1. Introduction
The development of the ocean and marine space is becoming an increasingly urgent task every year due to the presence of rich natural resources. Parts and equipment of marine and coastal infrastructure, partially or completely submerged in sea water, are exposed to an aggressive environment, which leads to their premature failure [
1,
2]. The synergistic effect of simultaneous mechanical wear and electrochemical or chemical corrosion (tribocorrosion) in most cases leads to even more serious damage due to the removal of the passivation film and increased corrosive attack [
3,
4]. Metallic materials are also subjected to the negative effects of the microbiological environment. When they are at rest, bacteria and microorganisms actively develop on their surfaces, which leads to further microbiological corrosion (MBC) [
5,
6].
Shipbuilding equipment such as propellers, hydraulic turbine blades, shafts, ship winches and parts of water and centrifugal pumps are made of high-strength hardened steels, such as AISI 420S [
7]. This material is characterized by high mechanical properties and moderate corrosion resistance [
8]. However, the critical components of such equipment are subjected to intensive wear [
9], which leads to the removal of the protective oxide film on the surface and an increase in the corrosion rate [
10,
11]. To minimize wear under tribocorrosion conditions, the material must possess high wear and corrosion resistance. One of the effective ways to increase wear and corrosion resistance, as well as impart antibacterial activity to materials, is the deposition of coatings [
12,
13,
14,
15,
16].
Chromium (Cr) is introduced into coatings to increase corrosion resistance and minimize pitting corrosion due to the formation of stable Cr
2O
3 oxide on the surface [
17]. Molybdenum (Mo) stabilizes and accelerates passive film formation and increases pitting resistance [
18,
19]. In a solution containing chlorine ions (Cl
−), Mo reduces the adsorption of these ions on the passive film due to the formation of Mo
6+ oxide, which reduces the penetration of Cl
− ions. The addition of Ag or Cu to the coating is one of the effective ways to impart antibacterial properties to the surface and, as a result, protect metal parts from biofouling [
20]. In addition, the introduction of Ag and Cu makes it possible to improve the tribological properties of coatings due to the formation of a tribolayer based on a solid lubricant during friction [
21,
22].
Carbides [
23,
24], nitrides [
25,
26] and transition metal carbonitrides [
27], as well as oxide phases [
28,
29] such as TaC, TiC, TiN, SiC, TiCN, TiO
2, Al
2O
3, ZrO
2, CeO
2, WO
3 and Si
3N
4 are often used as hard and wear-resistant structural components in protective coatings. Depending on the required composition, thickness, surface roughness and geometric shape of the items, methods such as electrospark deposition (ESD) in vacuum [
12,
30], CVD [
31,
32], PVD [
33,
34], laser cladding [
35,
36], galvanic deposition [
37,
38] and many others are used to deposit protective coatings. Among the listed methods, one should single out the method of electrospark deposition (ESD), which is often used to deposit thick wear-resistant and corrosion-resistant coatings [
39]. Vacuum treatment not only allows one to minimize surface oxidation and obtain coatings with high adhesive strength, but also simultaneously implement electrode arc evaporation, which is not realized at atmospheric pressure [
40].
For the protection of steels from tribocorrosion, a promising approach is the development of composite coatings consisting of a corrosion-resistant metal matrix based on Fe, Cr, Ni and Mo, additionally alloyed with Cu or Ag to impart antibacterial properties, and reinforcing TaC inclusions. Tantalum carbide TaC was chosen because it has an electrochemical potential (E
corr = −58 mV) close to the values of a corrosion-resistant iron-based matrix, preventing the creation of additional galvanic pairs [
41]. The aim of this work was to obtain TaC-FeCrMoNi-(Cu/Ag) composite coatings by electrospark alloying in vacuum and to study their corrosion, tribocorrosion, and antibacterial properties in artificial seawater.
2. Materials and Methods
2.1. Electrodes
Electrodes for coating deposition were obtained by cold pressing on a Lab Econ 600 hydraulic press (Fontijne Grotnes BV, Vlaardingen, The Netherlands). For the manufacture of TaC-CrMoNi, TaC-CrMoNi-Cu and TaC-CrMoNi-Ag electrodes, the following powders were used: Cr (PKh-1S, <63 μm), Ni (PNK-0T2, <20 μm), Mo (PM99.95, <5 μm), TaC (MRTU 9-09-03443-77, <5 μm), Cu (PMS-1, <71 μm) and Ag (PS-3, <15 μm, purity 99.0%). The powders were mixed in an Activator-4M planetary mill (Zavod Khimicheskogo Mashinostroeniya, Russia) with a drum volume of 1 L at a powder/balls mass ratio of 1:6 for 4 h. As a result, powder mixtures of three compositions were obtained (at.%): 67.5TaC-12.5Mo-7.5Ni-12.5Cr, 65TaC-10Mo-5Ni-10Cr-10Ag and 65TaC-10Mo-5Ni-10Cr-10Cu. From the resulting mixtures, electrode blanks 7 × 7 × 50 mm3 in size were obtained at a pressing pressure of 250 MPa. The materials were then sintered in a VE-3-16 vacuum furnace (LLC NPP VakETO, Moscow, Russia) at a pressure of 1 × 10−3 Pa and a temperature of 1300 °C for 60 min. After sintering, the electrodes were ground to obtain rods with a diameter of 6 mm. The composition of the electrodes was as follows (at.%): 37Ta-38C-11Cr-9Mo-5Ni, 36Ta-37C-9Cr-4Mo-4Ni-10Ag and 36Ta-37C-9Cr-8Mo-4Ni-6Cu.
2.2. Deposition of Coatings
Coatings were deposited using an original vacuum installation for electrospark processing [
12]. In a vacuum deposition chamber, a 3-axis CNC machine was installed, equipped with a rotating electrode unit and an insulated brush assembly for supplying voltage. The deposition regimes of the TaC-CrMoNi, TaC-CrMoNi-Cu and TaC-CrMoNi-Ag coatings were chosen after preliminary optimization. It was found that in order to obtain defect-free coatings (without cracks and pores) with high continuity and wear and corrosion resistance, the optimal deposition regimes are as follows: electrode rotation speed 1000 rpm, electrode scanning rate 500 mm/min, scanning step 0.5 mm, electrical pulse frequency 100 Hz, pulse voltage 100 V and pulse duration 50 μs. Before deposition, the vacuum chamber was evacuated to a pressure of 5 × 10
−3 Pa, after which Ar (99.993%) was supplied to the chamber and the operating pressure was maintained at 0.5 Pa. The composition of the as-deposited coatings was as follows (at.%): 34TaC-50Fe-11Cr-3Mo-2Ni, 31TaC-48Fe-10Cr-2Mo-2Ni-7Ag and 24TaC-51Fe-13Cr-3Mo-3Ni-6Cu.
Discs made of 420S steel (ø30 × 4 mm) were used as substrates. The 420S steel was chosen because it is martensitic steel capable of hardening and possesses moderate corrosion resistance, and in this work we mainly focus on highly loaded elements that require additional protection against corrosion and wear.
2.3. Coatings Characterization
The structural, elemental and phase composition of the electrodes and coatings were studied by scanning electron microscopy (SEM) using an S-3400N microscope (Hitachi) equipped with a NORAN energy-dispersive detector and X-ray diffraction (XRD) using a D8 Advance diffractometer (Bruker).
Hardness (H) and Young’s modulus (E) were evaluated by nanoindentation using a Nano Hardness Tester (CSM Instruments) on the coating cross-sections. Before measurements, the tip of the Berkovich diamond indenter was calibrated against fused quartz. The maximum indentation load was set at 8 mN. The measurement error did not exceed 10%. Resistance to cyclic, impact-dynamic loads was studied on an impact tester (CemeCon). The tests were carried out for 10
5 cycles at a frequency of 50 Hz (constant parameter of the device) and a load of 500 N. These regimes are optimal for evaluating the fatigue strength of “hard coating/hard substrate” systems and were successfully used in our previous works [
42]. A cemented carbide WC-6Co ball, 5 mm in diameter, was used as a counterbody due to its high strength and fracture toughness, which results in minimal wear of the counterbody during testing. Each sample was tested both in air and in artificial seawater (ASW).
Tribocorrosion properties were studied in ASW using a pin-on-disc tribometer (CSM Instruments) equipped with a rotating three-electrode cell, which allows electrochemical measurements to be carried out both under stationary conditions and during tribological tests. The cell was equipped with an Ag/AgCl reference electrode and an auxiliary Pt electrode. Potentiodynamic measurements were carried out using a VoltaLab PST050 potentiostat (Radiometer Analytical) with coating surface polarization from −200 to +1000 mV relative to the open corrosion potential (OCP) at a scanning rate of 1 mV/s. All potentials were recalculated and presented relative to the standard hydrogen electrode. The corrosion current density (CCD) was calculated using the Tafel equation. Artificial seawater was prepared according to ASTM D 1141-98 (
Table 1). Instead of cemented carbide, an alumina ball (Al
2O
3) with a diameter of 6 mm and a roughness of 0.8 µm was used as a counterbody in all ASW experiments because it is a non-conductive and corrosion resistant material and does not affect the corrosion potential measurements.
The tests were carried out at a load of 5 N, a sliding speed of 10 cm/s, and a test distance of 1000 m. The recovery rate of corrosion potential was observed during short pauses (up to 10 min) without rotation.
Coating wear rates were calculated by analyzing the wear track profiles using a Wyko-NT110 optical profiler (Veeco, Plainview, NY, USA). Due to the high roughness of the coatings, the relief of the coatings was smoothed to a roughness (Ra) of less than 1 µm before tribocorrosion tests in order to determine and compare the wear rates of the coatings.
The antibacterial activity of the coating was assessed by counting the number of colony-forming units (CFUs) of gram-positive Bacillus cereus Arc30 strain (collection of the State Scientific Center for Applied Microbiology and Biotechnology). Bacterial strains were cultivated on Mueller Hinton solid nutrient agar (HiMedia, India). Prior to the experiments, all plates were sterilized by UV radiation (exposure 60 min, distance 15 cm). One sample of each type was placed in 4 mL of saline, and two wells without samples with 4 mL of saline were used as controls (K). Simultaneously, 0.05 mL of a suspension of a 3-h broth culture of the test strain in saline with a cell concentration of about 2 × 106 CFUs/mL was added to all wells. The samples were incubated in a thermostat at 37 °C. All manipulations were carried out under aseptic conditions, the slightest contact between the samples being avoided. After 0 h, 3 h, 8 h and 24 h, 0.04 mL was taken from each well to determine the concentration of CFUs. Determination of the concentration of CFUs/mL was carried out according to the method of decimal dilutions in 0.3 mL of saline. A 0.01 mL bacterial suspension was inoculated from each dilution into Petri dishes with Mueller Hinton Agar nutrient medium (HiMedia, India), dried in a closed dish at room temperature for 10 min and cultured at 37 °C for 24 h.
4. Discussion
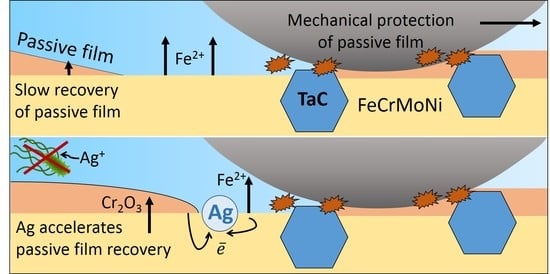
Comparing the data of the electrochemical and tribocorrosion tests, we can draw the following conclusions. The corrosion resistance of coating 1 is mostly determined by the quality of the passive film since TaC carbides are characterized by electrochemical potential close to that of a metal matrix and the effect of galvanic pairs is minimal. In contrast, the Ag and Cu particles on the coatings’ surface are active cathodes. In the absence of friction, the Ag- and Cu-containing coatings are more corrosion resistant due to the formation of a higher quality surface passivating film than that of coating 1. However, the distribution of metal particles over the coating surfaces is different. Silver is represented by relatively small particles (up to 2 μm) evenly distributed over the surface. Copper is found both on the surface, in the form of larger particles up to 10 µm, and in the bulk of the coating. In addition, it should be taken into account that Ag particles, unlike Cu, are covered with a thin film of silver chloride. These differences in the composition and distribution of particles determine the coatings’ resistance to corrosion.
On the one hand, the presence of Ag and Cu particles leads to the formation of a stable passivating film on the metal matrix. On the other hand, an excess cathode potential at the (Ag, Cu)/matrix interface can lead to the breakdown of the passivating film and the appearance of pitting defects. Large Cu particles form a large potential difference at the interface, which leads to the breakdown of the passivating film and the dissolution of the matrix around the particles. As a result, a relatively high corrosion current and strong pitting corrosion are observed in coating 3Cu. The Ag-containing coating is characterized by a minimal corrosion current and the absence of pitting corrosion due to the screening of the Ag particles by a chloride film. In this case, there is an optimal combination of the passivation effect of the metal matrix and the low Ag/matrix interface currents that prevents pitting corrosion.
Under tribological contact conditions, the passivating film is constantly removed, the measured potential is determined by the metal matrix potential without the passivating film, and the contribution of passivated regions is small. In this case, the corrosion rate is determined by the recovery rate of the passivating film after its removal by the counterpart material. The maximum drop in potential is observed for SS, since the Cr content, as the main passivating element, is lower compared with the coatings. In the coatings, the recovery rate of the passivating film is determined by the activity of the Ag and Cu particles in the wear track. In coating 1, the potentials between the TaC carbide (−58 mV) and the matrix (−87 mV) are relatively close, so the efficiency of the carbide grains as cathodes is minimal, resulting in the maximum potential drop among the tested coatings. The presence of Cu in the wear track accelerates the surface passivation and results in less potential shift. The minimum potential drop is observed for the Ag-doped coatings and is explained by the fact that Ag has a more positive potential than Cu (i.e., it is a more efficient cathode), and the chloride film that shields Ag under stationary conditions is constantly removed, which accelerates the passivating film recovery. Thus, coating 2Ag demonstrates the maximum resistance under both stationary corrosion and tribocorrosion conditions, since Ag acts as an effective cathode with respect to the metal matrix and promotes more efficient matrix passivation [
49].
The coating behavior under cyclic impact-dynamic loads in ASW is different from that under tribological contact conditions. In both cases, the materials are subjected to abrasion, which leads to continuous wear of the passivating film, and corrosive attack, i.e., both types of tests evaluate the resistance of a material to friction, wear, and corrosion. The obtained results show that under impact conditions, the material hardness is the determining factor, and intensive wear of the soft coating components (metallic Ag, Cu and matrix) leads to the leveling of the anticorrosion effects associated with them. In contrast, under tribocontact conditions, an increase in hardness and Youngs modulus (in the following order: 2Ag → 3Cu → 1) leads to a gradual increase both in CoF and wear rate values. In accordance with [
50], one may expect that the CoF and wear rate in a “spherical counterbody–plane surface” system would decrease. The observed controversial effect relates to the fact that the decrease in mechanical properties is due to the addition of soft metals (Ag and Cu) to coating 1, which form particles or a discontinuous layer on the surface and act as solid lubricants in sea water. Hence, Ag and Cu not only increase corrosion resistance, but also act as a solid lubricant, resulting in reduced friction and wear.
The potential application of the developed coatings is the protection of steel products operating in aggressive environments containing suspensions of abrasive particles from tribocorrosion, cavitation and biological effects, typical for the infrastructure of the river and sea fleet, hydraulic structures, the equipment of the oil and gas industry and the chemical industry, and manufacturing and sanitary equipment in urban environments.
5. Conclusions
The vacuum electrospark deposition method was successfully applied to produce relatively thick (50–75 μm) tribocorrosion-resistant Ag- and Cu-doped TaC-FeCrMoNi coatings on the surface of AISI 420S steel. The coatings have a composite structure, in which TaC grains 0.2–5.0 µm in size are uniformly distributed in Fe-based metal matrices. Doping with Ag led to the formation of surface particles 1–4 μm in size, while Cu was observed both on the surface and inside the coatings.
The improved corrosion resistance of the coatings was due to the high Cr content and the minimal galvanic effect between the TaC grains and the Fe-based matrix. The Ag-doped coating demonstrated the most positive corrosion potential value and the lowest current density, and it did not show signs of pitting corrosion after exposure to artificial seawater for 20 days.
The coatings had an increased hardness and wear resistance compared with the substrate and withstood 105 cycles in impact-dynamic tests in air and in artificial seawater at a load of 1000 N. The Ag- and Cu-doped coatings showed accelerated passivation in tribocorrosion tests.
The Ag-containing coating had the lowest CoF (0.2–0.25) and the lowest wear rate (1.6 × 10−6 mm3/Nm) in artificial seawater due to the Ag particles acting as a solid lubricant. The Ag- and Cu-doped coatings showed accelerated passivation in tribocorrosion tests.
All coatings exhibited a pronounced bactericidal effect against Bacillus cereus Arc30 strain, while the best characteristics were observed for the TaC-(CrMoNi) and TaC-(CrMoNi)-Ag coatings.