Fe–Si–Al Coatings with Stable Wear Resistance Prepared by Laser Cladding Industrial Wastes
Abstract
:1. Introduction
2. Materials and Methods
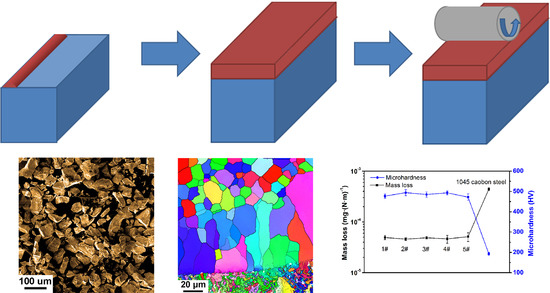
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Fu, Z.K.; Ding, H.H.; Wang, W.J.; Liu, Q.Y.; Guo, J.; Zhu, M.H. Investigation on microstructure and wear characteristic of laser cladding Fe-based alloy on wheel/rail materials. Wear 2015, 330, 592–599. [Google Scholar] [CrossRef]
- Guo, Y.X.; Shang, X.J.; Liu, Q.B. Microstructure and properties of in-situ tin reinforced laser cladding CoCr2FeNiTix high-entropy alloy composite coatings. Surf. Coat. Technol. 2018, 344, 353–358. [Google Scholar] [CrossRef]
- Mao, J.; Feng, A.; Cheng, B.; Li, Z.; Wu, H.; Huang, Y.; Zhang, H. Microstructure and friction and wear properties of Fe-based alloy coatings by laser cladding. Hot Work. Technol. 2017, 46, 139–142. [Google Scholar]
- Hua, H.; Ting, Z.; Ma, M.; Liu, W. Microstructure and wear resistance of laser cladding particulate reinforced Fe-based composite coating on railway steel. J. Laser Appl. 2017, 29, 022503. [Google Scholar]
- Kumar, S.; Mandal, A.; Das, A.K.; Dixit, A.R. Parametric study and characterization of AlN–Ni–Ti6Al4V composite cladding on titanium alloy. Surf. Coat. Technol. 2018, 349, 37–49. [Google Scholar] [CrossRef]
- Wang, K.M.; Chang, B.H.; Lei, Y.P.; Fu, H.G.; Lin, Y.H. Effect of cobalt on microstructure and wear resistance of Ni-based alloy voating fabricated by laser cladding. Metals 2017, 7, 12. [Google Scholar] [CrossRef]
- Yang, J.; Huang, J.H.; Fan, D.Y.; Chen, S.H. Microstructure and wear properties of Fe–6wt.%Cr–0.55wt.%C–xwt.%Nb laser cladding coating and the mechanism analysis. Mater. Des. 2015, 88, 1031–1041. [Google Scholar] [CrossRef]
- Bai, L.; Le, G.; Liu, X.; Li, J.; Xi, S.; Li, X. Grain morphologies and microstructures of laser melting deposited V-5Cr-5Ti alloys. J. Alloys Compd. 2018, 745, 716–724. [Google Scholar] [CrossRef]
- Chen, J.L.; Zhou, Y.J.; Shi, C.; Mao, D.H. Microscopic analysis and electrochemical behavior of Fe-based coating produced by laser cladding. Metals 2017, 7, 9. [Google Scholar] [CrossRef]
- Weng, Z.K.; Wang, A.H.; Wang, Y.Y.; Xiong, D.H.; Tang, H.Q. Diode laser cladding of Fe–based alloy on ductile cast iron and related interfacial behavior. Surf. Coat. Technol. 2016, 286, 64–71. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, C.S.; Qian, S.N.; Yu, Q.; Dong, C. Microstructure and wear resistance of laser-clad (Co, Ni)(61.2)B26.2Si7.8Ta4.8 coatings. Metals 2017, 7, 7. [Google Scholar] [CrossRef]
- Zhang, H.; Chong, K.; Zhao, W.; Sun, Z.P. Effects of pulse parameters on in-situ Ti-V carbides size and properties of Fe-based laser cladding layers. Surf. Coat. Technol. 2018, 344, 163–169. [Google Scholar] [CrossRef]
- Li, G.; Cui, Y.; Zhang, N.; Wang, X.; Xie, J.L. The precipitation in annealing and its effect on permittivity of Fe-Si-Al powders. Physica B 2016, 481, 1–7. [Google Scholar] [CrossRef]
- Fernandes, L.; Silva, F.J.G.; Paiva, O.C.; Baptista, A.; Pinto, G. Minimizing the adhesion effects in food packages forming by the use of advanced coatings. Procedia Manuf. 2018, 17, 886–894. [Google Scholar] [CrossRef]
- Fernandes, L.; Silva, F.J.G.; Andrade, M.F.; Alexandre, R.; Baptista, A.P.M.; Rodrigues, C. Improving the punch and die wear behavior in tin coated steel stamping process. Surf. Coat. Technol. 2017, 332, 174–189. [Google Scholar] [CrossRef]
- Krajewski, W.K.; Greer, A.L.; Krajewski, P.K. Trends in the development of high-aluminium zinc alloys of stable structure and properties. Arch. Metall. Mater. 2013, 58, 845–847. [Google Scholar] [CrossRef]
- Kou, S. Welding Metallurgy; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2003. [Google Scholar]
- Li, Y.J.; Dong, S.Y.; Yan, S.X.; He, P.; Xu, B.S. Phase evolution of ductile iron during laser cladding processing. Surf. Coat. Technol. 2018, 339, 37–47. [Google Scholar] [CrossRef]
- Zhao, X.; Yang, X.L.; Jing, T.F. Effect of initial microstructure on warm deformation behavior of 45 steel. J. Iron Steel Res. Int. 2012, 19, 75–78. [Google Scholar] [CrossRef]
- Lu, S.Y.; Yao, K.F.; Chen, Y.B.; Wang, M.H.; Liu, X.; Ge, X.Y. The effect of tempering temperature on the microstructure and electrochemical properties of a 13wt.% Cr-type martensitic stainless steel. Electrochim. Acta 2015, 165, 45–55. [Google Scholar] [CrossRef]
- Wang, Y.F.; Li, G.; Wang, C.S.; Xia, Y.L.; Sandip, B.; Dong, C. Microstructure and properties of laser clad Zr-based alloy coatings on Ti substrates. Surf. Coat. Technol. 2004, 176, 284–289. [Google Scholar] [CrossRef]
- Lee, Y.S.; Ha, S.; Park, J.H.; Lee, S.B. Structure-dependent mechanical behavior of copper thin films. Mater. Charact. 2017, 128, 68–74. [Google Scholar] [CrossRef]
- Raynor, D.; Silcock, J.M. Strengthening mechanisms in γ′ precipitating alloys. Met. Sci. J. 1970, 4, 121–130. [Google Scholar] [CrossRef]
- Andrade, M.F.C.; Martinho, R.P.; Silva, F.J.G.; Alexandre, R.J.D.; Baptista, A.P.M. Influence of the abrasive particles size in the micro-abrasion wear tests of TiAlSiN thin coatings. Wear 2009, 267, 12–18. [Google Scholar] [CrossRef] [Green Version]
- Pereira, D.; Gandra, J.; Pamies-Teixeira, J.; Miranda, R.M.; Vilaca, P. Wear behaviour of steel coatings produced by friction surfacing. J. Mater. Process. Technol. 2014, 214, 2858–2868. [Google Scholar] [CrossRef]
- Deng, X.K.; Zhang, G.J.; Wang, T.; Ren, S.; Bai, Z.L.; Cao, Q. Investigations on microstructure and wear resistance of Fe-Mo alloy coating fabricated by plasma transferred arc cladding. Surf. Coat. Technol. 2018, 350, 480–487. [Google Scholar] [CrossRef]
- Chen, X.; Qin, X.; Zhu, Z.; Gao, K. Microstructural evolution and wear properties of the continual local induction cladding NiCrBSi coatings. J. Mater. Process. Technol. 2018, 262, 257–268. [Google Scholar] [CrossRef]
- Wang, Z. Microstructure and wear resistance of laser-cladding NiCrSiB coating. Spec. Cast. Nonferr. Alloys 2013, 33, 509–511. [Google Scholar]
- Liu, X.; Liu, H.; Wang, D.; Wang, E.P.; Liu, W.J.; Yao, K.F.; Chen, N. Metallic glass-strengthened thermoplastic elastomer composites. Physica E 2017, 90, 37–41. [Google Scholar] [CrossRef]









| Sample No. | Laser Powder (W) | Scan Speed (mm/min) | Heat Import (kJ) |
|---|---|---|---|
| Specimen 1 | 800 | 500 | 403 |
| Specimen 2 | 1000 | 500 | 468 |
| Specimen 3 | 1200 | 300 | 744 |
| Specimen 4 | 1200 | 500 | 432 |
| Specimen 5 | 1200 | 700 | 319 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, X.; Ma, B.-B.; Hu, L.-W.; Li, J.-F.; Qu, F.-S.; Le, G.-M.; Li, X.-Y. Fe–Si–Al Coatings with Stable Wear Resistance Prepared by Laser Cladding Industrial Wastes. Metals 2019, 9, 96. https://doi.org/10.3390/met9010096
Liu X, Ma B-B, Hu L-W, Li J-F, Qu F-S, Le G-M, Li X-Y. Fe–Si–Al Coatings with Stable Wear Resistance Prepared by Laser Cladding Industrial Wastes. Metals. 2019; 9(1):96. https://doi.org/10.3390/met9010096
Chicago/Turabian StyleLiu, Xue, Bin-Bin Ma, Li-Wei Hu, Jin-Feng Li, Feng-Sheng Qu, Guo-Min Le, and Xiu-Yan Li. 2019. "Fe–Si–Al Coatings with Stable Wear Resistance Prepared by Laser Cladding Industrial Wastes" Metals 9, no. 1: 96. https://doi.org/10.3390/met9010096
APA StyleLiu, X., Ma, B. -B., Hu, L. -W., Li, J. -F., Qu, F. -S., Le, G. -M., & Li, X. -Y. (2019). Fe–Si–Al Coatings with Stable Wear Resistance Prepared by Laser Cladding Industrial Wastes. Metals, 9(1), 96. https://doi.org/10.3390/met9010096