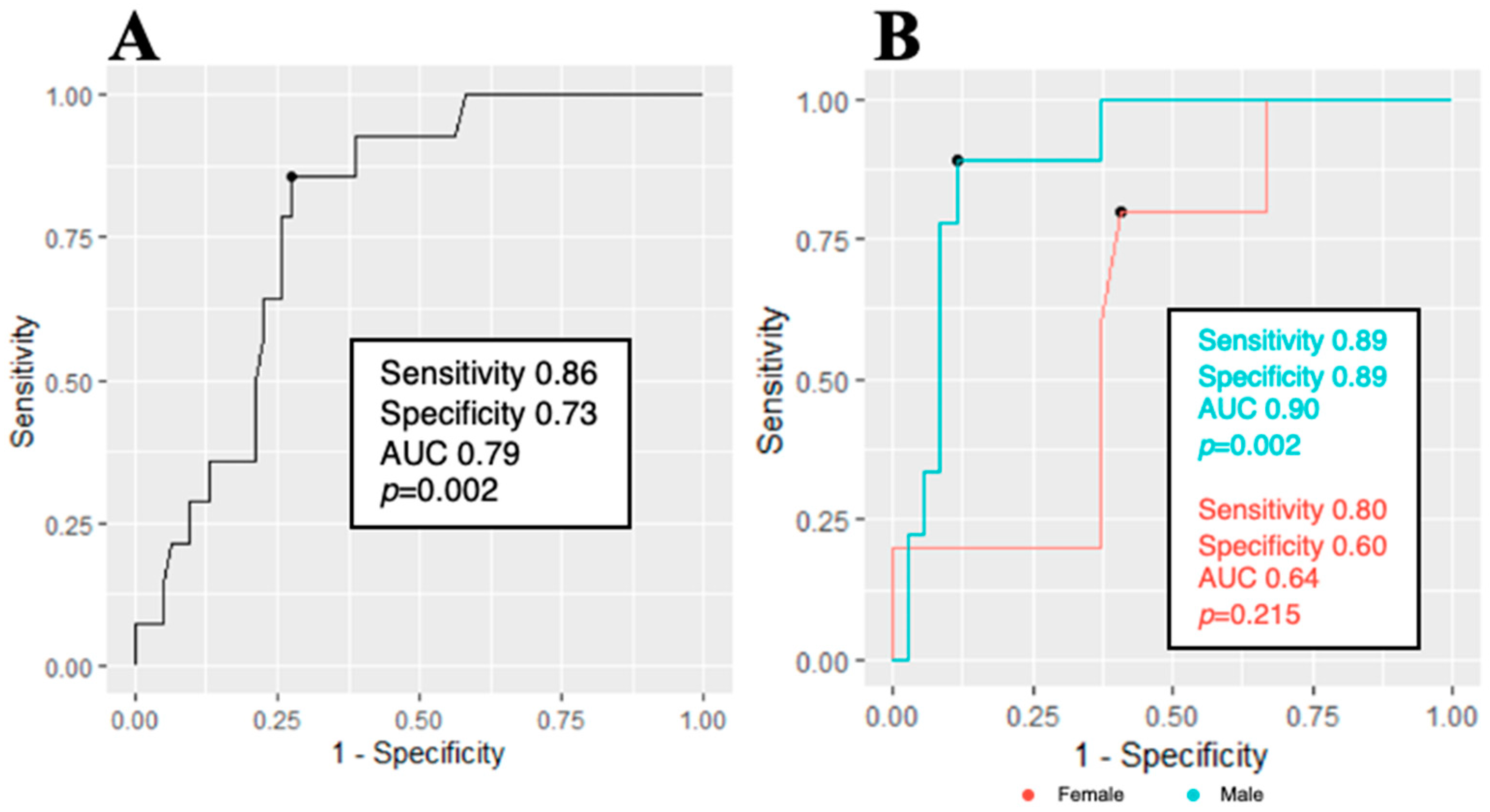
3.2. Data Comparison between Patients with and without an RAS Strain Pattern
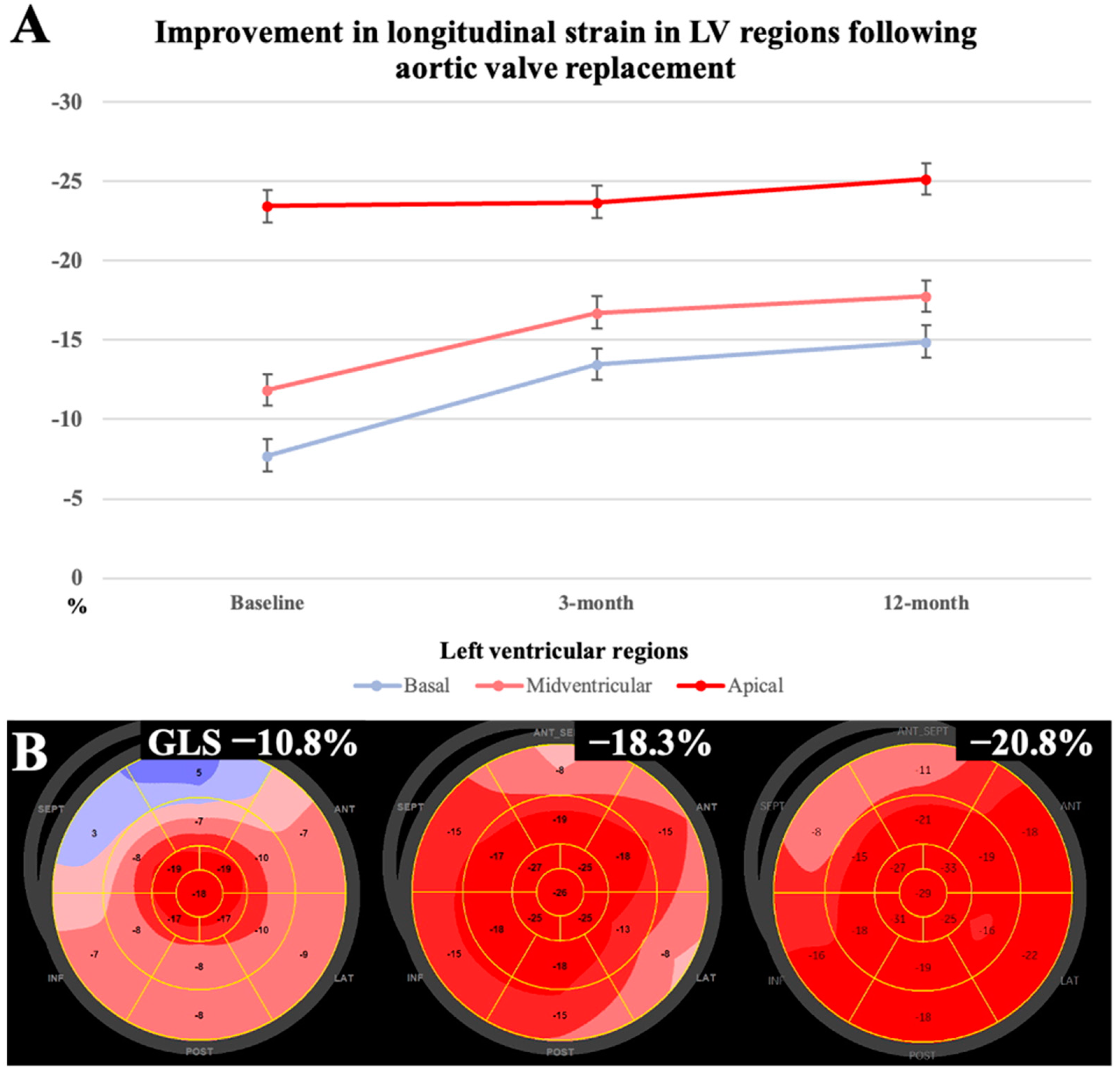
3.3. Follow-Up Echocardiography Data
4. Discussion
4.1. CA Links with AS
4.2. Consequences of AS-Induced Remodeling
4.3. GLS as a Prognostic Marker
4.4. RAS Strain Pattern after SAVR
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AS | aortic stenosis |
| ATTR-CA | transthyretin cardiac amyloidosis |
| AV | aortic valve |
| AVA | aortic valve area |
| SAVR | surgical aortic valve replacement |
| BNP | brain natriuretic peptide |
| BSA | body surface area |
| CA | cardiac amyloidosis |
| CAD | coronary artery disease |
| CKD | chronic kidney disease |
| CVF | collagen volume fraction |
| CMR | cardiovascular magnetic resonance |
| ECG | electrocardiography |
| ECV | extracellular volume |
| GLS | global longitudinal strain |
| Hs–Tn–I | high-sensitivity troponin I |
| LGE | late gadolinium enhancement |
| LV | left ventricle |
| LVEF | left ventricular ejection fraction |
| MI | myocardial infarction |
| NYHA | New York Heart Association |
| RAS | relative apical sparing |
| STE | speckle-tracking echocardiography |
| TAVI | transcatheter aortic valve implantation |
| T1 | T1 myocardial relaxation |
References
- Rapezzi, C.; Quarta, C.C.; Guidalotti, P.L.; Longhi, S.; Pettinato, C.; Leone, O.; Ferlini, A.; Salvi, F.; Gallo, P.; Gagliardi, C.; et al. Usefulness and Limitations of 99mTc-3,3-Diphosphono-1,2-Propanodicarboxylic Acid Scintigraphy in the Aetiological Diagnosis of Amyloidotic Cardiomyopathy. Eur. J. Nucl. Med. Mol. Imaging 2011, 38, 470–478. [Google Scholar] [CrossRef] [PubMed]
- Dumesnil, J.G.; Pibarot, P.; Carabello, B. Paradoxical Low Flow and/or Low Gradient Severe Aortic Stenosis despite Preserved Left Ventricular Ejection Fraction: Implications for Diagnosis and Treatment. Eur. Heart J. 2010, 31, 281–289. [Google Scholar] [CrossRef] [PubMed]
- Galat, A.; Guellich, A.; Bodez, D.; Slama, M.; Dijos, M.; Zeitoun, D.M.; Milleron, O.; Attias, D.; Dubois-Randé, J.-L.; Mohty, D.; et al. Aortic Stenosis and Transthyretin Cardiac Amyloidosis: The Chicken or the Egg? Eur. Heart J. 2016, 37, 3525–3531. [Google Scholar] [CrossRef] [PubMed]
- Balciunaite, G.; Rimkus, A.; Zurauskas, E.; Zaremba, T.; Palionis, D.; Valeviciene, N.; Aidietis, A.; Serpytis, P.; Rucinskas, K.; Sogaard, P.; et al. Transthyretin Cardiac Amyloidosis in Aortic Stenosis: Prevalence, Diagnostic Challenges, and Clinical Implications. Hell. J. Cardiol. 2020, 61, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Damy, T.; Judge, D.P.; Kristen, A.V.; Berthet, K.; Li, H.; Aarts, J. Cardiac Findings and Events Observed in an Open-Label Clinical Trial of Tafamidis in Patients with Non-Val30Met and Non-Val122Ile Hereditary Transthyretin Amyloidosis. J. Cardiovasc. Transl. Res. 2015, 8, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Nitsche, C.; Scully, P.R.; Patel, K.P.; Kammerlander, A.A.; Koschutnik, M.; Dona, C.; Wollenweber, T.; Ahmed, N.; Thornton, G.D.; Kelion, A.D.; et al. Prevalence and Outcomes of Concomitant Aortic Stenosis and Cardiac Amyloidosis. J. Am. Coll. Cardiol. 2021, 77, 128–139. [Google Scholar] [CrossRef] [PubMed]
- Rosenblum, H.; Masri, A.; Narotsky, D.L.; Goldsmith, J.; Hamid, N.; Hahn, R.T.; Kodali, S.; Vahl, T.; Nazif, T.; Khalique, O.K.; et al. Unveiling Outcomes in Coexisting Severe Aortic Stenosis and Transthyretin Cardiac Amyloidosis. Eur. J. Heart Fail. 2021, 23, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Scully, P.R.; Patel, K.P.; Treibel, T.A.; Thornton, G.D.; Hughes, R.K.; Chadalavada, S.; Katsoulis, M.; Hartman, N.; Fontana, M.; Pugliese, F.; et al. Prevalence and Outcome of Dual Aortic Stenosis and Cardiac Amyloid Pathology in Patients Referred for Transcatheter Aortic Valve Implantation. Eur. Heart J. 2020, 41, 2759–2767. [Google Scholar] [CrossRef]
- Chacko, L.; Martone, R.; Bandera, F.; Lane, T.; Martinez-Naharro, A.; Boldrini, M.; Rezk, T.; Whelan, C.; Quarta, C.; Rowczenio, D.; et al. Echocardiographic Phenotype and Prognosis in Transthyretin Cardiac Amyloidosis. Eur. Heart J. 2020, 41, 1439–1447. [Google Scholar] [CrossRef]
- Mitchell, C.; Rahko, P.S.; Blauwet, L.A.; Canaday, B.; Finstuen, J.A.; Foster, M.C.; Horton, K.; Ogunyankin, K.O.; Palma, R.A.; Velazquez, E.J. Guidelines for Performing a Comprehensive Transthoracic Echocardiographic Examination in Adults: Recommendations from the American Society of Echocardiography. J. Am. Soc. Echocardiogr. 2019, 32, 1–64. [Google Scholar] [CrossRef]
- Lang, R.M.; Badano, L.P.; Mor-Avi, V.; Afilalo, J.; Armstrong, A.; Ernande, L.; Flachskampf, F.A.; Foster, E.; Goldstein, S.A.; Kuznetsova, T.; et al. Recommendations for Cardiac Chamber Quantification by Echocardiography in Adults: An Update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J. Am. Soc. Echocardiogr. 2015, 28, 1–39.e14. [Google Scholar] [CrossRef] [PubMed]
- Abecasis, J.; Lopes, P.; Santos, R.R.; Maltês, S.; Guerreiro, S.; Ferreira, A.; Freitas, P.; Ribeiras, R.; Andrade, M.J.; Manso, R.T.; et al. Prevalence and Significance of Relative Apical Sparing in Aortic Stenosis: Insights from an Echo and Cardiovascular Magnetic Resonance Study of Patients Referred for Surgical Aortic Valve Replacement. Eur. Heart J. Cardiovasc. Imaging 2023, 24, 1033–1042. [Google Scholar] [CrossRef] [PubMed]
- Vahanian, A.; Beyersdorf, F.; Praz, F.; Milojevic, M.; Baldus, S.; Bauersachs, J.; Capodanno, D.; Conradi, L.; De Bonis, M.; De Paulis, R.; et al. 2021 ESC/EACTS Guidelines for the Management of Valvular Heart Disease. Eur. Heart J. 2022, 43, 561–632. [Google Scholar] [CrossRef] [PubMed]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research Electronic Data Capture (REDCap)—A Metadata-Driven Methodology and Workflow Process for Providing Translational Research Informatics Support. J. Biomed. Inform. 2009, 42, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Baumgartner, H.; Hung, J.; Bermejo, J.; Chambers, J.B.; Edvardsen, T.; Goldstein, S.; Lancellotti, P.; LeFevre, M.; Miller, F.; Otto, C.M. Recommendations on the Echocardiographic Assessment of Aortic Valve Stenosis: A Focused Update from the European Association of Cardiovascular Imaging and the American Society of Echocardiography. J. Am. Soc. Echocardiogr. 2017, 30, 372–392. [Google Scholar] [CrossRef] [PubMed]
- Voigt, J.-U.; Pedrizzetti, G.; Lysyansky, P.; Marwick, T.H.; Houle, H.; Baumann, R.; Pedri, S.; Ito, Y.; Abe, Y.; Metz, S.; et al. Definitions for a Common Standard for 2D Speckle Tracking Echocardiography: Consensus Document of the EACVI/ASE/Industry Task Force to Standardize Deformation Imaging. Eur. Heart J. Cardiovasc. Imaging 2015, 16, 1–11. [Google Scholar] [CrossRef]
- Balčiūnaitė, G.; Besusparis, J.; Palionis, D.; Žurauskas, E.; Skorniakov, V.; Janušauskas, V.; Zorinas, A.; Zaremba, T.; Valevičienė, N.; Šerpytis, P.; et al. Exploring Myocardial Fibrosis in Severe Aortic Stenosis: Echo, CMR and Histology Data from FIB-AS Study. Int. J. Cardiovasc. Imaging 2022, 38, 1555–1568. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: https://www.R-project.org/ (accessed on 12 September 2018).
- Koyama, J.; Ray-Sequin, P.A.; Falk, R.H. Longitudinal Myocardial Function Assessed by Tissue Velocity, Strain, and Strain Rate Tissue Doppler Echocardiography in Patients with AL (Primary) Cardiac Amyloidosis. Circulation 2003, 107, 2446–2452. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.-N.; Liu, Y.-N.; Cheng, X.-Q.; Liu, H.-Y.; Zhang, J.; Li, L.; Sun, J.; Gao, Y.-P.; Lu, R.-R.; Gao, Y.-P.; et al. Relative Apical Sparing Obtained with Speckle Tracking Echocardiography Is Not a Sensitive Parameter for Diagnosing Light-Chain Cardiac Amyloidosis. Quant. Imaging Med. Surg. 2024, 14, 2357–2369. [Google Scholar] [CrossRef]
- Yang, C.-H.; Takeuchi, M.; Nabeshima, Y.; Yamashita, E.; Izumo, M.; Ishizu, T.; Seo, Y. Prognostic Value of Apical Sparing of Longitudinal Strain in Patients with Symptomatic Aortic Stenosis. Acta Cardiol. Sin. 2022, 38, 341–351. [Google Scholar] [CrossRef]
- Santos, R.R.; Abecasis, J.; Maltes, S.; Mendes, G.S.; Guerreiro, S.; Padrao, C.; Freitas, P.; Ferreira, A.; Ribeiras, R.; Andrade, M.J.; et al. Relative Apical Sparing in Severe Aortic Stenosis: Does It Mean Concomitant Amyloid Cardiomyopathy? Eur. Heart J. 2022, 43, ehac544.137. [Google Scholar] [CrossRef]
- Robin, G.; Cognet, T.; Bouisset, F.; Cariou, E.; Méjean, S.; Pradel, S.; Colombat, M.; Galinier, M.; Carrié, D.; Lairez, O.; et al. Value of Longitudinal Strain to Identify Wild-Type Transthyretin Amyloidosis in Patients with Aortic Stenosis. Circ. J. 2021, 85, 1494–1504. [Google Scholar] [CrossRef]
- Thakker, J.; Okwuosa, T.M. The Limitations of Apical Sparing as an Echocardiographic Predictor of Cardiac Amyloidosis. J. Am. Coll. Cardiol. 2024, 83, 4393. [Google Scholar] [CrossRef]
- Phelan, D.; Collier, P.; Thavendiranathan, P.; Popović, Z.B.; Hanna, M.; Plana, J.C.; Marwick, T.H.; Thomas, J.D. Relative Apical Sparing of Longitudinal Strain Using Two-Dimensional Speckle-Tracking Echocardiography Is Both Sensitive and Specific for the Diagnosis of Cardiac Amyloidosis. Heart 2012, 98, 1442–1448. [Google Scholar] [CrossRef]
- De Gaspari, M.; Sinigiani, G.; De Michieli, L.; Della Barbera, M.; Rizzo, S.; Thiene, G.; Iliceto, S.; Perazzolo Marra, M.; Mele, D.; Basso, C.; et al. Relative Apical Sparing in Cardiac Amyloidosis Is Not Always Explained by an Amyloid Gradient. Eur. Heart J. Cardiovasc. Imaging 2023, 24, 1258–1268. [Google Scholar] [CrossRef]
- Rapezzi, C.; Fontana, M. Relative Left Ventricular Apical Sparing of Longitudinal Strain in Cardiac Amyloidosis: Is It Just Amyloid Infiltration? JACC Cardiovasc. Imaging 2019, 12, 1174–1176. [Google Scholar] [CrossRef] [PubMed]
- Ternacle, J.; Bodez, D.; Guellich, A.; Audureau, E.; Rappeneau, S.; Lim, P.; Radu, C.; Guendouz, S.; Couetil, J.-P.; Benhaiem, N.; et al. Causes and Consequences of Longitudinal LV Dysfunction Assessed by 2D Strain Echocardiography in Cardiac Amyloidosis. JACC Cardiovasc. Imaging 2016, 9, 126–138. [Google Scholar] [CrossRef]
- Jaiswal, V.; Agrawal, V.; Khulbe, Y.; Hanif, M.; Huang, H.; Hameed, M.; Shrestha, A.B.; Perone, F.; Parikh, C.; Gomez, S.I.; et al. Cardiac Amyloidosis and Aortic Stenosis: A State-of-the-Art Review. Eur. Heart J. Open 2023, 3, oead106. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Naharro, A.; Treibel, T.A.; Abdel-Gadir, A.; Bulluck, H.; Zumbo, G.; Knight, D.S.; Kotecha, T.; Francis, R.; Hutt, D.F.; Rezk, T.; et al. Magnetic Resonance in Transthyretin Cardiac Amyloidosis. J. Am. Coll. Cardiol. 2017, 70, 466–477. [Google Scholar] [CrossRef]
- Fung, M.J.; Thomas, L.; Leung, D.Y. Alterations in Layer-Specific Left Ventricular Global Longitudinal and Circumferential Strain in Patients with Aortic Stenosis: A Comparison of Aortic Valve Replacement versus Conservative Management Over a 12-Month Period. J. Am. Soc. Echocardiogr. 2019, 32, 92–101. [Google Scholar] [CrossRef]
- Algranati, D.; Kassab, G.S.; Lanir, Y. Why Is the Subendocardium More Vulnerable to Ischemia? A New Paradigm. Am. J. Physiol. Heart Circ. Physiol. 2011, 300, H1090–H1100. [Google Scholar] [CrossRef]
- Thornton, G.D.; Bennett, J.; Gama, F.; Nitsche, C.; Mravljak, R.; Abiodun, A.T.; Aziminia, N.; Xue, H.; Pierce, I.; Lloyd, G.; et al. Global Myocardial Ischaemia in Severe Aortic Stenosis Reverses Early after Aortic Valve Replacement. Eur. Heart J. Cardiovasc. Imaging 2023, 24, jead119.058. [Google Scholar] [CrossRef]
- Büchi, M.; Hess, O.M.; Murakami, T.; Krayenbuehl, H.P. Left Ventricular Wall Stress Distribution in Chronic Pressure and Volume Overload: Effect of Normal and Depressed Contractility on Regional Stress-Velocity Relations. Basic. Res. Cardiol. 1990, 85, 367–383. [Google Scholar] [CrossRef]
- Fung, M.J.; Leung, D.Y.; Thomas, L. Differential Myocardial Fibre Involvement by Strain Analysis in Patients with Aortic Stenosis. Heart Lung Circ. 2018, 27, 1357–1367. [Google Scholar] [CrossRef] [PubMed]
- Ng, A.C.T.; Prihadi, E.A.; Antoni, M.L.; Bertini, M.; Ewe, S.H.; Ajmone Marsan, N.; Leung, D.Y.; Delgado, V.; Bax, J.J. Left Ventricular Global Longitudinal Strain Is Predictive of All-Cause Mortality Independent of Aortic Stenosis Severity and Ejection Fraction. Eur. Heart J. Cardiovasc. Imaging 2018, 19, 859–867. [Google Scholar] [CrossRef]
- Weidemann, F.; Herrmann, S.; Störk, S.; Niemann, M.; Frantz, S.; Lange, V.; Beer, M.; Gattenlöhner, S.; Voelker, W.; Ertl, G.; et al. Impact of Myocardial Fibrosis in Patients with Symptomatic Severe Aortic Stenosis. Circulation 2009, 120, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Ricci, F.; Ceriello, L.; Khanji, M.Y.; Dangas, G.; Bucciarelli-Ducci, C.; Di Mauro, M.; Fedorowski, A.; Zimarino, M.; Gallina, S. Prognostic Significance of Cardiac Amyloidosis in Patients with Aortic Stenosis: A Systematic Review and Meta-Analysis. JACC Cardiovasc. Imaging 2021, 14, 293–295. [Google Scholar] [CrossRef] [PubMed]
- Dinh, W.; Nickl, W.; Smettan, J.; Kramer, F.; Krahn, T.; Scheffold, T.; Coll Barroso, M.T.; Brinkmann, H.; Koehler, T.; Lankisch, M.; et al. Reduced Global Longitudinal Strain in Association to Increased Left Ventricular Mass in Patients with Aortic Valve Stenosis and Normal Ejection Fraction: A Hybrid Study Combining Echocardiography and Magnetic Resonance Imaging. Cardiovasc. Ultrasound 2010, 8, 29. [Google Scholar] [CrossRef]
- Treibel, T.A.; Kozor, R.; Fontana, M.; Torlasco, C.; Reant, P.; Badiani, S.; Espinoza, M.; Yap, J.; Diez, J.; Hughes, A.D.; et al. Sex Dimorphism in the Myocardial Response to Aortic Stenosis. JACC Cardiovasc. Imaging 2018, 11, 962–973. [Google Scholar] [CrossRef]
- Treibel, T.A.; Badiani, S.; Lloyd, G.; Moon, J.C. Multimodality Imaging Markers of Adverse Myocardial Remodeling in Aortic Stenosis. JACC Cardiovasc. Imaging 2019, 12, 1532–1548. [Google Scholar] [CrossRef]
- Treibel, T.A.; Fontana, M.; Gilbertson, J.A.; Castelletti, S.; White, S.K.; Scully, P.R.; Roberts, N.; Hutt, D.F.; Rowczenio, D.M.; Whelan, C.J.; et al. Occult Transthyretin Cardiac Amyloid in Severe Calcific Aortic Stenosis. Circulation: Cardio-vascular Imaging 2016, 9, e005066. [Google Scholar] [CrossRef] [PubMed]
- Abecasis, J.; Gomes Pinto, D.; Ramos, S.; Masci, P.G.; Cardim, N.; Gil, V.; Félix, A. Left Ventricular Remodeling in Degenerative Aortic Valve Stenosis. Curr. Probl. Cardiol. 2021, 46, 100801. [Google Scholar] [CrossRef] [PubMed]
- Anan, R.; Imoto, T.; Onizuka, K.; Watanabe, H.; Mori, W.; Murakoso, M. Concentric Hypertrophy Geometry Is a Significant Determinant of Impaired Global Longitudinal Strain in Patients with Normal Cardiac Structure and Function. Heliyon 2023, 9, e16252. [Google Scholar] [CrossRef]
- Abecasis, J.; Mendes, G.; Ferreira, A.; Andrade, M.J.; Ribeiras, R.; Ramos, S.; Masci, P.G.; Gil, V. Relative Apical Sparing in Patients with Severe Aortic Stenosis: Prevalence and Significance. Eur. Heart J. 2020, 41, ehaa946.1987. [Google Scholar] [CrossRef]
- Demirelli, S.; Degirmenci, H.; Bilen, H.; Ermis, E.; Duman, H.; Arisoy, A.; Bakirci, E.M.; Ipek, E.; Askin, L. Left Ventricular Mechanics in Behcet’s Disease: A Speckle Tracking Echocardiographic Study. Bosn. J. Basic. Med. Sci. 2014, 14, 160–164. [Google Scholar] [PubMed]
- Russo, C.; Jin, Z.; Elkind, M.S.V.; Rundek, T.; Homma, S.; Sacco, R.L.; Di Tullio, M.R. Prevalence and Prognostic Value of Subclinical Left Ventricular Systolic Dysfunction by Global Longitudinal Strain in a Community-Based Cohort. Eur. J. Heart Fail. 2014, 16, 1301–1309. [Google Scholar] [CrossRef] [PubMed]
- Hoque, M.S.; Ahmed, C.M.; Osmany, D.F.; Banerjee, S.K. Evaluation of Subclinical LV Systolic Dysfunction by Global Longitudinal Strain Using 2D-Speckle Tracking Echocardiography in Patients Having Angina with Normal Epicardial Coronary Arteries. Eur. Heart J. 2022, 43, ehab849.009. [Google Scholar] [CrossRef]
- Al-Rashid, F.; Totzeck, M.; Saur, N.; Jánosi, R.A.; Lind, A.; Mahabadi, A.A.; Rassaf, T.; Mincu, R.-I. Global Longitudinal Strain Is Associated with Better Outcomes in Transcatheter Aortic Valve Replacement. BMC Cardiovasc. Disord. 2020, 20, 267. [Google Scholar] [CrossRef] [PubMed]
- Kaler, G.P.S.; Mahla, R.; Mahla, H.; Choudhary, S.; Singh, G.; Patel, R.; Kaler, N. Speckle Tracking Echocardiographic Assessment of Left Ventricular Function by Myocardial Strain before and after Aortic Valve Replacement. J. Saudi Heart Assoc. 2022, 33, 353–363. [Google Scholar] [CrossRef] [PubMed]
- Vollema, E.M.; Sugimoto, T.; Shen, M.; Tastet, L.; Ng, A.C.T.; Abou, R.; Marsan, N.A.; Mertens, B.; Dulgheru, R.; Lancellotti, P.; et al. Association of Left Ventricular Global Longitudinal Strain with Asymptomatic Severe Aortic Stenosis: Natural Course and Prognostic Value. JAMA Cardiol. 2018, 3, 839–847. [Google Scholar] [CrossRef]
- Carstensen, H.G.; Larsen, L.H.; Hassager, C.; Kofoed, K.F.; Jensen, J.S.; Mogelvang, R. Basal Longitudinal Strain Predicts Future Aortic Valve Replacement in Asymptomatic Patients with Aortic Stenosis. Eur. Heart J. Cardiovasc. Imaging 2016, 17, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Saito, M.; Wake, D.; Higaki, R.; Inaba, S.; Morioka, H.; Sumimoto, T. 4920Prognostic Implication of Relative Apical Sparing Pattern in Non-Ischemic Patients with Diffuse Left Ventricular Hypertrophy. Eur. Heart J. 2018, 39, ehy566.4920. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, M.; Chen, H.; Li, H. Prognostic Value of Global Longitudinal Strain in Asymptomatic Aortic Stenosis: A Systematic Review and Meta-Analysis. Front. Cardiovasc. Med. 2022, 9, 778027. [Google Scholar] [CrossRef] [PubMed]
- Bohbot, Y.; de Meester de Ravenstein, C.; Chadha, G.; Rusinaru, D.; Belkhir, K.; Trouillet, C.; Pasquet, A.; Marechaux, S.; Vanoverschelde, J.-L.; Tribouilloy, C. Relationship Between Left Ventricular Ejection Fraction and Mortality in Asymptomatic and Minimally Symptomatic Patients with Severe Aortic Stenosis. JACC Cardiovasc. Imaging 2019, 12, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Di Pietro, E.; Frittitta, V.; Motta, S.; Strazzieri, O.; Valvo, R.; Reddavid, C.; Costa, G.; Tamburino, C. Treatment in Patients with Severe Asymptomatic Aortic Stenosis: Is It Best Not to Wait? Eur. Heart J. Suppl. 2022, 24, I170–I174. [Google Scholar] [CrossRef] [PubMed]
- Le, T.-T.; Huang, W.; Singh, G.K.; Toh, D.-F.; Ewe, S.H.; Tang, H.C.; Loo, G.; Bryant, J.A.; Ang, B.; Tay, E.L.-W.; et al. Echocardiographic Global Longitudinal Strain Is Associated with Myocardial Fibrosis and Predicts Outcomes in Aortic Stenosis. Front. Cardiovasc. Med. 2021, 8, 750016. [Google Scholar] [CrossRef] [PubMed]
- Zafirovska, P.; Rosalia, R.A.; Ismail, L.G.; Matveeva, N.; Mitrev, Z. Preoperative Left Ventricular Global Longitudinal Strain Identifies Aortic Stenosis Patients with Improved Postoperative Recovery of Left Ventricular Geometry: A Prospective Cohort Study. Braz. J. Cardiovasc. Surg. 2022, 37, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.K.; Fortuni, F.; Kuneman, J.H.; Vollema, E.M.; van der Kley, F.; Marsan, N.A.; Delgado, V.; Bax, J.J. Changes in Computed-Tomography-Derived Segmental Left Ventricular Longitudinal Strain after Transcatheter Aortic Valve Implantation. Am. J. Cardiol. 2023, 198, 95–100. [Google Scholar] [CrossRef]
- Nitsche, C.; Koschutnik, M.; Donà, C.; Radun, R.; Mascherbauer, K.; Kammerlander, A.; Heitzinger, G.; Dannenberg, V.; Spinka, G.; Halavina, K.; et al. Reverse Remodeling Following Valve Replacement in Coexisting Aortic Stenosis and Transthyretin Cardiac Amyloidosis. Circ. Cardiovasc. Imaging 2022, 15, e014115. [Google Scholar] [CrossRef]

| Variables | All Patients (n = 77) | RAS Strain Pattern-Positive Patients (n = 14) | RAS Strain Pattern-Negative Patients (n = 63) | p-Value | |
|---|---|---|---|---|---|
| Age, years | 70 (62–73) | 67 (63–71) | 71 (62–73) | 0.415 | |
| Sex, female | 45 (58) | 9 (64) | 36 (57) | 0.849 | |
| BSA, m2 | 1.9 ± 0.2 | 1.9 ± 0.1 | 1.9 ± 0.2 | 0.822 | |
| Comorbidities | |||||
| Hypertension | 69 (90) | 13 (92) | 56 (88) | 0.597 | |
| Dyslipidemia | 61 (79) | 9 (64) | 52 (83) | 0.128 | |
| Unobstructive CAD | 30 (39) | 4 (29) | 26 (41) | 0.378 | |
| Diabetes mellitus | 12 (16) | 4 (27) | 8 (13) | 0.139 | |
| Atrial fibrillation | 2 (3) | 0 (0) | 2 (3) | 0.499 | |
| Symptoms and functional status | |||||
| Dyspnoea | 54 (70) | 9 (64) | 45 (71) | 0.597 | |
| Chest pain | 35 (47) | 8 (57) | 27 (45) | 0.413 | |
| Syncope | 8 (10) | 3 (21) | 5 (8) | 0.135 | |
| 6MWT, m | 369 (300–420) | 318 (284–399) | 369 (332–420) | 0.220 | |
| NYHA functional class | I | 15 (19) | 1 (7) | 14 (22) | 0.561 * |
| II | 22 (29) | 5 (36) | 17 (27) | ||
| III | 37 (48) | 8 (57) | 29 (46) | ||
| IV | 3 (4) | 0 (0) | 3 (5) | ||
| Risk scores | |||||
| STS-PROM, % | 1.6 (1.2–2.4) | 1.8 (1.4–2.2) | 1.6 (1.0–2.5) | 0.629 | |
| EuroSCORE II, % | 1.1 (0.7–1.6) | 1.0 (0.8–1.5) | 1.1 (0.7–1.7) | 0.771 | |
| ECG parameters | |||||
| S–L voltage index, mm | 31.2 ± 10 | 35.8 (29.5–44) | 30.1 (23–37.8) | 0.058 | |
| QRS duration, ms | 94 (86–102) | 96.0 ± (92.5–102.0) | 91.0 (86.0–99.5) | 0.224 | |
| Echocardiography data | |||||
| AV area index, cm2/m2 | 0.45 ± 0.1 | 0.41 ± 0.1 | 0.45 ± 0.1 | 0.163 | |
| Peak AV velocity, m/s | 4.9 ± 0.6 | 5.3 ± 0.7 | 4.8 ± 0.6 | 0.005 | |
| Mean AV gradient, mmHg | 54.9 (45–70) | 70 ± 20 | 56 ± 14 | 0.013 | |
| Low gradient AS | 8 (10) | 1 (7) | 7 (11) | 0.660 | |
| IVSd, mm | 12.8 ± 1.7 | 14.6 ± 1.1 | 12.4 ± 1.5 | <0.001 | |
| LVEdd, mm | 50 (47–55) | 51.8 ± 4.4 | 51.2 ± 5.6 | 0.384 | |
| LVEsd, mm | 32 (29–35) | 34.5 ± 6 | 32 ± 6 | 0.124 | |
| E/A ratio | 1.2 ± 0.5 | 1.4 ± 0.6 | 1.1 ± 0.4 | 0.042 | |
| E deceleration time, ms | 245 (212–308) | 223 (200–265) | 251 (223–314) | 0.190 | |
| Posterior wall diameter, mm | 12 (10–12) | 12 (12–14) | 11 (10–12) | 0.001 | |
| E/e’ septal | 17 (13–21) | 17.1 (16–18) | 16 (13–20) | 0.238 | |
| E/e’ lateral | 13 (11–17) | 15 (11–17) | 13 (11–17) | 0.804 | |
| E/e’ mean | 15 (12–18) | 16 ± 7 | 16 ± 6 | 0.717 | |
| LA volume index, mL/m2 | 48.6 ± 12 | 52 ± 14 | 48 ± 12 | 0.253 | |
| PASP, mmHg | 33 (29–42) | 39 ± 18 | 38 ± 14 | 0.826 | |
| RV S’, cm/s | 12 (10–13) | 12 (10–13) | 11 (10–13) | 0.771 | |
| TAPSE | 22 ± 3 | 21 ± 3 | 22 ± 3 | 0.057 | |
| GLS, % | −18 ± 5 | −14.9 ± 3 | −18.7 ± 5 | 0.002 | |
| GLS >−15% | 16 (21) | 7 (50) | 9 (14) | 0.006 | |
| CMR data | |||||
| IVSd, mm | 13.3 (12–15) | 14.8 ± 1 | 12.9 ± 2 | 0.004 | |
| LVEdd, mm | 50.4 ± 6 | 54 ± 4 | 50 ± 6 | 0.006 | |
| LVEsd, mm | 33.6 ± 8 | 37 ± 7 | 33 ± 8 | 0.140 | |
| LVEDV, mL | 136 (113–160) | 149 ± 30 | 143 ± 348 | 0.265 | |
| LVESV, mL | 41.5 (28–61) | 56 (46–73) | 36 (26–56) | 0.062 | |
| LV stroke volume index, mL/m2 | 46 (42–53) | 44 (43–48) | 47 (41–55) | 0.452 | |
| LVEF, % | 69.6 (61–75) | 62 ± 12 | 68 ± 13 | 0.040 | |
| LVEF < 50% | 8 (10) | 8 (20) | 0 (0) | 0.089 | |
| LV mass index, g/m2 | 91.3 (76–119) | 125 ± 28 | 91 ± 32 | 0.001 | |
| RVEDV, mL | 120 (108–140) | 119 (103–136) | 120 (109–140) | 0.734 | |
| RVESV, mL | 44 (36–59) | 40 (30–52) | 45 (37–59) | 0.163 | |
| RVEF, % | 62.9 (57–67) | 64.7 (61–72) | 61.7 (57–66) | 0.119 | |
| Native T1, ms | 959.2 ± 34 | 971 ± 36 | 956 ± 33 | 0.156 | |
| Post-contrast T1, ms | 352 (327–363) | 353 (332–363) | 351 (327–361) | 0.533 | |
| LGE prevalence | 57 (74) | 34 (85) | 23 (62) | 0.175 | |
| ECV, % | 22.6 ± 3 | 23.4 ± 3 | 22.3 ± 3 | 0.292 | |
| ECV index, %/m2 | 12 (11–13) | 13 (12–13) | 12 (11–13) | 0.454 | |
| Histology data (n = 71) | |||||
| CVF, % | 12.4 (8–22) | 10.8 (7–17) | 15.9 (8–23) | 0.232 | |
| CVF subendocardial, % | 21.8 (10–35) | 17.4 (9–24) | 21.8 (12–38) | 0.108 | |
| Surgery | |||||
| Tissue valve | 71 (92) | 14 (100) | 57 (91) | 0.229 | |
| Mechanical valve | 6 (8) | 0 (0) | 6 (10) | 0.229 | |
| Aortic intervention | 2 (3) | 0 (0) | 2 (3) | 0.499 | |
| Serum biomarkers | |||||
| BNP, pg/L | 142 (67–362) | 409 (161–961) | 119 (66–245) | 0.032 | |
| Hs-Tn-I, pg/L | 10 (5–18) | 15 (13–29) | 9 (5–18) | 0.026 | |
| Creatinine, μmol/L | 76 ± 16 | 73 ± 17 | 76 ± 16 | 0.491 | |
| eGFR, mL/min/1.73 m2 | 85 (69–90) | 87 (71–90) | 85 (70–90) | 0.533 | |
| Echocardiography Data | Baseline (n = 14) | 3-Month Follow-Up (n = 14) | 12-Month Follow-Up (n = 14) | p-Value |
|---|---|---|---|---|
| AV area index, cm2/m2 | 0.4 ± 0.1 | 1.3 ± 0.3 | 1.2 ± 0.3 | <0.001 |
| Peak AV velocity, m/s | 5.3 ± 0.7 | 2.2 ± 0.4 | 2.3 ± 0.4 | <0.001 |
| Mean AV gradient, mm Hg | 65.3 (56.2–84.1) | 9.25 (8.0–10.8) | 10.4 (8.2–12.6) | <0.001 |
| IVSd, mm | 14.5 (14.0–15.0) | 11.5 (11.0–14.0) | 11.5 (11.0–12.0) | <0.001 |
| LVEdd, mm | 51.5 (49.0–54.5) | 50.5 (48.5–53.8) | 50.0 (46.5–52.0) | 0.699 |
| LVEsd, mm | 34.2 ± 5.7 | 33.8 ± 5.9 | 31.9 ± 3.6 | 0.131 |
| E/e’ septal | 17.6 (16.1–20.1) | 16.6 (11.4–18.2) | 15.6 (12.7–21.5) | 0.092 |
| E/e’ lateral | 14.7 (11.2–17.0) | 8.43 (6.56–10.8) | 8.88 (6.60–11.8) | 0.011 |
| E/e’ mean | 17.1 ± 7.2 | 11.7 ± 3.7 | 11.9 ± 4.5 | 0.004 |
| LA volume index, mL/m2 | 52.0 ± 13.4 | 45.4 ± 8.5 | 43.0 ± 10.8 | 0.009 |
| RV S’, cm/s | 12 (10–13) | 9 (8–10) | 9 (9–11) | 0.009 |
| GLS, % | 14.9 ± 3 | 18.3 ± 2 | 19.7 ± 2 | <0.001 |
| RAS strain pattern, n (%) | 14 (100) | 0 (0) | 0 (0) | – |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramanauskaitė, D.; Balčiūnaitė, G.; Palionis, D.; Besusparis, J.; Žurauskas, E.; Janušauskas, V.; Zorinas, A.; Valevičienė, N.; Sogaard, P.; Glaveckaitė, S. The Relative Apical Sparing Strain Pattern in Severe Aortic Valve Stenosis: A Marker of Adverse Cardiac Remodeling. J. Pers. Med. 2024, 14, 707. https://doi.org/10.3390/jpm14070707
Ramanauskaitė D, Balčiūnaitė G, Palionis D, Besusparis J, Žurauskas E, Janušauskas V, Zorinas A, Valevičienė N, Sogaard P, Glaveckaitė S. The Relative Apical Sparing Strain Pattern in Severe Aortic Valve Stenosis: A Marker of Adverse Cardiac Remodeling. Journal of Personalized Medicine. 2024; 14(7):707. https://doi.org/10.3390/jpm14070707
Chicago/Turabian StyleRamanauskaitė, Dovilė, Giedrė Balčiūnaitė, Darius Palionis, Justinas Besusparis, Edvardas Žurauskas, Vilius Janušauskas, Aleksejus Zorinas, Nomeda Valevičienė, Peter Sogaard, and Sigita Glaveckaitė. 2024. "The Relative Apical Sparing Strain Pattern in Severe Aortic Valve Stenosis: A Marker of Adverse Cardiac Remodeling" Journal of Personalized Medicine 14, no. 7: 707. https://doi.org/10.3390/jpm14070707
APA StyleRamanauskaitė, D., Balčiūnaitė, G., Palionis, D., Besusparis, J., Žurauskas, E., Janušauskas, V., Zorinas, A., Valevičienė, N., Sogaard, P., & Glaveckaitė, S. (2024). The Relative Apical Sparing Strain Pattern in Severe Aortic Valve Stenosis: A Marker of Adverse Cardiac Remodeling. Journal of Personalized Medicine, 14(7), 707. https://doi.org/10.3390/jpm14070707






