A Review on Processing–Microstructure–Property Relationships of Al-Si Alloys: Recent Advances in Deformation Behavior
Abstract
:1. Introduction
2. Advancements in Al-Si Alloys
2.1. Overview
2.2. Applications of Al-Si Alloys
2.2.1. Automotive Applications
Internal Combustion Engine Vehicles
Hybrid and Electric Vehicles
2.2.2. Aeronautical and Aerospace Applications
2.3. Achievements for the Pathway to Net-Zero Emissions

3. Fabrication Techniques
3.1. Metal Casting
3.1.1. Gravity Casting
Sand Casting and Precision Sand Casting
Permanent-Mold Casting
Lost Foam Casting
Investment Casting
3.1.2. High-Pressure Die-Casting
3.1.3. Low-Pressure Die-Casting
3.1.4. Squeeze Casting
3.2. Semi-Solid Metal Processing
3.3. Additive Manufacturing

3.4. Recycling Al-Si Alloys (Secondary Al Alloys)
3.5. Designated Heat Treatment for Al-Si Alloys
4. Microstructures
4.1. Binary Al-Si Alloy Systems
4.1.1. Major Alloying Elements
4.1.2. Impurities
4.2. Principles of Solidification
4.2.1. Solidification Sequence
4.2.2. Microstructural Evolution
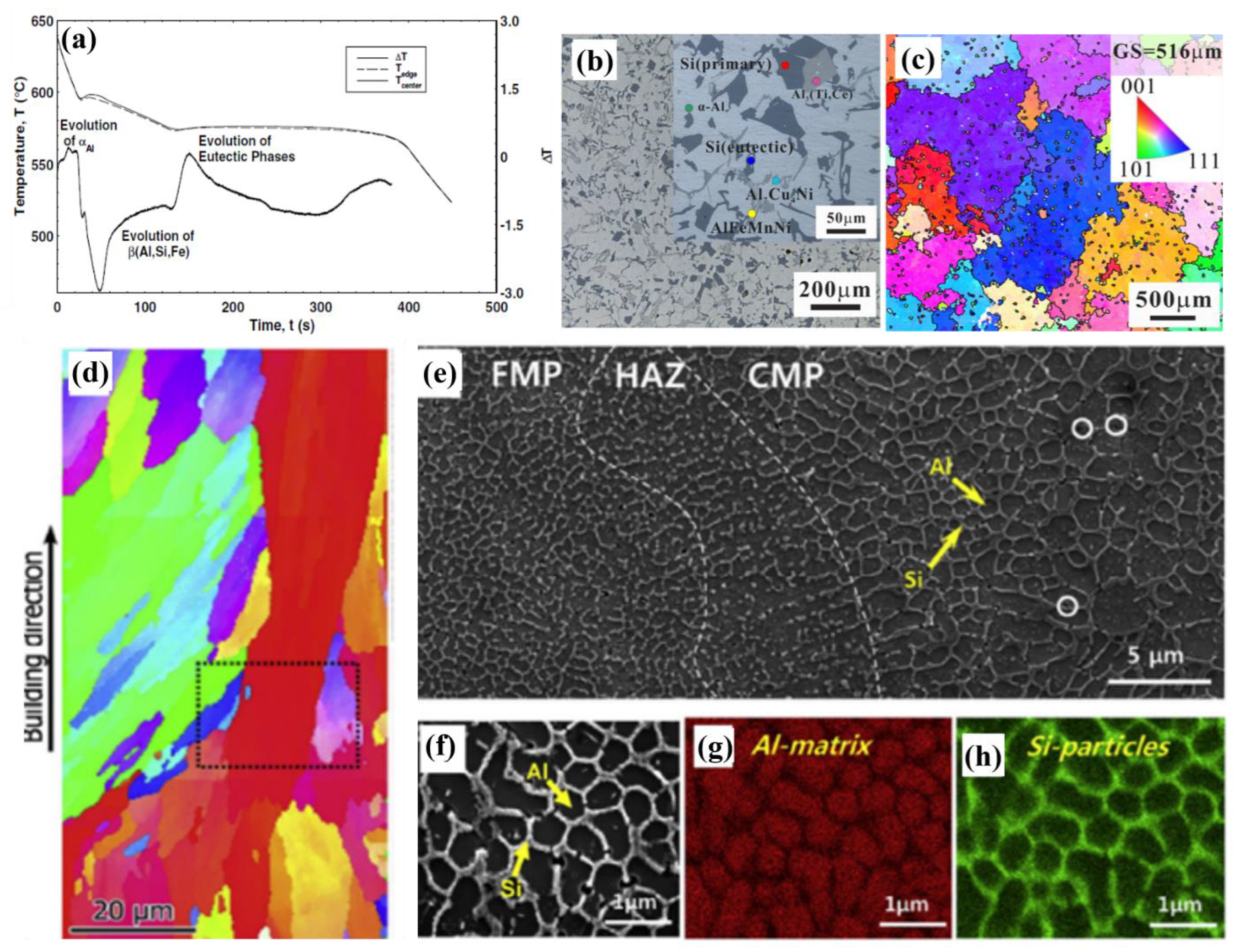
4.3. Grain Refinement
4.4. Modification of Si Morphology

4.5. Precipitation Phenomena in Al-Si Alloys
of metastable phase → Formation of equilibrium precipitate phase.
β″→ Rod-shaped β ′ → Plate-shaped β (Mg2Si) phase.
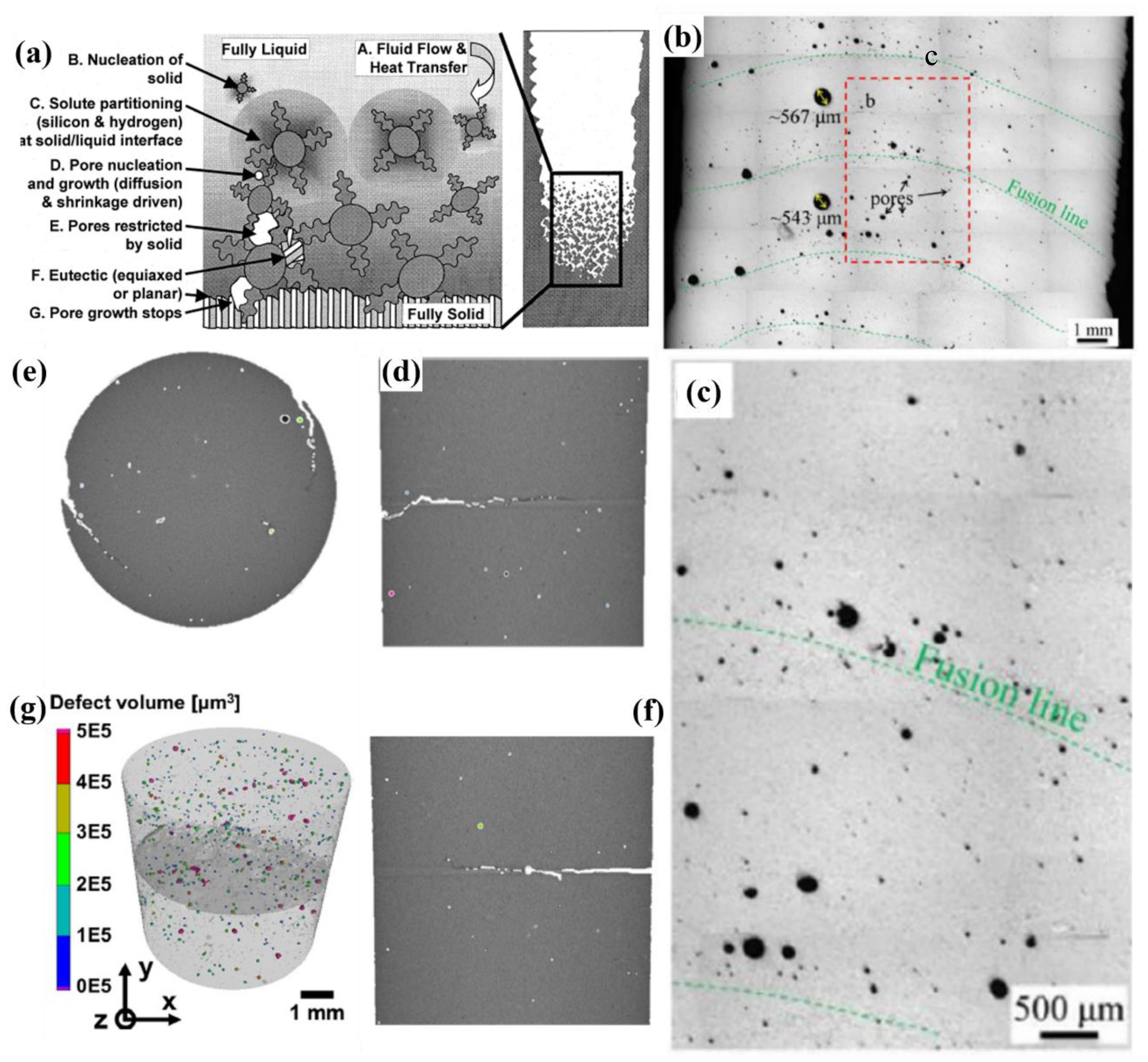
4.6. Porosity and Shrinkage Formation
5. Static Mechanical Properties
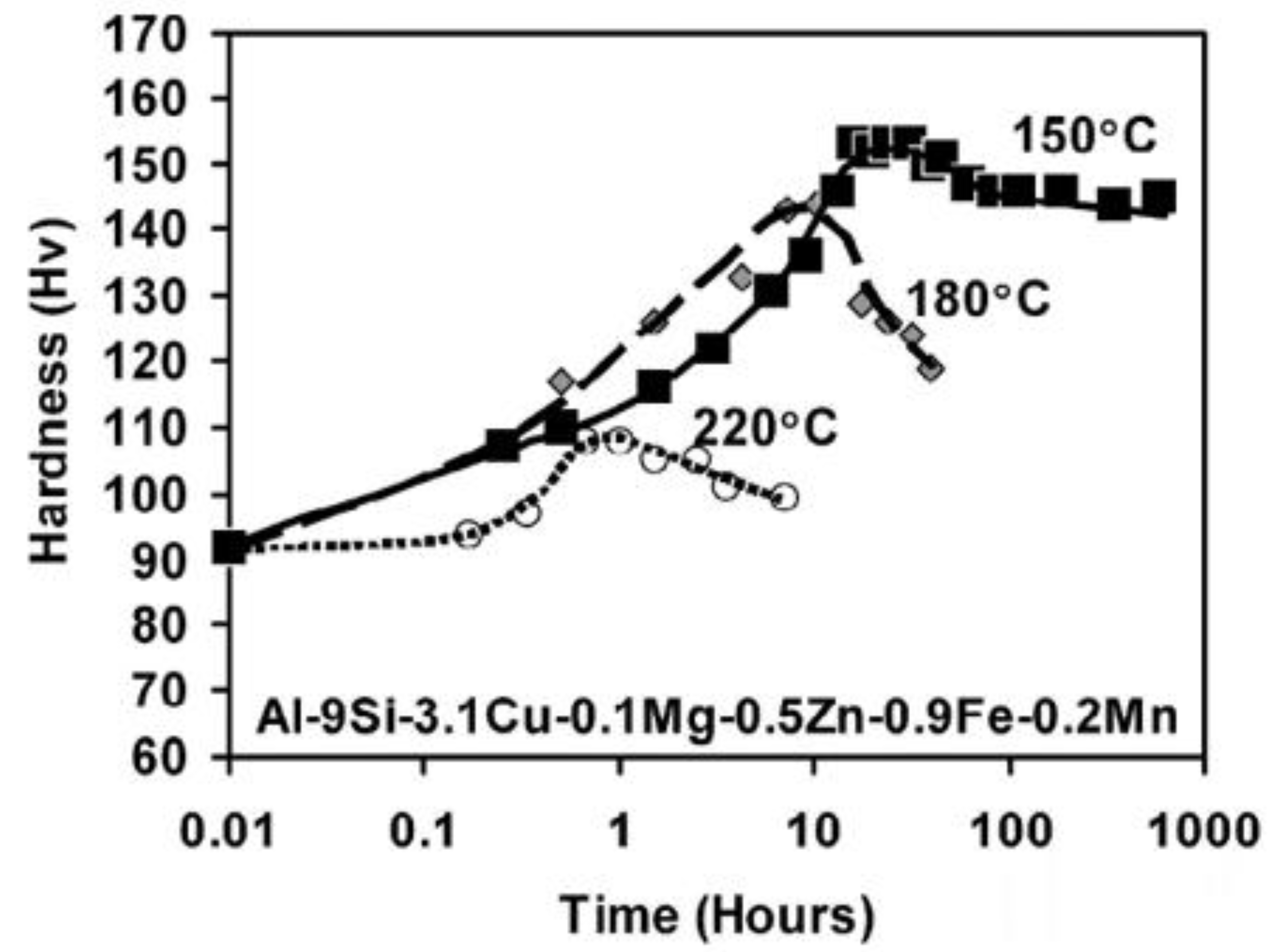
5.1. Elasticity and Hardness
5.2. Tensile and Compressive Properties
5.2.1. Effect of Si Content
5.2.2. Effect of Heat Treatment
5.2.3. Effect of Solidification Rate
5.2.4. Strain Hardening
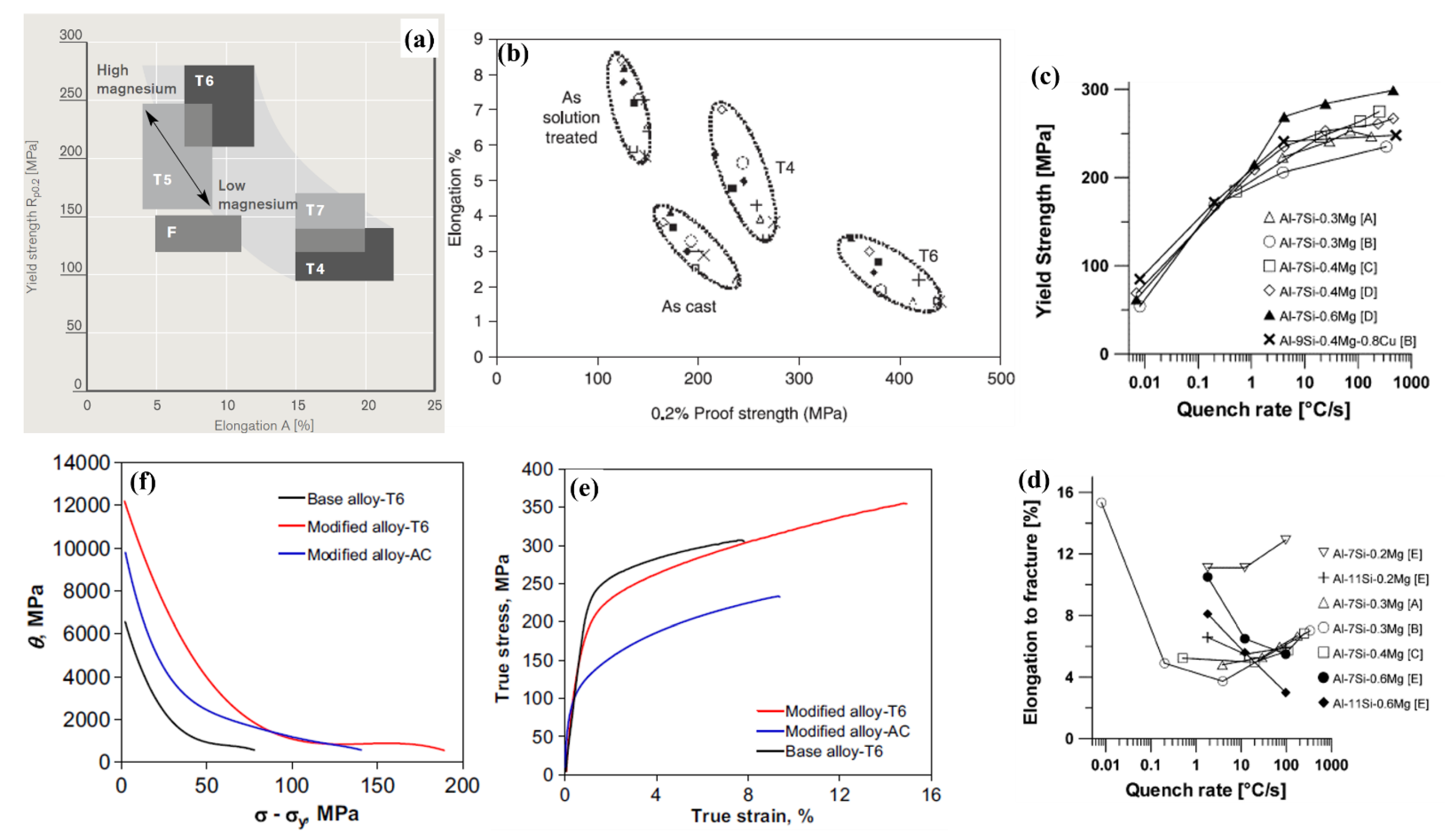
5.2.5. High Temperature Response
5.2.6. Fracture Behavior
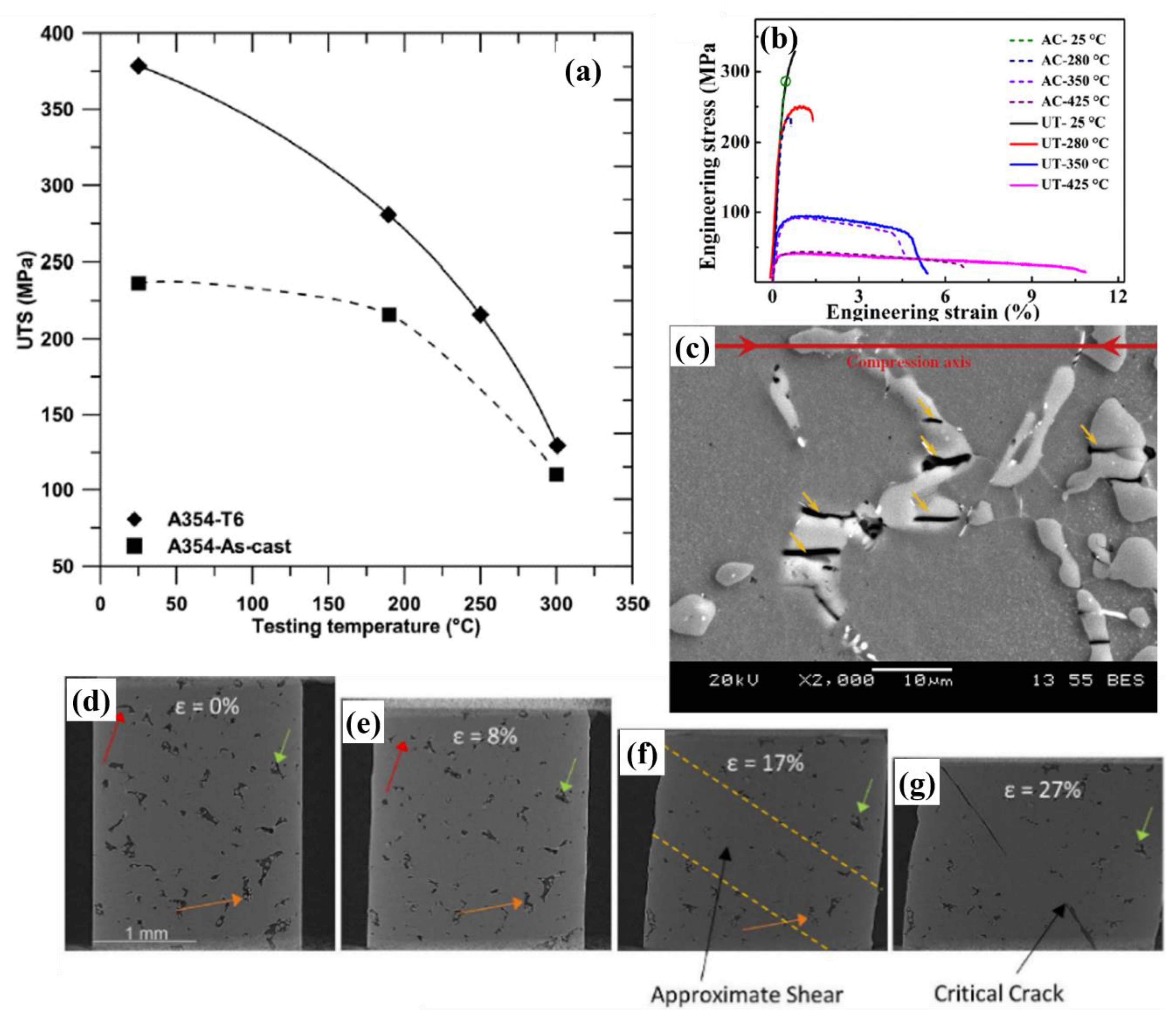
6. Fatigue Behavior of Al-Si Alloys
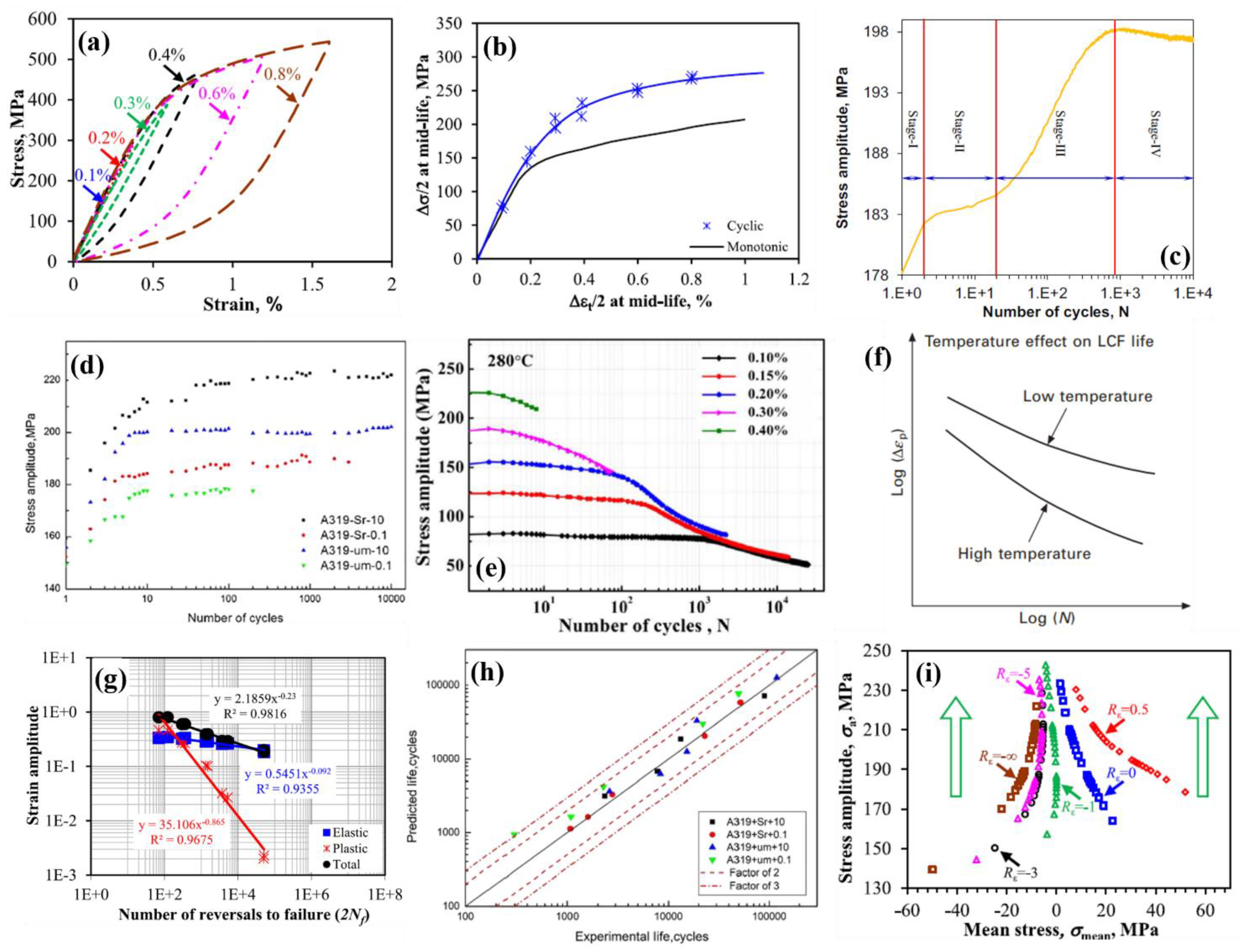
6.1. High-Cycle Fatigue
6.2. Low-Cycle Fatigue
6.2.1. Assessment of Damage in LCF
6.2.2. Existing Models to Predict Fatigue Life
6.3. Effect of Mean Stress
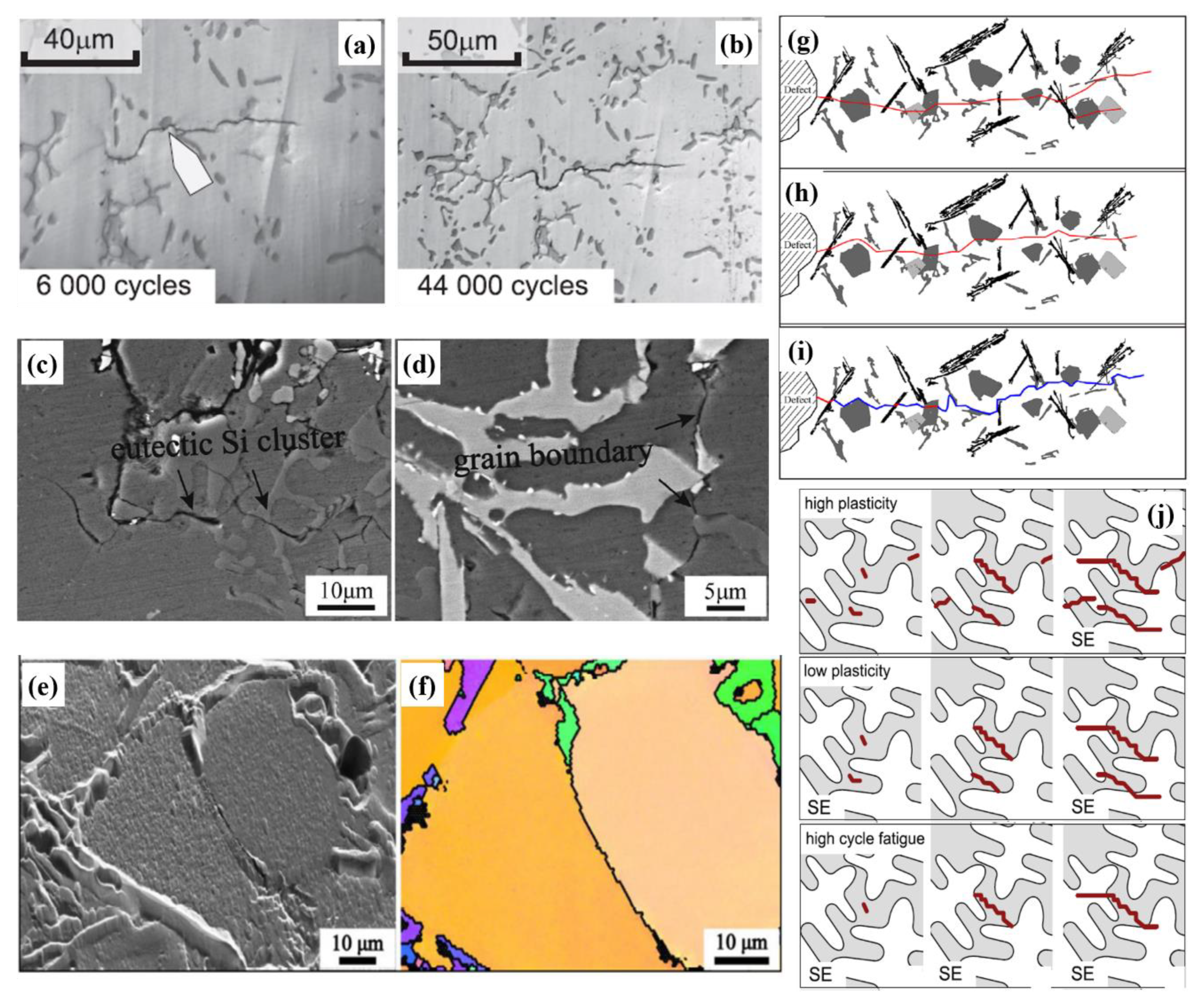
6.4. Fatigue Crack Initiation and Growth Mechanisms
7. Concluding Remarks and Future Outlook
7.1. Summary
- Al-Si alloys with other alloying elements, such as Cu, Mg, Mn, Ni, etc., show excellent castability and heat treatability. Impurities such as Fe in these alloys have a significant impact on ductility and fatigue properties, for which they should be minimized. With increasing demand for hybrid and EVs, newer Al-Si alloys with the addition of transition metals, rare-earth elements, Ag, Sn, Cu, etc., have been developed to improve the thermal stability and electrical conductivity, along with strength and elongation.
- The manufacturing practices of Al-Si alloys range considerably from casting to 3D printing (additively manufactured) alloys. Obtaining near-net-shaped products can be seen as a major objective for automotive and aerospace castings. Pressure die-casting techniques such as HPDC and LPDC, along with semi-solid metal processes, produce a variety of alloys with different microstructural features, depending upon mold parameters, cooling rate, vibration dynamics, etc., which control alloy performance.
- Emerging techniques of additive manufacturing, such as WAAM, L-PBF, and others have also led to cellular microstructures that behave differently than cast alloys under mechanical loading. However, porosity issues in these alloys currently limit their applicability at an industrial scale.
- The mechanical properties of these alloys under static and cyclic loading are closely related to their microstructures, intermetallics, porosities, surface features, and casting defects, etc. The presence of an Al-Si eutectic structure (modified or unmodified) imitates the behavior of particulate-reinforced composites, where the morphology of Si particles controls the failure mechanisms of the alloy. Like composite materials, the enhancement of Young’s modulus is a feature of Al-Si alloys with a Si content of >5%, which is controlled by the volume fraction of eutectic Si particles.
- High-cycle and low-cycle fatigue mechanisms of alloys vary based on alloy composition, production route, sample size, heat-treatment conditions, and microstructures. It has been observed in many studies that alloys with higher static ductility perform better under strain-controlled LCF, while stress-controlled HCF strength is mainly associated with its static strength.
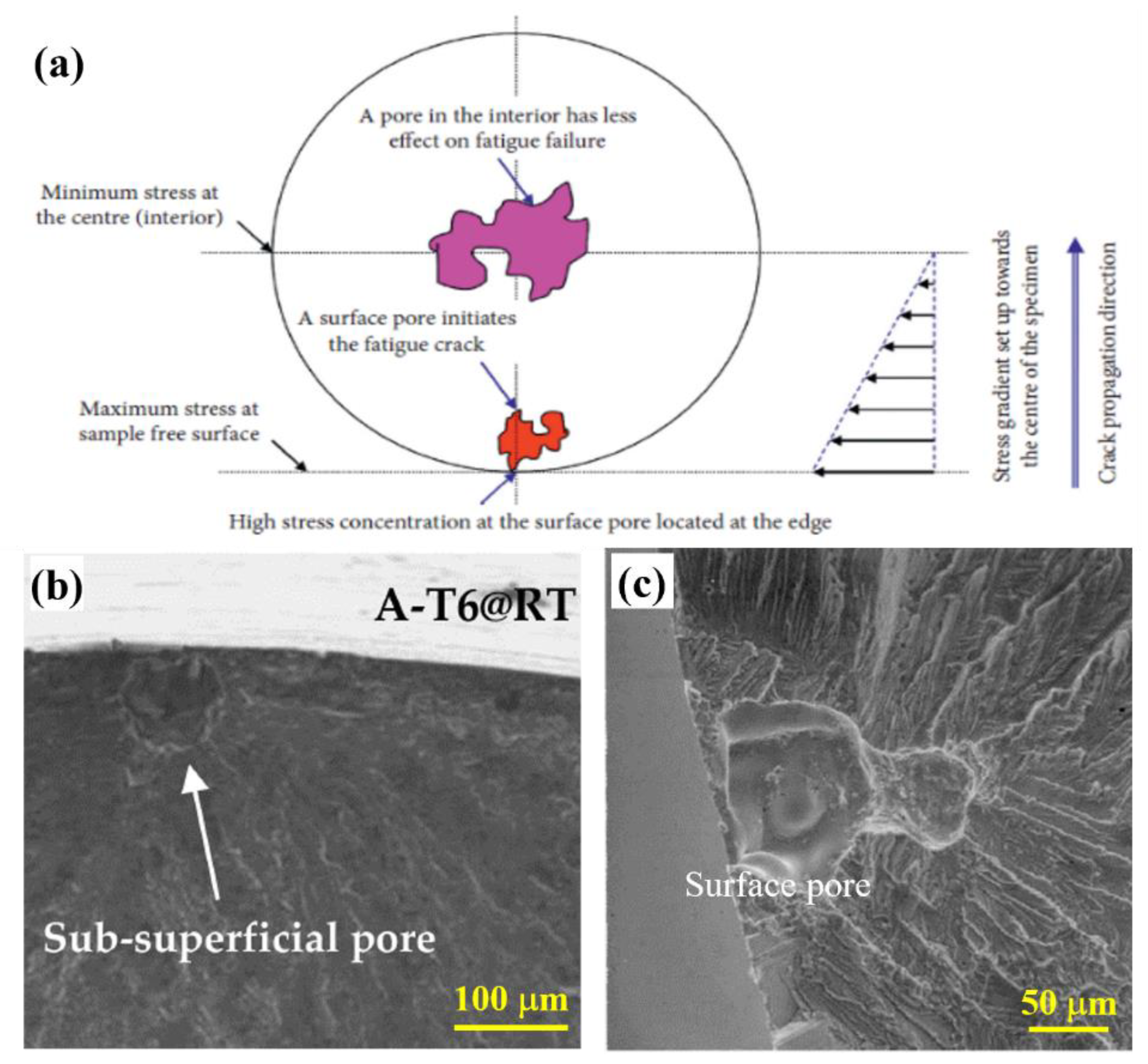
- Damage mechanisms under fatigue were assessed based on the strain-energy density stored in the material during cyclic loading. Initiation of fatigue cracks occurred from multiple slip band intrusions and extrusions formed on the surface of the alloys due to the higher stress concentration. Failure also heavily depends upon the amount of surface or near-surface defects in the fabricated alloys. Methods to minimize the presence of defects in fabricated Al-Si alloys include controlling the solidification rate, alloy composition, surface treatment, eutectic morphology, intermetallics, and avoiding unwanted pores.
7.2. Future Outlook
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Net Zero by 2050. Available online: https://www.iea.org/reports/net-zero-by-2050 (accessed on 20 January 2023).
- Luo, A.A.; Sachdev, A.K.; Apelian, D. Alloy Development and Process Innovations for Light Metals Casting. J. Mater. Process. Technol. 2022, 306, 117606. [Google Scholar] [CrossRef]
- Net-Zero Emissions by 2050. Available online: https://www.canada.ca/en/services/environment/weather/climatechange/climate-plan/net-zero-emissions-2050.html (accessed on 20 January 2023).
- For a Livable Climate: Net-Zero Commitments Must Be Backed by Credible Action. Available online: https://www.un.org/en/climatechange/net-zero-coalition (accessed on 20 January 2023).
- Tran, M.-K.; Bhatti, A.; Vrolyk, R.; Wong, D.; Panchal, S.; Fowler, M.; Fraser, R. A Review of Range Extenders in Battery Electric Vehicles: Current Progress and Future Perspectives. World Electr. Veh. J. 2021, 12, 54. [Google Scholar] [CrossRef]
- Riley, C. The Great Electric Car Race Is Just Beginning. Available online: https://www.cnn.com/interactive/2019/08/business/electric-cars-audi-volkswagen-tesla/ (accessed on 20 January 2023).
- DuckerFrontier. 2020 North America Light Vehicle Aluminum Content and Outlook; DuckerFrontier: Washington, DC, USA, 2020. [Google Scholar]
- Aluminum Market Size And Forecast. Available online: https://www.verifiedmarketresearch.com/product/aluminum-market/ (accessed on 20 January 2023).
- Automotive Aluminum Market Size, Share & Trends Analysis Report by End Use (Passenger Cars, Light Commercial Vehicles, Heavy Commercial Vehicles), by Application, and Segment Forecasts, 2019–2025. Available online: https://www.grandviewresearch.com/industry-analysis/automotive-aluminum-market (accessed on 20 January 2023).
- Djukanovic, G. International Aluminium 2019 Conference—Key Takeaways. Available online: https://aluminiuminsider.com/ (accessed on 20 January 2023).
- Aluminium Alloys Market. Available online: https://www.factmr.com/report/aluminium-alloys-market (accessed on 20 January 2023).
- Hybrid Electric Vehicles Market. Available online: https://www.factmr.com/report/270/hybrid-electric-vehicles-market (accessed on 20 January 2023).
- Aluminum Silicon Alloy Market Overview. Available online: https://dataintelo.com/report/aluminum-silicon-alloy-market/ (accessed on 20 January 2023).
- Berlanga-Labari, C.; Biezma-Moraleda, M.V.; Rivero, P.J. Corrosion of Cast Aluminum Alloys: A Review. Metals 2020, 10, 1384. [Google Scholar] [CrossRef]
- Santamaría, J.A.; Sertucha, J.; Redondo, A.; Lizarralde, I.; Ochoa de Zabalegui, E.; Rodríguez, P. Towards the Prediction of Tensile Properties in Automotive Cast Parts Manufactured by LPDC with the A356.2 Alloy. Metals 2022, 12, 656. [Google Scholar] [CrossRef]
- Ganesh, M.R.S.; Reghunath, N.; Levin, M.J.; Prasad, A.; Doondi, S.; Shankar, K.V. Strontium in Al–Si–Mg Alloy: A Review. Met. Mater. Int. 2022, 28, 1–40. [Google Scholar] [CrossRef]
- Gursoy, O.; Timelli, G. Lanthanides: A Focused Review of Eutectic Modification in Hypoeutectic Al–Si Alloys. J. Mater. Res. Technol. 2020, 9, 8652–8666. [Google Scholar] [CrossRef]
- Adamane, A.R.; Arnberg, L.; Fiorese, E.; Timelli, G.; Bonollo, F. Influence of Injection Parameters on the Porosity and Tensile Properties of High-Pressure Die Cast Al-Si Alloys: A Review. Int. J. Met. 2015, 9, 43–53. [Google Scholar] [CrossRef]
- Vijeesh, V.; Narayan Prabhu, K. Review of Microstructure Evolution in Hypereutectic Al–Si Alloys and Its Effect on Wear Properties. Trans. Indian Inst. Met. 2014, 67, 1–18. [Google Scholar] [CrossRef]
- Campbell, J.; Tiryakioğlu, M. Review of Effect of P and Sr on Modification and Porosity Development in Al–Si Alloys. Mater. Sci. Technol. 2010, 26, 262–268. [Google Scholar] [CrossRef]
- Campbell, J. Perspective Chapter: A Personal Overview of Casting Processes. In Casting Processes and Modelling of Metallic Materials; IntechOpen: London, UK, 2021. [Google Scholar]
- Viswanathan, S.; Apelian, D.; DasGupta, R.; Gywn, M.; Jorstad, J.L.; Monroe, R.W. (Eds.) ASM Handbook Volume 15: Casting; ASM International: Novelty, OH, USA, 2008; ISBN 978-0-87170-711-6. [Google Scholar]
- Rowe, J. Advanced Materials in Automotive Engineering; Woodhead Publishing: Cambridge, UK, 2012; ISBN 9781845695613. [Google Scholar]
- Robles Hernandez, F.C.; Herrera Ramírez, J.M.; Mackay, R. Applications in the Automotive and Aerospace Industries. In Al-Si Alloys; Springer International Publishing: Cham, Switzerland, 2017; pp. 163–171. [Google Scholar]
- Afroz, L.; Das, R.; Qian, M.; Easton, M.; Brandt, M. Fatigue Behaviour of Laser Powder Bed Fusion (L-PBF) Ti–6Al–4V, Al–Si–Mg and Stainless Steels: A Brief Overview. Int. J. Fract. 2022, 235, 3–46. [Google Scholar] [CrossRef]
- Li, Y.; Hu, A.; Fu, Y.; Liu, S.; Shen, W.; Hu, H.; Nie, X. Al Alloys and Casting Processes for Induction Motor Applications in Battery-Powered Electric Vehicles: A Review. Metals 2022, 12, 216. [Google Scholar] [CrossRef]
- Czerwinski, F. Thermal Stability of Aluminum Alloys. Materials 2020, 13, 3441. [Google Scholar] [CrossRef]
- Czerwinski, F. Cerium in Aluminum Alloys. J. Mater. Sci. 2020, 55, 24–72. [Google Scholar] [CrossRef]
- Robles Hernandez, F.C.; Herrera Ramírez, J.M.; Mackay, R. Al-Si Alloys; Springer International Publishing: Cham, Switzerland, 2017; ISBN 978-3-319-58379-2. [Google Scholar]
- Brochu, M.; Verreman, Y.; Ajersch, F.; Bucher, L. Fatigue Behavior of Semi-Solid Cast Aluminum: A Critical Review. Solid State Phenom. 2008, 141–143, 725–730. [Google Scholar] [CrossRef]
- Koutiri, I.; Bellett, D.; Morel, F.; Augustins, L.; Adrien, J. High Cycle Fatigue Damage Mechanisms in Cast Aluminium Subject to Complex Loads. Int. J. Fatigue 2013, 47, 44–57. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, R.T.; Imholte, D.D.; Rios, O.R.; Weiss, D.; Sims, Z.; Stromme, E.; McCall, S.K. Anticipating Impacts of Introducing Aluminum-Cerium Alloys into the United States Automotive Market. Resour. Conserv. Recycl. 2019, 144, 340–349. [Google Scholar] [CrossRef]
- Javidani, M.; Larouche, D. Application of Cast Al–Si Alloys in Internal Combustion Engine Components. Int. Mater. Rev. 2014, 59, 132–158. [Google Scholar] [CrossRef]
- Torres, R.; Esparza, J.; Velasco, E.; Garcia-Luna, S.; Colas, R. Characterisation of an Aluminium Engine Block. Int. J. Microstruct. Mater. Prop. 2006, 1, 129. [Google Scholar] [CrossRef]
- Fan, Y.; Makhlouf, M.M. The Al-Al3Ni Eutectic Reaction: Crystallography and Mechanism of Formation. Metall. Mater. Trans. A 2015, 46, 3808–3812. [Google Scholar] [CrossRef]
- Ye, H. An Overview of the Development of Al-Si-Alloy Based Material for Engine Applications. J. Mater. Eng. Perform. 2003, 12, 288–297. [Google Scholar] [CrossRef]
- Czerwinski, F. Current Trends in Automotive Lightweighting Strategies and Materials. Materials 2021, 14, 6631. [Google Scholar] [CrossRef] [PubMed]
- Czerwinski, F.; Kasprzak, W.; Sediako, D.; Emadi, D.; Shaha, S.; Friedman, J.; Chen, D.L. High-Temperature Aluminum Alloys for Automotive Powertrains; Cast Aluminum Alloys Were Developed with High-Temperature Tensile and Fatigue Strengths to Withstand Elevated-Temperature Applications in Modern Engines. Adv. Mater. Process. 2016, 174, 16–21. [Google Scholar]
- Esmeralda, A.G.; Arenas-García, H.; Rodríguez, A.F.; Talamantes-Silva, J.; Torres, R.; Garza-Montes-de-Oca, N.F.; Colás, R. Thermal Diffusivity of Al-Si Cast Alloys for Internal Combustion Engines. Thermochim. Acta 2019, 675, 172–179. [Google Scholar] [CrossRef]
- Rheinfelden Alloys GmbH & Co. KG. Alloys Alloys for High Pressure Die Casting; Rheinfelden Alloys GmbH & Co. KG: Rheinfelden, Germany, 2015; pp. 1–60. [Google Scholar]
- Hartlieb, M. Aluminum Alloys for Structural Die Casting. Die Cast. Eng. 2013, 57, 40–43. [Google Scholar]
- Pezda, J.; Jezierski, J. Non-Standard T6 Heat Treatment of the Casting of the Combustion Engine Cylinder Head. Materials 2020, 13, 4114. [Google Scholar] [CrossRef] [PubMed]
- Aluminium in Cars: Unlocking the Lightweighting Potential; Belgium. 2021. Available online: https://european-aluminium.eu/wp-content/uploads/2022/10/aluminium-in-cars-unlocking-the-lightweighting-potential.pdf (accessed on 20 January 2023).
- Baser, T.A.; Umay, E.; Akinci, V. New Trends in Aluminum Die Casting Alloys for Automotive Applications. Eurasia Proc. Sci. Technol. Eng. Math. 2022, 21, 79–87. [Google Scholar] [CrossRef]
- Constellium Supplying Aluminum Solutions for Audi E-Tron GT EV. Available online: https://www.greencarcongress.com/2021/06/20210630-constellium.html (accessed on 20 January 2023).
- LGC-Industrials Electric Vehicles: Making Them Lighter, Safer and More Efficient with Aluminum Alloys. Available online: https://www.armi.com/blog/electric-vehicles-making-them-lighter-safer-and-more-efficient-with-aluminum-alloys (accessed on 20 January 2023).
- Gomes, L.F.; Silva, B.L.; da Silva, P.S., Jr.; Garcia, A.; Spinelli, J.E. Ag-Containing Aluminum-Silicon Alloys as an Alternative for as-Cast Components of Electric Vehicles. Mater. Res. Express 2021, 8, 016527. [Google Scholar] [CrossRef]
- Lambert, F. Tesla Invents New Aluminum Alloys for Die Casting Electric Car Parts. Available online: https://electrek.co/2020/02/07/tesla-aluminum-alloys-die-casting-in-electric-car-parts/ (accessed on 20 January 2023).
- Gialanella, S.; Malandruccolo, A. Aerospace Alloys; Topics in Mining, Metallurgy and Materials Engineering; Springer International Publishing: Cham, Switzerland, 2020; ISBN 978-3-030-24439-2. [Google Scholar]
- Zaporozhets, O.; Isaienko, V.; Synylo, K. PARE Preliminary Analysis of ACARE FlightPath 2050 Environmental Impact Goals. CEAS Aeronaut. J. 2021, 12, 653–667. [Google Scholar] [CrossRef]
- Svendsen, A. Aluminum Continues Unprecedented Growth in Automotive Applications. Available online: https://www.lightmetalage.com/news/industry-news/automotive/aluminum-continues-unprecedented-growth-in-automotive-applications/ (accessed on 20 January 2023).
- CO2 Performance of New Passenger Cars in Europe. Available online: https://www.eea.europa.eu/ims/co2-performance-of-new-passenger (accessed on 20 January 2023).
- Robles Hernandez, F.C.; Herrera Ramírez, J.M.; Mackay, R. Metal Casting Process. In Al-Si Alloys; Springer International Publishing: Cham, Switzerland, 2017; pp. 49–81. [Google Scholar]
- Favi, C.; Germani, M.; Mandolini, M. Analytical Cost Estimation Model in High Pressure Die Casting. Procedia Manuf. 2017, 11, 526–535. [Google Scholar] [CrossRef]
- MacKay, R.; Szablewski, D. The Use of the Weibull Statistical Method to Assess the Reliability of a Development Engineered Casting Component. Int. J. Met. 2010, 4, 31–45. [Google Scholar] [CrossRef]
- El Khoukhi, D.; Morel, F.; Saintier, N.; Bellett, D.; Osmond, P.; Le, V.-D.; Adrien, J. Scatter and Size Effect in High Cycle Fatigue of Cast Aluminum-Silicon Alloys: A Comprehensive Experimental Investigation. Procedia Struct. Integr. 2022, 38, 611–620. [Google Scholar] [CrossRef]
- Azadi, M.; Bahmanabadi, H.; Gruen, F.; Winter, G. Evaluation of Tensile and Low-Cycle Fatigue Properties at Elevated Temperatures in Piston Aluminum-Silicon Alloys with and without Nano-Clay-Particles and Heat Treatment. Mater. Sci. Eng. A 2020, 788, 139497. [Google Scholar] [CrossRef]
- Liu, H.; Pang, J.; Wang, M.; Li, S.; Zhang, Z. High-Cycle Fatigue Behavior and Damage Mechanism of Multiphase Al-Si Piston Alloy at Room and Elevated Temperatures. Adv. Eng. Mater. 2018, 20, 1700972. [Google Scholar] [CrossRef]
- Campbell, J. Stop Pouring, Start Casting. Int. J. Met. 2012, 6, 7–18. [Google Scholar] [CrossRef]
- Farhang Mehr, F.; Cockcroft, S.; Reilly, C.; Maijer, D. Investigation of the Efficacy of a Water-Cooled Chill on Enhancing Heat Transfer at the Casting-Chill Interface in a Sand-Cast A319 Engine Block. J. Mater. Process. Technol. 2020, 286, 116789. [Google Scholar] [CrossRef]
- Ge, B.; Liu, X.; He, G.; Le, P.; Wen, Z.; Wang, Q. Quantitative Relationship between Microstructure Characteristics and Fatigue Parameters of A319 Casting Alloy. Fatigue Fract. Eng. Mater. Struct. 2020, 43, 605–616. [Google Scholar] [CrossRef]
- Zhou, Z.; Liu, X.; He, G.; Liao, Y.; Huang, Z.; Pan, J.; Li, J.; Wang, Q. A Comparison of Uniaxial and Multiaxial Non-Proportional Fatigue Properties in Cast Al-Si-Cu-T6 Alloys Solidified at Two Cooling Rates: Fatigue Behavior, Fracture Characteristics and Dislocation Evolution. Mater. Charact. 2022, 189, 111957. [Google Scholar] [CrossRef]
- Die Casting-Design Guide, Materials, Advantages and Disadvantages. Available online: https://www.engineeringclicks.com/die-casting/ (accessed on 20 January 2023).
- Elhadari, H.A.; Patel, H.A.; Chen, D.L.; Kasprzak, W. Tensile and Fatigue Properties of a Cast Aluminum Alloy with Ti, Zr and V Additions. Mater. Sci. Eng. A 2011, 528, 8128–8138. [Google Scholar] [CrossRef]
- De Mori, A.; Timelli, G.; Fabrizi, A.; Berto, F. Influence of Cu Content on the Microstructure and High-Temperature Tensile and Fatigue Properties of Secondary AlSi7Mg0.3VZr Alloys. Mater. Sci. Eng. A 2021, 816, 141310. [Google Scholar] [CrossRef]
- Tenkamp, J.; Koch, A.; Knorre, S.; Krupp, U.; Michels, W.; Walther, F. Influence of the Microstructure on the Cyclic Stress-Strain Behaviour and Fatigue Life in Hypo-Eutectic Al-Si-Mg Cast Alloys. MATEC Web Conf. 2018, 165, 15004. [Google Scholar] [CrossRef]
- Dos Santos, A.; Hosdez, J.; Limodin, N.; El Bartali, A.; Tandjaoui, A.; Witz, J.-F.; Niclaeys, C.; Quaegebeur, P.; Najjar, D. 2D and 3D Characterization of Damage Mechanisms in A319 Alloy. Exp. Mech. 2022, 63, 377–382. [Google Scholar] [CrossRef]
- Dezecot, S.; Maurel, V.; Buffiere, J.-Y.; Szmytka, F.; Koster, A. 3D Characterization and Modeling of Low Cycle Fatigue Damage Mechanisms at High Temperature in a Cast Aluminum Alloy. Acta Mater. 2017, 123, 24–34. [Google Scholar] [CrossRef]
- Pattnaik, S.; Karunakar, D.B.; Jha, P.K. Developments in Investment Casting Process—A Review. J. Mater. Process. Technol. 2012, 212, 2332–2348. [Google Scholar] [CrossRef]
- Investment Casting. Available online: https://thelibraryofmanufacturing.com/investment_casting.html (accessed on 20 January 2023).
- Siaminwe, L.; Clegg, A.J. Effect of Processing Variables on Structure and Tensile Properties of Investment Cast Al–Si–Mg Casting Alloy. Mater. Sci. Technol. 1999, 15, 812–820. [Google Scholar] [CrossRef]
- Dezecot, S.; Brochu, M. Microstructural Characterization and High Cycle Fatigue Behavior of Investment Cast A357 Aluminum Alloy. Int. J. Fatigue 2015, 77, 154–159. [Google Scholar] [CrossRef]
- Schmahl, M.; Märten, A.; Zaslansky, P.; Fleck, C. Nanofatigue Behaviour of Single Struts of Cast A356.0 Foam: Cyclic Deformation, Nanoindent Characteristics and Sub-Surface Microstructure. Mater. Des. 2020, 195, 109016. [Google Scholar] [CrossRef]
- Barbosa, J.; Puga, H. Ultrasonic Melt Processing in the Low Pressure Investment Casting of Al Alloys. J. Mater. Process. Technol. 2017, 244, 150–156. [Google Scholar] [CrossRef]
- Lim, C.S.; Clegg, A.J.; Loh, N.L. The Reduction of Dendrite ARM Spacing Using a Novel Pressure-Assisted Investment Casting Approach. J. Mater. Process. Technol. 1997, 70, 99–102. [Google Scholar] [CrossRef]
- Niu, X.P.; Hu, B.H.; Pinwill, I.; Li, H. Vacuum Assisted High Pressure Die Casting of Aluminium Alloys. J. Mater. Process. Technol. 2000, 105, 119–127. [Google Scholar] [CrossRef]
- Wang, J.; Jiao, X.Y.; Xie, H.; Deng, B.; Xiong, S.M. Crack Configuration Feature and Fracture Surface Difference for High Pressure Die Casting Hypereutectic Al-Si Alloys in High Cycle Fatigue. Int. J. Fatigue 2021, 153, 106469. [Google Scholar] [CrossRef]
- Lumley, R.N. Progress on the Heat Treatment of High Pressure Die Castings. In Fundamentals of Aluminium Metallurgy; Elsevier: Amsterdam, The Netherlands, 2011; pp. 262–303. [Google Scholar]
- Lumley, R.N.; Deeva, N.; Larsen, R.; Gembarovic, J.; Freeman, J. The Role of Alloy Composition and T7 Heat Treatment in Enhancing Thermal Conductivity of Aluminum High Pressure Diecastings. Metall. Mater. Trans. A 2013, 44, 1074–1086. [Google Scholar] [CrossRef]
- Lumley, R.N.; Griffiths, J.R. Fatigue Resistance of Heat Treated Aluminium High Pressure Die-Castings. Adv. Mater. Res. 2008, 41–42, 99–104. [Google Scholar] [CrossRef]
- Lumley, R. The Development of High Strength and Ductility in High-Pressure Die-Cast Al-Si-Mg Alloys from Secondary Sources. JOM 2019, 71, 382–390. [Google Scholar] [CrossRef]
- Yan, F. Development of High Strength Al-Mg2Si-Mg Based Alloy for High Pressure Diecasting Process. Ph.D. Thesis, Brunel University, London, UK, 2014. [Google Scholar]
- Musk, E. Tesla Annual Shareholder Meeting Presentation. In Proceedings of the Tesla Annual Shareholder Meeting, Fremont, CA, USA, 22 September 2020. [Google Scholar]
- Casarotto, F.; Franke, A.J.; Franke, R. High-Pressure Die-Cast (HPDC) Aluminium Alloys for Automotive Applications. In Advanced Materials in Automotive Engineering; Elsevier: Amsterdam, The Netherlands, 2012; pp. 109–149. [Google Scholar]
- Dash, S.S.; Li, D.J.; Zeng, X.Q.; Li, D.Y.; Chen, D.L. Low-Cycle Fatigue Behavior of Silafont®-36 Automotive Aluminum Alloy: Effect of Negative Strain Ratio. Mater. Sci. Eng. A 2022, 852, 143701. [Google Scholar] [CrossRef]
- Dash, S.S.; Li, D.J.; Zeng, X.Q.; Li, D.Y.; Chen, D.L. Cyclic Deformation Behavior and Fatigue Life Prediction of an Automotive Cast Aluminum Alloy: A New Method of Determining Intrinsic Fatigue Toughness. Fatigue Fract. Eng. Mater. Struct. 2022, 45, 725–738. [Google Scholar] [CrossRef]
- Dash, S.S.; Li, D.J.; Zeng, X.Q.; Chen, D.L. Heterogeneous Microstructure and Deformation Behavior of an Automotive Grade Aluminum Alloy. J. Alloys Compd. 2021, 870, 159413. [Google Scholar] [CrossRef]
- Fu, P.; Luo, A.A.; Jiang, H.; Peng, L.; Yu, Y.; Zhai, C.; Sachdev, A.K. Low-Pressure Die Casting of Magnesium Alloy AM50: Response to Process Parameters. J. Mater. Process. Technol. 2008, 205, 224–234. [Google Scholar] [CrossRef]
- Jorstad, J.L. Permanent Mold: Casting Processes. Adv. Mater. Process. 2008, 166, 30–34. [Google Scholar]
- Counter Pressure Technology. Available online: http://cpcmachines.com/technology/ (accessed on 20 January 2023).
- Jiang, W.; Fan, Z.; Liu, D.; Wu, H. Influence of Gas Flowrate on Filling Ability and Internal Quality of A356 Aluminum Alloy Castings Fabricated Using the Expendable Pattern Shell Casting with Vacuum and Low Pressure. Int. J. Adv. Manuf. Technol. 2013, 67, 2459–2468. [Google Scholar] [CrossRef]
- Zhang, B.; Maijer, D.M.; Cockcroft, S.L. Development of a 3-D Thermal Model of the Low-Pressure Die-Cast (LPDC) Process of A356 Aluminum Alloy Wheels. Mater. Sci. Eng. A 2007, 464, 295–305. [Google Scholar] [CrossRef]
- Lee, C. Do Effects of Microporosity on Tensile Properties of A356 Aluminum Alloy. Mater. Sci. Eng. A 2007, 464, 249–254. [Google Scholar] [CrossRef]
- Miller, A.E.; Maijer, D.M. Investigation of Erosive-Corrosive Wear in the Low Pressure Die Casting of Aluminum A356. Mater. Sci. Eng. A 2006, 435–436, 100–111. [Google Scholar] [CrossRef] [Green Version]
- Song, J.-Y.; Park, J.-C.; Jeong, B.-H.; Ahn, Y.-S. Fatigue Behaviour of A356 Aluminium Alloy for Automotive Wheels. Int. J. Cast Met. Res. 2012, 25, 26–30. [Google Scholar] [CrossRef]
- Merlin, M.; Timelli, G.; Bonollo, F.; Garagnani, G.L. Impact Behaviour of A356 Alloy for Low-Pressure Die Casting Automotive Wheels. J. Mater. Process. Technol. 2009, 209, 1060–1073. [Google Scholar] [CrossRef]
- Huang, J.-M.; Zhao, H.-D.; Chen, Z.-M. Microstructure and Properties of A356 Alloy Wheels Fabricated by Low-Pressure Die Casting with Local Squeeze. J. Mater. Eng. Perform. 2019, 28, 2137–2146. [Google Scholar] [CrossRef]
- Lee, K.; Kwon, Y.N.; Lee, S. Effects of Eutectic Silicon Particles on Tensile Properties and Fracture Toughness of A356 Aluminum Alloys Fabricated by Low-Pressure-Casting, Casting-Forging, and Squeeze-Casting Processes. J. Alloys Compd. 2008, 461, 532–541. [Google Scholar] [CrossRef]
- Jiang, W.; Fan, Z.; Liu, D.; Liao, D.; Dong, X.; Zong, X. Correlation of Microstructure with Mechanical Properties and Fracture Behavior of A356-T6 Aluminum Alloy Fabricated by Expendable Pattern Shell Casting with Vacuum and Low-Pressure, Gravity Casting and Lost Foam Casting. Mater. Sci. Eng. A 2013, 560, 396–403. [Google Scholar] [CrossRef]
- Park, C.; Kim, S.; Kwon, Y.; Lee, Y.; Lee, J. Mechanical and Corrosion Properties of Rheocast and Low-Pressure Cast A356-T6 Alloy. Mater. Sci. Eng. A 2005, 391, 86–94. [Google Scholar] [CrossRef]
- Corbit, S.A.; DasGupta, R. Squeeze Cast Automotive Applications and Squeeze Cast Aluminum Alloy Properties. In Proceedings of the International Congress & Exposition, Detroit, MI, USA, 23–26 February 1998. [Google Scholar]
- Ghomashchi, M.; Vikhrov, A. Squeeze Casting: An Overview. J. Mater. Process. Technol. 2000, 101, 1–9. [Google Scholar] [CrossRef]
- Chen, G.; Yang, M.; Jin, Y.; Zhang, H.; Han, F.; Chen, Q.; Zhao, Z. Ultrasonic Assisted Squeeze Casting of a Wrought Aluminum Alloy. J. Mater. Process. Technol. 2019, 266, 19–25. [Google Scholar] [CrossRef]
- Sivasankaran, S.; Ramkumar, K.R.; Ammar, H.R.; Al-Mufadi, F.A.; Alaboodi, A.S.; Irfan, O.M. Microstructural Evolutions and Enhanced Mechanical Performance of Novel Al-Zn Die-Casting Alloys Processed by Squeezing and Hot Extrusion. J. Mater. Process. Technol. 2021, 292, 117063. [Google Scholar] [CrossRef]
- Li, R.; Liu, L.; Zhang, L.; Sun, J.; Shi, Y.; Yu, B. Effect of Squeeze Casting on Microstructure and Mechanical Properties of Hypereutectic Al-xSi Alloys. J. Mater. Sci. Technol. 2017, 33, 404–410. [Google Scholar] [CrossRef]
- Shabani, M.O.; Baghani, A.; Khorram, A.; Heydari, F. Evaluation of Fracture Mechanisms in Al-Si Metal Matrix Nanocomposites Produced by Three Methods of Gravity Sand Casting, Squeeze Casting and Compo Casting in Semi-Solid State. Silicon 2020, 12, 2977–2987. [Google Scholar] [CrossRef]
- Patel, H.A.; Chen, D.L.; Bhole, S.D.; Sadayappan, K. Microstructure and Tensile Properties of Thixomolded Magnesium Alloys. J. Alloys Compd. 2010, 496, 140–148. [Google Scholar] [CrossRef]
- Patel, H.A.; Chen, D.L.; Bhole, S.D.; Sadayappan, K. Low Cycle Fatigue Behavior of a Semi-Solid Processed AM60B Magnesium Alloy. Mater. Des. 2013, 49, 456–464. [Google Scholar] [CrossRef]
- Patel, H.A.; Chen, D.L.; Bhole, S.D.; Sadayappan, K. Cyclic Deformation and Twinning in a Semi-Solid Processed AZ91D Magnesium Alloy. Mater. Sci. Eng. A 2010, 528, 208–219. [Google Scholar] [CrossRef]
- Pan, Q.; Apelian, D. Semisolid Metal Processing. In ASM Handbook Volume 15: Casting; ASM International: Novelty, OH, USA, 2008. [Google Scholar]
- Pola, A.; Tocci, M.; Kapranos, P. Microstructure and Properties of Semi-Solid Aluminum Alloys: A Literature Review. Metals 2018, 8, 181. [Google Scholar] [CrossRef] [Green Version]
- Qi, M.; Kang, Y.; Li, J.; Wulabieke, Z.; Xu, Y.; Li, Y.; Liu, A.; Chen, J. Microstructures Refinement and Mechanical Properties Enhancement of Aluminum and Magnesium Alloys by Combining Distributary-Confluence Channel Process for Semisolid Slurry Preparation with High Pressure Die-Casting. J. Mater. Process. Technol. 2020, 285, 116800. [Google Scholar] [CrossRef]
- Dey, A.K.; Poddar, P.; Singh, K.K.; Sahoo, K.L. Mechanical and Wear Properties of Rheocast and Conventional Gravity Die Cast A356 Alloy. Mater. Sci. Eng. A 2006, 435–436, 521–529. [Google Scholar] [CrossRef]
- Ma, C.I.; Lee, H.D.; Kim, D.U. Mechanical and Microstructural Properties Investigation on Rheocast Automotive Parts Using A356 Alloy. Solid State Phenom. 2006, 116–117, 489–492. [Google Scholar] [CrossRef]
- Guo, H.-M.; Yang, X.-J.; Wang, J.-X. Pressurized Solidification of Semi-Solid Aluminum Die Casting Alloy A356. J. Alloys Compd. 2009, 485, 812–816. [Google Scholar] [CrossRef]
- Lü, S.; Wu, S.; Dai, W.; Lin, C.; An, P. The Indirect Ultrasonic Vibration Process for Rheo-Squeeze Casting of A356 Aluminum Alloy. J. Mater. Process. Technol. 2012, 212, 1281–1287. [Google Scholar] [CrossRef]
- Chen, W.; Thornley, L.; Coe, H.G.; Tonneslan, S.J.; Vericella, J.J.; Zhu, C.; Duoss, E.B.; Hunt, R.M.; Wight, M.J.; Apelian, D.; et al. Direct Metal Writing: Controlling the Rheology through Microstructure. Appl. Phys. Lett. 2017, 110, 094104. [Google Scholar] [CrossRef]
- Li, P.-H.; Guo, W.-G.; Yuan, K.-B.; Su, Y.; Wang, J.-J.; Lin, X.; Li, Y.-P. Effects of Processing Defects on the Dynamic Tensile Mechanical Behavior of Laser-Solid-Formed Ti-6Al-4 V. Mater. Charact. 2018, 140, 15–29. [Google Scholar] [CrossRef]
- Brandl, E.; Heckenberger, U.; Holzinger, V.; Buchbinder, D. Additive Manufactured AlSi10Mg Samples Using Selective Laser Melting (SLM): Microstructure, High Cycle Fatigue, and Fracture Behavior. Mater. Des. 2012, 34, 159–169. [Google Scholar] [CrossRef]
- Read, N.; Wang, W.; Essa, K.; Attallah, M.M. Selective Laser Melting of AlSi10Mg Alloy: Process Optimisation and Mechanical Properties Development. Mater. Des. 2015, 65, 417–424. [Google Scholar] [CrossRef] [Green Version]
- Attar, H.; Calin, M.; Zhang, L.C.; Scudino, S.; Eckert, J. Manufacture by Selective Laser Melting and Mechanical Behavior of Commercially Pure Titanium. Mater. Sci. Eng. A 2014, 593, 170–177. [Google Scholar] [CrossRef]
- Ferrar, B.; Mullen, L.; Jones, E.; Stamp, R.; Sutcliffe, C.J. Gas Flow Effects on Selective Laser Melting (SLM) Manufacturing Performance. J. Mater. Process. Technol. 2012, 212, 355–364. [Google Scholar] [CrossRef]
- Murr, L.E.; Gaytan, S.M.; Ramirez, D.A.; Martinez, E.; Hernandez, J.; Amato, K.N.; Shindo, P.W.; Medina, F.R.; Wicker, R.B. Metal Fabrication by Additive Manufacturing Using Laser and Electron Beam Melting Technologies. J. Mater. Sci. Technol. 2012, 28, 1–14. [Google Scholar] [CrossRef]
- Hitzler, L.; Sert, E.; Merkel, M.; Öchsner, A.; Werner, E. Fracture Toughness and Fatigue Strength of Selective Laser Melted Aluminium–Silicon: An Overview. In TMS 2019 148th Annual Meeting & Exhibition Supplemental Proceedings; Springer: Cham, Switzerland, 2019; pp. 407–412. [Google Scholar]
- Awd, M.; Siddique, S.; Walther, F. Microstructural Damage and Fracture Mechanisms of Selective Laser Melted Al-Si Alloys under Fatigue Loading. Theor. Appl. Fract. Mech. 2020, 106, 102483. [Google Scholar] [CrossRef]
- Frazier, W.E. Metal Additive Manufacturing: A Review. J. Mater. Eng. Perform. 2014, 23, 1917–1928. [Google Scholar] [CrossRef]
- He, C.; Wei, J.; Li, Y.; Zhang, Z.; Tian, N.; Qin, G.; Zuo, L. Improvement of Microstructure and Fatigue Performance of Wire-Arc Additive Manufactured 4043 Aluminum Alloy Assisted by Interlayer Friction Stir Processing. J. Mater. Sci. Technol. 2023, 133, 183–194. [Google Scholar] [CrossRef]
- Aboulkhair, N.T.; Simonelli, M.; Parry, L.; Ashcroft, I.; Tuck, C.; Hague, R. 3D Printing of Aluminium Alloys: Additive Manufacturing of Aluminium Alloys Using Selective Laser Melting. Prog. Mater. Sci. 2019, 106, 100578. [Google Scholar] [CrossRef]
- Trevisan, F.; Calignano, F.; Lorusso, M.; Pakkanen, J.; Aversa, A.; Ambrosio, E.; Lombardi, M.; Fino, P.; Manfredi, D. On the Selective Laser Melting (SLM) of the AlSi10Mg Alloy: Process, Microstructure, and Mechanical Properties. Materials 2017, 10, 76. [Google Scholar] [CrossRef] [Green Version]
- Michi, R.A.; Plotkowski, A.; Shyam, A.; Dehoff, R.R.; Babu, S.S. Towards High-Temperature Applications of Aluminium Alloys Enabled by Additive Manufacturing. Int. Mater. Rev. 2022, 67, 298–345. [Google Scholar] [CrossRef]
- Johnson, Q.C.; Laursen, C.M.; Spear, A.D.; Carroll, J.D.; Noell, P.J. Analysis of the Interdependent Relationship between Porosity, Deformation, and Crack Growth during Compression Loading of LPBF AlSi10Mg. Mater. Sci. Eng. A 2022, 852, 143640. [Google Scholar] [CrossRef]
- Körber, S.; Völkl, R.; Glatzel, U. 3D Printed Polymer Positive Models for the Investment Casting of Extremely Thin-Walled Single Crystals. J. Mater. Process. Technol. 2021, 293, 117095. [Google Scholar] [CrossRef]
- Taylor, J.A. Iron-Containing Intermetallic Phases in Al-Si Based Casting Alloys. Procedia Mater. Sci. 2012, 1, 19–33. [Google Scholar] [CrossRef] [Green Version]
- Ceschini, L.; Boromei, I.; Morri, A.; Seifeddine, S.; Svensson, I.L. Microstructure, Tensile and Fatigue Properties of the Al–10%Si–2%Cu Alloy with Different Fe and Mn Content Cast under Controlled Conditions. J. Mater. Process. Technol. 2009, 209, 5669–5679. [Google Scholar] [CrossRef]
- Cinkilic, E.; Ridgeway, C.D.; Yan, X.; Luo, A.A. A Formation Map of Iron-Containing Intermetallic Phases in Recycled Cast Aluminum Alloys. Metall. Mater. Trans. A 2019, 50, 5945–5956. [Google Scholar] [CrossRef]
- Becker, H.; Bergh, T.; Vullum, P.E.; Leineweber, A.; Li, Y. Effect of Mn and Cooling Rates on α-, β- and δ-Al–Fe–Si Intermetallic Phase Formation in a Secondary Al–Si Alloy. Materialia 2019, 5, 100198. [Google Scholar] [CrossRef]
- Basak, C.B.; Meduri, A.; Hari Babu, N. Influence of Ni in High Fe Containing Recyclable Al-Si Cast Alloys. Mater. Des. 2019, 182, 108017. [Google Scholar] [CrossRef]
- Bösch, D.; Pogatscher, S.; Hummel, M.; Fragner, W.; Uggowitzer, P.J.; Göken, M.; Höppel, H.W. Secondary Al-Si-Mg High-Pressure Die Casting Alloys with Enhanced Ductility. Metall. Mater. Trans. A 2015, 46, 1035–1045. [Google Scholar] [CrossRef]
- Tillov, E.; Chalupov, M.; Hurtalov, L. Evolution of Phases in a Recycled Al-Si Cast Alloy During Solution Treatment. In Scanning Electron Microscopy; IntechOpen: London, UK, 2012. [Google Scholar]
- Velasco, E.; Nino, J. Recycling of Aluminium Scrap for Secondary Al-Si Alloys. Waste Manag. Res. J. Sustain. Circ. Econ. 2011, 29, 686–693. [Google Scholar] [CrossRef]
- Benedyk, J.C. International Temper Designation Systems for Wrought Aluminum Alloys. Light Met. Age Int. 2010, 26–30. [Google Scholar]
- Warmuzek, M. Aluminum-Silicon Casting Alloys Atlas of Microfractographs; ASM International: Novelty, OH, USA, 2004; ISBN 0871707942. [Google Scholar]
- Robles Hernandez, F.C.; Herrera Ramírez, J.M.; Mackay, R. Al-Si Alloys, Minor, Major, and Impurity Elements. In Al-Si Alloys; Springer International Publishing: Cham, Switzerland, 2017; pp. 1–15. [Google Scholar]
- Campbell, J. Castings; Elsevier: Amsterdam, The Netherlands, 2003. [Google Scholar]
- Elmadagli, M.; Perry, T.; Alpas, A.T. A Parametric Study of the Relationship between Microstructure and Wear Resistance of Al–Si Alloys. Wear 2007, 262, 79–92. [Google Scholar] [CrossRef]
- Hernandez, F.C.R.; Djurdjevic, M.B.; Kierkus, W.T.; Sokolowski, J.H. Calculation of the Liquidus Temperature for Hypo and Hypereutectic Aluminum Silicon Alloys. Mater. Sci. Eng. A 2005, 396, 271–276. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, Z.; Ma, Y.; Jin, Y. Effect of Fe and Mn Additions on Microstructure and Wear Properties of Spray-Deposited Al–20Si Alloy. Mater. Lett. 2004, 58, 2442–2446. [Google Scholar] [CrossRef]
- Dash, S.S.; Li, D.J.; Zeng, X.Q.; Li, D.Y.; Chen, D.L. Deformation Behavior of a Newly-Developed T4-Treated Al–Si Die Cast Alloy. Mater. Sci. Eng. A 2022, 866, 144283. [Google Scholar] [CrossRef]
- Dash, S.S.; Li, D.J.; Zeng, X.Q.; Li, D.Y.; Chen, D.L. Monotonic and Cyclic Deformation Behavior of a Silafont®-36 Cast Aluminum Alloy in an Overaged Condition. In Proceedings of the 61st Conference of Metallurgists, COM 2022; Springer International Publishing: Cham, Switzerland, 2023; pp. 15–19. [Google Scholar]
- Liu, F.; Yu, F.; Zhao, D.; Gao, L. Fatigue Behavior of an Al-12.7Si-0.7Mg Alloy Processed by Extrusion and Heat Treatment. Front. Mater. 2021, 8, 667771. [Google Scholar] [CrossRef]
- Bogdanoff, T.; Lattanzi, L.; Merlin, M.; Ghassemali, E.; Seifeddine, S. The Influence of Copper Addition on Crack Initiation and Propagation in an Al–Si–Mg Alloy during Cyclic Testing. Materialia 2020, 12, 100787. [Google Scholar] [CrossRef]
- Zhou, Z.; Liu, X.; Le, P.; Liao, Y.; He, G.; Wang, Q.; He, Q. Effect of Heat Treatment and Loading Path on Non-Proportional Fatigue Behavior and Fracture Characteristics of an Al-Si Casting Alloy. J. Mater. Eng. Perform. 2022, 1–13. [Google Scholar] [CrossRef]
- Robles Hernandez, F.C. Improvement in Functional Characteristics of Aluminum-Silicon Cast Components through the Utilization of a Novel Electromagnetic Treatment of Liquid Melts. Ph.D. Thesis, University of Windsor, Windsor, ON, Canada, 2004. [Google Scholar]
- Andilab, B.; Emadi, P.; Ravindran, C. Casting and Characterization of A319 Aluminum Alloy Reinforced with Graphene Using Hybrid Semi-Solid Stirring and Ultrasonic Processing. Materials 2022, 15, 7232. [Google Scholar] [CrossRef] [PubMed]
- McDonald, S.D.; Nogita, K.; Dahle, A.K. Eutectic Nucleation in Al–Si Alloys. Acta Mater. 2004, 52, 4273–4280. [Google Scholar] [CrossRef]
- Riahi, A.R.; Alpas, A.T. Fracture of Silicon-Rich Particles during Sliding Contact of Al–Si Alloys. Mater. Sci. Eng. A 2006, 441, 326–330. [Google Scholar] [CrossRef]
- Robles Hernandez, F.C.; Herrera Ramírez, J.M.; Mackay, R. Principles of Solidification. In Al-Si Alloys; Springer International Publishing: Cham, Switzerland, 2017; pp. 173–210. [Google Scholar]
- Haque, M.; Maleque, M. Effect of Process Variables on Structure and Properties of Aluminium–Silicon Piston Alloy. J. Mater. Process. Technol. 1998, 77, 122–128. [Google Scholar] [CrossRef]
- Kapranos, P.; Kirkwood, D.H.; Atkinson, H.V.; Rheinlander, J.T.; Bentzen, J.J.; Toft, P.T.; Debel, C.P.; Laslaz, G.; Maenner, L.; Blais, S.; et al. Thixoforming of an Automotive Part in A390 Hypereutectic Al–Si Alloy. J. Mater. Process. Technol. 2003, 135, 271–277. [Google Scholar] [CrossRef]
- Dinnis, C.M.; Dahle, A.K.; Taylor, J.A.; Otte, M.O. The Influence of Strontium on Porosity Formation in Al-Si Alloys. Metall. Mater. Trans. A 2004, 35, 3531–3541. [Google Scholar] [CrossRef]
- Rao, J.; Zhang, J.; Liu, R.; Zheng, J.; Yin, D. Modification of Eutectic Si and the Microstructure in an Al-7Si Alloy with Barium Addition. Mater. Sci. Eng. A 2018, 728, 72–79. [Google Scholar] [CrossRef]
- Cai, Q.; Mendis, C.L.; Chang, I.T.H.; Fan, Z. Effect of Short T6 Heat Treatment on the Microstructure and the Mechanical Properties of Newly Developed Die-Cast Al–Si–Mg–Mn Alloys. Mater. Sci. Eng. A 2020, 788, 139610. [Google Scholar] [CrossRef]
- Gan, J.; Du, J.; Wen, C.; Zhang, G.; Shi, M.; Yuan, Z. The Effect of Fe Content on the Solidification Pathway, Microstructure and Thermal Conductivity of Hypoeutectic Al–Si Alloys. Int. J. Met. 2022, 16, 178–190. [Google Scholar] [CrossRef]
- Shen, X.; Liu, S.; Wang, X.; Cui, C.; Gong, P.; Zhao, L.; Han, X.; Li, Z. Effect of Cooling Rate on the Microstructure Evolution and Mechanical Properties of Iron-Rich Al–Si Alloy. Materials 2022, 15, 411. [Google Scholar] [CrossRef]
- Mohammed, S.M.A.K.; Li, D.J.; Zeng, X.Q.; Chen, D.L. Low-cycle Fatigue Behavior of a Newly Developed Cast Aluminum Alloy for Automotive Applications. Fatigue Fract. Eng. Mater. Struct. 2019, 42, 1912–1926. [Google Scholar] [CrossRef]
- Shankar, S.; Riddle, Y.W.; Makhlouf, M.M. Nucleation Mechanism of the Eutectic Phases in Aluminum–Silicon Hypoeutectic Alloys. Acta Mater. 2004, 52, 4447–4460. [Google Scholar] [CrossRef]
- Zhao, B.; Xing, S.; Sun, H.; Yan, G.; Gao, W.; Ou, L. Effect of Rare-Earth La on Microstructure and Mechanical Properties of Al7Si4CuMg Alloys Prepared by Squeeze Casting. J. Mater. Sci. 2022, 57, 12064–12083. [Google Scholar] [CrossRef]
- Wang, R.; Lu, W.; Hogan, L.M. Twin Related Silicon Crystals in Al–Si Alloys and Their Growth Mechanism. Mater. Sci. Technol. 1995, 11, 441–449. [Google Scholar] [CrossRef]
- Li, J.H.; Albu, M.; Ludwig, T.; Matsubara, Y.; Hofer, F.; Arnberg, L.; Tsunekawa, Y.; Schumacher, P. Modification of Eutectic Si in Al-Si Based Alloys. Mater. Sci. Forum 2014, 794–796, 130–136. [Google Scholar] [CrossRef]
- Nogita, K.; McDonald, S.D.; Dahle, A.K. Eutectic Modification of Al-Si Alloys with Rare Earth Metals. Mater. Trans. 2004, 45, 323–326. [Google Scholar] [CrossRef] [Green Version]
- Mackay, R.; Sokolowski, J. Comparison Between Wedge Test Castings and Component Engine Block Casting Properties. Int. J. Met. 2010, 4, 33–50. [Google Scholar] [CrossRef]
- Gall, K.; Yang, N.; Horstemeyer, M.; McDowell, D.L.; Fan, J. The Debonding and Fracture of Si Particles during the Fatigue of a Cast Al-Si Alloy. Metall. Mater. Trans. A 1999, 30, 3079–3088. [Google Scholar] [CrossRef]
- Wang, M.; Pang, J.C.; Liu, H.Q.; Li, S.X.; Zhang, Z.F. Property Optimization of Low-Cycle Fatigue in Al-Si Piston Alloy at Elevated Temperatures by Ultrasonic Melt Treatment. J. Mater. Res. Technol. 2019, 8, 4556–4568. [Google Scholar] [CrossRef]
- Aversa, A.; Marchese, G.; Saboori, A.; Bassini, E.; Manfredi, D.; Biamino, S.; Ugues, D.; Fino, P.; Lombardi, M. New Aluminum Alloys Specifically Designed for Laser Powder Bed Fusion: A Review. Materials 2019, 12, 1007. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, J.; Wang, X.Q.; Wang, W.; Attallah, M.M.; Loretto, M.H. Microstructure and Strength of Selectively Laser Melted AlSi10Mg. Acta Mater. 2016, 117, 311–320. [Google Scholar] [CrossRef] [Green Version]
- Kim, D.-K.; Woo, W.; Hwang, J.-H.; An, K.; Choi, S.-H. Stress Partitioning Behavior of an AlSi10Mg Alloy Produced by Selective Laser Melting during Tensile Deformation Using in Situ Neutron Diffraction. J. Alloys Compd. 2016, 686, 281–286. [Google Scholar] [CrossRef]
- Hadadzadeh, A.; Baxter, C.; Amirkhiz, B.S.; Mohammadi, M. Strengthening Mechanisms in Direct Metal Laser Sintered AlSi10Mg: Comparison between Virgin and Recycled Powders. Addit. Manuf. 2018, 23, 108–120. [Google Scholar] [CrossRef]
- Hadadzadeh, A.; Amirkhiz, B.S.; Mohammadi, M. Contribution of Mg2Si Precipitates to the Strength of Direct Metal Laser Sintered AlSi10Mg. Mater. Sci. Eng. A 2019, 739, 295–300. [Google Scholar] [CrossRef]
- Ervina Efzan, M.N.; Kong, H.J.; Kok, C.K. Review: Effect of Alloying Element on Al-Si Alloys. Adv. Mater. Res. 2013, 845, 355–359. [Google Scholar] [CrossRef]
- Brodova, I.G.; Popel, P.S.; Eskin, G.I. Liquid Metal Processing; CRC Press: Boca Raton, FL, USA, 2001; ISBN 9781482264913. [Google Scholar]
- Kendig, K.L.; Miracle, D.B. Strengthening Mechanisms of an Al-Mg-Sc-Zr Alloy. Acta Mater. 2002, 50, 4165–4175. [Google Scholar] [CrossRef]
- de Souza Baptista, L.A.; Paradela, K.G.; Ferreira, I.L.; Garcia, A.; Ferreira, A.F. Experimental Study of the Evolution of Tertiary Dendritic Arms and Microsegregation in Directionally Solidified Al–Si–Cu Alloys Castings. J. Mater. Res. Technol. 2019, 8, 1515–1521. [Google Scholar] [CrossRef]
- Barrirero, J. Eutectic Modification of Al-Si Casting Alloys; Linköping Studies in Science and Technology. Dissertations; Linköping University Electronic Press: Linköping, Sweden, 2019; Volume 2014, ISBN 9789175190075. [Google Scholar]
- Lu, Y.; Godlewski, L.A.; Zindel, J.W.; Lee, A. Use of Reactive Nanostructured Chemicals for Refinement of Si Eutectic in an Aluminum Casting Alloy. J. Mater. Sci. 2019, 54, 12818–12832. [Google Scholar] [CrossRef]
- Sigworth, G.; Campbell, J.; Jorstad, J. The Modification of Al-Si Casting Alloys: Important Practical and Theoretical Aspects. Int. J. Met. 2009, 3, 65–78. [Google Scholar] [CrossRef]
- McDonald, S.D.; Dahle, A.K.; Taylor, J.A.; StJohn, D.H. Eutectic Grains in Unmodified and Strontium-Modified Hypoeutectic Aluminum-Silicon Alloys. Metall. Mater. Trans. A 2004, 35, 1829–1837. [Google Scholar] [CrossRef]
- Tiedje, N.S.; Hattel, J.; Taylor, J.A.; Easton, M.A. A Solidification Model for Unmodified, Na-Modified and Sr-Modified Al-Si Alloys. IOP Conf. Ser. Mater. Sci. Eng. 2012, 27, 012033. [Google Scholar] [CrossRef] [Green Version]
- Abboud, J.; Mazumder, J. Developing of Nano Sized Fibrous Eutectic Silicon in Hypereutectic Al–Si Alloy by Laser Remelting. Sci. Rep. 2020, 10, 12090. [Google Scholar] [CrossRef]
- Qu, S.J.; Feng, A.H.; Geng, L.; Shen, J.; Chen, D.L. Silicon Nitride Whisker-Reinforced Aluminum Matrix Composites: Twinning and Precipitation Behavior. Metals 2020, 10, 420. [Google Scholar] [CrossRef] [Green Version]
- Prukkanon, W.; Srisukhumbowornchai, N.; Limmaneevichitr, C. Modification of Hypoeutectic Al–Si Alloys with Scandium. J. Alloys Compd. 2009, 477, 454–460. [Google Scholar] [CrossRef]
- Zhu, G.L.; Gu, N.J.; Zhou, B.J. Effects of Combining Na and Sr Additions on Eutectic Modification in Al-Si Alloy. IOP Conf. Ser. Mater. Sci. Eng. 2017, 230, 012015. [Google Scholar] [CrossRef] [Green Version]
- Eguskiza, S.; Niklas, A.; Fernández-Calvo, A.I.; Santos, F.; Djurdjevic, M. Study of Strontium Fading in Al-Si-Mg AND Al-Si-Mg-Cu Alloy by Thermal Analysis. Int. J. Met. 2015, 9, 43–50. [Google Scholar] [CrossRef]
- Zhang, W.; Ma, S.; Wei, Z.; Bai, P. The Relationship between Residual Amount of Sr and Morphology of Eutectic Si Phase in A356 Alloy. Materials 2019, 12, 3222. [Google Scholar] [CrossRef] [Green Version]
- Hernandez-Sandoval, J.; Garza-Elizondo, G.H.; Samuel, A.M.; Valtiierra, S.; Samuel, F.H. The Ambient and High Temperature Deformation Behavior of Al–Si–Cu–Mg Alloy with Minor Ti, Zr, Ni Additions. Mater. Des. 2014, 58, 89–101. [Google Scholar] [CrossRef]
- Shaha, S.K.; Czerwinski, F.; Kasprzak, W.; Friedman, J.; Chen, D.L. Thermal Stability of (AlSi) (ZrVTi) Intermetallic Phases in the Al–Si–Cu–Mg Cast Alloy with Additions of Ti, V, and Zr. Thermochim. Acta 2014, 595, 11–16. [Google Scholar] [CrossRef]
- Zamani, M.; Morini, L.; Ceschini, L.; Seifeddine, S. The Role of Transition Metal Additions on the Ambient and Elevated Temperature Properties of Al-Si Alloys. Mater. Sci. Eng. A 2017, 693, 42–50. [Google Scholar] [CrossRef]
- Shaha, S.K.; Czerwinski, F.; Kasprzak, W.; Friedman, J.; Chen, D.L. Monotonic and Cyclic Deformation Behavior of the Al–Si–Cu–Mg Cast Alloy with Micro-Additions of Ti, V and Zr. Int. J. Fatigue 2015, 70, 383–394. [Google Scholar] [CrossRef]
- Abdelaziz, M.H.; Elgallad, E.M.; Doty, H.W.; Valtierra, S.; Samuel, F.H. Melting and Solidification Characteristics of Zr-, Ni-, and Mn-Containing 354-Type Al-Si-Cu-Mg Cast Alloys. Philos. Mag. 2019, 99, 1633–1655. [Google Scholar] [CrossRef]
- Rahimian, M.; Amirkhanlou, S.; Blake, P.; Ji, S. Nanoscale Zr-Containing Precipitates; a Solution for Significant Improvement of High-Temperature Strength in Al-Si-Cu-Mg Alloys. Mater. Sci. Eng. A 2018, 721, 328–338. [Google Scholar] [CrossRef] [Green Version]
- Asghar, G.; Peng, L.; Fu, P.; Yuan, L.; Liu, Y. Role of Mg2Si Precipitates Size in Determining the Ductility of A357 Cast Alloy. Mater. Des. 2020, 186, 108280. [Google Scholar] [CrossRef]
- Ebhota, W.S.; Jen, T.-C. Intermetallics Formation and Their Effect on Mechanical Properties of Al-Si-X Alloys. In Intermetallic Compounds—Formation and Applications; IntechOpen: London, UK, 2018. [Google Scholar]
- Roy, S.; Allard, L.F.; Rodriguez, A.; Watkins, T.R.; Shyam, A. Comparative Evaluation of Cast Aluminum Alloys for Automotive Cylinder Heads: Part I-Microstructure Evolution. Metall. Mater. Trans. A 2017, 48, 2529–2542. [Google Scholar] [CrossRef]
- Roy, S.; Allard, L.F.; Rodriguez, A.; Porter, W.D.; Shyam, A. Comparative Evaluation of Cast Aluminum Alloys for Automotive Cylinder Heads: Part II-Mechanical and Thermal Properties. Metall. Mater. Trans. A 2017, 48, 2543–2562. [Google Scholar] [CrossRef]
- Chen, R.; Xu, Q.; Guo, H.; Xia, Z.; Wu, Q.; Liu, B. Modeling the Precipitation Kinetics and Tensile Properties in Al-7Si-Mg Cast Aluminum Alloys. Mater. Sci. Eng. A 2017, 685, 403–416. [Google Scholar] [CrossRef]
- Zhang, J.; Cinkilic, E.; Huang, X.; Wang, G.G.; Liu, Y.; Weiler, J.P.; Luo, A.A. Optimization of T5 Heat Treatment in High Pressure Die Casting of Al–Si–Mg–Mn Alloys by Using an Improved Kampmann-Wagner Numerical (KWN) Model. Mater. Sci. Eng. A 2023, 865, 144604. [Google Scholar] [CrossRef]
- Lee, P.; Chirazi, A.; See, D. Modeling Microporosity in Aluminum–Silicon Alloys: A Review. J. Light Met. 2001, 1, 15–30. [Google Scholar] [CrossRef]
- Haselhuhn, A.S.; Sanders, P.G.; Pearce, J.M. Hypoeutectic Aluminum–Silicon Alloy Development for GMAW-Based 3-D Printing Using Wedge Castings. Int. J. Met. 2017, 11, 843–856. [Google Scholar] [CrossRef] [Green Version]
- Amirkhanlou, S.; Ji, S. Casting Lightweight Stiff Aluminum Alloys: A Review. Crit. Rev. Solid State Mater. Sci. 2020, 45, 171–186. [Google Scholar] [CrossRef]
- Lasagni, F.; Degischer, H.P. Enhanced Young’s Modulus of Al-Si Alloys and Reinforced Matrices by Co-Continuous Structures. J. Compos. Mater. 2010, 44, 739–755. [Google Scholar] [CrossRef]
- Lasagni, F.; Lasagni, A.; Marks, E.; Holzapfel, C.; Mücklich, F.; Degischer, H.P. Three-Dimensional Characterization of ‘as-Cast’ and Solution-Treated AlSi12(Sr) Alloys by High-Resolution FIB Tomography. Acta Mater. 2007, 55, 3875–3882. [Google Scholar] [CrossRef]
- Jeong, C.-Y. Effect of Alloying Elements on High Temperature Mechanical Properties for Piston Alloy. Mater. Trans. 2012, 53, 234–239. [Google Scholar] [CrossRef] [Green Version]
- Lados, D.A.; Apelian, D.; Wang, L. Aging Effects on Heat Treatment Response and Mechanical Properties of Al-(1 to 13 Pct)Si-Mg Cast Alloys. Metall. Mater. Trans. B 2011, 42, 181–188. [Google Scholar] [CrossRef] [Green Version]
- Lumley, R. Design of Secondary Alloy Compositions for High Performance Aluminium Pressure Diecastings. Mater. Sci. Forum 2011, 693, 247–255. [Google Scholar] [CrossRef]
- Lumley, R.N.; Polmear, I.J.; Curtis, P.R. Rapid Heat Treatment of Aluminum High-Pressure Diecastings. Metall. Mater. Trans. A 2009, 40, 1716–1726. [Google Scholar] [CrossRef]
- Liu, M.; Zheng, R.; Xiao, W.; Yu, X.; Peng, Q.; Ma, C. Concurrent Enhancement of Strength and Ductility for Al-Si Binary Alloy by Refining Si Phase to Nanoscale. Mater. Sci. Eng. A 2019, 751, 303–310. [Google Scholar] [CrossRef]
- Bernsztejn, L.; Zajmowskij, W.A. Struktura I Własnoscimechanicznemetali (Structure and Mechanical Properties of Metals), Wyd. Nauk. Warsaw 1973. [Google Scholar]
- Suryawanshi, J.; Prashanth, K.G.; Scudino, S.; Eckert, J.; Prakash, O.; Ramamurty, U. Simultaneous Enhancements of Strength and Toughness in an Al-12Si Alloy Synthesized Using Selective Laser Melting. Acta Mater. 2016, 115, 285–294. [Google Scholar] [CrossRef]
- Prashanth, K.G.; Scudino, S.; Klauss, H.J.; Surreddi, K.B.; Löber, L.; Wang, Z.; Chaubey, A.K.; Kühn, U.; Eckert, J. Microstructure and Mechanical Properties of Al–12Si Produced by Selective Laser Melting: Effect of Heat Treatment. Mater. Sci. Eng. A 2014, 590, 153–160. [Google Scholar] [CrossRef]
- Banerjee, D.; Williams, J.C. Perspectives on Titanium Science and Technology. Acta Mater. 2013, 61, 844–879. [Google Scholar] [CrossRef]
- Yang, K.V.; Rometsch, P.; Jarvis, T.; Rao, J.; Cao, S.; Davies, C.; Wu, X. Porosity Formation Mechanisms and Fatigue Response in Al-Si-Mg Alloys Made by Selective Laser Melting. Mater. Sci. Eng. A 2018, 712, 166–174. [Google Scholar] [CrossRef]
- Nikanorov, S.P.; Osipov, V.N.; Regel, L.I. Structural and Mechanical Properties of Directionally Solidified Al-Si Alloys. J. Mater. Eng. Perform. 2019, 28, 7302–7323. [Google Scholar] [CrossRef]
- Gomes, L.F.; Kugelmeier, C.L.; Garcia, A.; Della Rovere, C.A.; Spinelli, J.E. Influences of Alloying Elements and Dendritic Spacing on the Corrosion Behavior of Al–Si–Ag Alloys. J. Mater. Res. Technol. 2021, 15, 5880–5893. [Google Scholar] [CrossRef]
- Ghassemali, E.; Riestra, M.; Bogdanoff, T.; Kumar, B.S.; Seifeddine, S. Hall-Petch Equation in a Hypoeutectic Al-Si Cast Alloy: Grain Size vs. Secondary Dendrite Arm Spacing. Procedia Eng. 2017, 207, 19–24. [Google Scholar] [CrossRef]
- Ceschini, L.; Morri, A.; Toschi, S.; Seifeddine, S. Room and High Temperature Fatigue Behaviour of the A354 and C355 (Al–Si–Cu–Mg) Alloys: Role of Microstructure and Heat Treatment. Mater. Sci. Eng. A 2016, 653, 129–138. [Google Scholar] [CrossRef]
- Hosch, T.; Napolitano, R.E. The Effect of the Flake to Fiber Transition in Silicon Morphology on the Tensile Properties of Al–Si Eutectic Alloys. Mater. Sci. Eng. A 2010, 528, 226–232. [Google Scholar] [CrossRef]
- Tiryakioğlu, M.; Shuey, R.T. Quench Sensitivity of an Al-7 Pct Si-0.6 Pct Mg Alloy: Characterization and Modeling. Metall. Mater. Trans. B 2007, 38, 575–582. [Google Scholar] [CrossRef]
- Sjölander, E.; Seifeddine, S. The Heat Treatment of Al–Si–Cu–Mg Casting Alloys. J. Mater. Process. Technol. 2010, 210, 1249–1259. [Google Scholar] [CrossRef] [Green Version]
- Pedersen, L.; Arnberg, L. The Effect of Solution Heat Treatment and Quenching Rates on Mechanical Properties and Microstructures in AlSiMg Foundry Alloys. Metall. Mater. Trans. A 2001, 32, 525–532. [Google Scholar] [CrossRef]
- Zhang, D.L.; Zheng, L. The Quench Sensitivity of Cast Al-7 Wt Pct Si-0.4 Wt Pct Mg Alloy. Metall. Mater. Trans. A 1996, 27, 3983–3991. [Google Scholar] [CrossRef]
- Shaha, S.K.; Czerwinski, F.; Kasprzak, W.; Friedman, J.; Chen, D.L. Effect of Mn and Heat Treatment on Improvements in Static Strength and Low-Cycle Fatigue Life of an Al–Si–Cu–Mg Alloy. Mater. Sci. Eng. A 2016, 657, 441–452. [Google Scholar] [CrossRef]
- Tiryakioğlu, M. The Effect of Solution Treatment and Artificial Aging on the Work Hardening Characteristics of a Cast Al–7%Si–0.6%Mg Alloy. Mater. Sci. Eng. A 2006, 427, 154–159. [Google Scholar] [CrossRef]
- Tiryakioğlu, M.; Alexopoulos, N.D. The Effect of Artificial Aging on Tensile Work Hardening Characteristics of a Cast Al-7 Pct Si-0.55 Pct Mg (A357) Alloy. Metall. Mater. Trans. A 2008, 39, 2772–2780. [Google Scholar] [CrossRef]
- Kocks, U.F.; Mecking, H. Physics and Phenomenology of Strain Hardening: The FCC Case. Prog. Mater. Sci. 2003, 48, 171–273. [Google Scholar] [CrossRef]
- Wang, C.X.; Yu, F.X.; Zhao, D.Z.; Zhao, X.; Zuo, L. Effect of Si Content on Deformation Behavior of DC Cast Al-Si Alloys. Adv. Mater. Res. 2013, 652–654, 1080–1083. [Google Scholar] [CrossRef]
- Dash, S.S.; Li, D.J.; Zeng, X.Q.; Li, D.Y.; Chen, D.L. On the Origin of Deformation Mechanisms in a Heterostructured Aluminum Alloy via Slip Trace and Lattice Rotation Analyses. Mater. Sci. Eng. A 2023, 867, 144723. [Google Scholar] [CrossRef]
- Wang, Q.G.; Cáceres, C.H. On the Strain Hardening Behaviour of Al-Si-Mg Casting Alloys. Mater. Sci. Eng. A 1997, 234–236, 106–109. [Google Scholar] [CrossRef]
- Chen, B.; Moon, S.K.; Yao, X.; Bi, G.; Shen, J.; Umeda, J.; Kondoh, K. Strength and Strain Hardening of a Selective Laser Melted AlSi10Mg Alloy. Scr. Mater. 2017, 141, 45–49. [Google Scholar] [CrossRef]
- Hernandez Sandoval, J. Improving the Performance of 354 Type Alloy. Ph.D. Thesis, Université du Québec à Chicoutimi, Chicoutimi, QC, Canada, 2010. [Google Scholar]
- Rakhmonov, J.; Timelli, G.; Bonollo, F. The Effect of Transition Elements on High-Temperature Mechanical Properties of Al-Si Foundry Alloys-A Review. Adv. Eng. Mater. 2016, 18, 1096–1105. [Google Scholar] [CrossRef]
- Mohamed, A.M.A.; Samuel, F.H.; Kahtani, S. Al Microstructure, Tensile Properties and Fracture Behavior of High Temperature Al–Si–Mg–Cu Cast Alloys. Mater. Sci. Eng. A 2013, 577, 64–72. [Google Scholar] [CrossRef]
- Colombo, M.; Gariboldi, E.; Morri, A. Er Addition to Al-Si-Mg-Based Casting Alloy: Effects on Microstructure, Room and High Temperature Mechanical Properties. J. Alloys Compd. 2017, 708, 1234–1244. [Google Scholar] [CrossRef]
- Jeon, J.H.; Shin, J.H.; Bae, D.H. Si Phase Modification on the Elevated Temperature Mechanical Properties of Al-Si Hypereutectic Alloys. Mater. Sci. Eng. A 2019, 748, 367–370. [Google Scholar] [CrossRef]
- Hyer, H.; Zhou, L.; Mehta, A.; Park, S.; Huynh, T.; Song, S.; Bai, Y.; Cho, K.; McWilliams, B.; Sohn, Y. Composition-Dependent Solidification Cracking of Aluminum-Silicon Alloys during Laser Powder Bed Fusion. Acta Mater. 2021, 208, 116698. [Google Scholar] [CrossRef]
- Qian, L.; Toda, H.; Akahori, T.; Niinomi, M.; Kobayashi, T.; Nishido, S. Numerical Simulation of Fracture of Model Al-Si Alloys. Metall. Mater. Trans. A 2005, 36, 2979–2992. [Google Scholar] [CrossRef]
- Vardanyan, V.H.; Zhang, Z.; Alhafez, I.A.; Urbassek, H.M. Cutting of Al/Si Bilayer Systems: Molecular Dynamics Study of Twinning, Phase Transformation, and Cracking. Int. J. Adv. Manuf. Technol. 2020, 107, 1297–1307. [Google Scholar] [CrossRef] [Green Version]
- Shaha, S.K.; Czerwinski, F.; Kasprzak, W.; Friedman, J.; Chen, D.L. Microstructure and Mechanical Properties of Al–Si Cast Alloy with Additions of Zr–V–Ti. Mater. Des. 2015, 83, 801–812. [Google Scholar] [CrossRef]
- Wang, Q.G. Microstructural Effects on the Tensile and Fracture Behavior of Aluminum Casting Alloys A356/357. Metall. Mater. Trans. A 2003, 34, 2887–2899. [Google Scholar] [CrossRef]
- Gall, K.; Horstemeyer, M.; Van Schilfgaarde, M.; Baskes, M. Atomistic Simulations on the Tensile Debonding of an Aluminum–Silicon Interface. J. Mech. Phys. Solids 2000, 48, 2183–2212. [Google Scholar] [CrossRef]
- Ma, Z.; Samuel, A.M.; Doty, H.W.; Valtierra, S.; Samuel, F.H. Effect of Fe Content on the Fracture Behaviour of Al–Si–Cu Cast Alloys. Mater. Des. 2014, 57, 366–373. [Google Scholar] [CrossRef]
- Lee, F.T.; Major, J.F.; Samuel, F.H. Effect of Silicon Particles on the Fatigue Crack Growth Characteristics of Al-12 Wt Pct Si-0.35 Wt Pct Mg-(0 to 0.02) Wt Pct Sr Casting Alloys. Metall. Mater. Trans. A 1995, 26, 1553–1570. [Google Scholar] [CrossRef]
- Shaha, S.K.; Czerwinski, F.; Kasprzak, W.; Friedman, J.; Chen, D.L. Effect of Zr, V and Ti on Hot Compression Behavior of the Al–Si Cast Alloy for Powertrain Applications. J. Alloys Compd. 2014, 615, 1019–1031. [Google Scholar] [CrossRef]
- Ren, P.; Huang, W.; Zuo, Z.; Li, D.; Zhao, C.; Yan, K. High Cycle Fatigue Analysis and Modelling of Cast Al–Si Alloys Extracted from Cylinder Heads Considering Microstructure Characteristics. J. Mater. Res. Technol. 2022, 19, 3004–3017. [Google Scholar] [CrossRef]
- Moon, M.S.; Yoo, M.H.; Kim, K.W.; Song, J.H.; Oh, J.H. Study on the Mechanical Properties of Commercial Vehicle Wheel through the Molten Forged on the A356 Alloy with a Multi-Cavity Fabrication Process. In Light Metals 2021; Springer: Cham, Switzerland, 2021; pp. 871–877. [Google Scholar]
- Tian, D.D.; Liu, X.S.; He, G.Q.; Shen, Y.; Lv, S.Q.; Wang, Q.G. Low Cycle Fatigue Behavior of Casting A319 Alloy under Two Different Aging Conditions. Mater. Sci. Eng. A 2016, 654, 60–68. [Google Scholar] [CrossRef]
- Dahdah, N.; Limodin, N.; El Bartali, A.; Witz, J.F.; Seghir, R.; Charkaluk, E.; Buffiere, J.Y. Damage Investigation in A319 Aluminium Alloy by X-ray Tomography and Digital Volume Correlation during In Situ High-Temperature Fatigue Tests. Strain 2016, 52, 324–335. [Google Scholar] [CrossRef] [Green Version]
- Ren, P.; Song, W.; Zhong, G.; Huang, W.; Zuo, Z.; Zhao, C.; Yan, K. High-Cycle Fatigue Failure Analysis of Cast Al-Si Alloy Engine Cylinder Head. Eng. Fail. Anal. 2021, 127, 105546. [Google Scholar] [CrossRef]
- Wu, Y.; Liao, H.; Tang, Y. Enhanced High-Cycle Fatigue Strength of Al–12Si–4Cu-1.2Mn-T6 Cast Aluminum Alloy at Room Temperature and 350 C. Mater. Sci. Eng. A 2021, 825, 141917. [Google Scholar] [CrossRef]
- Robles Hernandez, F.C.; Herrera Ramírez, J.M.; Mackay, R. Mechanical Properties. In Al-Si Alloys; Springer International Publishing: Cham, Switzerland, 2017; pp. 133–162. [Google Scholar]
- Xia, F.; Gao, X.S.; Liang, M.X.; Guo, Y.C.; Li, J.P.; Yang, Z.; Wang, J.L.; Zhang, L.L. Effect of Thermal Exposure on Microstructure and High-Temperature Fatigue Life of Al-Si Piston Alloys. J. Mater. Res. Technol. 2020, 9, 12926–12935. [Google Scholar] [CrossRef]
- Konečná, R.; Fintova, S.; Nicoletto, G.; Riva, E. High Temperature Fatigue Strength and Quantitative Metallography of an Eutectic Al-Si Alloy for Piston Application. Key Eng. Mater. 2013, 592–593, 627–630. [Google Scholar] [CrossRef]
- Wu, M.-Z.; Zhang, J.-W.; Zhang, Y.-B.; Wang, H.-Q. Effects of Mg Content on the Fatigue Strength and Fracture Behavior of Al-Si-Mg Casting Alloys. J. Mater. Eng. Perform. 2018, 27, 5992–6003. [Google Scholar] [CrossRef]
- Huter, P.; Renhart, P.; Oberfrank, S.; Schwab, M.; Grün, F.; Stauder, B. High- and Low-Cycle Fatigue Influence of Silicon, Copper, Strontium and Iron on Hypo-Eutectic Al–Si–Cu and Al–Si–Mg Cast Alloys Used in Cylinder Heads. Int. J. Fatigue 2016, 82, 588–601. [Google Scholar] [CrossRef]
- De Mori, A.; Timelli, G.; Berto, F.; Fabrizi, A. High Temperature Fatigue of Heat Treated Secondary AlSi7Cu3Mg Alloys. Int. J. Fatigue 2020, 138, 105685. [Google Scholar] [CrossRef]
- Konečná, R.; Nicoletto, G.; Kunz, L.; Riva, E. The Role of Elevated Temperature Exposure on Structural Evolution and Fatigue Strength of Eutectic AlSi12 Alloys. Int. J. Fatigue 2016, 83, 24–35. [Google Scholar] [CrossRef]
- Liu, H.; Pang, J.; Wang, M.; Li, J.; Guo, Y.; Li, S.; Zhang, Z. The Influence of Defect and Temperature on the Fatigue Behaviours of Al-Si-Cu-Mg-Ni Alloy. Fatigue Fract. Eng. Mater. Struct. 2019, 42, 2372–2382. [Google Scholar] [CrossRef]
- Wang, Q.; Apelian, D.; Lados, D. Fatigue Behavior of A356-T6 Aluminum Cast Alloys. Part I. Effect of Casting Defects. J. Light Met. 2001, 1, 73–84. [Google Scholar] [CrossRef]
- Ammar, H.R.; Samuel, A.M.; Doty, H.W.; Samuel, F.H. The Influence of Hot Isostatic Pressing on the Fatigue Life of Al–Si–Cu–Mg 354-T6 Casting Alloy. Int. J. Met. 2022, 16, 1315–1326. [Google Scholar] [CrossRef]
- Rezanezhad, S.; Azadi, M.; Azadi, M. Influence of Heat Treatment on High-Cycle Fatigue and Fracture Behaviors of Piston Aluminum Alloy Under Fully-Reversed Cyclic Bending. Met. Mater. Int. 2021, 27, 860–870. [Google Scholar] [CrossRef]
- Tenkamp, J.; Blinn, B.; Beck, T.; Walther, F. Microstructure- and Plasticity-Based Fatigue and Defect Tolerance Assessment of Age-Hardenable Al-Si Cast Alloys in LCF and HCF Regime. Int. J. Fatigue 2023, 166, 107240. [Google Scholar] [CrossRef]
- Ceschini, L.; Morri, A.; Toschi, S.; Bjurenstedt, A.; Seifeddine, S. Influence of Sludge Particles on the Fatigue Behavior of Al-Si-Cu Secondary Aluminium Casting Alloys. Metals 2018, 8, 268. [Google Scholar] [CrossRef] [Green Version]
- Samuel, A.; Zedan, Y.; Doty, H.; Songmene, V.; Samuel, F.H. A Review Study on the Main Sources of Porosity in Al-Si Cast Alloys. Adv. Mater. Sci. Eng. 2021, 2021, 1921603. [Google Scholar] [CrossRef]
- Ceschini, L.; Morri, A.; Morri, A.; Gamberini, A.; Messieri, S. Correlation between Ultimate Tensile Strength and Solidification Microstructure for the Sand Cast A357 Aluminium Alloy. Mater. Des. 2009, 30, 4525–4531. [Google Scholar] [CrossRef]
- Liu, G.; Blake, P.; Ji, S. Effect of Zr on the High Cycle Fatigue and Mechanical Properties of Al–Si–Cu–Mg Alloys at Elevated Temperatures. J. Alloys Compd. 2019, 809, 151795. [Google Scholar] [CrossRef]
- Baek, M.-S.; Kreethi, R.; Park, T.-H.; Sohn, Y.; Lee, K.-A. Influence of Heat Treatment on the High-Cycle Fatigue Properties and Fatigue Damage Mechanism of Selective Laser Melted AlSi10Mg Alloy. Mater. Sci. Eng. A 2021, 819, 141486. [Google Scholar] [CrossRef]
- Xin, Q. Durability and Reliability in Diesel Engine System Design. In Diesel Engine System Design; Elsevier: Amsterdam, The Netherlands, 2013; pp. 113–202. [Google Scholar]
- Emami, A.R.; Begum, S.; Chen, D.L.; Skszek, T.; Niu, X.P.; Zhang, Y.; Gabbianelli, F. Cyclic Deformation Behavior of a Cast Aluminum Alloy. Mater. Sci. Eng. A 2009, 516, 31–41. [Google Scholar] [CrossRef]
- Chen, S.; Liu, K.; Chen, X.-G. Elevated-Temperature Low-Cycle Fatigue Behaviors of Al-Si 356 and 319 Foundry Alloys. In Light Metals 2019; Chesonis, C., Ed.; Springer International Publishing: Cham, Switzerland, 2019; pp. 251–257. [Google Scholar]
- Koh, S.K.; Oh, S.J.; Li, C.; Ellyin, F. Low-Cycle Fatigue Life of SiC-Particulate-Reinforced Al-Si Cast Alloy Composites with Tensile Mean Strain Effects. Int. J. Fatigue 1999, 21, 1019–1032. [Google Scholar] [CrossRef]
- Chen, S.; Liu, K.; Chen, X.-G. Effect of Mo on Elevated-Temperature Low-Cycle Fatigue Behavior of Al-Si 356 Cast Alloy. In Light Metals 2020; Tomsett, A., Ed.; Springer International Publishing: Cham, Switzerland, 2020; pp. 261–266. [Google Scholar]
- Shaha, S.K.; Czerwinski, F.; Kasprzak, W.; Friedman, J.; Chen, D.L. Improving High-Temperature Tensile and Low-Cycle Fatigue Behavior of Al-Si-Cu-Mg Alloys Through Micro-Additions of Ti, V, and Zr. Metall. Mater. Trans. A 2015, 46, 3063–3078. [Google Scholar] [CrossRef]
- Fan, K.L.; Liu, X.S.; He, G.Q.; Chen, H. Elevated Temperature Low Cycle Fatigue of a Gravity Casting Al–Si–Cu Alloy Used for Engine Cylinder Heads. Mater. Sci. Eng. A 2015, 632, 127–136. [Google Scholar] [CrossRef]
- Bosefilho, W.; Defreitas, E.; Dasilva, V.; Milan, M.; Spinelli, D. Al–Si Cast Alloys under Isothermal and Thermomechanical Fatigue Conditions. Int. J. Fatigue 2007, 29, 1846–1854. [Google Scholar] [CrossRef]
- Wang, M.; Pang, J.C.; Li, S.X.; Zhang, Z.F. Low-Cycle Fatigue Properties and Life Prediction of Al-Si Piston Alloy at Elevated Temperature. Mater. Sci. Eng. A 2017, 704, 480–492. [Google Scholar] [CrossRef]
- Hazeli, K.; Askari, H.; Cuadra, J.; Streller, F.; Carpick, R.W.; Zbib, H.M.; Kontsos, A. Microstructure-Sensitive Investigation of Magnesium Alloy Fatigue. Int. J. Plast. 2015, 68, 55–76. [Google Scholar] [CrossRef]
- Begum, S.; Chen, D.L.; Xu, S.; Luo, A.A. Low Cycle Fatigue Properties of an Extruded AZ31 Magnesium Alloy. Int. J. Fatigue 2009, 31, 726–735. [Google Scholar] [CrossRef]
- Lin, X.Z.; Chen, D.L. Strain Controlled Cyclic Deformation Behavior of an Extruded Magnesium Alloy. Mater. Sci. Eng. A 2008, 496, 106–113. [Google Scholar] [CrossRef]
- Borrego, L.; Abreu, L.; Costa, J.; Ferreira, J. Analysis of Low Cycle Fatigue in AlMgSi Aluminium Alloys. Eng. Fail. Anal. 2004, 11, 715–725. [Google Scholar] [CrossRef] [Green Version]
- Tenkamp, J.; Stern, F.; Walther, F. Uniform Fatigue Damage Tolerance Assessment for Additively Manufactured and Cast Al-Si Alloys: An Elastic-Plastic Fracture Mechanical Approach. Addit. Manuf. Lett. 2022, 3, 100054. [Google Scholar] [CrossRef]
- Le, P.W.; Liu, X.S.; He, G.Q.; Ge, B.; Weng, Z.Z.; Wang, Q.G. Effect of HIPing and Degassing on the Low Cycle Fatigue Behavior of A319 Cast Alloy. Mater. Res. Express 2019, 6, 106552. [Google Scholar] [CrossRef]
- Huang, H.; Dong, Y.; Xing, Y.; Jia, Z.; Liu, Q. Low Cycle Fatigue Behaviour at 300 °C and Microstructure of Al-Si-Mg Casting Alloys with Zr and Hf Additions. J. Alloys Compd. 2018, 765, 1253–1262. [Google Scholar] [CrossRef]
- Kamal, M.; Rahman, M.M. Advances in Fatigue Life Modeling: A Review. Renew. Sustain. Energy Rev. 2018, 82, 940–949. [Google Scholar] [CrossRef]
- Fatemi, A.; Shamsaei, N. Multiaxial Fatigue: An Overview and Some Approximation Models for Life Estimation. Int. J. Fatigue 2011, 33, 948–958. [Google Scholar] [CrossRef]
- Li, H.; Jing, H.; Xu, L.; Han, Y.; Zhao, L.; Rong, J.; Tang, Z.; Xiao, B.; Zhang, Y.; Luo, Z.; et al. Cyclic Damage Behavior of Sanicro 25 Alloy at 700 °C: Dispersed Damage and Concentrated Damage. Int. J. Plast. 2019, 116, 91–117. [Google Scholar] [CrossRef]
- Zhang, Q.; Zuo, Z.; Liu, J. High-Temperature Low-Cycle Fatigue Behaviour of a Cast Al-12Si-CuNiMg Alloy. Fatigue Fract. Eng. Mater. Struct. 2013, 36, 623–630. [Google Scholar] [CrossRef]
- Kandil, F.A.; Brown, M.W.; Miller, K.J. Biaxial Low-Cycle Fatigue Failure of 316 Stainless Steel at Elevated Temperatures. In Mechanical Behaviour and Nuclear Applications of Stainless Steel at Elevated Temperatures; Maney Publishing: Leeds, UK, 1982. [Google Scholar]
- Wang, C.H.; Brown, M.W. A Path-Independent Parameter for Fatigue under Proportional and Non-Proportional Loading. Fatigue Fract. Eng. Mater. Struct. 1993, 16, 1285–1297. [Google Scholar] [CrossRef]
- Łagoda, T.; Vantadori, S.; Głowacka, K.; Kurek, M.; Kluger, K. Using the Smith-Watson-Topper Parameter and Its Modifications to Calculate the Fatigue Life of Metals: The State-of-the-Art. Materials 2022, 15, 3481. [Google Scholar] [CrossRef] [PubMed]
- Santecchia, E.; Hamouda, A.M.S.; Musharavati, F.; Zalnezhad, E.; Cabibbo, M.; El Mehtedi, M.; Spigarelli, S. A Review on Fatigue Life Prediction Methods for Metals. Adv. Mater. Sci. Eng. 2016, 2016, 9573524. [Google Scholar] [CrossRef] [Green Version]
- Kauzlarich, J.J. The Palmgren-Miner Rule Derived. In Tribology Series; Elsevier: Amsterdam, The Netherlands, 1989; pp. 175–179. [Google Scholar]
- Zhu, S.-P.; Huang, H.-Z.; He, L.-P.; Liu, Y.; Wang, Z. A Generalized Energy-Based Fatigue–Creep Damage Parameter for Life Prediction of Turbine Disk Alloys. Eng. Fract. Mech. 2012, 90, 89–100. [Google Scholar] [CrossRef]
- Zhu, S.-P.; Huang, H.-Z.; Wang, Z.-L. Fatigue Life Estimation Considering Damaging and Strengthening of Low Amplitude Loads under Different Load Sequences Using Fuzzy Sets Approach. Int. J. Damage Mech. 2011, 20, 876–899. [Google Scholar] [CrossRef]
- Azadi, M. Effects of Strain Rate and Mean Strain on Cyclic Behavior of Aluminum Alloys under Isothermal and Thermo-Mechanical Fatigue Loadings. Int. J. Fatigue 2013, 47, 148–153. [Google Scholar] [CrossRef]
- Wang, M.; Pang, J.C.; Liu, H.Q.; Li, S.X.; Zhang, M.X.; Zhang, Z.F. Effect of Constraint Factor on the Thermo-Mechanical Fatigue Behavior of an Al-Si Eutectic Alloy. Mater. Sci. Eng. A 2020, 783, 139279. [Google Scholar] [CrossRef]
- Wang, M.; Pang, J.; Liu, X.; Wang, J.; Liu, Y.; Li, S.; Zhang, Z. Optimization of Thermo-Mechanical Fatigue Life for Eutectic Al–Si Alloy by the Ultrasonic Melt Treatment. Materials 2022, 15, 7113. [Google Scholar] [CrossRef]
- Dowling, N.E. Mean Stress Effects in Stress-Life and Strain-Life Fatigue; SAE International: Warrendale, PA, USA, 2004. [Google Scholar]
- Arcari, A.; De Vita, R.; Dowling, N.E. Mean Stress Relaxation during Cyclic Straining of High Strength Aluminum Alloys. Int. J. Fatigue 2009, 31, 1742–1750. [Google Scholar] [CrossRef]
- Arcari, A.; Dowling, N.E. Modeling Mean Stress Relaxation in Variable Amplitude Loading for 7075-T6511 and 7249-T76511 High Strength Aluminum Alloys. Int. J. Fatigue 2012, 42, 238–247. [Google Scholar] [CrossRef]
- Chu, C.-C. Comparison of Mean Stress Correction Methods for Fatigue Life Prediction; SAE International: Warrendale, PA, USA, 2000. [Google Scholar]
- Ince, A. A Generalized Mean Stress Correction Model Based on Distortional Strain Energy. Int. J. Fatigue 2017, 104, 273–282. [Google Scholar] [CrossRef]
- Ince, A.; Glinka, G. A Modification of Morrow and Smith-Watson-Topper Mean Stress Correction Models. Fatigue Fract. Eng. Mater. Struct. 2011, 34, 854–867. [Google Scholar] [CrossRef]
- Dowling, N.E. Mean Stress Effects in Strain-Life Fatigue. Fatigue Fract. Eng. Mater. Struct. 2009, 32, 1004–1019. [Google Scholar] [CrossRef]
- Ince, A. A Mean Stress Correction Model for Tensile and Compressive Mean Stress Fatigue Loadings. Fatigue Fract. Eng. Mater. Struct. 2017, 40, 939–948. [Google Scholar] [CrossRef]
- Hao, H.; Ye, D.; Chen, Y.; Feng, M.; Liu, J. A Study on the Mean Stress Relaxation Behavior of 2124-T851 Aluminum Alloy during Low-Cycle Fatigue at Different Strain Ratios. Mater. Des. 2015, 67, 272–279. [Google Scholar] [CrossRef]
- Branco, R.; Costa, J.D.; Borrego, L.P.; Wu, S.C.; Long, X.Y.; Zhang, F.C. Effect of Strain Ratio on Cyclic Deformation Behaviour of 7050-T6 Aluminium Alloy. Int. J. Fatigue 2019, 129, 105234. [Google Scholar] [CrossRef]
- Fan, K.L.; Liu, X.S.; He, G.Q.; Cheng, H.; Lv, S.Q. Strain Ratio Effects on Low-Cycle Fatigue Behavior of Gravity Cast Al-Si-Cu Alloys. J. Mater. Eng. Perform. 2015, 24, 3942–3950. [Google Scholar] [CrossRef]
- Sangid, M.D. The Physics of Fatigue Crack Initiation. Int. J. Fatigue 2013, 57, 58–72. [Google Scholar] [CrossRef]
- Stinville, J.C.; Charpagne, M.A.; Cervellon, A.; Hemery, S.; Wang, F.; Callahan, P.G.; Valle, V.; Pollock, T.M. On the Origins of Fatigue Strength in Crystalline Metallic Materials. Science 2022, 377, 1065–1071. [Google Scholar] [CrossRef] [PubMed]
- Basinski, Z.S.; Basinski, S.J. Fundamental Aspects of Low Amplitude Cyclic Deformation in Face-Centred Cubic Crystals. Prog. Mater. Sci. 1992, 36, 89–148. [Google Scholar] [CrossRef]
- Déprés, C.; Robertson, C.F.; Fivel, M.C. Crack Initiation in Fatigue: Experiments and Three-Dimensional Dislocation Simulations. Mater. Sci. Eng. A 2004, 387–389, 288–291. [Google Scholar] [CrossRef]
- Mughrabi, H. Microstructural Mechanisms of Cyclic Deformation, Fatigue Crack Initiation and Early Crack Growth. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2015, 373, 20140132. [Google Scholar] [CrossRef] [Green Version]
- Blochwitz, C.; Tirschler, W. Twin Boundaries as Crack Nucleation Sites. Cryst. Res. Technol. 2005, 40, 32–41. [Google Scholar] [CrossRef]
- Kitagawa, H. Applicability of Fracture Mechanics to Very Small Cracks or the Cracks in the Early Stage. In Proceedings of the 2nd ICM, Cleveland, OH, USA, 16– 24 August 1976; pp. 627–631. [Google Scholar]
- Stolarz, J.; Madelaine-Dupuich, O.; Magnin, T. Microstructural Factors of Low Cycle Fatigue Damage in Two Phase Al–Si Alloys. Mater. Sci. Eng. A 2001, 299, 275–286. [Google Scholar] [CrossRef]
- Joyce, M.R.; Styles, C.M.; Reed, P.A.S. Elevated Temperature Short Crack Fatigue Behaviour in near Eutectic Al–Si Alloys. Int. J. Fatigue 2003, 25, 863–869. [Google Scholar] [CrossRef]
- McDowell, D.; Gall, K.; Horstemeyer, M.; Fan, J. Microstructure-Based Fatigue Modeling of Cast A356-T6 Alloy. Eng. Fract. Mech. 2003, 70, 49–80. [Google Scholar] [CrossRef] [Green Version]
- Xue, Y.; Burton, C.L.; Horstemeyer, M.F.; McDowell, D.L.; Berry, J.T. Multistage Fatigue Modeling of Cast A356-T6 and A380-F Aluminum Alloys. Metall. Mater. Trans. B 2007, 38, 601–606. [Google Scholar] [CrossRef]
- Xue, Y.; El Kadiri, H.; Horstemeyer, M.F.; Jordon, J.B.; Weiland, H. Micromechanisms of Multistage Fatigue Crack Growth in a High-Strength Aluminum Alloy. Acta Mater. 2007, 55, 1975–1984. [Google Scholar] [CrossRef]






| Process Element | Die-Casting | Precision Sand Casting | Precision Sand Casting + Crank Chill |
|---|---|---|---|
| Alloy | Al-10Si-2.5Cu-1.0Fe max | Al-7Si-3.5Cu-0.4Fe max | Al-8Si-3Cu-0.6Fe max |
| Metal temperature | 680–700 °C | 730–760 °C | 730 °C |
| Modifier/grain refiner | None | None | Sr/TiB |
| Casting yield | 70–75% | 73% | 55% |
| Soundness | Low | Medium | High |
| Typical scrap | 10–15% In-house 3–5% Customer | 3–5% In-house 0.1–0.3% Customer | 3–5% In-house 0.1–0.3% Customer |
| Heat treatment | T5 | T5 | T5 and T7 |
| Durability | Low | Medium | High |
| Designated Symbol | Definition |
|---|---|
| F | As-cast or fabricated and no mechanical properties specified |
| O | Annealed to obtain lowest-strength temper |
| W | Solution heat-treated |
| T1 | Cooled from an elevated temperature-shaping process and naturally aged |
| T2 | Cooled from an elevated temperature-shaping process, cold-worked, and naturally aged |
| T3 | Solution heat-treated, cold-worked, and naturally aged |
| T4 | Solution heat-treated and naturally aged |
| T5 | Cooled from an elevated temperature-shaping process and artificially aged |
| T6 | Solution heat-treated and artificially aged |
| T7 | Solution heat-treated and artificially overaged |
| T8 | Solution heat-treated, cold-worked, and artificially overaged |
| T9 | Solution heat-treated, artificially overaged, and cold-worked |
| T10 | Cooled from an elevated temperature-shaping process, cold-worked, and artificially aged |
| Alloy | Fabrication Method | Chemical Composition (in wt.%) | Temper | Temperature, °C | Tensile Properties | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Si | Mg | Mn | Fe | Ni | Cu | Ti | Sr | Others, Each | YS, MPa | UTS, MPa | % El | E, GPa | ||||
| Silafont®-36 [87] | Vacuum-assisted-HPDC | 10.3 | 0.3 | 0.6 | 0.09 | - | - | 0.07 | 0.01 | Zn, <0.001 | F | RT | 169 ± 6 | 314 ± 3 | 6 ± 2 | 80 ± 2 |
| Silafont®-36 [148] | T4 | RT | 142 ± 11 | 222 ± 33 | 11 ± 5 | 72 ± 3 | ||||||||||
| Silafont®-36 [149] | T7 | RT | 130 ± 9 | 174 ± 21 | 7 ± 3 | 72 ± 3 | ||||||||||
| 360 alloy [82] | HPDC | 9–10 | 0.5 | 0.4 | 2 | 0.5 | 0.6 | - | - | Zn, 0.5; Sn, 0.2 | F | RT | 170 | 305 | 3 | - |
| A360.0 alloy [82] | HPDC | 9–10 | 0.5 | 0.4 | 1.3 | 0.5 | 0.6 | - | - | Zn, 0.5; Sn, 0.2 | F | RT | 165 | 315 | 4 | - |
| 380 alloy [82] | HPDC | 8–9 | 0.1 | 0.5 | 2 | 0.5 | 3.0–4.0 | - | - | Zn, 3; Sn, 0.4 | F | RT | 160 | 315 | 3 | - |
| A380.0 alloy [82] | HPDC | 8–9 | 0.1 | 0.5 | 1.3 | 0.5 | 3.0–4.0 | - | - | Zn, 3; Sn, 0.4 | F | RT | 160 | 325 | 4 | - |
| 383 alloy [82] | HPDC | 10–12 | 0.1 | 0.5 | 1.3 | 0.3 | 2.0–3.0 | - | - | Zn, 3; Sn, 0.2 | F | RT | 150 | 310 | 4 | - |
| 384 alloy [82] | HPDC | 11–12 | 0.1 | 0.5 | 1.3 | 0.5 | 3.0–4.5 | - | - | Zn, 3; Sn, 0.4 | F | RT | 165 | 330 | 3 | - |
| 390 alloy [82] | HPDC | 16–18 | 0.5–0.7 | 0.1 | 1.3 | - | 4.0–5.0 | 0.2 | - | Zn, 0.1 | F | RT | 240 | 280 | <1 | - |
| B390.0 alloy [82] | HPDC | 16–18 | 0.5–0.7 | 0.5 | 1.3 | 0.1 | 4.0–5.0 | 0.2 | - | Zn, 1.5 | F | RT | 250 | 315 | <1 | - |
| 392 alloy [82] | HPDC | 18–20 | 0.8–1.2 | 0.2–0.6 | 1.5 | 0.5 | 0.4–0.8 | 0.2 | - | Zn, 0.5; Sn, 0.3 | F | RT | 270 | 290 | <1 | - |
| 413 alloy [82] | HPDC | 11–13 | 0.1 | 0.4 | 2 | 0.5 | 1 | - | - | Zn, 0.5; Sn, 0.2 | F | RT | 145 | 295 | 3 | - |
| A413.0 alloy [82] | HPDC | 11–13 | 0.1 | 0.4 | 1.3 | 0.5 | 1 | - | - | Zn, 0.5; Sn, 0.2 | F | RT | 130 | 290 | 4 | - |
| C443.0 alloy [82] | HPDC | 5–6 | 0.1 | 0.4 | 2 | 0.5 | 0.6 | - | - | Zn, 0.5; Sn, 0.2 | F | RT | 95 | 230 | 9 | - |
| A356 alloy [81] | PMC | 7–8 | 0.4 | <0.1 | <0.2 | - | <0.2 | - | - | Zn, <0.1 | T61 | RT | 179 | 262 | 5 | - |
| 356 alloy [81] | PMC | 7 | 0.3 | - | 0.2 | - | 0.05 | - | - | - | T6 | RT | 152 | 228 | 3 | - |
| Al-Si alloy [81] | HPDC | 8 | 0.2 | 0.5 | 0.4 | - | 0.1 | - | - | Zn, 0.4 | F | RT | 115 ± 1 | 264 ± 3 | 9 ± 1 | - |
| T4 | RT | 98 ± 1 | 230 ± 3 | 16 ± 2 | - | |||||||||||
| Al-Si-Zn alloy [81] | HPDC | 8.8 | 0.4 | 0.5 | 0.3 | - | 0.5 | - | - | Zn, 1.5 | F | RT | 147 ± 2 | 300 ± 8 | 6 ± 1 | - |
| T4 | RT | 159 ± 3 | 313 ± 2 | 11 ± 1 | - | |||||||||||
| Silafont®-36 [40] | HPDC | 10–12 | 0.1–0.5 | 0.5–0.8 | <0.2 | - | <0.03 | 0.04–0.2 | 0.01–0.03 | Zn, <0.07; P, <0.001 | F | RT | 120–150 | 250–290 | 5–11 | - |
| 0.3 | T4 | RT | 133 | 229 | 4 | - | ||||||||||
| 0.3 | T6 | RT | 250 | 299 | 1 | - | ||||||||||
| T7 | RT | 120–170 | 200–240 | 15–20 | - | |||||||||||
| Silafont®-38 [40] | HPDC | 8–10 | 0.1–0.5 | 0.5–0.8 | <0.2 | - | 0.1–0.4 | <0.2 | 0.01 | Zn, 0.1–0.4 | T6 | RT | 230–270 | 300–345 | 6–9 | - |
| T6 | RT | 180–200 | 250–275 | 8–10 | - | |||||||||||
| Castaman®-35 [40] | HPDC | 10–11 | 0.2–0.5 | 0.5–0.8 | <0.2 | - | <0.03 | 0.2 | - | Zn, 0.1 | F | RT | 120–150 | 200–270 | 4–9 | - |
| T6 | RT | 180–280 | 250–340 | 6–12 | - | |||||||||||
| Castasil®-37 [40] | HPDC | 9–11 | <0.1 | 0.4–0.6 | <0.2 | - | <0.1 | <0.2 | 0.02 | Zn, <0.1; Mo, 0.2; Zr, 0.2 | F | RT | 120–150 | 260–300 | 10–14 | - |
| Thermodur®-73 [40] | Sand or gravity casting or HPDC | 10–12 | 1.8–2.3 | <0.4 | <0.15 | 2–2.3 | 2–2.3 | 0.1 | - | Zn, 0.1 | T5 | RT | 130–155 | 250–280 | 1–2 | - |
| Castasil®-21 [40] | HPDC | 8–9 | <0.03 | <0.01 | 0.5–0.7 | - | <0.02 | <0.01 | 0.02 | Zn, <0.07 | F | RT | 90–100 | 200–230 | 6–9 | - |
| O | RT | 80–90 | 170–190 | 9–14 | - | |||||||||||
| A380 alloy [80] | HPDC | 9 | 0.1 | 0.2 | 0.9 | 0.1 | 3.1 | - | - | Zn, 0.5; Sn, 0.01 | F | RT | 180 | 355 | 6.1 | - |
| T4 | RT | 225 | 419 | 11.3 | - | |||||||||||
| T6 | RT | 354 | 466 | 6.6 | - | |||||||||||
| Al-12.7Si-0.7Mg alloy [150] | Direct chill casting + Hot extrusion | 12.7 | 0.7 | - | - | - | - | - | - | - | T1 | RT | 175 | 265 | 15.5 | - |
| T4 | RT | 188 | 316 | 15.2 | - | |||||||||||
| T6 | RT | 300 | 348 | 6.2 | - | |||||||||||
| Al-7.0Si-0.3Mg-V-Zr alloy [65] | PMC with steel | 6.5 | 0.3 | 0.3 | 0.4 | 0.01 | 0.06 | 0.1 | - | Zn, 0.05; Bi; Cr; Ca; V, 0.3; Zr, 0.2 | T6 | 20 | 228 ± 3 | 273 ± 3 | 3.5 | - |
| T6 | 300 | 96 ± 2 | 100 ± 3 | 11 ± 2 | - | |||||||||||
| Al-7.0Si-3.0Cu-0.3Mg-V-Zr alloy [65] | PMC with steel | 6.4 | 0.3 | 0.3 | 0.5 | 0.01 | 3.7 | 0.11 | - | Zn, 0.06; Bi; Cr; Ca; V, 0.3; Zr, 0.2 | T6 | 20 | 353 ± 2 | 383 ± 5 | 1 | - |
| T6 | 300 | 108 ± 2 | 116 ± 3 | 11 ± 1 | - | |||||||||||
| A357 alloy [72] | Investment casting | 7.1 | 0.6 | <0.01 | 0.06 | - | 0.01 | 0.13 | 0.001 | Zn, <0.01 | T6 | RT | 272 | 331 | 5.9 | - |
| 7.1 | 0.6 | <0.01 | 0.06 | - | 0.01 | 0.1 | 0.01 | Zn, <0.01 | T6 | RT | 299 | 353 | 7.6 | - | ||
| 7.1 | 0.6 | <0.01 | 0.06 | - | 0.01 | 0.1 | 0.01 | Zn, <0.01 | HIP + T6 | RT | 327 | 361 | 6.5 | - | ||
| Al-7.0Si-0.5Cu-0.3Mg alloy [56] | Gravity casting | 7 | 0.3 | - | - | - | 0.5 | - | - | - | T7 | RT | 260 ± 2 | 304 ± 4 | 5 ± 1 | 77 ± 6 |
| Al-7.0Si-0.3Mg alloy [56] | Lost foam casting | 7 | 0.3 | - | - | - | - | - | - | - | T7 | RT | 240 ± 5 | 251 ± 6 | 0.8 | 68 ± 5 |
| Al-Si piston alloy [58] | Sand casting | 10.8 | 0.7 | 0.4 | 0.2 | 2.7 | 4.2 | 0.2 | - | Zn, 0.1; Ce, 0.2 | T6 | 25 | - | 289 | 0.1 | - |
| T6 | 350 | 73 | 92 | 6 | - | |||||||||||
| T6 | 425 | 35 | 42 | 12 | - | |||||||||||
| Al-Mg-Si alloy [151] | PMC | 6.8 | 0.4 | - | 0.1 | - | 0 | 0.07 | 0.03 | - | F | RT | 107 | 255 | 19 | - |
| 7 | 0.4 | - | 0.1 | - | 0.5 | 0.07 | 0.02 | F | RT | 117 | 281 | 15 ± 2 | - | |||
| 7.1 | 0.4 | - | 0.1 | - | 1.6 | 0.07 | 0.02 | F | RT | 136 | 317 | 14 | - | |||
| 6.9 | 0.4 | - | 0.2 | - | 3.2 | 0.08 | 0.02 | F | RT | 149 | 252 ± 9 | 2 | - | |||
| Al-Si-Cu alloy [77] | HPDC | 17.1 | - | - | - | - | 4.2 | - | - | - | F | RT | 236 | 318 | - | 75 |
| 17.3 | - | - | - | - | 2.4 | - | - | F | RT | 199 | 298 | - | 72 | |||
| 20 | - | - | - | - | 2.6 | - | - | F | RT | 234 | 297 | - | 80 | |||
| EN AC-AlSi7Mg0.3 alloy [66] | PMC with steel | 7.5 | 0.3 | 0.002 | 0.1 | - | - | 0.2 | - | - | T6/A | RT | 221 | 303 | 11 | 73 |
| T6/B | RT | 233 | 301 | 11 | 73 | |||||||||||
| A319 alloy [61] | Precision sand casting | 6.2 | 0.3 | 0.3 | 0.5 | 0.3 | 3.4 | - | 0.02 | Zn, 0.8; Sn, 0.1 | HIP + T6 | RT | 249 | 315 | 2.5 | 75 |
| 6.2 | 0.3 | 0.3 | 0.5 | 0.3 | 3.4 | - | 0.02 | Zn, 0.8; Sn, 0.1 | HIP + T6 | RT | 216 | 227 | 2.4 | 76 | ||
| 6.2 | 0.3 | 0.3 | 0.5 | 0.3 | 3.4 | - | - | Zn, 0.8; Sn, 0.1 | HIP + T6 | RT | 205 | 275 | 2.5 | 76 | ||
| 6.2 | 0.3 | 0.3 | 0.5 | 0.3 | 3.4 | - | - | Zn, 0.8; Sn, 0.1 | HIP + T6 | RT | 202 | 220 | 1.4 | 76 | ||
| AlSi12CuNiMg piston alloy [57] | Gravity casting | 12.7 | 1 | 0.1 | 0.6 | 0.8 | 1.2 | - | - | Zn, 0.16 | T6 | 25 | 106 ± 10 | 237 ± 16 | 1.5 | 86 ± 6 |
| T6 | 250 | 99 | 153 | 3.9 | 75 | |||||||||||
| T6 | 300 | 80 | 103 | 12.4 | 65 | |||||||||||
| A319 alloy [152] | Gravity casting | 6.4 | 0.4 | 0.3 | 0.5 | 0.02 | 3.1 | 0.04 | 0.02 | Zn, 0.5 | F | RT | 224 ± 4 | 234 ± 5 | 2.3 | 78 ± 2 |
| T4 | RT | 257 ± 5 | 264 ± 5 | 2 | 78 ± 1 | |||||||||||
| T6 | RT | 276 ± 5 | 282 ± 7 | 1.8 | 76 ± 1 | |||||||||||
| T7 | RT | 198 ± 3 | 216 ± 5 | 3.1 | 75 ± 1 | |||||||||||
| A319 alloy [62] | Precision sand casting | 6.2 | - | - | - | - | 3.4 | - | - | - | T6 (HSCR) | RT | 227 | 295 | 2.6 | 76 |
| T6 (LCSR) | RT | 207 | 209 | 1.5 | 76 | |||||||||||
| A356 alloy [64] | PMC | 7 | 0.3 | - | - | - | - | - | - | - | T6 | RT | 185 | 260 | 5 | - |
| A319 alloy [64] | PMC | 6 | - | - | - | - | 3.5 | - | - | - | T6 | RT | 185 | 275 | 3 | - |
| Al-Si-Cu-Zr-V alloy [64] | PMC with steel | 7 | 0.5 | - | 0.1 | - | 1 | 0.1 | - | - | T6 | RT | 297 ± 5 | 340 ± 4 | 2.8 | - |
| PMC with steel | 7 | 0.5 | - | 0.1 | - | 1 | 0.1 | - | Zr, 0.2 | T6 | RT | 301 ± 2 | 337 ± 7 | 2.3 | - | |
| PMC with steel | 7 | 0.5 | - | 0.12 | - | 1 | 0.1 | - | Zr, 0.2; V, 0.3 | T6 | RT | 309 ± 3 | 335 ± 12 | 2 | - | |
| Aural 2 [2] | Die-casting | 9.5–11.5 | 0.3 | 0.5–0.6 | 0.15–0.2 | - | <0.03 | <0.08 | 0.01–0.02 | <0.03 | F | RT | 140 | 310 | 8.6 | - |
| T5 | RT | 189–230 | 303–339 | 8–9 | - | |||||||||||
| Aural 3 [2] | Die-casting | 9.5–11.5 | 0.4–0.6 | 0.5–0.6 | 0.2 | - | <0.03 | <0.08 | 0.01–0.16 | <0.03 | F | RT | 130–160 | 250–310 | 4–8 | - |
| T5 | RT | 190–240 | 300–340 | 4–7 | - | |||||||||||
| Aural 5 [2] | Die-casting | 6.5–9.5 | 0.1–0.6 | 0.3–0.6 | 0.2 | - | <0.03 | <0.08 | 0.03 | <0.03 | F | RT | 125–145 | 245–265 | 9–12 | - |
| T5 | RT | 120–160 | 190–260 | 6–12 | - | |||||||||||
| Mercalloy A368 [2] | 8.5–9.5 | 0.1–0.3 | 0.3–0.4 | 0.3 | - | 0.3 | 0.2 | 0.05–0.07 | Zn, <0.1 | F | RT | 125–140 | 260–275 | 10–12 | - | |
| T6 | RT | 185–200 | 280–295 | 14–18 | - | |||||||||||
| Mercalloy A367 [2] | Die-casting | 8.5–9.5 | 0.3–0.5 | 0.3–0.4 | <0.3 | - | - | <0.2 | 0.05–0.07 | <0.05 | F | RT | 115 | 270 | 8.1 | - |
| T5 | RT | 170–205 | 295–310 | 5–9 | - | |||||||||||
| T6 | RT | 230–245 | 295–340 | 8–10 | - | |||||||||||
| Mercalloy A362 [2] | Die-casting | 10.5–11.5 | 0.5–0.7 | 0.3–0.4 | 0.4 | <0.1 | <0.2 | <0.2 | 0.05–0.07 | - | F | RT | 125–140 | 260–275 | 9–11 | - |
| T6 | RT | 230–250 | 295–315 | 14–16 | - | |||||||||||
| Alcoa EZCast 370 [2] | Die-casting | 6.0–9.0 | 0.2–0.8 | 0.1–0.8 | <0.2 | - | - | <0.2 | 0.02 | <0.05 | F | RT | 105–140 | 250–280 | 8–13 | - |
| T5 | RT | 150–220 | 245–310 | 6–10 | - | |||||||||||
| T4 | RT | 100–120 | 205–235 | 18–23 | - | |||||||||||
| T6 | RT | 135–250 | 195–300 | 7–16 | - | |||||||||||
| ER4043 [127] | WAAM | 5 | - | - | 0.14 | - | 0.03 | 0.02 | - | Zn, 0.02 | AD | RT | 82 | 164 | 13.8 | - |
| FSP | RT | 88 | 148 | 33.2 | - | |||||||||||
| Alloy | Primary α-Al | Nanohardness, GPa | Q Phase Area, % | θ Phase Area, % | |
|---|---|---|---|---|---|
| Cu at Center, wt.% | Cu at Edge, wt.% | ||||
| Cu 0 | - | - | 0.66 ± 0.02 | - | - |
| Cu 0.5 | 0.11 ± 0.01 | 0.11 ± 0.02 | 0.67 ± 0.02 | 0.5 | - |
| Cu 1.5 | 0.37 ± 0.03 | 0.51 ± 0.07 | 0.74 ± 0.02 | 1.4 | 0.8 |
| Cu 3.0 | 0.74 ± 0.04 | 0.9 ± 0.13 | 0.87 ± 0.02 | 2.2 | 2.6 |
| Alloy/Alloy System | Fabrication Method | Condition | Porosity (%) | Stress Ratio, Rσ | Loading Mode | Temperature, °C | Fatigue Strength, MPa | Test Cycles |
|---|---|---|---|---|---|---|---|---|
| Al-12Si [260] | Gravity casting | F | - | - | Uniaxial | RT | 80 | 1.00 × 107 |
| F | - | - | Uniaxial | 250 | 50 | 1.00 × 107 | ||
| F | - | - | Uniaxial | 300 | 40 | 1.00 × 107 | ||
| Al-7.0Si-0.3Mg [261] | Gravity casting | T6 | - | −1 | Uniaxial | RT | 60 | 1.00 × 107 |
| Al-7.0Si-0.6Mg [261] | Gravity casting | T6 | - | −1 | Uniaxial | RT | 80 | 1.00 × 107 |
| Al-7.0Si-0.5Mg [262] | NEMAK Rotacast© process | T79 | <0.01 | −1 | Uniaxial | RT | 82 | 1.00 × 107 |
| Al-7.0Si-3.0Cu-0.3Mg [263] | PMC with steel | T6 | - | 0.01 | Uniaxial | RT | 108 | 1.50 × 106 |
| T6 | - | 0.01 | Uniaxial | 200 | 124 | 1.50 × 106 | ||
| T6 | - | 0.01 | Uniaxial | 300 | 72 | 1.50 × 106 | ||
| Al-12.0Si-0.9Cu-0.35Mg-0.5Fe- 0.3Ni-0.1Ti-0.2Mn -0.2Zn [264] | Gravity casting | T6 | - | - | Uniaxial | RT | 90 | 1.00 × 107 |
| T6 | - | - | Uniaxial | 250 | 78 | 1.00 × 107 | ||
| T6 | - | - | Uniaxial | 300 | 60 | 1.00 × 107 | ||
| T6 | - | - | Uniaxial | 350 | 27 | 1.00 × 107 | ||
| Al-10.4Si-3.1Cu-0.9Mg- 2.0Ni-0.2Ti-0.5Mn [265] | Sand casting | T6 | - | −1 | Uniaxial | 350 | 42.5 | 1.00 × 107 |
| T6 | - | −1 | Uniaxial | 425 | 32.5 | 1.00 × 107 | ||
| Al-12.0Si-4.0Cu-1.2Mn [257] | PMC | T6 | - | −1 | Uniaxial | RT | 125 | 1.00 × 106 |
| T6 | - | −1 | Uniaxial | 350 | 47.5 | 1.00 × 106 | ||
| Al-11.4Si-4.1Cu-2.1Ni -0.8Mg-0.2Fe-0.2Zr [259] | Casting | T6 | - | - | Rotating bending | 350 | 50 | 8.21 × 106 |
| T6 + 350 °C for 100 h | - | Rotating bending | 350 | 50 | 2.03 × 107 | |||
| T6 + 425 °C for 100 h | - | Rotating bending | 350 | 50 | 2.24 × 107 | |||
| A380/Al-Si-Cu [80] | HPDC | F | 0.12 ± 0.06 | 0.1 | Uniaxial | RT | 205 | 1.00 × 107 |
| T4 | - | 0.1 | Uniaxial | RT | 240 | 1.00 × 107 | ||
| T6 | 0.13 ± 0.05 | 0.1 | Uniaxial | RT | 260 | 1.00 × 107 | ||
| Al-7.0Si-0.3Mg-V-Zr [65] | PMC with steel | T6 | - | 0.01 | Uniaxial | 20 | 120 | 1.50 × 106 |
| T6 | - | 0.01 | Uniaxial | 300 | 50 | 1.50 × 106 | ||
| Al-7.0Si-3.0Cu-0.3Mg-V-Zr [65] | PMC with steel | T6 | - | 0.01 | Uniaxial | 20 | 104 | 1.50 × 106 |
| T6 | - | 0.01 | Uniaxial | 300 | 64 | 1.50 × 106 | ||
| A357 alloy [72] | Investment casting | T6 | 5 mm2 | 0.1 | Uniaxial | RT | 73 | 1.00 × 106 |
| Sr addition + T6 | 9.5 mm2 | 0.1 | Uniaxial | RT | 80 | 1.00 × 106 | ||
| Sr addition + HIP + T6 | - | 0.1 | Uniaxial | RT | 107 | 1.00 × 106 | ||
| Al-Si-Cu-Ni alloy [58] | Sand casting | T6 | - | −1 | Uniaxial | 25 | 79.5 | 1.00 × 107 |
| T6 | - | −1 | Uniaxial | 350 | 39.5 | 1.00 × 107 | ||
| T6 | - | −1 | Uniaxial | 425 | 33.5 | 1.00 × 107 |
| Alloys | Heat Treatment/Test Condition | Cyclic Yield Strength, MPa | Cyclic Strain-Hardening Exponent | Cyclic Strength Coefficient, MPa | Fatigue Strength Coefficient, MPa | Fatigue Strength Exponent | Fatigue Ductility Coefficient | Fatigue Ductility Exponent | Fatigue Damage Transition Exponent |
|---|---|---|---|---|---|---|---|---|---|
| σ′y | n′ | K′ | σ′f | b | ε′f | c | b | ||
| Silafont®-36 alloy [86] | F/RT | 238 | 0.1 | 464 | 418 | −0.09 | 0.35 | −0.86 | 1.05 |
| Silafont®-36 alloy [148] | T4/RT | 208 | 0.01 | 408 | 332 | −0.08 | 0.12 | −0.79 | 1.16 |
| A356 alloy [276] | T5/RT | - | 0.15 | 502 | 345 | −0.1 | 0.05 | −0.57 | - |
| T6/RT | - | 0.24 | 1628 | 800 | −0.15 | 0.03 | −0.55 | - | |
| ModT6/RT | - | 0.21 | 1105 | 584 | −0.12 | 0.03 | −0.52 | - | |
| Al-10Si alloy [278] | T4/RT | - | 0.06 | 336 | - | - | - | - | - |
| Al-Si-Cu-Mg alloy [230] | T6/RT | 265 | 0.14 | 630 | 285 | −0.03 | 0.01 | −0.25 | - |
| Al-Si-Cu-Mg-Mn alloy [230] | T6/RT | 322 | 0.11 | 677 | 561 | −0.1 | 0.09 | −0.85 | - |
| Al-Si-Cu-Mg-Ti alloy [64] | T6/RT | 330 | 0.24 | 1758 | 560 | −0.13 | 0.004 | −0.43 | - |
| Al-Si-Cu-Mg-Ti-Zr alloy [64] | T6/RT | 344 | 0.27 | 2183 | 648 | −0.15 | 0.005 | −0.46 | - |
| Al-Si-Cu-Mg-Ti-Zr-V alloy [64] | T6/RT | 347 | 0.28 | 2393 | 609 | −0.14 | 0.005 | −0.43 | - |
| Al-Si-Cu-Mg-Ti-V-Zr alloy [280] | F/RT | 262 | 0.09 | 474 | 386 | −0.08 | 0.09 | −0.77 | - |
| A319 + Sr + HSCR alloy [61] | T6/RT | - | 0.1404 | 834.3 | 822.8 | −0.13 | 0.79 | −0.88 | - |
| A319 + Sr + LSCR alloy [61] | T6/RT | - | 0.1141 | 458.9 | 379.7 | −0.09 | 0.17 | −0.76 | - |
| A319 + HSCR alloy [61] | T6/RT | - | 0.1627 | 703.4 | 780.1 | −0.14 | 0.49 | −0.72 | - |
| A319 + LSCR alloy [61] | T6/RT | - | 0.1006 | 504.5 | 489.1 | −0.11 | 0.01 | −0.53 | - |
| A319 + HSCR alloy [62] | T6/RT | - | 0.23 | 1930.2 | - | - | - | - | - |
| A319 + LSCR alloy [62] | T6/RT | - | 0.16 | 639.6 | - | - | - | - | - |
| A356 alloy [277] | T7/300 °C | - | 0.12 | 134.52 | 141.7 | −0.11 | 0.07 | −0.52 | - |
| A319 alloy [277] | T7/300 °C | - | 0.26 | 418.41 | 175.1 | −0.1 | 0.02 | −0.39 | - |
| A356 + Mo alloy [279] | T7/300 °C | - | 0.12 | 158.53 | 105.7 | −0.07 | 0.06 | −0.57 | - |
| Al-14Si piston alloy [283] | T6/280 °C | - | - | - | 282 | −0.15 | 0.002 | −0.11 | 3.39 |
| T6/350 °C | - | - | - | 179 | −0.15 | 0.04 | −0.32 | 2.23 | |
| T6/425 °C | - | - | - | 76 | −0.11 | 0.68 | −0.64 | 1.66 | |
| A356 alloy from rim of wheel [95] | T6/RT | - | - | - | 585 ± 1.2 | −0.11 ± 0.01 | - | - | - |
| A356 alloy from spoke of wheel [95] | T6/RT | - | - | - | 969 ± 1.3 | −0.17 ± 0.02 | - | - | - |
| A319 alloy-HIP [289] | T6/RT | - | - | - | 968.68 | −0.14 | 0.47 | −1.05 | - |
| Al-Si-Cu alloy [281] | T6/150 °C | - | - | - | - | - | 0.002 | −0.23 | - |
| T6/250 °C | - | - | - | - | - | 0.002 | −0.24 | - | |
| Al-12Si-Cu-Ni-Mg alloy [294] | T5/200 °C | - | - | - | 286.8 | - | 0.02 | - | - |
| T5/350 °C | - | - | - | 216.8 | - | 0.04 | - | - | |
| T5/400 °C | - | - | - | 111.4 | - | 0.05 | - | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dash, S.S.; Chen, D. A Review on Processing–Microstructure–Property Relationships of Al-Si Alloys: Recent Advances in Deformation Behavior. Metals 2023, 13, 609. https://doi.org/10.3390/met13030609
Dash SS, Chen D. A Review on Processing–Microstructure–Property Relationships of Al-Si Alloys: Recent Advances in Deformation Behavior. Metals. 2023; 13(3):609. https://doi.org/10.3390/met13030609
Chicago/Turabian StyleDash, Soumya Sobhan, and Daolun Chen. 2023. "A Review on Processing–Microstructure–Property Relationships of Al-Si Alloys: Recent Advances in Deformation Behavior" Metals 13, no. 3: 609. https://doi.org/10.3390/met13030609
APA StyleDash, S. S., & Chen, D. (2023). A Review on Processing–Microstructure–Property Relationships of Al-Si Alloys: Recent Advances in Deformation Behavior. Metals, 13(3), 609. https://doi.org/10.3390/met13030609







