Characterization of the Anodic Film and Corrosion Resistance of an A535 Aluminum Alloy after Intermetallics Removal by Different Etching Time
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Surface Preparation
2.3. Anodizing and Sealing Treatment
2.4. Characterization of Anodic Oxide Films
2.5. Vickers Microhardness
2.6. Corrosion Testing
3. Results and Discussion
3.1. Surface Pretreatment Characterization
3.2. Characterization of Anodic Oxide Film on a Cast A535 Alloy
3.2.1. Surface Morphology of Anodic Oxide Films
3.2.2. Thickness and Hardness of the Anodic Oxide Film on an A535 Alloy
3.3. Corrosion Resistance
3.3.1. Corrosion under Salt Spray Exposure
3.3.2. Electrochemical Corrosion Behavior
4. Conclusions
- A suitable alkaline etching time of 4 min can effectively remove intermetallic particles from the matrix surface, providing the grooved surface with higher roughness for anodic oxide film growth. In addition, at a lower etching time, the corroded surface showed local pitting, which resulted from the remaining intermetallics that reacted with the electrolyte and increased the positive charges in the corroded area.
- Etching the surface of the alloy for 4 min assured lower porosity and more uniform anodic film with a sufficiently large thickness of up to 70 µm, with the maximum hardness of 550 HV.
- The corrosion resistance of an A535 alloy can be improved by sealing with stearic acid, which provided better quality of the sealed layer than NiF2 due to the fewer reaction products and improved wettability.
- After a salt spray test for 336 h, the samples sealed with stearic did not show pitting sites and corrosion products.
- The corrosion behavior evaluated by polarization fitting curves revealed that the anodic oxide film sealed with stearic had better corrosion resistance compared to the anodic oxide film sealed with NiF2, which was quantified by the lower corrosion current density (Icorr).
- Alkaline etching can remove intermetallic particles at the surface of an A535 alloy.
- Remaining intermetallic phase on the alkaline etched surface was responsible for the current density changing, which lead to pitting corrosion.
- Etching for 4 min produces better surface adhesion of the anodic film and fewer voids.
- An anodized A535 alloy sealed by stearic acid has better corrosion resistance.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fasoyinu, F.; Cousineau, D.; Sahoo, M. Marrying Almag 535 to the permanent mold process. Mod. Cast. 2005, 95, 43–45. [Google Scholar]
- Fasoyinu, F.; Thomson, J.; Castles, T.; Sahoo, M. Mechanical Properties and Metallography of Al-Mg Alloy 535.0. AFS Trans. 2003, 3, 1–13. [Google Scholar]
- Kaufman, J.G.; Rooy, E.L. Aluminum Alloy Castings, Properties, Processes and Applications; ASM International: Almere, The Netherlands, 2004. [Google Scholar]
- Speidel, M.O. Stress corrosion cracking of aluminum alloys. Met. Mater. Trans. A 1975, 6, 631–651. [Google Scholar] [CrossRef]
- Jones, R.H.; Baer, D.R.; Danielson, M.J.; Vetrano, J.S. Role of Mg in the stress corrosion cracking of an Al-Mg alloy. Met. Mater. Trans. A 2001, 32, 1699–1711. [Google Scholar] [CrossRef]
- Baer, D.R.; Windisch, C., Jr.; Engelhard, M.H.; Danielson, M.J.; Jones, R.H.; Vetrano, J.S. Influence of Mg on the corrosion of Al. J. Vac. Sci. Technol. A Vac. Surf. Film. 2000, 18, 131–136. [Google Scholar] [CrossRef]
- Yasakau, K.; Zheludkevich, M.; Ferreira, M. Role of intermetallics in corrosion of aluminum alloys. Smart corrosion protection. In Intermetallic Matrix Composites; Elsevier: Amsterdam, The Netherlands, 2018; pp. 425–462. [Google Scholar] [CrossRef]
- Li, Z.; Yi, D.; Tan, C.; Wang, B. Investigation of the stress corrosion cracking behavior in annealed 5083 aluminum alloy sheets with different texture types. J. Alloys Compd. 2019, 817, 152690. [Google Scholar] [CrossRef]
- Yan, J.; Heckman, N.M.; Velasco, L.; Hodge, A.M. Improve sensitization and corrosion resistance of an Al-Mg alloy by optimization of grain boundaries. Sci. Rep. 2016, 6, 26870. [Google Scholar] [CrossRef] [Green Version]
- Zhang, P.; Zuo, Y.; Zhao, X.; Tang, Y.; Zhang, X. Correlation between microhardness and microstructure of anodic film on 2024 aluminum alloy. J. Wuhan Univ. Technol. Sci. Ed. 2015, 30, 586–590. [Google Scholar] [CrossRef]
- Li, S.-M.; Li, Y.-D.; Zhang, Y.; Liu, J.-H.; Yu, M. Effect of intermetallic phases on the anodic oxidation and corrosion of 5A06 aluminum alloy. Int. J. Miner. Met. Mater. 2015, 22, 167–174. [Google Scholar] [CrossRef] [Green Version]
- Abdel-Gawad, S.A.; Osman, W.M.; Fekry, A.M. Characterization and corrosion behavior of anodized Aluminum alloys for military industries applications in artificial seawater. Surf. Interfaces 2018, 14, 314–323. [Google Scholar] [CrossRef]
- Iewkitthayakorn, I.; Janudom, S.; Mahathaninwong, N.; Karrila, S.; Wannasin, J. Anodizing parameters for superheated slurry cast 7075 aluminum alloys. Trans. Nonferrous Met. Soc. China 2019, 29, 1200–1210. [Google Scholar] [CrossRef]
- Zhu, B. On the Influence of Si on Anodising and Mechanical Properties of Cast Aluminium Alloys. Ph.D. Thesis, Jönköping University, Jönköping, Sweden, 2017. [Google Scholar]
- Berlanga-Labari, C.; Biezma-Moraleda, M.V.; Rivero, P.J. Corrosion of Cast Aluminum Alloys: A Review. Metals 2020, 10, 1384. [Google Scholar] [CrossRef]
- Konieczny, J.; Labisz, K.; Polok-Rubiniec, M.; Włodarczyk-Fligier, A. Influence of Aluminium Alloy Anodizing and Casting Methods on Structure and Functional Properties. Arch. Met. Mater. 2016, 61, 1337–1342. [Google Scholar] [CrossRef] [Green Version]
- Jothi, V.; Adesina, A.Y.; Kumar, A.M.; Al-Aqeeli, N.; Ram, J.N. Influence of an anodized layer on the adhesion and surface protective performance of organic coatings on AA2024 aerospace Al alloy. Prog. Org. Coat. 2019, 138, 105396. [Google Scholar] [CrossRef]
- Bruera, F.; Kramer, G.; Vera, M.; Ares, A. Evaluation of surface pretreatment stages of Al 1050 to obtain nanostructured anodic films. Superlattices Microstruct. 2019, 130, 103–116. [Google Scholar] [CrossRef]
- Leontiev, A.; Roslyakov, I.; Napolskii, K. Complex influence of temperature on oxalic acid anodizing of aluminium. Electrochim. Acta 2019, 319, 88–94. [Google Scholar] [CrossRef]
- Aggerbeck, M.; Canulescu, S.; Dirscherl, K.; Johansen, V.E.; Engberg, S.; Schou, J.; Ambat, R. Appearance of anodised aluminium: Effect of alloy composition and prior surface finish. Surf. Coat. Technol. 2014, 254, 28–41. [Google Scholar] [CrossRef]
- Yu, S.; Wang, L.; Wu, C.; Feng, T.; Cheng, Y.; Bu, Z.; Zhu, S. Studies on the corrosion performance of an effective and novel sealing anodic oxide coating. J. Alloys Compd. 2020, 817, 153257. [Google Scholar] [CrossRef]
- Ofoegbu, S.U.; Fernandes, F.A.; Pereira, A.B. The Sealing Step in Aluminum Anodizing: A Focus on Sustainable Strategies for Enhancing Both Energy Efficiency and Corrosion Resistance. Coatings 2020, 10, 226. [Google Scholar] [CrossRef] [Green Version]
- Cartigny, V.; Veys-Renaux, D.; Desenne, P.; Rocca, E. Rapid sealing of an alumina nanoporous network grown by anodizing and dye-filled. Surf. Coat. Technol. 2019, 364, 369–376. [Google Scholar] [CrossRef]
- Boisier, G.; Lamure, A.; Pébère, N.; Portail, N.; Villatte, M. Corrosion protection of AA2024 sealed anodic layers using the hydrophobic properties of carboxylic acids. Surf. Coat. Technol. 2009, 203, 3420–3426. [Google Scholar] [CrossRef] [Green Version]
- Jothi, V.; Adesina, A.Y.; Kumar, A.M.; Ram, J.S.N. Influence of Organic Acids on the Surface and Corrosion Resistant Behavior of Anodized Films on AA2024 Aerospace Alloys in Artificial Seawater. Met. Mater. Int. 2019, 26, 1611–1620. [Google Scholar] [CrossRef]
- El-Hameed, A.M.A.; Abdel-Aziz, Y.A.; El-Tokhy, F.S. Anodic Coating Characteristics of Different Aluminum Alloys for Spacecraft Materials Applications. Mater. Sci. Appl. 2017, 8, 197–208. [Google Scholar] [CrossRef] [Green Version]
- Jo, H.; Lee, S.; Kim, D.; Lee, J. Low Temperature Sealing of Anodized Aluminum Alloy for Enhancing Corrosion Resistance. Materials 2020, 13, 4904. [Google Scholar] [CrossRef]
- Chankitmunkong, S.; Eskin, D.G.; Limmaneevichitr, C.; Pandee, P.; Kengkla, N.; Athchasiri, J.; Tanawansombat, T.; Parnlasarn, N.; Diewwanit, O. Effect of Homogenization on Anodic Film and Electrochemical Behavior of an A535 Alloy After Sealing with Stearic Sealant. In Light Metals; Springer: Berlin/Heidelberg, Germany, 2022; pp. 221–227. [Google Scholar] [CrossRef]
- Shang, Y.; Wang, L.; Liu, Z.; Niu, D.; Wang, Y.; Liu, C. The Effects of Different Sealing Techniques for Anodic Film of Al-12.7 Si 0.7 Mg Alloys. Int. J. Electrochem. Sci. 2016, 11, 5234–5344. [Google Scholar] [CrossRef]
- Medvedev, O.S.; Alyasova, E.E.; Besprozvannaya, R.E.; Gadzhiev, A.A.; Krivova, V.V.; Kondratev, A.S.; Kim, A.E.; Novikov, P.A.; Popovich, A.A. Influence of Alloying Elements on the Mechanical Properties of Anodized Aluminum and on the Adhesion of Copper Metallization. Materials 2021, 14, 7028. [Google Scholar] [CrossRef]
- Li, K.; Li, W.; Yi, A.; Zhu, W.; Liao, Z.; Chen, K.; Li, W. Tuning the Surface Characteristic of Al-Si Alloys and Its Impacts on the Formation of Micro Arc Oxidation Layers. Coatings 2021, 11, 453. [Google Scholar] [CrossRef]
- Sadeghpour-Motlagh, M.; Mokhtari-Zonouzi, K.; Aghajani, H.; Kakroudi, M.G. Effects of Etching Time and NaOH Concentration on the Production of Alumina Nanowires Using Porous Anodic Alumina Template. J. Mater. Eng. Perform. 2014, 23, 2007–2014. [Google Scholar] [CrossRef]
- Jin, Z.; Cai, C.; Hashimoto, T.; Yuan, Y.; Kang, D.; Hunter, J.; Zhou, X. Alkaline etching and desmutting of aluminium alloy: The behaviour of Mg2Si particles. J. Alloys Compd. 2020, 842, 155834. [Google Scholar] [CrossRef]
- Jin, Z.; Cai, C.; Yuan, Y.; Kang, D.; Hunter, J.; Zhou, X. The behaviour of AA5754 and AA5052 aluminium alloys in alkaline etching solution: Similarity and difference. Mater. Charact. 2020, 171, 110768. [Google Scholar] [CrossRef]
- Shi, H.; Yu, M.; Liu, J.; Rong, G.; Du, R.; Wang, J.; Li, S. Effect of alkaline etching on microstructure and anticorrosion performance of anodic film on Al-Mg-Si alloy. Corros. Sci. 2020, 169, 108642. [Google Scholar] [CrossRef]
- Kaseem, M.; Yang, H.W.; Ko, Y.G. Toward a nearly defect-free coating via high-energy plasma sparks. Sci. Rep. 2017, 7, 2378. [Google Scholar] [CrossRef] [PubMed]
- Zuo, Y.; Zhao, P.-H.; Zhao, J.-M. The influences of sealing methods on corrosion behavior of anodized aluminum alloys in NaCl solutions. Surf. Coat. Technol. 2003, 166, 237–242. [Google Scholar] [CrossRef]
- Kalantary, M.R.; Gabe, D.R.; Ross, D.H. A model for the mechanism of nickel fluoride cold sealing of anodized aluminium. J. Appl. Electrochem. 1992, 22, 268–276. [Google Scholar] [CrossRef]
- Boisier, G.; Pébère, N.; Druez, C.; Villatte, M.; Suel, S. FESEM and EIS study of sealed AA2024 T3 anodized in sulfuric acid electrolytes: Influence of tartaric acid. J. Electrochem. Soc. 2008, 155, C521. [Google Scholar] [CrossRef] [Green Version]
- Saleema, N.; Sarkar, D.; Paynter, R.; Gallant, D.; Eskandarian, M. A simple surface treatment and characterization of AA 6061 aluminum alloy surface for adhesive bonding applications. Appl. Surf. Sci. 2012, 261, 742–748. [Google Scholar] [CrossRef] [Green Version]
- Kocabaş, M.; Curioni, M.; Cansever, N. The effect of anodic oxidation conditions on the structure of dual pre-treatment electroless nickel coating on AA1050 surface. Surf. Coat. Technol. 2019, 364, 321–328. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.; Kim, Y.; Jang, H.; Chung, W. Cr2O3 sealing of anodized aluminum alloy by heat treatment. Surf. Coat. Technol. 2012, 243, 34–38. [Google Scholar] [CrossRef]
- Cheng, T.-C.; Chou, C.-C. The Electrical and Mechanical Properties of Porous Anodic 6061-T6 Aluminum Alloy Oxide Film. J. Nanomater. 2015, 2015, 371405. [Google Scholar] [CrossRef]
- Mihajlović, S.; Sekulić, Ž.; Daković, A.; Vučinić, D.; Jovanović, V.; Stojanović, J. Surface properties of natural calcite filler treated with stearic acid. Ceram. Silikáty 2009, 53, 268–275. [Google Scholar]
- Zhang, X.; Wu, G.; Peng, X.; Li, L.; Feng, H.; Gao, B.; Huo, K.; Chu, P.K. Mitigation of Corrosion on Magnesium Alloy by Predesigned Surface Corrosion. Sci. Rep. 2015, 5, 17399. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Usman, B.J.; Scenini, F.; Curioni, M. Corrosion Testing of Anodized Aerospace Alloys: Comparison Between Immersion and Salt Spray Testing using Electrochemical Impedance Spectroscopy. J. Electrochem. Soc. 2020, 167, 041505. [Google Scholar] [CrossRef]
- Materia, T. Corrosion of Aluminum and Its Alloys: Forms of Corrosion. Total Mater. 2008. Available online: http://www.keytometals.com/page.aspx?ID=CheckArticle&site=ktn&NM=187 (accessed on 25 June 2022).
- Wu, Y.; Zhao, W.; Wang, W.; Wang, L.; Xue, Q. Novel anodic oxide film with self-sealing layer showing excellent corrosion resistance. Sci. Rep. 2017, 7, 1344. [Google Scholar] [CrossRef]
- Shulman, G.P.; Bauman, A. Organic acid sealants for anodized aluminum—A new method for corrosion protection. Met. Finish. 1995, 93, 16–19. [Google Scholar] [CrossRef]
- Golru, S.S.; Attar, M.; Ramezanzadeh, B. Effects of different surface cleaning procedures on the superficial morphology and the adhesive strength of epoxy coating on aluminium alloy 1050. Prog. Org. Coat. 2015, 87, 52–60. [Google Scholar] [CrossRef]
- Brito, C.; Vida, T.; Freitas, E.; Cheung, N.; Spinelli, J.E.; Garcia, A. Cellular/dendritic arrays and intermetallic phases affecting corrosion and mechanical resistances of an Al–Mg–Si alloy. J. Alloys Compd. 2016, 673, 220–230. [Google Scholar] [CrossRef]
- Zaid, B.; Saidi, D.; Benzaid, A.; Hadji, S. Effects of pH and chloride concentration on pitting corrosion of AA6061 aluminum alloy. Corros. Sci. 2008, 50, 1841–1847. [Google Scholar] [CrossRef]
- Malina, J.; Radošević, J. Influence of NaCl concentration on pitting corrosion of extruded Al-Mg-Si alloy AA6060. Zaštita Mater. 2015, 56, 47–51. [Google Scholar] [CrossRef]
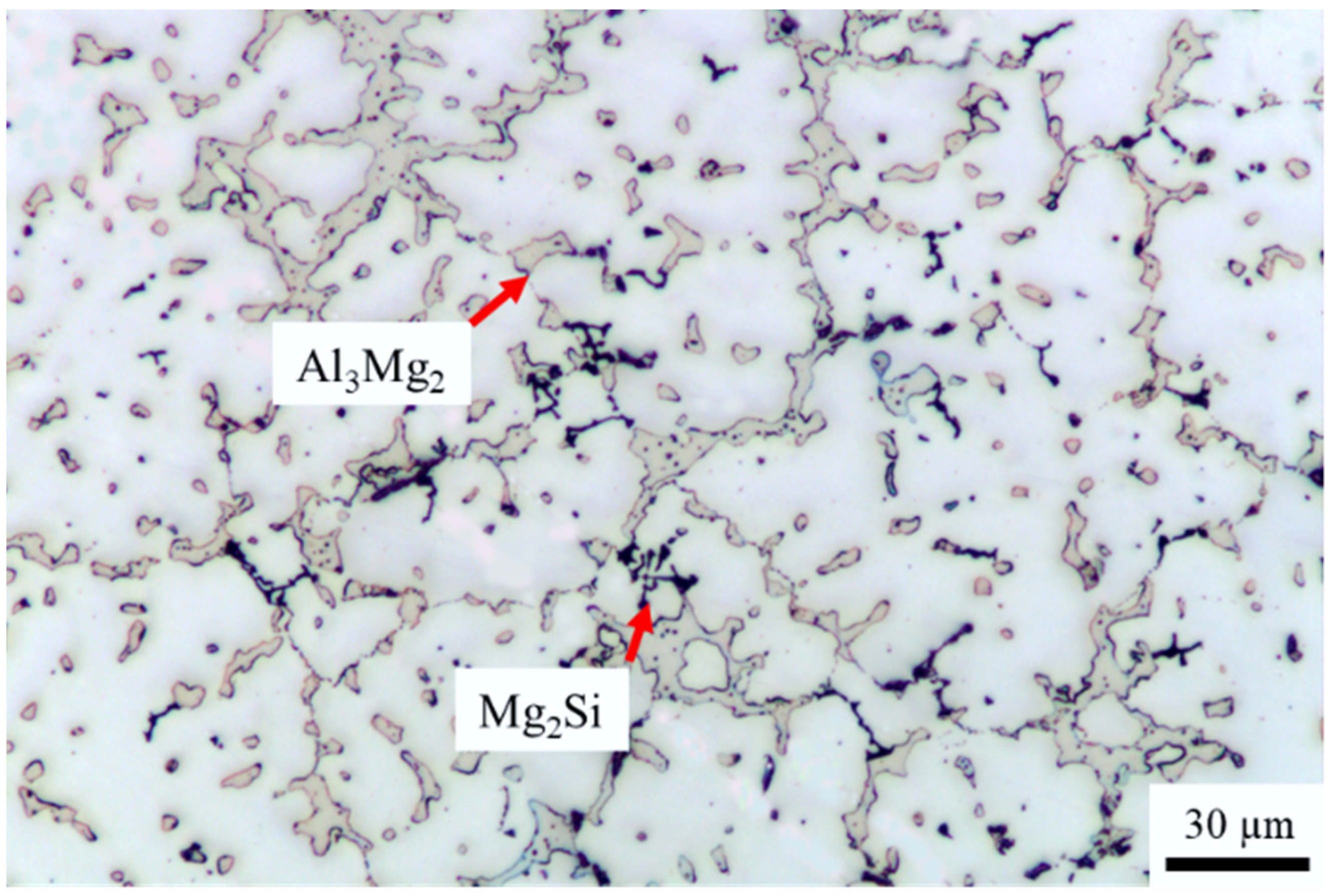















| Element | Al | Si | Mg | Fe | Mn | Ti | Cr | Cu |
|---|---|---|---|---|---|---|---|---|
| Balance | 0.2 | 7.03 | 0.15 | 0.17 | 0.13 | <0.01 | <0.01 |
| Sealing Method | Condition |
|---|---|
| Cold sealing with nickel fluoride (NiF2) | 1.2 g L−1, pH 5.5–6.5, 25 °C, 20 min. Post treatment, deionized water, pH 5.5–6.5, 60 °C, 15 min. |
| Hydrothermal sealing with stearic acid (C17H35CO2H) | 100 wt.% stearic acid, 100 °C, 30 min, air dry |
| Samples | Alkaline Etching Time (min) | Anodizing | Sealing Method |
|---|---|---|---|
| As cast | - | - | - |
| 2-A | 2 | ✓ | - |
| 4-A | 4 | ✓ | - |
| 2-N | 2 | ✓ | NiF2 |
| 4-N | 4 | ✓ | NiF2 |
| 2-S | 2 | ✓ | Stearic acid |
| 4-S | 4 | ✓ | Stearic acid |
| Specimens | βa (V/dec) | βc (V/dec) | Ecorr, Obs (V) | Icorr (mA/cm2) |
|---|---|---|---|---|
| 2-N | 0.57982 | −1.55570 | −0.58 | 9.40 × 10−7 |
| 4-N | 0.47716 | −0.83352 | −0.85 | 6.53 × 10−7 |
| 2-S | 0.24037 | −0.62977 | −0.53 | 8.15 × 10−9 |
| 4-S | 0.35701 | −1.06010 | −0.35 | 5.89 × 10−9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chankitmunkong, S.; Eskin, D.; Limmaneevichitr, C.; Kengkla, N.; Diewwanit, O. Characterization of the Anodic Film and Corrosion Resistance of an A535 Aluminum Alloy after Intermetallics Removal by Different Etching Time. Metals 2022, 12, 1140. https://doi.org/10.3390/met12071140
Chankitmunkong S, Eskin D, Limmaneevichitr C, Kengkla N, Diewwanit O. Characterization of the Anodic Film and Corrosion Resistance of an A535 Aluminum Alloy after Intermetallics Removal by Different Etching Time. Metals. 2022; 12(7):1140. https://doi.org/10.3390/met12071140
Chicago/Turabian StyleChankitmunkong, Suwaree, Dmitry Eskin, Chaowalit Limmaneevichitr, Nattarat Kengkla, and Onnjira Diewwanit. 2022. "Characterization of the Anodic Film and Corrosion Resistance of an A535 Aluminum Alloy after Intermetallics Removal by Different Etching Time" Metals 12, no. 7: 1140. https://doi.org/10.3390/met12071140






