Corrosion Behavior of TC4 Titanium Alloys in Al–Li Alloy Melt
Abstract
:1. Introduction
2. Experimental
2.1. Material Preparation
2.2. Characterizations and Testing
3. Results
3.1. DRL Microstructure and Composition
3.2. Growth Kinetics of Diffusion Reaction Layer
3.3. Effect of Lithium on the Dissolution Rate of Titanium
4. Discussion
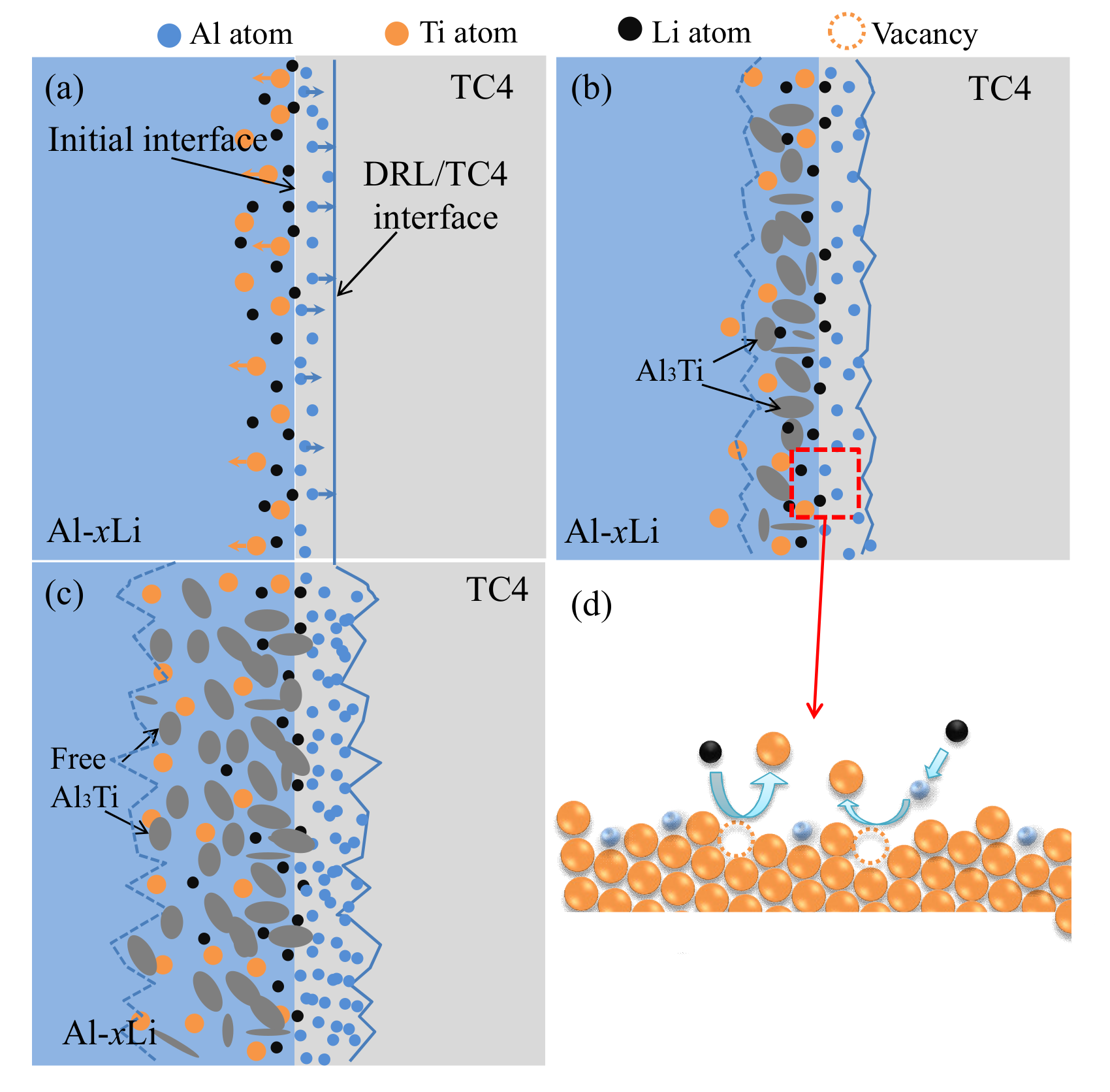
4.1. The Structure Evolution of DRL
- (1)
- Formation ofAl3Ti layer at the interface of solid TC4 and liquid Al–xLi alloy:
- (2)
- Formation of granular Al3Ti near Al3Ti layer:
- (3)
- Growth of Al3Ti layer and generation of small Al3Ti particles:
4.2. Influence Mechanism of Lithium on Diffusion Reaction Layer
5. Conclusions
- (1).
- After immersing at 680 °C to ~720 °C for 0.5 h to ~2 h, a diffusion reaction layer (DRL) composed of an Al3Ti compound was formed at the liquid/solid interface of Al–Li alloy melt/TC4 alloy. The DRL thickness increased with increases in holding time, temperature, and lithium content in the melt.
- (2).
- The lithium reduced the activation energy of the diffusion reaction and increased the ability of melt to dissolve the TC4 titanium alloy. At 700 °C, the growth kinetic equation of DRL is as follows:
- (3).
- At 700 °C, with the increase of lithium content in aluminum–lithium alloy melt, the dissolution rate of Ti increased significantly. When the holding time reaches 3 h, the Ti content in Al–2Li alloy melt is 0.105 wt%.
- (4).
- Application of crucibles and tools made of TC4 titanium alloy to smelt aluminum–lithium alloys can effectively eliminate the pollution of the melt caused by the dissolution of iron-containing appliances in the melt. Under the conventional melting temperature and time conditions of aluminum–lithium alloy, the titanium content in the melt is kept at a low level, and the TC4 titanium alloy appliances are not severely affected by the dissolution and corrosion of the aluminum–lithium alloy.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Rioja, R.J.; Liu, J. The Evolution of Al-Li Base Products for Aerospace and Space Applications. Metall. Mater. Trans. A 2012, 43, 3325–3337. [Google Scholar] [CrossRef]
- Yu, N.; Shang, J.; Cao, Y. Comparative Analysis of Al-Li Alloy and Aluminum Honeycomb Panel for Aerospace Application by Structural Optimization. Math. Probl. Eng. 2015, 2015, 815257. [Google Scholar] [CrossRef] [Green Version]
- Williams, J.C.; Starke, E.A. Progress in structural materials for aerospace systems. Acta Mater. 2003, 51, 5775–5799. [Google Scholar] [CrossRef]
- Akhtar, N.; Akhtar, W.; Wu, S.J. Melting and casting of lithium containing aluminium alloys. Int. J. Cast Met. Res. 2015, 28, 1–8. [Google Scholar] [CrossRef]
- Niraj, N.; Govind, K.; Suseelan, N. Studies on Al-Cu-Li-Mg-Ag-Zr alloy processed through vacuum induction melting (VIM) technique. Mater. Sci. Eng. A 2007, 454, 500–507. [Google Scholar]
- Wang, J.H.; Yi, D.Q.; Su, X.P. Solution kinetic of Al3Fe and Al9FeNi phases in aluminum allou melt. Trans. Nonferrous Met. Soc. China 2007, 17, 591–595. [Google Scholar]
- Song, D.-F.; Wang, S.-C.; Zhao, Y.-L.; Liu, S.-H.; DU, Y.; Kang, Y.-H.; Wang, Z.; Zhang, W.-W. Effect of melt holding on morphological evolution and sedimentation behavior of iron-rich intermetallic phases in Al-Si-Fe-Mn-Mg alloy. Trans. Nonferrous Met. Soc. China 2020, 30, 1–13. [Google Scholar] [CrossRef]
- Singh, H. Hot corrosion studies on plasma spray coatings over some Ni- and Fe- based super-alloys. J. Therm. Spray Technol. 2005, 14, 311–312. [Google Scholar]
- Ji, S.; Yang, W.; Gao, F.; Watson, D.; Fan, Z. Effect of iron on the microstructure and mechanical property of Al–Mg–Si–Mn and Al–Mg–Si die cast alloys. Mater. Sci. Eng. A 2013, 564, 130–139. [Google Scholar] [CrossRef]
- Zhou, H.; Cai, Q.; Wu, H.B.; Shan, Y.; Liu, X. Effect of Ni on the Microstructure and Mechanical Property of Annealing and Tempering Pre-hardened Plastic Mould Steel. New Technol. New Process 2012, 3, 67–70. [Google Scholar]
- Sun, Y.M. Elimination Measures of Harmful Iron Element in Aluminum Alloy. Foundry Technol. 2009, 30, 520–522. [Google Scholar]
- Wang, F.; Wang, X.; Cui, J. Effect of Low-Frequency Electromagnetic Casting on Micro-Structure and Macro-Segregation of 5A90 Alloy Ingots. Materials 2020, 13, 2720. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.-Y.; Li, L.; Bai, P.-K.; Jin, Y.; Wu, L.-Y.; Li, J.; Guan, R.-G.; Qu, H.-Q. The heat treatment influence on the microstructure and hardness of TC4 titanium alloy manufactured via selective laser melting. Materials 2018, 11, 1318. [Google Scholar] [CrossRef] [Green Version]
- Banerji, A.; Reif, W. Development of Al-Ti-C grain refiners containing TiC. Metall. Trans. A 1986, 17, 2127. [Google Scholar] [CrossRef]
- Ogawa, T.; Hasunuma, S.; Shirawachi, T. Effect of Chemical Composition and Relative Humidity on the Humid Gas Stress Corrosion Cracking of Aluminum Alloys. J. High Press. Inst. Jpn. 2019, 57, 24–33. [Google Scholar] [CrossRef]
- Wang, X.; Liao, X.; Ma, C.; Zhang, S.; Liu, Y.; Chen, H. Effects of chemical composition on the corrosion behavior of A7N01S-T5 Al alloys. Int. J. Mod. Phys. B 2015, 29, 27. [Google Scholar] [CrossRef]
- Delijic, K.; Markoli, B. The influence of the chemical composition on the corrosion performances of a medium strength Al-Mg-Si (6XXX) type alloys. Metall. Mater. Eng. 2014, 20, 131–140. [Google Scholar] [CrossRef] [Green Version]
- Okamoto, H. Al-Ti (aluminum-titanium). J. Phase Equilib. 1993, 14, 764. [Google Scholar] [CrossRef]
- Jiang, S.; Li, S. Formation mechanism and prediction of new phases in binary metallic liquid/solid interface. Rare Met. 2011, 30, 486–491. [Google Scholar] [CrossRef]
- Jiang, S.Y.; Li, S.C.; Zhang, L. Microstructure evolution of Al–Ti liquid–solid interface. Trans. Nonferrous Met. Soc. China 2013, 23, 3545–3552. [Google Scholar] [CrossRef]
- Huo, P.; Zhao, Z.; Bai, P.; Yuan, X.; Wang, Q.; Zhao, R.; Zhang, L.; Du, W.; Han, B.; Wang, Y. Deformation evolution and fracture mechanism of porous TC4 alloy scaffolds fabricated using selective laser melting under uniaxial compression. J. Alloys Compd. 2021, 861, 158529. [Google Scholar] [CrossRef]
- Li, H.; Yan, L.; Meng, L.; Li, Y.; Ai, F.; Zhang, H.; Jiang, Z. Effect of Ni on the microstructure and diffusion behavior at the interface of WC/high-speed steel composites. Metals 2021, 11, 341. [Google Scholar] [CrossRef]
- Gu, C.Q. Foundation of Materials Engineering; Beijing Machinery Industry Press: Beijing, China, 2003; p. 288. [Google Scholar]
- Wu, Y.J.; Lan, T. Study on Infiltration Combustion Synthesis (ICS) of TiAl Intermetallic Compound. Rare Met. Mater. Eng. 1996, 25, 17. [Google Scholar]
- Zhang, Y.S.; Zhao, S.M. Study on the Li diffusion coefficient measurement in liquid Al. Acta Chim. 1990, 4, 690–693. [Google Scholar]







| Element | Ti | Al | V | Fe | Si | Li |
|---|---|---|---|---|---|---|
| TC4 | 90.0 | 5.7 | 4.2 | 0 | ||
| Al–1Li | 98.9 | <0.05 | <0.05 | 0.96 | ||
| Al–2Li | 98.0 | <0.05 | <0.05 | 1.93 |
| Point | A | B | C |
|---|---|---|---|
| Al | 74.28 | 74.52 | 74.59 |
| Ti | 25.64 | 25.38 | 25.24 |
| V | 0.08 | 0.10 | 0.17 |
| T/°C | t/min | d/μm | ||
|---|---|---|---|---|
| Al | Al–1Li | Al–2Li | ||
| 680 | 30 | 3 | 4.4 | 25.4 |
| 60 | 3.7 | 7.0 | 85.7 | |
| 120 | 4.7 | 10.4 | 116.2 | |
| 700 | 30 | 3.2 | 5.2 | 33.4 |
| 60 | 4.3 | 8.9 | 103.6 | |
| 120 | 5.1 | 12.4 | 146.5 | |
| 720 | 30 | 4.6 | 7.2 | 42 |
| 60 | 5.4 | 11.2 | 140.8 | |
| 120 | 6.4 | 17.3 | 186.2 | |
| Melt | c/wt% | |||
|---|---|---|---|---|
| 30 min | 60 min | 120 min | 180 min | |
| Al | 0.018 | 0.027 | 0.030 | 0.038 |
| Al–Li | 0.029 | 0.037 | 0.042 | 0062 |
| Al–2Li | 0.035 | 0.057 | 0.095 | 0.105 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, F.; Wang, X.; Yan, Q.; Cui, J. Corrosion Behavior of TC4 Titanium Alloys in Al–Li Alloy Melt. Metals 2021, 11, 794. https://doi.org/10.3390/met11050794
Wang F, Wang X, Yan Q, Cui J. Corrosion Behavior of TC4 Titanium Alloys in Al–Li Alloy Melt. Metals. 2021; 11(5):794. https://doi.org/10.3390/met11050794
Chicago/Turabian StyleWang, Fuyue, Xiangjie Wang, Qiang Yan, and Jianzhong Cui. 2021. "Corrosion Behavior of TC4 Titanium Alloys in Al–Li Alloy Melt" Metals 11, no. 5: 794. https://doi.org/10.3390/met11050794







