Research Status of Electrolytic Preparation of Rare Earth Metals and Alloys in Fluoride Molten Salt System: A Mini Review of China
Abstract
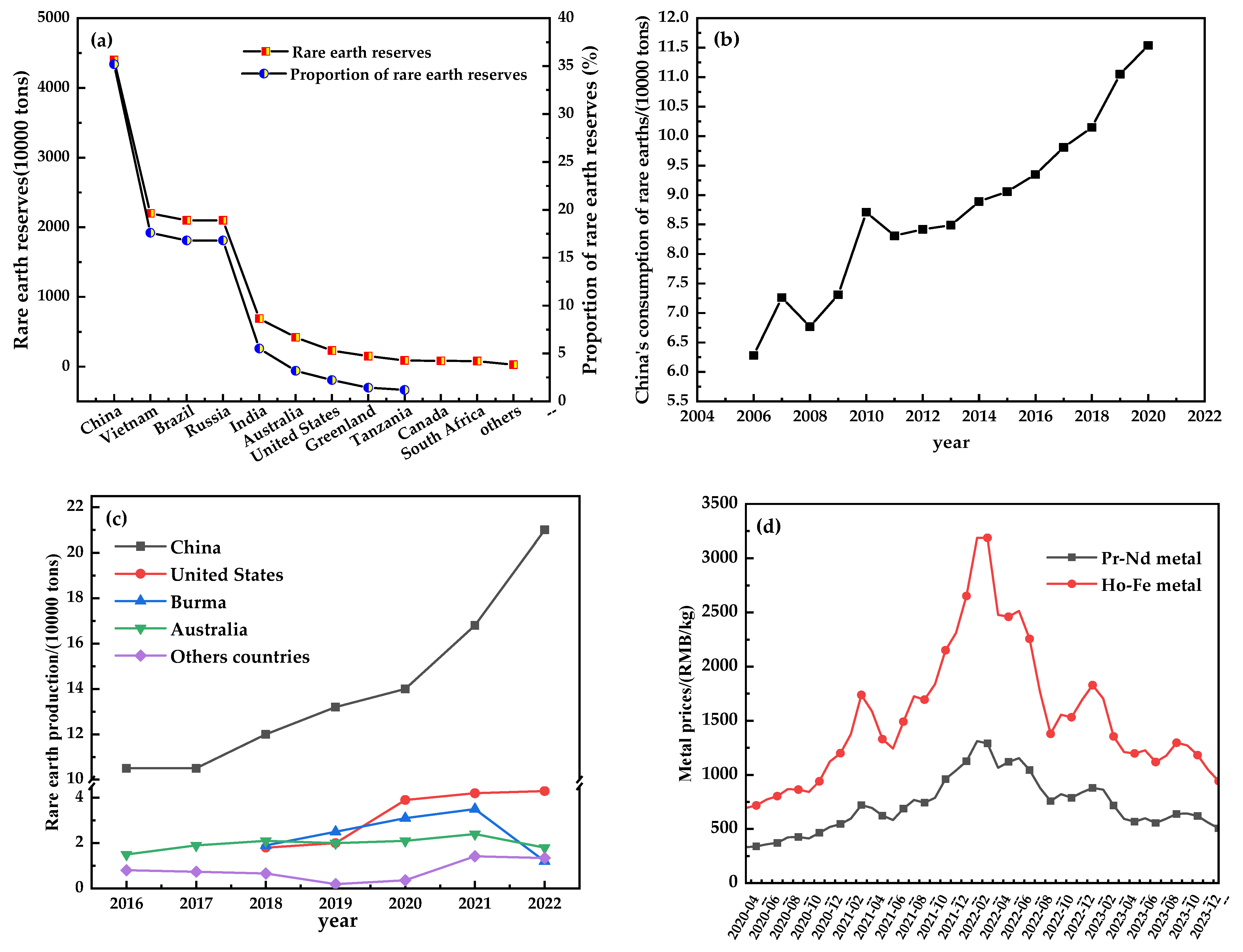
:1. Introduction
2. Comparison of Pyrometallurgical Methods for Rare Earth Metals
REC2(s) = RE(s) + 2C(s)
| Metal | Melting Point/K | Boiling Point/K | The Temperature at 133.3 Pa Vapor Pressure/K | Evaporation Rate at 133.3 Pa Vapor Pressure/[g/(cm2·h)] | REF3 Melting Point/K | RE2O3 Melting Point/K | Mineral Characteristics Group | Main Preparation Method |
|---|---|---|---|---|---|---|---|---|
| La | 1193 | 3743 | 2490 | 53 | 1763 | 2490 | Light rare earth | Molten salt electrolysis |
| Ce | 1071 | 3743 | 2420 | 53 | 1710 | 2415 | ||
| Pr | 1204 | 3403 | 2241 | 56 | 1668 | 2400 | ||
| Nd | 1283 | 3303 | 2032 | 60 | 1647 | 2484 | ||
| Sm | 1345 | 2173 | 1237 | 83 | 1579 | 2603 | Lanthanum or carbonthermal reduction | |
| Eu | 1095 | 1713 | 1110 | 90 | 1549 | 2668 | ||
| Gd | 1584 | 3273 | 2295 | 59 | 1504 | 2663 | Heavy rare earth | Calcium thermal reduction |
| Tb | 1633 | 3073 | 2212 | 60 | 1445 | 2663 | ||
| Dy | 1682 | 2873 | 1712 | 71 | 1427 | 2664 | ||
| Ho | 1743 | 2873 | 1799 | 69 | 1416 | 2673 | ||
| Er | 1795 | 3173 | 1879 | 68 | 1413 | - | ||
| Tm | 1818 | 2003 | 1368 | 83 | 1431 | 2684 | Lanthanum or carbonthermal reduction | |
| Yb | 1097 | 1703 | 924 | 108 | 1430 | - | ||
| Lu | 1929 | 3603 | 2371 | 61 | 1455 | - | Calcium thermal reduction | |
| Y | 1796 | 3203 | 1355 | 43 | 1425 | - |
| Method | Main Raw Materials | Product Components | Production Efficiency | Production Process |
|---|---|---|---|---|
| Calcium thermal reduction method [21,22] | REF3, Ca | The purity of rare earth metals is between 95.5% and 99.95%. | The yield of rare earth metals is about 95–98%. | Low investment, short process, and a large amount of slag generated from calcium thermal reduction. |
| Lithium thermal reduction [23] | RECl3, Li | The purity of rare earth metals can reach more than 99.9%. | The yield of rare earth metals reaches more than 95%. | The quality of the prepared rare earth metals is better than that of calcium thermal reduction. Rare earth chlorides are very hygroscopic compounds. |
| Carbonaceous reduction [24,25] | RE2O3, C | Not indicated | The yield of metals reaches 59.37% | High processing cost and discontinuous production. |
| Lanthanum thermal reduction [26,27,28] | RE2O3, La | The purity of rare earth metals reaches over 99.9%. | The recovery of rare earth metals reaches over 90%. | High temperature and pressure conditions require high-quality materials and performance of the equipment. |
| Chloride system molten salt electrolysis method [29,30] | RECl3-MCl(M = Li, K) | Electrolytic rare earth metals with a purity of 95–98% | The yield of rare earth metals is about 85–90%. Single rare earth current efficiency is around 70%. | Generates toxic chlorine gas, low metal recovery. |
| Fluoride system molten salt electrolysis method [30] | REF3-RE2O3-LiF | Electrolytic rare earth metals with a purity of 95–98% | The yield of rare earth metals is about 90–95%. Current efficiency is about 70–80%. | The current efficiency and rare earth metal yield are high; the electrolysis temperature is high, and the anode produces greenhouse gases such as CO2, CF4, and C2F6. |
3. Research Status of Fluoride Molten Salt System
3.1. Preparation of Individual Rare Earth Metal
3.1.1. Lanthanum Metal
3.1.2. Cerium Metal
3.1.3. Praseodymium Metal
3.1.4. Neodymium Metal
3.2. Preparation of Rare Earth Alloys
3.2.1. RE–Mg Alloys
3.2.2. RE–Al Alloys
3.2.3. RE–Ni Alloys
| RE | Molten Salt | Electrode | Electrolysis Time | Temperature | Applied Current Density/Current/ Potential (vs. Pt) | RE Alloys |
|---|---|---|---|---|---|---|
| Ce [40] | 52.66 wt.% LiF-47.34 wt.% CaF2-CeF3 | Ni | 60 min | 1093 K | −1.5 V | CeNi5 |
| Eu [76] | 55.56 wt.% LiF-44.44 wt.% CaF2-EuF3-AlF3 | W | 20 min | 1113 K | −1.1 V | EuAl4 |
| Nd [76] | 55.56 wt.% LiF-44.44 wt.% CaF2-NdF3-AlF3 | W | 20 min | 1133 K | −1.33 V, −1.49 V, −1.69 V, −1.88 V | Nd3Al11, NdAl3, NdAl2, Nd3Al |
| Ce [77] | 55.56 wt.% LiF-44.44 wt.% CaF2-CeF3-AlF3 | W | 20 min | 1113 K | −1.25 V, −1.43 V, −1.79 V | Ce3Al11, CeAl3, (Ce3Al, CeAl) |
| Sm [77] | 55.56 wt.% LiF-44.44 wt.% CaF2-NdF3-AlF3 | W | 20 min | 1113 K | −1.45 V | SmAl3 |
| Sm [78] | LiF-CaF2-Sm2O3-AlF3 | W | 180 min | 1223 K | −731.4 mA/cm2 | SmAl2, SmAl3 |
| Sm [96] | 52.66 wt.% LiF-47.34 wt.% CaF2 | Ni | 60 min | 1120 K | −200 mA/cm2 | Sm3Ni, SmNi2 |
| Nd [97] | 52.66 wt.% LiF-47.34 wt.% CaF2-NdF3 | Ni | 120 min | 1113 K | 35 mA/cm2, 0.65 V | NdNi2, NdNi3 |
| Gd [97] | 52.66 wt.% LiF-47.34 wt.% CaF2-GdF3 | Ni | 120 min | 1113 K | 35 mA/cm2, 0.65 V | GdNi2, GdNi |
| Sm [97] | 52.66 wt.% LiF-47.34 wt.% CaF2-SmF3 | Ni | 120 min | 1113 K | 35 mA/cm2, 0.7 V | SmNi2, SmNi3 |
| Dy [98] | 55.56 wt.% LiF-44.44 wt.% CaF2-DyF3 | Ni | 60 min | 1113 K | −15 mA/cm2 | Dy2Ni17, DyNi4 |
| Dy [98] | 55.56 wt.% LiF-44.44 wt.% CaF2-DyF3 | Ni | 60 min | 1113 K | −15 mA/cm2 | Dy2Ni17, DyNi4 |
| Nd [99] | 57.84 wt.% LiF-42.16 wt.% CaF2-NdF3 | Ni | 75 min | 1123 K | 0.15 V,0.35 V (vs. Li+/Li) | NdNi2, NdNi3 |
| Nd [99] | 57.84 wt.% LiF-42.16 wt.% CaF2-NdF3 | Ni | 50 min | 1123 K | 0.60 V (vs. Li+/Li) | NdNi5 |
| Pr [102] | 57.84 wt.% LiF-42.16 wt.% CaF2-PrF3 | Ni | 90 min /180 min | 1123 K | 0.2 V, 0.36 V, 0.6 V(vs. Li+/Li) | PrNi2, PrNi3, PrNi5 |
| Dy [113] | 57.84 wt.% LiF-42.16 wt.% CaF2-DyF3 | Fe | 30 min | 1123 K | 0.1 V (vs. Li+/Li) | DyFe2 |
3.2.4. Nd Alloys
3.3. Discussion and Summary
3.3.1. Electrolysis Parameters
3.3.2. Treatment of Anode Gases
3.3.3. Design of Rare Earth Cell
4. Prospects
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Gao, L.Y.; Deng, Y.H.; Huang, R.; Yang, K.Z.; Huang, M.S. Research and Application Status and Development Trend of Rare Earth Functional Materials. Rare Met. Cem. Carbides 2023, 51, 59–64. [Google Scholar]
- Zhang, W.J.; Tong, X.; Xie, X. A Review on Research of Rare Earths Separation and Purification Technology. J. Chin. Soc. Rare Earths 2022, 40, 24–37. [Google Scholar]
- Hu, J.L.; Xue, D.F. Research Progress on the Characteristics of Rare Earth lons and Rare Earth Functional Materials. Chin. J. Appl. Chem. 2020, 37, 245–255. [Google Scholar]
- U.S. Geological Survey. Mineral Commodity Summaries 2023; U.S. Geological Survey: Reston, VA, USA, 2023; p. 210. [CrossRef]
- U.S. Geological Survey. Mineral Commodity Summaries 2022; U.S. Geological Survey: Reston, VA, USA, 2022; p. 202. [CrossRef]
- U.S. Geological Survey. Mineral Commodity Summaries 2021; U.S. Geological Survey: Reston, VA, USA, 2021; p. 200. [CrossRef]
- U.S. Geological Survey. Mineral Commodity Summaries 2020; U.S. Geological Survey: Reston, VA, USA, 2020; p. 200. [CrossRef]
- U.S. Geological Survey. Mineral Commodity Summaries 2019; U.S. Geological Survey: Reston, VA, USA, 2019; p. 200. [CrossRef]
- U.S. Geological Survey. Mineral Commodity Summaries 2018; U.S. Geological Survey: Reston, VA, USA, 2018; p. 200. [CrossRef]
- Shenzhen Foresight Industry Research Institute Co., Ltd. Sina Finance. Analysis of the Supply and Demand Situation in China’s Rare Earth Industry Market in 2022. 22 December 2021. Available online: https://finance.sina.com.cn/roll/2021-12-22/doc-ikyamrmz0499220.shtml (accessed on 6 February 2024).
- Zhang, S.J.; Zhang, L.W.; Zhang, Y.W.; Shang, L.; Li, J.B. Summarize on rare earth mineral resources and their distribution at home and abroad. Inorg. Chem. Ind. 2020, 52, 9–16. [Google Scholar]
- Association of China Rare Earth Industry. Dilute Price Action. 31 October 2023. Available online: https://www.acreiorg.cn/article/b65ba59e-4d03-4708-85a1-692cled11ea9 (accessed on 25 December 2023).
- Liu, S.L.; Fan, H.R.; Liu, X. Global rare earth elements projects: New developments and supply chains. Ore Geol. Rev. 2023, 157, 105428. [Google Scholar] [CrossRef]
- Cheng, S.; Li, W.; Han, Y. Recent process developments in beneficiation and metallurgy of rare earths: A review. J. Rare Earths 2023, in press. [CrossRef]
- The State Council Information Office of the People’s Republic of China. China’s Rare Earths: Status and Policies; People’s Publishing House: Beijing, China, 2012.
- Qiu, L.H. China Rare Earth Industry Safety Assessment and Countermeasures. Master’s Thesis, Jiangxi University of Science and Technology, Ganzhou, China, 2020. [Google Scholar]
- Liao, C.F. Rare Earth Metallurgy; Metallurgical Industry Press: Beijing, China, 2019. [Google Scholar]
- Li, M.; Liu, Z.G.; Zhang, X.W.; Chang, H.W. Modern Metallurgy of Rare Earth; Science Press: Beijing, China, 2016. [Google Scholar]
- Chen, Y.F.; Zhu, Z.Q.; Xu, Y.Y.; Wu, S.Y.; Lai, H.S. Study on the Preparation of Aluminum-Yttrium Master Alloy by Recovering Yttrium Element from Yttrium Smelting Slag. J/OL. Chin. Soc. Rare Earths 2024, 1–15. [Google Scholar]
- Gupta, C.K.; Krishnamurthy, N. Extractive Metallurgy of Rare Earths; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Liu, S.B.; Han, X.L.; Zhang, Y.J.; Liao, J.W.; Chen, X.K. Preparation of Gadolinium by Calcium Thermal Reduction. Fujian Metall. 2023, 52, 23–25. [Google Scholar]
- Cheng, W.; Huang, M.S.; Wang, Z.J.; Yang, L.H. Preparation of High-purity Lanthanum by Calciothermic Reduction. Min. Metall. Eng. 2013, 33, 104–106+109. [Google Scholar]
- Cheng, W.; Li, Z.A.; Chen, D.H.; Pang, S.M.; Wang, Z.Q.; Wang, X.S. Preparation of High Purity Lanthanum by Combined Method of Lithium. Thermal Reduction and Vacuum Distillation. Chin. J. Rare Met. 2011, 35, 781–785. [Google Scholar]
- Chen, H.L.; Xie, K.Q.; Zhang, Y.; Liang, K.; Yang, K.; Gao, L.; Fu, D.Z. Experimental Study on Vacuum Carbothermal Reduction of Ga2O3. Chin. J. Vac. Sci. Technol. 2023, 43, 60–65. [Google Scholar]
- Liu, G.H. Rare Earth Materials; Chemical Industry Press: Beijing, China, 2007. [Google Scholar]
- Yang, H.; Kang, J.H.; Huang, W.L. The Vacuam Distllation Experiment of Metal Sm. J. Gannan Norm. Univ. 2001, 6, 56–57. [Google Scholar]
- Zhang, X.H.; Zhao, E.X.; Miao, X.C.; Liu, Y.B.; Chen, G.H.; Dong, Y.Y. Preparation of high purity ytterbium metal by once reduction distillation. Met. Funct. Mater. 2020, 27, 28–33. [Google Scholar]
- Liu, Y.J.; Yan, S.H. The development of rare earth pyrometallurgy technology in China. Rare Earth Inf. 2003, 4, 2–8. [Google Scholar]
- Liu, Y.B.; Chen, G.H.; Yu, B.; Huang, H.T.; Zhang, Q.J.; Zhang, W.C. Research Progress of Preparation of Rare Earth Metals by Molten Salt Electrolysis. Rare Earth 2021, 42, 133–143. [Google Scholar]
- Pang, S.M.; Yan, S.A.; Li, Z.A.; Chen, D.H.; Xu, L.H.; Zhao, B. Development on Molten Salt Electrolytic Methods and Technology for Preparing Rare Earth Metals and Alloys in China. Rare Met. 2011, 35, 440–450. [Google Scholar]
- Han, W.; Li, M.; Zhang, M.L. Progress in preparation of rare earth metals and alloys by electrodeposition in molten salts. Rare Met. 2016, 35, 811–825. [Google Scholar] [CrossRef]
- Zhao, Z.L.; Zhang, Z.H.; Jiao, S.Z.; Liu, W.H. Lanthanum, cerium and praseodymium from fluoride systems by molten salt electrolysis process. Rare Earth 1986, 6, 44–49. [Google Scholar]
- Ministry of Ecology and Environment of the People’s Republic of China. Emission Standards of Pollutants from Rare Earths Industry. 1 October 2011. Available online: https://www.mee.gov.cn/ywgz/fgbz/bz/bzwb/shjbh/swrwpfbz/201102/t20110210_200521.shtml (accessed on 6 February 2024).
- Zhang, C.; Liang, X.Y.; Qin, F.; Zhang, Y.P.; Wang, B.G.; Fan, Z.Z. Evaluation and Assessment of the Implementation of the “Emission Standards of Pollutants from Rare Earths Industry”. In Proceedings of the Chinese Rare Earth Society, Rare Earth Chemistry and Hydrometallurgy Professional Committee, Chinese Rare Earth Society, Pyrometallurgy Professional Committee, Strategic Alliance for Innovation in Advanced Rare Earth Materials Industry, Ganzhou, China, 24 September 2014. [Google Scholar]
- Huang, K.; Guo, M.; Yan, F.; Gao, Y.F. Progress in the application of rare earth element lanthanum and its toxic effects. Rare Earth Inf. 2023, 3, 29–31. [Google Scholar]
- Guo, M.L.; Chen, Q.; Dai, W.H.; Ma, Y.; Xu, M.R.; Lu, B.L.; Zhu, Y.L.; Wang, L.H. Effects of Lanthanum Oxide Nanoparticles on Skotomorphogenesis of Plants. J. Chin. Soc. Rare Earths 2023, 41, 986–996. [Google Scholar]
- Wang, J.; Wu, B.L.; Wang, H.Y.; Zhu, Y.Y. Comparative study of inhibition to Smutans’ glucosyltransferase given bytrace element agents containing fluoride. Stomatology 2007, 27, 407–408. [Google Scholar]
- Massot, L.; Gibilaro, M.; Nicaise, J. Electrochemical behaviour of Lanthanum fluoride and Praseodymium fluoride on inert and reactive electrodes in molten LiF-CaF2. J. Fluor. Chem. 2021, 246, 109797. [Google Scholar] [CrossRef]
- Chesser, R.; Guo, S.; Zhang, J. Electrochemical behavior of dysprosium and lanthanum in molten LiF-NaF-KF (Flinak) salt. Ann. Nucl. Energy 2018, 120, 246–252. [Google Scholar] [CrossRef]
- Liang, X.; Chen, S.M.; Hong, K.; Chen, D.Y.; Li, Z.Q.; Lai, Y.B.; Xu, J.B. Solubility of La2O3 in LaF3-LiF Fluoride Molten Salts. J. Chin. Soc. Rare Earths 2021, 39, 594–600. [Google Scholar]
- Zhao, Y.F.; Wang, L.; Liu, W.H. Behavior of LaOF in Fluorine Salt Melt for the Preparation of Lanthanum by Electrolysis. Chin. J. Nonferrous Met. 1987, 3, 25–30. [Google Scholar]
- Jiang, Y.J.; Deng, Y.C. Discussion on the generation and recovery of HF gas during the molten salt electrolysis of rare earth oxides. Rare Earth 2016, 37, 149–151. [Google Scholar]
- Zhang, Z.H.; Zhao, L.Z.; Jiao, S.Z.; Wu, D.W. Research on the Preparation of Lanthanum Metal from Lanthanum Oxide by Electrolysis. Jiangxi Nonferrous Met. 1992, 3, 129–134. [Google Scholar]
- Lin, W.Q.; Xiao, Z.G. Research on the Preparation of High-Purity Metal Lanthanum by Molten Salt Electrolysis. Jiangxi Nonferrous Met. 2003, 1, 32–34. [Google Scholar]
- Wang, W.W. Fused Salt Electrorefining of Lanthanum. Chin. J. Rare Met. 2013, 37, 770–777. [Google Scholar]
- Li, B.C.; Li, S.J.; Kong, Y.P.; Chen, J.S.; Liu, K.R.; Han, Q. Dissolution Mechanism and Electrochemical Behavior of CeO2 in Molten CeF3-LiF-BaF2. Rare Met. Mater. Eng. 2020, 49, 749–754. [Google Scholar]
- Constantin, V.; Popescu, A.M.; Olteanu, M. Electrochemical studies on cerium(III) in molten fluoride mixtures. J. Rare Earths 2010, 28, 428–434. [Google Scholar] [CrossRef]
- Chandra, M.; Vandarkuzhali, S.; Ghosh, S. Redox behavior of cerium (III) in LiF–CaF2 eutectic melt. Electrochim. Acta 2011, 58, 150–156. [Google Scholar] [CrossRef]
- Gibilaro, M.; Massot, L.; Chamelot, P.; Taxil, P. Co-reduction of aluminum and lanthanide ions in molten fluorides: Application to cerium and samarium extraction from nuclear wastes. Electrochim. Acta 2009, 54, 5300–5306. [Google Scholar] [CrossRef]
- Lin, R.S.; Ye, G.A.; He, H.; Ouyang, Y.G.; Tang, H.B. Basic Research on the Process of Preparing Cerium Metal from Molten Salt Electrolysis of CeO2. Annu. Rep. China Inst. At. Energy 2009, 1, 345–347. [Google Scholar]
- Zhang, S.Q.; Wang, Y.D.; Wang, S.; Gao, Y.S.; Wang, X.M. Fluoride Molten Salt system for Electrolytic CeO2. J. Chin. Soc. Rare Earths 2015, 33, 455–460. [Google Scholar]
- Liu, Z.X.; Xu, Z.Q.; Wu, Y.F.; Li, Y.Q.; Dong, Y.F. Numerical simulation of the temperature field in a tank for the production of cerium metal by molten salt electrolysis. Rare Earths 2017, 38, 82–88. [Google Scholar]
- Li, M.; Wang, J.; Han, W.; Dong, Y.C. Molten salts electrolytic extraction of variable rare earth Tm using reactive Cu electrode. Sci. Sin. Chim. 2019, 49, 175–183. [Google Scholar] [CrossRef]
- Straka, M.; Korenko, M.; Szatmáry, L. Electrochemistry of praseodymium in LiF–CaF2. J. Radioanal. Nucl. Chem. 2011, 289, 591–593. [Google Scholar] [CrossRef]
- Yin, R.Y. Development of praseodymium metal preparation technology. Sichuan Rare Earth 2003, 3, 5–7. [Google Scholar]
- Electrolysis Group of Baotou Iron and Steel Metallurgical Research Institute. Electrolytic production of neodymium metal from neodymium oxide in fluoride molten salts. Rare Earths Niobium 1974, 3, 34–41. [Google Scholar]
- Bao, J.H. Study on improving the utilization of neodymium oxide during electrolysis. Rare Met. Express 2004, 7, 35–36. [Google Scholar]
- Mao, J.H.; Peng, G.H. Effects of Proportion of Electrolyte on 10KA Molten Salt Electrolyse in Producing Neodymium. Jiangxi Nonferrous Met. 2007, 3, 20–22. [Google Scholar]
- Zhang, X.L.; Deng, Z.M.; Hu, S.L. Low current density electrolysis of neodymium metal. J. Gannan Norm. Coll. 2006, 3, 66–68. [Google Scholar]
- Pang, Q.S.; Zhang, Y.K.; Huang, J.; Zhang, H. Analysis of Electrolytic Characteristics of Different Cathode Structures in Rare Earth Electrolytic Cells. Chin. Rare Earths 2019, 40, 17–23. [Google Scholar]
- Ji, Y.Z.; Xiao, F.X.; Sun, S.C.; Chen, J.Q.; Tu, G.F. Research Progress on Model Cell of High Current Rare Earth Reduction Cell. J. Chin. Soc. Rare Earths 2022, 40, 38–45. [Google Scholar]
- Chen, D.H.; Yan, S.H.; Li, Z.A.; Pang, S.M.; Xu, L.H.; Guo, X.Y. Key Technologies of 3000A Submerged-Liquid Cathodic Rare Earth Electrolysis Process. J. Chin. Soc. Rare Earths 2011, 29, 769–772. [Google Scholar]
- Chen, D.H.; Yan, S.H.; Li, Z.A.; Wang, Z.Q.; Pang, S.M.; Wang, X.S.; Xu, L.H. Liquid-Cathode Cell for Neodymium Electrolysis in NdF3-LiF-Nd2O3 Molten. J. Chin. Rare Earth Soc. 2009, 27, 302–305. [Google Scholar]
- Dysinger, D.K.; Murphy, J.E. Electrowinning of neodymium from a molten oxide-fluoride electrolyte. Bur. Mines Rep. Investig. 1994. [Google Scholar]
- Wei, X.M.; Pang, D.X.; Xu, X.R. Shape and Its Change of Carbon in Melting Salt Electrolyzed Nd. J. Funct. Mater. 1993, 6, 521–523. [Google Scholar]
- Xue, J.Q.; Liu, N.N.; Liu, Z.F.; Tang, C.B.; Bi, Q. Failure and Borate lmpregnation Protection of Graphite Anode forNeodymium Electrodeposition in Molten Salt. Rare Met. 2016, 40, 806–815. [Google Scholar]
- Abbasalizadeh, A.; Malfliet, A.; Seetharaman, S. Electrochemical Recovery of Rare Earth Elements from Magnets: Conversion of Rare Earth Based Metals into Rare Earth Fluorides in Molten Salts. Mater. Trans. 2017, 58, 400–405. [Google Scholar] [CrossRef]
- Yang, Y.S.; Lan, C.Q.; Guo, L.Y.; An, Z.Q.; Zhao, Z.W.; Li, B.W. Recovery of rare-earth element from rare-earth permanent magnet waste by electro-refining in molten fluorides. Sep. Purif. Technol. 2020, 233, 116030. [Google Scholar] [CrossRef]
- Hua, Z.S.; Wang, L.; Wang, J.; Xiao, Y.X.; Yang, Y.X.; Zhao, Z.; Liu, M.J. Extraction of rare earth elements from NdFeB scrap by AlF3–NaF melts. Mater. Sci. Technol. 2015, 31, 1007–1010. [Google Scholar] [CrossRef]
- Guo, T.; Wang, S.D.; Ye, X.S.; Li, Q.; Liu, H.N.; Guo, M.; Wu, Z.J. Research progress in the preparation of rare earth alloys by molten salt electrolysis method. Sci. Sin. Chim. 2012, 42, 1328–1336. [Google Scholar]
- Wang, X.; Jiao, Y.F.; Liao, C.F. Research status and development prospect of preparation for heavy rare earth metals and alloys by electroreduction in nonaqueous solvent. Nonferrous Met. Sci. Eng. 2018, 9, 99–104. [Google Scholar]
- Li, K.; Cao, Y.C.; Chen, G.H.; Liu, Y.B.; Yu, B.; Zhao, E.X. Study on Electrochemical Behavior of Gd-Mg Alloy Prepared by Co Deposition in Molten Salt. Chin. Rare Earths 2018, 39, 132–137. [Google Scholar]
- Peng, G.H.; Guo, X.F.; Qiu, C.Z.; Han, B.J.; Fang, L.; Zhang, S.L. Preparation of Gd-Mg Master Alloy by Co-electrodeposition Method in Fluoride Molten Salt. J. Kunming Univ. Sci. Technol. (Sci. Technol.) 2010, 35, 16–19+26. [Google Scholar]
- Yang, Q.S.; Chen, J.J.; Xie, J.Q. Preparation of Mg-Nd Intermediate Alloy by Electrolyzing in Fluoride Smelt Salt. Rare Met. 2007, 31, 45–49. [Google Scholar]
- Soare, V.; Burada, M.; Ostvold, T.; Kontoyannis, C.; Stefanidaki, E. Study of the Mg-Nd alloy obtained by electrolysis in molten oxifluoride media. J. Min. Metall. Sect. B Metall. 2003, 39, 209–221. [Google Scholar] [CrossRef]
- Soare, V.; Gurgu, C.; Burada, M. Producion of a Mg-Nd Alloy by Direct Electrolysis from an Oxifluoride Melt. Can. Metall. Q. 2006, 45, 153–160. [Google Scholar] [CrossRef]
- Deng, W.P.; Zeng, X.T.; Chi, X.D. Electrolytic production of magnesium-yttrium alloys and yttrium metal by molten salt electrolysis. Rare Met. 1997, 18, 59–62. [Google Scholar]
- He, S.; Li, Z.A.; Yan, S.H.; Wang, Z.Q.; Pang, S.M.; Chen, B.Y. Cathode Process in Electrolytic Codeposition of Y-Mg Alloy in Molten Fluoride. J. Chin. Soc. Rare Earths 2007, 25, 120–123. [Google Scholar]
- Yang, S.H.; Yang, F.L.; Liao, C.F.; Li, M.Z.; Wang, X. Electrodeposition of magnesium-yttrium alloys by molten salt electrolysis. J. Rare Earths 2010, 28, 385–388. [Google Scholar] [CrossRef]
- Leng, X.W.; Tang, H.; Liao, C.F.; Wang, X. Investigation on viscosity of electrolysis system for preparing Al-Cu-Y alloy via molten salt electrolysis. Nonferrous Met. Sci. Eng. 2019, 10, 8–11. [Google Scholar]
- Wang, X.; Liao, C.F.; Jiao, Y.F.; Tang, H. Cathodic reduction process of Al-Cu-Y alloy in fluoride-oxide eutectic system via molten salt electrolysis. J. Rare Earths 2018, 36, 324–330. [Google Scholar] [CrossRef]
- Wang, X.; Liao, C.F.; Jiao, Y.F.; Tang, H. Study on Cathodic Reduction Process During the Preparation of Al-Cu-Y Alloy by Molten Salt Electrolysis via Fluoride-Oxide System. Mater. Rev. 2017, 31, 50–54. [Google Scholar]
- Liao, C.F.; Luo, L.S.; Wang, X.; Tang, H. Preparation for A-Nd intermediate alloy by molten-salt electrolysis method and its mechanism. J. Chin. Soc. Rare Earths 2015, 25, 3523–3529. [Google Scholar]
- Jiao, Y.F.; Wang, X.; Liao, C.F.; Jia, S.; Tang, H.; Cai, B.Q.; Sun, Q.C. Density of Na3AIF6-AIF3-LiF-MgF2-Al2O3-Sm2O3 molten salt melt for Al-Sm alloy. J. Rare Earths 2018, 36, 190–196. [Google Scholar] [CrossRef]
- Liao, C.F.; Jiao, Y.F.; Wang, X.; Cai, B.Q.; Sun, Q.C.; Tang, H. Electrical conductivity optimization of the Na3AlF6–Al2O3–Sm2O3 molten salts system for Al–Sm intermediate binary alloy production. Int. J. Miner. Metall. Mater. 2017, 24, 1034–1042. [Google Scholar] [CrossRef]
- Sun, B.L.; Zhai, Y.C.; Tian, Y.W. Electrolytic production of aluminum-scandium alloy in fluoride salt system. Chin. J. Rare Met. 1998, 22, 32–35. [Google Scholar]
- Yang, S. Study on the Production of Aluminum-Scandium Alloy by Electrolysis. Ph.D. Thesis, Zhengzhou University, Zhengzhou, China, 2003. [Google Scholar]
- Yang, S.; Li, Q.; Gu, S.Q. Dissolution properties of scandium oxide in cryolite alumina system. Chin. J. Rare Met. 2003, 27, 418–420. [Google Scholar]
- Liu, X.; Xue, J.; Guo, Z. Segregation behaviors of Sc and unique primary Al3Sc in Al-Sc alloys prepared by molten salt electrolysis. J. Mater. Sci. Technol. 2019, 35, 1422–1431. [Google Scholar] [CrossRef]
- Li, L.X.; Wang, T.S.; Huang, X.L.; Huang, J.D. Research Progress on the Preparation of Al-Sc Master Alloy by Molten SaltElectrolysis Method. Mater. Rep. 2018, 32, 3768–3773. [Google Scholar]
- Liu, X.; Guo, H.; Xue, J.L.; Zhang, Y.N.; Li, X. Microstructure and mechanical properties of as casted Al-Si-Sc-Ce alloys prepared by molten salt electrolysis with varied cooling rate. J. Alloys Compd. 2023, 947, 169703. [Google Scholar] [CrossRef]
- Zhu, C.W.; Liu, X.; Cao, Z.M.; Xue, J.L.; Guo, Z.C. Microstructure and Properties of Al-M-Sc Alloy Prepared by Cathode Al-Liquid Electrolysis. Rare Met. 2022, 46, 419–427. [Google Scholar]
- Gibilaro, M.; Massot, L.; Chamelot, P.; Taxil, P. Study of neodymium extraction in molten fluorides by electrochemical co-reduction with aluminum. J. Nucl. Mater. 2008, 382, 39–45. [Google Scholar] [CrossRef]
- Gibilaro, M.; Massot, L.; Chamelot, P.; Cassayre, L.; Taxil, P. Electrochemical extraction of europium from molten fluoride media. Electrochim. Acta 2009, 55, 281–287. [Google Scholar] [CrossRef]
- Gao, Y.; Shi, Y.K.; Liu, X.L.; Huang, C.; Li, B. Cathodic Behavior of Samarium(III) and Sm-Al alloys Preparation in Fluorides Melts. Electrochim. Acta 2016, 190, 208–214. [Google Scholar] [CrossRef]
- Gal, L.; Charbonnier, V.; Zhang, J.X. Optimization of the La substitution by Mg in the La2Ni7 hydride-forming system for use as negative electrode in Ni-MH battery. Int. J. Hydrogen Energy 2015, 40, 17017–17020. [Google Scholar] [CrossRef]
- Wu, R.; Yuan, H.P.; Liu, Y.R.; Hou, Z.Y.; Li, Z.N.; Wang, S.M.; Jiang, L.J.; Hao, L. Effect of carbon coating on electrochemical properties of AB3.5-type La-Y-Ni-based hydrogen storage alloys. J. Rare Earths 2022, 40, 1264–1271. [Google Scholar] [CrossRef]
- Qi, Y.; Li, H.X.; Wan, C.B. Research Progress of Element Substitution Applied in La-Mg-NiHydrogen Storage Alloy. Rare Earth 2023, 44, 117–126. [Google Scholar]
- Luo, N.; Peng, G.H. Electrochemical Preparation of La-Mg-Ni Alloys in Molten Fluorides. J. Gannan Norm. Univ. 2018, 39, 50–53. [Google Scholar]
- Wang, X.; Liao, C.F.; Wang, R.X.; Sun, Q.C. Characterization and Preparation of Ni-Yb Alloy by Molten Salt Electrolysis in Fluoride Melt. Mater. Rep. 2019, 33, 750–753. [Google Scholar]
- Zhang, Y.J.; Cai, B.Q.; Wang, X.; Wang, R.X.; Shi, Z.N. Dissolution and structural evolution of Yb2O3 in eutectic LiF–YbF3 molten salts. J. Fluor. Chem. 2023, 269, 110–145. [Google Scholar]
- Wang, Y.X.; Lai, H.S.; Wen, X.Q.; Liu, W.W.; Hong, K. Preparation of YNi alloy by molten salt electrolysis in fluoride system. Nonferrous Met. Sci. Eng. 2021, 12, 126–130. [Google Scholar]
- Yu, B.; Huang, H.T.; Zhang, Q.J.; Yan, Q.C.; Kang, J.; Liu, Y.B. Preparation of Y-Ni Alloys by Molten Salt Electrolysis. Rare Earth 2021, 42, 81–86. [Google Scholar]
- Massot, L.; Chamelot, P.; Taxil, P. Cathodic behavior of samarium(III) in LiF–CaF2 media on molybdenum and nickel electrodes. Electrochim. Acta 2005, 50, 5510–5517. [Google Scholar] [CrossRef]
- Chamelot, P.; Massot, L.; Hamel, C.; Nourry, C.; Taxil, P. Feasibility of the electrochemical way in molten fluorides for separating thorium and lanthanides and extracting lanthanides from the solvent. J. Nucl. Mater. 2007, 360, 64–74. [Google Scholar] [CrossRef]
- Saïla, A.; Gibilaro, M.; Massot, L.; Chamelot, P.; Taxil, P.; Affoune, A.M. Electrochemical behaviour of dysprosium(III) in LiF–CaF2 on Mo, Ni and Cu electrodes. J. Electroanal. Chem. 2010, 642, 150–156. [Google Scholar] [CrossRef]
- Nohira, T.; Kobayashi, S.; Kobayashi, K.; Hagiwara, R.; Oishi, T.; Konishi, H. Electrochemical Formation of Nd-Ni Alloys in Molten LiF-CaF2-NdF3. ECS Trans. 2010, 33, 205–212. [Google Scholar] [CrossRef]
- Watanabe, Y.; Norikawa, Y.; Yasuda, K. Electrochemical Dy-Alloying Behaviors of Inconel and Hastelloy in Molten LiF–CaF2–DyF3. Mater. Trans. 2019, 60, 379–385. [Google Scholar] [CrossRef]
- Kobayashi, S.; Nohira, T.; Kobayashi, K.; Yasuda, K.; Hagiwara, R.; Oishi, T.; Konishi, H. Electrochemical Formation of Dy-Ni Alloys in Molten LiF-CaF2 -DyF3. J. Electrochem. Soc. 2012, 159, 193–197. [Google Scholar] [CrossRef]
- Yasuda, K.; Kondo, K.; Nohira, T.; Hagiwara, R. Electrochemical Formation of Pr–Ni Alloys in LiF–CaF2–PrF3 and NaCl–KCl–PrCl3 Melts. J. Electrochem. Soc. 2014, 161, 3097–3104. [Google Scholar] [CrossRef]
- Yasuda, K.; Oishi, T.; Kagotani, T. Electrochemical Dy-alloying behaviors of Ni-based alloys in molten LiF–CaF2–DyF3 and LiCl–KCl–DyCl3: Effects of temperature and electrolysis potential. J. Alloys Compd. 2021, 889, 161605. [Google Scholar] [CrossRef]
- Du, C. Development of Highly Selective Recovery System of Rare Earth Elements from Ce-Containing NdFeB. Master’s Thesis, Jiangxi University of Science and Technology, Ganzhou, China, 2021. [Google Scholar]
- Kawaguchi, K.; Nohira, T. Electrochemical Formation of Dy–Fe Alloys in Molten LiF–CaF2–DyF3. J. Electrochem. Soc. 2023, 170, 102504. [Google Scholar] [CrossRef]
- Zhou, A.G.; Chen, Y.X.; Peng, S.H.; Wang, J. Effects of Silicon on Pr-Nd Alloy Production by Molten Salt Electrolytic Method. China Tungsten Ind. 2012, 27, 44–46. [Google Scholar]
- Lu, X.N.; Zhang, S.Z.; Xie, X.R.; Wen, H.R. On the effective control of non-rare earth impurity in the preparation of praseodymium neodymium alloy by 25 kA molten salt electrolysis. Nonferrous Met. Sci. Eng. 2015, 6, 10–15. [Google Scholar]
- Du, S.L.; Shen, J.C.; Tang, D.X. Molten salt electrolysis of praseodymium-rich praseodymium oxides for the synthesis of NdPrFe alloys. J. Rare Earths 1994, 18, 167–171. [Google Scholar]
- Wu, Y.F.; Ma, S.Y.; Wang, Z.F.; Bian, X.; Liu, Z.X.; Liu, Y.L. Formation mechanism and countermeasures of nodules at furnace bottom of15 kA rare earth molten salt electrolysis cell. China Nonferrous Metall. 2023, 52, 81–87. [Google Scholar]
- Yu, L.Z. Molten salt electrolysis process for Nd-Fe alloys. Rare Earth Inf. 1992, 8, 8–9. [Google Scholar]
- Makaseev, Y.; Buinovskiy, A.; Zhitkov, S.; Kartashov, E.; Sofronov, V. Production of Nd-Fe Foundry Alloy by Electrolysis in Molten Salts. MATEC Web Conf. 2017, 96, 9. [Google Scholar] [CrossRef]
- Yu, B.; Liu, Y.B.; Zhang, Q.J.; Huang, H.T.; Kang, J.; Yan, Q.C. Preparation of Pr-Nd-Ce Alloys by Molten Salt Electrolysis. Nonferrous Met. (Extr. Metall.) 2021, 4, 90–95. [Google Scholar]
- Chen, G.H.; Wang, X.Q.; Liu, Y.B.; Zhao, E.X.; Yu, B.; Li, K. Preparation of Pr-Nd-Dy Alloys by Molten Salt Electrolysis. Rare Earth 2015, 36, 80–84. [Google Scholar]
- Chen, G.H.; Cao, Y.C.; Liu, Y.B.; Li, K.; Chen, Y.X.; Yu, B. Preparation of Pr-Nd-Gd Alloys by Molten Salt Electrolysis. J. Chin. Soc. Rare Earths 2015, 33, 206–210. [Google Scholar]
- Holcombe, B.; Sinclair, N.; Wasalathanthri, R.; Mainali, B.; Guarr, E.; Baker, A.A.; Usman, S.O.; Kim, E.; Sen-Britain, S.; Jin, H.; et al. Sustainable and Energy-Efficient Production of Rare-Earth Metals via Chloride-Based Molten Salt Electrolysis. ACS Sustain. Chem. Eng. 2024, 12, 4186–4193. [Google Scholar] [CrossRef]
- Akolkar, R. Perspective—Is Sustainable Electrowinning of Neodymium Metal Achievable? J. Electrochem. Soc. 2022, 169, 043501. [Google Scholar] [CrossRef]
- Sinclair, N.S.; Wasalathanthri, R.; Mainali, B.; Holcombe, B.; Baker, A.; Kim, E.; Orhan, A.; Mccall, S.; Akolkar, R. (Invited) Rare Earth Metal Production Via Chloride Based Molten-Salt Electrolysis. ECS Meet. Abstr. 2023; MA2023-01, 1522. [Google Scholar]
- Sinclair, N.S.; Holcombe, B.P.; Baker, A.; Kim, E.; Mccall, S.; Akolkar, R. Neodymium Metal Production Via Chloride Based Molten-Salt Electrolysis. ECS Meet. Abstr. 2023, MA2023-02, 1338. [Google Scholar] [CrossRef]
- Liu, S.Z.; Chen, L.Y.; Li, B.; Wang, L.L.; Yan, B.; Liu, M.G. Anode processes for Nd electrowinning from LiF-NdF3-Nd2O3 melt. Electrochim. Acta 2014, 147, 82–86. [Google Scholar] [CrossRef]
- Gibilaro, M.; Remazeilles, C.; Massot, L.; Chamelot, P. Process Optimization to Avoid Perfluorocarbon Emission During Neodymium Rare Earth Electrolysis in Molten LiF-NdF3-Nd2O3. J. Electrochem. Soc. 2022, 169, 083501. [Google Scholar] [CrossRef]
- Zhang, L.Z.; Wang, X.F.; Gong, B. Perfluorocarbon emissions from electrolytic reduction of rare earth metals in fluoride/oxide system. Atmos. Pollut. Res. 2017, 9, 61–65. [Google Scholar] [CrossRef]
- Lin, R.S.; He, H.; Ye, G.A.; Tang, H.B. Study on Anode Process in Molten Salt Electrolysis of CeO2. Chin. Rare Earths 2016, 37, 137–143. [Google Scholar]
- Vogel, H.; Friedrich, B. Reducing Greenhouse Gas Emission from the Neodymium Oxide Electrolysis. Part II: Basics of a Process Control Avoiding PFC Emission. Int. J. Nonferrous Metall. 2017, 6, 27–46. [Google Scholar] [CrossRef]
- Vogel, H.; Flerus, B.; Stoffner, F.; Friedrich, B. Reducing Greenhouse Gas Emission from the Neodymium Oxide Electrolysis. Part I: Analysis of the Anodic Gas Formation. J. Sustain. Metall. 2017, 3, 99–107. [Google Scholar] [CrossRef]
- Du, J.H.; Xi, Z.P.; Ju, H.; Cai, T.X.; Zhang, Y.P.; Wu, H.R.; Liu, S.Y.; Sun, B.L.; Duan, S.J. Development of Nonconsumable Anode for Rare Earth Molten Salt Electrolysis. Rare Earth 2001, 22, 65–67. [Google Scholar]
- Zhu, J.D.; Hu, J.B.; Xiao, H.; Yang, L.F.; Yang, M.S.; Wang, S.C.; Zhang, J.J. Aluminum-based metal organic frameworks for greenhouse gases CF4 and C2F6 capture with excellent capacity and selectivity. Sep. Purif. Technol. 2024, 331, 125614. [Google Scholar] [CrossRef]
- Xiao, S.; Wang, F.G.; Cui, H.; Xia, Y.L.; Chen, J.Y.; Xie, S.J.; Tang, J. Adsorbent exploration of CaSO4 for C4F7N/N2-based insulation devices from experimental and theoretical insights. CSEE J. Power Energy Syst. 2022, 1–9. [Google Scholar]
- Zhang, X.X.; Deng, Z.T.; Fu, M.L.; Zhuo, R.; Xiao, S.; Li, W. Effect of Small Amount of O2 on Breakdown and Decomposition Characteristics of c-C4F8/N2 Mixture Gas. High Volt. Eng. 2019, 45, 708–715. [Google Scholar]
- Zhang, H.X.; Lv, X.J.; Zhong, S.P.; Wang, J.E.; Chen, H.; Liu, S.; Zeng, X.P.; Jian, Y.Z. Electric Field Numerical Simulation and Cathode Structure Optimization of Rare Earth Electrolysis Cell. J. Chin. Soc. Rare Earths 2020, 38, 667–676. [Google Scholar]
- Lian, H.; Pan, Y.; Guo, H.T.; Sheng, P.J.; Liu, Y.; Kong, X.M.; Zhang, Z.J.; Ai, Y.Z.; Yang, P.L.; Sun, Z.J.; et al. A Large-Scale Water-Cooled Anode Conductor Plate Device for Rare Earth Molten Salt Electrolysis Cells. Chinese Patent CN113802150A, 17 December 2021. [Google Scholar]
- Lian, H.; Pan, Y.; Guo, H.T.; Liu, Y.; Kong, X.M.; Zhang, Z.J.; Ai, Y.Z.; Sun, Z.J.; Liu, W.J.; Hou, L.; et al. A Large-Sized Cathode Rare Earth Molten Salt Electrolysis Cell. Chinese Patent CN113337851A, 3 September 2021. [Google Scholar]
- Northeastern University. A Covered Upper-Inserted Cathode Rare Earth Electrolysis Cell. Chinese Patent CN115354364A, 18 November 2022.
- Central South University. A New Type of Rare Earth Electrolysis Cell. Chinese Patent CN105256337A, 20 January 2016.
- Central South University. A New Type of Rare Earth Molten Salt Electrolysis Cell with a Novel Electrode Structure. Chinese Patent CN105624736B, 9 January 2018.
- Xue, J.Q.; Zhang, J.; Tang, C.B.; Bi, Q.; Guo, Z.Y. Simulation and Design of Enclosed Rare Earth Electrolytic Cell with Bottom-Liquid Cathode. Rare Met. Cem. Carbides 2014, 42, 19–22+30. [Google Scholar]
- Zhang, J. The Technology Design Research on Liquid Cathode Molten Salt Electrolysis of Nd. Master’s Thesis, Xi’an University of Architecture and Technology, Xi’an, China, 2015. [Google Scholar]
- Li, F. The Design and Optimization Research on 6kA Bottom Liquid Cathode Structure Electrolytic Cell of Neodymium. Master’s Thesis, Xi’an University of Architecture and Technology, Xi’an, China, 2020. [Google Scholar]
- Northeastern University. A Bottom Cathode Rare Earth Electrolysis Cell. Chinese Patent CN115074781A, 20 September 2022.
- Li, Y.L.; Hu, X.L.; Xu, J.F.; Zhou, L.S.; Tang, C. Potential Analytical Solution of Electric Potential under lnfluence of ElectricDouble Layer in Bottom Cathode Electrolyzer. Guangzhou Chem. Ind. 2023, 51, 44–47. [Google Scholar]
- Wang, J.; Wang, C.H.; Tu, G.F.; Ren, Y.Q.; Mi, L. Heat Balance Calculation in 10kA Bottom-Cathode-Structure Rare Earth Electrolysis Cell. Chin. Rare Earths 2008, 29, 61–63. [Google Scholar]
- Wang, J.; Zhang, Z.L.; Tu, G.F.; Wu, W.Y. Simulation of the Electric Field in 10kA Bottom-cathode-structure Rare Earth Electrolytic Cell. Chin. Rare Earths 2010, 31, 36–39. [Google Scholar]
- Bao, G.Z.; Wu, Y.F.; Ma, S.Y.; Wang, Z.F.; Dong, Y.F.; Liu, Z.X. The Impact of Electric Field and Flow Field on Electrolysis Efficiency in Rare Earth Electrolysis Cells. In Proceedings of the 4th Youth Academic Conference of the Chinese Society of Rare Earths, Ganzhou, China, 19 May 2023. [Google Scholar]
- Wen, T.G.; Zhang, B.; Zhang, J.W.; Li, M.Z.; Yang, S.H. Numerical simulation of multiphase flow in 6 kA neodymium electrolytic cell. Nonferrous Met. Sci. Eng. 2023, 14, 706–715. [Google Scholar]
- Zhang, K.Y.; Jiang, W.Z.; Lv, Y.; Wang, S.Z.; Tang, J.W.; Wang, J.X. Numerical Simulation of the Effect of Anode Slotting on BubbleMovement in Bottom-cathode Rare Earth Electrolytic Cell. Chin. Rare Earths 2023, 44, 18–27. [Google Scholar]
- Dong, Y.F.; Liu, Z.X.; Wu, Y.F.; Xu, L.Y.; He, J.H. Simulation and Optimization of Polar Distance in 60kA Bottom-Cathode Rare Earth Electrolyzer. Nonferrous Met. (Extr. Metall.) 2013, 10, 32–35. [Google Scholar]
- Liu, Z.X.; Dong, Y.F.; Wu, Y.F.; He, J.H.; Xu, L.Y. Numerical Simulation of Effect of Anode Inclination on the Flow of the Molten Salt in 60kA Bottom-cathode Rare Earth Electrolytic Cell. Chin. Rare Earths 2013, 34, 21–24. [Google Scholar]
- Shi, F. Present Condition of Research and Developing Trend in the Rare Earth Electrolysis Cell. J. Chin. Soc. Rare Earths 2007, 25, 70–76. [Google Scholar]
- In 2015, China released a total of 23 national standards for rare earths. Sustain. Min. Metall. 2016, 32, 64–65.
- Ning, D.J. A Siphon Furnace Device for Rare Earth Metals. Chinese Patent CN116356380A, 30 June 2023. [Google Scholar]
- Wang, M. Process Design of Rare Earth Metal Smelting Production Line. Rare Met. Cem. Carbides 2023, 51, 13–17. [Google Scholar]
- Liang, B. Reserch on Key Technology of Siphon Extraction Method of Rare Earth Metal Molten Product Based on CFD. Master’s Thesis, Guilin University of Electronic Technology, Guilin, China, 2023. [Google Scholar]
- Sichuan Jiangtong Rare Earth Co., Ltd. A Casting Mold Suitable for Fluorine Removal Device by Siphon Smelting of Rare Earth. Chinese Patent CN217990857U, 9 December 2022. [Google Scholar]
- Baotou Shengyou Rare Earth Co., Ltd. Electrolysis Cell Siphon Recovery Device for Rare Earth Metals. Chinese Patent CN216378431U, 26 April 2022. [Google Scholar]









| Molten Salt System | Electrolysis Temperature | Electrolytic Raw Materials | Time of Electrolysis | Cathodic Current Density/(A/cm2) | Metal Quantity | Current Efficiency |
|---|---|---|---|---|---|---|
| 65 wt.% LaF3–23 wt.% LiF–12 wt.% BaF2 [41] | 1223 K | La2O3 | 60 min | 7 | 36.8 g | 85.21% |
| 1223 K | Tetragonal crystal system LaOF | 60 min | 7 | 38.1 g | 88.23% | |
| 1223 K | Rhombic hexagonal crystal system LaOF | 90 min | 7 | 46.7 g | 86.51% | |
| 70 wt.% LaF3–LiF–BaF2 [43] | 1223 K | La2O3 | 70 min | 8 | 45.3 g | 93.38% |
| LiF–LaF3 [44] | 1243–1253 K | Fluorescence-level purity La2O3 | 20 min | 5 | 71.8 g | 81.57% |
| Electrolyte System | 38 wt.% NdF3–35 wt.% LiF–27 wt.% CaF2 [64] | 89 wt.% NdF3–11 wt.% LiF [56] | 90 wt.% NdF3–10 wt.% LiF [59] | (88.9–90 wt.%) NdF3–LiF [62] |
|---|---|---|---|---|
| Electrode | Anode: graphite Cathode: tungsten rod | Anode: graphite electrolyzer Cathode: 6–8 mm diameter molybdenum rod | Anode: graphite Cathode: 28 mm diameter tungsten rod | Anode: graphite block Cathode: tungsten plate |
| Electrolysis temperature | 1303 K | 1308 K | 1343–1353 K | 1293 K |
| Cathode current density | 10.97 A/cm2 | 7 A/cm2 | 0.85–1.39 A/cm2 | 1.95 A/cm2 |
| Tank voltage | not indicated | 4.50–5.50 V | 4–6.50 V | 4.80–6 V |
| Electrolyzer structure | Anode and cathode up plug rare earth cell | Anode and cathode up plug rare earth cell | Anode and cathode up plug rare earth cell | Bottom cathode rare earth cell |
| Current efficiency | 52.7% | 62% | 57.60–87.10% | 75.36–85.75% |
| Rare earth direct yield | 38.60% | 88.50% | Not indicated | 95.20% |
| Rare Earth Metals/Rare Earth Alloys | Electrolyte | Raw Material | Electrolysis Temperature | Cathode Current Density | Current Efficiency | Metal Yield |
|---|---|---|---|---|---|---|
| La [41] | 65 wt.% LaF3–23 wt.% LiF–12 wt.% BaF2 | La2O3 | 1233 K | 7 A/cm2 | 85.21% | 99.29% |
| La [43] | 70 wt.% LaF3–LiF–BaF2 | La2O3 | 1223 K | 8 A/cm2 | 93.38% | 99.29% |
| La [44] | LaF3–LiF | La2O3 | 1263 K | 5 A/cm2 | 81.57% | not indicated |
| Ce [50] | 63 wt.% CeF3–21 wt.% LiF–16 wt.% BaF2 | CeO2 | 1173 K | 6.25 A/cm2 | 53 % | 94.70% |
| Pr [55] | PrF3–LiF | Pr6O11 | 1213–1233 K | 6 - 8 A/cm2 | 70-80% | 90% |
| Nd [56] | 89 wt.% NdF3–11 wt.% LiF | Nd2O3 | 1308 K | 7 A/cm2 | 62% | 88.50% |
| Nd [64] | 38 wt.% NdF3–35 wt.% LiF–27 wt.% CaF2 | Nd2O3 | 1303 K | 10.97 A/cm2 | 52.70% | 38.60% |
| Nd [59] | 90 wt.% NdF3–10 wt.% LiF | Nd2O3 | 1343–1353 K | 0.85–3.79 A/cm2 | 57.60–87.10% | not indicated |
| Nd [62] | 88.9 wt.% NdF3–11.1 wt.% LiF | Nd2O3 | 1233–1293 K | 1.95 A/cm2 | 74.30–85.75% | 93.10–95.20% |
| Nd–Mg [74] | NdF3–LiF–BaF2–Na3AlF6 | Nd2O3 | 1323 K | 6–7 A/cm2 | 60–72.60% | 84.2–95.2% |
| Y–Ni [102] | 85 wt.% YF3–15 wt.% LiF | Y2O3 | 1273 K | 10 A/cm2 | 72.80% | not indicated |
| Pr–Nd [114] | 90 wt.% (Pr, Nd)F3–10 wt.% LiF | REO (RE = Nd, Pr) | 1273–1373 K | 4.5–6.5 A/cm2 | 57.40-85% | not indicated |
| Pr–Nd [116] | 65 wt.% NdF3–15 wt.% BaF2–20 wt.% LiF | REO (RE = Nd, Pr) | 1303 K | 8–13 A/cm2 | 57% | 90% |
| Pr–Nd–(6–8 wt.%) Dy [121] | (87.50–90.91 wt.%)REF3–LiF | REO (RE = Pr, Nd, Dy) | 1263–1353 K | 4–8 A/cm2 | 76.59% | 93.20–98.12% |
| Pr–Nd–(10–15 wt.%) Gd [122] | 17.85 wt.% GdF3–69.03 wt.% (Pr, Nd)F3–13.12 wt.% LiF | REO (RE = Pr, Nd, Dy) | 1283–1373 K | 5 - 9 A/cm2 | 75.50–79.60% | 93.20–98.00% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liao, C.; Que, L.; Fu, Z.; Deng, P.; Li, A.; Wang, X.; Chen, S. Research Status of Electrolytic Preparation of Rare Earth Metals and Alloys in Fluoride Molten Salt System: A Mini Review of China. Metals 2024, 14, 407. https://doi.org/10.3390/met14040407
Liao C, Que L, Fu Z, Deng P, Li A, Wang X, Chen S. Research Status of Electrolytic Preparation of Rare Earth Metals and Alloys in Fluoride Molten Salt System: A Mini Review of China. Metals. 2024; 14(4):407. https://doi.org/10.3390/met14040407
Chicago/Turabian StyleLiao, Chunfa, Lianghua Que, Zanhui Fu, Pan Deng, Alin Li, Xu Wang, and Shumei Chen. 2024. "Research Status of Electrolytic Preparation of Rare Earth Metals and Alloys in Fluoride Molten Salt System: A Mini Review of China" Metals 14, no. 4: 407. https://doi.org/10.3390/met14040407





