Surface Decarburization of the Hypo-Eutectoid Carbon Steel C45 during Annealing in Steady Air at Temperatures T > AC1
Abstract
:1. Introduction
2. Theoretical Basics
2.1. Metallurgical Theory of the Decarburization of Hypo-Eutectoid Carbon Steel at T > AC1
2.2. Theoretical Overview of the Oxidation of Hypo-Eutectoid Carbon Steel at T > AC1
2.2.1. Oxidation of Iron
2.2.2. Oxidation of Carbon
3. Experimental
4. Results and Discussion
4.1. Oxidation of the Surface of C45 Steel
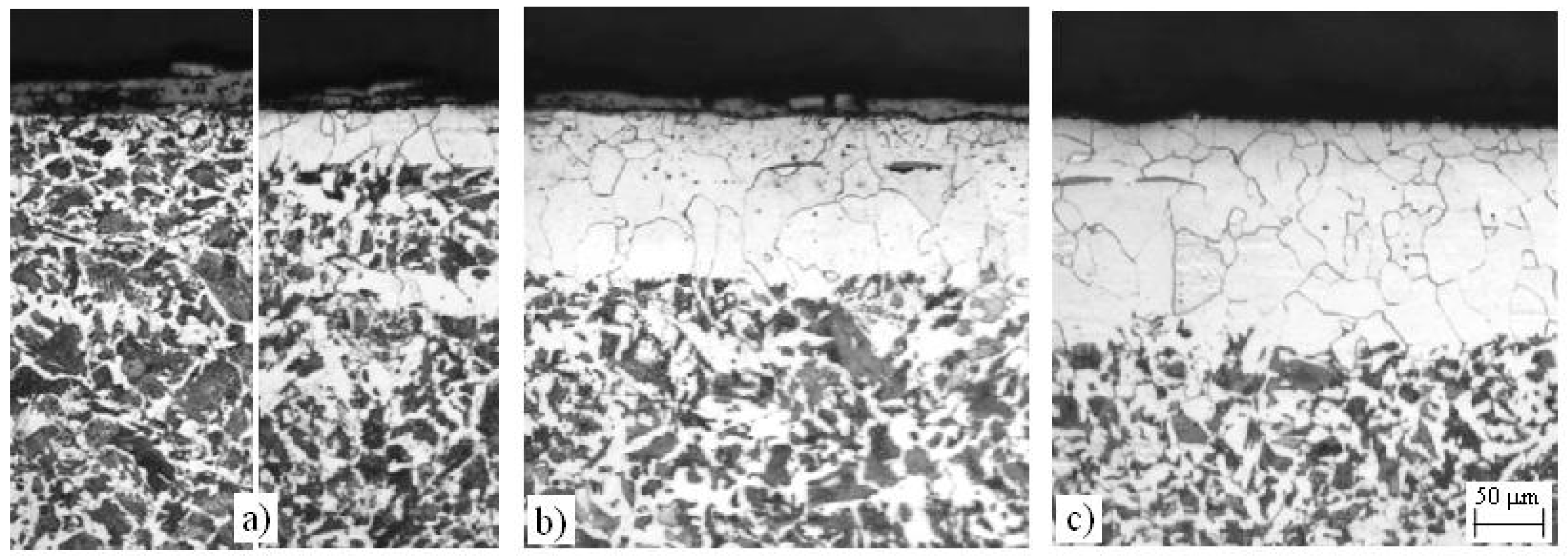
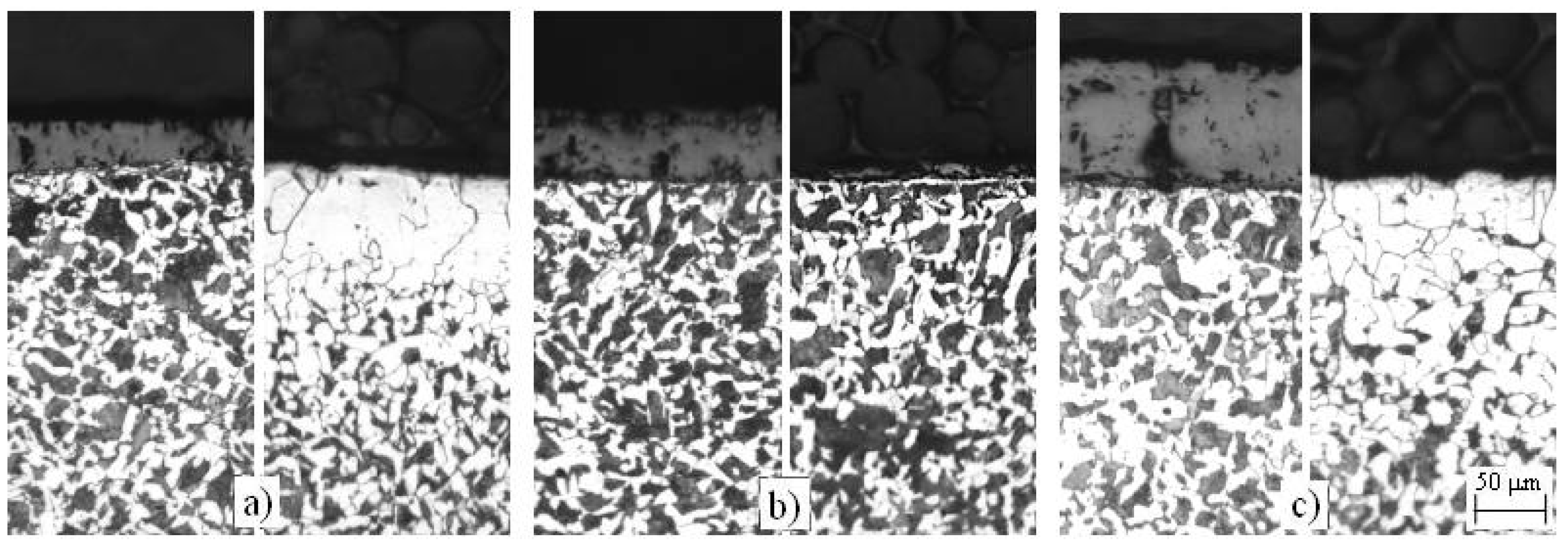
4.2. Decarburization of the Surface of C45 Steel
4.2.1. Annealing at Temperature T = 760 °C
4.2.2. Annealing at Temperature T = 850 °C
4.2.3. Annealing at Temperature T = 950 °C
4.2.4. Annealing at Temperature T = 1100 °C
4.3. Theoretical Assessment of the Thickness of the Decarburized Layer
Method of Calculating the Thickness of the Decarburized Layer
5. Conclusions
- The oxidation, the maximum visible decarburization of the surface and the completely decarburized ferrite layer grow with longer annealing times in accordance with the parabolic law of growth. The thickness of the decarburized layer increases exponentially with higher temperatures. The reasons for some global and local discrepancies in practice (total absence of decarburization, thinner completely decarburized ferrite surface layer at higher temperatures, locally limited decarburization) must be searched for in the ratio of the rates of oxidation and decarburization, the different fittinsg of the oxide layer on the steel surface, the local peeling off and cracking of the oxide layer and with that the associated different rates of oxidation for the individual local segments, the effect of segregations on the kinetics of oxidation and decarburization, the effect of segregations on the local peeling off, and the cracking of the oxide layer.
- Under the annealing conditions Ta = 850 °C, ta = 1 h there is no visible decarburization. This means that under these conditions, the oxidation of the surface of C45 steel that was grinded prior to annealing is faster than the decarburization and that normalization and annealing for quenching can be performed in an air atmosphere. Because of the very locally limited minimal decarburization at Ta = 760 °C, ta = ½ h it is acceptable to also perform annealing for a dual-phase (ferrite + martensite) microstructure in an air atmosphere under these conditions. Under all other annealing conditions, it is necessary to introduce measures for the prevention of decarburization or to take into account the addition of thickness prior to the annealing, which is the sum of the thicknesses of both the oxidized layer and the visible decarburized layer.
- A completely ferrite surface layer without a partially decarburized layer exists only in the samples that were annealed at Ta = 760 °C (AC1 < Ta < AC3) in a dual-phase region (α + γ). This is a consequence of the specific course of the austenite decarburization at the inner boundary of the ferrite layer in conditions of constant and time-independent thermodynamic equilibrium between the ferrite and austenite grains in the whole volume of an annealed piece. These conditions do not allow the partial decarburization towards the interior to occur, as is the case with pieces annealed at Ta > AC3.
- A continuous, visible, decarburized ferrite layer forms only under the annealing conditions Ta = 760 °C, ta > 1 h, while at Ta = 1100 °C, ½ h ≤ ta ≤ 2 h (steel is very overheated here) there is a partially decarburized layer spread towards the interior under the continuous surface ferrite layer. Under all other annealing conditions there are only local decarburized surface layers in which there is, besides a partial decarburization, also a thin superficial, completely ferrite layer observed. This layer forms already during the annealing at the annealing temperatures AC1 < Ta < TG, while at Ta > TG it only forms later, during the cooling of the completely decarburized austenite. The thickest strongly decarburized surface layer is formed at Ta = 1100 °C, which is a consequence of the high temperatures and their effect on the oxidation and decarburization of the steel surface.
- Even though the Van-Ostrand-Dewey equation was developed for the decarburization of austenite (Ta > TG), the results show that it can be used, at least in certain time periods, for all annealing temperatures Ta > AC1, which widens the temperature region of its use considerably.
- With the method of trial and error, the Van-Ostrand-Dewey equation enables the adjustment of calculated values with experimental measurements and an assessment of the accuracy of the measurements for different experimental techniques. The metallographically and theoretically measured depths of decarburization with a carbon content of 0.91C0 to 0.98C0 in a ferrite-pearlite microstructure, formed during cooling in steady air, can be adjusted, in general, with a 93% to 98% accuracy.
- The most suitable equation for calculating the diffusion coefficient of carbon in austenite for the researched experimental state is also determined by the Van-Ostrand-Dewey equation. Because of the completely decarburized austenite in the surface layer, the equation that gives the lowest values for the diffusion coefficient of carbon in austenite at annealing temperatures Ta (K) is appropriate: (cm2/s); R = 8.314 J/mol·K.
- The equations used to calculate the thickness of oxidized steel dm and the absolute depth of the decarburization xA can also be used to precisely theoretically determine the visible decarburization of the C45 steel with a difference of both quantities = xA − dm. This shows that the equation for growth of the oxide layer dox on iron, from which the thickness of the oxidized iron dm is calculated, also describes the oxidation of non-alloyed steel in steady air relatively well, even though the literature stated a slower oxidation of non-alloyed steel in comparison with iron.
- Most types of annealing take place at temperatures T > AC3; therefore, the addition of thickness, which has to be more than the absolute depth of the decarburization x0.884, must be taken into account before annealing in steady air for C45 steel with C = 0.43 wt % to achieve the required hardness. In the case of the annealing conditions at which visible decarburization does not exist, the additional thickness only has to be greater than the calculated thickness of the oxidized steel dm. When annealing for the production of dual-phase steel under the annealing conditions Ta = 760 °C, ta ≥ 1 h, it is necessary to ensure the addition of thickness in the size of the absolute depth of decarburization xA = x0.976, because of the removal of the surface ferrite layer.
Author Contributions
Conflicts of Interest
References
- Leslie, W.C. The Physical Metallurgy of Steels; McGraw-Hill Inc.: London, UK, 1982; pp. 74–81. ISBN 0-07-066385-8. [Google Scholar]
- De Cooman, B.C.; Speer, J.G. Fundamentals of Steel Product Physical Metallurgy; AIST: Warrendale, PA, USA, 2011; pp. 61–64, 37–42. [Google Scholar]
- Billings, G.A. Oxidation and Decarburization Kinetics of Iron-Carbon Alloys in Carbon Dioxide-Carbon Monoxide Atmosphere. Master’s Thesis, McMaster University, Hamilton, ON, Canada, 1966. [Google Scholar]
- Marston, H.F.; Rose, A.J.; Abbot, R.E.; Bugdol, M.; Richardson, W. Oxidation and Decarburization of High Carbon Steels; Final Report; European Commission, Directorate-General Science, Research and Development: Luxembourg, 1998; pp. 14, 15, 52, 53, 65, 66. ISBN 92-828-4548-6. [Google Scholar]
- Mardon, C. The Austenitization and Decarburization of High Silicon Spring Steel. Ph.D. Thesis, University of Canterbury, Christchurch, New Zealand, 1998. [Google Scholar]
- Mayott, S.W. Analysis of the Effect of Reduced Oxygen Atmospheres on the Decarburization Depth of 300M Alloy Steel. Master’s Thesis, Rensselaer Polytechnic Institute, Department of Materials Science and Engineering, New York, NY, USA, 2010. [Google Scholar]
- Zhang, C.L.; Zhou, L.Y.; Liu, Y.Z. Surface decarburization characteristics and relation decarburized types and heating temperature of spring steel 60Si2MnA. Int. J. Miner. Metall. Mater. 2013, 20, 720–724. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, W.; Tong, Q.; Wang, L. Effects of Temperature and Oxygen Concentration on the Characteristics of Decarburization of 55SiCr Spring Steel. ISIJ Int. 2014, 54, 1920–1926. [Google Scholar] [CrossRef] [Green Version]
- Zhao, F.; Zhang, C.L.; Liu, Y.Z. Ferrite decarburization of high silicon spring steel in three temperature ranges. Arch. Metall. Mater. 2016, 61, 1715–1722. [Google Scholar] [CrossRef]
- Schumann, H. Metallographie; 12. Auflage; VEB-DVG: Leipzig, Germany, 1983; pp. 316–321, 368, 150–152. ISBN 3-342-00009-0. [Google Scholar]
- Zorc, M. Decarburization of Non-Alloy Medium Carbon Steel during Annealing in an Air Atmosphere. Bachelor’s Thesis, University of Ljubljana, Faculty of Natural Sciences and Engineering-Department of Materials and Metallurgy, Ljubljana, Slovenia, 2016. [Google Scholar]
- Hasegava, M. Ellingham Diagram. In Treatise on Process Metallurgy-Vol. 1: Process Fundamentals; Seetharaman, S., Ed.; Elsevier: Oxford, UK, 2013; pp. 507–516. ISBN 9870080969862. [Google Scholar]
- Vodopivec, F. Kovine in Zlitine (Metals and Alloys, in Slovenian); Inštitut za Kovinske Materiale in Tehnologijo: Ljubljana, Slovenia, 2002; pp. 159–164. ISBN 961-238-084-8. [Google Scholar]
- Chen, R.Y.; Yuen, W.Y.D. Review of the High-Temperature Oxidation of Iron and Carbon Steels in Air or Oxigen. Oxid. Met. 2003, 59, 433–468. [Google Scholar] [CrossRef]
- Kveder, A. (Ed.) Metalurški Priročnik (Handbook of Metallurgy, in Slovenian); Tehniška Založba Slovenije: Ljubljana, Slovenia, 1972; pp. 232, 233, 759. [Google Scholar]
- Liščić, B. Steel Heat Treatment. In Steel Heat Treatment Handbook, 2nd ed.; Totten, G.E., Ed.; CRC Press-Taylor & Francis Group: Boca Raton, FL, USA, 2007; pp. 277–414. ISBN 987-0-8493-8455-4. [Google Scholar]
- Gontarev, V. Teorija Metalurških Procesov (Theory of Metallurgical Processes, in Slovenian); Univerza v Ljubljani, NTF-Oddelek Za Materiale in Metalurgijo: Ljubljana, Slovenia, 2005; pp. 27–32. ISBN 961-6047-30-2. [Google Scholar]
- Naumann, F.K. Das Buch der Schadensfälle; Dr. Riederer-Verlag GmbH: Stuttgart, Germany, 1976; p. 181. [Google Scholar]
- Rose, A.; Peter, W.; Strassburg, W.; Rademacher, L. Atlas zur Wärmebehandlung der Stähle-Teil II; Verlag Stahleisen M.B.H.: Düsseldorf, Germany, 1961; p. II-101A. [Google Scholar]
- Vander Voort, G.F. Understanding and Measuring Decarburization. Adv. Mat. Process. 2015, 173, 22–27. [Google Scholar]
- Gegner, J. Diffusion Modelling of Decarburization Microhardness-Distance Profiles. Available online: https://www.ariel.ac.il/sites/conf/mmt/MMT-2006/Service_files/papers/Session_1/1-168_di.pdf (accessed on 28 April 2017).
- Choi, S.; van der Zwaag, S. Prediction of Decarburized Ferrite Depth of Hypoeutectoid Steel with Simultaneous Oxidation. ISIJ Int. 2012, 52, 549–558. [Google Scholar] [CrossRef] [Green Version]
- Cioffi, R.D. A Comparison Study on Depth of Decarburization and the Role of Stable Carbide Forming Elements in 1075 Plain Carbon Steel and 440A Stainless Steel; Rensselaer Polytechnic Institute, Department of Materials Science and Engineering: New York, NY, USA, 2012. [Google Scholar]
- Choi, S.; Lee, Y. An Approach to predict the Depth of the Decarburized Ferrite Layer of Spring Steel Based on Measured Temperature History of material during Cooling. ISIJ Int. 2014, 54, 1682–1689. [Google Scholar] [CrossRef]
- Birks, N. Mechanism of decarburization. In Decarburization; ISI Publication 133; The Iron and Steel Institute: London, UK, 1970; pp. 1–12. ISBN 0900497165. [Google Scholar]
- Wells, C.; Batz, W.; Mehl, R.F. Diffusion Coefficient of Carbon in Austenite. J. Met. 1950, 188, 553–560. [Google Scholar] [CrossRef]
- Bhadeshia, H.K.D.H.; Honeycombe, R.W.K. Steels Microstructure and Properties, 3rd ed.; Elsevier: Oxford, UK, 2006; pp. 11–12. ISBN 978-0-750-68084-4. [Google Scholar]
- Callister, W.D., Jr. Material Science and Engineering-An Introduction, 7th ed.; John Wiley & Sons, Inc.: New York, NY, USA, 2007; p. 119. ISBN 978-0-471-73696-7. [Google Scholar]
- Porter, D.A.; Easterling, K.E. Phase Transformations in Metals and Alloys, 2nd ed.; Chapman & Hall: London, UK, 1992; pp. 7, 74. ISBN 0-412-450030-5. [Google Scholar]
- Error Function Table. ECE 3610 Engineering Probability & Statistics. Available online: https://www.geophysik.uni-muenchen.de/Members/mohr/materials/ComputationalGeophysics2017/?searchterm=Function%20Table (accessed on 16 May 2017).
- Ericsson, T. Principles of Heat Treating of Steels. In ASM Handbook-Vol. 4: Heat Treating; Lampman, S.R., Zorc, T.B., Eds.; ASM International®: Materials Park, OH, USA, 2009; pp. 3–19. ISBN 978-0-87170-379-8. [Google Scholar]
- CEN Brussels, B. Standard EN 10083-2/2006: Steels for Quenching and Tempering—Part 2: Technical Delivery Conditions for Non Alloy Steels. ICS 77.140.10. 2006. [Google Scholar]
- Jaason, K.; Peetsalu, P.; Saarna, M.; Kulu, P.; Beilmann, J. Decarburisation Effect on Hardened Strip Steel Fastening Components. Mat. Sci. 2016, 22, 148–152. [Google Scholar] [CrossRef]









| Equation | Ta (°C/K) | 760/1033 | 850/1123 | 950/1223 | 1100/1373 |
|---|---|---|---|---|---|
| (3) | DCγ (cm2/s) | 2.236×10−8 | 7.880×10−8 | 2.570×10−7 | 1.096×10−6 |
| (4) | DCγ (cm2/s) | 7.546×10−9 | 3.000×10−8 | 1.097×10−7 | 5.382×10−7 |
| Ta (°C) | ta (s) | dm | xA | / | x0.884 | 0.884 | |||
|---|---|---|---|---|---|---|---|---|---|
| 760 | 1800 | 31 | 110 | 79 | 50 | 50 | 1.580 | 82;118 * | 51;87 * |
| 3600 | 43 | 156 | 113 | 120 | 120 | 0.942 | 116;167 * | 73;124 * | |
| 7200 | 61 | 221 | 160 | 150 | 150 | 1.067 | 164;236 * | 103;175 | |
| 850 | 1800 | 67 | 216 | 149 | 160 | 80 | 0.931 | 163 | 96 |
| 3600 | 95 | 305 | 210 | 3 | 3 | 70.00 | 231 | 136 | |
| 7200 | 134 | 432 | 298 | 280 | 70 | 1.064 | 326 | 192 | |
| 950 | 1800 | 141 | 337 | 196 | 200 | 40;75 # | 0.980 | 312 | 171 |
| 3600 | 199 | 477 | 278 | 260 | 60;100 # | 1.069 | 441 | 242 | |
| 7200 | 282 | 674 | 392 | 400 | 100;150 # | 0.980 | 624 | 342 | |
| 1100 | 1800 | 350 | 1027 | 677 | 650 | 200 # | 1.041 | 691 | 341 |
| 3600 | 477 | 1452 | 975 | 1000 | 260 # | 0.975 | 977 | 500 | |
| 7200 | 699 | 2054 | 1355 | 1400 | 300 # | 0.968 | 1382 | 683 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zorc, M.; Nagode, A.; Burja, J.; Kosec, B.; Zorc, B. Surface Decarburization of the Hypo-Eutectoid Carbon Steel C45 during Annealing in Steady Air at Temperatures T > AC1. Metals 2018, 8, 425. https://doi.org/10.3390/met8060425
Zorc M, Nagode A, Burja J, Kosec B, Zorc B. Surface Decarburization of the Hypo-Eutectoid Carbon Steel C45 during Annealing in Steady Air at Temperatures T > AC1. Metals. 2018; 8(6):425. https://doi.org/10.3390/met8060425
Chicago/Turabian StyleZorc, Matija, Aleš Nagode, Jaka Burja, Borut Kosec, and Borut Zorc. 2018. "Surface Decarburization of the Hypo-Eutectoid Carbon Steel C45 during Annealing in Steady Air at Temperatures T > AC1" Metals 8, no. 6: 425. https://doi.org/10.3390/met8060425







