Effects of Ni and Co on the Corrosion Resistance of Al-Si-Cu-Zn-Fe Alloys in NaCl Solution
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. SDAS and Secondary Phases under Slow and Fast Solidification
3.2. Potentiodynamic Polarization
3.3. Electrochemical Impedance Spectroscopy
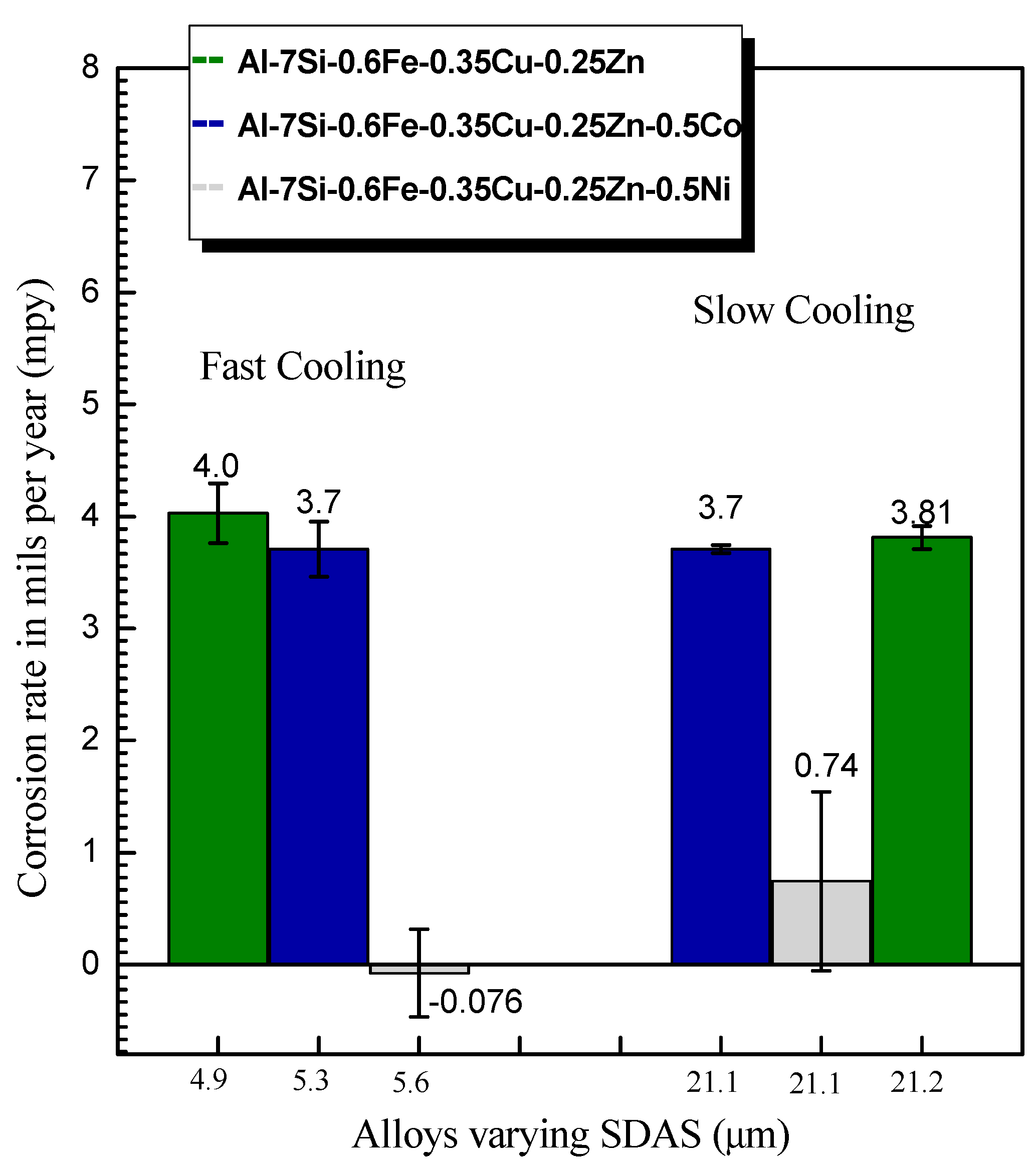
3.4. Immersion Corrosion Results
4. Conclusions
- The microstructures of the alloys were formed by the α-Al dendritic matrix, Si and AlFeSi/AlFeSi(Ni)/AlFeSi(Co) phases. The additions of either Co or Ni were not able to change the SDAS as compared to the non-modified alloy. The fast cooling process resulted in a reduction of SDAS of approximately four times.
- Considering the short elapsed time measurements, the currents associated with the Ecorr, i.e., the corrosion current density (icorr), appear to be highest for the Ni-containing alloy and lowest for the unmodified alloy. While not applying the Taefl extrapolation, the estimated icorr values of the Ni-containing alloy were found to be higher, ranging from 10−6 to 10−5 A/cm2. Moreover, the resistance to anodic polarization was marginally higher in the CC non-modified and Co-containing alloys in comparison to the corresponding DS samples.
- Due to the formation of a thick and dense alumina layer containing Ni in its inner layer, the Ni-containing alloy showed a lower corrosion rate under long exposure conditions (30 days). It is understood from the results of the present investigation that the addition of a small amount of Ni may be beneficial for longer exposure times to the saline electrolyte.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Kruger, J.; Lillard, R.S.; Streinz, C.C.; Moran, P.J. Effect of Microstructure on Passive Film Formation and Breakdown of Al-Ta Alloys. Faraday Discuss. 1992, 94, 127–136. [Google Scholar] [CrossRef]
- Natishan, P.M.; Mccafferty, E.; Hubler, G. Surface charge considerations in the pitting of ion-implanted aluminum. J. Electrochem. Soc. 1988, 135, 321–327. [Google Scholar] [CrossRef]
- Scully, J.R.; Knight, T.O.; Buchheit, R.G.; Peebles, D.E. Electrochemical characteristics of the Al2Cu, Al3Ta and Al3Zr intermetallic phases and their relevancy to the localized corrosion of Al alloys. Corros. Sci. 1993, 35, 185–195. [Google Scholar] [CrossRef]
- Birbilis, N.; Buchheit, R.G. Electrochemical Characteristics of Intermetallic Phases in Aluminum Alloys An Experimental Survey and Discussion. J. Electrochem. Soc. 2005, 152, 140–151. [Google Scholar] [CrossRef] [Green Version]
- Brown, G.M.; Shimizu, K.; Kobayashi, K.; Thompson, G.E.; Wood, G.C. The growth of chromate conversion coatings on high purity aluminium. Corros. Sci. 1993, 34, 1045–1054. [Google Scholar] [CrossRef]
- Shimizu, K.; Brown, G.; Kobayashi, K.; Skeldon, P.; Thompson, G.; Wood, G. Ultramicrotomy—A route towards the enhanced understanding of the corrosion and filming behaviour of aluminium and its alloys. Corros. Sci. 1998, 40, 1049–1072. [Google Scholar] [CrossRef]
- Ezuber, H.; El-Houd, A.; El-Shawesh, F. Materials & Design A study on the corrosion behavior of aluminum alloys in seawater. Mater. Des. 2008, 29, 801–805. [Google Scholar] [CrossRef]
- Meng, G.; Wei, L.; Zhang, T.; Shao, Y.; Wang, F.; Dong, C.; Li, X. Effect of microcrystallization on pitting corrosion of pure aluminium. Corros. Sci. 2009, 51, 2151–2157. [Google Scholar] [CrossRef]
- Kaiser, M.S.; Sabbir, S.H.; Rahman, M.; Kabir, M.S.; Nur, M. Al Effect of Fe, Ni and Cr on the corrosion behavior of hypereutectic Al-Si automotive alloy in 3.5% NaCl solution at different temperature and velocity. Mech. Eng. 2018, ME48, 11–17. [Google Scholar]
- Hossain, A.; Gulshan, F.; Syed, A.; Kurny, W. Electrochemical corrosion behavior of Ni-containing hypoeutectic Al-Si alloy. Electrochem. Sci. 2015, 5, 173–179. [Google Scholar] [CrossRef]
- Sani, A.S.; Aliyu, I.P.E. Effect of Chromium and Cobalt Additions on the Corrosion Resistance of Aluminium Silicon Iron Alloy (Al-Si- Fe). J. Sci. Eng. Res. 2012, 3, 1–10. [Google Scholar]
- Mirzaee-Moghadam, M.; Lashgari, H.R.; Zangeneh, S.; Rasaee, S.; Seyfor, M.; Asnavandi, M.; Mojtahedi, M. Dry sliding wear characteristics, corrosion behavior, and hot deformation properties of eutectic Al–Si piston alloy containing Ni-rich intermetallic compounds. Mater. Chem. Phys. 2022, 279, 125758. [Google Scholar] [CrossRef]
- Arthanari, S.; Jang, J.C.; Shin, K.S. Corrosion studies of high pressure die-cast Al-Si-Ni and Al-Si-Ni-Cu alloys. J. Alloys Compd. 2018, 749, 146–154. [Google Scholar] [CrossRef]
- Osório, W.R.; Cheung, N.; Spinelli, J.E.; Cruz, K.S.G.A. Microstructural modification by laser surface remelting and its effect on the corrosion resistance of an Al—9 wt % Si casting alloy. Appl. Surf. Sci. 2008, 254, 2763–2770. [Google Scholar] [CrossRef]
- Xavier, M.G.C.; Freitas, B.J.M.; Gouveia, G.L.; Garcia, A.; Spinelli, J.E. The Roles of Ni and Co in Dendritic Growth and Tensile Properties of Fe-Containing Al–Si–Cu–Zn Scraps under Slow and Fast Solidification Cooling. Adv. Eng. Mater. 2021, 2100822, 1–16. [Google Scholar] [CrossRef]
- Silva, B.L.; Cheung, N.; Garcia, A.; Spinelli, J.E. Thermal Parameters, Microstructure, and Mechanical Properties of Directionally Solidified Sn-0.7 wt.% Cu Solder Alloys Containing 0 ppm to 1000 ppm Ni. J. Electron. Mater. 2013, 42, 179–191. [Google Scholar] [CrossRef]
- Sivarupan, T.; Taylor, J.A.; Cáceres, C.H. SDAS, Si and Cu Content, and the Size of Intermetallics in Al-Si-Cu-Mg-Fe Alloys. Metall. Mater. Trans. A Phys. Metall. Mater. Sci. 2015, 46, 2082–2107. [Google Scholar] [CrossRef]
- Xavier, M.G.C.; Souza, T.M.G.; Cheung, N.; Garcia, A.; Spinelli, J.E. Effects of cobalt and solidification cooling rate on intermetallic phases and tensile properties of a -Cu, -Zn, -Fe containing Al-Si alloy. Int. J. Adv. Manuf. Technol. 2020, 107, 717–730. [Google Scholar] [CrossRef]
- Žihalová, M.; Bolibruchová, D.; Matejka, M. Change in the microstructure of alsi10mgmn alloy with higher iron content due to an increase in the amount of nickel. Teh. Vjesn. 2021, 28, 34–37. [Google Scholar] [CrossRef]
- Gaustad, G.; Olivetti, E.; Kirchain, R. Design for Recycling. J. Ind. Ecol. 2010, 14, 286–308. [Google Scholar] [CrossRef]
- Gaustad, G.; Olivetti, E.; Kirchain, R. Improving aluminum recycling: A survey of sorting and impurity removal technologies. Resour. Conserv. Recycl. 2012, 58, 79–87. [Google Scholar] [CrossRef]
- Silva, A.P.; Spinelli, J.E.; Mangelinck-noël, N.; Garcia, A. Microstructural development during transient directional solidification of hypermonotectic Al–Bi alloys. Mater. Des. 2010, 31, 4584–4591. [Google Scholar] [CrossRef]
- Vandersluis, E.; Lombardi, A.; Ravindran, C.; Bois-brochu, A.; Chiesa, F.; Mackay, R. Materials Science & Engineering A Factors in fl uencing thermal conductivity and mechanical properties in 319 Al alloy cylinder heads. Mater. Sci. Eng. A 2015, 648, 401–411. [Google Scholar] [CrossRef]
- Gündüz, M.; Çadirli, E. Directional solidification of aluminium-copper alloys. Mater. Sci. Eng. A 2002, 327, 167–185. [Google Scholar] [CrossRef]
- Mansfeld, F.; Fernandes, J.C.S. Impedance spectra for aluminum 7075 during the early stages of immersion in sodium chloride. Corros. Sci. 1993, 34, 2105–2108. [Google Scholar] [CrossRef]
- Moreto, J.A.; dos Santos, M.S.; Ferreira, M.O.A.; Carvalho, G.S.; Gelamo, R.V.; Aoki, I.V.; Taryba, M.; Bose Filho, W.W.; Fernandes, J.C.S. Corrosion and corrosion-fatigue synergism on the base metal and nugget zone of the 2524-T3 Al alloy joined by FSW process. Corros. Sci. 2021, 182, 109253. [Google Scholar] [CrossRef]
- Moreto, J.A.; Rossino, L.S.; Filho, W.W.B.; Marino, C.E.B.; Da Conceição Ferreira, M.; Taryba, M.; Fernandes, J.C.S. On the global and localised corrosion behaviour of the AA2524-T3 aluminium alloy used as aircraft fuselage skin. Mater. Res. 2019, 22, e20180280. [Google Scholar] [CrossRef] [Green Version]
- Moreto, J.A.; Gelamo, R.V.; Nascimento, J.P.L.; Taryba, M.; Fernandes, J.C.S. Improving the corrosion protection of 2524-T3-Al alloy through reactive sputtering Nb2O5 coatings. Appl. Surf. Sci. 2021, 556, 149750. [Google Scholar] [CrossRef]
- ASTM Standard guide for laboratory immersion corrosion testing of metals. ASTM Int. 2012, G31-12a, 1–10. [CrossRef]
- Zhang, Z.; Liu, M.-Y.; Breton, F.; Chen, X.-G. Microstructure and Mechanical Properties of AlSi10Mg Permanent Mould and High Pressure Vacuum Die Castings. In Proceedings of the 16th International Aluminum Alloys Conference (ICAA16), Montreal, QC, Canada, 17–21 June 2018; p. 399716. [Google Scholar]
- Yan, Q.; Song, B.; Shi, Y. Comparative study of performance comparison of AlSi10Mg alloy prepared by selective laser melting and casting. J. Mater. Sci. Technol. 2020, 41, 199–208. [Google Scholar] [CrossRef]
- Hwang, J.Y.; Doty, H.W.; Kaufman, M.J. The effects of Mn additions on the microstructure and mechanical properties of Al-Si-Cu casting alloys. Mater. Sci. Eng. A 2008, 488, 496–504. [Google Scholar] [CrossRef]
- Arthanari, S.; Jang, J.C.; Shin, K.S. Corrosion Behavior of High Pressure Die Cast Al-Ni and Al-Ni-Ca Alloys in 3.5% NaCl Solution. Corros. Sci. Technol. 2017, 3, 100–108. [Google Scholar]
- Martin, F.J.; Cheek, G.T.; O’Grady, W.E.; Natishan, P.M. Impedance studies of the passive film on aluminium. Corros. Sci. 2005, 47, 3187–3201. [Google Scholar] [CrossRef]
- Osório, W.R.; Goulart, P.R.; Garcia, A. Effect of silicon content on microstructure and electrochemical behavior of hypoeutectic Al–Si alloys. Mater. Lett. 2008, 62, 365–369. [Google Scholar] [CrossRef]
- Das, N.; Sengupta, P.; Abraham, G.; Arya, A.; Kain, V.; Dey, G.K. Development in corrosion resistance by microstructural refinement in Zr-16 SS 304 alloy using suction casting technique. Mater. Res. Bull. 2016, 80, 295–302. [Google Scholar] [CrossRef]
- Vargel, C. Corrosion of Aluminium; Elsevier: Amsterdam, The Netherlands, 2020; ISBN 9780080999258. [Google Scholar]
- Roche, V.; Koga, G.Y.; Matias, T.B.; Kiminami, C.S.; Bolfarini, C.; Botta, W.J.; Nogueira, R.P.; Jorge Junior, A.M. Degradation of biodegradable implants: The influence of microstructure and composition of Mg-Zn-Ca alloys. J. Alloys Compd. 2019, 774, 168–181. [Google Scholar] [CrossRef]












| Alloys | Solidification Severity | SDAS (μm) | Rs (Ω cm2) | Rf (kΩ cm2) | Qf (μF s (αf–1)) | αf | Rct (kΩ cm2) | Qdl (μF s (αdl–1)) | αdl | χ2/|Z| |
|---|---|---|---|---|---|---|---|---|---|---|
| Non-modified | Slow cooling | 21.2 | 31.6 | 45.9 | 4.3 | 0.84 | 100.9 | 14.4 | 0.98 | 0.09 |
| Ni-containing | Slow cooling | 21.1 | 31.7 | 10.0 | 13.7 | 0.86 | 67.3 | 56100 | 0.72 | 0.09 |
| Co-containing | Slow cooling | 21.1 | 29.8 | 30.4 | 3.3 | 0.88 | 111.3 | 18.4 | 0.84 | 0.04 |
| Non-modified | Fast cooling | 4.9 | 31.9 | 31.7 | 4.7 | 0.88 | 69.7 | 33.3 | 0.88 | 0.07 |
| Ni-containing | Fast cooling | 5.6 | 30.3 | 7.3 | 8.6 | 0.87 | 9.4 | 219.0 | 0.86 | 0.09 |
| Co-containing | Fast cooling | 5.3 | 31.1 | 31.7 | 10.7 | 0.85 | 62.7 | 51.5 | 0.88 | 0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xavier, M.G.C.; Freitas, B.J.M.; Koga, G.Y.; Spinelli, J.E. Effects of Ni and Co on the Corrosion Resistance of Al-Si-Cu-Zn-Fe Alloys in NaCl Solution. Metals 2022, 12, 645. https://doi.org/10.3390/met12040645
Xavier MGC, Freitas BJM, Koga GY, Spinelli JE. Effects of Ni and Co on the Corrosion Resistance of Al-Si-Cu-Zn-Fe Alloys in NaCl Solution. Metals. 2022; 12(4):645. https://doi.org/10.3390/met12040645
Chicago/Turabian StyleXavier, Marcella G. C., Brenda J. M. Freitas, Guilherme Y. Koga, and José E. Spinelli. 2022. "Effects of Ni and Co on the Corrosion Resistance of Al-Si-Cu-Zn-Fe Alloys in NaCl Solution" Metals 12, no. 4: 645. https://doi.org/10.3390/met12040645








