Compression and Tensile Testing of L-PBF Ti-6Al-4V Lattice Structures with Biomimetic Porosities and Strut Geometries for Orthopedic Implants
Abstract
:1. Introduction
2. Materials and Methods
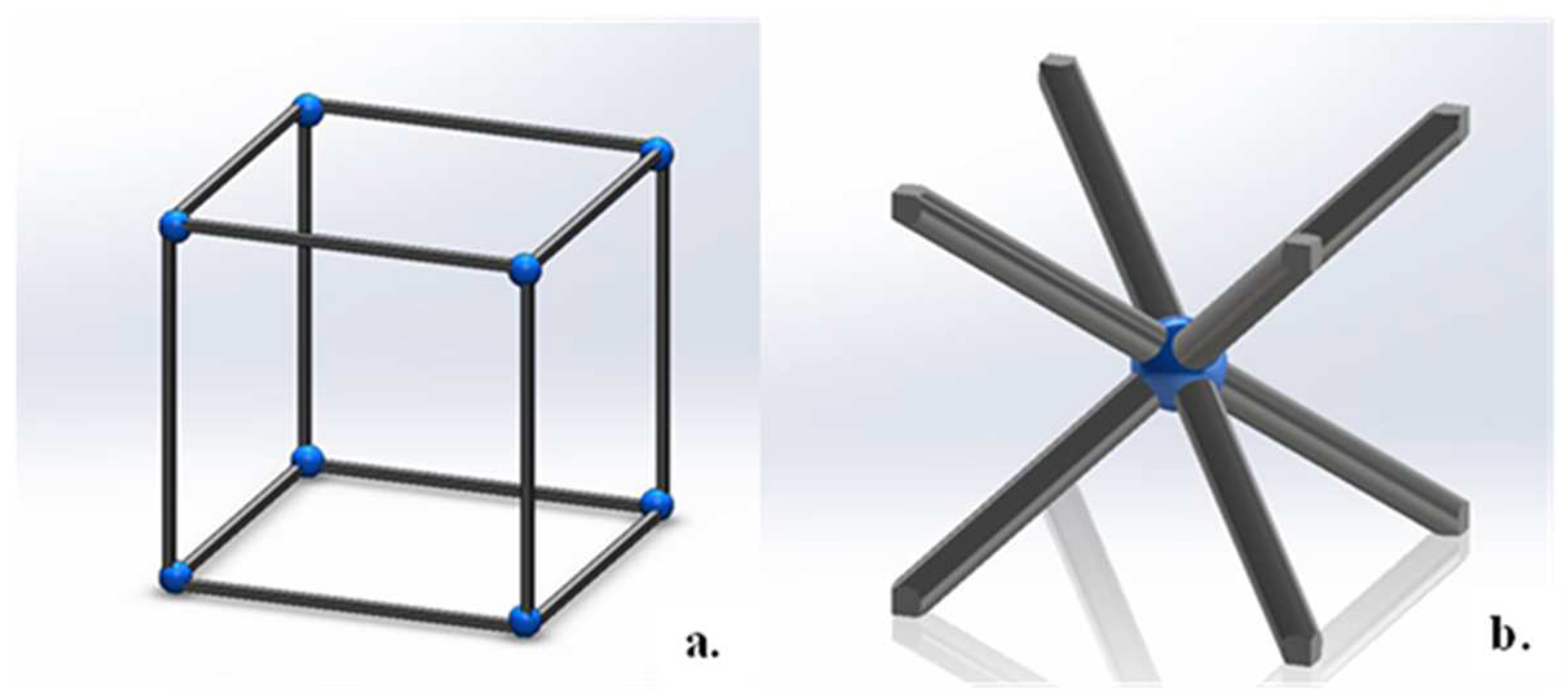
2.1. Design of Lattice Structures
2.2. Materials and AM Processing Parameters
2.3. SEM Analysis
2.4. Compression Testing
2.5. Tensile Testing
3. Results and Discussion
3.1. SEM Analysis
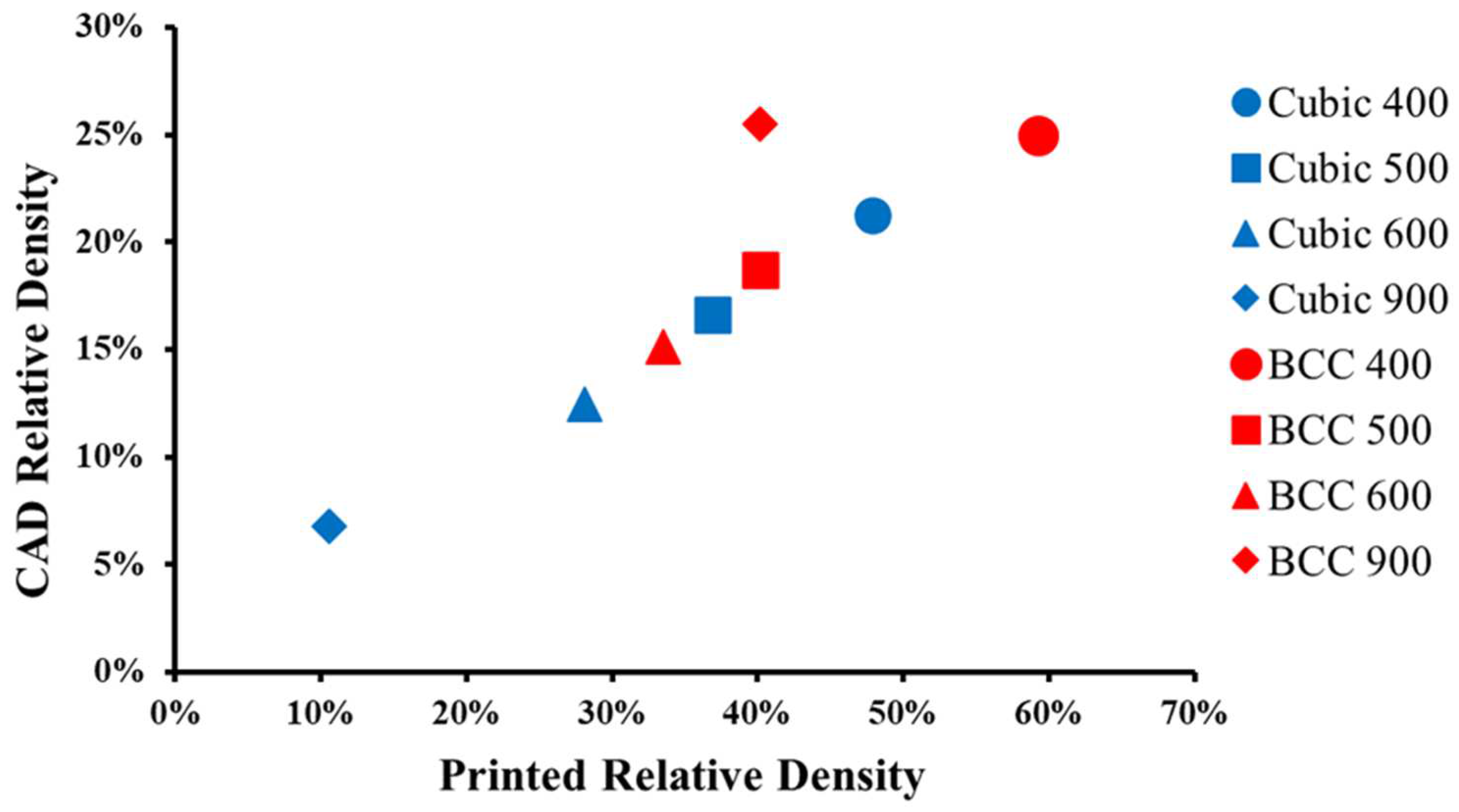
3.2. Relative Density
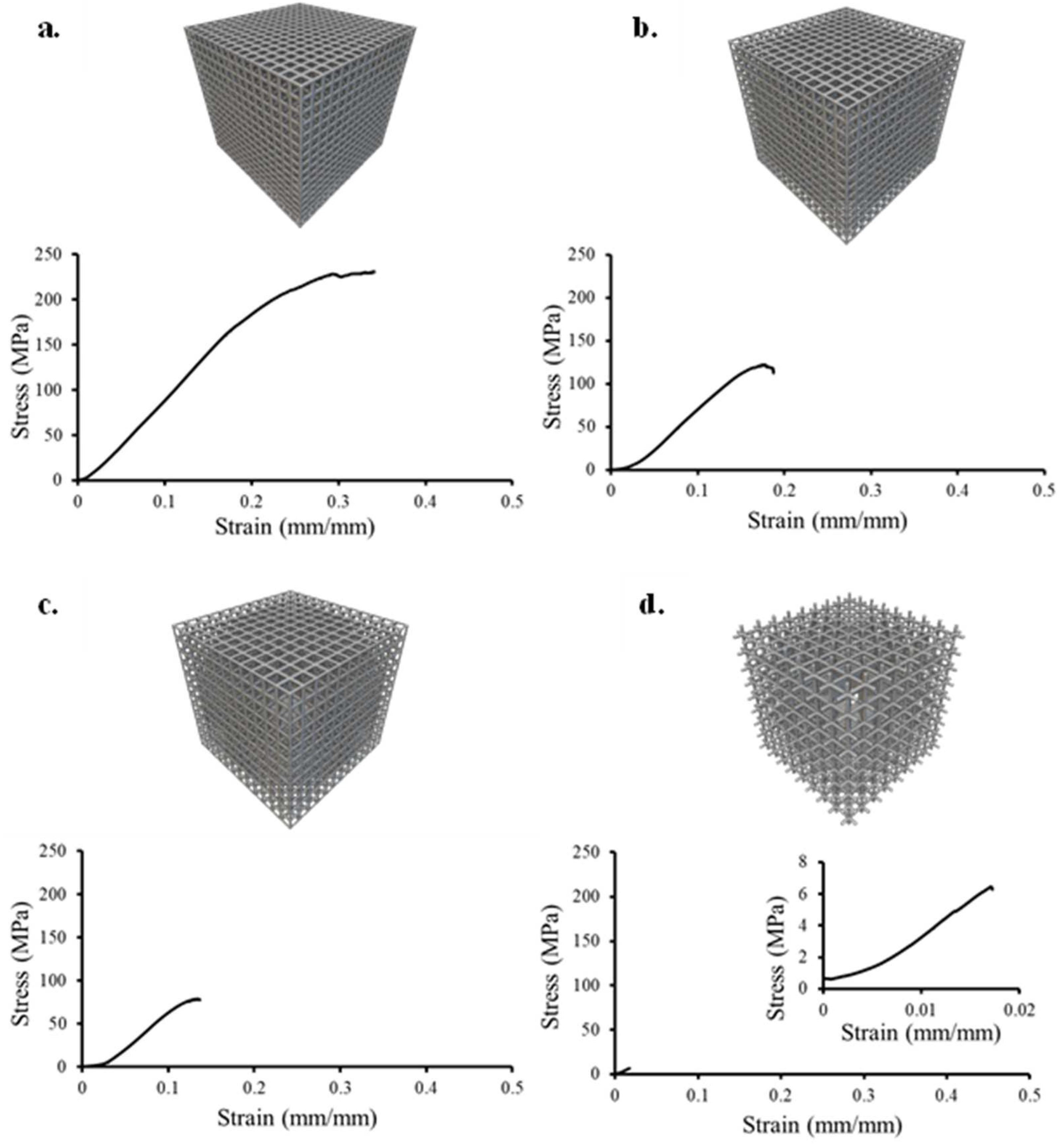
3.3. Compressive Response
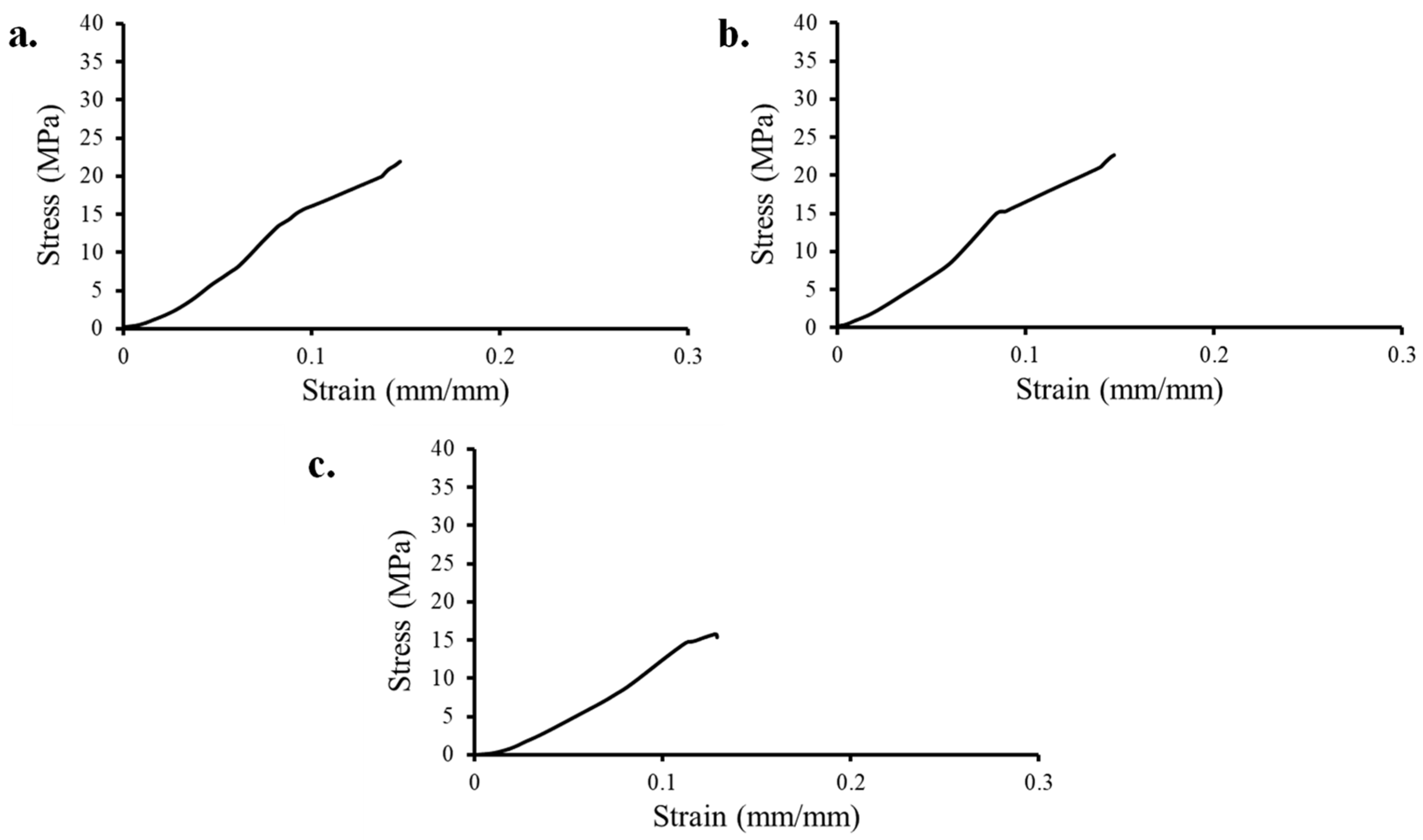
3.4. Tensile Response
3.5. Study Limitations
4. Conclusions
- Under compression, cubic lattices exhibit failure layer-by-layer, whereas BCC lattices display diagonal shear failure along the 45° direction of the struts. The elastic modulus range for cancellous bone (10 to 900 MPa) was matched by all pore sizes and unit cell geometries. Cubic 900 µm was the only lattice to satisfy the compressive yield strength (1.6 MPa) and compressive ultimate strength (6.9 MPa) of human cancellous bone (σy: 0.6 to 16.3 MPa and σmax: 0.1 to 16 MPa). BCC lattices displayed higher elastic modulus, compressive yield strength, and compressive ultimate strength than cubic lattices, due to higher relative density and superior unit cell geometry for compressive forces;
- The experimental tensile specimen exhibited numerous failures near the grips, indicating that an alternative design should be explored in the future for metallic lattice structures. The tensile elastic modulus (92.7–129.6 MPa) and tensile yield strength (3.4–7.1 MPa) of all tensile lattice specimens were within the range of human cancellous bone (E: 10 to 900 MPa and σy: 0.6 to 16.3 MPa);
- Structural characterization revealed that the relative density, weight, and strut diameter of selective laser melted parts were consistently larger than their corresponding CAD models for both cubic and BCC strut geometries due to selective laser melting resolution and unmelted powder adhesion.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yan, C.; Hao, L.; Hussein, A.; Young, P.; Raymont, D. Advanced Lightweight 316L Stainless Steel Cellular Lattice Structures Fabricated via Selective Laser Melting. Mater. Des. 2014, 55, 533–541. [Google Scholar] [CrossRef]
- Stoffelen, D.V.C.; Eraly, K.; Debeer, P. The Use of 3D Printing Technology in Reconstruction of a Severe Glenoid Defect: A Case Report with 2.5 Years of Follow-Up. J. Shoulder Elb. Surg. 2015, 24, e218–e222. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Qu, X.; Mao, Y.; Dai, K.; Zhu, Z. Custom Acetabular Cages Offer Stable Fixation and Improved Hip Scores for Revision THA With Severe Bone Defects. Clin. Orthop. Relat. Res. 2016, 474, 731–740. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.C.; Kumta, S.M.; Geel, N.V.; Demol, J. One-Step Reconstruction with a 3D-Printed, Biomechanically Evaluated Custom Implant after Complex Pelvic Tumor Resection. Comput. Aided Surg. Off. J. Int. Soc. Comput. Aided Surg. 2015, 20, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Dhiman, S.; Sidhu, S.; Singh, P.; Bahraminasab, M. Mechanobiological Assessment of Ti-6Al-4V Fabricated via Selective Laser Melting Technique: A Review. Rapid Prototyp. J. 2019, 25, 1266–1284. [Google Scholar] [CrossRef]
- Wang, X.; Xu, S.; Zhou, S.; Xu, W.; Leary, M.; Choong, P.; Qian, M.; Brandt, M.; Xie, Y.M. Topological Design and Additive Manufacturing of Porous Metals for Bone Scaffolds and Orthopaedic Implants: A Review. Biomaterials 2016, 83, 127–141. [Google Scholar] [CrossRef]
- Dallago, M.; Raghavendra, S.; Luchin, V.; Zappini, G.; Pasini, D.; Benedetti, M. The Role of Node Fillet, Unit-Cell Size and Strut Orientation on the Fatigue Strength of Ti-6Al-4V Lattice Materials Additively Manufactured via Laser Powder Bed Fusion. Int. J. Fatigue 2021, 142, 105946. [Google Scholar] [CrossRef]
- Soro, N.; Saintier, N.; Merzeau, J.; Veidt, M.; Dargusch, M.S. Quasi-Static and Fatigue Properties of Graded Ti–6Al–4V Lattices Produced by Laser Powder Bed Fusion (LPBF). Addit. Manuf. 2021, 37, 101653. [Google Scholar] [CrossRef]
- Bai, L.; Gong, C.; Chen, X.; Zheng, J.; Yang, J.; Li, K.; Sun, Y. Heterogeneous Compressive Responses of Addi-tively Manufactured Ti-6Al-4V Lattice Structures by Varying Geometric Parameters of Cells. Int. J. Mech. Sci. 2022, 214, 106922. [Google Scholar] [CrossRef]
- Tseng, S.-F.; Wang, I.-H.; Chang, C.-M.; Lee, C.-C.; Yeh, D.-Y.; Chen, T.-W.; Yeh, A.-C. Mechanical Character-istic Comparison of Additively Manufactured Ti–6Al–4V Lattice Structures in Biocompatible Bone Tissue Growth. Mater. Sci. Eng. A 2022, 857, 144045. [Google Scholar] [CrossRef]
- Daynes, S.; Lifton, J.; Lu, W.F.; Wei, J.; Feih, S. Fracture Toughness Characteristics of Additively Manufactured Ti–6Al–4V Lattices. Eur. J. Mech. A/Solids 2021, 86, 104170. [Google Scholar] [CrossRef]
- Jin, N.; Yan, Z.; Wang, Y.; Cheng, H.; Zhang, H. Effects of Heat Treatment on Microstructure and Mechanical Properties of Selective Laser Melted Ti-6Al-4V Lattice Materials. Int. J. Mech. Sci. 2021, 190, 106042. [Google Scholar] [CrossRef]
- Cutolo, A.; Engelen, B.; Desmet, W.; Van Hooreweder, B. Mechanical Properties of Diamond Lattice Ti–6Al–4V Structures Produced by Laser Powder Bed Fusion: On the Effect of the Load Direction. J. Mech. Behav. Biomed. Mater. 2020, 104, 103656. [Google Scholar] [CrossRef] [PubMed]
- Ridzwan, M.; Shuib, S.; Hassan, A.Y.; Ahmed Shokri, A.; Ibrahim, M. Problem of Stress Shielding and Improvement to the Hip Implant Designs: A Review. J. Med. Sci. 2007, 7, 460–467. [Google Scholar] [CrossRef]
- Oftadeh, R.; Perez-Viloria, M.; Villa-Camacho, J.C.; Vaziri, A.; Nazarian, A. Biomechanics and Mechanobiology of Trabecular Bone: A Review. J. Biomech. Eng. 2015, 137, 108021–1080215. [Google Scholar] [CrossRef] [PubMed]
- Kienapfel, H.; Sprey, C.; Wilke, A.; Griss, P. Implant Fixation by Bone Ingrowth. J. Arthroplast. 1999, 14, 355–368. [Google Scholar] [CrossRef] [PubMed]
- Biemond, J.E.; Aquarius, R.; Verdonschot, N.; Buma, P. Frictional and Bone Ingrowth Properties of Engineered Surface Topographies Produced by Electron Beam Technology. Arch. Orthop. Trauma Surg. 2011, 131, 711–718. [Google Scholar] [CrossRef] [PubMed]
- de Wild, M.; Schumacher, R.; Mayer, K.; Schkommodau, E.; Thoma, D.; Bredell, M.; Kruse Gujer, A.; Grätz, K.W.; Weber, F.E. Bone Regeneration by the Osteoconductivity of Porous Titanium Implants Manufactured by Selective Laser Melting: A Histological and Micro Computed Tomography Study in the Rabbit. Tissue Eng. Part. A 2013, 19, 2645–2654. [Google Scholar] [CrossRef] [PubMed]
- Sing, S.L.; An, J.; Yeong, W.Y.; Wiria, F.E. Laser and Electron-Beam Powder-Bed Additive Manufacturing of Metallic Implants: A Review on Processes, Materials and Designs. J. Orthop. Res. 2016, 34, 369–385. [Google Scholar] [CrossRef]
- Van Bael, S.; Chai, Y.C.; Truscello, S.; Moesen, M.; Kerckhofs, G.; Van Oosterwyck, H.; Kruth, J.-P.; Schrooten, J. The Effect of Pore Geometry on the In Vitro Biological Behavior of Human Periosteum-Derived Cells Seeded on Selective Laser-Melted Ti6Al4V Bone Scaffolds. Acta Biomater. 2012, 8, 2824–2834. [Google Scholar] [CrossRef]
- Markhoff, J.; Wieding, J.; Weissmann, V.; Pasold, J.; Jonitz-Heincke, A.; Bader, R. Influence of Different Three-Dimensional Open Porous Titanium Scaffold Designs on Human Osteoblasts Behavior in Static and Dynamic Cell Investigations. Materials 2015, 8, 5490–5507. [Google Scholar] [CrossRef] [PubMed]
- Valstar, E.R.; Nelissen, R.G.H.H.; Reiber, J.H.C.; Rozing, P.M. The Use of Roentgen Stereophotogrammetry to Study Micromotion of Orthopaedic Implants. ISPRS J. Photogramm. Remote Sens. 2002, 56, 376–389. [Google Scholar] [CrossRef]
- Obaton, A.-F.; Fain, J.; Djemaï, M.; Meinel, D.; Léonard, F.; Mahé, E.; Lécuelle, B.; Fouchet, J.-J.; Bruno, G. In Vivo XCT Bone Characterization of Lattice Structured Implants Fabricated by Additive Manufacturing. Heliyon 2017, 3, e00374. [Google Scholar] [CrossRef] [PubMed]
- Ran, Q.; Yang, W.; Hu, Y.; Shen, X.; Yu, Y.; Xiang, Y.; Cai, K. Osteogenesis of 3D Printed Porous Ti6Al4V Implants with Different Pore Sizes. J. Mech. Behav. Biomed. Mater. 2018, 84, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, N.; Fujibayashi, S.; Takemoto, M.; Sasaki, K.; Otsuki, B.; Nakamura, T.; Matsushita, T.; Kokubo, T.; Matsuda, S. Effect of Pore Size on Bone Ingrowth into Porous Titanium Implants Fabricated by Additive Manufacturing: An In Vivo Experiment. Mater. Sci. Eng. C 2016, 59, 690–701. [Google Scholar] [CrossRef] [PubMed]
- Van der Stok, J.; Van der Jagt, O.P.; Amin Yavari, S.; De Haas, M.F.P.; Waarsing, J.H.; Jahr, H.; Van Lieshout, E.M.M.; Patka, P.; Verhaar, J.A.N.; Zadpoor, A.A.; et al. Selective Laser Melting-Produced Porous Titanium Scaffolds Regenerate Bone in Critical Size Cortical Bone Defects. J. Orthop. Res. 2013, 31, 792–799. [Google Scholar] [CrossRef] [PubMed]
- Melchels, F.P.W.; Barradas, A.M.C.; van Blitterswijk, C.A.; de Boer, J.; Feijen, J.; Grijpma, D.W. Effects of the Architecture of Tissue Engineering Scaffolds on Cell Seeding and Culturing. Acta Biomater. 2010, 6, 4208–4217. [Google Scholar] [CrossRef] [PubMed]
- Wally, Z.J.; Haque, A.M.; Feteira, A.; Claeyssens, F.; Goodall, R.; Reilly, G.C. Selective Laser Melting Processed Ti6Al4V Lattices with Graded Porosities for Dental Applications. J. Mech. Behav. Biomed. Mater. 2019, 90, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Su, K.; Su, L.; Liang, P.; Ji, P.; Wang, C. The Effect of 3D-Printed Ti6Al4V Scaffolds with Various Macropore Structures on Osteointegration and Osteogenesis: A Biomechanical Evaluation. J. Mech. Behav. Biomed. Mater. 2018, 88, 488–496. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.Z.; Leary, M.; Tang, H.P.; Song, T.; Qian, M. Selective Electron Beam Manufactured Ti-6Al-4V Lattice Structures for Orthopedic Implant Applications: Current Status and Outstanding Challenges. Curr. Opin. Solid State Mater. Sci. 2018, 22, 75–99. [Google Scholar] [CrossRef]
- Yánez, A.; Cuadrado, A.; Martel, O.; Afonso, H.; Monopoli, D. Gyroid Porous Titanium Structures: A Versatile Solution to Be Used as Scaffolds in Bone Defect Reconstruction. Mater. Des. 2018, 140, 21–29. [Google Scholar] [CrossRef]
- Parthasarathy, J.; Starly, B.; Raman, S.; Christensen, A. Mechanical Evaluation of Porous Titanium (Ti6Al4V) Structures with Electron Beam Melting (EBM). J. Mech. Behav. Biomed. Mater. 2010, 3, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Choy, S.Y.; Sun, C.-N.; Leong, K.F.; Wei, J. Compressive Properties of Ti-6Al-4V Lattice Structures Fabricated by Selective Laser Melting: Design, Orientation and Density. Addit. Manuf. 2017, 16, 213–224. [Google Scholar] [CrossRef]
- Mullen, L.; Stamp, R.C.; Brooks, W.K.; Jones, E.; Sutcliffe, C.J. Selective Laser Melting: A Regular Unit Cell Approach for the Manufacture of Porous, Titanium, Bone in-Growth Constructs, Suitable for Orthopedic Applications. J. Biomed. Mater. Res. B Appl. Biomater. 2009, 89, 325–334. [Google Scholar] [CrossRef]
- He, M.; Li, Y.; Yin, J.; Sun, Q.; Xiong, W.; Li, S.; Yang, L.; Hao, L. Compressive Performance and Fracture Mechanism of Bio-Inspired Heterogeneous Glass Sponge Lattice Structures Manufactured by Selective Laser Melting. Mater. Des. 2022, 214, 110396. [Google Scholar] [CrossRef]
- Maskery, I.; Aboulkhair, N.T.; Aremu, A.O.; Tuck, C.J.; Ashcroft, I.A.; Wildman, R.D.; Hague, R.J.M. A Mechanical Property Evaluation of Graded Density Al-Si10-Mg Lattice Structures Manufactured by Selective Laser Melting. Mater. Sci. Eng. A 2016, 670, 264–274. [Google Scholar] [CrossRef]
- Wang, X.; Wang, C.; Zhou, X.; Wang, D.; Zhang, M.; Gao, Y.; Wang, L.; Zhang, P. Evaluating Lattice Mechanical Properties for Lightweight Heat-Resistant Load-Bearing Structure Design. Materials 2020, 13, 4786. [Google Scholar] [CrossRef] [PubMed]
- Bankoff, A.D.P. Biomechanical Characteristics of the Bone. In Human Musculoskeletal Biomechanics; Goswami, T., Ed.; InTech: London, UK, 2012; pp. 63–86. [Google Scholar]
- Papazoglou, D.P. Additively Manufactured Lattice Structures for Orthopedic Implants—Analysis of Mechanical Properties. In Proceedings of the ASTM ICAM, Virtual, 16–20 November 2020. [Google Scholar]
- Mazur, M.; Leary, M.; McMillan, M.; Sun, S.; Shidid, D.; Brandt, M. 5-Mechanical Properties of Ti6Al4V and AlSi12Mg Lattice Structures Manufactured by Selective Laser Melting (SLM). In Woodhead Publishing Series in Electronic and Optical Materials; Brandt, M.B.T.-L.A.M., Ed.; Woodhead Publishing: Sawston, UK, 2017; pp. 119–161. ISBN 978-0-08-100433-3. [Google Scholar]
- Papazoglou, D.P. Additively Manufactured Ti-6Al-4V Biomimetic Lattice Structures for Patient-Specific Orthopedic Implants: The Effect of Unit Cell Geometry, Pore Size, and Pulsed Electromagnetic Field Stimulation on the Osseointegration of MG-63 Cells in Vitro, Mechanical Properties, and Surface Characterization. Ph.D. Thesis, University of Dayton, Dayton, OH, USA, 2023. [Google Scholar]
- ASTM Standard F3456-22; Standard Guide for Powder Reuse Schema in Powder Bed Fusion Processes for Medical Applications for Additive Manufacturing Feedstock Materials. ASTM International: West Conshohocken, PA, USA, 2022.
- Porter, D.A.; Di Prima, M.A.; Badhe, Y.; Parikh, A.R. Nylon Lattice Design Parameter Effects on Additively Manufactured Structural Performance. J. Mech. Behav. Biomed. Mater. 2022, 125, 104869. [Google Scholar] [CrossRef]
- Ashby, M.F. The Properties of Foams and Lattices. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2005, 364, 15–30. [Google Scholar] [CrossRef]
- ASTM Standard E9-09; Standard Test Methods of Compression Testing of Metallic Materials at Room Temperature. ASTM International: West Conshohocken, PA, USA, 2018.
- ASTM Standard E8/E8M-22; Standard Test Methods for Tension Testing of Metallic Materials. ASTM International: West Conshohocken, PA, USA, 2022.
- Maconachie, T.; Leary, M.; Lozanovski, B.; Zhang, X.; Qian, M.; Faruque, O.; Brandt, M. SLM Lattice Structures: Properties, Performance, Applications and Challenges. Mater. Des. 2019, 183, 108137. [Google Scholar] [CrossRef]
- Yan, X.; Li, Q.; Yin, S.; Chen, Z.; Jenkins, R.; Chen, C.; Wang, J.; Ma, W.; Bolot, R.; Lupoi, R.; et al. Mechanical and in Vitro Study of an Isotropic Ti6Al4V Lattice Structure Fabricated Using Selective Laser Melting. J. Alloys Compd. 2019, 782, 209–223. [Google Scholar] [CrossRef]
- Sombatmai, A.; Uthaisangsuk, V.; Wongwises, S.; Promoppatum, P. Multiscale Investigation of the Influence of Geometrical Imperfections, Porosity, and Size-Dependent Features on Mechanical Behavior of Additively Manufactured Ti-6Al-4V Lattice Struts. Mater. Des. 2021, 209, 109985. [Google Scholar] [CrossRef]
- Vrána, R.; Koutecký, T.; Červinek, O.; Zikmund, T.; Pantělejev, L.; Kaiser, J.; Koutný, D. Deviations of the SLM Produced Lattice Structures and Their Influence on Mechanical Properties. Materials 2022, 15, 3144. [Google Scholar] [CrossRef] [PubMed]
- Martinez, M.A.F.; Balderrama, Í.D.F.; Karam, P.S.B.H.; de Oliveira, R.C.; de Oliveira, F.A.; Grandini, C.R.; Vicente, F.B.; Stavropoulos, A.; Zangrando, M.S.R.; Sant’Ana, A.C.P. Surface Roughness of Titanium Disks Influences the Adhesion, Proliferation and Differentiation of Osteogenic Properties Derived from Human. Int. J. Implant. Dent. 2020, 6, 46. [Google Scholar] [CrossRef] [PubMed]
- An, Y.; Draughn, R. Mechanical Testing of Bone and the Bone–Implant Interface; CRC Press: New York, NY, USA, 2000; ISBN 9780429125799. [Google Scholar]
- Dunham, C.E.; Takaki, S.E.; Johnson, J.A.; Dunning, C.E. Mechanical Properties of Cancellous Bone of the Distal Humerus. Clin. Biomech. 2005, 20, 834–838. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, C.J.; Beaupré, G.S.; Keller, T.S.; Carter, D.R. The Influence of Bone Volume Fraction and Ash Fraction on Bone Strength and Modulus. Bone 2001, 29, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Keaveny, T.M.; Morgan, E.F.; Niebur, G.L.; Yeh, O.C. Biomechanics of Trabecular Bone. Annu. Rev. Biomed. Eng. 2001, 3, 307–333. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Wang, L.; Zhang, H.; Maldar, A.; Zhu, G.; Chen, W.; Park, J.-S.; Wang, J.; Zeng, X. Effect of Heat Treatment on the Tensile Behavior of Selective Laser Melted Ti-6Al-4V by in Situ X-Ray Characterization. Acta Mater. 2020, 189, 93–104. [Google Scholar] [CrossRef]
- Alsalla, H.; Hao, L.; Smith, C. Fracture Toughness and Tensile Strength of 316L Stainless Steel Cellular Lattice Structures Manufactured Using the Selective Laser Melting Technique. Mater. Sci. Eng. A 2016, 669, 1–6. [Google Scholar] [CrossRef]
- Røhl, L.; Larsen, E.; Linde, F.; Odgaard, A.; Jørgensen, J. Tensile and Compressive Properties of Cancellous Bone. J. Biomech. 1991, 24, 1143–1149. [Google Scholar] [CrossRef]
- Kaya, G.; Yıldız, F.; Korkmaz, İ.H.; Kaymaz, İ.; Yetim, A.F.; Ergüder, T.O.; Şen, Ç. Effects of Process Parame-ters on Selective Laser Melting of Ti6Al4V-ELI Alloy and Parameter Optimization via Response Surface Method. Mater. Sci. Eng. A 2023, 885, 145581. [Google Scholar] [CrossRef]












| Lattice Type | Average Pore Size (µm) | Pore Size Deviation (%) | Average Strut Diameter (µm) | Strut Diameter Deviation (%) |
|---|---|---|---|---|
| Cubic P400 | 374.6 (±32.1) | −6.5 | 310.1 (±34.4) | 43.1 |
| Cubic P500 | 439.2 (±63.0) | −12.9 | 333.8 (±43.7) | 49.9 |
| Cubic P600 | 559.1 (±50.9) | −7.1 | 268.2 (±43.8) | 29 |
| Cubic P900 | 849.5 (±37.9) | −5.7 | 312.6 (±67.6) | 43.7 |
| BCC P400 | 219.2 (±49.1) | −58.3 | 428.5 (±37.0) | 72.6 |
| BCC P500 | 411.5 (±25.3) | −19.5 | 344.8 (±41.3) | 52.9 |
| BCC P600 | 533.6 (±37.1) | −11.8 | 375.5 (±30.1) | 60.8 |
| BCC P900 | 675.2 (±109.9) | −28.5 | 629.1 (±35.0) | 44.5 |
| Lattice Type | CAD Weight (g) | Produced Weight (g) | CAD Relative Density (%) | Produced Relative Density (%) | CAD Porosity (%) | Produced Porosity (%) |
|---|---|---|---|---|---|---|
| Cubic P400 | 0.9 | 2.3 | 21 | 52 | 79.4 | 49.2 |
| Cubic P500 | 0.7 | 1.6 | 17 | 40 | 83.8 | 63.3 |
| Cubic P600 | 0.5 | 1.2 | 12 | 31 | 87.9 | 27.9 |
| Cubic P900 | 0.2 | 0.6 | 7 | 12 | 93.1 | 92.1 |
| BCC 400 | 1.0 | 2.8 | 25 | 65 | 75.8 | 14.9 |
| BCC 500 | 0.8 | 1.7 | 19 | 44 | 81.9 | 61.6 |
| BCC 600 | 0.6 | 1.4 | 15 | 37 | 85.3 | 64.7 |
| BCC 900 | 1.0 | 1.7 | 25 | 44 | 75.2 | 77.8 |
| Lattice Type | Elastic Moduli—E (MPa) | Yield Strength—σy (MPa) | Compressive Strength—σmax (MPa) |
|---|---|---|---|
| Cubic P400 | 843.3 (±32.9) | 63.9 ± 11.2 | 221.2 ± 11.3 |
| Cubic P500 | 685.5 (±77.9) | 52.0 ± 21.2 | 111.6 ± 9.1 |
| Cubic P600 | 632.1 (±80.0) | 38.5 ± 7.1 | 78.5 ± 3.4 |
| Cubic P900 | 185.2 (±108.3) | 1.6 ± 0.5 | 6.9 ± 0.3 |
| BCC P400 | 996.1 (±244.8) | 73.4 ± 13.1 | - |
| BCC P500 | 712.8 (±105.0) | 58.4 ± 4.9 | 118.9 ± 2.7 |
| BCC P600 | 710.8 (±155.1) | 46.6 ± 1.2 | 103.9 ± 3.3 |
| BCC P900 | 753.2 (±86.9) | 34.1 ± 14.6 | 135.7 ± 1.4 |
| Lattice Type | Number of Samples | Elastic Moduli—E (MPa) | Yield Strength—σy (MPa) | Tensile Strength—σmax (MPa) |
|---|---|---|---|---|
| Cubic P400 | 0 | - | - | - |
| Cubic P500 | 2 | 108.3 (±8.1) | 7.1 (±2.6) | 35.5 (±2.3) |
| Cubic P600 | 3 | 129.6 (±3.1) | 5.3 (±0.5) | 29.2 (±2.6) |
| Cubic P900 | 3 | 110.9 (±8.6) | 4.5 (±0.3) | 14.0 (±1.1) |
| BCC P400 | 0 | - | - | - |
| BCC P500 | 2 | 109.2 (±0.8) | 4.7 (±0.5) | 21.9 (±2.3) |
| BCC P600 | 2 | 111.7 (±14.3) | 3.4 (±0.2) | 18.1 (±7.2) |
| BCC P900 | 2 | 92.7 (±4.2) | 4.2 (±0.8) | 13.3 (±6.7) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Papazoglou, D.P.; Neidhard-Doll, A.T.; Pinnell, M.F.; Erdahl, D.S.; Osborn, T.H. Compression and Tensile Testing of L-PBF Ti-6Al-4V Lattice Structures with Biomimetic Porosities and Strut Geometries for Orthopedic Implants. Metals 2024, 14, 232. https://doi.org/10.3390/met14020232
Papazoglou DP, Neidhard-Doll AT, Pinnell MF, Erdahl DS, Osborn TH. Compression and Tensile Testing of L-PBF Ti-6Al-4V Lattice Structures with Biomimetic Porosities and Strut Geometries for Orthopedic Implants. Metals. 2024; 14(2):232. https://doi.org/10.3390/met14020232
Chicago/Turabian StylePapazoglou, Dimitri P., Amy T. Neidhard-Doll, Margaret F. Pinnell, Dathan S. Erdahl, and Timothy H. Osborn. 2024. "Compression and Tensile Testing of L-PBF Ti-6Al-4V Lattice Structures with Biomimetic Porosities and Strut Geometries for Orthopedic Implants" Metals 14, no. 2: 232. https://doi.org/10.3390/met14020232
APA StylePapazoglou, D. P., Neidhard-Doll, A. T., Pinnell, M. F., Erdahl, D. S., & Osborn, T. H. (2024). Compression and Tensile Testing of L-PBF Ti-6Al-4V Lattice Structures with Biomimetic Porosities and Strut Geometries for Orthopedic Implants. Metals, 14(2), 232. https://doi.org/10.3390/met14020232






