Corrosion Behavior of API-X120 Carbon Steel Alloy in a GTL F-T Process Water Environment at Low COD Concentration
Abstract
:1. Introduction
2. Experimental Set-Up and Procedures
2.1. Material and Solution
2.1.1. Working Electrode
2.1.2. GTL Produced Water Characterization
2.2. Experimental Set-Up
2.3. Electrochemical Measurement
2.4. Morphology and Composition Analysis
3. Results and Discussion
3.1. Effect of Temperature.
3.1.1. Potentiodynamic Polarization
3.1.2. Electrochemical Impedance Spectroscopy (EIS) Data Analysis
3.2. Effect of Immersion Time
3.2.1. Potentiodynamic Polarization Curves
3.2.2. Electrochemical Impedance Spectroscopy (EIS)
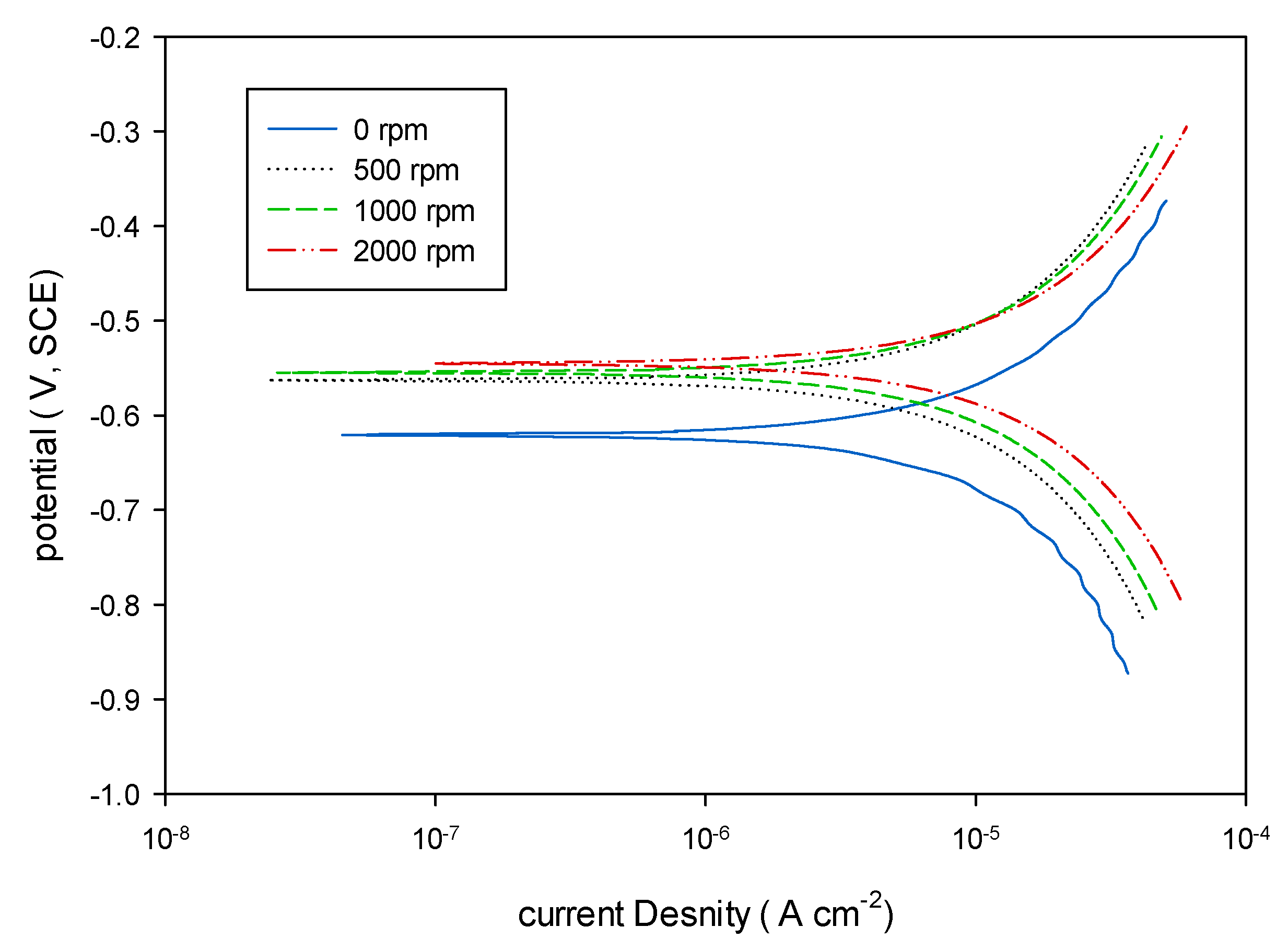
3.3. Effect of Rotation Speed
3.4. Morphology Analysis
3.4.1. Visual Observation
3.4.2. Scanning Electron Microscope (SEM) Analysis
3.5. XRD Analysis
3.6. XPS Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sönmez, E.; Kekre, S.; Scheller-Wolf, A.; Secomandi, N. Strategic analysis of technology and capacity investments in the liquefied natural gas industry. Eur. J. Oper. Res. 2013, 226, 100–114. [Google Scholar] [CrossRef]
- Neerkathalingam, P.S.; Van Vuuren, M.J. Process Wastewater Treatment and Management in Gas to Liquids Industries. In SPE Oil and Gas India Conference and Exhibition; Society of Petroleum Engineers: Richardson, TX, USA, 2010. [Google Scholar]
- Onwusogh, U. Feasibility of Produced Water Treatment and Reuse—Case Study of a GTL Plant. In Proceedings of the International Petroleum Technology Conference 2015, Doha, Qatar, 6–9 December 2015. [Google Scholar]
- Majone, M.; Aulenta, F.; Dionisi, D.; D’Addario, E.N.; Sbardellati, R.; Bolzonella, D.; Beccari, M. High-rate anaerobic treatment of Fischer–Tropsch wastewater in a packed-bed biofilm reactor. Water Res. 2010, 44, 2745–2752. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Han, H.; Han, Y.; Li, K.; Zhu, H. Enhanced treatment of Fischer-Tropsch (FT) wastewater using the up-flow anaerobic sludge blanket coupled with bioelectrochemical system: Effect of electric field. Bioresour. Technol. 2017, 232, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Ma, W.; Han, H.; Li, K.; Hao, X. Enhanced treatment of Fischer–Tropsch (FT) wastewater by novel anaerobic biofilm system with scrap zero valent iron (SZVI) assisted. Biochem. Eng. J. 2017, 117, 66–76. [Google Scholar] [CrossRef]
- Benamor, A.; Talkhan, A.G.; Nasser, M.; Hussein, I.; Okonkwo, P.C. Effect of temperature and fluid speed on the corrosion behavior of carbon steel pipeline in Qatari oilfield produced water. J. Electroanal. Chem. 2018, 808, 218–227. [Google Scholar] [CrossRef]
- Bahgat Radwan, A.; Sliem, M.H.; Okonkwo, P.C.; Shibl, M.F.; Abdullah, A.M. Corrosion inhibition of API X120 steel in a highly aggressive medium using stearamidopropyl dimethylamine. J. Mol. Liq. 2017, 236, 220–231. [Google Scholar] [CrossRef]
- Wang, X.; Song, X.; Chen, Y.; Wang, Z.; Zhang, L. Corrosion Behavior of X70 and X80 Pipeline Steels in Simulated Soil Solution. Int. J. Electrochem. Sci. 2018, 13, 6436–6450. [Google Scholar] [CrossRef]
- Paul, S.; Pattanayak, A.; Guchhait, S.K. Corrosion behavior of carbon steel in synthetically produced oil field seawater. Int. J. Metals 2014, 628505. [Google Scholar] [CrossRef] [Green Version]
- Din Yati, M.S.; Derman, M.; Isa, M.; Muhammad, M.M.; Nain, H. Electrochemical Characterisation of Hybrid Activators for Aluminium Sacrificial Anodes in Natural Sea Water. Def. S&T Tech. Bull. 2013, 6. [Google Scholar]
- Kugel, A.J.; Jarabek, L.E.; Daniels, J.W.; Vander Wal, L.J.; Ebert, S.M.; Jepperson, M.J.; Stafslien, S.J.; Pieper, R.J.; Webster, D.C.; Bahr, J. Combinatorial materials research applied to the development of new surface coatings XII: Novel, environmentally friendly antimicrobial coatings derived from biocide-functional acrylic polyols and isocyanates. J. Coat. Technol. Res. 2009, 6, 107–121. [Google Scholar] [CrossRef]
- Desimone, M.P.; Gordillo, G.; Simison, S.N. The effect of temperature and concentration on the corrosion inhibition mechanism of an amphiphilic amido-amine in CO 2 saturated solution. Corros. Sci. 2011, 53, 4033–4043. [Google Scholar] [CrossRef] [Green Version]
- Veawab, A.; Tontiwachwuthikul, P.; Chakma, A. Corrosion behavior of carbon steel in the CO2 absorption process using aqueous amine solutions. Ind. & Eng. Chem. Res. 1999, 38, 3917–3924. [Google Scholar]
- Zhao, B.; Sun, Y.; Yuan, Y.; Gao, J.; Wang, S.; Zhuo, Y.; Chen, C. Study on corrosion in CO2 chemical absorption process using amine solution. Energy Procedia 2011, 4, 93–100. [Google Scholar] [CrossRef] [Green Version]
- Porcayo-Calderon, J.; Escalera, L.M.; Canto, J.; Casales-Diaz, M.; Salinas-Bravo, V. Effect of the Temperature on the CO2-Corrosion of Ni3Al. Int. J. Electrochem. Sci. 2015, 10, 3136–3151. [Google Scholar]
- Gürten, A.A.; Keleş, H.; Bayol, E.; Kandemirli, F. The effect of temperature and concentration on the inhibition of acid corrosion of carbon steel by newly synthesized Schiff base. J. Ind. Eng. Chem. 2015, 27, 68–78. [Google Scholar] [CrossRef]
- Esmaily, M.; Shahabi-Navid, M.; Svensson, J.-E.; Halvarsson, M.; Nyborg, L.; Cao, Y.; Johansson, L.-G. Influence of temperature on the atmospheric corrosion of the Mg–Al alloy AM50. Corros. Sci. 2015, 90, 420–433. [Google Scholar] [CrossRef] [Green Version]
- Haile, T.; Sopkow, L.; Choi, L.; Wolodko, J.; Tsaprailis, H. Corrosivity of Produced and Make-up Water in Oil Sands Thermal Water Treatment Systems. In NACE International Corrosion Conference Proceedings; NACE International: Houston, TX, USA, 2016. [Google Scholar]
- Stern, M.; Geaby, A.L. Electrochemical polarization I. A theoretical analysis of the shape of polarization curves. J. Electrochem. Soc. 1957, 104, 56–63. [Google Scholar] [CrossRef]
- Melot, D.; Paugam, G.; Roche, M. Disbondments of pipeline coatings and their effects on corrosion risks. J. Prot. Coat. Linings 2009. Available online: https://pdfs.semanticscholar.org/e96f/faa69e0368a901b19f319923b262fb8fcbae.pdf (accessed on 20 March 2020).
- Wang, X.; Xu, J.; Sun, C.; Yan, M. Effect of oilfield produced water on corrosion of pipeline. Int. J. Electrochem. Sci. 2015, 10, 8656–8667. [Google Scholar]
- Leibig, M.; Halsey, T.C. The double layer impedance as a probe of surface roughness. Electrochim. Acta 1993, 38, 1985–1988. [Google Scholar] [CrossRef]
- Maocheng, Y.; Jin, X.; Libao, Y.U.; Tangqing, W.; Cheng, S.U.N.; Wei, K.E. EIS analysis on stress corrosion initiation of pipeline steel under disbonded coating in near-neutral pH simulated soil electrolyte. Corros. Sci. 2016, 110, 23–34. [Google Scholar] [CrossRef] [Green Version]
- Harris, M. Effects of Corrosion Inhibitor on the Corrosion Behavior of Low Carbon Steel in CO2 Environment. 2017. Available online: https://ideaexchange.uakron.edu/honors_research_projects/475/ (accessed on 8 March 2020).
- Mustafa, C.M.; Rahman, A.K.M.O.; Begum, D.A. Effects of time and temperature on the mild steel corrosion inhibition by molybdate and nitrite. Indian J. Chem. Technol. 1996, 3, 44–48. [Google Scholar]
- Keddam, M.; Mottos, O.R.; Takenouti, H. Reaction model for iron dissolution studied by electrode impedance I. Experimental results and reaction model. J. Electrochem. Soc. 1981, 128, 257–266. [Google Scholar] [CrossRef]
- Baig, M.; Ammar, H.R.; Seikh, A.H.; Alam, M.A.; Alharthi, N.H. Effect of Immersion Time and Temperature on Corrosion Behaviour of Nanocrystalline Al-Fe-Cr Alloy. J. Electrochem. Sci. 2017, 12, 3336–3349. [Google Scholar] [CrossRef]
- Lopes-Sesenes, R.; Dominguez-Patiño, G.F.; Gonzalez-Rodriguez, J.; Uruchurtu, J. Effect of flowing conditions on the corrosion inhibition of carbon steel by extract of buddleia perfoliata. Int. J. Electrochem. Sci. 2013, 8, 477–489. [Google Scholar]
- Tian, J.; Huang, H.; Pan, Z.-Q.; Zhou, H. Effect of flow velocity on corrosion behavior of AZ91D magnesium alloy at elbow of loop system. Trans. Nonferrous Metals Soc. China 2016, 26, 2857–2867. [Google Scholar] [CrossRef]
- Shen, S.; Yang, Y.-N.; Bian, Y.; Zhao, Y. Kinetics of CO2 absorption into aqueous basic amino acid salt: Potassium salt of lysine solution. Environ. Sci. & Technol. 2016, 50, 2054–2063. [Google Scholar]
- Ortega-Toledo, D.M.; Gonzalez-Rodriguez, J.G.; Casales, M.; Martinez, L.; Martinez-Villafañe, A. CO2 corrosion inhibition of X-120 pipeline steel by a modified imidazoline under flow conditions. Corros. Sci. 2011, 53, 3780–3787. [Google Scholar] [CrossRef]
- Kitamura, N.; Sakamoto, T.; Takebe, H. Effect of Reaction Layer on Water Durability in BaO–FeO–Fe2O3–P2O5 Glasses. Mater. Trans. 2018, 59, 437–442. [Google Scholar] [CrossRef] [Green Version]
- Sun, J.; Liu, W.; Chang, W.; Zhang, Z.; Li, Z.; Yu, T.; Lu, M. Characteristics and formation mechanism of corrosion scales on low—Chromium x65 steels in CO2 environment. Acta Metall Sin 2009, 45, 84–90. [Google Scholar]















| Unit | Contaminant |
|---|---|
| F-T reaction | Inorganic compounds and oxygenated hydrocarbons |
| Cooling tower blow down water | Dissolved solids, suspended solids and heavy metals |
| Steam generation unit blow-down water | Dissolved solids and minerals |
| Process area, equipment washing and maintenance activities | Oil, emulsified oil and other hydrocarbons |
| Parameter, Dimension, (SMWW Method) | Value | Used Method |
|---|---|---|
| TOC, mg/L | 125.4 | Total organic carbon analyser (TOC-L) |
| COD, mg/L | 530 | APHA 5220 C Closed Reflux, Titrimetric Method |
| Phenols, mg/L | 0.131 | HAC spectrophotometer |
| PH, (4500 H+. B) | 4.18 | Electrometric Method |
| Conductivity, μs/cm | 124.6 | Conductivity meter |
| Salinity, ppt | 0.06 | Standard method |
| TDS, mg/L | 38.5 | Standard method |
| TSS, mg/L (APHA 2540 B) | 2.5 | Total Suspended Solids Dried at 103–105 °C |
| Component | Unit | Concentration |
|---|---|---|
| Ammonium | ppm | 8.450 |
| Potassium | ppm | 0.058 |
| Calcium | ppm | 2.044 |
| Magnesium | ppm | 0.068 |
| Boron | ppb | <dl * |
| Vanadium | ppb | 0.018 |
| Iron | ppb | 31.144 |
| Copper | ppb | 0.314 |
| Barium | ppb | <dl |
| Temperature (K) | Immersion Time (min) | Rotation Speed (RPM) |
|---|---|---|
| 293, 303, 313, and 323 | 0, 30, 60, and 120 | 0, 500, 1000, and 2000 |
| Temperature (K) | 293 | 303 | 313 | 323 |
|---|---|---|---|---|
| Beta A (ba), (V·decade−1) | 0.189 | 0.24 | 0.222 | 0.270 |
| Beta C (bc), (V·decade−1) | 0.212 | 0.280 | 0.290 | 0.350 |
| Rp, [KΩ·cm2] | 5.691 | 4.619 | 2.562 | 2.256 |
| Corrosion Current, Icorr (µA) | 7.623 | 12.15 | 21.31 | 29.34 |
| Corrosion Current Density (µA·cm−2) | 9.26 | 14.76 | 25.89 | 35.65 |
| Corrosion Potential, Ecorr (mV) | −621.8 | −620.5 | −618.3 | −625.1 |
| Corrosion rate (mpy) | 4.238 | 6.753 | 11.85 | 16.31 |
| Temperature (K) | Rs | Rct | Qdl | |
|---|---|---|---|---|
| CPE | n-CPE | |||
| [Ω·cm2] | [Ω·cm2] | [Ω−1·cm−2·s−n] | ||
| 293 | 76 | 1126 | 5.41 × 10−5 | 0.78 |
| 303 | 63 | 723 | 5.95 × 10−5 | 0.733 |
| 313 | 48 | 653 | 5.10 × 10−5 | 0.784 |
| 323 | 32 | 412 | 6.53 × 10−5 | 0.78 |
| Time (min) | 0 | 30 | 60 | 120 |
|---|---|---|---|---|
| Beta A (ba), (V·decade−1) | 0.240 | 0.205 | 0.177 | 0.145 |
| Beta C (bc), (V·decade−1) | 0.280 | 0.214 | 0.187 | 0.154 |
| Rp, [KΩ·cm2] | 4.619 | 4.155 | 3.841 | 3.414 |
| Corrosion Current, Icorr (µA) | 12.15 | 10.94 | 10.28 | 9.498 |
| Corrosion Current Density (µA·cm−2) | 14.76 | 13.29 | 12.49 | 11.54 |
| Corrosion Potential, Ecorr (mV) | −620.5 | −616.8 | −611.7 | −607.1 |
| Corrosion rate (mpy) | 6.753 | 6.083 | 5.715 | 5.28 |
| Time (min) | Rs | Rct | Qdl | |
|---|---|---|---|---|
| CPE | n-CPE | |||
| [Ω·cm2] | [Ω·cm2] | [Ω−1c·m−2·s−n] | ||
| 0 | 63 | 723 | 5.95 × 10−5 | 0.733 |
| 30 | 57 | 854 | 6.24 × 10−5 | 0.787 |
| 60 | 44 | 904 | 6.74 × 10−5 | 0.805 |
| 120 | 38 | 1204 | 6.73 × 10−5 | 0.822 |
| Speed (rpm) | 0 | 500 | 1000 | 2000 |
|---|---|---|---|---|
| Beta A (ba), (V·decade−1) | 0.189 | 0.222 | 0.226 | 0.207 |
| Beta C (bc), (V·decade−1) | 0.212 | 0.223 | 0.224 | 0.218 |
| Rp, [KΩ·cm2] | 5.691 | 5.943 | 5.24 | 4.255 |
| Corrosion Current, Icorr (µA) | 7.623 | 8.128 | 9.322 | 10.83 |
| Corrosion Current Density (µA·cm−2) | 9.26 | 9.88 | 11.33 | 13.16 |
| Corrosion Potential, Ecorr (mV) | −621.8 | −563 | −555 | −545 |
| Corrosion rate (mpy) | 4.238 | 7.747 | 8.885 | 10.33 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ewis, D.; Talkhan, A.G.G.; Benamor, A.; Qiblawey, H.; Nasser, M.; Ba-Abbad, M.M.M.; El-Naas, M. Corrosion Behavior of API-X120 Carbon Steel Alloy in a GTL F-T Process Water Environment at Low COD Concentration. Metals 2020, 10, 707. https://doi.org/10.3390/met10060707
Ewis D, Talkhan AGG, Benamor A, Qiblawey H, Nasser M, Ba-Abbad MMM, El-Naas M. Corrosion Behavior of API-X120 Carbon Steel Alloy in a GTL F-T Process Water Environment at Low COD Concentration. Metals. 2020; 10(6):707. https://doi.org/10.3390/met10060707
Chicago/Turabian StyleEwis, Dina, Ahmed Gomaa Gomaa Talkhan, Abdelbaki Benamor, Hazim Qiblawey, Mustafa Nasser, Muneer M. M. Ba-Abbad, and Muftah El-Naas. 2020. "Corrosion Behavior of API-X120 Carbon Steel Alloy in a GTL F-T Process Water Environment at Low COD Concentration" Metals 10, no. 6: 707. https://doi.org/10.3390/met10060707







