Kinetic Mechanism of Hydrogen Absorption of AA6111 Alloys Melt
Abstract
:1. Introduction
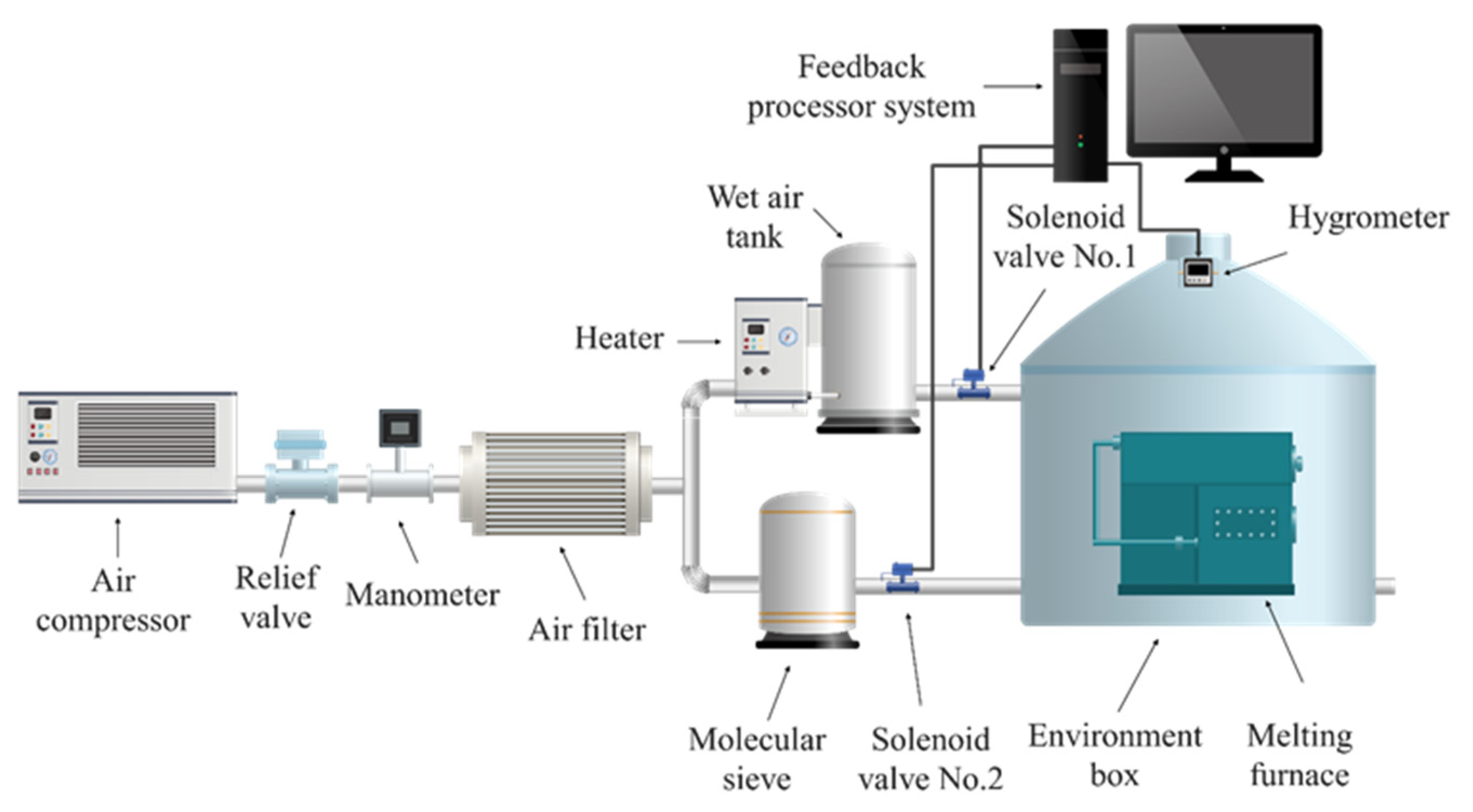
2. Materials and Methods
3. Results and Discussion
3.1. Analysis of Experimental Results
3.1.1. Effect of Melting Environment on the Hydrogen Absorption Process
3.1.2. Effect of Structural Change in Oxide Film on Hydrogen Absorption during the Heating Process
3.2. Modeling of the Hydrogen Absorption Process in Aluminum Melt
3.3. Calculation Result and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jakse, N.; Pasturel, A. Ab initio based understanding of diffusion mechanisms of hydrogen in liquid aluminum. Appl. Phys. Lett. 2014, 104, 3377. [Google Scholar] [CrossRef]
- Sergey, V.B.; Vladimir, N.B.; Ivan, Y.G.; Boris, P.K.; Natalia, P.K. Saturation dynamics of aluminum alloys with hydrogen. ARPN J. Eng. Appl. Sci. 2017, 12, 6243–6247. [Google Scholar]
- Yu, M.Z.; Cui, J.; Tang, Z.C.; Shen, Z.N.; Chen, X.Y.; Xu, Z.B.; Zeng, J.M. Effect of Er-Rich Precipitates on Microstructure and Electrochemical Behavior of the Al-5Zn-0.03In Alloy. Metals 2022, 12, 131. [Google Scholar] [CrossRef]
- Cui, J.; Tang, Z.C.; Yu, M.Z.; Hu, J.J.; Chen, X.Y.; Xu, Z.B.; Zeng, J.M. Effect of Heat Treatment on Microstructural Evolution and Microhardness Change of Al-5Zn-0.03In-1Er Alloy. Metals 2022, 12, 370. [Google Scholar] [CrossRef]
- Tang, Z.C.; Cui, J.; Yu, M.Z.; Zhu, W.Q.; Xu, Z.B.; Zeng, J.M.; Xu, T.; Yang, H.D.; Tan, Y.P.; Yang, B. A new insight on the diffusion growth mechanism of intermetallic compounds in Al-Er system. Mater. Des. 2022, 224, 111341. [Google Scholar] [CrossRef]
- Voigt, C.; Ditscherlein, L.; Werzner, E.; Zienert, T.; Nowak, R.; Peuker, U.; Sobczak, N.; Aneziris, C. Wettability of AlSi7Mg alloy on alumina, spinel, mullite and rutile and its influence on the aluminum melt filtration efficiency. Mater. Des. 2018, 150, 75–85. [Google Scholar] [CrossRef]
- Wu, R.Z.; Sun, B.D.; Shu, D.; Li, F.; Chen, H.L.; Yan, L.L.; Wang, J. Degassing of Aluminum Melt. Mater. Sci. Technol. 2006, 14, 218–221. [Google Scholar] [CrossRef]
- Haghayeghi, R.; Bahai, H.; Kapranos, P. Effect of ultrasonic argon degassing on dissolved hydrogen in aluminium alloy. Mater. Lett. 2012, 82, 230–232. [Google Scholar] [CrossRef]
- Harvey, J.P.; Chartrand, P. Modeling the Hydrogen Solubility in Liquid Aluminum Alloys. Metall. Mater. Trans. B 2010, 41, 908–924. [Google Scholar] [CrossRef]
- Jiang, G.R.; Liu, Y.; Li, Y.X.; Su, Y.Q.; Guo, J.J. A Model for Calculating Solubility of Hydrogen in Molten Aluminum Alloys. Acta Metall. Sin. 2008, 44, 129–133. [Google Scholar]
- Zhang, H.W.; Li, Y.X.; Liu, Y. Hydrogen Solubility in Pure Metals for Gasar Process. Acta Metall. Sin. 2007, 43, 113–118. [Google Scholar]
- Anyalebechi, P.N. Hydrogen Solubility in Liquid and Solid Pure Aluminum—Critical Review of Measurement Methodologies and Reported Values. Mater. Sci. Appl. 2022, 13, 158–212. [Google Scholar] [CrossRef]
- Anyalebechi, P.N. Analysis of the effects of alloying elements on hydrogen solubility in liquid aluminum alloys. Scr. Metall. Et Mater. 1995, 33, 1209–1216. [Google Scholar] [CrossRef]
- Sigworth, G.K.; Engh, T.A. Chemical and kinetic factors related to hydrogen removal from aluminum. Metall. Trans. B 1982, 13, 447–460. [Google Scholar] [CrossRef]
- Jang, H.S.; Kang, H.J.; Yoon, P.; Lee, G.; Shin, S. Effects of Mg Content on Hydrogen Content and Melt Quality of Al-Mg Alloys. Met.—Open Access Metall. J. 2019, 9, 1235. [Google Scholar] [CrossRef] [Green Version]
- Muhammet, U.; Remzi, e.; Derya, D.; Murat, T. Characterization of the Effect of Melt Treatments on Melt Quality in Al-7wt %Si-Mg Alloys. Met.—Open Access Metall. J. 2017, 7, 157. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Lv, X.; Torgerson, A.T.; Long, M. Removal of Impurity Elements from Molten Aluminum: A Review. Miner. Process. Extr. Metall. Rev. 2011, 32, 150–228. [Google Scholar] [CrossRef]
- Kim, M.J.; Yun, J.P.; Yang, J.; Choi, S.J.; Kim, D.E. Prediction of the Temperature of Liquid Aluminum and the Dissolved Hydrogen Content in Liquid Aluminum with a Machine Learning Approach. Met.—Open Access Metall. J. 2020, 10, 330. [Google Scholar] [CrossRef] [Green Version]
- Zeng, J.; Ping, G.; Wang, Y. Investigation of Inner Vacuum Sucking method for degassing of molten aluminum. Mater. Sci. Eng. B 2012, 177, 1717–1720. [Google Scholar] [CrossRef]
- Sun, Y.Z.; Li, S.C.; Zhang, G.Q.; Qiu, H.X.; Yu, C.; Zhang, G.W. Research on Absorbing Hydrogen Special Property of Liquid State ZL101 Aluminium Alloys. Acta Met. Sin. 1999, 9, 939–941. [Google Scholar]
- Guo, Y.J. Effect of Casting Temperature on Hydrogen Content in Aluminum Alloy. Alum. Fabr. 2008, 1, 39–40. [Google Scholar]
- Tiryakiolu, M. On estimating the hydrogen solubility in solid aluminium alloys and the hydrogen solubility difference between the liquidus and solidus temperatures. Int. J. Cast Met. Res. 2021, 34, 83–87. [Google Scholar] [CrossRef]
- Li, P.J.; Zeng, D.B.; Jia, J. Kinetics of absorbing hydrogen of Al melt. Trans. Nonferrous Met. Soc. China 2000, 10, 261–263. [Google Scholar]
- Fu, G.S.; Kang, J.X. Interactive Mechanism Between Inclusions and Hydrogen in Molten Aluminum. Chin. J. Nonferrous Met. 1999, 1, 51–56. [Google Scholar] [CrossRef]
- Zhou, D.S.; Long, W.; Xu, C.J.; Zhang, H.H. Hydrogen Separation Behavior of A356 Aluminum Alloy During Solidification. Heat Treat. 2013, 28, 33–36. [Google Scholar]
- Liu, Y.; Dai, Y.; Wang, J.; Shu, D.; Sun, B. Structure of Liquid Aluminum and Hydrogen Absorption. J. Wuhan Univ. Technol.—Mater. Sci. Ed. 2011, 26, 93–97. [Google Scholar] [CrossRef]
- Liu, Y.; Dai, Y.B.; Wang, J.; Shu, D.; Sun, B.D. Hydrogen Diffusion in Aluminum Melts: An ab initio Molecular Dynamics Study. J. Wuhan Univ. Technol.—Mater. Sci. Ed. 2012, 27, 560–567. [Google Scholar] [CrossRef]
- Fox, S.; Campbell, J. Visualisation of oxide film defects during solidification of aluminium alloys. Scr. Mater. 2000, 43, 881–886. [Google Scholar] [CrossRef]
- Raiszadeh, R.; Griffiths, W.D. The Effect of Holding Liquid Aluminum Alloys on Oxide Film Content. Metall. Mater. Trans. B 2011, 42, 133–143. [Google Scholar] [CrossRef]
- Raiszadeh, R.; Griffiths, W.D. A Semi-empirical Mathematical Model to Estimate the Duration of the Atmosphere within a Double Oxide Film Defect in Pure Aluminum Alloy. Metall. Mater. Trans. B 2008, 39, 298–303. [Google Scholar] [CrossRef]
- Scherf, A.; Janda, D.; Baghaie Yazdi, M.; Li, X.; Stein, F.; Heilmaier, M. Oxidation Behavior of Binary Aluminium-Rich Fe–Al Alloys with a Fine-Scaled, Lamellar Microstructure. Oxid. Met. 2015, 83, 559–574. [Google Scholar] [CrossRef]
- Shmelev, V.; Yang, H.; Yim, C. Hydrogen Generation by Reaction of Molten Aluminum with Water Steam. Int. J. Hydrogen Energy 2016, 41, 14562–14572. [Google Scholar] [CrossRef]
- Gyarmati, G.; Fegyverneki, G.; Mende, T.; Tokár, M. Characterization of the Double Oxide Film Content of Liquid Aluminum Alloys by Computed Tomography. Mater. Charact. 2019, 157, 109925. [Google Scholar] [CrossRef]
- Griffiths, W.D.; Caden, A.J.; El-Sayed, M.A. An Investigation into Double Oxide Film Defects in Aluminium Alloys. Mater. Sci. Forum 2014, 783–786, 142–147. [Google Scholar] [CrossRef]
- Hart, R.K. The Oxidation of Aluminium in Dry and Humid Oxygen Atmospheres. Proc. R. Soc. Lond. 1956, 236, 68–88. [Google Scholar]
- Zhang, Q.Y.; Sun, D.K.; Zhang, S.H.; Wang, H.; Zhu, M.F. Modeling of microporosity formation and hydrogen concentration evolution during solidification of an Al-Si alloy. Chin. Phys. B 2020, 29, 078104. [Google Scholar] [CrossRef]
- Dong, Y.H.; Shuai, S.S.; Zheng, T.X.; Cao, J.W.; Chen, C.Y. In-Situ observation of solid-liquid interface transition during directional solidification of Al-Zn alloy via X-ray Imaging. J. Mater. Sci. Technol. 2020, 39, 113–123. [Google Scholar] [CrossRef]
- Moore, B. Basic Physical Chemistry, 3rd ed.; Prentice-Hall: New York, NY, USA, 1983; pp. 217–298. [Google Scholar]
- Yang, Q.Q.; Liu, Y.; Li, Y.X. Hydrogen diffusion coefficient in liquid metals evaluated by solid–gas eutectic unidirectional solidification. Trans. Nonferrous Met. Soc. China 2014, 24, 4030–4037. [Google Scholar] [CrossRef]








| Elements | Si | Mg | Cu | Fe | Mn | Zn | Ti | Cr | Al |
|---|---|---|---|---|---|---|---|---|---|
| Composition | 0.85 | 0.73 | 0.58 | 0.38 | 0.25 | 0.13 | 0.08 | 0.07 | Bal. |
| Lots | Holding Temperature (K) | Relative Humidity (%) |
|---|---|---|
| A1 | 973 | 30 |
| A2 | 50 | |
| A3 | 80 | |
| A4 | 1023 | 30 |
| A5 | 50 | |
| A6 | 80 | |
| A7 | 1073 | 30 |
| A8 | 50 | |
| A9 | 80 |
| Equipment Name | Model | Manufacturer |
|---|---|---|
| Precision electronic balance | AG285 | Mettler Toledo, Zurich, Switzerland |
| Melting furnace | K4/13 | Nabertherm, Lilienthal, Germany |
| Electro-thermostatic blast oven | 101-1-BS | Shanghai Furnace, Shanghai, China |
| Optical microscope | DMRE | Leica, Wetzler, Germany |
| Hydrogen analyzer | Hyscan II | MechaTech, Bristol, UK |
| The high-temperature laser scanning confocal | VL2000DX-SVF18SP | Yonekura, Yokohama, Japan |
| Humidity adjusting device | — | — |
| Parameters | Measured Parameters | Calculated Parameters | ||||
|---|---|---|---|---|---|---|
| Lots | ||||||
| A1 | 4.5 | 5.7 | 3.754 | 1.046 | 5.207 | |
| A2 | 5.0 | 6.1 | 3.843 | 1.105 | 8.179 | |
| A3 | 5.5 | 6.6 | 4.249 | 1.228 | 5.729 | |
| A4 | 4.8 | 5.9 | 4.136 | 1.026 | 5.568 | |
| A5 | 5.6 | 6.7 | 4.561 | 1.026 | 6.125 | |
| A6 | 6.4 | 8.0 | 4.894 | 1.019 | 6.637 | |
| Temperature (K) | Relative Humidity (%) | Mathematical Model |
|---|---|---|
| 973 | 30 | |
| 50 | ||
| 80 | ||
| 1023 | 30 | |
| 50 | ||
| 80 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, B.; Lu, W.; Zhang, G.; Tan, Y.; Wu, X.; Hu, J.; Xu, Z.; Tang, H.; Wang, J.; Zhu, G.; et al. Kinetic Mechanism of Hydrogen Absorption of AA6111 Alloys Melt. Metals 2023, 13, 236. https://doi.org/10.3390/met13020236
Yang B, Lu W, Zhang G, Tan Y, Wu X, Hu J, Xu Z, Tang H, Wang J, Zhu G, et al. Kinetic Mechanism of Hydrogen Absorption of AA6111 Alloys Melt. Metals. 2023; 13(2):236. https://doi.org/10.3390/met13020236
Chicago/Turabian StyleYang, Bo, Weihong Lu, Guoqing Zhang, Yapeng Tan, Xiaocong Wu, Jiajin Hu, Zhengbing Xu, Hongqun Tang, Junsheng Wang, Guoliang Zhu, and et al. 2023. "Kinetic Mechanism of Hydrogen Absorption of AA6111 Alloys Melt" Metals 13, no. 2: 236. https://doi.org/10.3390/met13020236






