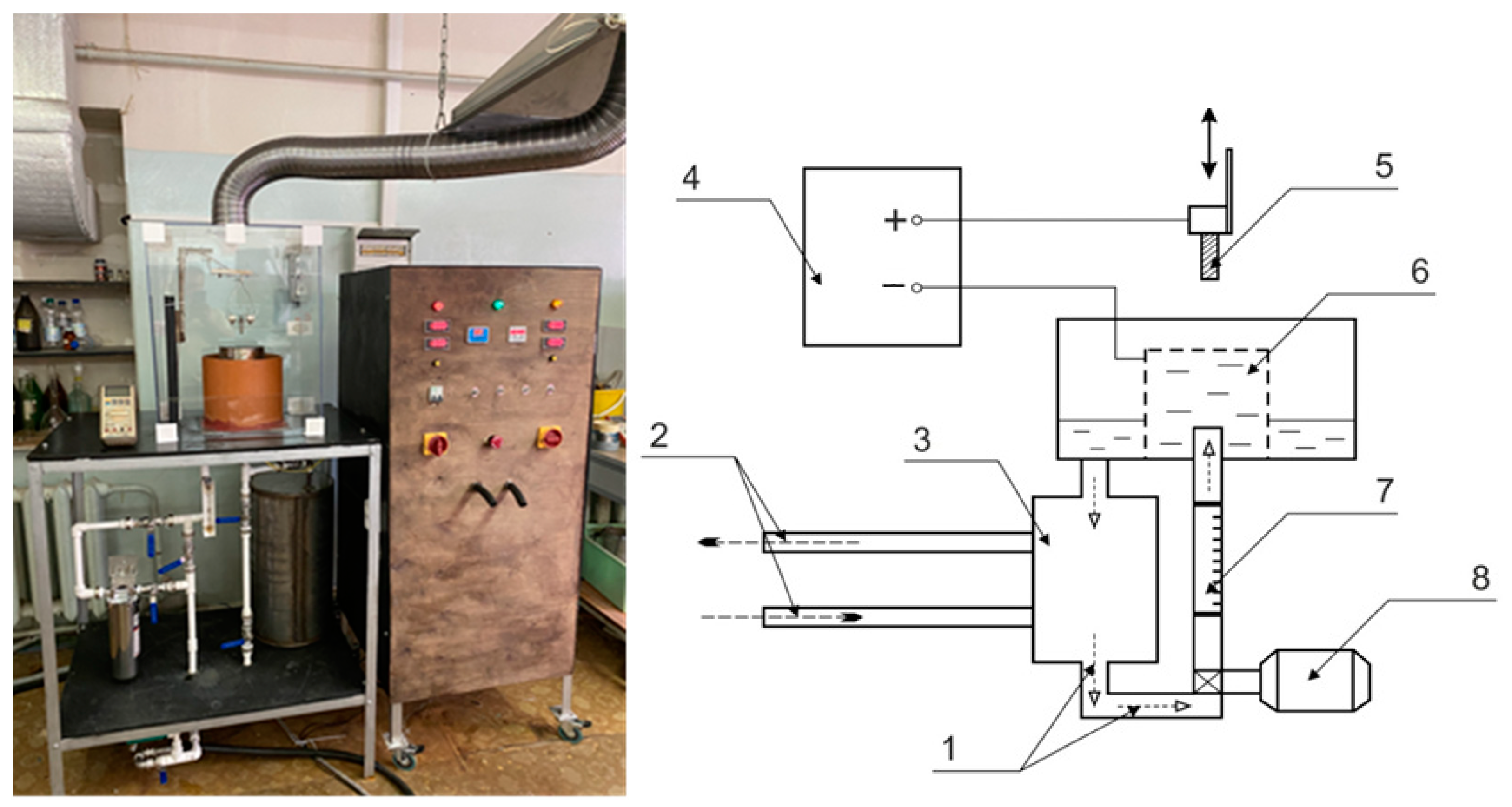
1. Introduction
Stainless steels are used as structural and functional materials in products operating under aggressive conditions in the food, chemical, and thermal power engineering industries, among others. Products made of this material are characterized by high ductility, toughness, heat resistance and corrosion resistance but low strength and hardness. To harden stainless steels, surface plastic deformation technologies aimed at creating grain boundaries and substructural hardening are effectively used, for example, in ultrasonic strain engineering technology [
1] and ultrasonic shot peening [
2,
3]. The disadvantage of deformation surface treatments is the significant increase in roughness and the need for subsequent finishing [
1,
2,
3]. Laser shock peening increases the thickness of the hardened layer to 2 mm and have been proven to solve this problem [
4,
5,
6].
Alternatives to the mechanical treatment of the metal surface include physical and chemical methods of surface hardening, such as plasma electrolytic treatment, which has a complex effect on the operational properties of metal products [
7,
8,
9]. The majority of studies cover the cathodic variant of plasma electrolytic diffusion saturation variety. Thus, during the cathodic plasma electrolytic nitriding (PEN) of 316L steel in a solution of ammonium nitrate and potassium hydroxide, the formation of FeN
0.076 and Fe
3O
4 in the surface layer was revealed [
10]. After PEN, the 10–15 μm thick oxide layer containing 39–41% oxygen is formed on the surface of the steel in a carbamide electrolyte. The maximum nitrogen content in steel of 0.68% is no longer detected at a depth of 30 μm. The cathodic nitriding of austenitic stainless steel in solutions of ammonium carbonate leads to the formation of nitrides (Fe
2N, Fe
4N, CrN and Cr
2N) and oxides (Fe
3O
4 and Cr
2O
3) of iron and chromium, and the microstructure of the nitrided layer contains a nitride zone and an internal nitriding zone [
11]. As the temperature increases, more high-nitrogen nitride, Fe
2N, is formed. The nitriding of stainless steel has shown a positive result for increasing wear and corrosion resistance. After the PEN of 316L steel in solutions of carbamide or ammonium nitrate with the addition of potassium hydroxide, the dry friction coefficient with a corundum ball decreases from 0.19 in the untreated sample to 0.13 with an increase in wear resistance of 4.4–10 times [
10]. The PEN of 304, 316L and 430 stainless steels in sodium nitrite solution proved to be an effective method of inhibiting pitting corrosion in 0.5M sodium chloride solution [
12].
After the cathodic plasma electrolytic carburizing (PEC) of 12Cr18Ni10Ti steel, FeO iron oxides were detected in the glycerin electrolyte [
13]. An increase in the applied voltage increased the degree of grain grinding to an extent to which nanoscale crystals formed alongside the increase in surface roughness [
14]. After the carburizing of 12Cr18Ni10Ti steel, the compression of ferrite and austenite crystal lattices was observed, which occurs due to the displacement of the lines (110) α-Fe and (111) γ-Fe. Additional phases of Fe
3O
4, (Cr,Fe)
7C
3, Fe
15Cr
4Ni
2, CrN and CrFe were detected in the PEC of 304 austenitic steel in a chloride-glycerin electrolyte [
15] and 403 stainless martensitic steel, including Fe
3O
4, CrFe, FeO and CrC [
16]. Cathodic carburizing during pulse treatment (250~600 V; 1500 Hz) in an electrolyte of glycerin and sodium chloride on 1Cr18Ni9Ti steel forms a hardened layer with a thickness of 0.2 mm and a microhardness up to 513 HV for 3–5 min [
17]. After the cathodic carburizing of 304 steel in an electrolyte of glycerin (80%) and potassium chloride at a voltage of 350 V for 3 min, the thickness of the layer hardened to 762 HV reaches 0.085 mm [
18].
In the cathodic plasma electrolytic nitrocarburizing (PENC) of 316L stainless steel in a carbamide electrolyte, the main phase appears as austenite nitrogen [
19]. Additionally, oxides and oxygen-containing phases, including NiFe
2O
4, FeCr
2O
4 [
20], Fe
2O
3, Fe
3O
4, Cr
2O
4 [
21] and Fe(Fe,Cr)
2O
4 [
22,
23], nitrides, including Fe
3N [
21], CrN and Cr
2N [
23], carbides, including Cr
3C
2 and Cr
7C
3 [
23], and silicon dioxide [
21] are detected. The cathodic PENC of 304 steel in carbamide electrolytes increased microhardness up to 1380 HV with an increase in surface roughness from 0.025 to 0.14 μm and a 4.2-fold increase in wear resistance [
19]. In one study, 316L steel after cathodic PENC in carbamide electrolytes with various additives had an increase in microhardness up to 1200 HV and 50 times the wear resistance [
22] and, in another, an increase up to 1600 HV and 4.5 times the wear resistance [
23].
The disadvantage of cathode plasma electrolytic treatment is the low controllability of the technological process and consequent properties. In addition, the increase in surface roughness that accompanies the cathodic treatment option requires additional finishing. The anodic version of plasma electrolytic saturation as a way to increase the operational properties of stainless steel products has not been practically considered in the literature. This option of plasma electrolytic treatment, however, in addition to hardening and increasing wear and corrosion resistance allows us to reduce surface roughness and exclude subsequent finishing treatment [
24,
25,
26,
27]. In this paper, the possibility of increasing hardness and wear resistance of the stainless steel surface with various types of anodic plasma electrolytic diffusion saturation (nitriding, boriding and carburizing) is considered.
4. Discussion
Surface morphology during anodic plasma electrolytic treatment in aqueous electrolytes is determined by the competition of the processes of high-temperature oxidation, which leads to the formation of an oxide layer with a growth of roughness on the surface, and the anodic dissolution of the treated material, which leads to the alignment of the surface profile and a decrease in roughness [
38,
39,
40]. In the considered cases of the anodic diffusion saturation of austenitic stainless steel samples, in general, the prevalence of anodic dissolution is observed, alongside a decrease in surface roughness. This determines the fundamental difference between the result of the anodic and cathodic treatment shown in the Introduction. At the same time, at high saturation temperatures, the intensive oxidation of the surface is observed, partially compensating for the decrease in the weight of the samples and the roughness during anodic dissolution. Under these conditions, the surface morphology will be determined by the structural features of the oxide layers—pores (after PEB) and areas with traces of the detachment of the fragile oxidized material (after PEC at high temperatures) are visually observed. Similar morphological features were observed after the PEN [
27,
41], PEB [
42,
43] and PEC [
36,
37] of carbon steels, which determine the general mechanism of the processes of the high-temperature oxidation and the anodic dissolution of the surfaces of carbon and high-alloy steels.
In contrast to the plasma electrolytic treatment of carbon steels that do not contain alloying additives, the surface hardening of austenitic stainless steel with a low carbon content develops due to the formation of inclusion compounds in the form of carbides and nitrides. In this case, an increase in microhardness reaches the depth of phase transformations up to 20–25 microns, which is observed during PEN (
Figure 12). During PEC, when quenching with the formation of martensite or the consolidation of the crystal lattice is possible, due to the presence of carbon diffusion at a greater depth than nitrogen, the surface layer hardens up to 80 μm (
Figure 14).According to this mechanism, the hardening of low-carbon unalloyed steels is possible during anodic PEC [
36,
37] and PENC [
39,
40,
44]. The results of PEB clearly showed that the absence of inclusion compounds in the surface layer does not lead to an increase in microhardness (
Figure 13), while the PEB of medium carbon steel (0.45 wt.% C) makes it possible to harden the surface to 1800 HV [
42,
43].
Despite the decrease in the hardness of samples after PEN with an increase in the processing temperature, their wear resistance does not decrease and the relationship between hardness and wear resistance in the function of nitriding temperature is not one-digit. Wear resistance is affected by the formation of nitride particles and the formation of a low level of micro-deformations in the lattice. Nitrides formed in the diffusion layer can be incoherent, coherent or semi-coherent. Coherent and semi-coherent nitrides lead to the greater deformation of the matrix than incoherent ones. Plastic deformation plays a leading role in the wear process. At a higher nitriding temperature, the matrix of the diffusion layer apparently has greater plasticity, which significantly reduces the level of micro-deformations of the crystal lattice of the iron matrix. Therefore, wear resistance does not decrease following a decrease in hardness. The level of weight wear within the margin of error does not change.
Samples after PEB show the maximum level of weight wear and the highest friction coefficient after treatment at temperatures of 800 and 950 °C, which corresponds to the cases of the most developed pores on the surface (
Figure 4). The X-ray analysis of the sample after saturation (
Figure 10) shows the presence of FeO on the surface, which can lead to both an increase in the friction coefficient and friction weight losses.
The minimum weight wear after PEC is achieved by processing at 900 °C. It is influenced by two factors, including the maximum hardness (
Figure 14) and a large number of oxides (
Figure 5c), among which Fe
3O
4—a highly effective lubricant—appears, according to X-ray analysis (
Figure 11) [
45].
All samples after PEN, PEB and PEC show a correlation of the friction coefficient with the Kragelsky–Kombalov criterion, which is a generalized dimensionless criterion for surface roughness. With a predominance of plastic deformations in the tribo-conjugation, the molecular component of the external friction coefficient does not depend on the microgeometry of the surface. In addition, the deformation component of the friction coefficient increases with an increase in complex Δ. The friction cumulative coefficient also increases with an increase in Kragelsky–Kombalov criterion. In all plasma electrolytic treatment sessions, the maximum Kragelsky-Kombalov criterion on the friction track correlates with the highest friction coefficient of this sample.
The Kragelsky–Kombalov criterion determines the bearing capacity of the roughness profile. The smaller Δ is, the higher the bearing capacity of the roughness profile.
Table 4,
Table 5 and
Table 6 show that the loss in weight during friction is smaller in samples with lower values of the Kragelsky–Kombalov criterion. The maximum value Δ on the friction track of an untreated sample (0.989) corresponds to weight loss during friction of 23.2 ± 0.3 g.
PEN at all temperatures reduces Δ 2.4 to 2.6 times, and the weight loss due to friction at 1 km falls 46–58 times. PEB at 850 and 900 °C shows a 2.3–2.4 times lower value of Δ, and weight decreases 12.9 and 7.0 times, respectively. At 800 and 950 °C, values of Δ increase; in addition, a pronounced porosity of the oxide surface, leading to a strong increase in weight losses, becomes of great importance. After PEC, the friction on the track is less than double compared to an untreated sample, and such a roughness profile provides weight losses per 1 km of friction 232 times as small as in an untreated sample.
The relief of a rough surface also influences the friction coefficient via the distribution of material along the height of a single protrusion, that is, the shape and size of the protrusion. With a decrease in the radii of the curvature of the vertices of the microfoils, their deeper penetration into the volume of the material occurs in absolute magnitude, and the friction coefficient (the deformation part) increases, which is confirmed by
Table 4,
Table 5 and
Table 6.
Friction bonds in the process of friction after treatment are broken as a result of the plastic displacement of the counter body material, as indicated by the value of the relative insertion h/r < 0.1 in all cases. The type of wear can be characterized as fatigue wear with boundary friction and plastic contact for samples after all the described types of processing.
The assumption about the type of wear and the nature of the destruction of friction bonds is confirmed by the values of the Greenwood–Williamson complex parameter, which are greater than 3 for all the described types of processing (
Table 4,
Table 5 and
Table 6).
The actual contact area differs significantly from the nominal (geometric contact area of counterbody with the sample). The actual contact area has a minimum value of 1% (PEN at 800 °C,
Table 1) of the nominal value and a maximum of 20% (PEB at 800 °C,
Table 5) for treated samples versus 24% for untreated.
In all cases of processing, an unsaturated plastic contact is realized during the friction process. With this type of contact, the deformation of micro-dimensions does not influence the load increase and the number of protrusions increases with the load increase. As can be seen from
Table 4,
Table 5 and
Table 6, the number of protrusions that come into contact in the tribo-connection is always smaller than the number of protrusions on the contour area.
The highest values of the friction coefficients after plasma electrolytic treatment are demonstrated by samples with PEN. Moreover, the values of their friction coefficients after all nitriding temperatures are greater than those of the untreated sample, but the weight wear is two orders smaller than that of the untreated one. Equation (16) serves to explain this fact. The actual contact area
Ar of samples with PEN is the smallest of all experimental series of samples and varies from 1 to 8% of the nominal depending on the PEN temperature (
Table 4). For comparison, the actual contact area of samples after PEB is 19–20% of the nominal (
Table 5) samples with that of PEC being 9–13% of the nominal (
Table 6) samples at different temperatures. A strong decrease in the actual contact area of PEN samples at values of absolute penetration
h, which is comparable to that of other series, leads to low values of friction losses of weight per 1 km at fairly high values of the coefficient of friction.
Thus, the study showed a number of fundamental differences between the results of anodic plasma electrolytic saturation with light elements of austenitic stainless steel from the cathodic treatment option. In particular, with smaller prolonged saturation, as with the anodic treatment, there is no accumulation of high concentrations of diffusant atoms in the surface layer or a formation of inclusion compounds with a high content of nitrogen and carbon, such as Fe
2-3N during the cathodic nitriding of steel; 12Cr18Ni10Ti [
6], Fe
2N, Fe
4N, CrN and Cr
2N during the cathodic nitriding of 316L steel [
9]; and (Cr,Fe)
7C
3 and CrN during the cathodic cementation of 304 steel [
15]. Nevertheless, after anodic PEC, the microhardness value of the diffusion layers (700 HV) exceeds that obtained by cathodic carburizing (513 HV [
17]), and the results of the tribological tests showed an improvement in the wear resistance index by two orders of magnitude, significantly exceeding the results on the friction of nitrided 316L steel by the cathodic method [
8]. The analysis of friction track microtopology showed the correlation of the Kragelsky–Kombalov criterion and the friction coefficient. All this testifies to the complex influence of the hardness of the reinforced layer and the composition and morphology of the surface. Thus, the effectiveness of the use of anodic plasma electrolytic treatment to increase the hardness and wear resistance of austenitic stainless steel is shown.