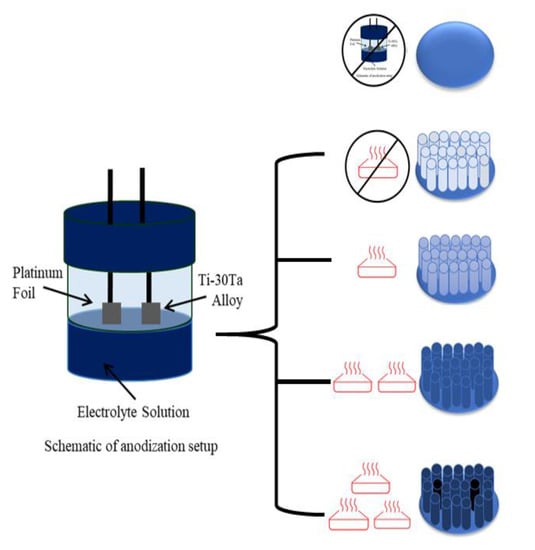
Influence of Annealing Temperature on Corrosion Resistance of TiO2 Nanotubes Grown on Ti–30Ta Alloy
Abstract
:1. Introduction
2. Materials and Methods
2.1. Alloy Processing
2.2. Nanotubes Growth
2.3. Electrochemical Behavior
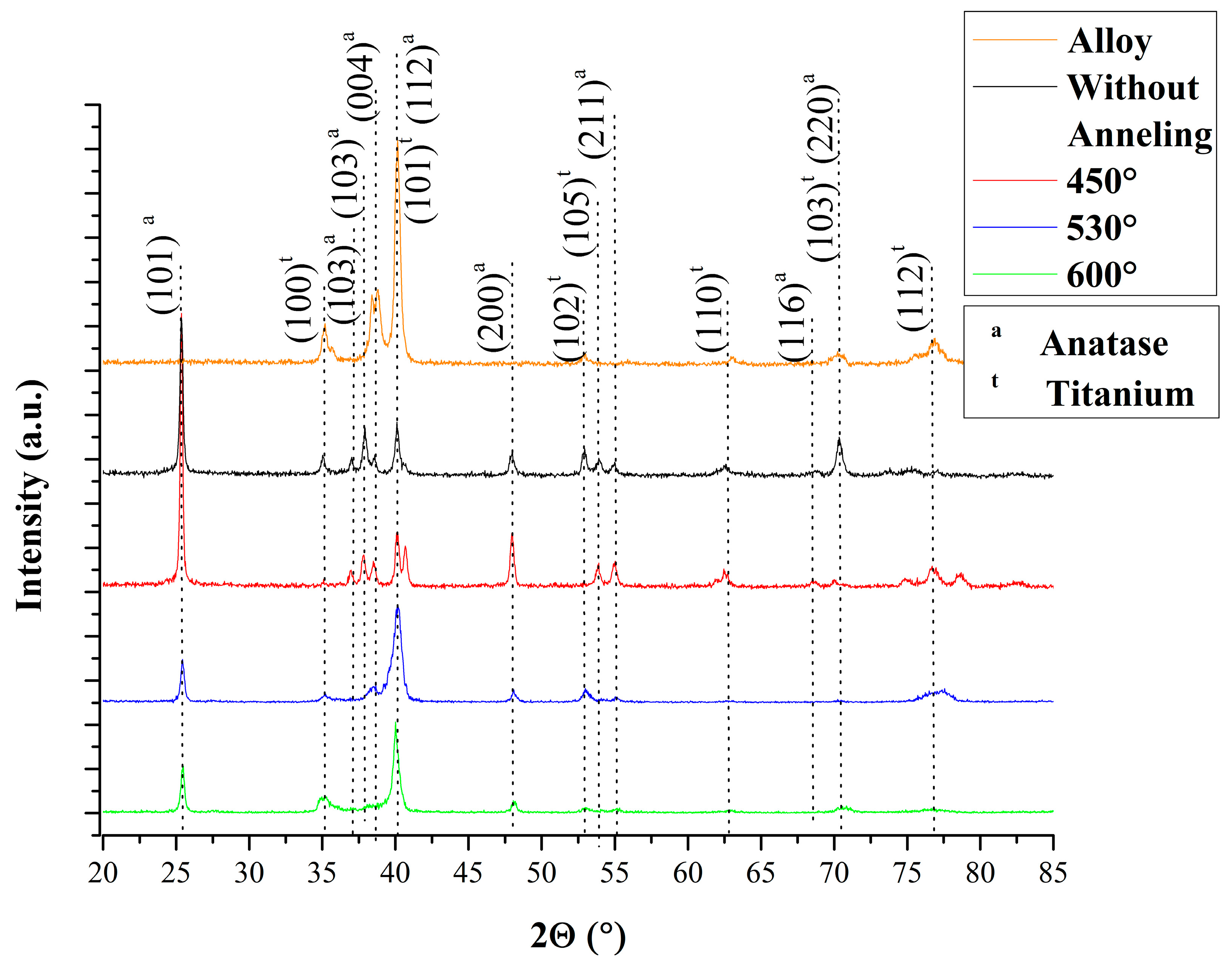
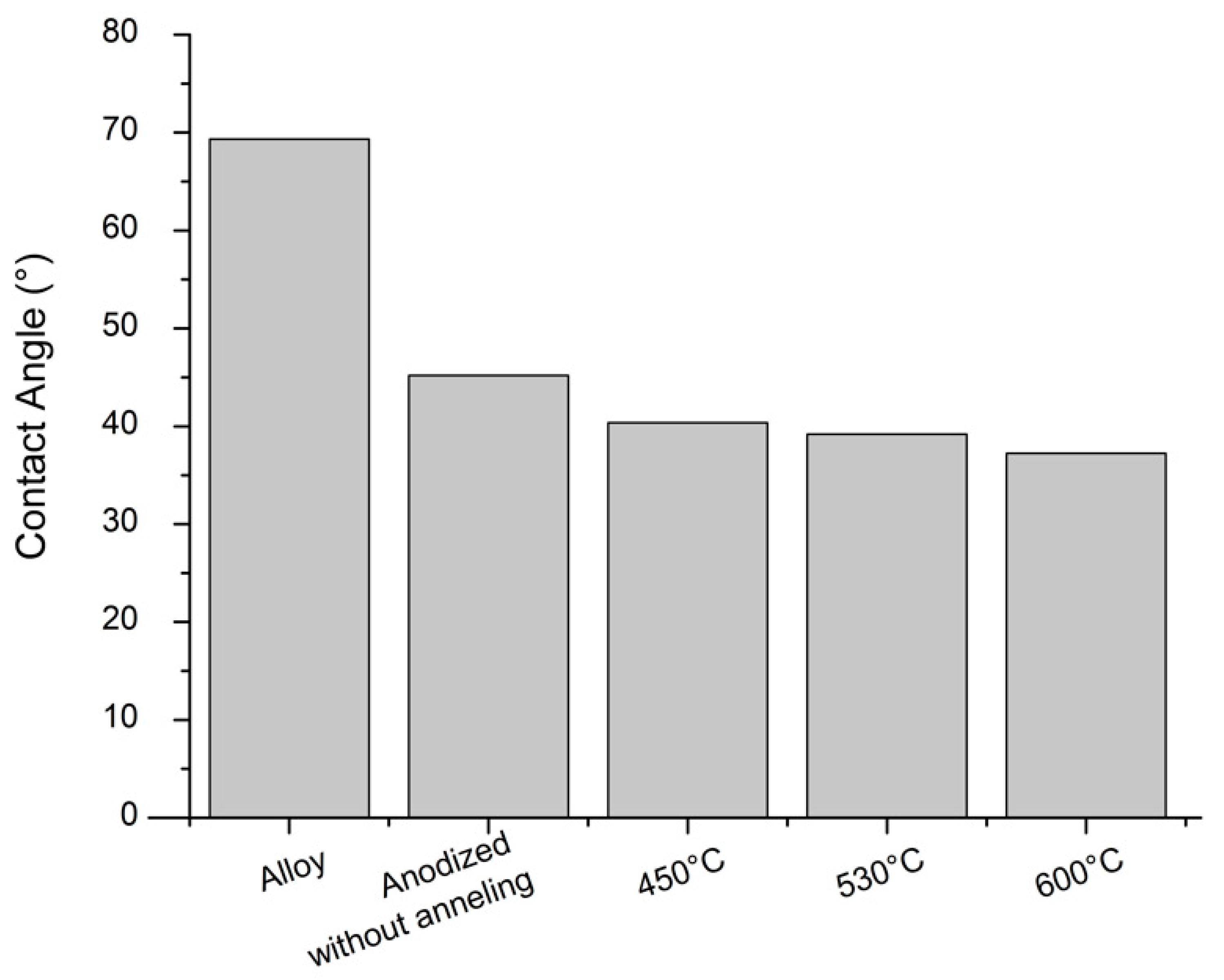
2.4. Sample Characterization
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Verma, R.P. Materials Today: Proceedings Titanium based biomaterial for bone implants: A mini review. Mater. Today Proc. 2020, 2–5. [Google Scholar] [CrossRef]
- Chouirfa, H.; Bouloussa, H.; Migonney, V.; Falentin-Daudré, C. Review of titanium surface modification techniques and coatings for antibacterial applications. Acta Biomater. 2019, 83, 37–54. [Google Scholar] [CrossRef]
- Wilson, J. Metallic biomaterials: State of the art and new challenges. Fundam. Biomater. Met. 2018, 1–33. [Google Scholar] [CrossRef]
- Pandey, A.; Awasthi, A.; Saxena, K.K. Metallic implants with properties and latest production techniques: A review. Adv. Mater. Process. Technol. 2020, 6, 405–440. [Google Scholar] [CrossRef]
- Domingues Goncalves, A.; Balestri, W.; Reinwald, Y. Biomedical Implants for Regenerative Therapies. In Biomaterials [Working Title]; IntechOpen: London, UK, 2020. [Google Scholar]
- Zhou, Y.L.; Niinomi, M.; Akahori, T.; Fukui, H.; Toda, H. Corrosion resistance and biocompatibility of Ti-Ta alloys for biomedical applications. Mater. Sci. Eng. A 2005, 398, 28–36. [Google Scholar] [CrossRef]
- Capellato, P.; Claro, A.P.R.A.; Silva, G.; Zavaglia, C.A.C. Antimicrobial effect of TiO2 nanotubes coating for dental implant. Dent. Mater. 2018, 34, e21. [Google Scholar] [CrossRef]
- Kim, S.E.; Jeong, H.W.; Hyun, Y.T.; Lee, Y.T.; Jung, C.H.; Kim, S.K.; Song, J.S.; Lee, J.H. Elastic modulus and in vitro biocompatibility of Ti− xNb and Ti− xTa alloys. Met. Mater. Int. 2007, 13, 145–149. [Google Scholar] [CrossRef]
- Capellato, P.; Escada, A.L.A.; Popat, K.C.; Claro, A.P.R.A. Interaction between mesenchymal stem cells and Ti-30Ta alloy after surface treatment. J. Biomed. Mater. Res. Part A 2014, 102, 2147–2156. [Google Scholar] [CrossRef]
- Kulkarni, M.; Flašker, A.; Lokar, M.; Mrak-Poljšak, K.; Mazare, A.; Artenjak, A.; Čučnik, S.; Kralj, S.; Velikonja, A.; Schmuki, P.; et al. Binding of plasma proteins to titanium dioxide nanotubes with different diameters. Int. J. Nanomed. 2015, 10, 1359–1373. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Rath, B.; Tingart, M.; Eschweiler, J. Role of implants surface modification in osseointegration: A systematic review. J. Biomed. Mater. Res. Part A 2020, 108, 470–484. [Google Scholar] [CrossRef] [Green Version]
- Jedrzejowski, P.; Vetrone, F.; Zalzal, S.; Sarkissian, A.; Clair, S.; Richert, L.; Perepichka, D.F.; Wuest, J.D.; Yi, J.-H.; Variola, F.; et al. Improving Biocompatibility of Implantable Metals by Nanoscale Modification of Surfaces: An Overview of Strategies, Fabrication Methods, and Challenges. Small 2009, 5, 996–1006. [Google Scholar] [CrossRef]
- Fan, X.; Feng, B.; Liu, Z.; Tan, J.; Zhi, W.; Lu, X.; Wang, J.; Weng, J. Fabrication of TiO2 nanotubes on porous titanium scaffold and biocompatibility evaluation in vitro and in vivo. J. Biomed. Mater. Res. Part A 2012, 100, 3422–3427. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, M.T.; Khan, Z.A.; Siddiquee, A.N. Surface Modifications of Titanium Materials for developing Corrosion Behavior in Human Body Environment: A Review. Procedia Mater. Sci. 2014, 6, 1610–1618. [Google Scholar] [CrossRef] [Green Version]
- Capellato, P.; Sachs, D.; Claro, A.P.R.A.; Silva, G.; Zavaglia, C.A.C. Comparative research of bacteria gram-negative and positive on ti-30ta alloy. Dent. Mater. 2019, 35, e6–e7. [Google Scholar] [CrossRef]
- Capellato, P.; Smith, B.S.; Popat, K.C.; Alves Claro, A.P.R. Cellular Functionality on Nanotubes of Ti-30Ta Alloy; Materials Science Forum: Foz de Iguaçu, Brazil, 2015; Volume 805, ISBN 9783038352365. [Google Scholar]
- Capellato, P.; Smith, B.S.; Popat, K.C.; Claro, A.P.R.A. Fibroblast functionality on novel Ti30Ta nanotube array. Mater. Sci. Eng. C 2012, 32, 2060–2067. [Google Scholar] [CrossRef]
- Capellato, P.; Silva, G.; Popat, K.; Simon-Walker, R.; Alves Claro, A.P.; Zavaglia, C. Cell investigation of Adult Human dermal fibroblasts on PCL nanofibers/TiO2 nanotubes Ti-30Ta alloy for biomedical application. Artif. Organs 2020, 44, 877–882. [Google Scholar] [CrossRef]
- Abdeen, D.H.; El Hachach, M.; Koc, M.; Atieh, M.A. A review on the corrosion behaviour of nanocoatings on metallic substrates. Materials 2019, 12, 210. [Google Scholar] [CrossRef] [Green Version]
- Manam, N.S.; Harun, W.S.W.; Shri, D.N.A.; Ghani, S.A.C.; Kurniawan, T.; Ismail, M.H.; Ibrahim, M.H.I. Study of corrosion in biocompatible metals for implants: A review. J. Alloys Compd. 2017, 701, 698–715. [Google Scholar] [CrossRef] [Green Version]
- Uwais, Z.A.; Hussein, M.A.; Samad, M.A.; Al-Aqeeli, N. Surface Modification of Metallic Biomaterials for Better Tribological Properties: A Review. Arab. J. Sci. Eng. 2017, 42, 4493–4512. [Google Scholar] [CrossRef]
- Asri, R.I.M.; Harun, W.S.W.; Samykano, M.; Lah, N.A.C.; Ghani, S.A.C.; Tarlochan, F.; Raza, M.R. Corrosion and surface modification on biocompatible metals: A review. Mater. Sci. Eng. C 2017, 77, 1261–1274. [Google Scholar] [CrossRef] [Green Version]
- Trincă, L.C.; Mareci, D.; Solcan, C.; Fântânariu, M.; Burtan, L.; Vulpe, V.; Hriţcu, L.D.; Souto, R.M. In vitro corrosion resistance and in vivo osseointegration testing of new multifunctional beta-type quaternary TiMoZrTa alloys. Mater. Sci. Eng. C 2020, 108, 110485. [Google Scholar] [CrossRef]
- Mendis, S.; Xu, W.; Tang, H.P.; Jones, L.A.; Liang, D.; Thompson, R.; Choong, P.; Brandt, M.; Qian, M. Characteristics of oxide films on Ti-(10–75)Ta alloys and their corrosion performance in an aerated Hank’s balanced salt solution. Appl. Surf. Sci. 2020, 506, 145013. [Google Scholar] [CrossRef]
- Capellato, P.; Riedel, N.A.; Williams, J.D.; Machado, J.P.B.; Ketul Popat, K.C.; Alves Claro, A.P.R. Ion Bean Etching on ti-30ta Alloy for Biomedical Application; Materials Science Forum: Foz de Iguaçu, Brazil, 2015; Volume 805, ISBN 9783038352365. [Google Scholar]
- Eduardo Muzzio, N.; Azzaroni, O.; Moya, S.E.; Ángel Pasquale, M. Concepts for Designing Tailored Thin Film Surfaces with Potential Biological Applications. In Multilayer Thin Films—Versatile Applications for Materials Engineering; IntechOpen: London, UK, 2020. [Google Scholar]
- Jaroenworaluck, A.; Regonini, D.; Bowen, C.R.; Stevens, R. A microscopy study of the effect of heat treatment on the structure and properties of anodised TiO 2 nanotubes. Appl. Surf. Sci. 2010, 256, 2672–2679. [Google Scholar] [CrossRef]
- Regonini, D.; Jaroenworaluck, A.; Stevens, R.; Bowen, C.R. Effect of heat treatment on the properties and structure of TiO2 nanotubes: Phase composition and chemical composition. Surf. Interface Anal. 2010, 42, 139–144. [Google Scholar] [CrossRef]
- Mansoorianfar, M.; Mansourianfar, M.; Fathi, M.; Bonakdar, S.; Ebrahimi, M.; Zahrani, E.M.; Hojjati-Najafabadi, A.; Li, D. Surface modification of orthopedic implants by optimized fluorine-substituted hydroxyapatite coating: Enhancing corrosion behavior and cell function. Ceram. Int. 2020, 46, 2139–2146. [Google Scholar] [CrossRef]
- Kulkarni, M.; Mazare, A.; Schmuki, P.; Iglic, A. Influence of anodization parameters on morphology of TiO2 nanostructured surfaces. Adv. Mater. Lett. 2016, 7, 23–28. [Google Scholar] [CrossRef] [Green Version]
- Kulkarni, M.; Mazare, A.; Gongadze, E.; Perutkova, Š.; Kralj-Iglič, V.; Milošev, I.; Mozetič, M. Titanium nanostructures for biomedical applications. Nanotechnology 2015, 26, 062002. [Google Scholar] [CrossRef]
- Zhou, Y.L.; Niinomi, M.; Akahori, T. Decomposition of martensite α″ during aging treatments and resulting mechanical properties of Ti-Ta alloys. Mater. Sci. Eng. A 2004, 384, 92–101. [Google Scholar] [CrossRef]
- Das, K.; Bose, S.; Bandyopadhyay, A. TiO2 nanotubes on Ti: Influence of nanoscale morphology on bone cell–materials interaction. J. Biomed. Mater. Res. Part A Off. J. Soc. Biomater. Jpn. Soc. Biomater. Aust. Soc. Biomater. Korean Soc. Biomater. 2009, 90, 225–237. [Google Scholar] [CrossRef]
- Khrunyk, Y.Y.; Belikov, S.V.; Tsurkan, M.V.; Vyalykh, I.V.; Markaryan, A.Y.; Karabanalov, M.S.; Popov, A.A.; Wysokowski, M. Surface-Dependent Osteoblasts Response to TiO2 Nanotubes of Different Crystallinity. Nanomaterials 2020, 10, 320. [Google Scholar] [CrossRef] [Green Version]
- Geetha, M.; Singh, A.K.; Asokamani, R.; Gogia, A.K. Ti based biomaterials, the ultimate choice for orthopaedic implants—A review. Prog. Mater. Sci. 2009, 54, 397–425. [Google Scholar] [CrossRef]
- Bayram, C.; Demirbilek, M.; Yalçin, E.; Bozkurt, M.; Doǧan, M.; Denkbaş, E.B. Osteoblast response on co-modified titanium surfaces via anodization and electrospinning. Appl. Surf. Sci. 2014, 288, 143–148. [Google Scholar] [CrossRef]
- Porter, J.R.; Henson, A.; Ryan, S.; Popat, K.C. Biocompatibility and Mesenchymal Stem Cell Response to Poly(ɛ-Caprolactone) Nanowire Surfaces for Orthopedic Tissue Engineering. Tissue Eng. Part A 2009, 15, 2547–2559. [Google Scholar] [CrossRef]
- Devgan, S.; Sidhu, S.S. Evolution of surface modification trends in bone related biomaterials: A review. Mater. Chem. Phys. 2019, 233, 68–78. [Google Scholar] [CrossRef]
- LIU, X.; Chu, P.K.; DING, C. Surface modification of titanium, titanium alloys, and related materials for biomedical applications. Mater. Sci. Eng. R Rep. 2004, 47, 49–121. [Google Scholar] [CrossRef] [Green Version]
- Dai, N.; Zhang, L.-C.; Zhang, J.; Chen, Q.; Wu, M. Corrosion behavior of selective laser melted Ti-6Al-4 V alloy in NaCl solution. Corros. Sci. 2016, 102, 484–489. [Google Scholar] [CrossRef]
- Vasilescu, C.; Drob, S.I.; Calderon Moreno, J.M.; Osiceanu, P.; Popa, M.; Vasilescu, E.; Marcu, M.; Drob, P. Long-term corrosion resistance of new Ti-Ta-Zr alloy in simulated physiological fluids by electrochemical and surface analysis methods. Corros. Sci. 2015, 93, 310–323. [Google Scholar] [CrossRef]
- Fomby, P.; Cherlin, A.J.; Hadjizadeh, A.; Doillon, C.J.; Sueblinvong, V.; Weiss, D.J.; Bates, J.H.T.; Gilbert, T.; Liles, W.C.; Lutzko, C.; et al. Anodic oxide nanotube layers on Ti–Ta alloys: Substrate composition, microstructure and self-organization on two-size scales. Corros. Sci. 2006, 12, 181–204. [Google Scholar] [CrossRef]
- Kirkland, N.T.; Lespagnol, J.; Birbilis, N.; Staiger, M.P. A survey of bio-corrosion rates of magnesium alloys. Corros. Sci. 2010, 52, 287–291. [Google Scholar] [CrossRef]
- Saji, V.S.; Choe, H.C. Electrochemical corrosion behaviour of nanotubular Ti–13Nb–13Zr alloy in Ringer’s solution. Corros. Sci. 2009, 51, 1658–1663. [Google Scholar] [CrossRef]
- Qadir, M.; Lin, J.; Biesiekierski, A.; Li, Y.; Wen, C. Effect of Anodized TiO2-Nb2O5-ZrO2 Nanotubes with Different Nanoscale Dimensions on the Biocompatibility of a Ti35Zr28Nb Alloy. ACS Appl. Mater. Interfaces 2020, 12, 6776–6787. [Google Scholar] [CrossRef] [PubMed]
- Chernozem, R.V.; Surmeneva, M.A.; Ignatov, V.P.; Peltek, O.O.; Goncharenko, A.A.; Muslimov, A.R.; Timin, A.S.; Tyurin, A.I.; Ivanov, Y.F.; Grandini, C.R.; et al. Comprehensive Characterization of Titania Nanotubes Fabricated on Ti-Nb Alloys: Surface Topography, Structure, Physicomechanical Behavior, and a Cell Culture Assay. ACS Biomater. Sci. Eng. 2020, 6, 1487–1499. [Google Scholar] [CrossRef]

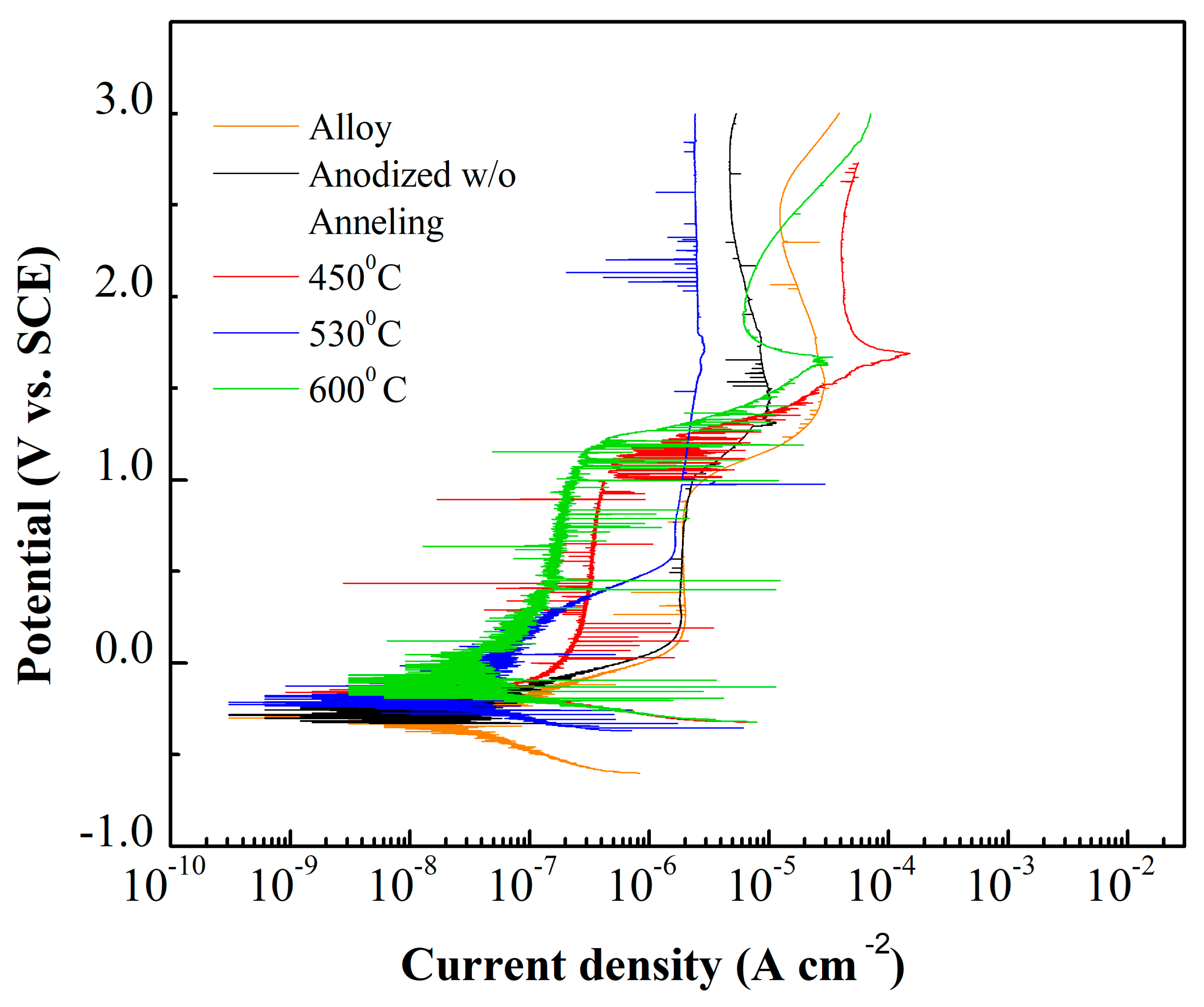
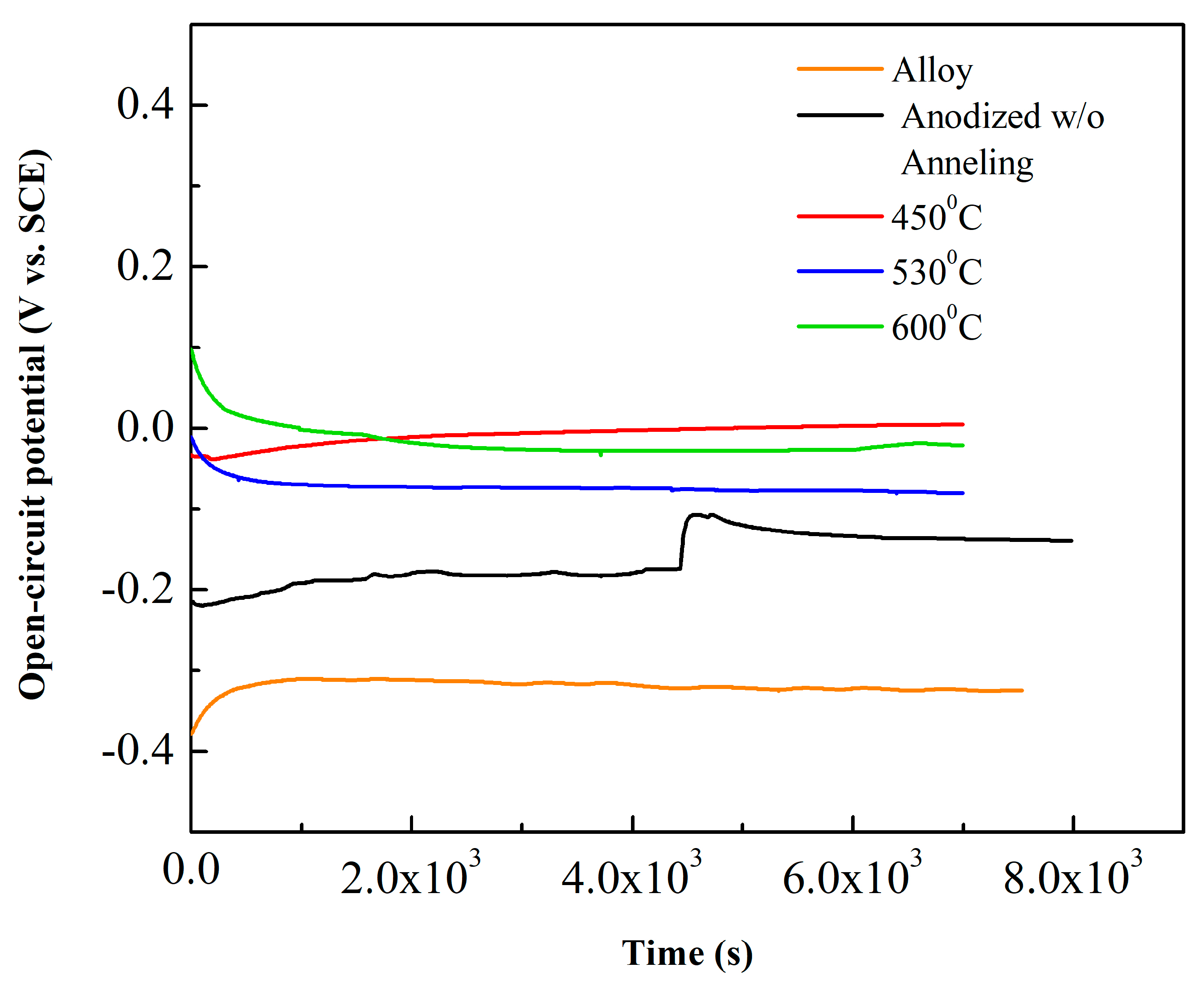
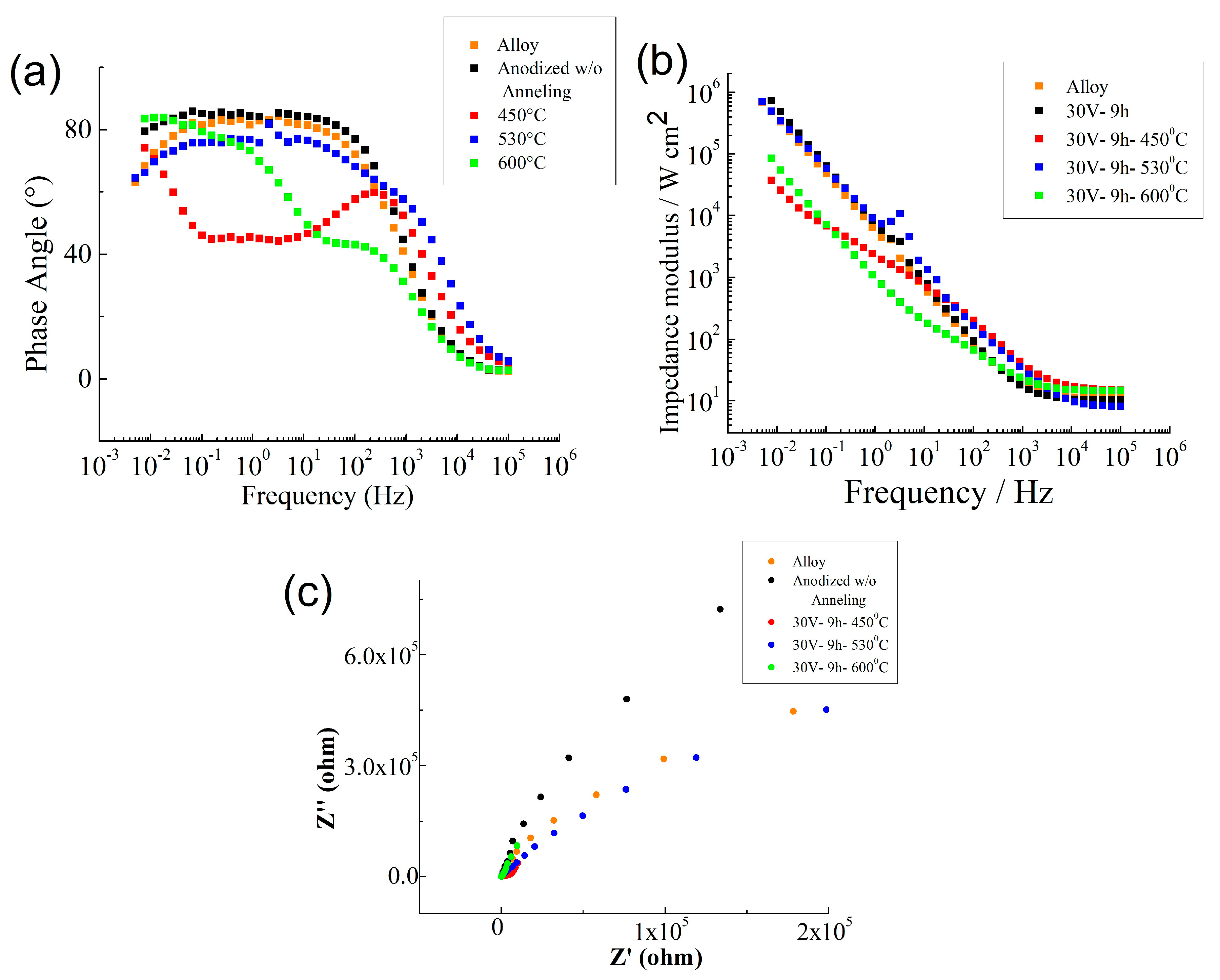
| Values | Impedance Modulus (Ω cm2) | Corrosion Current Density (A cm−2) | Corrosion Potential (V) |
|---|---|---|---|
| Alloy | 3.81 × 106 | 2.84 × 10−8 | −0.299 |
| Anodized w/o Anneling | 5.66 × 105 | 5.80 × 10−8 | −0.288 |
| 450 °C | 2.98 × 104 | 3.68 × 10−8 | −0.284 |
| 530 °C | 3.98 × 105 | 4.90 × 10−8 | −0.221 |
| 600 °C | 6.29 × 104 | 2.76 × 10−8 | −0.122 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Capellato, P.; Sachs, D.; Vilela, F.B.; Melo, M.M.; Silva, G.; Rodrigues, G.; Zavaglia, C.A.d.C.; Nakazato, R.Z.; Claro, A.P.R.A. Influence of Annealing Temperature on Corrosion Resistance of TiO2 Nanotubes Grown on Ti–30Ta Alloy. Metals 2020, 10, 1106. https://doi.org/10.3390/met10081106
Capellato P, Sachs D, Vilela FB, Melo MM, Silva G, Rodrigues G, Zavaglia CAdC, Nakazato RZ, Claro APRA. Influence of Annealing Temperature on Corrosion Resistance of TiO2 Nanotubes Grown on Ti–30Ta Alloy. Metals. 2020; 10(8):1106. https://doi.org/10.3390/met10081106
Chicago/Turabian StyleCapellato, Patricia, Daniela Sachs, Filipe Bueno Vilela, Mirian M. Melo, Gilbert Silva, Geovani Rodrigues, Cecilia A. de C. Zavaglia, Roberto Z. Nakazato, and Ana Paula R. A. Claro. 2020. "Influence of Annealing Temperature on Corrosion Resistance of TiO2 Nanotubes Grown on Ti–30Ta Alloy" Metals 10, no. 8: 1106. https://doi.org/10.3390/met10081106
APA StyleCapellato, P., Sachs, D., Vilela, F. B., Melo, M. M., Silva, G., Rodrigues, G., Zavaglia, C. A. d. C., Nakazato, R. Z., & Claro, A. P. R. A. (2020). Influence of Annealing Temperature on Corrosion Resistance of TiO2 Nanotubes Grown on Ti–30Ta Alloy. Metals, 10(8), 1106. https://doi.org/10.3390/met10081106