1. Introduction
Over the past few years, materials based on intermetallic compounds are increasingly used thanks to their wide range of properties. For example, shape memory alloys (e.g., NiTi) are very attractive, as well as materials for high-temperature use, such as NiAl or TiAl. Other examples are FeAl or Fe
3Al intermetallics, which are used in strongly oxidizing or sulphidizing environments in combination with high temperatures [
1,
2,
3,
4]. Intermetallic compounds based on Fe-Al are considered to be very promising materials for industrial applications due to their low cost and high specific strength combined with excellent creep resistance, as well as their oxidation resistance at high temperatures [
2,
5,
6].
Fe-Al-Si system is of high interest because iron is the most common impurity in Al alloys, while Si is used as an alloying element [
7]. Impurities come to Al mainly during recycling, when some iron-containing material cannot be magnetically separated from Al completely [
8]. Within cast Al-Si alloys, Fe and Si form hard and brittle ternary phases, which influence corrosion and wear resistance [
7]. One author´s previous work [
4] showed that the Fe-Al-Si phases need not be considered as detrimental phases in Al alloys, but can instead be used for advantages in high-temperature service, because the addition of Si to binary Fe-Al system improves oxidation resistance and thermal stability [
9,
10]. Based on this fact, a completely new grade of Fe-Al-Si alloys has been developed at UCT Prague during the past few years. The Fe-Al-Si alloy containing 20 wt. % of Al and 20 wt. % of Si was determined to be the most oxidation-resistant composition [
10].
While the attractive properties of Fe-Al and Fe-Al-Si alloys are becoming increasingly evident, the technology is still insufficient to process them reliably and efficiently in order to maximize their mechanical properties. The already tested or practically applied methods include wire-arc additive manufacturing [
11], air/vacuum induction melting [
12], electroslag remelting [
13], and hot pressing [
14,
15]. Mechanical alloying (MA) is a promising route for preparation of Fe-Al and Fe-Al-Si alloys. The preparation via MA, usually carried out as high-energy ball milling, is a powder processing technique that allows the preparation of alloys with the final phase composition starting from blended elemental powder mixtures. The MA leads to the reduction of the input powder size thanks to the high kinetic energy of the milling balls. Furthermore, other phenomena also occur during the process, such as local welding of particles by plastic deformation, friction forces and diffusion, structure refinement due to severe plastic deformation, as well as the formation of solid solutions and chemical compounds (intermetallics) [
16,
17]. Products prepared by MA under appropriate conditions usually possess an ultrafine-grained nanocrystalline or amorphous microstructure [
18]. Since grain size reduction often improves physical and mechanical properties in many cases, the nanocrystalline materials are very attractive for many applications. The nanocrystalline structure may result in higher hardness, strength or even ductility of brittle materials which results in better ductility of intermetallics [
15]. Recently, we have developed an innovative MA process of ultra-high energy mechanical alloying [
19]. Our development is based on the following facts:
This means that the ultra-high energy mechanical alloying process combines the advantages of mechanical alloying and self-propagating high-temperature synthesis, where the intermetallics are prepared by thermal activation. During this process we are able to produce intermetallics in 2–4 h [
19]. This is more rapid than the milling times needed with other processing parameters reported in the literature to achieve similar phase composition and microstructure [
20,
21,
22,
23]. The above defined ultra-high energy mechanical alloying process is more efficient than the use of dedicated high-energy milling devices using 1200–2000 rpm [
19].
For the estimation of the energy supplied to the powder during the milling process we can use the following statements: When the energy in mechanical alloying process is calculated, only the kinetic energy of the balls is taken into account. The energy caused by each collision in conventional mechanical alloying processes was calculated to be 10–400 J/hit or 0.01–0.8 W/g of the balls, depending on the degree of filling (or ball-to powder ratio) and rotational velocity [
24,
25,
26]. This leads to the generation of the total energy of approx. 200 W in the case of our milling device operated under standard conditions. This could lead to the increase of temperature to approx. 300 °C [
26].
However, in our milling process we don’t avoid friction, as we apply a low amount of powder and use no lubricant. There are studies which show that some intermetallics can be generated in the friction mode only [
27], because the generated temperature is a key factor for the formation of intermetallics. From our previous results with our ultra-high energy mechanical alloying process, we can conclude that the temperature localized in the contact point between powder, ball and wall exceeds 650 °C, because this temperature is needed for the formation of some intermetallics as the Ni-Ti and Fe-Al binary phases prepared in our previous paper [
19]. The required temperature was determined by DTA heating curve of the powder mixtures of pure metals previously [
16]. Hence it can be expected that the energy level including friction and the use of high ball-to-powder ratio is much more than two times higher (approx. 400–500 W). Current high speed mills reach the estimated energy usage of approx. 400 W [
24].
Recently, there were published several studies about the application of conventional mechanical alloying in Fe-Al binary system. Krasnowski et al. [
21], in their paper on Fe- 50 at. % Al, described changes in the phase composition of the mechanically alloyed powder, and consequently the influence of the milling time on the crystallite size, the lattice strain and the lattice parameter. Also, Haghighi et al. [
22] studied the mentioned system and the grain size as well as the lattice parameter depending on the milling time. Both teams, though working with different milling conditions, observed the same trend that the powder particle size increased at the initial stage, but then it began to decrease and in the end it remained constant. The reason for the decrease of the particle size is the deformation strengthening, which causes the increase of the strength and the decrease of plasticity, as well as the formation of intermetallics, which substantially increase brittleness.
This work aims to optimize conditions of innovative ultra-high energy mechanical alloying process for the preparation of Fe-Al-Si alloys and to describe the chemical processes during the mechanical alloying of these alloys in dependence on the content of Al and Si. The thermal stability of the powders after mechanical alloying is described in order to predict the sintering behavior.
3. Results and Discussion
In the case of FeAl32 binary alloy, the mechanical alloying led to the temporary formation of FeAl
3 phase after 0.5 h of milling [
28]. This phase, as well as Fe
2Al
5 are known as transient phases formed by Fe + Al reaction in reactive sintering synthesis of FeAl phase [
29]. It was then converted to FeAl ordered phase (B2 structure) by the reaction with iron and only the FeAl phase was detected after 1 h of milling. Further prolongation of the milling did not cause any changes in the phase composition [
28].
The addition of Si slowed down the phase development. After one hour of milling, there were still detectable amounts of Al and Si in the FeAl35Si5 alloy, see
Figure 1 and
Table 1. The mechanical alloying for this powder mixture started by the formation of Fe
2Al
5 phase, as can be seen in the XRD pattern acquired after milling for 0.5–2 h. This phase formed instead of FeAl
3, probably due to the lower ratio of Al to Fe in FeAl35Si5 than in the FeAl32 alloy. It also could have happened due to the fact that the presence of silicon extending the region of stability of the Fe
2Al
5 phase, as presented in the Fe-Al-Si phase diagram [
30]. The single-phase FeAl alloy is obtained after milling for 6 h (
Figure 1 and
Table 1).
At the initial stage of milling of FeAl30Si10 powder blend, only the Fe
2Si phase formed after 1 h (
Figure 2 and
Table 2). It implies that the greater addition of Si inhibits the formation of the Fe
2Al
5 phase. The unreacted initial components (Al and Si) were found even after 2 h of milling together with the formed FeAl phase. Longer milling durations produced single-phase FeAl. The same conclusion was made previously during observation of the mechanism of self-propagating high-temperature synthesis (SHS), where the formation of the Fe
2Al
5 phase was completely suppressed by the addition of a reasonable amount of Si [
10]. The formation of Fe
2Al
5 phase during SHS is not desirable because it generates higher porosity in Fe-Al alloys due to enormous changes in lattice parameters during the chemical reaction from pure elemental powders to Fe
2Al
5 and then to FeAl [
29,
31]. The consequences of the presence of this phase in mechanical alloying are unknown. It could be expected that it causes internal porosity of the powder by the same mechanism as in reactive sintering. However, this porosity could be lowered by mechanical alloying itself (healing of internal porosity by plastic deformation) or during subsequent pressure-assisted sintering of the mechanically alloyed powder.
In the case of FeAl20Si20 alloy, mechanical alloying for 1 h did not yield any intermetallics. As the process continued, the Fe
3Si, FeSi and FeAl phases were produced after 2 h and remained unchanged up to 6 h of MA. Firstly, they coexisted with unreacted Fe and Si. After 4 h and longer milling procedure, the unreacted components were not detected by XRD (
Figure 3 and
Table 3). The phase composition of FeAl20Si20 alloy powder after mechanical alloying is completely different from the Fe-Al-Si equilibrium phase diagram, where the ternary phases are expected for alloy of this composition [
32,
33]. The reason why binary phases are formed instead probably lies in the fact that the powders of two elements can get easily into contact during milling. In addition, mechanical alloying usually increases the mutual solubility of the elements in solid solutions, as well as in secondary solid solutions (i.e., solution of elements in an intermetallic compound) [
34]. Therefore the silicides and aluminides might be substituted by Al and Si, respectively. This fact was proved by the shift of diffraction lines of all phases in milled powders, as compared with the reference patterns in PDF4 database (
Table 4). In the case of the FeAl phase, the shift to lower values of the lattice parameter indicates the partial substitution of Al and/or Fe by Si, because Si has a lower atomic radius than both Al and Fe. The alloy´s composition makes is likely that Si substitutes for Fe in the FeAl phase. In FeAl20Si20, the lattice parameter of the Fe
3Si phase is comparable with the table value or a bit higher at certain stages of the MA process, which indicates that Si is probably substituted partially by Al with higher atomic radius than Si. However, this hypothesis cannot be verified using the applied equipment due to ultrafine microstructure of the alloys.
The microstructural development of powders during mechanical alloying is presented in
Figure 4,
Figure 5 and
Figure 6. It is evident that after the first half hour of mechanical alloying the microstructure is composed of welded lamellae of starting powders—Fe, Al. Si is very fine because of its brittleness and it is firmly anchored between the lamellae. The phases in early stages of the process are relatively coarse and hence they can be identified by EDS analysis, see
Table 5. The lamellae are mutually enriched by the other elements as a result of diffusion promoted by the temperature increase due to friction and collisions during milling. With the increasing duration of the process, the width of the lamellae decreases, and hence the diffusion path of the elements is shorter and a homogeneous structure is created as a result. The final microstructure (
Figure 4,
Figure 5 and
Figure 6c,d) is very fine, and therefore the EDS detector cannot be used to recognize the individual intermetallic phases. After longer process duration (
Figure 4d,
Figure 5d and
Figure 6d), the contamination by chromium from the stainless steel milling vial and balls could be observed as the white particles in the powder. In
Figure 4, the development of porosity is visible during longer process durations (
Figure 4c,d). It confirms the above stated hypothesis that the temporary formation of Fe
2Al
5 phase and its transformation to FeAl phase with a totally different crystal structure causes the evolution of pores. The alloys FeAl30Si10 and FeAl20Si20 (
Figure 5 and
Figure 6) tend to crack during milling due to the higher brittleness of silicon-rich phases, which appear during the process (
Figure 2 and
Figure 3). The SEM images of the milled powders in
Figure 4,
Figure 5 and
Figure 6 also confirm that the silicon slows down the milling process because while the lamellar structure disappears completely in FeAl35Si5 and FeAl30Si10 powder blends in 2 h of mechanical alloying, in FeAl20Si20 the lamellar structure persists partially for up to 4 h of milling.
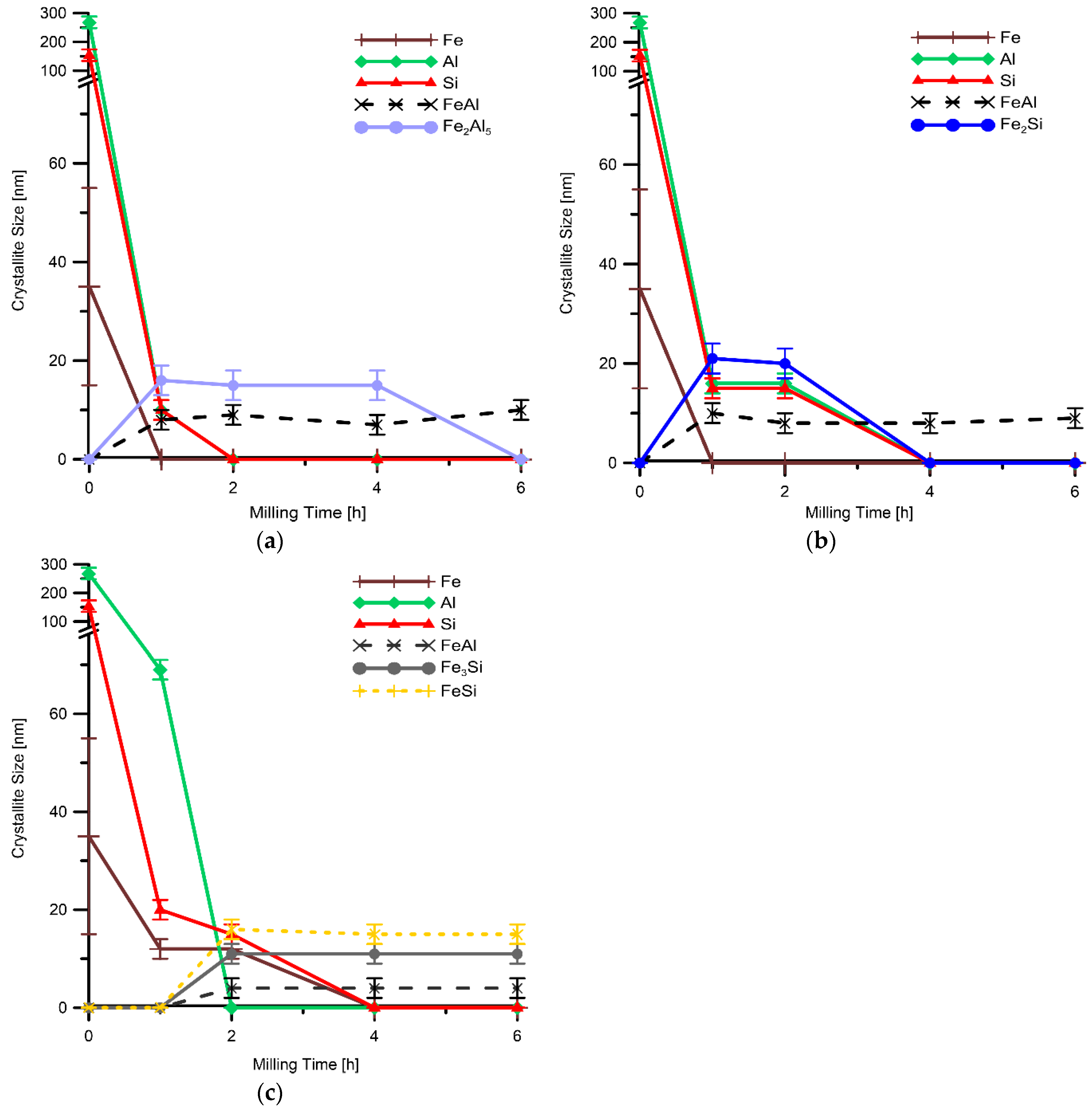
The crystallite size determined by XRD and calculated by Rietveld method showed that the structure of all elemental powders (Fe, Al, Si) has been significantly refined at the very beginning of mechanical alloying. Once the intermetallics are formed their crystallite size is then almost independent of the milling duration, reaching 10−20 nm (see
Figure 7a−c). The crystallite size of the transient Fe
2Al
5 phase is a bit higher than the one of FeAl phase. The reason for that is probably the fact that Fe
2Al
5 phase exhibits almost no plasticity in contrast to B2 FeAl phase. Due to plasticity, the grain refinement by severe plastic deformation during milling could occur, leading to finer structure. The same trend is observable for iron silicides, where lower ductility could also be expected when the content of Si in the compound increases. However, the trend is less significant, probably due to the generally lower plasticity of silicides than aluminides.
The average particle size of powders before milling is presented in
Figure 8. Between 0 and 2 h an increase in the particle size occurred, followed by a decrease of the particle size due to two contradictory processes. The formation of cold welds between particles led to an increase in particle size, while the cracking of powder particles is caused by deformation strengthening. Between 2 and 4 h of mechanical alloying, the size of the powder particles decreased because of cracking as a result of the formation of brittle intermetallic phases and continual deformation of powders. After exceeding 4 h of milling, the particle size remained almost constant, showing a balance between continual cracking and cold welding.
In order to determine the thermal stability of the mechanically alloyed powders and to predict the subsequent sintering behavior, DTA of the milled powders has been carried out. Using the DTA analysis, the possible problems during sintering, such as phase transformations which could cause the volume changes, the appearance of unwanted phases with undesirable properties or partial melting of the alloy due to the existence of metastable lower-melting phases could be predicted. These results have general validity for the optimization of further consolidation by any method including spark plasma sintering, hot isostatic pressing or even free sintering. The heating curves presented in
Figure 9 exhibit weak exothermic effects at approx. 500–630 °C and 800–1050 °C depending on the powder composition. Therefore, the powders of all alloys after mechanical alloying were annealed at 630 and 1030 °C in order to explain the observed thermal effects. It was found that the reaction below 630 °C is associated with the formation of Fe
2Al
5 phase (
Table 6). This intermetallic compound probably formed because of the depletion of the silicides by Al during annealing. As stated above, the binary silicides and aluminides are supersaturated by Al and Si, respectively. This depletion could lead to the formation of another aluminide phase, which has been previously determined as a typical product of a direct reaction of iron with Al [
29]. On the other hand, the reactions at the temperatures close to 1030 °C are connected with the formation of Fe
3Si silicide in FeAl35Si5 and FeAl30Si10 alloys, probably due to the Si escape from the aluminide phase. In the case of the FeAl20Si20 alloy, ternary phase Fe
2Al
3Si
3 forms in the powder during annealing at 1030 °C. From these findings, it can be predicted that the porosity will be formed if free sintering would be applied due to the transient Fe
2Al
5 phase, which has a big misfit of lattice parameters with the other Fe-Al and Fe-Si phases [
36]. The pressure-assisted sintering, such as hot isostatic pressing or spark plasma sintering can be highly recommended. The highly brittle behavior could be expected in the case of the FeAl20Si20 alloy due to the presence of Fe-Al-Si ternary phase in the compact.