Microstructures and Tensile Fracture Behavior of 2219 Wrought Al–Cu Alloys with Different Impurity of Fe
Abstract
1. Introduction
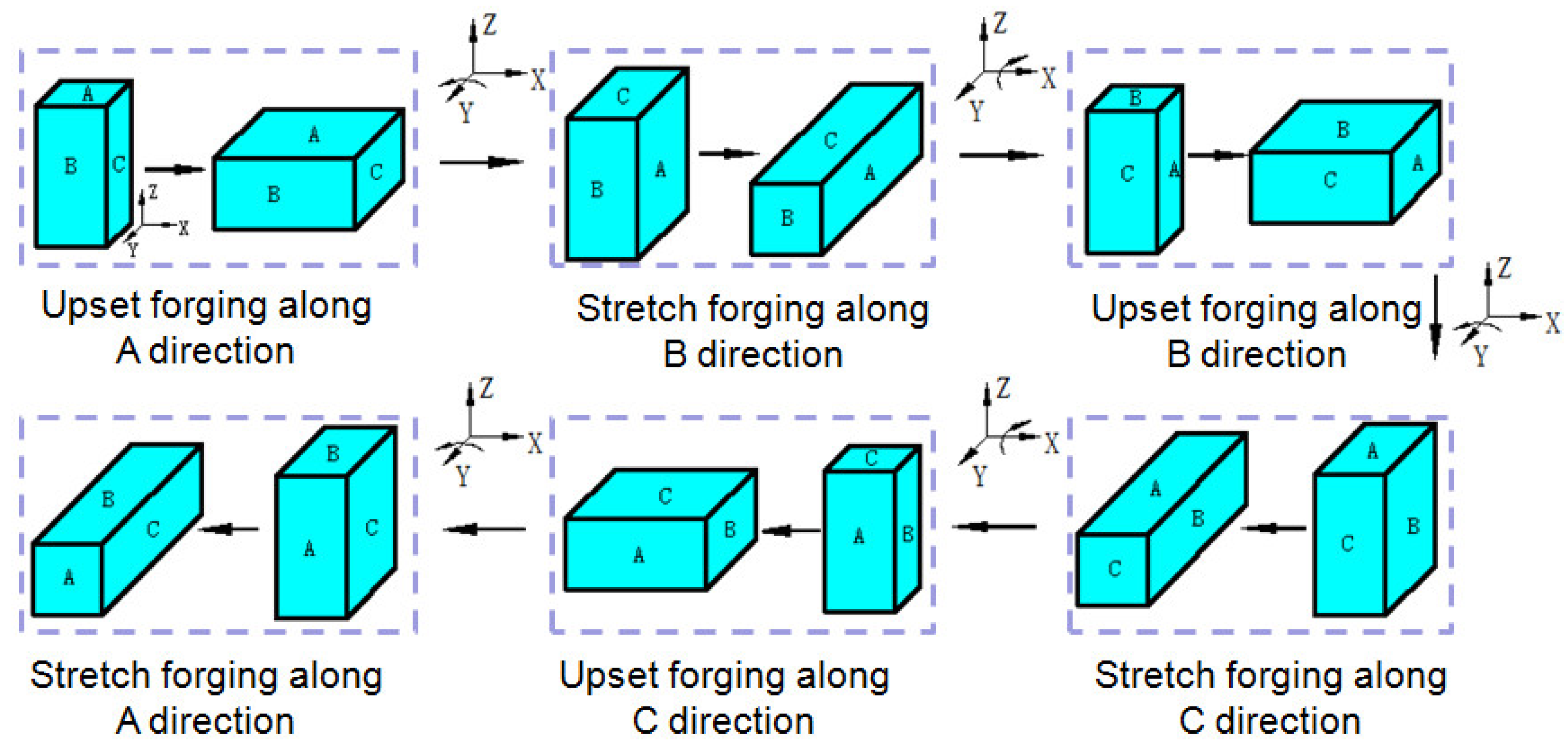
2. Materials and Methods
3. Results
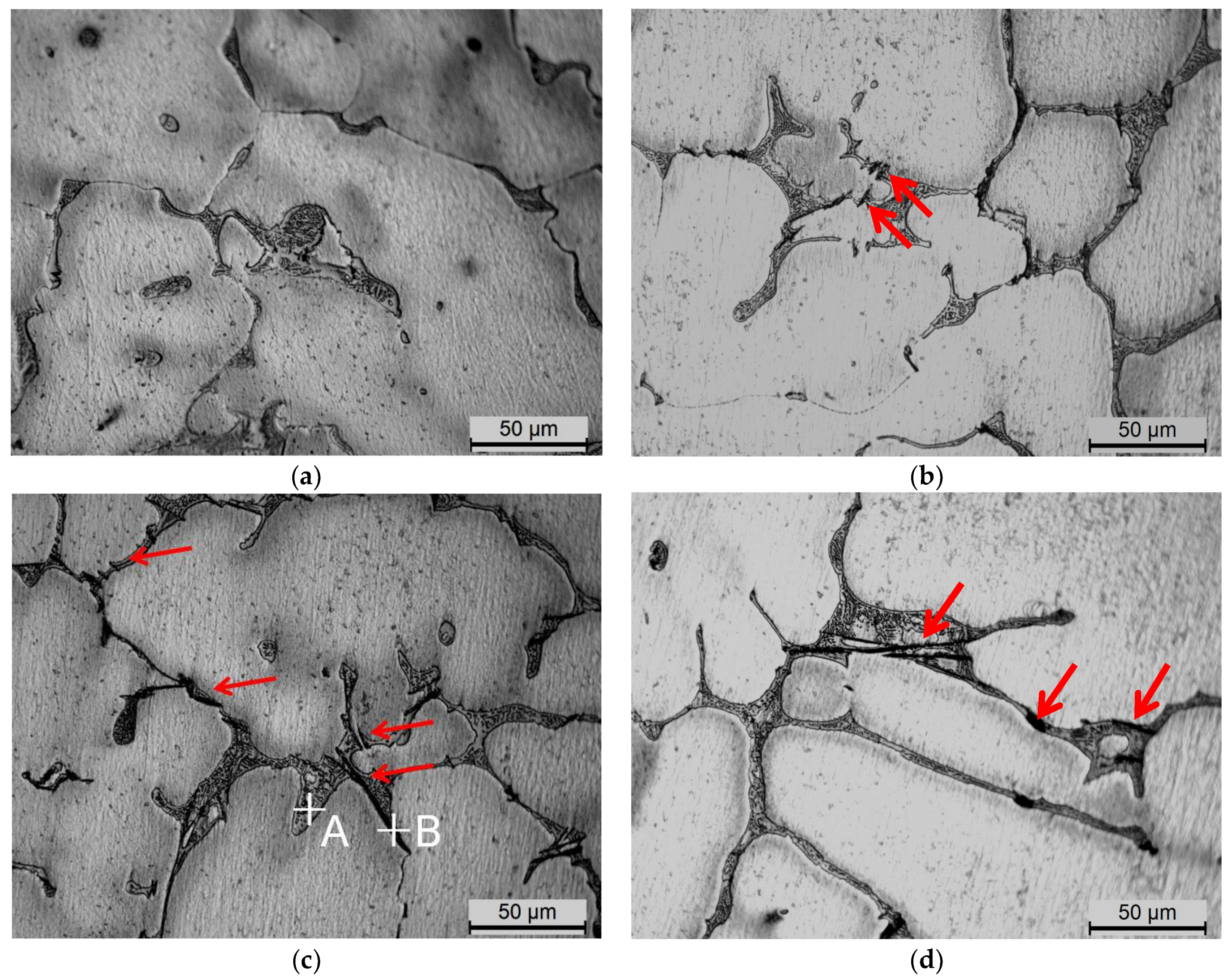
3.1. The Microstructure Evolution of Fe-Rich Intermetallics
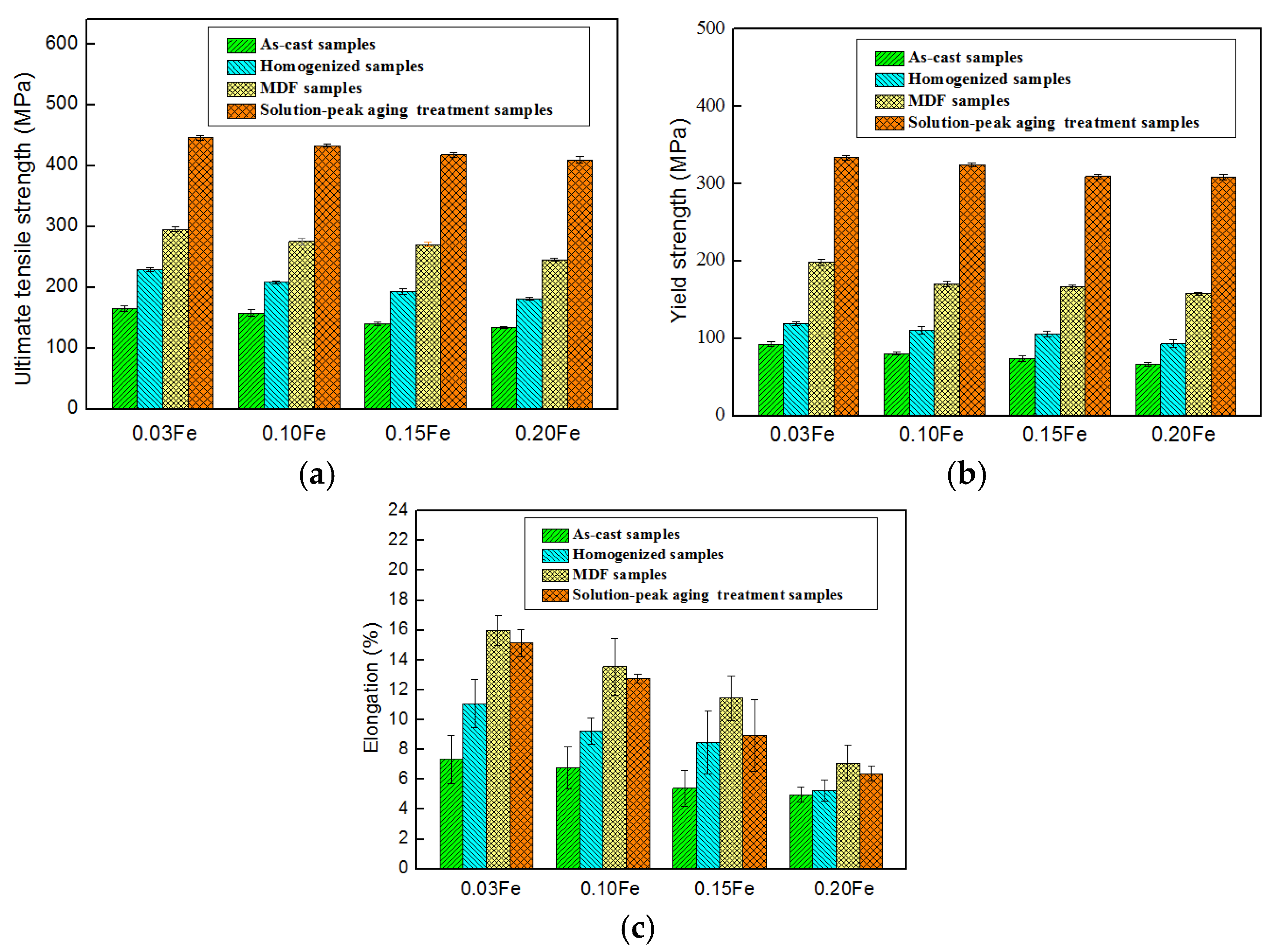
3.2. Mechanical Properties and Tensile Fracture Morphology
4. Discussion
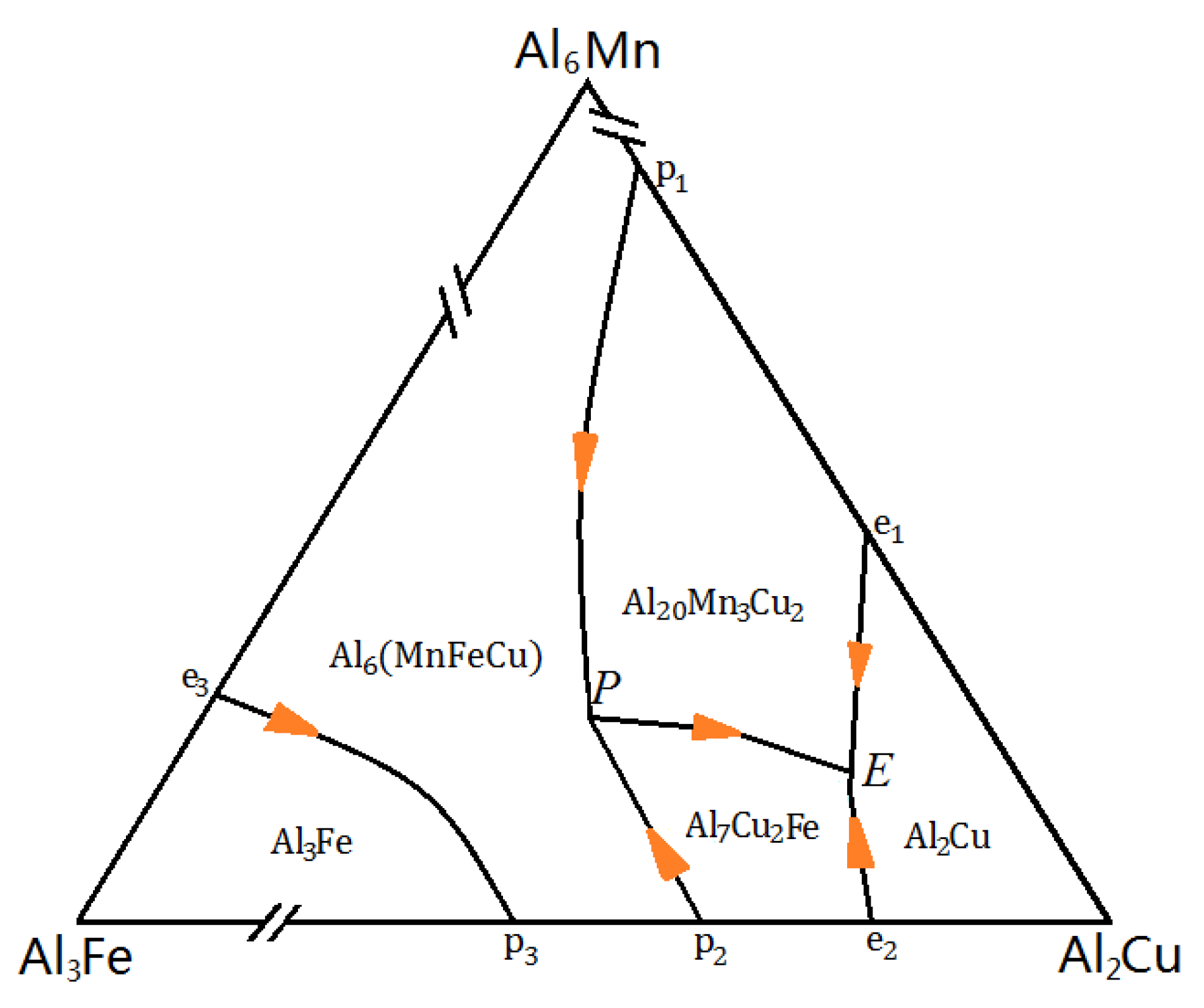
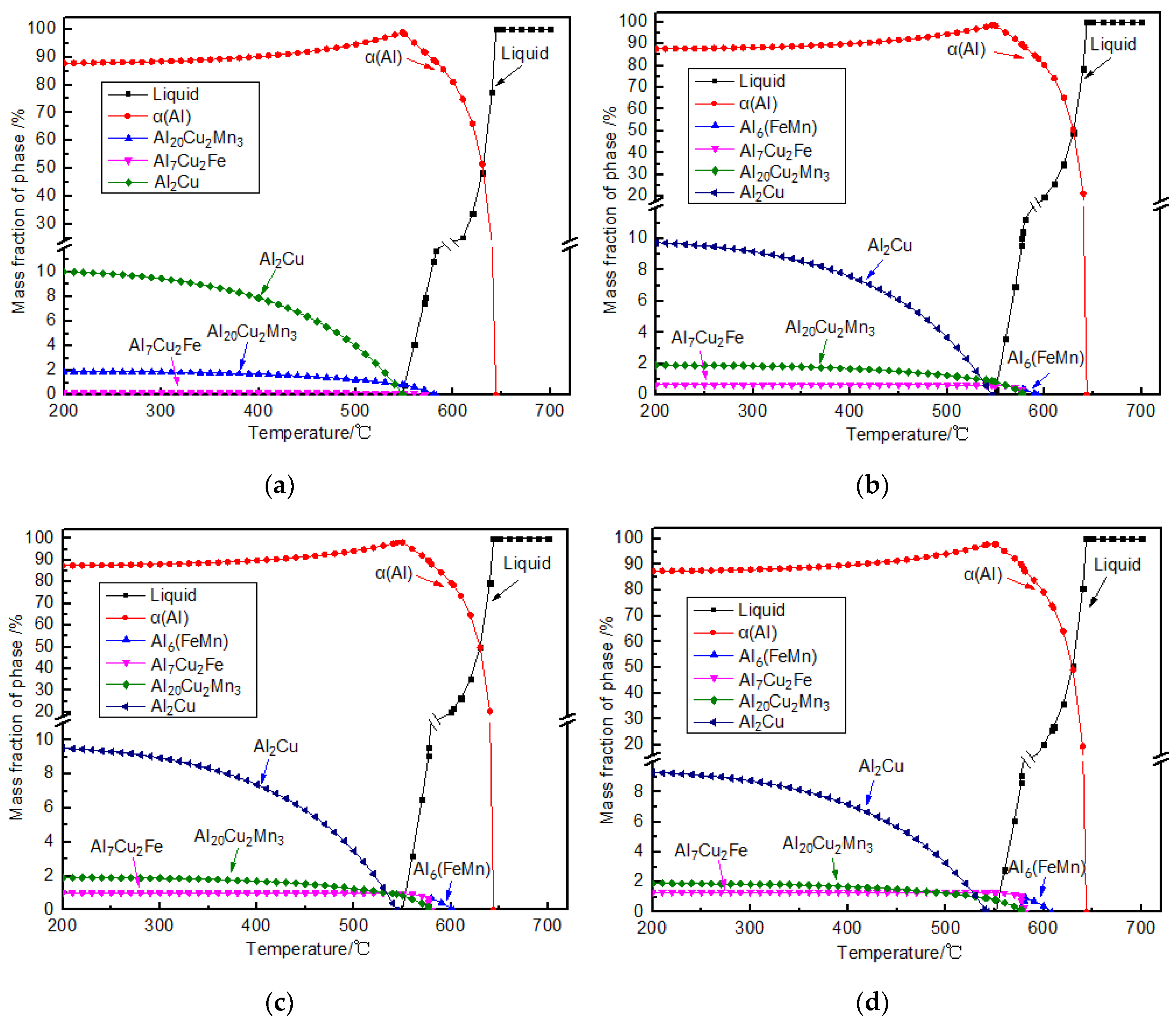
4.1. Fe-Rich Intermetallics Analysis
4.2. Effect of the Fe-Rich Intermetallic Particles on Tensile Fracture Behavior of 2219 Al–Cu Alloys
5. Conclusions
- (1)
- When the Fe content was less than 0.03 wt.%, the main constituents were Al2Cu intermetallics. As the Fe content increased to 0.10 wt.%, a new needle-like Al7Cu2Fe or Al7Cu2(Fe, Mn) phase presented. Further increase in the Fe content, the characteristic of the needle-like Al7Cu2Fe or Al7Cu2(Fe, Mn) intermetallics did not change, whereas their sizes became longer and wider.
- (2)
- The fragmented Al7Cu2Fe or Al7Cu2(Fe, Mn) intermetallics were obtained during multidirectional forging process. However, they were un-dissolved in the α-Al matrix in subsequent solution treatment due to the low tolerance of Fe in Al–Cu alloys. The sharp edges of the fragmented Al7Cu2Fe or Al7Cu2(FeMn) particles can act as crack initiators and then as crack propagation paths because they were subjected to higher stress concentrations during deformation.
- (3)
- For all the samples, the same trend of UTS, YS, and EL variation from the processes of as-cast to peak aging stage. The as-cast samples presented relatively low values of UTS/YS/EL, i.e., 165.35/92.56 MPa and 7.34%, 157.61/80.67 MPa and 6.79%, 140.29/74.44 MPa and 5.41%, and 133.77/66.52 MPa and 4.98% as a function of the Fe content ranging from 0.03 to 0.20 wt.%. The MDF samples possessed the maximum EL values, i.e., 15.99%, 13.56%, 11.46%, and 7.09% corresponding to the Fe contents of 0.03, 0.10, 0.15, and 0.20 wt.%, respectively. The solution-peak aging treatment significantly increased the UTS/YS values by at least 270/90 MPa, respectively, compared with the as-cast condition.
- (4)
- For peak aging condition, the UTS, YS, and EL values decreased with the increase of Fe content. For 0.03 wt.% Fe alloy, the UTS, YS, and EL values were 445.64 MPa, 333.76 MPa, and 15.14%, respectively. Increasing the Fe content from 0.03 to 0.20 wt.%, the UTS, YS, and EL decreased by 36 MPa, 25 MPa, and 57.92%, respectively.
Author Contributions
Funding
Conflicts of Interest
References
- He, H.; Yi, Y.-P.; Huang, S.-Q.; Zhang, Y. Effects of deformation temperature on second-phase particles and mechanical properties of 2219 Al-Cu alloy. Mater. Sci. Eng. A 2018, 712, 414–423. [Google Scholar] [CrossRef]
- Xu, D.F.; Chen, K.H.; Chen, Y.Q.; Chen, S.Y. Evolution of the second-phase particles and their effect on tensile fracture behavior of 2219 Al-xCu alloys. Metals 2020, 10, 197. [Google Scholar] [CrossRef]
- Dong, F.; Yi, Y.; Huang, C.; Huang, S. Influence of cryogenic deformation on second-phase particles, grain structure, and mechanical properties of Al–Cu–Mn alloy. J. Alloys Compd. 2020, 827, 154300. [Google Scholar] [CrossRef]
- Backerud, L.; Chai, G.; Tamminen, J. Solidification Characteristics of Aluminum Alloys; American Foundrymen’s Society: Schaumburg, IL, USA, 1990; p. 266. [Google Scholar]
- Zhang, W.; Lin, B.; Zhang, D.; Li, Y. Microstructures and mechanical properties of squeeze cast Al–5.0Cu–0.6Mn alloys with different Fe content. Mater. Des. 2013, 52, 225–233. [Google Scholar] [CrossRef]
- Liu, K.; Cao, X.; Chen, X.-G. Solidification of Iron-Rich Intermetallic Phases in Al-4.5Cu-0.3Fe Cast Alloy. Met. Mater. Trans. A 2010, 42, 2004–2016. [Google Scholar] [CrossRef]
- Tseng, C.-J.; Lee, S.-L.; Wu, T.-F.; Lin, J.-C. Effects of Fe Content on Microstructure and Mechanical Properties of A206 Alloy. Mater. Trans. JIM 2000, 41, 708–713. [Google Scholar] [CrossRef]
- Lin, B.; Xu, R.; Li, H.; Xiao, H.; Zhang, W.; Li, S. Development of high Fe content squeeze cast 2A16 wrought Al alloys with enhanced mechanical properties at room and elevated temperatures. Mater. Charact. 2018, 142, 389–397. [Google Scholar] [CrossRef]
- Tseng, C.-J.; Lee, S.-L.; Tsai, S.-C.; Cheng, C.-J. Effects of manganese on microstructure and mechanical properties of A206 alloys containing iron. J. Mater. Res. 2002, 17, 2243–2250. [Google Scholar] [CrossRef]
- Liu, K.; Cao, X.; Chen, X.-G. Effect of Mn, Si, and Cooling Rate on the Formation of Iron-Rich Intermetallics in 206 Al-Cu Cast Alloys. Met. Mater. Trans. A 2012, 43, 1231–1240. [Google Scholar] [CrossRef]
- Xu, R.; Lin, B.; Li, H.Y.; Xiao, H.Q.; Zhao, Y.L.; Zhang, W.W. Microstructure evolution and mechanical properties of Al-6.5Cu-0.6Mn-0.5Fe alloys with different Si additions. Trans. Nonferrous Met. Soc. China 2019, 29, 1583–1591. [Google Scholar] [CrossRef]
- Zhang, W.; Lin, B.; Fan, J.; Zhang, D.; Li, Y. Microstructures and mechanical properties of heat-treated Al–5.0Cu–0.5Fe squeeze cast alloys with different Mn/Fe ratio. Mater. Sci. Eng. A 2013, 588, 366–375. [Google Scholar] [CrossRef]
- Liu, Y.; Luo, L.; Han, C.; Ou, L.; Wang, J.; Liu, C. Effect of Fe, Si and Cooling Rate on the Formation of Fe- and Mn-rich Intermetallics in Al–5Mg–0.8Mn Alloy. J. Mater. Sci. Technol. 2016, 32, 305–312. [Google Scholar] [CrossRef]
- Lin, Y.; Wu, B.; Li, S.; Mao, S.; Liu, X.; Zhang, Y.; Wang, L. The quantitative relationship between microstructure and mechanical property of a melt spun Al–Mg alloy. Mater. Sci. Eng. A 2015, 621, 212–217. [Google Scholar] [CrossRef]
- Lichioiu, I.; Peter, I.; Varga, B.; Rosso, M. Preparation and Structural Characterization of Rapidly Solidified Al–Cu Alloys. J. Mater. Sci. Technol. 2014, 30, 394–400. [Google Scholar] [CrossRef]
- Chobaut, N.; Carron, D.; Drezet, J.-M. Characterisation of precipitation upon cooling of an AA2618 Al–Cu–Mg alloy. J. Alloys Compd. 2016, 654, 56–62. [Google Scholar] [CrossRef]
- Lin, B.; Zhang, W.; Zhao, Y.; Li, Y. Solid-state transformation of Fe-rich intermetallic phases in Al–5.0Cu–0.6Mn squeeze cast alloy with variable Fe contents during solution heat treatment. Mater. Charact. 2015, 104, 124–131. [Google Scholar] [CrossRef]
- Lin, B.; Zhang, W.W. Effect of heat treatment on morphology of Fe-rich intermetallics in Al–Cu alloys. Mater. Sci. Technol. 2016, 33, 738–743. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, R.; Chen, P.; Li, X.; Liu, Z. Microstructural evolution of Al2Cu phase and mechanical properties of the large-scale Al alloy components under different consecutive manufacturing processes. J. Alloys Compd. 2019, 808, 151634. [Google Scholar] [CrossRef]
- Zhang, L.; Li, X.; Li, R.; Jiang, R.; Zhang, L. Effects of high-intensity ultrasound on the microstructures and mechanical properties of ultra-large 2219 Al alloy ingot. Mater. Sci. Eng. A 2019, 763, 138–154. [Google Scholar] [CrossRef]
- Liu, Z.; Li, R.; Jiang, R.; Zhang, L.; Li, X. Scalable Ultrasound-Assisted Casting of Ultra-large 2219 Al Alloy Ingots. Met. Mater. Trans. A 2019, 50, 1146–1152. [Google Scholar] [CrossRef]
- Peng, H.; Li, R.; Li, X.; Ding, S.; Chang, M.; Liao, L.; Zhang, Y.; Chen, P. Effect of Multi-Source Ultrasonic on Segregation of Cu Elements in Large Al–Cu Alloy Cast Ingot. Materials 2019, 12, 2828. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Yi, Y.-P.; Huang, S.-Q.; Zhang, Y. Effects of cold predeformation on dissolution of second-phase Al2Cu particles during solution treatment of 2219 Al–Cu alloy forgings. Mater. Charact. 2018, 135, 18–24. [Google Scholar] [CrossRef]
- Lu, Y.; Wang, J.; Li, X.; Li, W.; Li, R.; Zhou, D. Effects of pre-deformation on the microstructures and corrosion behavior of 2219 aluminum alloys. Mater. Sci. Eng. A 2018, 723, 204–211. [Google Scholar] [CrossRef]
- Belov, N.A.; Eskin, D.G.; Aksenov, A.A. Multicomponent Phase Diagrams, Applications for Commercial Aluminum Alloys; Elsevier: London, UK, 2005. [Google Scholar]
- Tang, K.; Du, Q.; Li, Y. Modelling microstructure evolution during casting, homogenization and ageing heat treatment of Al-Mg-Si-Cu-Fe-Mn alloys. Calphad 2018, 63, 164–184. [Google Scholar] [CrossRef]
- Li, Y.; Arnberg, L. A eutectoid phase transformation for the primary intermetallic particle from Alm(Fe,Mn) to Al3(Fe,Mn) in AA5182 alloy. Acta Mater. 2004, 52, 2945–2952. [Google Scholar] [CrossRef]
- Sharma, V.; Kumar, K.S.; Rao, B.N.; Pathak, S. Effect of microstructure and strength on the fracture behavior of AA2219 alloy. Mater. Sci. Eng. A 2009, 502, 45–53. [Google Scholar] [CrossRef]
- Elgallad, E.M.; Zhang, Z.; Chen, X.-G. Effect of two-step aging on the mechanical properties of AA2219 DC cast alloy. Mater. Sci. Eng. A 2015, 625, 213–220. [Google Scholar] [CrossRef]
- An, L.; Cai, Y.; Liu, W.; Yuan, S.; Zhu, S.; Meng, F. Effect of pre-deformation on microstructure and mechanical properties of 2219 aluminum alloy sheet by thermomechanical treatment. Trans. Nonferrous Met. Soc. China 2012, 22, 370–375. [Google Scholar] [CrossRef]
- Liu, L.; Wu, Y.; Gong, H.; Dong, F.; Ahmad, A. Modified kinetic model for describing continuous dynamic recrystallization behavior of Al 2219 alloy during hot deformation process. J. Alloys Compd. 2020, 817, 153301. [Google Scholar] [CrossRef]
- Pugh, S. Relations between the elastic moduli and the plastic properties of polycrystalline pure metals. Philos. Mag. 1954, 45, 823–843. [Google Scholar] [CrossRef]
- Li, C.M.; Zeng, S.M.; Chen, Z.Q.; Cheng, N.P.; Chen, T.X. First-principles calculations of elastic and thermodynamic properties of the four main intermetallic phases in Al–Zn–Mg–Cu alloys. Comput. Mater. Sci. 2014, 93, 210–220. [Google Scholar] [CrossRef]
- Tian, J.; Yu-Hong, Z.; Hou, H.; Han, P. First-principles investigation of the structural, mechanical and thermodynamic properties of Al 2 Cu phase under various pressure and temperature conditions. Solid State Commun. 2017, 268, 44–50. [Google Scholar] [CrossRef]
- Xie, D.; Guo, H.; Yang, X.; Ma, T. First-principles study of structural, mechanical, and electronic properties of typical iron-containing phases in Al-Cu alloys under different pressures. Phys. B Condens. Matter 2019, 555, 112–117. [Google Scholar] [CrossRef]
- Tian, J.; Yu-Hong, Z.; Wen, Z.; Hou, H.; Han, P. Physical properties and Debye temperature of Al 7 Cu 2 Fe alloy under various pressures analyzed by first-principles. Solid State Commun. 2017, 257, 6–10. [Google Scholar] [CrossRef]






| Sample No. | Cu | Mn | Fe | Si | Mg | V | Zr | Al |
|---|---|---|---|---|---|---|---|---|
| 0.03 wt.% Fe | 5.87 | 0.362 | 0.026 | <0.0005 | ≤0.02 | 0.070 | 0.138 | Bal. |
| 0.10 wt.% Fe | 5.90 | 0.359 | 0.101 | <0.0005 | ≤0.02 | 0.068 | 0.136 | Bal. |
| 0.15 wt.% Fe | 5.89 | 0.361 | 0.147 | <0.0005 | ≤0.02 | 0.036 | 0.130 | Bal. |
| 0.20 wt.% Fe | 5.88 | 0.362 | 0.195 | <0.0005 | ≤0.02 | 0.059 | 0.135 | Bal. |
| Point | Elements | |||
|---|---|---|---|---|
| Al | Cu | Fe | Mn | |
| A | 67.96 | 32.04 | - | - |
| B | 71.01 | 20.62 | 6.64 | 1.73 |
| C | 73.28 | 17.42 | 7.61 | 1.69 |
| D | 72.54 | 27.46 | - | - |
| Reactions | T (°C) |
|---|---|
| L→α-Al | 651–648 |
| L→α-Al+Al6(FeMn) | 608–577 |
| L+ Al6(FeMn)→α-Al+Al20Mn3Cu2+Al7Cu2Fe | 597–576 |
| L→α-Al +Al20Mn3Cu2+Al7Cu2Fe | 587–537 |
| L→α-Al+Al2Cu+Al20Mn3Cu2+Al7Cu2Fe | 547–540 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, D.; Zhu, C.; Xu, C.; Chen, K. Microstructures and Tensile Fracture Behavior of 2219 Wrought Al–Cu Alloys with Different Impurity of Fe. Metals 2021, 11, 174. https://doi.org/10.3390/met11010174
Xu D, Zhu C, Xu C, Chen K. Microstructures and Tensile Fracture Behavior of 2219 Wrought Al–Cu Alloys with Different Impurity of Fe. Metals. 2021; 11(1):174. https://doi.org/10.3390/met11010174
Chicago/Turabian StyleXu, Daofen, Changjun Zhu, Chengfu Xu, and Kanghua Chen. 2021. "Microstructures and Tensile Fracture Behavior of 2219 Wrought Al–Cu Alloys with Different Impurity of Fe" Metals 11, no. 1: 174. https://doi.org/10.3390/met11010174
APA StyleXu, D., Zhu, C., Xu, C., & Chen, K. (2021). Microstructures and Tensile Fracture Behavior of 2219 Wrought Al–Cu Alloys with Different Impurity of Fe. Metals, 11(1), 174. https://doi.org/10.3390/met11010174




