Comparative Study on Electronic Structure and Optical Properties of α-Fe2O3, Ag/α-Fe2O3 and S/α-Fe2O3
Abstract
1. Introduction
2. Computational Methods and Models
2.1. Computational Methods
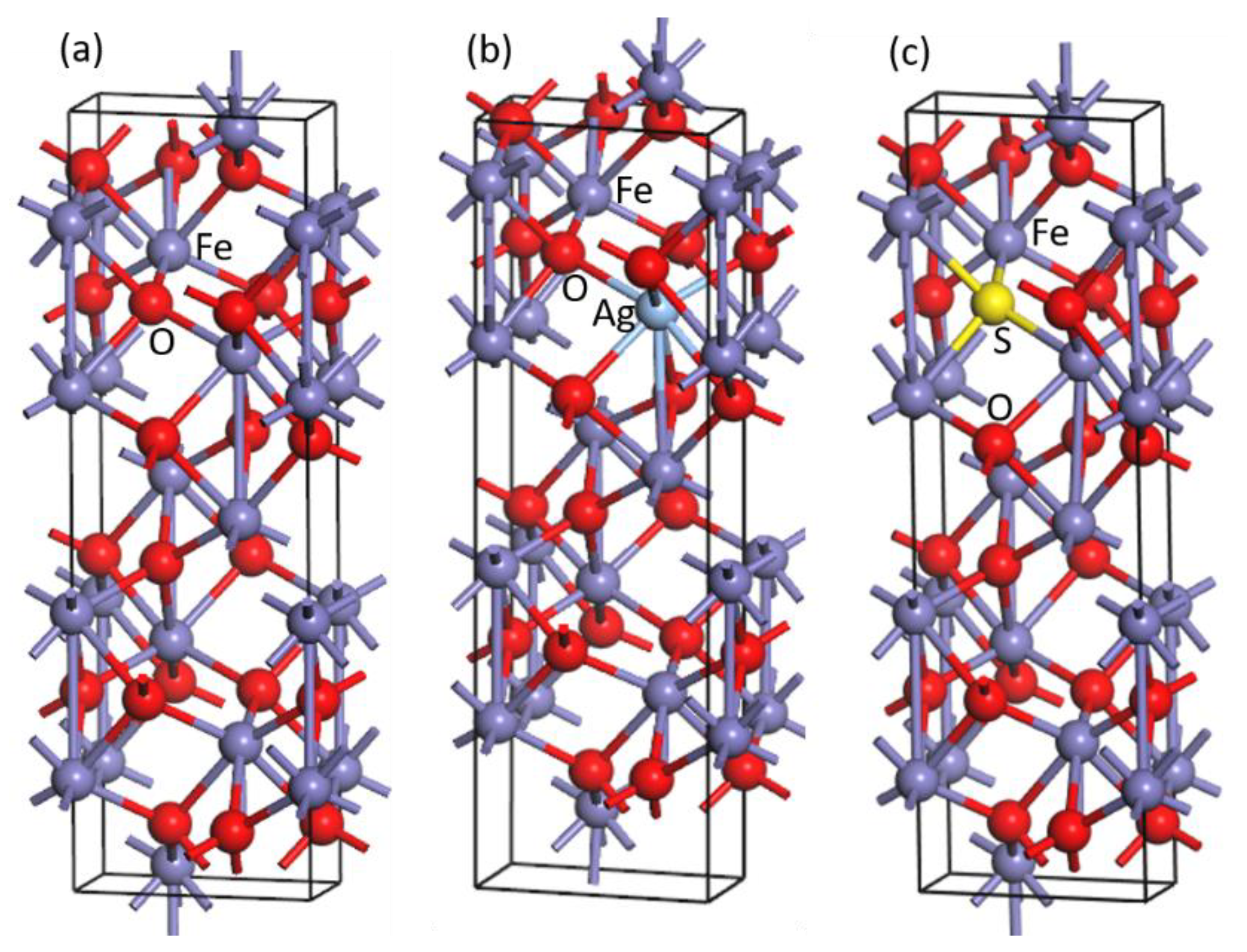
2.2. Computational Models
3. Results and Discussion
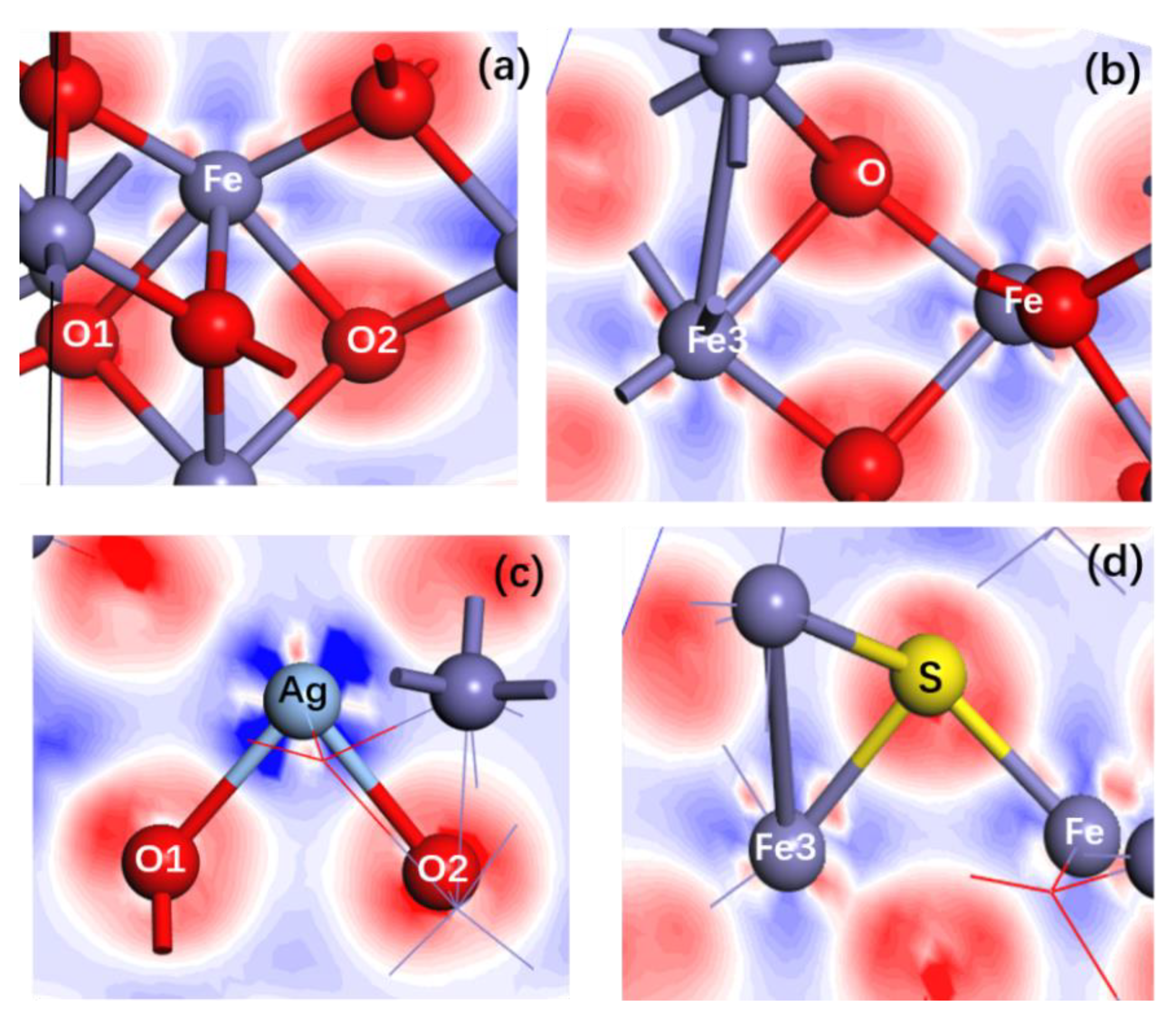
3.1. Structural Changes of Ag- and S-Doped α-Fe2O3
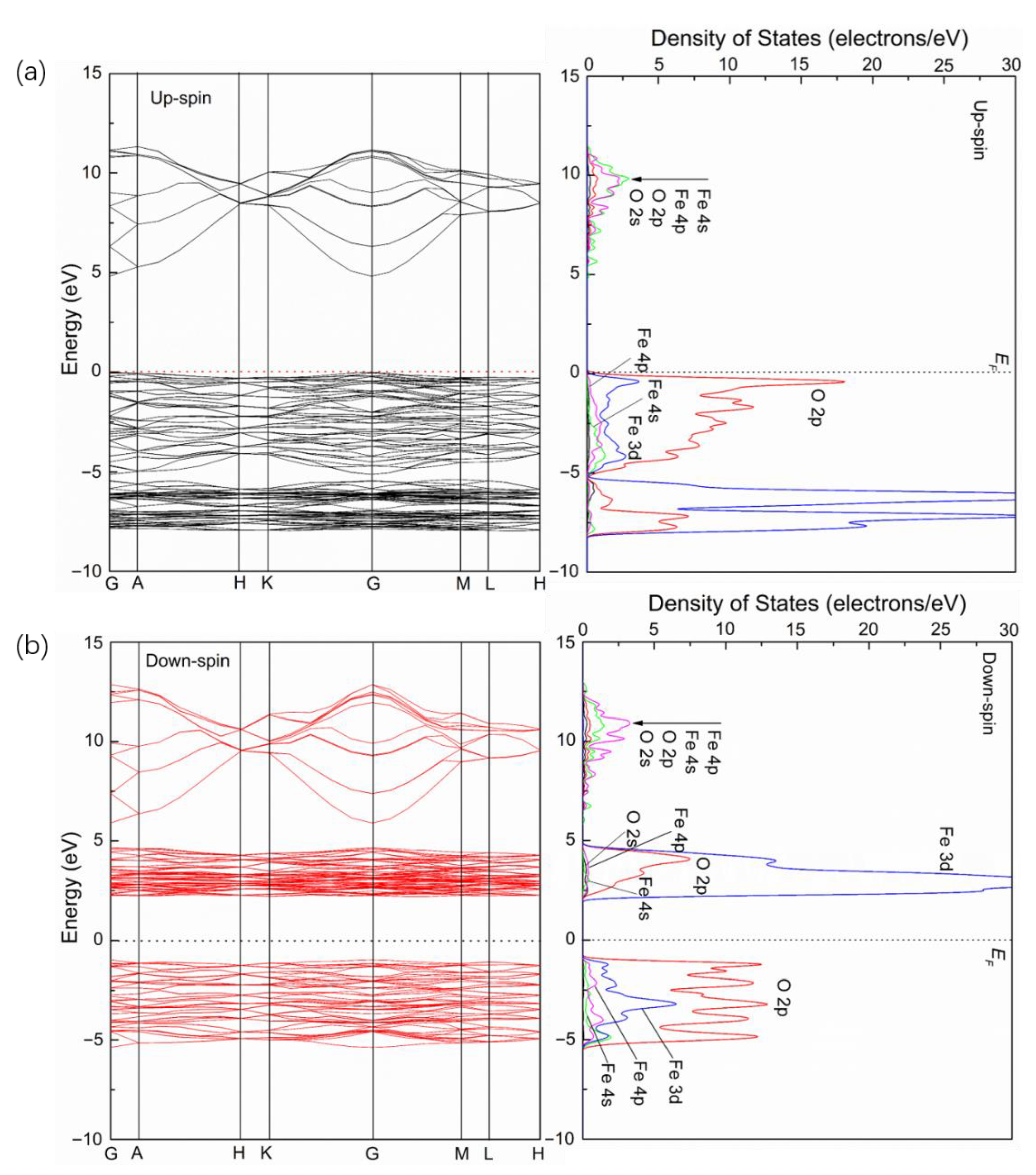
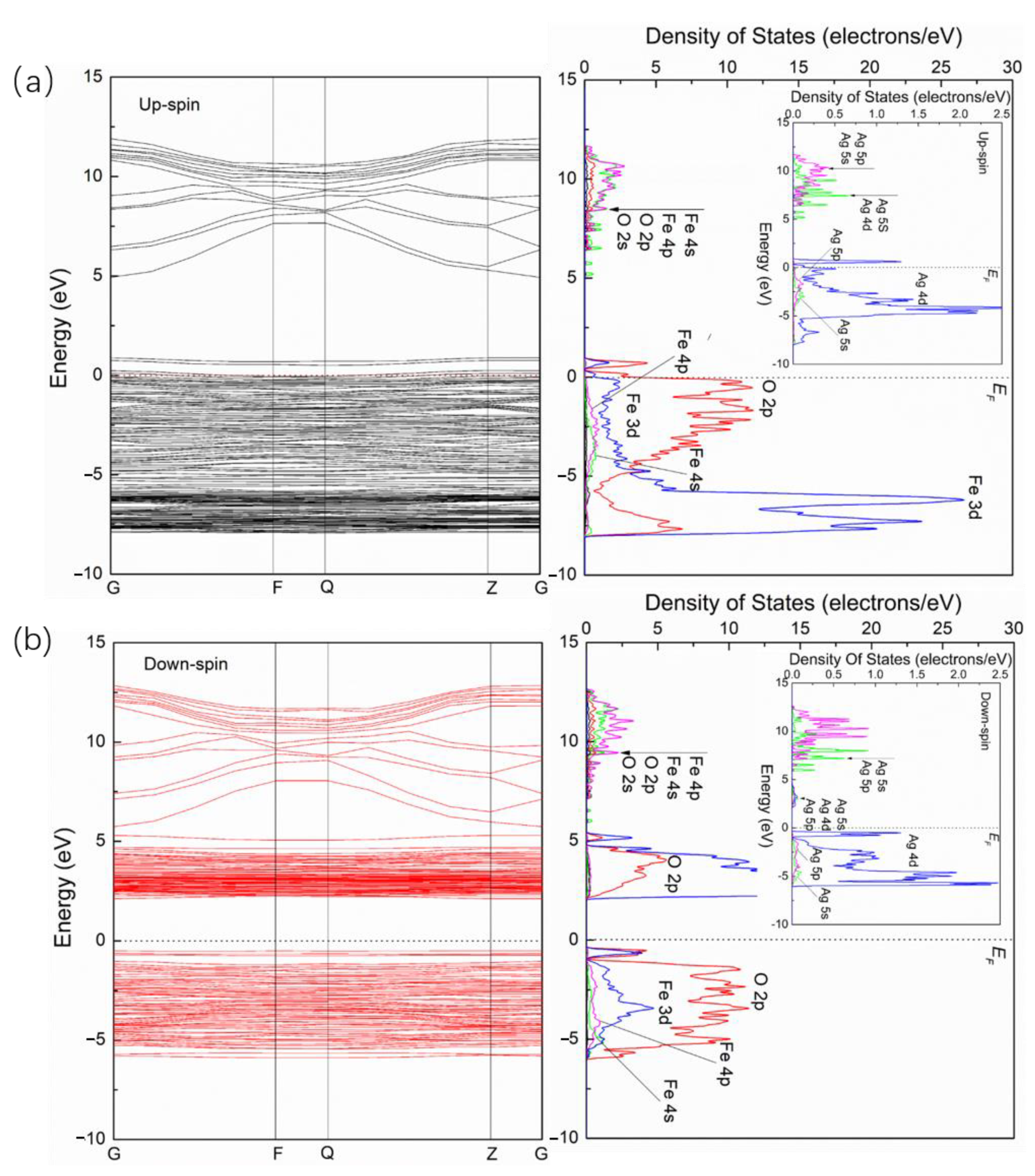
3.2. Electronic Structures of Pure, Ag- Doped and S-Doped α-Fe2O3
3.3. Optical Property of Pure, Ag-Doped and S-Doped α-Fe2O3
4. Conclusions
- (1)
- The doping of Ag and S results in a large change of the crystal structure of α-Fe2O3. The lengths of all Ag-O and S-Fe bonds are larger than those of corresponding O-Fe bonds before Ag and S doping, which induces the expansion of the α-Fe2O3 crystal.
- (2)
- The band gaps of α-Fe2O3 decrease after Ag and S doping. For Ag/α-Fe2O3, the energy levels near the Fermi level for the upspin and downspin bands are split and form small DOS peaks, respectively. The main contribution of Ag in the conduction band is from Ag 5p and Ag 5s orbitals and from Ag 4d orbital in the valence band. For S/α-Fe2O3, the energy levels near the Fermi level for the upspin and downspin bands are also split; however, two DOS peaks are formed, respectively. The main contribution of S is from S 3p with a few contributions of S 3s.
- (3)
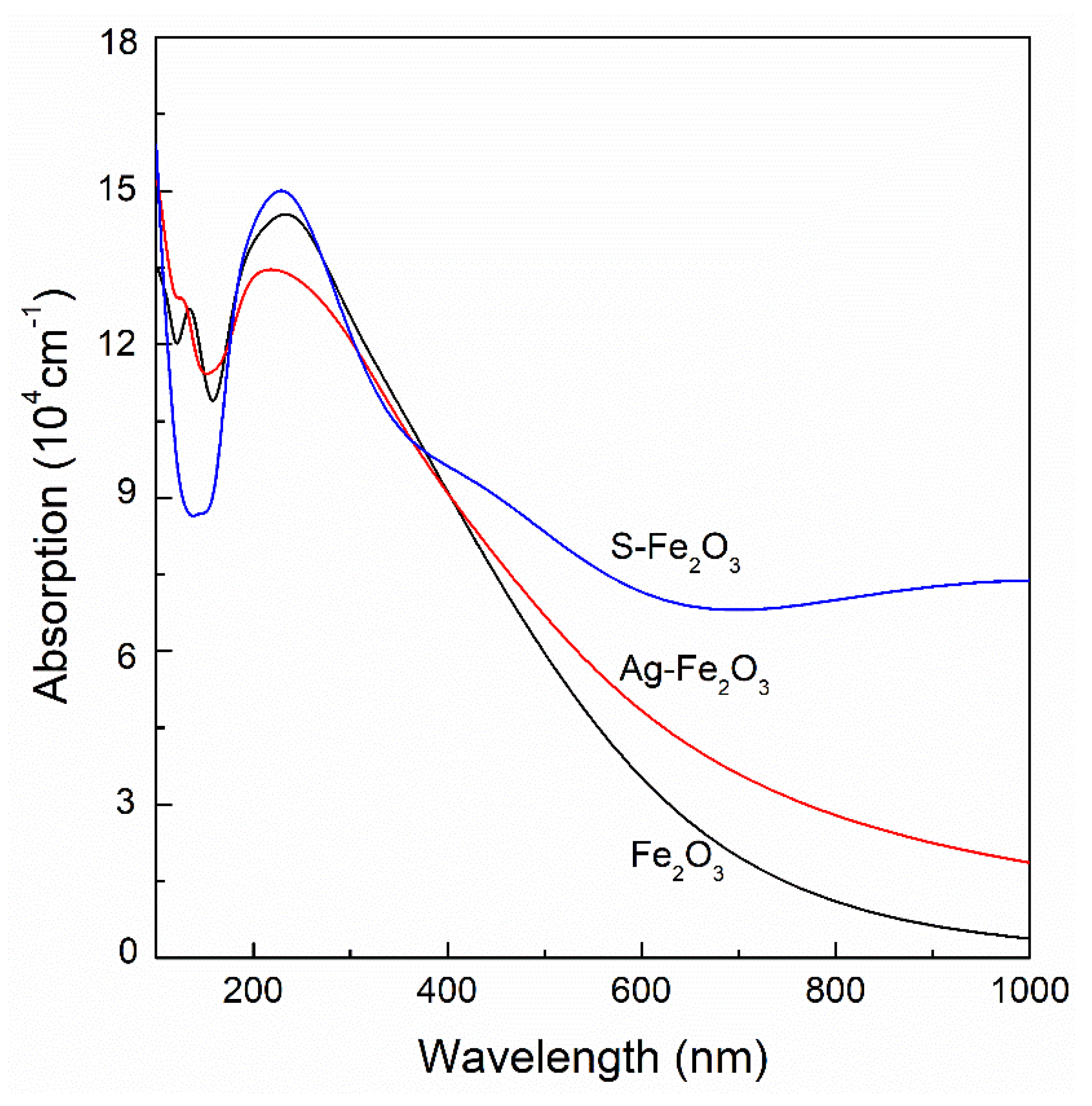
- The absorption peaks of Ag-doped and S-doped α-Fe2O3 shift slightly to a short wavelength accompanying the increased peak intensity of S/α-Fe2O3 and decreased peak intensity of Ag/α-Fe2O3. When the wavelength is greater than 380 nm, the optical absorptions of Ag- and S-doped α-Fe2O3 in the visible range are stronger than that of pure α-Fe2O3, and the optical absorption of S-doped α-Fe2O3 is stronger than that of Ag-doped α-Fe2O3.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ewing, T.; Ha, P.T.; Beyenal, H. Evaluation of long-term performance of sediment microbial fuel cells and the role of natural resources. Appl. Energy 2017, 192, 490–497. [Google Scholar] [CrossRef]
- Fujishima, A.; Honda, K. Electrochemical Photolysis of Water at a Semiconductor Electrode. Nature 1972, 238, 37–38. [Google Scholar] [CrossRef] [PubMed]
- Wei, T.; Zhu, Y.N.; An, X.; Liu, L.M.; Cao, X.; Liu, H.; Qu, J. Defect modulation of z-scheme TiO2/Cu2O photocatalysts for durable water splitting. ACS Catal. 2019, 9, 8346–8354. [Google Scholar] [CrossRef]
- Dong, Z.; Ding, D.; Li, T.; Ning, C. Ni-doped TiO2 nanotubes photoanode for enhanced photoelectrochemical water splitting, Appl. Surf. Sci. 2018, 443, 321–328. [Google Scholar] [CrossRef]
- Wei, Y.; Wang, J.; Yu, R.; Wan, J.; Wang, D. Constructing SrTiO3-TiO2 heterogeneous hollow multi-shelled structures for enhanced solar water splitting. Angew. Chem. Int. Ed. 2019, 58, 1422–1426. [Google Scholar] [CrossRef]
- Barroso, M.; Mesa, C.A.; Pendlebury, S.R.; Cowan, A.J.; Hisatomi, T.; Sivula, K.; Graetzel, M.; Klug, D.R.; Durrant, J.R. Dynamics of photogenerated holes in surface modified α-Fe2O3 photoanodes for solar water splitting. Proc. Natl. Acad. Sci. USA 2019, 109, 15640–15645. [Google Scholar] [CrossRef] [PubMed]
- Al-Ku Haili, M.F.; Saleem, M.; Durrani, S.M.A. Optical properties of iron oxide (α-Fe2O3) thin films deposited by the reactive evaporation of iron. J. Alloy. Compd. 2012, 521, 178–182. [Google Scholar] [CrossRef]
- Hung, W.H.; Peng, C.J.; Yang, C.R.; Li, C.J.; Shyue, J.J.; Chang, P.C.; Tseng, C.M.; Juan, P.C. Exploitation of a spontaneous spatial charge separation effect in plasmonic polyhedral α-Fe2O3 nanocrystal photoelectrodes for hydrogen production. Nano Energy 2016, 30, 523–530. [Google Scholar] [CrossRef]
- Leduc, J.; Goenuellue, Y.; Ghamgosar, P.; You, S.; Mouzon, J.; Choi, H.; Vomiero, A.; Grosch, M.; Mathur, S. Electronically-coupled phase boundaries in α-Fe2O3/Fe3O4 nanocomposite photoanodes for enhanced water oxidation. ACS Appl. Nano Mater. 2019, 2, 334–342. [Google Scholar] [CrossRef]
- Wanaguru, P.; An, J.; Zhang, Q. DFT+U study of ultrathin α-Fe2O3 nanoribbons from (110) and (104) surfaces. J. Appl. Phys. 2016, 119, 084302. [Google Scholar] [CrossRef]
- Li, L.; Liu, C.; Qiu, Y.; Mitsuzak, N.; Chen, Z. The influence of the hydrothermal temperature and time on morphology and photoelectrochemical response of α-Fe2O3 photoanode. J. Alloy. Compd. 2017, 969, 980–987. [Google Scholar] [CrossRef]
- Balbuena, J.; Cruz-yusta, M.; Pastor, A.; Sánchez, L. α-Fe2O3/SiO2 composites for the enhanced photocatalytic NO oxidation. J. Alloy. Compd. 2018, 735, 1553–1561. [Google Scholar] [CrossRef]
- Dare-Edwards, M.P.; Goodenough, J.B.; Hamnett, A.; Trevellick, P.R. Electrochemistry and photoelectrochemistry of iron(III) oxide. J. Chem. Soc. Faraday Trans. 1983, 79, 2027–2041. [Google Scholar] [CrossRef]
- Marschall, R. Semiconductor composites: Strategies for enhancing charge carrier separation to improve photocatalytic activity. Adv. Funct. Mater. 2014, 24, 2421–2440. [Google Scholar] [CrossRef]
- Cong, Y.; Chen, M.; Xu, T.; Zhang, Y.; Wang, Q. Tantalum and aluminum co-doped iron oxide as a robust photocatalyst for water oxidation. Appl. Catal. B Environ. 2014, 147, 733–740. [Google Scholar] [CrossRef]
- Ordal, M.A.; Long, L.L.; Bell, R.J.; Bell, S.E.; Bell, R.R. Optical properties of the metals Al, Co, Cu, Au, Fe, Pb, Ni, Pd, Pt, Ag, Ti, and W in the infrared and far infrared. Appl. Opt. 1983, 22, 1099–1119. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wang, G.; Chen, D.; Lv, X.; Li, J. Tuning photoelectrochemical performances of Ag-TiO2 nanocomposites via reduction/oxidation of Ag. Chem. Mater. 2008, 77, 87–95. [Google Scholar]
- Zhang, Y.; Jiang, S.; Song, W.; Zhou, P.; Ji, H.; Ma, W.; Hao, W.; Chen, C.; Zhao, J. Nonmetal P-doped hematite photoanode with enhanced electron morbility and high water oxidation activity. Energy Environ. Sci. 2015, 8, 1231–1236. [Google Scholar] [CrossRef]
- Carraro, G.; Barreca, D.; Bekermann, D.; Montini, T.; Gasparotto, A.; Gombac, V.; Maccato, C.; Fornasiero, P. Supported F-doped α-Fe2O3 nanomaterials: Synthesis, characterization and photo-assisted H2 production. J. Nanosci. Nanotechnol. 2013, 13, 4962–4968. [Google Scholar] [CrossRef] [PubMed]
- Payne, M.C.; Teter, M.P.; Allan, D.C.; Arias, T.A.; Joannopoulos, J.D. Iterative minimization techniques for ab initio total-energy calculations: Molecular dynamics and conjugate gradients. Rev. Mod. Phys. 1992, 64, 1045–1097. [Google Scholar] [CrossRef]
- Kresse, G.; Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 1995, 6, 15–50. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef]
- Pozun, Z.D.; Henkelman, G. Hybrid density functional theory band structure engineering in hematite. J. Chem. Phys. 2011, 134, 224706. [Google Scholar] [CrossRef] [PubMed]
- Paterson, E. Iron oxides in the laboratory. Preparation and characterization. Clay Miner. 1992, 27, 393. [Google Scholar] [CrossRef]
- Krén, E.; Szabó, P.; Konczos, G. Neutron diffraction studies on the (1−x) Fe2O3−xRh2O3 system. Phys. Lett. 1965, 19, 103–104. [Google Scholar] [CrossRef]


| k-Point | System Energy (103eV) | Lattice Parameters (Å) | |
|---|---|---|---|
| a (b) | c | ||
| 1 × 1 × 1 | −73.64 | 5.099 | 14.156 |
| 2 × 2 × 1 | −73.74 | 5.094 | 14.145 |
| 2 × 2 × 2 | −73.82 | 5.090 | 14.140 |
| 3 × 3 × 1 | −73.86 | 5.088 | 14.138 |
| 3 × 3 × 2 | −73.87 | 5.087 | 14.138 |
| Computational Functions | System Energy (103·eV) | Lattice Parameters (Å) | |
|---|---|---|---|
| a (b) | c | ||
| GGA-PBE | −72.23 | 5.0886 | 14.449 |
| GGA-RPBE | −72.84 | 5.0901 | 14.248 |
| GGA-PW91 | −73.86 | 5.0875 | 14.138 |
| GGA-WC | −73.09 | 5.0879 | 14.219 |
| Convergence Tolerance Parameters | Tolerances |
|---|---|
| Maximum energy change | 2.0 × 10−5 eV·atom−1 |
| Maximum force | 0.05 eV·Å−1 |
| Maximum stress | 0.1 GPa |
| Maximum displacement | 0.002 Å |
| Self-consistent field (SCF) | 2.0 × 10−6 eV·atom−1 |
| Species | Atoms | Charge/e |
|---|---|---|
| Pure α-Fe2O3 | O | −0.760 |
| O1 | −0.760 | |
| O2 | −0.760 | |
| O3 | −0.760 | |
| O4 | −0.760 | |
| O5 | −0.760 | |
| Fe1 | 1.140 | |
| Fe2 | 1.140 | |
| Fe3 | 1.140 | |
| Fe4 | 1.140 | |
| Ag-doped α-Fe2O3 | Ag | 1.730 |
| O | −0.690 | |
| O1 | −0.720 | |
| O2 | −0.740 | |
| O3 | −0.720 | |
| O4 | −0.680 | |
| O5 | −0.690 | |
| S-doped α-Fe2O3 | S | −0.230 |
| Fe | 0.870 | |
| Fe1 | 0.860 | |
| Fe2 | 0.910 | |
| Fe3 | 0.910 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, C.; Li, B.; Zhou, X.; Chen, J.; Tang, H. Comparative Study on Electronic Structure and Optical Properties of α-Fe2O3, Ag/α-Fe2O3 and S/α-Fe2O3. Metals 2021, 11, 424. https://doi.org/10.3390/met11030424
Zhao C, Li B, Zhou X, Chen J, Tang H. Comparative Study on Electronic Structure and Optical Properties of α-Fe2O3, Ag/α-Fe2O3 and S/α-Fe2O3. Metals. 2021; 11(3):424. https://doi.org/10.3390/met11030424
Chicago/Turabian StyleZhao, Cuihua, Baishi Li, Xi Zhou, Jianhua Chen, and Hongqun Tang. 2021. "Comparative Study on Electronic Structure and Optical Properties of α-Fe2O3, Ag/α-Fe2O3 and S/α-Fe2O3" Metals 11, no. 3: 424. https://doi.org/10.3390/met11030424
APA StyleZhao, C., Li, B., Zhou, X., Chen, J., & Tang, H. (2021). Comparative Study on Electronic Structure and Optical Properties of α-Fe2O3, Ag/α-Fe2O3 and S/α-Fe2O3. Metals, 11(3), 424. https://doi.org/10.3390/met11030424






