Oral Care in Head and Neck Radiotherapy: Proposal for an Oral Hygiene Protocol
Abstract
1. Head and Neck Cancer Epidemiology
2. Purpose of the Study
3. Materials and Methodology
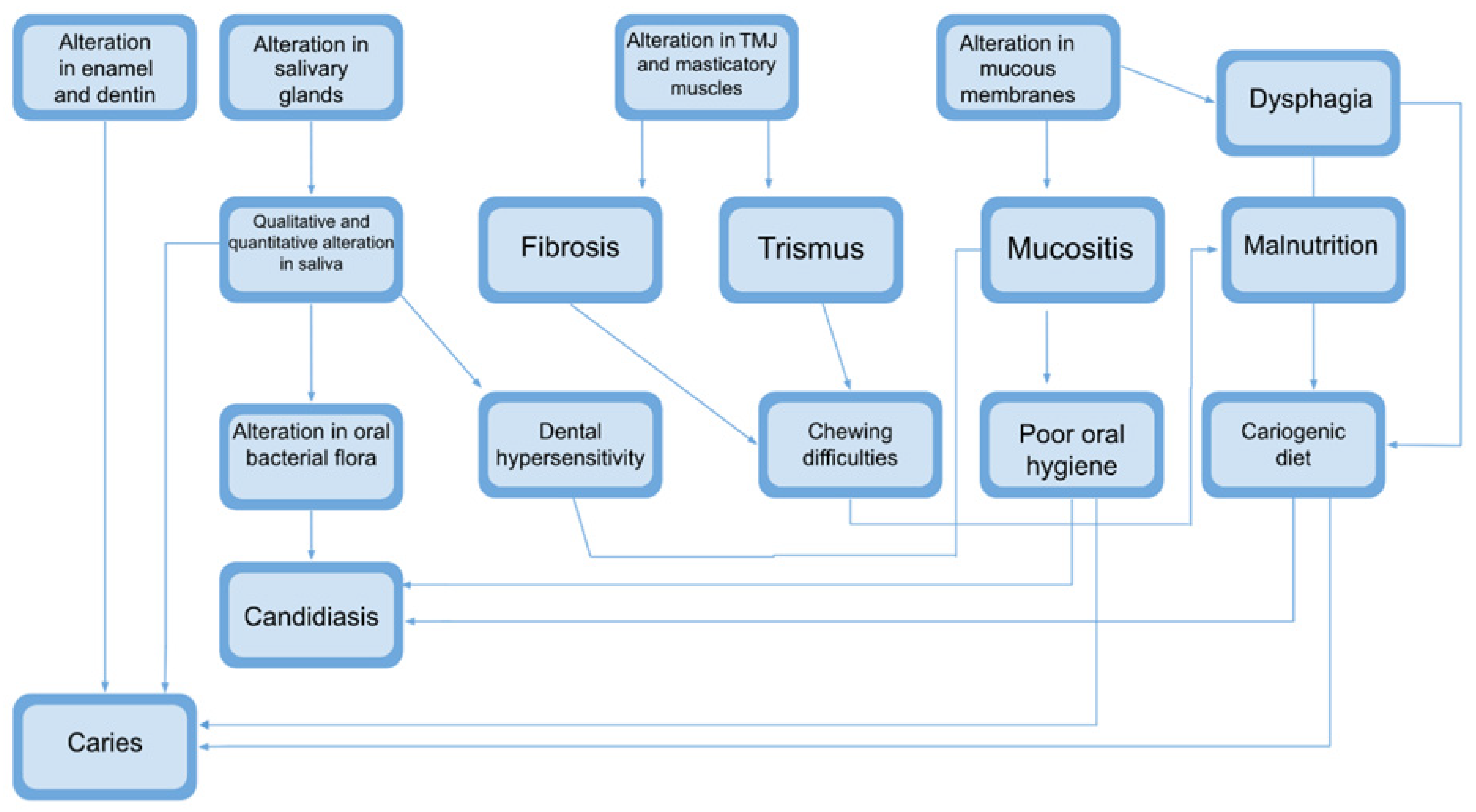
4. Results
5. Discussion
5.1. Oral Mucositis
5.2. Oral Candidiasis
- The expression of cell wall proteins (such as adhesins and secreted aspartate-proteinases) facilitates adhesion to epithelial cells and the extracellular matrix. This process is further enhanced by the hydrophobicity of the cell membrane, which enables the generation of exceptionally strong electrostatic forces [51];
- The expression of surface antigens similar to molecules of the human complement system allows the microorganism to camouflage itself and evade the host’s immune response, which is already weakened by the lysis of cytokines involved in the host’s cellular defense by aspartate proteinases secreted [52];
- The ability to transition from the blastoconidia phenotype to hyphae, which enhances its tissue invasion capacity and hinders its phagocytosis [53];
- Biofilm formation not only enhances surface adhesion but also provides protection and promotes the development of more virulent complex fungal communities, such as C. tropicalis and C. glabrata [54].
5.3. Xerostomia
5.4. Caries
5.5. Osteoradionecrosis
5.6. Fibrosis
- Laser photobiomodulation [86];
- Jaw mobilization devices, which are beneficial for expanding the interincisal opening and reducing complications associated with trismus [87];
- Pharmacological means, through the use of pentoxifylline and tocopherol, which, due to their anti-inflammatory and vasodilatory actions, are capable of enhancing the blood and oxygen supply to tissues, thereby reducing fibrogenesis. Additionally, botulinum toxin injections can prevent spasms of the masticatory muscles at the base of trismus [88].
5.7. Protocol for Dental Management of a Head and Neck Cancer Patient
5.8. Home Oral Hygiene and Recommended Devices
5.9. Professional Oral Hygiene Sessions
5.10. Strength and Limitations of the Present Study
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Mathers, C.; Parkin, D.M.; Piñeros, M.; Znaor, A.; Bray, F. Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int. J. Cancer. 2019, 144, 1941–1953. [Google Scholar] [CrossRef] [PubMed]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424, Erratum in: CA Cancer J. Clin. 2020, 70, 313. [Google Scholar] [CrossRef]
- Windon, M.J.; D’Souza, G.; Rettig, E.M.; Westra, W.H.; van Zante, A.; Wang, S.J.; Ryan, W.R.; Mydlarz, W.K.; Ha, P.K.; Miles, B.A.; et al. Increasing prevalence of human papillomavirus-positive oropharyngeal cancers among older adults. Cancer 2018, 124, 2993–2999. [Google Scholar] [CrossRef] [PubMed]
- Del Mistro, A.; Frayle, H.; Menegaldo, A.; Favaretto, N.; Gori, S.; Nicolai, P.; Spinato, G.; Romeo, S.; Tirelli, G.; da Mosto, M.C.; et al. Age-independent increasing prevalence of human papillomavirus-driven oropharyngeal carcinomas in North-East Italy. Sci. Rep. 2020, 10, 9320. [Google Scholar] [CrossRef]
- Baboci, L.; Boscolo-Rizzo, P.; Holzinger, D.; Bertorelle, R.; Biasini, L.; Michel, A.; Schmitt, M.; Spinato, G.; Bussani, R.; Alemany, L.; et al. Evidence of the causal role of human papillomavirus type 58 in an oropharyngeal carcinoma. Virol. J. 2013, 10, 334. [Google Scholar] [CrossRef]
- Fung, S.Y.; Lam, J.W.; Chan, K.C. Clinical utility of circulating Epstein-Barr virus DNA analysis for the management of nasopharyngeal carcinoma. Chin. Clin. Oncol. 2016, 5, 18. [Google Scholar] [CrossRef]
- Chen, A.A.; Gheit, T.; Stellin, M.; Lupato, V.; Spinato, G.; Fuson, R.; Menegaldo, A.; Mckay-Chopin, S.; Dal Cin, E.; Tirelli, G.; et al. Oncogenic DNA viruses found in salivary gland tumors. Oral Oncol. 2017, 75, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Donà, S.; Borsetto, D.; Fussey, J.; Biscaro, V.; Vian, E.; Spinato, G.; Menegaldo, A.; Da Mosto, M.C.; Rigoli, R.; Polesel, J.; et al. Association between hepatitis C and B viruses and head and neck squamous cell carcinoma. J. Clin. Virol. 2019, 121, 104209. [Google Scholar] [CrossRef] [PubMed]
- Pulte, D.; Brenner, H. Changes in survival in head and neck cancers in the late 20th and early 21st century: A period analysis. Oncologist 2010, 15, 994–1001. [Google Scholar] [CrossRef]
- Topchu, I.; Bychkov, I.; Roshchina, E.; Makhov, P.; Boumber, Y. PIP4K2B Protein Regulation by NSD1 in HPV-Negative Head and Neck Squamous Cell Carcinoma. Cancers 2024, 16, 1180. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Osazuwa-Peters, N.; Simpson, M.C.; Zhao, L.; Boakye, E.A.; Olomukoro, S.I.; Deshields, T.; Loux, T.M.; Varvares, M.A.; Schootman, M. Suicide risk among cancer survivors: Head and neck versus other cancers. Cancer 2018, 124, 4072–4079. [Google Scholar] [CrossRef] [PubMed]
- Spinato, G.; Stellin, M.; Azzarello GBonazza, D.; Zanconati, F.; Politi, D.; Cocuzza, S.; Di Mauro, P.; Ausoni, S.; Tonoli, G.; Costantini, G.; et al. Multicenter research into the quality of life of patients with advanced oropharyngeal carcinoma with long-term survival associated with human papilloma virus. Oncol. Lett. 2017, 14, 185–193. [Google Scholar] [CrossRef] [PubMed] [PubMed Central][Green Version]
- Tirelli, G.; Boscolo Nata, F.; Gatto, A.; Bussani, R.; Spinato, G.; Zacchigna, S.; Piovesana, M. Intraoperative margin control in transoral approach for oral and oropharyngeal cancer. Laryngoscope 2019, 129, 1810–1815. [Google Scholar] [CrossRef] [PubMed]
- Cegolon, L.; Mastrangelo, G.; Emanuelli, E.; Camerotto, R.; Spinato, G.; Frezza, D. Early Negativization of SARS-CoV-2 infection by nasal spray of seawater plus additives: The Renaissance open-label controlled clinical trial. Pharmaceutics 2022, 14, 2502. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Nagy, K.N.; Sonkodi, I.; Szöke, I.; Nagy, E.; Newman, H.N. The microflora associated with human oral carcinomas. Oral Oncol. 1998, 34, 304–308. [Google Scholar] [CrossRef]
- Kurkivuori, J.; Salaspuro, V.; Kaihovaara, P.; Kari, K.; Rautemaa, R.; Grönroos, L.; Meurman, J.H.; Salaspuro, M. Acetaldehyde production from ethanol by oral streptococci. Oral Oncol. 2007, 43, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Anjali, K.; Arun, A.B.; Bastian, T.S.; Parthiban, R.; Selvamani, M.; Adarsh, H. Oral microbial profile in oral cancer patients before and after radiation therapy in a cancer care center—A prospective study. J. Oral Maxillofac. Pathol. 2020, 24, 117–124. [Google Scholar] [PubMed]
- Hong, B.-Y.; Sobue, T.; Choquette, L.; Dupuy, A.K.; Thompson, A.; Burleson, J.A.; Salner, A.L.; Schauer, P.K.; Joshi, P.; Fox, E.; et al. Chemotherapy-induced oral mucositis is associated with detrimental bacterial dysbiosis. Microbiome 2019, 7, 66. [Google Scholar] [CrossRef]
- Laheij, A.M.; de Soet, J.J. Can the oral microflora affect oral ulcerative mucositis? Curr. Opin. Support. Palliat. Care 2014, 8, 180–187. [Google Scholar] [CrossRef]
- Ludovichetti, F.S.; Zuccon, A.; Positello, P.; Zerman, N.; Gracco, A.; Stellini, E.; Mazzoleni, S. Preventive oral hygiene and ventilator-associated pneumonia in paediatric intensive care unit. Eur. J. Paediatr. Dent. 2022, 23, 298–302. [Google Scholar]
- Reyes-Gibby, C.C.; Wang, J.; Zhang, L.; Peterson, C.B.; Do, K.-A.; Jenq, R.R.; Shelburne, S.; Shah, D.P.; Chambers, M.S.; Hanna, E.Y.; et al. Oral microbiome and onset of oral mucositis in patients with squamous cell carcinoma of the head and neck. Cancer 2020, 126, 5124–5136. [Google Scholar] [CrossRef] [PubMed]
- Sonis, S.T. The pathobiology of mucositis. Nat. Rev. Cancer 2004, 4, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Tong, H.C.; Gao, X.J.; Dong, X.Z. Non-mutans streptococci in patients receiving radiotherapy in the head and neck area. Caries Res. 2003, 37, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Ausoni, S.; Boscolo-Rizzo, P.; Singh, B.; Da Mosto, M.C.; Spinato, G.; Tirelli, G.; Spinato, R.; Azzarello, G. Targeting cellular and molecular drivers of head and neck squamous cell carcinoma: Current options and emerging perspectives. Cancer Metastasis Rev. 2016, 35, 413–426. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hongsheng, Y. Different responses of tumor and normal cells to low-dose radiation. Contemp. Oncol. 2013, 17, 356–362. [Google Scholar]
- Kumagai, T.; Rahman, F.; Smith, A.M. The microbiome and radiation induced bowel injury: Evidence for potential mechanistic role in disease pathogenesis. Nutrients 2018, 10, 1405. [Google Scholar] [CrossRef]
- Boscolo-Rizzo, P.; Rampazzo, E.; Perissinotto, E.; Piano, M.A.; Giunco, S.; Baboci, L.; Spinato, G.; Spinato, R.; Tirelli, G.; Da Mosto, M.C.; et al. Telomere shortening in mucosa surrounding the tumor: Biosensor of field cancerization and prognostic marker of mucosal failure in head and neck squamous cell carcinoma. Oral Oncol. 2015, 51, 500–507. [Google Scholar] [CrossRef] [PubMed]
- Squier, C.A.; Kremer, M.J. Biology of oral mucosa and esophagus. J. Natl. Cancer Inst. Monogr. 2001, 29, 7–15. [Google Scholar] [CrossRef]
- Daugėlaitė, G.; Užkuraitytė, K.; Jagelavičienė, E.; Filipauskas, A. Prevention and treatment of chemotherapy and radiotherapy induced oral mucositis. Medicina 2019, 55, 25. [Google Scholar] [CrossRef]
- Nicolatou-Galitis, O.; Bossi, P.; Orlandi, E.; Bensadoun, R.-J. The role of Benzydamine in prevention and treatment of chemoradiotherapy-induced mucositis. Support. Care Cancer 2021, 29, 5701–5709. [Google Scholar] [CrossRef]
- Elad, S.; Cheng, K.K.; Lalla, R.V.; Yarom, N.; Hong, C.; Logan, R.M.; Bowen, J.; Gibson, R.; Saunders, D.P.; Zadik, Y.; et al. MASCC/ISOO clinical practice guidelines for the management of mucositis secondary to cancer therapy. Cancer 2020, 126, 4423–4431, Erratum in: Cancer 2021, 127, 3700. [Google Scholar] [CrossRef] [PubMed]
- Naidu, M.U.; Ramana, G.V.; Ratnam, S.V.; Sudhavani, T.; Naidu, K.J.; Roy, P.; Suresh, P.; Rani, P.U.; Mohan, I.K. A randomised, double-blind, parallel, placebo-controlled study to evaluate the efficacy of MF 5232 (Mucotrol), a concentrated oral gel wafer, in the treatment of oral mucositis. Drugs R D 2005, 6, 291–298. [Google Scholar] [CrossRef]
- Boscolo-Rizzo, P.; Tirelli, G.; Mantovani, M.; Baggio, V.; Lupato, V.; Spinato, G.; Gava, A.; Da Mosto, M.C. Non-endemic locoregionally advanced nasopharyngeal carcinoma: Long-term outcome after induction plus concurrent chemoradiotherapy in everyday clinical practice. Eur. Arch. Otorhinolaryngol. 2015, 272, 3491–3498. [Google Scholar] [CrossRef] [PubMed]
- Saunders, D.P.; Rouleau, T.; Cheng, K.; Yarom, N.; Kandwal, A.; Joy, J.; Bektas Kayhan, K.; van de Wetering, M.; Brito-Dellan, N.; Kataoka, T.; et al. Systematic review of antimicrobials, mucosal coating agents, anesthetics, and analgesics for the management of oral mucositis in cancer patients and clinical practice guidelines. Support. Care Cancer 2020, 28, 2473–2484. [Google Scholar] [CrossRef]
- Schubert, M.M.; Eduardo, F.P.; Guthrie, K.A.; Franquin, J.-C.; Bensadoun, R.-J.; Migliorati, C.A.; Lloid, C.M.; Eduardo, C.P.; Walter, N.-F.; Marques, M.M.; et al. A phase III randomized double-blind placebo-controlled clinical trial to determine the efficacy of low level laser therapy for the prevention of oral mucositis in patients undergoing hematopoietic cell transplantation. Support. Care Cancer 2007, 15, 1145–1154. [Google Scholar] [CrossRef]
- Hahm, E.; Kulhari, S.; Arany, P. Targeting the pain, inflammation and immune (P11) axis: Plausible rationale for PBM. Photon. Lasers Med. 2012, 1, 241–254. [Google Scholar] [CrossRef]
- Johansson, J.-E.; Bratel, J.; Hardling, M.; Heikki, L.; Mellqvist, U.-H.; Hasséus, B. Cryotherapy as prophylaxis against oral mucositis after high-dose melphalan and autologous stem cell transplantation for myeloma: A randomised, open-label, Phase 3, non-inferiority trial. Bone Marrow Transplant. 2019, 54, 1482–1488. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, K.M. The effect of olive leaf extract in decreasing the expression of two pro-inflammatory cytokines in patients receiving chemotherapy for cancer. A randomized clinical trial. Saudi Dent. J. 2013, 25, 141–147. [Google Scholar] [CrossRef]
- Yarom, N.; Hovan, A.; Bossi, P.; Ariyawardana, A.; Jensen, S.B.; Gobbo, M.; Saca-Hazboun, H.; Kandwal, A.; Majorana, A.; Ottaviani, G.; et al. Systematic review of natural and miscellaneous agents for the management of oral mucositis in cancer patients and clinical practice guidelines—Part 1: Vitamins, minerals, and nutritional supplements. Support. Care Cancer 2019, 27, 3997–4010. [Google Scholar] [CrossRef]
- Yarom, N.; Ariyawardana, A.; Hovan, A.; Barasch, A.; Jarvis, V.; Jensen, S.B.; Zadik, Y.; Elad, S.; Bowen, J.; Lalla, R.V.; et al. Systematic review of natural agents for the management of oral mucositis in cancer patients. Support. Care Cancer 2013, 21, 3209–3221. [Google Scholar] [CrossRef]
- Fidler, P.; Loprinzi, C.L.; O’Fallon, J.R.; Leitch, J.M.; Lee, J.K.; Hayes, D.L.; Novotny, P.; Clemens-Schutjer, D.; Bartel, J.; Michalak, J.C. Prospective evaluation of a chamomile mouthwash for prevention of 5-FU-induced oral mucositis. Cancer 1996, 77, 522–525. [Google Scholar] [CrossRef]
- You, W.C.; Hsieh, C.C.; Huang, J.T. Effect of extracts from indigowood root (Isatis indigotica Fort.) on immune responses in radiation-induced mucositis. J. Altern. Complement. Med. 2009, 15, 771–778. [Google Scholar] [CrossRef] [PubMed]
- Babaee, N.; Moslemi, D.; Khalilpour, M.; Vejdani, F.; Moghadamnia, Y.; Bijani, A.; Baradaran, M.; Kazemi, M.T.; Khalilpour, A.; Pouramir, M.; et al. Antioxidant capacity of calendula officinalis flowers extract and prevention of radiation induced oro- pharyngeal mucositis in patients with head and neck cancers: A randomized controlled clinical study. DARU 2009, 21, 18. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Wang, P.; Ouyang, H.; Wang, J.; Sun, L.; Li, Y.; Liu, D.; Jiang, Z.; Wang, B.; Pan, Z. Efficacy of traditional Chinese medicine in treatment and prophylaxis of radiation-induced oral mucositis in patients receiving radiotherapy: A randomized controlled trial. Integr. Cancer Ther. 2018, 17, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.H.; Yuan, B.; Liu, Q.F.; Wang, Q. Study of qingre liyan decoction in treating and preventing acute radioactive oral mucositis. Chin. J. Integr. Med. 2007, 13, 280–284. [Google Scholar] [CrossRef]
- Rezaeipour, N.; Jafari, F.; Rezaeizadeh, H.; Nasseri, M.; Kamalinejad, M.; Ghobadi, A.; Shamsipour, M.; Zargaran, A.; Ameri, A. Efficacy of a Persian medicine herbal compound (Alcea digitata Alef and Malva sylvestris L.) on prevention of radiation induced acute mucositis in patients with head and neck cancer: A pilot study. Int. J. Cancer Manag. 2017, 10, 5812. [Google Scholar] [CrossRef]
- Lalla, R.V.; Bowen, J.; Barasch, A.; Elting, L.; Epstein, J.; Keefe, D.M.; McGuire, D.B.; Migliorati, C.; Nicolatou-Galitis, O.; Peterson, D.E.; et al. MASCC/ISOO clinical practice guidelines for the management of mucositis secondary to cancer therapy. Cancer 2014, 120, 1453–1461. [Google Scholar] [CrossRef]
- Boscolo-Rizzo, P.; D’Alessandro, A.; Polesel, J.; Borsetto, D.; Tofanelli, M.; Deganello, A.; Tomasoni, M.; Nicolai, P.; Bossi, P.; Spinato, G.; et al. Different inflammatory blood markers correlate with specific outcomes in incident HPV-negative head and neck squamous cell carcinoma: A retrospective cohort study. BMC Cancer 2022, 22, 243. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ellepola, A.N.; Samaranayake, L.P. Oral candidal infections and antimycotics. Crit. Rev. Oral Biol. Med. 2000, 11, 172–198. [Google Scholar] [CrossRef]
- McCullough, M.J.; Ross, B.C.; Reade, P.C. Candida albicans: A review of its history, taxonomy, epidemiology, virulence attributes, and methods of strain differentiation. Int. J. Oral Maxillofac. Surg. 1996, 25, 136–144. [Google Scholar] [CrossRef]
- Gow, N.A.; Brown, A.J.; Odds, F.C. Fungal morphogenesis and host invasion. Curr. Opin. Microbiol. 2002, 5, 366–371. [Google Scholar] [CrossRef] [PubMed]
- Pizzo, G.; Giuliana, G.; Milici, M.E.; Giangreco, R. Effect of dietary carbohydrates on the in vitro epithelial adhesion of Candida albicans, Candida tropicalis, and Candida krusei. New Microbiol. 2000, 23, 63–71. [Google Scholar]
- Jayatilake, J.A.M.S.; Samaranayake, Y.H.; Cheung, L.K.; Samaranayake, L.P. Quantitative evaluation of tissue invasion by wild type, hyphal and SAP mutants of Candida albicans, and non-albicans Candida species in reconstituted human oral epithelium. J. Oral Pathol. Med. 2006, 35, 484–491. [Google Scholar] [CrossRef]
- Silva, S.; Henriques, M.; Martins, A.; Oliveira, R.; Williams, D.; Azeredo, J. Biofilms of non-Candida albicans Candida species: Quantification, structure and matrix composition. Med. Mycol. 2009, 47, 681–689. [Google Scholar] [CrossRef] [PubMed]
- MacNeill, S.; Rindler, E.; Walker, A.; Brown, A.R.; Cobb, C.M. Effects of tetracycline hydrochloride and chlorhexidine gluconate on Candida albicans. An in vitro study. J. Clin. Periodontol. 1997, 24, 753–760. [Google Scholar] [CrossRef] [PubMed]
- Budtz-Jörgensen, E. Etiology, pathogenesis, therapy, and prophylaxis of oral yeast infections. Acta Odontol. Scand. 1990, 48, 61–69. [Google Scholar] [CrossRef]
- Lan, X.; Chan, J.Y.; Pu, J.J.; Qiao, W.; Pang, S.; Yang, W.-F.; Wong, K.C.; Kwong, D.L.; Su, Y.-X. Saliva electrolyte analysis and xerostomia-related quality of life in nasopharyngeal carcinoma patients following intensity-modulated radiation therapy. Radiother. Oncol. 2020, 150, 97–103. [Google Scholar] [CrossRef]
- Chaudhury, N.M.; Proctor, G.B.; Karlsson, N.G.; Carpenter, G.H.; Flowers, S.A. Reduced Mucin-7 (Muc7) sialylation and altered saliva rheology in Sjögren’s syndrome associated oral dryness. Mol. Cell Proteom. 2016, 15, 1048–1059. [Google Scholar] [CrossRef]
- Kielbassa, A.M.; Munz, I.; Bruggmoser, G.; Schulte-Mönting, J. Effect of demineralization and remineralization on microhardness of irradiated dentin. J. Clin. Dent. 2002, 13, 104–110. [Google Scholar]
- Moore, C.; McLister CMc Cardwell, C.; O’Neill, C.; Donnelly, M.; McKenna, G. Dental caries following radiotherapy for head and neck cancer: A systematic review. Oral Oncol. 2020, 100, 104484. [Google Scholar] [CrossRef]
- Kim, J.H.; Ahn, H.J.; Choi, J.H.; Jung, D.W.; Kwon, J.S. Effect of 0.1% pilocarpine mouthwash on xerostomia: Double-blind, randomised controlled trial. J. Oral Rehabil. 2014, 41, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Gomez, D.R.; Zhung, J.E.; Gomez, J.; Chan, K.; Wu, A.J.; Wolden, S.L.; Pfister, D.G.; Shaha, A.; Shah, J.P.; Kraus, D.H.; et al. Intensity-modulated radiotherapy in postoperative treatment of oral cavity cancers. Int. J. Radiat. Oncol. Biol. Phys. 2009, 73, 1096–1103. [Google Scholar] [CrossRef] [PubMed]
- Hey, J.; Seidel, J.; Schweyen, R.; Paelecke-Habermann, Y.; Vordermark, D.; Gernhardt, C.; Kuhnt, T. The influence of parotid gland sparing on radiation damages of dental hard tissues. Clin. Oral Investig. 2013, 17, 1619–1625. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-A.; Wang, C.-C.; Wong, Y.-K.; Wang, C.-P.; Jiang, R.-S.; Lin, J.-C.; Chen, C.-C.; Liu, S.-A. Osteoradionecrosis of mandible bone in patients with oral cancer--associated factors and treatment outcomes. Head Neck 2016, 38, 762–768. [Google Scholar] [CrossRef]
- Kubota, H.; Miyawaki, D.; Mukumoto, N.; Ishihara, T.; Matsumura, M.; Hasegawa, T.; Akashi, M.; Kiyota, N.; Shinomiya, H.; Teshima, M.; et al. Risk factors for osteoradionecrosis of the jaw in patients with head and neck squamous cell carcinoma. Radiat. Oncol. 2021, 16, 1. [Google Scholar] [CrossRef]
- Aarup-Kristensen, S.; Hansen, C.R.; Forner, L.; Brink, C.; Eriksen, J.G.; Johansen, J. Osteoradionecrosis of the mandible after radiotherapy for head and neck cancer: Risk factors and dose-volume correlations. Acta Oncol. 2019, 58, 1373–1377. [Google Scholar] [CrossRef]
- Lee, I.J.; Koom, W.S.; Lee, C.G.; Kim, Y.B.; Yoo, S.W.; Keum, K.C.; Kim, G.E.; Choi, E.C.; Cha, I.H. Risk factors and dose-effect relationship for mandibular osteoradionecrosis in oral and oropharyngeal cancer patients. Int. J. Radiat. Oncol. Biol. Phys. 2009, 75, 1084–1091. [Google Scholar] [CrossRef]
- Huang, T.-L.; Chien, C.-Y.; Tsai, W.-L.; Liao, K.-C.; Chou, S.-Y.; Lin, H.-C.; Dean Luo, S.; Lee, T.-F.; Lee, C.-H.; Fang, F.-M. Long-term late toxicities and quality of life for survivors of nasopharyngeal carcinoma treated with intensity-modulated radiotherapy versus non-intensity-modulated radiotherapy. Head Neck 2016, 38 (Suppl. S1), E1026–E1032. [Google Scholar] [CrossRef]
- Nabil, S.; Samman, N. Risk factors for osteoradionecrosis after head and neck radiation: A systematic review. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2012, 113, 54–69. [Google Scholar] [CrossRef]
- Lazim, N.M.; Spinato, G.; Abdullah, K.; Boscolo-Rizzo, P. Survival rates and quality of life of nasopharyngeal carcinoma patients. In An Evidence-Based Approach to the Management of Nasopharyngeal Cancer; Abdullah, B., Balasubramanian, A., Lazim, N.M., Eds.; Academic Press: Cambridge, MA, USA, 2020; pp. 255–276. ISBN 9780128144039. [Google Scholar] [CrossRef]
- Marx, R.E. A new concept in the treatment of osteoradionecrosis. J. Oral Maxillofac. Surg. 1983, 41, 351–357. [Google Scholar] [CrossRef]
- Campisia, G.; Bedognib, A.; Di Fedea, O.; Vescovic, P.; Fuscod, V.; Lo Muzioe, L. Osteonecrosis of the jaw related to bisphosphonates, denosumab and anti-angiogenics in cancer and osteoporotic patients: Diagnosis and management. Dent. Cadmos 2013, 81, 566–589. [Google Scholar]
- Delanian, S.; Depondt, J.; Lefaix, J.-L. Major healing of refractory mandible osteoradionecrosis after treatment combining pentoxifylline and tocopherol: A phase II trial. Head Neck 2005, 27, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Lyons, A.J.; Brennan, P.A. Pentoxifylline—A review of its use in osteoradionecrosis. Br. J. Oral Maxillofac. Surg. 2017, 55, 230–234. [Google Scholar] [CrossRef] [PubMed]
- Da Mosto, M.C.; Lupato, V.; Romeo, S.; Spinato, G.; Addonisio, G.; Baggio, V.; Gava, A.; Boscolo-Rizzo, P. Is neck dissection necessary after induction plus concurrent chemoradiotherapy in complete responder head and neck cancer patients with pretherapy advanced nodal disease? Ann. Surg. Oncol. 2013, 20, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Straub, J.M.; New, J.; Hamilton, C.D.; Lominska, C.; Shnayder, Y.; Thomas, S.M. Radiation-induced fibrosis: Mechanisms and implications for therapy. J. Cancer Res. Clin. Oncol. 2015, 141, 1985–1994. [Google Scholar] [CrossRef]
- Mat Lazim, N.; Kandhro, A.H.; Menegaldo, A.; Spinato, G.; Verro, B.; Abdullah, B. Autofluorescence image-guided endoscopy in the management of upper aerodigestive tract tumors. Int. J. Environ. Res. Public Health 2022, 20, 159. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Baudelet, M.; Van den Steen, L.; Tomassen, P.; Bonte, K.; Deron, P.; Huvenne, W.; Rottey, S.; De Neve, W.; Sundahl, N.; Van Nuffelen, G.; et al. Very late xerostomia, dysphagia, and neck fibrosis after head and neck radiotherapy. Head Neck 2019, 41, 3594–3603. [Google Scholar] [CrossRef]
- Machtay, M.; Moughan, J.; Trotti, A.; Garden, A.S.; Weber, R.S.; Cooper, J.S.; Forastiere, A.; Ang, K.K. Factors associated with severe late toxicity after concurrent chemoradiation for locally advanced head and neck cancer: An RTOG analysis. J. Clin. Oncol. 2008, 26, 3582–3589. [Google Scholar] [CrossRef] [PubMed]
- Hirota, S.; Tsujino, K.; Oshitani, T.; Hishikawa, Y.; Takada, Y.; Kono, M.; Abe, M. Subcutaneous fibrosis after whole neck irradiation. Int. J. Radiat. Oncol. Biol. Phys. 2002, 52, 937–943. [Google Scholar] [CrossRef]
- Dijkstra, P.U.; Huisman, P.M.; Roodenburg, J.L. Criteria for trismus in head and neck oncology. Int. J. Oral Maxillofac. Surg. 2006, 35, 337–342. [Google Scholar] [CrossRef]
- Nagaraja, S.; Kadam, S.A.; Selvaraj, K.; Ahmed, I.; Javarappa, R. Trismus in head and neck cancer patients treated by telecobalt and effect of early rehabilitation measures. J. Cancer Res. Ther. 2016, 12, 685–688. [Google Scholar] [PubMed]
- Lee, L.-Y.; Chen, S.-C.; Chen, W.-C.; Huang, B.-S.; Lin, C.-Y. Postradiation trismus and its impact on quality of life in patients with head and neck cancer. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2015, 119, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Gebre-Medhin, M.; Haghanegi, M.; Robért, L.; Kjellén, E.; Nilsson, P. Dose-volume analysis of radiation-induced trismus in head and neck cancer patients. Acta Oncol. 2016, 55, 1313–1317. [Google Scholar] [CrossRef] [PubMed]
- Shao, C.-H.; Chiang, C.-C.; Huang, T.-W. Exercise therapy for cancer treatment-induced trismus in patients with head and neck cancer: A systematic review and meta-analysis of randomized controlled trials. Radiother. Oncol. 2020, 151, 249–255. [Google Scholar] [CrossRef]
- Zecha, J.A.; Raber-Durlacher, J.E.; Nair RGEpstein, J.B.; Elad, S.; Hamblin, M.R.; Barasch, A.; Migliorati, C.A.; Milstein, D.M.; Genot, M.T.; Lansaat, L. Low-level laser therapy/photobiomodulation in the management of side effects of chemoradiation therapy in head and neck cancer: Part 2: Proposed applications and treatment protocols. Support. Care Cancer 2016, 24, 2793–2805. [Google Scholar] [CrossRef]
- Bensadoun, R.-J.; Riesenbeck, D.; Lockhart, P.B.; Elting, L.S.; Spijkervet, F.K.; Brennan, M.T.; Trismus Section, Oral Care Study Group, Multinational Association for Supportive Care in Cancer (MASCC)/International Society of Oral Oncology (ISOO). A systematic review of trismus induced by cancer therapies in head and neck cancer patients. Support. Care Cancer 2010, 18, 1033–1038. [Google Scholar] [CrossRef]
- Hartl, D.M.; Cohen, M.; Juliéron, M.; Marandas, P.; Janot, F.; Bourhis, J. Botulinum toxin for radiation-induced facial pain and trismus. Otolaryngol. Head Neck Surg. 2008, 138, 459–463. [Google Scholar]
- Peterson, D.E.; Doerr, W.; Hovan, A.; Pinto, A.; Saunders, D.; Elting, L.S.; Spijkervet, F.K.; Brennan, M.T. Osteoradionecrosis in cancer patients: The evidence base for treatment-dependent frequency, current management strategies, and future studies. Support. Care Cancer 2010, 18, 1089–1098. [Google Scholar] [CrossRef]
- Sulistiyani, E.; Brimson, J.M.; Chansaenroj, A.; Sariya, L.; Urkasemsin, G.; Oonsiri, S.; Tencomnao, T.; Vacharaksa, A.; Chaisuparat, R.; Ferreira, J.N. Epigallocatechin-3-gallate protects pro-acinar epithelia against salivary gland radiation injury. Int. J. Mol. Sci. 2021, 22, 3162. [Google Scholar] [CrossRef]

| Acute Complications | Late Complications |
|---|---|
| Oral mucositis | Osteoradionecrosis |
| Disruption of the oral microbiome | Caries |
| Candidiasis | Skin fibrosis |
| Xerostomia | Muscles fibrosis |
| Dysphagia | Joint fibrosis |
| Dysgeusia |
| Toothpaste | Mouthwash | Drugs | Other | |
|---|---|---|---|---|
| From 15 days before of the start of treatment radiotherapy | Fluoridated, SLS-free | Fluorinated, fortified with CPC and chlorhexidine | Mousse with fluoride (2 times/day) | |
| Onset of mucositis | Fluorinated, SLS-free, fortified with chlorhexidine and CPC | Added with Benzidamine (1–2 times/day) Karyosite 2 times/day | ||
| Onset of candidiasis | Fluorinated, SLS-free, fortified with chlorhexidine and CPC | Fluorinated, with chlorhexidine (1 time/day, away from Fluconazole) | Fluconazole suspension 50 mg/5 mL (2 times/day) | |
| Onset of candidiasis (particularly particularly if HCC) | Fluorinated, SLS-free, NOT fortified with chlorhexidine | Fluorinated, NOT fortified with chlorhexidine | Fluorinated, NOT fortified with chlorhexidine Nystatin suspension 100,000 I.U. | |
| Onset of xerostomia | Fluorinated, SLS-free, fortified with chlorhexidine and CPC | Fluorinated, fortified with chlorhexidine (3 times/day) | Substitute for saliva gel or spray with hyaluronan (3–4 times/day) | |
| After the end of the treatment radiotherapy | Fluorinated, SLS-free | Fluorinated, fortified with CPC Karyosite 2 times/day | Mousse with fluoride (2 times/week) |
| Ablation Tartar | Fluoroprophylaxis | Measurement Aperture Maximum Mouth | Rationale and Instructions IOD | |
|---|---|---|---|---|
| 15 days before of the start of the treatment | YES | YES | YES | YES |
| 7 days before of the start of the tratment | As needed | If patient has not used fluorinated mousse | NO | As needed |
| 1 time per week until the end of treatment RT | As needed | If patient has not used fluorinated mousse | YES | As needed |
| 1 months from the end of the treatment RT (1st follow-up) | YES | YES | YES | As needed |
| 2 months from the end of the treatment RT (2nd follow-up) | YES | YES | YES | As needed |
| 3 months from the end of the treatment RT (3rd follow-up) | YES | YES | YES | As needed |
| Recalls of hygiene professional every 3–4 months | YES | NO | YES | As needed |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spinato, G.; Schiavon, V.; Torvilli, S.; Carraro, S.; Amato, F.; Daloiso, A.; Di Fiore, A.; Favero, V.; Franz, L.; Marioni, G.; et al. Oral Care in Head and Neck Radiotherapy: Proposal for an Oral Hygiene Protocol. J. Pers. Med. 2024, 14, 1013. https://doi.org/10.3390/jpm14091013
Spinato G, Schiavon V, Torvilli S, Carraro S, Amato F, Daloiso A, Di Fiore A, Favero V, Franz L, Marioni G, et al. Oral Care in Head and Neck Radiotherapy: Proposal for an Oral Hygiene Protocol. Journal of Personalized Medicine. 2024; 14(9):1013. https://doi.org/10.3390/jpm14091013
Chicago/Turabian StyleSpinato, Giacomo, Valentina Schiavon, Sara Torvilli, Stefano Carraro, Federica Amato, Antonio Daloiso, Adolfo Di Fiore, Vittorio Favero, Leonardo Franz, Gino Marioni, and et al. 2024. "Oral Care in Head and Neck Radiotherapy: Proposal for an Oral Hygiene Protocol" Journal of Personalized Medicine 14, no. 9: 1013. https://doi.org/10.3390/jpm14091013
APA StyleSpinato, G., Schiavon, V., Torvilli, S., Carraro, S., Amato, F., Daloiso, A., Di Fiore, A., Favero, V., Franz, L., Marioni, G., de Filippis, C., Fabbris, C., Emanuelli, E., & Nicolai, P. (2024). Oral Care in Head and Neck Radiotherapy: Proposal for an Oral Hygiene Protocol. Journal of Personalized Medicine, 14(9), 1013. https://doi.org/10.3390/jpm14091013











