The Interactions between Polyphenols and Microorganisms, Especially Gut Microbiota
Abstract
1. Introduction
2. The Structure and Role of Polyphenols
3. The Impact of Polyphenols on Microorganisms and the Mechanism of Their Action
4. Mechanism of Antibacterial Activity of Polyphenols
- Reactions with proteins;
- Inhibition of nucleic acid synthesis by bacterial cells or DNA damage;
- Interaction with the bacterial cell wall or inhibition of cell wall formation;
- Alteration of cytoplasmic membrane function, such as modifications of the membrane permeability or fluidity, cytoplasmic membrane damage and—in the result—the membrane disruption;
- Inhibition of energy metabolism;
- Changes in cell attachment and inhibition of biofilm formation;
- Substrate and metal deprivation.
4.1. Reactions with Proteins
4.2. Inhibition of Bacterial DNA Synthesis and Interaction with Nucleic Acids
4.3. Interaction with the Bacterial Cell Wall or Inhibition of Cell Wall Formation
4.4. Alteration of Cytoplasmic Membrane Function
4.5. Inhibition of Energy Metabolism
4.6. The Inhibition of Biofilm Formation and Interfering with Bacterial Quorum Sensing
4.7. Substrate Deprivation
4.8. The Relationship between Polyphenols Structure and Antibacterial Activity
4.9. The Impact of Food Matrix on Polyphenol Activity
5. Polyphenols Biotransformation by Intestinal Bacteria
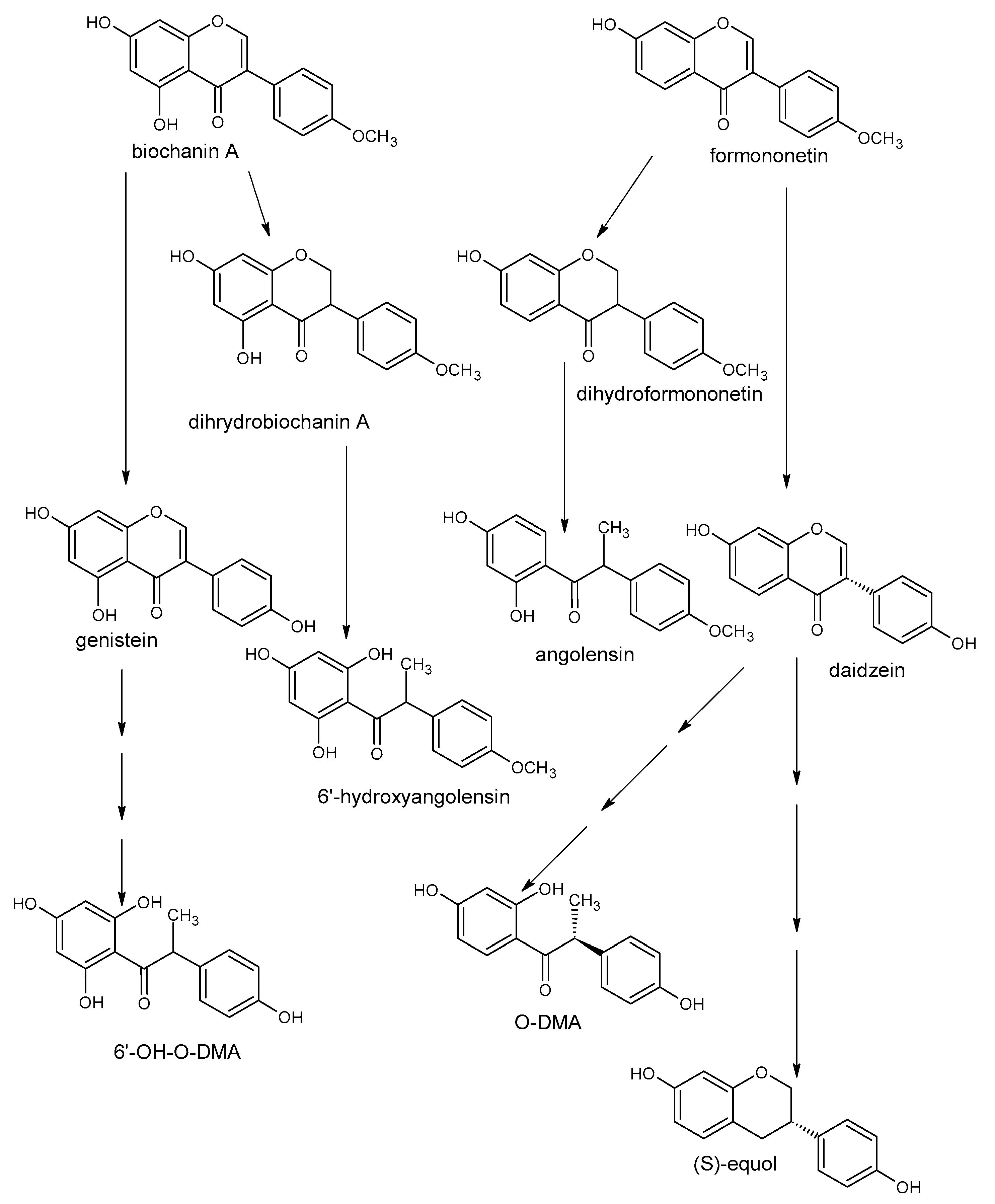
5.1. Isoflavonoids
5.2. Other Phytoestrogens
5.3. Bacterial Transformation of Anthocyanidins
5.4. Metabolism of Procyanidins and Catechins by Intestinal Bacteria
5.5. The Bacterial Metabolism of Flavones and Flavonols
5.6. Microbial Catabolism of Phenolic Acids
5.7. Bacterial Metabolism of Resveratrol and Curcumin
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hooper, L.V.; Gordon, J.I. Commensal host-bacterial relationships in the gut. Science 2001, 292, 1115–1118. [Google Scholar] [CrossRef]
- Zhu, B.; Wang, X.; Li, L. Human gut microbiome: The second genome of human body. Protein Cell 2010, 1, 718–725. [Google Scholar] [CrossRef]
- Qin, J.; Li, R.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, N.; Levenez, F.; Yamada, T.; et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 2010, 464, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Eckburg, P.B.; Bik, E.M.; Bernstein, C.N.; Purdom, E.; Dethlefsen, L.; Sargent, M.; Gill, S.R.; Nelson, K.E.; Relman, D.A. Diversity of the human intestinal microbial flora. Science 2005, 308, 1635–1638. [Google Scholar] [CrossRef]
- Lagier, J.-C.; Armougom, F.; Million, M.; Hugon, P.; Pagnier, I.; Robert, C.; Bittar, F.; Fournous, G.; Gimenez, G.; Maraninchi, M.; et al. Microbial culturomics: Paradigm shift in the human gut microbiome study. Clin. Microbiol. Infect. 2012, 18, 1185–1193. [Google Scholar] [CrossRef]
- Duda-Chodak, A.; Tarko, T.; Satora, P.; Sroka, P. Interaction of dietary compounds, especially polyphenols, with the intestinal microbiota: A review. Eur. J. Nutr. 2015, 54, 325–341. [Google Scholar] [CrossRef]
- Conlon, M.A.; Bird, A.R. The impact of diet and lifestyle on gut microbiota and human health. Nutrients 2015, 7, 17–44. [Google Scholar] [CrossRef]
- Thursby, E.; Juge, N. Introduction to the human gut microbiota. Biochem. J. 2017, 474, 1823–1836. [Google Scholar] [CrossRef]
- Hermon-Taylor, J. Gut pathogens: Invaders and turncoats in a complex cosmos. Gut Pathog. 2009, 1, 3. [Google Scholar] [CrossRef][Green Version]
- Acton, D.S.; Plat-Sinnige, M.J.; van Wamel, W.; de Groot, N.; van Belkum, A. Intestinal carriage of Staphylococcus aureus: How does its frequency compare with that of nasal carriage and what is its clinical impact? Eur. J. Clin. Microbiol. Infect. Dis. 2009, 28, 115–127. [Google Scholar] [CrossRef]
- Kamada, N.; Chen, G.Y.; Inohara, N.; Núñez, G. Control of pathogens and pathobionts by the gut microbiota. Nat. Immunol. 2013, 14, 685–690. [Google Scholar] [CrossRef]
- Rowland, I.; Gibson, G.; Heinken, A.; Scott, K.; Swann, J.; Thiele, I.; Tuohy, K. Gut microbiota functions: Metabolism of nutrients and other food components. Eur. J. Nutr. 2018, 57, 1–24. [Google Scholar] [CrossRef]
- Possemiers, S.; Bolca, S.; Verstraete, W.; Heyerick, A. The intestinal microbiome: A separate organ inside the body with the metabolic potential to influence the bioactivity of botanicals. Fitoterapia 2011, 82, 53–66. [Google Scholar] [CrossRef]
- Hervert-Hernández, D.; Goñi, I. Dietary polyphenols and human gut microbiota: A review. Food Rev. Int. 2011, 27, 154–169. [Google Scholar] [CrossRef]
- Man, A.W.C.; Zhou, Y.; Xia, N.; Li, H. Involvement of gut microbiota, microbial metabolites and interaction with polyphenol in host immunometabolism. Nutrients 2020, 12, 3054. [Google Scholar] [CrossRef]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.E.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly)phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid. Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef]
- Saarela, M.; Lähteenmäki, L.; Crittendenb, R.; Salminenc, S.; Mattila-Sandholm, T. Gut bacteria and health foods—The European perspective. Int. J. Food Microbiol. 2002, 78, 99–117. [Google Scholar] [CrossRef]
- Marchesi, J.R.; Adams, D.H.; Fava, F.; Hermes, G.D.A.; Hirschfield, G.M.; Hold, G.; Quraishi, M.N.; Kinross, J.; Smidt, H.; Tuohy, K.M.; et al. The gut microbiota and host health: A new clinical frontier. Gut 2016, 65, 330–339. [Google Scholar] [CrossRef]
- Cardona, F.; Andrés-Lacueva, C.; Tulipani, S.; Tinahones, F.J.; Queipo-Ortuño, M.I. Benefits of polyphenols on gut microbiota and implications in human health. J. Nutr. Biochem. 2013, 24, 1415–1422. [Google Scholar] [CrossRef]
- Górniak, I.; Bartoszewski, R.; Króliczewski, J. Comprehensive review of antimicrobial activities of plant flavonoids. Phytochem. Rev. 2019, 18, 241–272. [Google Scholar] [CrossRef]
- Piasecka, A.; Jedrzejczak-Rey, N.; Bednarek, P. Secondary metabolites in plant innate immunity: Conserved function of divergent chemicals. New Phytol. 2015, 206, 948–964. [Google Scholar] [CrossRef]
- Mao, X.; Xiao, X.; Chen, D.; Yu, B.; He, J. Tea and its components prevent cancer: A review of the redox-related mechanism. Int. J. Mol. Sci. 2019, 20, 5249. [Google Scholar] [CrossRef]
- Schaffer, S.; Asseburg, H.; Kuntz, S.; Muller, W.E.; Eckert, G.P. Effects of polyphenols on brain ageing and Alzheimer’s disease: Focus on mitochondria. Mol. Neurobiol. 2012, 46, 161–178. [Google Scholar] [CrossRef]
- Guasch-Ferré, M.; Merino, J.; Sun, Q.; Fitó, M.; Salas-Salvadó, J. Dietary polyphenols, mediterranean diet, prediabetes, and Type 2 diabetes: A narrative review of the evidence. Oxid. Med. Cell. Longev. 2017, 2017, 1–16. [Google Scholar] [CrossRef]
- Kim, Y.; Keogh, J.B.; Clifton, P.M. Polyphenols and glycemic control. Nutrients 2016, 8, 17. [Google Scholar] [CrossRef]
- Öztürk, E.; Arslan, A.K.K.; Yerer, M.B.; Bishayee, A. Resveratrol and diabetes: A critical review of clinical studies. Biomed. Pharmacother. 2017, 95, 230–234. [Google Scholar] [CrossRef]
- Roleira, F.M.F.; Tavares-da-Silva, E.J.; Varela, C.L.; Costa, S.C.; Silva, T.; Garrido, J.; Borges, F. Plant derived and dietary phenolic antioxidants: Anticancer properties. Food Chem. 2015, 183, 235–258. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Moustaid-Moussa, N.; Chen, L.; Mo, H.; Shastri, A.; Su, R.; Bapat, P.; Kwun, I.; Shen, C.-L. Novel insights of dietary polyphenols and obesity. J. Nutr. Biochem. 2014, 25, 1–18. [Google Scholar] [CrossRef]
- Fraga, C.G.; Croft, K.D.; Kennedy, D.O.; Tomás-Barberán, F.A. The effects of polyphenols and other bioactives on human health. Food Funct. 2019, 10, 514–528. [Google Scholar] [CrossRef] [PubMed]
- Oppedisano, F.; Maiuolo, J.; Gliozzi, M.; Musolino, V.; Carresi, C.; Nucera, S.; Scicchitano, M.; Scarano, F.; Bosco, F.; Macrì, R.; et al. The potential for natural antioxidant supplementation in the early stages of neurodegenerative disorders. Int. J. Mol. Sci. 2020, 21, 2618. [Google Scholar] [CrossRef]
- Silva, R.F.M.; Pogaĉnik, L. Polyphenols from food and natural products: Neuroprotection and safety. Antioxidants 2020, 9, 61. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Yau, Y.F.; Leung, K.S.; El-Nezami, H.; Lee, J.C.-Y. Interaction of polyphenols as antioxidant and anti-inflammatory compounds in brain–liver–gut axis. Antioxidants 2020, 9, 669. [Google Scholar] [CrossRef] [PubMed]
- Stalmach, A.; Mullen, W.; Nagai, C.; Crozier, A. On-line HPLC analysis of the antioxidant activity of phenolic compounds in brewed, paper-filtered coffee. Braz. J. Plant Physiol. 2006, 18, 253–262. [Google Scholar] [CrossRef]
- Ma, J.-N.; Ma, C.-M. Antifungal inhibitory activities of caffeic and quinic acid derivatives. In Coffee in Health and Disease Prevention; Preedy, V.R., Ed.; Elsevier Inc.; Academic Press: London, UK, 2015; pp. 635–641. [Google Scholar] [CrossRef]
- Heleno, S.A.; Martins, A.; Queiroz, M.J.R.P.; Ferreira, I.C.F.R. Bioactivity of phenolic acids: Metabolites versus parent compounds: A review. Food Chem. 2015, 173, 501–513. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Pan, Q.H.; Shi, Y.; Duan, C.Q. Biosynthesis and genetic regulation of proanthocyanidins in plants. Molecules 2008, 13, 2674–2703. [Google Scholar] [CrossRef]
- Clifford, M.N.; Jaganath, I.B.; Ludwig, I.A.; Crozier, A. Chlorogenic acids and the acyl-quinic acids: Discovery, biosynthesis, bioavailability and bioactivity. Nat. Prod. Rep. 2017, 34, 1391–1421. [Google Scholar] [CrossRef]
- Tasdemir, D.; Lack, G.; Brun, R.; Ruedi, P.; Scapozza, L.; Perozzo, R. Inhibition of Plasmodium falciparum fatty acid biosynthesis: Evaluation of FabG, FabZ, and FabI as drug targets for flavonoids. J. Med. Chem. 2006, 49, 3345–3353. [Google Scholar] [CrossRef]
- Routray, W.; Orsat, V. Blueberries and their anthocyanins: Factors affecting biosynthesis and properties. Comprehen. Rev. Food Sci. Food Saf. 2011, 10, 303–320. [Google Scholar] [CrossRef]
- Naoi, M.; Wu, Y.; Shamoto-Nagai, M.; Maruyama, W. Mitochondria in neuroprotection by phytochemicals: Bioactive polyphenols modulate mitochondrial apoptosis system, function and structure. Int. J. Mol. Sci. 2019, 20, 2451. [Google Scholar] [CrossRef]
- Qabaha, K.I. Antimicrobial and free radical scavenging activities of five Palestinian medicinal plants. Afr. J. Tradit. Complement. Altern. Med. 2013, 10, 101–108. [Google Scholar] [CrossRef][Green Version]
- Veličković, D.T.; Randjelović, N.V.; Ristić, M.S.; Veličković, A.S.; Šmelcerović, A.A. Chemical constituents and antimicrobial activity of the ethanol extracts obtained from the flower, leaf and stem of Salvia officinalis L. J. Serb. Chem. Soc. 2003, 68, 17–24. [Google Scholar] [CrossRef]
- Bozyel, M.E.; Şenturan, M.; Benek, A.; Bozyel, E.M.; Canli, K.; Altuner, E.M. In vitro antimicrobial activity screening of Heliotropium europaeum against wide range of microorganisms and multi drug resistant (MDR) bacteria. Eur. J. Biomed. Pharmac. Sci. 2019, 6, 113–117. [Google Scholar]
- Al-Juraifani, A.A. Antimicrobial activity of some medicinal plants used in Saudi Arabia. CJPAS 2011, 5, 509–1512. [Google Scholar]
- Al-Bayati, F.A.; Sulaiman, K.D. In vitro antimicrobial activity of Salvadora persica L. extracts against some isolated oral pathogens in Iraq. Turk. J. Biol. 2008, 32, 57–62. [Google Scholar]
- Rasooli, I.; Shayegh, S.; Taghizadeh, M.; Astaneh, S.D.A. Phytotherapeutic prevention of dental biofilm formation. Phytother. Res. 2008, 22, 1162–1167. [Google Scholar] [CrossRef] [PubMed]
- Puupponen-Pimia, R.; Nohynek, L.; Meier, C.; Kahkonen, M.; Heinonen, M.; Hopia, A.; Oksman-Caldentey, K.-M. Antimicrobial properties of phenolic compounds from berries. J. Appl. Microbiol. 2001, 90, 494–507. [Google Scholar] [CrossRef] [PubMed]
- Dall’Agnol, R.; Ferraz, A.; Bernardi, A.P.; Albring, D.; Nör, C.; Sarmento, L.; Lamb, L.; Hass, M.; von Poser, G.; Schapoval, E.E.S. Antimicrobial activity of some Hypericum species. Phytomedicine 2003, 10, 511–516. [Google Scholar] [CrossRef]
- Rauha, J.-P.; Remes, S.; Heinonen, M.; Hopia, A.; Kahkonen, M.; Kujala, T.; Pihlaja, K.; Vuorela, H.; Vuorela, P. Antimicrobial effects of Finnish plant extracts containing flavonoids and other phenolic compounds. Int. J. Food Microbiol. 2000, 56, 3–12. [Google Scholar] [CrossRef]
- Singh, R.; Shushni, M.A.M.; Belkheir, A. Antibacterial and antioxidant activities of Mentha piperita L. Arab. J. Chem. 2015, 8, 322–328. [Google Scholar] [CrossRef]
- Osawa, K.; Saeki, T.; Yasuda, H.; Hamashima, H.; Sasatsu, M.; Arai, T. The antibacterial activities of peppermint oil and green tea polyphenols, alone and in combination, against enterohemorrhagic Escherichia coli. Biocontrol Sci. 1999, 4, 1–7. [Google Scholar] [CrossRef]
- Vattem, D.A.; Lina, Y.-T.; Ghaedianb, R.; Shetty, K. Cranberry synergies for dietary management of Helicobacter pylori infections. Process Biochem. 2005, 40, 1583–1592. [Google Scholar] [CrossRef]
- Si, W.; Gong, J.; Tsao, R.; Kalab, M.; Yang, R.; Yin, Y. Bioassay-guided purification and identification of antimicrobial components in Chinese green tea extract. J. Chromatogr. A 2006, 1125, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.-S.; Chung, H.-S. Antibacterial activities of phenolic components from Camellia sinensis L. on pathogenic microorganisms. J. Food Sci. Nutr. 2007, 12, 135–140. [Google Scholar] [CrossRef]
- Ankolekar, C.; Johnson, D.; da Silva Pinto, M.; Johnson, K.; Labbe, R.; Shetty, K. Inhibitory potential of tea polyphenolics and influence of extraction time against Helicobacter pylori and lack of inhibition of beneficial lactic acid bacteria. J. Med. Food 2011, 14, 1321–1329. [Google Scholar] [CrossRef] [PubMed]
- Kohda, C.; Yanagawa, Y.; Shimamura, T. Epigallocatechin gallate inhibits intracellular survival of Listeria monocytogenes in macrophages. Biochem. Biophys. Res. Commun. 2008, 365, 310–315. [Google Scholar] [CrossRef]
- Nakayama, M.; Shimatani, K.; Ozawa, T.; Shigemune, N.; Tomiyama, D.; Yui, K.; Katsuki, M.; Ikeda, K.; Nonaka, A.; Miyamoto, T. Mechanism for the antibacterial action of epigallocatechin gallate (EGCg) on Bacillus subtilis. Biosci. Biotechnol. Biochem. 2015, 79, 845–854. [Google Scholar] [CrossRef]
- Coppo, E.; Marchese, A. Antbacterial activity of polyphenols. Curr. Pharm. Biotechnol. 2014, 15, 380–390. [Google Scholar] [CrossRef]
- Tzounis, X.; Vulevic, J.; Kuhnle, G.G.C.; George, T.; Leonczak, J.; Gibson, G.R.; Kwik-Uribe, C.; Spencer, J.P.E. Flavanol monomer-induced changes to the human faecal microflora. Br. J. Nutr. 2008, 99, 782–792. [Google Scholar] [CrossRef]
- Xie, Y.; Chen, J.; Xiao, A.; Liu, L. Antibacterial activity of polyphenols: Structure-activity relationship and influence of hyperglycemic condition. Molecules 2017, 22, 1913. [Google Scholar] [CrossRef]
- Bae, E.A.; Han, M.J.; Kim, D.H. In vitro anti-Helicobacter pylori activity of some flavonoids and their metabolites. Planta Med. 1999, 65, 442–443. [Google Scholar] [CrossRef]
- Rodríguez Vaquero, M.J.; Alberto, M.R.; Manca de Nadra, M.C. Influence of phenolic compounds from wines on the growth of Listeria monocytogenes. Food Control 2007, 18, 587–593. [Google Scholar] [CrossRef]
- Duda-Chodak, A. The inhibitory effect of polyphenols on human gut microbiota. J. Physiol. Pharmacol. 2012, 63, 497–503. [Google Scholar] [PubMed]
- Cueva, C.; Moreno-Arribas, M.V.; Martín-Álvarez, P.J.; Bills, G.; Vicente, M.F.; Basilio, A.; Rivas, C.L.; Requena, T.; Rodríguez, J.M.; Bartolome, B. Antimicrobial activity of phenolic acids against commensal, probiotic and pathogenic bacteria. Res. Microbiol. 2010, 161, 372–382. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Kokubo, R.; Sakaino, M. Antimicrobial activities of eucalyptus leaf extracts and flavonoids from Eucalyptus maculata. Lett. Appl. Microbiol. 2004, 39, 60–64. [Google Scholar] [CrossRef]
- Roldán, L.P.; Díaz, G.J.; Duringer, J.M. Composition and antibacterial activity of essential oils obtained from plants of the Lamiaceae family against pathogenic and beneficial bacteria. Rev. Colomb. Cienc. Pec. 2010, 23, 451–461. [Google Scholar]
- Adamczak, A.; Ożarowski, M.; Karpiński, T.M. Antibacterial activity of some flavonoids and organic acids widely distributed in plants. J. Clin. Med. 2020, 9, 109. [Google Scholar] [CrossRef]
- Tabasco, R.; Sánchez-Patán, F.; Monagas, M.; Bartolomé, B.; Moreno-Arribas, M.V.; Peláez, C.; Requena, T. Effect of grape polyphenols on lactic acid bacteria and bifidobacteria growth: Resistance and metabolism. Food Microbiol. 2011, 28, 1345–1352. [Google Scholar] [CrossRef]
- Gwiazdowska, D.; Juś, K.; Jasnowska-Małecka, J.; Kluczyńska, K. The impact of polyphenols on Bifidobacterium growth. Acta Biochim. Pol. 2015, 62, 895–901. [Google Scholar] [CrossRef]
- Lee, H.C.; Jenner, A.M.; Low, C.S.; Lee, Y.K. Effect of tea phenolics and their aromatic fecal bacterial metabolites on intestinal microbiota. Res. Microbiol. 2006, 157, 876–884. [Google Scholar] [CrossRef]
- Dolara, P.; Luceri, C.; De Filippo, C.; Femia, A.P.; Giovannelli, L.; Caderni, G.; Cecchini, C.; Silvi, S.; Orpianesi, C.; Cresci, A. Red wine polyphenols influence carcinogenesis, intestinal microflora, oxidative damage and gene expression profiles of colonic mucosa in F344 rats. Mutat. Res. 2005, 591, 237–246. [Google Scholar] [CrossRef]
- Smith, A.H.; Zoetendal, E.; Mackie, R.I. Bacterial mechanisms to overcome inhibitory effects of dietary tannins. Microb. Ecol. 2005, 50, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Henning, S.M.; Summanen, P.H.; Lee, R.-P.; Yang, J.; Finegold, S.M.; Heber, D.; Li, Z. Pomegranate ellagitannins stimulate the growth of Akkermansia muciniphila in vivo. Anaerobe 2017, 43, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.-I.; Yi, L.; Zhang, Y.; Zhou, X.; Ran, L.; Yang, J.; Zhu, J.; Zhang, Q.; Mi, M. Resveratrol attenuates trimethylamine-N-oxide (TMAO)-induced atherosclerosis by regulating TMAO synthesis and bile acid metabolism via remodeling of the gut microbiota. mBio 2016, 7, 02210–02215. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Y.; Sun, J.; Xia, S.; Tang, X.; Le, Y.S.G. Effects of resveratrol on gut microbiota and fat storage in a mouse model with high-fat-inducedobesity. Food Funct. 2014, 5, 1241–1249. [Google Scholar] [CrossRef] [PubMed]
- Masumoto, S.; Terao, A.; Yamamoto, Y.; Mukai, T.; Miura, T.; Shoji, T. Non-absorbable apple procyanidins prevent obesity associated with gut microbial and metabolomic changes. Sci. Rep. 2016, 6, 31208. [Google Scholar] [CrossRef] [PubMed]
- Roopchand, D.E.; Carmody, R.N.; Kuhn, P.; Moskal, K.; Rojas-Silva, P.; Turnbaugh, P.J.; Raskin, I. Dietary polyphenols promote growth of the gut bacterium Akkermansia muciniphila and attenuate high-fat diet–induced metabolic syndrome. Diabetes 2015, 64, 2847–2858. [Google Scholar] [CrossRef] [PubMed]
- Fogliano, V.; Corollaro, M.L.; Vitaglione, P.; Napolitano, A.; Ferracane, R.; Travaglia, F.; Arlorio, M.; Costabile, A.; Klinder, A.; Gibson, G. In vitro bioaccessibility and gut biotransformation of polyphenols present in the water-insoluble cocoa fraction. Mol. Nutr. Food Res. 2011, 55, S44–S55. [Google Scholar] [CrossRef] [PubMed]
- Queipo-Ortuño, M.I.; Boto-Ordóñez, M.; Murri, M.; Gomez-Zumaquero, J.M.; Clemente-Postigo, M.; Estruch, R.; Diaz, F.C.; Andrés-Lacueva, C.; Tinahones, F.J. Influence of red wine polyphenols and ethanol on the gut microbiotaecology and biochemical biomarkers. Am. J. Clin. Nutr. 2012, 95, 1323–1334. [Google Scholar] [CrossRef] [PubMed]
- Vendrame, S.; Guglielmetti, S.; Riso, P.; Arioli, S.; Klimis-Zacas, D.; Porrini, M. Six-week consumption of a wild blueberry powder drink increases bifidobacteria in the human gut. J. Agric. Food Chem. 2011, 59, 12815–12820. [Google Scholar] [CrossRef]
- Moreno-Indias, I.; Sánchez-Alcoholado, L.; Pérez-Martínez, P.; Andrés-Lacueva, C.; Cardona, F.; Tinahones, F.; Queipo-Ortuño, M.I. Red wine polyphenols modulate fecal microbiota and reduce markers of the metabolic syndrome in obese patients. Food Funct. 2016, 7, 1775–1787. [Google Scholar] [CrossRef]
- Bustos, I.; Garcia-Cayuela, T.; Hernández-Ledesma, B.; Peláez, C.; Requena, T.; Martinez-Cuesta, M.C. Effect of flavan-3-ols on the adhesion of potential probiotic Lactobacilli to intestinal cells. J. Agric. Food Chem. 2012, 60, 9082–9088. [Google Scholar] [CrossRef] [PubMed]
- Volstatova, T.; Marsik, P.; Rada, V.; Geigerova, M.; Havlik, J. Effect of apple extracts and selective polyphenols on the adhesion of potential probiotic strains of Lactobacillus gasseri R and Lactobacillus casei FMP. J. Funct. Foods 2017, 35, 391–397. [Google Scholar] [CrossRef]
- Howell, A.B. Bioactive compounds in cranberries and their role in prevention of urinary tract infections. Mol. Nutr. Food Res. 2007, 51, 732–737. [Google Scholar] [CrossRef] [PubMed]
- Foo, L.Y.; Lu, Y.; Howell, A.B.; Vorsa, N. The structure of cranberry proanthocyanidins which inhibit adherence of uropathogenic P-fimbriated Escherichia coli in vitro. Phytochemistry 2000, 54, 173–181. [Google Scholar] [CrossRef]
- Sánchez-Patán, F.; Cueva, C.; Monagas, M.; Walton, G.E.; Gibson, G.R.M.; Quintanilla-López, J.E.; Lebrón-Aguilar, R.; Martín-Álvarez, P.J.; Moreno-Arribas, M.V.; Bartolomé, B. In vitro fermentation of a red wine extract by human gut microbiota: Changes in microbial groups and formation of phenolic metabolites. J. Agric. Food Chem. 2012, 60, 2136–2147. [Google Scholar] [CrossRef]
- Hervert-Hernández, D.; Pintado, C.; Rotger, R.; Goñi, I. Stimulatory role of grape pomace polyphenols on Lactobacillus acidophilus growth. Int. J. Food Microbiol. 2009, 136, 119–122. [Google Scholar] [CrossRef]
- Vivas, N.; Lonvaud-Funel, A.; Glories, Y. Effect of phenolic acids and anthocyanins on growth, viability and malolactic activity of a lactic acid bacterium. Food Microbiol. 1997, 14, 291–300. [Google Scholar] [CrossRef]
- Naito, Y.; Uchiyama, K.; Takagi, T. A next-generation beneficial microbe: Akkermansia muciniphila. J. Clin. Biochem. Nutr. 2018, 63, 33–35. [Google Scholar] [CrossRef]
- Dehghanbanadaki, H.; Aazami, H.; Raftar, S.K.A.; Ashrafian, F.; Ejtahed, H.-S.; Hashemi, E.; Tavassol, H.Z.; Badi, S.A.; Siadat, S.D. Global scientific output trend for Akkermansia muciniphila research: A bibliometric and scientometric analysis. BMC Med. Inform. Decis. Mak. 2020, 20, 291. [Google Scholar] [CrossRef]
- Farhadi, F.; Khameneh, B.; Iranshahi, M.; Iranshahy, M. Antibacterial activity of flavonoids and their structure–activity relationship: An update review. Phytother. Res. 2019, 33, 13–40. [Google Scholar] [CrossRef]
- Mickymaray, S.; Alfaiz, F.A.; Paramasivam, A. Efficacy and mechanisms of flavonoids against the emerging opportunistic nontuberculous Mycobacteria. Antibiotics 2020, 9, 450. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.K.; Kumar, S. Perspective on Plant Products as Antimicrobial Agents: A Review. Pharmacologia 2013, 4, 469–480. [Google Scholar] [CrossRef]
- Papuc, C.; Goran, G.V.; Predescu, C.N.; Nicorescu, V.; Stefan, G. Plant polyphenols as antioxidant and antibacterial agents for shelf-life extension of meat and meat products: Classification, structures, sources, and action mechanisms. Compr. Rev. Food Sci. Food Saf. 2017, 16, 1243–1268. [Google Scholar] [CrossRef] [PubMed]
- Cushnie, T.P.T.; Lamb, A.J. Antimicrobial activity of flavonoids. Int. J. Antimicrob. Agents 2005, 26, 343–356. [Google Scholar] [CrossRef] [PubMed]
- Cushnie, T.P.T.; Lamb, A.J. Recent advances in understanding the antibacterial properties of flavonoids. Int. J. Antimicrob. Agents 2011, 38, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Matilla-Cuenca, L.; Gil, C.; Cuesta, S.; Rapún-Araiz, B.; Žiemytė, M.; Mira, A.; Lasa, I.; Valle, J. Antibiofilm activity of flavonoids on staphylococcal biofilms through targeting BAP amyloids. Sci. Rep. 2020, 10, 18968. [Google Scholar] [CrossRef]
- Xie, Y.; Yang, W.; Tang, F.; Chen, X.; Ren, L. Antibacterial activities of flavonoids: Structure-activity relationship and mechanism. Curr. Med. Chem. 2015, 22, 132–149. [Google Scholar] [CrossRef]
- Kumar, S.; Pandey, A.K. Chemistry and biological activities of flavonoids: An overview. Sci. World J. 2013, 162750. [Google Scholar] [CrossRef]
- Brudzynski, K.; Maldonado-Alvarez, L. Polyphenol-protein complexes and their consequences for the redox activity, structure and function of honey. A current view and new hypothesis—A review. Pol. J. Food Nutr. Sci. 2015, 65, 71–80. [Google Scholar] [CrossRef]
- Seczyk, Ł.; Świeca, M.; Kapusta, I.; Gawlik-Dziki, U. Protein–phenolic interactions as a factor affecting the physicochemical properties of White Bean Proteins. Molecules 2019, 24, 408. [Google Scholar] [CrossRef]
- Boath, A.S.; Grussu, D.; Stewart, D.; McDougall, G.J. Berry polyphenols inhibit digestive enzymes: A source of potential health benefits? Food Dig. 2012, 3, 1–7. [Google Scholar] [CrossRef]
- Gonçalves, S.; Romano, A. Inhibitory properties of phenolic compounds against enzymes linked with human diseases. In Phenolic Compounds—Biological Activity; Soto-Hernandez, M., Palma-Tenango, M., Garcia-Mateos, M., Eds.; IntechOpen: Rijeka, Croatia, 2017; pp. 99–118. ISBN 978-953-51-5090-9. [Google Scholar] [CrossRef]
- Martinez-Gonzalez, A.I.; Díaz-Sánchez, Á.G.; de la Rosa, L.A.; Vargas-Requena, C.L.; Bustos-Jaimes, I.; Alvarez-Parrilla, E. Polyphenolic compounds and digestive enzymes: In vitro non-covalent interactions. Molecules 2017, 22, 669. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Wang, Y.; Miao, M. Inhibition of α-amylase by polyphenolic compounds: Substrate digestion, binding interactions and nutritional intervention. Trends Food Sci. Technol. 2020, 104, 190–207. [Google Scholar] [CrossRef]
- Tadera, K.; Minami, Y.; Takamatsu, K.; Matsuoka, T. Inhibition of alpha-glucosidase and alpha-amylase by flavonoids. J. Nutr. Sci. Vitaminol. 2006, 52, 149–153. [Google Scholar] [CrossRef]
- Cowan, M.M. Plant products as antimicrobial agents. Clin. Microbiol. Rev. 1999, 12, 564–582. [Google Scholar] [CrossRef]
- Yang, N.Y.; Hinner, M.J. Getting across the cell membrane: An overview for small molecules, peptides, and proteins. Methods Mol. Biol. 2015, 1266, 29–53. [Google Scholar] [CrossRef]
- Hu, P.; Huang, P.; Chen, M.W. Curcumin reduces Streptococcus mutans biofilm formation by inhibiting sortase A activity. Arch. Oral Biol. 2013, 58, 1343–1348. [Google Scholar] [CrossRef]
- Kang, S.S.; Kim, J.-G.; Lee, T.-H.; Oh, K.-B. Flavonols inhibit sortases and sortase-mediated Staphylococcus aureus clumping to fibrynogen. Biol. Pharm. Bull. 2006, 29, 1751–1755. [Google Scholar] [CrossRef]
- Edziri, H.; Mastouri, M.; Mahjoub, M.A.; Mighri, Z.; Mahjoub, A.; Verschaeve, L. Antibacterial, antifungal and cytotoxic activities of two flavonoids from Retama raetam flowers. Molecules 2012, 17, 7284–7293. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, T.; Wang, K.; Hou, C.; Cai, S.; Huang, Y.; Du, Z.; Huang, H.; Kong, J.; Chen, Y. Baicalein inhibits Staphylococcus aureus biofilm formation and the quorum sensing system in vitro. PLoS ONE 2016, 11, e0153468. [Google Scholar] [CrossRef]
- Yoda, Y.; Hu, Z.-Q.; Zhao, W.-H.; Shimamura, T. Different susceptibilities of Staphylococcus and Gram-negative rods to epigallocatechin gallate. J. Infect. Chemother. 2004, 10, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, M.; Shimatani, K.; Ozawa, T.; Shigemune, N.; Tsugukuni, T.; Tomiyama, D.; Kurahachi, M.; Nonaka, A.; Miyamoto, T. A study of the antibacterial mechanism of catechins: Isolation and identification of Escherichia coli cell surface proteins that interact with epigallocatechin gallate. Food Sci. Biotechnol. 2013, 33, 433–439. [Google Scholar] [CrossRef]
- Taylor, P.W.; Hamilton-Miller, J.M.T.; Stapleton, P.D. Antimicrobial properties of green tea catechins. Food Sci. Technol. Bull. 2005, 2, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Fowler, Z.L.; Shah, K.; Panepinto, J.C.; Jacobs, A.; Koffas, M.A.G. Development of non-natural flavanones as antimicrobial agents. PLoS ONE 2011, 6, e25681. [Google Scholar] [CrossRef] [PubMed]
- Omosa, L.K.; Midiwo, J.O.; Mbaveng, A.T.; Tankeo, S.B.; Seukep, J.A.; Voukeng, I.K.; Dzotam, J.K.; Isemeki, J.; Derese, S.; Omolle, R.A.; et al. Antibacterial activities and structure—Activity relationships of a panel of 48 compounds from Kenyan plants against multidrug resistant phenotypes. SpringerPlus 2016, 5, 901. [Google Scholar] [CrossRef] [PubMed]
- Randhawa, H.K.; Hundal, K.K.; Ahirrao, P.N.; Jachak, S.M.; Nandanwar, H.S. Efflux pump inhibitory activity of flavonoids isolated from Alpinia calcarata against methicillin-resistant Staphylococcus aureus. Biologia 2016, 71, 484–493. [Google Scholar] [CrossRef]
- Brown, A.K.; Papaemmanouil, A.; Bhowruth, V.; Bhatt, A.; Dover, L.G.; Besra, G.S. Flavonoid inhibitors as novel antimycobacterial agents targeting Rv0636, a putative dehydratase enzyme involved in Mycobacterium tuberculosis fatty acid synthase II. Microbiology 2007, 153, 3314–3322. [Google Scholar] [CrossRef]
- Zhang, Y.M.; Rock, C.O. Evaluation of epigallocatechin gallate and related plant polyphenols as inhibitors of the FabG and FabI reductases of bacterial type II fatty-acid synthase. J. Biol. Chem. 2004, 279, 30994–31001. [Google Scholar] [CrossRef]
- Xiao, Z.-T.; Zhu, Q.; Zhang, H.-Y. Identifying antibacterial targets of flavonoids by comparative genomics and molecular modelling. Open J. Genom. 2014, 3, 1–8. [Google Scholar] [CrossRef]
- Zhang, L.; Kong, Y.; Wu, D.; Zhang, H.; Wu, J.; Chen, J.; Ding, J.; Hu, L.; Jiang, H.; Shen, X. Three flavonoids targeting the β-hydroxyacyl-acyl carrier protein dehydratase from Helicobacter pylori: Crystal structure characterization with enzymatic inhibition assay. Protein Sci. 2008, 17, 1971–1978. [Google Scholar] [CrossRef]
- Jeong, K.-W.; Lee, J.-Y.; Kang, D.-I.; Lee, J.-U.; Shin, S.Y.; Kim, Y. Screening of flavonoids as candidate antibiotics against Enterococcus faecalis. J. Nat. Prod. 2009, 72, 719–724. [Google Scholar] [CrossRef] [PubMed]
- Hertel, W.; Peschel, G.; Ozegowski, J.-H.; Müller, P.-J. Inhibitory effects of triterpenes and flavonoids on the enzymatic activity of hyaluronic acid-splitting enzymes. Arch. Pharm. Chem. Life Sci. 2006, 339, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Brahmachari, G.; Mandal, N.C.; Jash, S.K.; Roy, R.; Mandal, L.C.; Mukhopadhyay, A.; Biswajit Behera, B.; Majhi, S.; Mondal, A.; Gangopadhyay, A. Evaluation of the antimicrobial potential of two flavonoids isolated from Limnophila plants. Chem. Biodivers. 2011, 8, 1139–1151. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Z.-P.; Shi, D.-H.; Li, H.-Q.; Zhang, L.N.; Xua, C.; Zhua, H.-L. Polyphenols based on isoflavones as inhibitors of Helicobacter pylori urease. Bioorg. Med. Chem. 2007, 15, 3703–3710. [Google Scholar] [CrossRef]
- Ulrey, R.K.; Barksdale, S.M.; Zhou, W.; van Hoek, M.L. Cranberry proanthocyanidins have anti-biofilm properties against. BMC Complement. Altern. Med. 2014, 14, 499. [Google Scholar] [CrossRef]
- Mori, A.; Nishino, C.; Enoki, N.; Tawata, S. Antibacterial activity and mode of action of plant flavonoids against Proteus vulgaris and Staphylococcus aureus. Phytochem. 1987, 26, 2231–2234. [Google Scholar] [CrossRef]
- Lou, Z.; Wang, H.; Rao, S.; Sun, J.; Ma, C.; Li, J. p-Coumaric acid kills bacteria through dual damage mechanisms. Food Control 2012, 25, 550–554. [Google Scholar] [CrossRef]
- Dzoyem, J.P.; Hamamoto, H.; Ngameni, B.; Ngadjui, B.T.; Sekimizu, K. Antimicrobial action mechanism of flavonoids from Dorstenia species. Drug Discov. Ther. 2013, 7, 66–72. [Google Scholar] [CrossRef][Green Version]
- Suriyanarayanan, B.; Shanmugam, K.; Santhosh, R.S. Synthetic quercetin inhibits mycobacterial growth possibly by interacting with DNA gyrase. Rom. Biotechnol. Lett. 2013, 18, 8587–8593. [Google Scholar]
- Bandele, O.J.; Clawson, S.J.; Osheroff, N. Dietary polyphenols as topoisomerase II poisons: B ring and C ring substituents determine the mechanism of enzyme-mediated DNA cleavage enhancement. Chem. Res. Toxicol. 2008, 21, 1253–1260. [Google Scholar] [CrossRef]
- Bandele, O.J.; Osheroff, N. (−)-Epigallocatechin gallate, a major constituent of green tea, poisons human type II topoisomerases. Chem. Res. Toxicol. 2008, 21, 936–943. [Google Scholar] [CrossRef] [PubMed]
- Oblak, M.; Kotnik, M.; Solmajer, T. Discovery and Development of ATPase inhibitors of DNA gyrase as antibacterial agents. Curr. Med. Chem. 2007, 14, 2033–2047. [Google Scholar] [CrossRef] [PubMed]
- Plaper, A.; Golob, M.; Hafner, I.; Oblak, M.; Solmajer, T.; Jerala, R. Characterization of quercetin binding site on DNA gyrase. Biochem. Biophys. Res. Commun. 2003, 306, 530–536. [Google Scholar] [CrossRef]
- Gradišar, H.; Pristovšek, P.; Plaper, A.; Jerala, R. Green Tea catechins inhibit bacterial DNA gyrase by interaction with its ATP binding site. J. Med. Chem. 2007, 50, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Ohemeng, K.A.; Podlogar, B.L.; Nguyen, V.N.; Bernstein, J.I.; Krause, H.M.; Hilliard, J.J.; Barrett, J.F. DNA gyrase inhibitory and antimicrobial activities of some diphenic acid monohydroxamides. J. Med. Chem. 1997, 40, 3292–3296. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Lu, Y.; Zang, X.; Wu, T.; Qi, X.J.; Pan, S.; Xu, X. 3D-QSAR and docking studies of flavonoids as potent Escherichia coli inhibitors. Sci. Rep. 2016, 6, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Bernard, F.-X.; Sable, S.; Cameron, B.; Provost, J.; Desnottes, J.-F.; Crouzet, J.; Blanche, F. Glycosylated flavones as selective inhibitors of topoisomerase IV. Antimicrob. Agents Chemother. 1997, 41, 992–998. [Google Scholar] [CrossRef] [PubMed]
- Arima, H.; Hitoshi Ashida, H.; Danno, G. Rutin-enhanced antibacterial activities of flavonoids against Bacillus cereus and Salmonella enteritidis. Biosci. Biotechnol. Biochem. 2002, 66, 1009–1014. [Google Scholar] [CrossRef]
- Khan, N.S.; Ahmad, A.; Hadi, S.M. Anti-oxidant, pro-oxidant properties of tannic acid and its binding to DNA. Chem. Biol. Interact. 2000, 125, 177–189. [Google Scholar] [CrossRef]
- Cetin-Karaca, H.; Newman, M.C. Antimicrobial efficacy of plant phenolic compounds against Salmonella and Escherichia coli. Food Biosci. 2015, 11, 8–16. [Google Scholar] [CrossRef]
- Stepanović, S.; Antić, N.; Dakić, I.; Švabić-Vlahović, M. In vitro antimicrobial activity of propolis and synergism between propolis and antimicrobial drugs. Microbiol. Res. 2003, 158, 353–357. [Google Scholar] [CrossRef] [PubMed]
- Ikigai, H.; Nakae, T.; Hara, Y.; Shimamura, T. Bactericidal catechins damage the lipid bilayer. Biochim. Biophys. Acta 1993, 1147, 132–136. [Google Scholar] [CrossRef]
- Vattem, D.A.; Lin, Y.-T.; Labbe, R.G.; Shetty, K. Phenolic antioxidant mobilization in cranberry pomace by solid-state bioprocessing using food grade fungus Lentinus edodes and effect on antimicrobial activity against select food borne pathogens. Innov. Food Sci. Emerg. Technol. 2004, 5, 81–91. [Google Scholar] [CrossRef]
- Wu, D.; Kong, Y.; Han, C.; Chen, J.; Hu, L.; Jiang, H.; Shen, X. d-Alanine:d-alanine ligase as a new target for the flavonoids quercetin and apigenin. Int. J. Antimicrob. Agents 2008, 32, 421–426. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Z.-P.; Wei, W.; Liu, Q.; Wang, P.-F.; Luo, X.; Chen, F.-Y.; Cao, Y.; Huang, H.-X.; Liua, M.-M.; Zhu, H.L. C-7 modified flavonoids as novel tyrosyl-tRNA synthetase inhibitors. RSC Adv. 2017, 7, 6193–6201. [Google Scholar] [CrossRef]
- Cai, W.; Fu, Y.; Zhang, W.; Chen, X.; Zhao, J.; Song, W.; Li, Y.; Huang, Y.; Wu, Z.; Sun, R.; et al. Synergistic effects of baicalein with cefotaxime against Klebsiella pneumonia through inhibiting CTX-M-1 gene expression. BMC Microbiol. 2016, 16, 181. [Google Scholar] [CrossRef]
- Farooq, S.; Wahab, A.-T.; Fozing, C.D.A.; Rahman, A.-U.; Choudhary, M.I. Artonin I inhibits multidrug resistance in Staphylococcus aureus and potentiates the action of inactive antibiotics in vitro. J. Appl. Microbiol. 2014, 117, 996–1011. [Google Scholar] [CrossRef]
- Zhao, W.-H.; Hu, Z.-Q.; Okubo, S.; Hara, Y.; Shimamura, T. Mechanism of synergy between epigallocatechin gallate and β-lactams against Methicillin-Resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 2001, 45, 1737–1742. [Google Scholar] [CrossRef]
- Delehanty, J.B.; Johnson, B.J.; Hickey, T.E.; Pons, T.; Ligler, F.S. Plant proanthocyanidins bind to and neutralize bacterial lipopolysaccharides. In Naval Research Laboratory Reviews; 2008; pp. 101–107. [Google Scholar]
- Nohynek, L.J.; Alakomi, H.L.; Kähkönen, M.P.; Heinonen, M.; Helander, I.M.; Oksman-Caldentey, K.-M.; Puupponen-Pimiä, R.H. Berry phenolics: Antimicrobial properties and mechanisms of action against severe human pathogens. Nutr. Cancer 2006, 54, 18–32. [Google Scholar] [CrossRef]
- Hartmann, M.; Berditsch, M.; Hawecker, J.; Ardakani, M.F.; Gerthsen, D.; Ulrich, A.S. Damage of the bacterial cell envelope by antimicrobial peptides gramicidin S and PGLa as revealed by transmission and scanning electron microscopy. Antimicrob. Agents Chemother. 2010, 54, 3132–3142. [Google Scholar] [CrossRef]
- Reygaert, W.C. The antimicrobial possibilities of green tea. Front. Microbiol. 2014, 5, 434. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Woo, E.-R.; Lee, D.G. Apigenin induces cell shrinkage in Candida albicans by membrane perturbation. FEMS Yeast Res. 2018, 18, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Perumal, S.; Mahmud, R.; Ismail, S. Mechanism of action of isolated caffeic acid and epicatechin 3-gallate from Euphorbia hirta against Pseudomonas aeruginosa. Pharmacogn. Mag. 2017, 13 (Suppl. 2), S311–S315. [Google Scholar] [CrossRef]
- Matijašević, D.; Pantić, M.; Rašković, B.; Pavlović, V.; Duvnjak, D.; Sknepnek, A.; Nikšić, M. The antibacterial activity of Coriolus versicolor methanol extract and its effect on ultrastructural changes of Staphylococcus aureus and Salmonella Enteritidis. Front. Microbiol. 2016, 7, 1226. [Google Scholar] [CrossRef] [PubMed]
- Fathima, A.; Rao, J.R. Selective toxicity of Catechin—A natural flavonoid towards bacteria. Appl. Microbiol. Biotechnol. 2016, 100, 6395–6402. [Google Scholar] [CrossRef]
- Tagousop, C.N.; Tamokou, J.d.D.; Ekom, S.E.; Ngnokam, D.; Voutquenne-Nazabadioko, L. Antimicrobial activities of flavonoid glycosides from Graptophyllum grandulosum and their mechanism of antibacterial action. BMC Complement. Altern. Med. 2018, 18, 252. [Google Scholar] [CrossRef]
- Ollila, F.; Halling, K.; Vuorela, P.; Vuorela, H.; Slotte, J.P. Characterization of flavonoid–biomembrane interactions. Arch. Biochem. Biophys. 2002, 399, 103–108. [Google Scholar] [CrossRef]
- Chabot, S.; Bel-Rhlid, R.; Chenevert, R.; Piche, Y. Hyphal growth promotion in vitro of the VA mycorrhizal fungus, Gigaspora margarita Becker & Hall, by the activity of structurally specific flavonoid compounds under CO2 -enriched conditions. New Phytol. 1992, 122, 461–467. [Google Scholar]
- Tsuchiya, H. Stereospecificity in membrane effects of catechins. Chem. Biol. Inter. 2001, 134, 41–54. [Google Scholar] [CrossRef]
- Carson, C.F.; Mee, B.J.; Riley, T.V. Mechanism of action of Melaleuca alternifolia (Tea Tree) oil on Staphylococcus aureus determined by time-kill, lysis, leakage, and salt tolerance assays and electron microscopy. Antimicrob. Agents Chemother. 2002, 46, 1914–1920. [Google Scholar] [CrossRef]
- Tsuchiya, H. Membrane interactions of phytochemicals as their molecular mechanism applicable to the discovery of drug leads from plants. Molecules 2015, 20, 18923–18966. [Google Scholar] [CrossRef]
- Selvaraj, S.; Krishnaswamy, S.; Devashya, V.; Sethuraman, S.; Krishnan, U.M. Influence of membrane lipid composition on flavonoid–membrane interactions: Implications on their biological activity. Progr. Lipid Res. 2015, 58, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Arora, A.; Byrem, T.M.; Nair, M.G.; Strasburg, G.M. Modulation of liposomal membrane fluidity by flavonoids and isoflavonoids. Arch. Biochem. Biophys. 2000, 373, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Van Dijk, C.; Driessen, A.J.M.; Recourt, K. The Uncoupling efficiency and affinity of flavonoids for vesicles. Biochem. Pharmacol. 2000, 60, 1593–1600. [Google Scholar] [CrossRef]
- Wu, T.; He, M.; Zang, X.; Zhou, Y.; Qio, T.; Pan, S.; Xu, X. A structure-activity relationship study of flavonoids as inhibitors of E. coli by membrane interaction effect. Biochim. Biophys. Acta 2013, 1828, 2751–2756. [Google Scholar] [CrossRef]
- He, M.; Wu, T.; Pan, S.; Xu, X. Antimicrobial mechanism of flavonoids against Escherichia coli ATCC25922 by model membrane study. Appl. Surf. Sci. 2014, 305, 515–521. [Google Scholar] [CrossRef]
- Mun, S.-H.; Joung, D.-K.; Kim, S.-B.; Park, S.-J.; Seo, Y.-S.; Gong, R.; Choi, J.-G.; Shin, D.-W.; Rho, J.-R.; Kang, O.-H.; et al. The mechanism of antimicrobial activity of sophoraflavanone B against methicillin-resistant Staphylococcus aureus. Foodborne Pathog. Dis. 2014, 11, 234–239. [Google Scholar] [CrossRef]
- Savoia, D. Plant-derived antimicrobial compounds: Alternatives to antibiotic. Future Microbiol. 2012, 7, 979–990. [Google Scholar] [CrossRef]
- Chinnam, N.; Dadi, P.K.; Sabri, S.A.; Ahmad, M.; Kabir, M.A.; Ahmad, Z. Dietary bioflavonoids inhibit Escherichia coli ATP synthase in a differential manner. Int. J. Biol. Macromol. 2010, 46, 478–486. [Google Scholar] [CrossRef]
- Dadi, P.K.; Ahmad, M.; Ahmad, Z. Inhibition of ATPase activity of Escherichia coli ATP synthase by polyphenols. Int. J. Biol. Macromol. 2009, 45, 72–79. [Google Scholar] [CrossRef]
- Gill, A.O.; Holley, R.A. Inhibition of membrane bound ATPases of Escherichia coli and Listeria monocytogenes by plant oil aromatics. Int. J. Food Microbiol. 2006, 111, 170–174. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, Z.; Laughlin, T.F.; Kady, I.O. Thymoquinone inhibits Escherichia coli ATP synthase and cell growth. PLoS ONE 2015, 10, e0127802. [Google Scholar] [CrossRef] [PubMed]
- Haraguchi, H.; Tanimoto, K.; Tamura, Y.; Mizutanit, K.; Kinoshita, T. Mode of antibacterial action of retrochalcones from Glycyrrhiza inflata. Phytochemistry 1998, 48, 125–129. [Google Scholar] [CrossRef]
- Kim, H.-S.; Quon, M.J.; Kim, J. New insights into the mechanisms of polyphenols beyond antioxidant properties; lessons from the green tea polyphenol, epigallocatechin 3-gallate. Redox Biol. 2014, 2, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Castillo-Juárez, I.; Maeda, T.; Mandujano-Tinoco, E.A.; Tomás, M.; Pérez-Eretza, B.; García-Contreras, S.J.; Wood, T.K.; García-Contreras, R. Role of quorum sensing in bacterial infections. World J. Clin. Cases 2015, 3, 575–598. [Google Scholar] [CrossRef] [PubMed]
- Packiavathy, I.A.S.V.; Priya, S.; Pandian, S.K.; Ravi, A.V. Inhibition of biofilm development of uropathogens by curcumin—An anti-quorum sensing agent from Curcuma longa. Food Chem. 2014, 148, 453–460. [Google Scholar] [CrossRef]
- Wang, T.-Y.; Li, Q.; Bi, K.-S. Bioactive flavonoids in medicinal plants: Structure, activity and biological fate. Asian J. Pharm. Sci. 2018, 13, 12–23. [Google Scholar] [CrossRef]
- Gopu, V.; Meena, C.K.; Shetty, P.H. Quercetin influences quorum sensing in food borne bacteria: In-vitro and in-silico evidence. PLoS ONE 2015, 10, e0134684. [Google Scholar] [CrossRef]
- Al Azzaz, J.; Al Tarraf, A.; Heumann, A.; Da Silva Barreira, D.; Laurent, J.; Assifaoui, A.; Rieu, A.; Guzzo, J.; Lapaquette, P. Resveratrol favors adhesion and biofilm formation of Lacticaseibacillus paracasei subsp. paracasei Strain ATCC334. Int. J. Mol. Sci. 2020, 21, 5423. [Google Scholar] [CrossRef]
- Lee, J.-H.; Regmi, S.C.; Kim, J.-A.; Cho, M.H.; Yun, H.; Lee, C.-S.; Lee, J. Apple flavonoid phloretin inhibits Escherichia coli O157:H7 biofilm formation and ameliorates colon inflammation in rats. Infect. Immun. 2011, 79, 4819–4827. [Google Scholar] [CrossRef]
- Serra, D.O.; Mika, F.; Richter, A.M.; Hengge, R. The green tea polyphenol EGCG inhibits E. coli biofilm formation by impairing amyloid curli fibre assembly and downregulating the biofilm regulator CsgD via the r E -dependent sRNA RybB. Mol. Microbiol. 2016, 101, 136–151. [Google Scholar] [CrossRef] [PubMed]
- Hengge, R. Targeting bacterial biofilms by the Green Tea polyphenol EGCG. Molecules 2019, 24, 2403. [Google Scholar] [CrossRef] [PubMed]
- Bai, L.; Takagi, S.; Ando, T.; Yoneyama, H.; Ito, K.; Mizugai, H.; Isogai, E. Antimicrobial activity of tea catechin against canine oral bacteria and the functional mechanisms. J. Vet. Med. Sci. 2016, 78, 1439–1445. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-Y.; Su, T.Y.; Wang, M.Y.; Yang, S.-F.; Mar, K.; Hung, S.-L. Inhibitory effects of tea catechin epigallocatechin-3-gallate against biofilms formed from Streptococcus mutans and a probiotic Lactobacillus strain. Arch. Oral Biol. 2018, 94, 69–77. [Google Scholar] [CrossRef]
- Veloz, J.J.; Alvear, M.; Salazar, L.A. Antimicrobial and antibiofilm activity against Streptococcus mutans of individual and mixtures of the main polyphenolic compounds found in chilean propolis. BioMed Res. Int. 2019, 2019, 7602343. [Google Scholar] [CrossRef]
- Xu, X.; Zhou, X.D.; Wu, C.D. Tea catechin epigallocatechin gallate inhibits Streptococcus mutans biofilm formation by suppressing gtf genes. Arch. Oral Biol. 2012, 57, 678–683. [Google Scholar] [CrossRef]
- Xu, X.; Zhou, X.D.; Wu, C.D. The tea catechin epigallocatechin gallate suppresses cariogenic virulence factors of Streptococcus mutans. Antimicrob. Agents Chemother. 2011, 55, 1229–1236. [Google Scholar] [CrossRef]
- Sivaranjani, M.; Gowrishankar, S.; Kamaladevi, A.; Pandian, S.K.; Balamurugan, K.; Ravi, A.V. Morin inhibits biofilm production and reduces the virulence of Listeria monocytogenes—An in vitro and in vivo approach. Int. J. Food Microbiol. 2016, 237, 73–82. [Google Scholar] [CrossRef]
- Upadhyay, A.; Upadhyaya, I.; Kollanoor-Johny, A.; Venkitanarayanan, K. Antibiofilm effect of plant derived antimicrobials on Listeria monocytogenes. Food Microbiol. 2013, 36, 79–89. [Google Scholar] [CrossRef]
- Borges, A.; Saavedra, M.J.; Simões, M. The activity of ferulic and gallic acids in biofilm prevention and control of pathogenic bacteria. J. Bioadh. Biofilm Res. 2012, 28, 755–767. [Google Scholar] [CrossRef]
- Borges, A.; Ferreira, C.; Saavedra, M.J.; Simões, M. Antibacterial activity and mode of action of ferulic and gallic acids against pathogenic bacteria. Microb. Drug Resist. 2013, 19, 256–265. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Cazares, N.; García-Contreras, R.; Pérez-López, M.; Castillo-Juárez, I. Phenolic compounds with anti-virulence properties. In Phenolic Compounds—Biological Activity; Soto-Hernandez, M., Palma-Tenango, M., del Rosario Garcia-Mateos, M., Eds.; IntechOpen: Rijeka, Croatia, 2017; pp. 139–167. [Google Scholar] [CrossRef]
- Daglia, M. Polyphenols as antimicrobial agents. Curr. Opin. Biotechnol. 2012, 23, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Scalbert, A. Antimicrobial properties of tannins. Phytochemistry 1991, 30, 3875–3883. [Google Scholar] [CrossRef]
- Field, S.A.; Lettinga, G. Toxicity of tannic compounds to microorganisms. In Plant Polyphenols, 1st ed.; Hemingway, R.W., Laks, P.E., Eds.; Plenum Press: New York, NY, USA, 1992; pp. 673–692. [Google Scholar]
- Chung, K.T.; Lu, Z.; Chou, M.W. Mechanism of inhibition of tannic acid and related compounds on the growth of intestinal bacteria. Food Chem. Toxicol. 1998, 36, 1053–1060. [Google Scholar] [CrossRef]
- Andrews, S.C.; Robinson, A.K.; Rodriguez-Quiñones, F. Bacterial iron homeostasis. FEMS Microbiol. Rev. 2003, 27, 215–237. [Google Scholar] [CrossRef]
- Perron, N.R.; Brumaghim, J.L. A Review of the antioxidant mechanisms of polyphenol compounds related to iron binding. Cell Biochem. Biophys. 2009, 53, 75–100. [Google Scholar] [CrossRef]
- Fernandez, M.T.; Mira, M.L.; Florencio, M.H.; Jennings, K.R. Iron and copper chelation by flavonoids: An electrospray mass spectrometry study. J. Inorg. Biochem. 2002, 92, 105–111. [Google Scholar] [CrossRef]
- Mladěnka, P.; Macáková, K.; Filipský, T.; Zatloukalová, L.; Jahodář, L.; Bovicelli, P.; Silvestri, I.P.; Hrdina, R.; Saso, L. In vitro analysis of iron chelating activity of flavonoids. J. Inorg. Biochem. 2011, 105, 693–701. [Google Scholar] [CrossRef]
- Rajakovich, L.J.; Balskus, E.P. Metabolic functions of the human gut microbiota: The role of metalloenzymes. Nat. Prod. Rep. 2019, 36, 593. [Google Scholar] [CrossRef]
- Wang, W.-L.; Chai, S.C.; Huang, M.; He, H.-Z.; Hurley, T.D.; Ye, Q.-Z. Discovery of inhibitors of Escherichia coli methionine aminopeptidase with the Fe(II)-form selectivity and antibacterial activity. J. Med. Chem. 2008, 51, 6110–6120. [Google Scholar] [CrossRef]
- Lagha, A.B.; Haas, B.; Grenier, D. Tea polyphenols inhibit the growth and virulence properties of Fusobacterium nucleatum. Sci. Rep. 2017, 7, 44815. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.; Tan, K.S. Effects of epigallocatechin gallate against Enterococcus faecalis biofilm and virulence. Arch. Oral Biol. 2015, 60, 393–399. [Google Scholar] [CrossRef] [PubMed]
- Echeverría, J.; Opazo, J.; Mendoza, L.; Urzúa, A.; Wilkens, M. Structure-activity and lipophilicity relationships of selected antibacterial natural flavones and flavanones of Chilean flora. Molecules 2017, 22, 608. [Google Scholar] [CrossRef]
- Botta, B.; Vitali, A.; Menendez, P.; Misiti, D.; Delle Monache, G. Prenylated flavonoids: Pharmacology and biotechnology. Curr. Med. Chem. 2005, 12, 713–739. [Google Scholar] [CrossRef] [PubMed]
- Cermak, P.; Olsovska, J.; Mikyska, A.; Dusek, M.; Kadleckova, Z.; Vanicek, J.; Nyc, O.; Sigler, K.; Bostikova, V.; Bostik, P. Strong antimicrobial activity of xanthohumol and other derivatives from hops (Humulus lupulus L.) on gut anaerobic bacteria. APMIS 2017, 125, 1033–1038. [Google Scholar] [CrossRef] [PubMed]
- Stompor, M.; Zarowska, B. Antimicrobial activity of xanthohumol and its selected structural analogues. Molecules 2016, 21, 608. [Google Scholar] [CrossRef] [PubMed]
- Jamil, S.; Lathiff, S.M.A.; Abdullah, S.A.; Jemaon, N.; Sirat, H.M. Antimicrobial flavonoids from Artocarpus anisophyllus Miq. and Artocarpus lowii King. J. Tekchnologi 2014, 71, 95–99. [Google Scholar] [CrossRef]
- Campos, F.M.; Couto, J.A.; Figueiredo, A.R.; Tóth, I.V.; Rangel, A.O.S.S.; Hogg, T.A. Cell membrane damage induced by phenolic acids on wine lactic acid bacteria. Int. J. Food Microbiol. 2009, 135, 144–151. [Google Scholar] [CrossRef]
- Thakur, D.; Das, S.C.; Sabhapondit, S.; Tamuly, P.; Deka, D.K. Antimicrobial activities of Tocklai vegetative tea clones. Indian J. Microbiol. 2011, 51, 450–455. [Google Scholar] [CrossRef][Green Version]
- Puljula, E.; Walton, G.; Woodward, M.J.; Karonen, M. Antimicrobial activities of ellagitannins against Clostridiales perfringens, Escherichia coli, Lactobacillus plantarum and Staphylococcus aureus. Molecules 2020, 25, 3714. [Google Scholar] [CrossRef]
- Tarko, T.; Duda-Chodak, A.; Wajda, Ł.; Satora, P.; Sroka, P.; Semik-Szczurak, D. Application of principal component analysis for optimization of polyphenol extraction from alternative plant sources. J. Food Nutr. Res. 2017, 56, 61–72. [Google Scholar]
- Boeing, J.S.; Barizão, E.O.; Costa e Silva, B.; Montanher, F.P.; de Cinque Almeida, V.; Visentainer, J.V. Evaluation of solvent effect on the extraction of phenolic compounds and antioxidant capacities from the berries: Application of principal component analysis. Chem. Cent. J. 2014, 8, 48. [Google Scholar] [CrossRef] [PubMed]
- Borges, A.; José, H.; Homem, V.; Simões, M. Comparison of techniques and solvents on the antimicrobial and antioxidant potential of extracts from Acacia dealbata and Olea europaea. Antibiotics 2020, 9, 48. [Google Scholar] [CrossRef] [PubMed]
- Tarko, T.; Duda-Chodak, A.; Soszka, A. Changes in phenolic compounds and antioxidant activity of fruit musts and fruit wines during simulated digestion. Molecules 2020, 25, 5574. [Google Scholar] [CrossRef] [PubMed]
- Renard, C.M.G.C.; Watrelot, A.A.; Le Bourvellec, C. Interactions between polyphenols and polysaccharides: Mechanisms and consequences in food processing and digestion. Trends Food Sci. Technol. 2017, 60, 43–51. [Google Scholar] [CrossRef]
- Spencer, J.P.E. Metabolism of tea flavonoids in the gastrointestinal tract. J. Nutr. 2003, 133, 3255S–3261S. [Google Scholar] [CrossRef]
- Thomas, M.; Thibault, J.-F. Cell-wall polysaccharides in the fruits of Japanese quince (Chaenomeles japonica): Extraction and preliminary characterisation. Carbohydr. Polym. 2002, 49, 345–355. [Google Scholar] [CrossRef]
- Ma, Y.; Luo, J.; Xu, Y. Co-preparation of pectin and cellulose from apple pomace by a sequential process. J. Food. Sci. Technol. 2019, 56, 4091–4100. [Google Scholar] [CrossRef]
- Bindon, K.A.; Bacic, A.; Kennedy, J.A. Tissue-specific and developmental modifications of grape cell walls influence the adsorption of proanthocyanidins. J. Agric. Food Chem. 2012, 60, 9249–9260. [Google Scholar] [CrossRef]
- Phan, A.D.T.; Flanagan, B.M.; D’Arcy, B.R.; Gidley, M.J. Binding selectivity of dietary polyphenols to different plant cell wall components: Quantification and mechanism. Food Chem. 2017, 233, 216–227. [Google Scholar] [CrossRef]
- Voragen, A.G.J.; Coenen, G.-J.; Verhoef, R.P.; Schols, H.A. Pectin, a versatile polysaccharide present in plant cell walls. Struct. Chem. 2009, 20, 263–275. [Google Scholar] [CrossRef]
- Thomas, M.; Guillemin, F.; Guillon, F.; Thibault, J.-F. Pectins in the fruits of Japanese quince (Chaenomeles japonica). Carbohydr. Polym. 2003, 53, 361–372. [Google Scholar] [CrossRef]
- Liu, Y.; Ying, D.; Sanguansri, L.; Cai, Y.; Le, X. Adsorption of catechin onto cellulose and its mechanism study: Kinetic models, characterization and molecular simulation. Food Res. Int. 2018, 112, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Selma, M.V.; Espin, J.C.; Tomás-Barberán, F.A. Interaction between phenolics and gut microbiota: Role in human health. J. Agric. Food Chem. 2009, 57, 6485–6501. [Google Scholar] [CrossRef] [PubMed]
- Healey, G.R.; Murphy, R.; Brough, L.; Butts, C.A.; Coad, J. Interindividual variability in gut microbiota and host response to dietary interventions. Nutr Rev. 2017, 75, 1059–1080. [Google Scholar] [CrossRef]
- Gross, G.; Jacobs, D.M.; Peters, S.; Possemiers, S.; van Duynhoven, J.; Vaughan, E.E.; van de Wiele, T. In vitro bioconversion of polyphenols from black tea and red wine/grape juice by human intestinal microbiota displays strong interindividual variability. J. Agric. Food Chem. 2010, 58, 10236–10246. [Google Scholar] [CrossRef]
- Hwang, C.S.; Kwak, H.S.; Lim, H.J.; Lee, S.H.; Kang, Y.S.; Choe, T.B.; Hur, H.G.; Han, K.O. Isoflavone metabolites and their in vitro dual functions: They can act as an estrogenic agonist or antagonist depending on the estrogen concentration. J. Steroid. Biochem. Mol. Biol. 2006, 101, 246–253. [Google Scholar] [CrossRef]
- Frankenfeld, C.L. O-desmethylangolensin: The importance of equol’s lesser known cousin to human health. Adv. Nutr. 2011, 2, 317–324. [Google Scholar] [CrossRef]
- Vázquez, L.; Llórez, A.B.; Redruello, B.; Mayo, B. Metabolism of soy isoflavones by intestinal bacteria: Genome analysis of an Adlercreutzia equolifaciens strain that does not produce equol. Biomolecules 2020, 10, 950. [Google Scholar] [CrossRef]
- Murota, K.; Nakamura, Y.; Uehara, M. Flavonoid metabolism: The interaction of metabolites and gut microbiota. Biosci. Biotechnol. Biochem. 2018, 82, 600–610. [Google Scholar] [CrossRef]
- Mace, T.A.; Ware, M.B.; King, S.A.; Loftus, S.; Farren, M.R.; McMichael, E.; Scoville, S.; Geraghty, C.; Young, G.; Carson, W.E., III; et al. Soy isoflavones and their metabolites modulate cytokine-induced natural killer cell function. Sci. Rep. 2019, 9, 5068. [Google Scholar] [CrossRef] [PubMed]
- Marín, L.; Miguélez, E.M.; Villar, C.J.; Lombó, F. Bioavailability of dietary polyphenols and gut microbiota metabolism: Antimicrobial properties. Biomed Res. Int. 2015, 2015, 905215. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Kim, S.-I.; Han, J.; Wang, X.-L.; Song, D.-G.; Kim, S.-U. Stereospecific biotransformation of dihydrodaidzein into (3S)-equol by the human intestinal bacterium Eggerthella strain Julong 732. Appl. Environ. Microbiol. 2009, 75, 3062–3068. [Google Scholar] [CrossRef] [PubMed]
- Braune, A.; Blaut, M. Bacterial species involved in the conversion of dietary flavonoids in the human gut. Gut Microbes 2016, 7, 216–234. [Google Scholar] [CrossRef] [PubMed]
- Decroos, K.; Vanhemmens, S.; Cattoir, S.; Boon, N.; Verstraete, W. Isolation and characterisation of an equol-producing mixed microbial culture from a human faecal sample and its activity under gastrointestinal conditions. Arch. Microbiol. 2005, 183, 45–55. [Google Scholar] [CrossRef]
- Hur, H.-G.; Beger, R.D.; Heinze, T.M.; Lay, J.O., Jr.; Freeman, J.P.; Dore, J.; Rafii, F. Isolation of an anaerobic intestinal bacterium capable of cleaving the C-ring of the isoflavonoid daidzein. Arch. Microbiol. 2002, 178, 8–12. [Google Scholar] [CrossRef]
- Rossi, M.; Amaretti, A.; Roncaglia, L.; Leonardi, A.; Raimondi, S. Dietary isoflavones and intestinal microbiota: Metabolism and transformation into bioactive compounds. In Isoflavones Biosynthesis, Occurence and Health Effects; Thompson, M.J., Ed.; Nova Science Publishers Inc.: New York, NY, USA, 2010; pp. 137–216. ISBN 9781617281136. [Google Scholar]
- Schoefer, L.; Mohan, R.; Braune, A.; Birringer, M.; Blaut, M. Anaerobic C-ring cleavage of genistein and daidzein by Eubacterium ramulus. FEMS Microbiol. Lett. 2002, 208, 197–202. [Google Scholar] [CrossRef]
- Kim, M.; Han, J.; Kim, S.-U. Isoflavone daidzein: Chemistry and bacterial metabolism. J. Appl. Biol. Chem. 2008, 51, 253–261. [Google Scholar] [CrossRef]
- Matthies, A.; Clavel, T.; Gütschow, M.; Engst, W.; Haller, D.; Blaut, M.; Braune, A. Conversion of daidzein and genistein by an anaerobic bacterium newly isolated from the mouse intestine. Appl. Environ. Microbiol. 2008, 74, 4847–4852. [Google Scholar] [CrossRef]
- Matthies, A.; Loh, G.; Blaut, M.; Braune, A. Daidzein and genistein are converted to equol and 5-hydroxy-equol by human intestinal Slackia isoflavoniconvertens in gnotobiotic rats. J. Nutr. 2012, 142, 40–46. [Google Scholar] [CrossRef]
- Wang, X.-L.; Kim, H.-K.; Kang, S.-I.; Kim, S.-U.; Hur, H.-G. Production of phytoestrogen S-equol from daidzein in mixed culture of two anaerobic bacteria. Arch. Microbiol. 2007, 187, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, S.-i.; Suzuki, T. Isolation and characterization of a novel equol-producing bacterium from human feces. Biosci. Biotechnol. Biochem. 2008, 72, 2660–2666. [Google Scholar] [CrossRef] [PubMed]
- Tamura, M.; Tsushida, T.; Shinohara, K. Isolation of an isoflavone-metabolizing, Clostridium-like bacterium, strainTM-40, from human faeces. Anaerobe 2007, 13, 32–35. [Google Scholar] [CrossRef]
- Jin, J.-S.; Kitahara, M.; Sakamoto, M.; Hattori, M.; Benno, Y. Slackia equolifaciens sp. nov., a human intestinal bacterium capable of producing equol. Int. J. Syst. Evol. Microbiol. 2010, 60, 1721–1724. [Google Scholar] [CrossRef] [PubMed]
- Shimada, Y.; Yasuda, S.; Takahashi, M.; Hayashi, T.; Miyazawa, N.; Sato, I.; Abiru, Y.; Uchiyama, S.; Hishigaki, H. Cloning and expression of a novel NADP(H)-dependent daidzein reductase, an enzyme involved in the metabolism of daidzein, from equol-producing Lactococcus Strain 20-92. Appl. Environ. Microbiol. 2010, 76, 5892–5901. [Google Scholar] [CrossRef]
- Wang, X.L.; Kim, K.T.; Lee, J.H.; Hur, H.G.; Kim, S.I. C-Ring cleavage of isoflavones daidzein and genistein by a newly-isolated human intestinal bacterium Eubacterium ramulus Julong 601. J. Microbiol. Biotechnol. 2004, 14, 766–771. [Google Scholar]
- Hur, H.-G.; Lay, J.O., Jr.; Beger, R.D.; Freeman, J.P.; Rafii, F. Isolation of human intestinal bacteria metabolizing the natural isoflavone glycosides daidzin and genistin. Arch. Microbiol. 2000, 174, 422–428. [Google Scholar] [CrossRef]
- Jin, J.-S.; Nishihata, T.; Kakiuchi, N.; Hattori, M. Biotransformation of C-glucosylisoflavone puerarin to estrogenic (3S)-equol in co-culture of two human intestinal bacteria. Biol. Pharm. Bull. 2008, 31, 1621–1625. [Google Scholar] [CrossRef]
- Hur, H.-G.; Raffi, F. Biotransformation of the isoflavonoids biochanin A, formononetin, and glycitein by Eubacterium limosum. FEMS Microbiol. Lett. 2000, 192, 21–25. [Google Scholar] [CrossRef]
- Heinonen, S.-M.; Wähälä, K.; Adlercreutz, H. Identification of urinary metabolites of the red clover isoflavones formononetin and biochanin A in human subjects. J. Agric. Food Chem. 2004, 52, 6802–6809. [Google Scholar] [CrossRef]
- Carreau, C.; Flouriot, G.; Bennetau-Pelissero, C.; Potier, M. Enterodiol and enterolactone, two major diet-derived polyphenol metabolites have different impact on ER_ transcriptional activation in human breast cancer cells. J. Steroid Biochem. Mol. 2008, 110, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.-Q. Mammalian phytoestrogens: Enterodiol and enterolactone. J. Chromatogr. B 2002, 777, 289–309. [Google Scholar] [CrossRef]
- Dinkova-Kostova, A.T.; Gang, D.R.; Davin, L.B.; Bedgar, D.L.; Chu, A.; Lewis, N.G. (1)-pinoresinol/(1)-lariciresinol reductase from Forsythia intermedia. J. Biol. Chem. 1996, 271, 29473–29482. [Google Scholar] [CrossRef] [PubMed]
- Landete, J.M. Plant and mammalian lignans: A review of source, intake, metabolism, intestinal bacteria and health. Food Res. Int. 2012, 46, 410–424. [Google Scholar] [CrossRef]
- Yoder, S.C.; Lancaster, S.M.; Hullar, M.A.J.; Lampe, J.W. Gut microbial metabolism of plant lignans: Influence on human health. In Diet-Microbe Interactions in the Gut. Effects on Human Health and Disease; Tuohy, K., Del Rio, D., Eds.; Elsevier Inc.: Oxford, UK, 2015; pp. 103–117. [Google Scholar] [CrossRef]
- Wang, L.-Q.; Meselhy, M.R.; Li, Y.; Qin, G.-W.; Hattori, M. Human intestinal bacteria capable of transforming secoisolariciresinol diglucoside to mammalian lignans, enterodiol and enterolactone. Chem. Pharm. Bull. 2000, 48, 1606–1610. [Google Scholar] [CrossRef] [PubMed]
- Clavel, T.; Lippman, R.; Gavini, F.; Dore´, J.; Blaut, M. Clostridium saccharogumia sp. nov. and Lactonifactor longoviformis gen. nov., sp. nov., two novel human faecal bacteria involved in the conversion of the dietary phytoestrogen secoisolariciresinol diglucoside. Syst. Appl. Microbiol. 2007, 30, 16–26. [Google Scholar] [CrossRef]
- Clavel, T.; Henderson, G.; Alpert, C.-A.; Philippe, C.; Rigottier-Gois, L.; Doré, J.; Blaut, M. Intestinal bacterial communities that produce active estrogen-like compounds enterodiol and enterolactone in humans. Appl. Environ. Microbiol. 2005, 71, 6077–6085. [Google Scholar] [CrossRef]
- Peñalvo, J.L.; Heinonen, S.-M.; Aura, A.-M.; Adlercreutz, H. Dietary sesamin is converted to enterolactone in humans. J. Nutr. 2005, 135, 1056–1062. [Google Scholar] [CrossRef]
- Villalba, K.J.O.; Barka, F.V.; Pasos, C.V.; Rodríguez, P.E. Food ellagitannins: Structure, metabolomic fate, and biological properties. In Tannins—Structural Properties, Biological Properties and Current Knowledge Food Ellagitannins: Structure, Metabolomic Fate, and Biological Properties; Aires, A., Ed.; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Wu, S.; Tian, L. Diverse phytochemicals and bioactivities in the ancient fruit and modern functional food pomegranate (Punica granatum). Molecules 2017, 22, 1606. [Google Scholar] [CrossRef]
- Larrosa, M.; Tomás-Barberán, F.A.; Espín, J.C. The dietary hydrolysable tannin punicalagin releases ellagic acid that induces apoptosis in human colon adenocarcinoma Caco-2 cells by using the mitochondrial pathway. J. Nutr. Biochem. 2006, 17, 611–625. [Google Scholar] [CrossRef]
- Selma, M.V.; Beltrán, D.; García-Villalba, R.; Espín, J.C.; Tomás-Barberán, F.A. Description of urolithin production capacity from ellagic acid of two human intestinal Gordonibacter species. Food Funct. 2014, 5, 1779–1784. [Google Scholar] [CrossRef] [PubMed]
- Selma, M.V.; Tomás-Barberán, F.A.; Beltran, D.; Garcıa-Villalba, R.; Espın, J.C. Gordonibacter urolithinfaciens sp. nov., a urolithin-producing bacterium isolated from the human gut. Int. J. Syst. Evol. Microbiol. 2014, 64, 2346–2352. [Google Scholar] [CrossRef] [PubMed]
- Beltrȧn, D.; Romo-Vaquero, M.; Espín, J.C.; Tomȧs-Barberȧn, F.A.; Selma, M.V. Ellagibacter isourolithinifaciens gen. nov., sp. nov., a new member of the family Eggerthellaceae, isolated from human gut. Int. J. Syst. Evol. Microbiol. 2018, 68, 1707–1712. [Google Scholar] [CrossRef] [PubMed]
- Espín, J.C.; Larrosa, M.; García-Conesa, M.T.; Tomás-Barberán, F. Biological significance of urolithins, the gut microbial ellagic acid-derived metabolites: The evidence so far. Evid.-Based Complementary Altern. Med. 2013, 2013, 1–16. [Google Scholar] [CrossRef]
- Tomás-Barberán, F.A.; González-Sarrías, A.; García-Villalba, R.; Núñez-Sánchez, M.A.; Selma, M.V.; García-Conesa, M.T.; Espín, J.C. Urolithins, the rescue of “old” metabolites to understand a “new” concept: Metabotypes as a nexus among phenolic metabolism, microbiota dysbiosis, and host health status. Mol. Nutr. Food Res. 2017, 61, 1500901. [Google Scholar] [CrossRef]
- Kawabata, K.; Yoshioka, Y.; Terao, J. Role of intestinal microbiota in the bioavailability and physiological functions of dietary polyphenols. Molecules 2019, 24, 370. [Google Scholar] [CrossRef]
- Possemiers, S.; Heyerick, A.; Robbens, V.; Keukeleire, D.D.; Verstraete, W. Activation of proestrogens from hops (Humulus lupulus L.) by intestinal microbiota; conversion of isoxanthohumol into 8-prenylnaringenin. J. Agric. Food Chem. 2005, 53, 6281–6288. [Google Scholar] [CrossRef]
- Paraiso, I.L.; Plagmann, L.S.; Yang, L.; Zielke, R.; Gombart, A.F.; Maier, C.S.; Sikora, A.E.; Blakemore, P.R.; Stevens, J.F. Reductive metabolism of xanthohumol and 8-prenylnaringenin by the intestinal bacterium Eubacterium ramulus. Mol. Nutr. Food Res. 2019, 63, 1800923. [Google Scholar] [CrossRef]
- Nikolic, D.; Li, Y.; Chadwick, L.R.; Pauli, G.F.; van Breemen, R.B. Metabolism of xanthohumol and isoxanthohumol, prenylated flavonoids from hops (Humulus lupulus L.), by human liver microsomes. J. Mass Spectrom. 2005, 40, 289–299. [Google Scholar] [CrossRef]
- Pepper, M.S.; Hazel, S.J.; Hümpel, M.; Schleuning, W.-D. 8-prenylnaringenin, a novel phytoestrogen, inhibits angiogenesis in vitro and in vivo. J. Cell. Physiol. 2004, 199, 98–107. [Google Scholar] [CrossRef]
- Hameed, A.S.S.; Rawat, P.S.; Meng, X.; Liu, W. Biotransformation of dietary phytoestrogens by gut microbes: A review on bidirectional interaction between phytoestrogen metabolism and gut microbiota. Biotechnol. Adv. 2020, 43, 107576. [Google Scholar] [CrossRef] [PubMed]
- Clifford, M.N. Review. Anthocyanins—Nature, occurrence and dietary burden. J. Sci. Food Agric. 2000, 80, 1063–1072. [Google Scholar] [CrossRef]
- Aura, A.-M.; Martin-Lopez, P.; O’Leary, K.A.; Williamson, G.; Oksman-Caldentey, K.-M.; Poutanen, K.; Santos-Buelga, C. In vitro metabolism of anthocyanins by human gut microflora. Eur. J. Nutr. 2005, 44, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Keppler, K.; Humpf, H.-U. Metabolism of anthocyanins and their phenolic degradation products by the intestinal microflora. Bioorg. Med. Chem. 2005, 13, 5195–5205. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Barrio, R.; Edwards, C.A.; Crozier, A. Colonic catabolism of ellagitannins, ellagic acid, and raspberry anthocyanins: In vivo and in vitro studies. Drug Metab. Dispos. 2011, 39, 1680–1688. [Google Scholar] [CrossRef]
- Zhu, Y.; Sun, H.; He, S.; Lou, Q.; Yu, M.; Tang, M.; Tu, L. Metabolism and prebiotics activity of anthocyanins from black rice (Oryza sativa L.) in vitro. PLoS ONE 2018, 13, e0195754. [Google Scholar] [CrossRef]
- Ávila, M.; Hidalgo, M.; Sánchez-Moreno, C.; Pelaez, C.; Requena, T.; de Pascual-Teresa, S. Bioconversion of anthocyanin glycosides by Bifidobacteria and Lactobacillus. Food Res. Int. 2009, 42, 1453–1461. [Google Scholar] [CrossRef]
- Hidalgo, M.; Oruna-Concha, J.; Kolida, S.; Walton, G.E.; Kallithraka, S.; Spencer, J.P.E.; Gibson, G.; de Pascual-Teresa, S. Metabolism of anthocyanins by human gut microflora and their influence on gut bacterial growth. J. Agric. Food Chem. 2012, 60, 3882–3890. [Google Scholar] [CrossRef]
- Fleschhut, J.; Kratzer, F.; Rechkemmer, G.; Kulling, S.E. Stability and biotransformation of various dietary anthocyanins in vitro. Eur. J. Nutr. 2006, 45, 7–18. [Google Scholar] [CrossRef]
- Racova, Z.; Anzenbacherova, E.; Papouskova, B.; Poschner, S.; Kucova, P.; Gausterer, J.C.; Gabor, F.; Kolar, M.; Anzenbacher, P. Metabolite profiling of natural substances in human: in vitro study from fecal bacteria to colon carcinoma cells (Caco-2). J. Nutr. Biochem. 2020, 85, 108482. [Google Scholar] [CrossRef]
- Xie, L.; Lee, S.G.; Vance, T.M.; Wang, Y.; Kim, B.; Lee, J.-Y.; Chun, O.K.; Bolling, B.W. Bioavailability of anthocyanins and colonic polyphenol metabolites following consumption of aronia berry extract. Food Chem. 2016, 211, 860–868. [Google Scholar] [CrossRef]
- Chen, Y.; Li, Q.; Zhao, T.; Zhang, Z.; Mao, G.; Feng, W.; Wu, X.; Yang, L. Biotransformation and metabolism of three mulberry anthocyanin monomers by rat gut microflora. Food Chem. 2017, 237, 887–894. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Christman, L.M.; Li, R.; Gu, L. Synergic interactions between polyphenols and gut microbiota in mitigating inflammatory bowel diseases. Food Funct. 2020, 11, 4878–4891. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, P.; Araújo, P.; Oliveira, J.; Fraga, I.; Pissarra, J.; Amaral, C. Metabolic pathways of degradation of malvidin-3-O-monoglucoside by Candida oleophila. Int. Biodeterior. Biodegrad. 2019, 144, 104768. [Google Scholar] [CrossRef]
- Sánchez-Patán, F.; Cueva, C.; Monagas, M.; Walton, G.E.; Gibson, G.R.; Martín-Álvarez, P.J.; Moreno-Arribas, M.V.; Bartolomé, B. Gut microbial catabolism of grape seed flavan-3-ols by human faecal microbiota. Targetted analysis of precursor compounds, intermediate metabolites and end-products. Food Chem. 2012, 131, 337–347. [Google Scholar] [CrossRef]
- Mena, P.; Calani, L.; Bruni, R.; Del Rio, D. Bioactivation of high-molecular-weight polyphenols by the gut microbiome. In Diet-Microbe Interactions in the Gut. Effects on Human Health and Disease; Tuohy, K., Del Rio, D., Eds.; Elsevier Inc.: Oxford, UK, 2015; pp. 73–101. [Google Scholar] [CrossRef]
- Kutschera, M.; Engst, W.; Blaut, M.; Braune, A. Isolation of catechin-converting human intestinal bacteria. J. Appl. Microbiol. 2011, 111, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Appeldoorn, M.M.; Vincken, J.-P.; Gruppen, H.; Hollman, P.C.H. Procyanidin dimers A1, A2, and B2 are absorbed without conjugation or methylation from the small intestine of rats. J. Nutr. 2009, 139, 1469–1473. [Google Scholar] [CrossRef] [PubMed]
- Déprez, S.; Brezillon, C.; Rabot, S.; Philippe, C.; Mila, I.; Lapierre, C.; Scalbert, A. Polymeric proanthocyanidins are catabolized by human colonic microflora into low-molecular-weight phenolic acids. J. Nutr. 2000, 130, 2733–2738. [Google Scholar] [CrossRef]
- Ou, K.; Sarnoski, P.; Schneider, K.R.; Song, K.; Khoo, C.; Gu, L. Microbial catabolism of procyanidins by human gut microbiota. Mol. Nutr. Food Res. 2014, 58, 2196–2205. [Google Scholar] [CrossRef]
- Monagas, M.; Urpi-Sarda, M.; Sánchez-Patán, F.; Llorach, R.; Garrido, I.; Gómez-Cordovés, C.; Andres-Lacueva, C.; Bartolomé, B. Insights into the metabolism and microbial biotransformation of dietary flavan-3-ols and the bioactivity of their metabolites. Food Funct. 2010, 1, 233–253. [Google Scholar] [CrossRef]
- Alakomi, H.L.; Puupponen-Pimiä, R.; Aura, A.M.; Helander, I.M.; Nohynek, L.; Oksman-Caldentey, K.M.; Saarela, M. Weakening of Salmonella with selected microbial metabolites of berry-derived phenolic compounds and organic acids. J. Agric. Food Chem. 2007, 55, 3905–3912. [Google Scholar] [CrossRef] [PubMed]
- Braune, A.; Blaut, M. Deglycosylation of puerarin and other aromatic C-glucosides by a newly isolated human intestinal bacterium. Environ. Microbiol. 2011, 13, 482–494. [Google Scholar] [CrossRef] [PubMed]
- Ahn, H.J.; You, H.L.; Park, M.S.; Li, Z.; Choe, D.; Johnston, T.V.; Ku, S.; Ji, G.E. Microbial biocatalysis of quercetin-3-glucoside and isorhamnetin-3-glucoside in Salicornia herbacea and their contribution to improved anti-inflammatory activity. RSC Adv. 2020, 10, 5339–5350. [Google Scholar] [CrossRef]
- Schoefer, L.; Mohan, R.; Schwiertz, A.; Braune, A.; Blaut, M. Anaerobic degradation of flavonoids by Clostridium orbiscindens. Appl. Environ. Microbiol. 2003, 69, 5849–5854. [Google Scholar] [CrossRef]
- Aura, A.-M. Microbial metabolism of dietary phenolic compounds in the colon. Phytochem. Rev. 2008, 7, 407–429. [Google Scholar] [CrossRef]
- Corrêa, T.A.F.; Rogero, M.M.; Hassimotto, N.M.A.; Lajolo, F.M. The two-way polyphenols-microbiota interactions and their effects on obesity and related metabolic diseases. Front. Nutr. 2019, 6, 188. [Google Scholar] [CrossRef]
- Hanske, L.; Loh, G.; Sczesny, S.; Blaut, M.; Braune, A. The bioavailability of apigenin-7-glucoside is influenced by human intestinal microbiota in rats. J. Nutr. 2009, 139, 1095–1102. [Google Scholar] [CrossRef]
- Schneider, H.; Blaut, M. Anaerobic degradation of flavonoids by Eubacterium ramulus. Arch. Microbiol. 2000, 173, 71–75. [Google Scholar] [CrossRef]
- Nunes, C.; Almeida, L.; Laranjinha, J. Synergistic inhibition of respiration in brain mitochondria by nitric oxide and dihydroxyphenylacetic acid (DOPAC): Implications for Parkinson’s disease. Neurochem. Int. 2005, 47, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Najmanová, I.; Pourová, J.; Vopršalová, M.; Pilařová, V.; Semecký, V.; Nováková, L.; Mladěnka, P. Flavonoid metabolite 3-(3-hydroxyphenyl)propionic acid formed by human microflora decreases arterial blood pressure in rats. Mol. Nutr. Food Res. 2016, 60, 981–991. [Google Scholar] [CrossRef] [PubMed]
- Rechner, A.R.; Smith, M.A.; Kuhnle, G.; Gibson, G.R.; Debnam, E.S.; Srai, S.K.S.; Moore, K.P.; Rice-Evans, C.A. Colonic metabolism of dietary polyphenols: Influence of structure on microbial fermentation products. Free Rad. Biol. Med. 2004, 36, 212–225. [Google Scholar] [CrossRef] [PubMed]
- Sova, M.; Saso, L. Natural sources, pharmacokinetics, biological activities and health benefits of hydroxycinnamic acids and their metabolites. Nutrients 2020, 12, 2190. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Gao, W.; Chen, X.; Wang, H. The effect of bioprocessing on the phenolic acid composition and antioxidant activity of wheat bran. Cereal Chem. 2014, 91, 255–261. [Google Scholar] [CrossRef]
- Farah, A.; Monteiro, M.; Donangelo, C.M.; Lafay, S. Chlorogenic acids from green coffee extract are highly bioavailable in humans. J. Nutr. 2008, 138, 2309–2315. [Google Scholar] [CrossRef] [PubMed]
- Gonthier, M.P.; Verny, M.A.; Besson, C.; Rémésy, C.; Scalbert, A. Chlorogenic acid bioavailability largely depends on its metabolism by the gut microflora in rats. J. Nutr. 2003, 133, 1853–1859. [Google Scholar] [CrossRef] [PubMed]
- García-Villalba, R.; Beltrán, D.; Frutos, M.D.; Selma, M.V.; Espín, J.C.; Tomás-Barberán, F.A. Metabolism of different dietary phenolic compounds by the urolithin-producing human-gut bacteria Gordonibacter urolithinfaciens and Ellagibacter isourolithinifaciens. Food Funct. 2020, 11, 7012–7022. [Google Scholar] [CrossRef]
- Mills, C.E.; Tzounis, X.; Oruna-Concha, M.J.; Mottram, D.S.; Gibson, G.R.; Spencer, J.P. In vitro colonic metabolism of coffee and chlorogenic acid results in selective changes in human faecal microbiota growth. Br. J. Nutr. 2015, 113, 1220–1227. [Google Scholar] [CrossRef]
- Monteiro, M.; Farah, A.; Perrone, D.; Trugo, L.C.; Donangelo, C. Chlorogenic acid compounds from coffee are differentially absorbed and metabolized in humans. J. Nutr. 2007, 137, 2196–2201. [Google Scholar] [CrossRef]
- Ogawa, M. Coffee and hippuric acid. In Coffee in Health and Disease Prevention; Preedy, V.R., Ed.; Academic Press: Cambridge, MA, USA, 2014; pp. 209–216. [Google Scholar]
- Jung, C.M.; Heinze, T.M.; Schnackenberg, L.K.; Mullis, L.B.; Elkins, S.A.; Elkins, C.A.; Steele, R.S.; Sutherland, J.B. Interaction of dietary resveratrol with animal-associated bacteria. FEMS Microbiol. Lett. 2009, 297, 266–273. [Google Scholar] [CrossRef]
- Bode, L.M.; Bunzel, D.; Huch, M.; Cho, G.S.; Ruhland, D.; Bunzel, M.; Bub, A.; Franz, C.M.; Kulling, S.E. In vivo and in vitro metabolism of trans-resveratrol by human gut microbiota. Am. J. Clin. Nutr. 2013, 97, 295–309. [Google Scholar] [CrossRef]
- Springer, M.; Moco, S. Resveratrol and its human metabolites-effects on metabolic health and obesity. Nutrients 2019, 11, 143. [Google Scholar] [CrossRef] [PubMed]
- Chaplin, A.; Carpéné, C.; Mercader, J. Resveratrol, metabolic syndrome, and gut microbiota. Nutrients 2018, 10, 1651. [Google Scholar] [CrossRef] [PubMed]
- Ito-Nagahata, T.; Kurihara, C.; Hasebe, M.; Ishii, A.; Yamashita, K.; Iwabuchi, M.; Sonoda, M.; Fukuhara, K.; Sawada, R.; Matsuoka, A.; et al. Stilbene analogs of resveratrol improve insulin resistance through activation of AMPK. Biosci. Biotechnol. Biochem. 2013, 77, 1229–1235. [Google Scholar] [CrossRef] [PubMed]
- Jarosova, V.; Vesely, O.; Marsik, P.; Jaimes, J.D.; Smejkal, K.; Kloucek, P.; Havlik, J. Metabolism of stilbenoids by human faecal microbiota. Molecules 2019, 24, 1155. [Google Scholar] [CrossRef]
- Scazzocchio, B.; Minghetti, L.; D’Archivio, M. Interaction between gut microbiota and curcumin: A new key of understanding for the health effects of curcumin. Nutrients 2020, 12, 2499. [Google Scholar] [CrossRef]
- Tan, S.; Calani, L.; Bresciani, L.; Dall’asta, M.; Faccini, A.; Augustin, M.A.; Gras, S.L.; Del Rio, D. The degradation of curcuminoids in a human faecal fermentation model. Int. J. Food Sci. Nutr. 2015, 66, 790–796. [Google Scholar] [CrossRef]
- Hassaninasab, A.; Hashimoto, Y.; Tomita-Yokotani, K.; Kobayashi, M. Discovery of the curcumin metabolic pathway involving a unique enzyme in an intestinal microorganism. Proc. Natl. Acad. Sci. USA 2011, 108, 6615–6620. [Google Scholar] [CrossRef]
- Burapan, S.; Kim, M.; Han, J. Curcuminoid demethylation as an alternative metabolism by human intestinal microbiota. J. Agric. Food Chem. 2017, 65, 3305–3310. [Google Scholar] [CrossRef]
- Bresciani, L.; Favari, C.; Calani, L.; Francinelli, V.; Riva, A.; Petrangolini, G.; Allegrini, P.; Mena, P.; Del Rio, D. The effect of formulation of curcuminoids on their metabolism by human colonic microbiota. Molecules 2020, 25, 940. [Google Scholar] [CrossRef]
- Anand, P.; Kunnumakkara, A.J.; Newman, R.A.; Aggarwal, B.B. Bioavailability of curcumin: Problems and promises. Mol. Pharmac. 2007, 4, 807–818. [Google Scholar] [CrossRef]
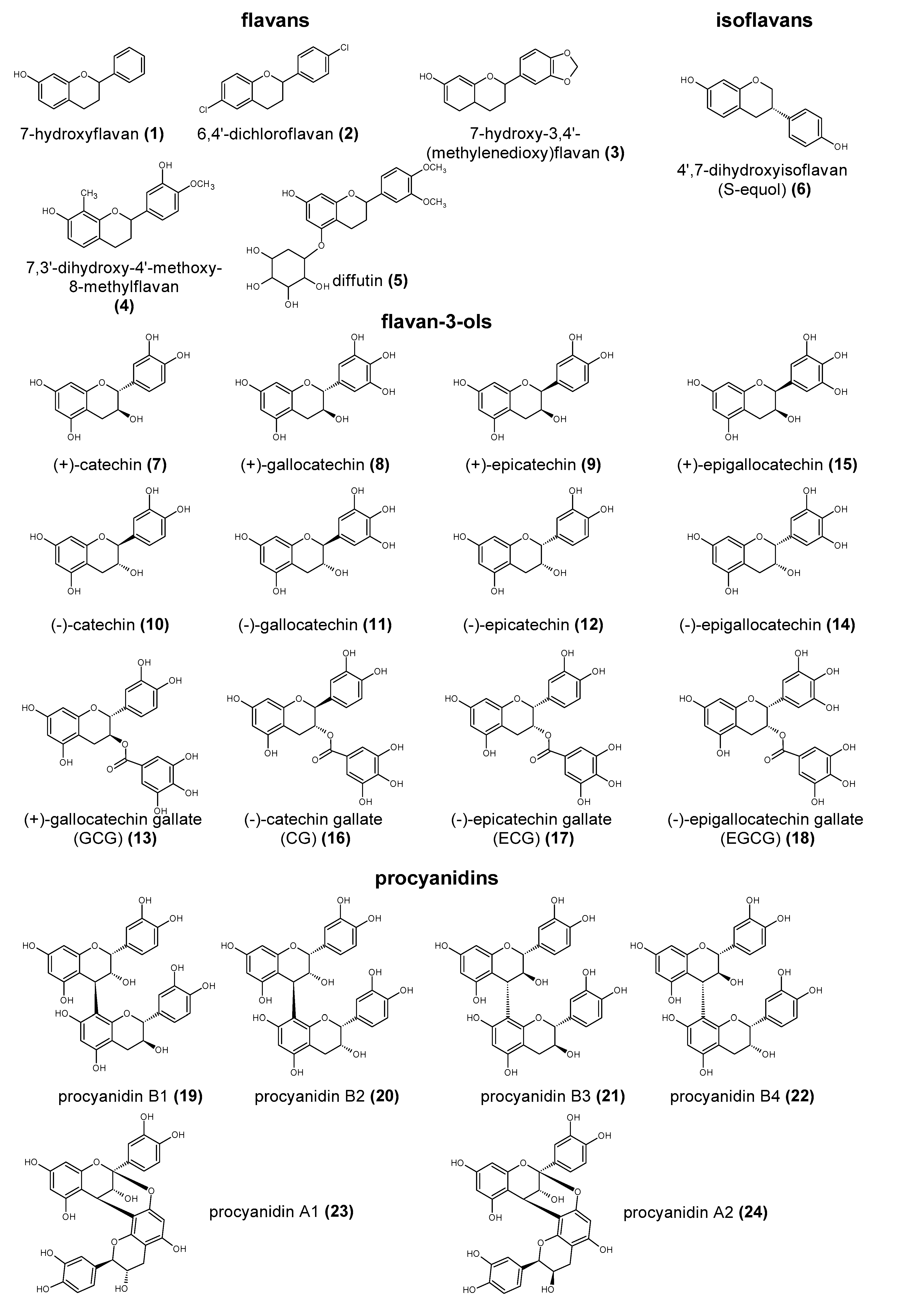
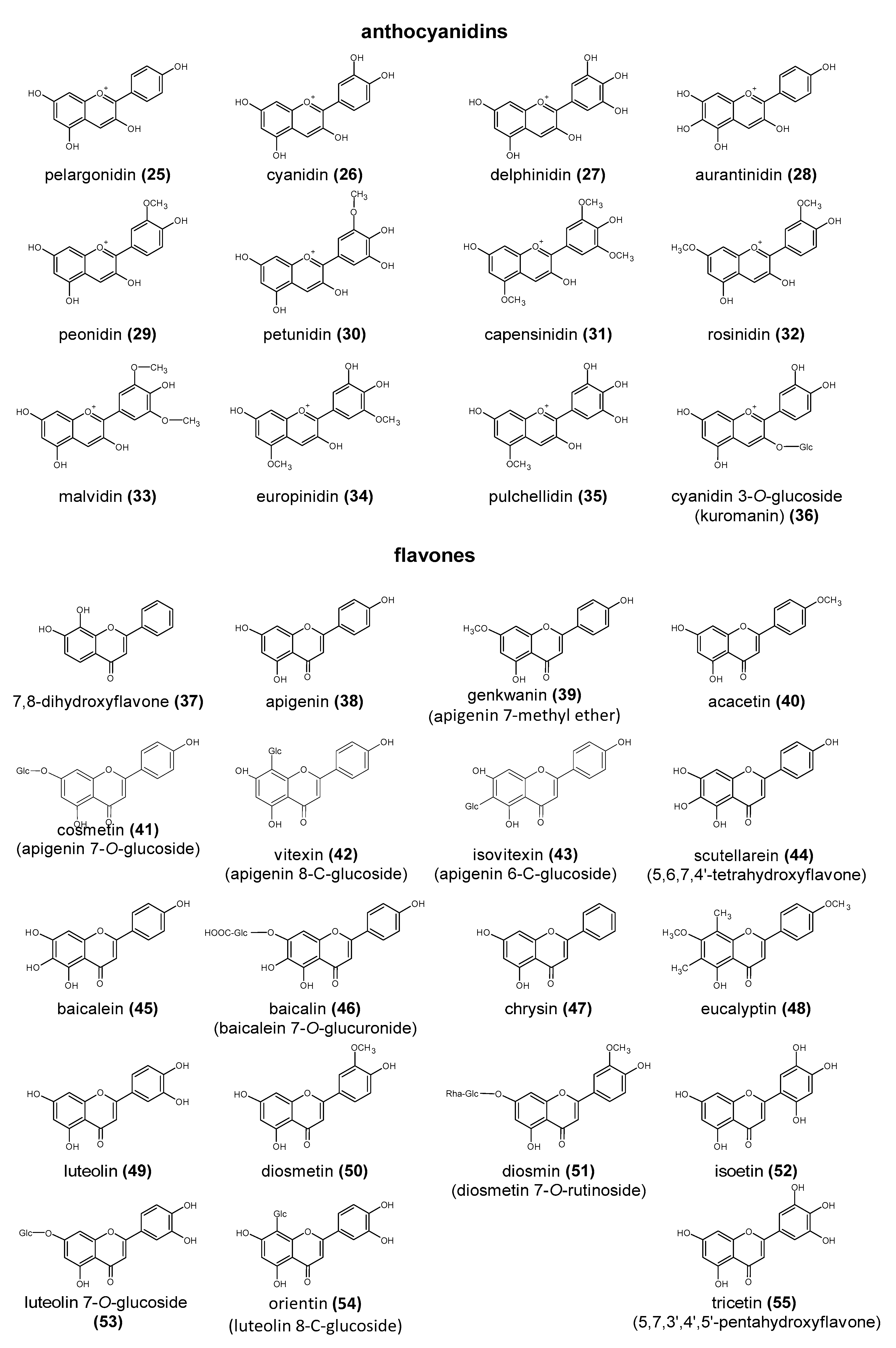
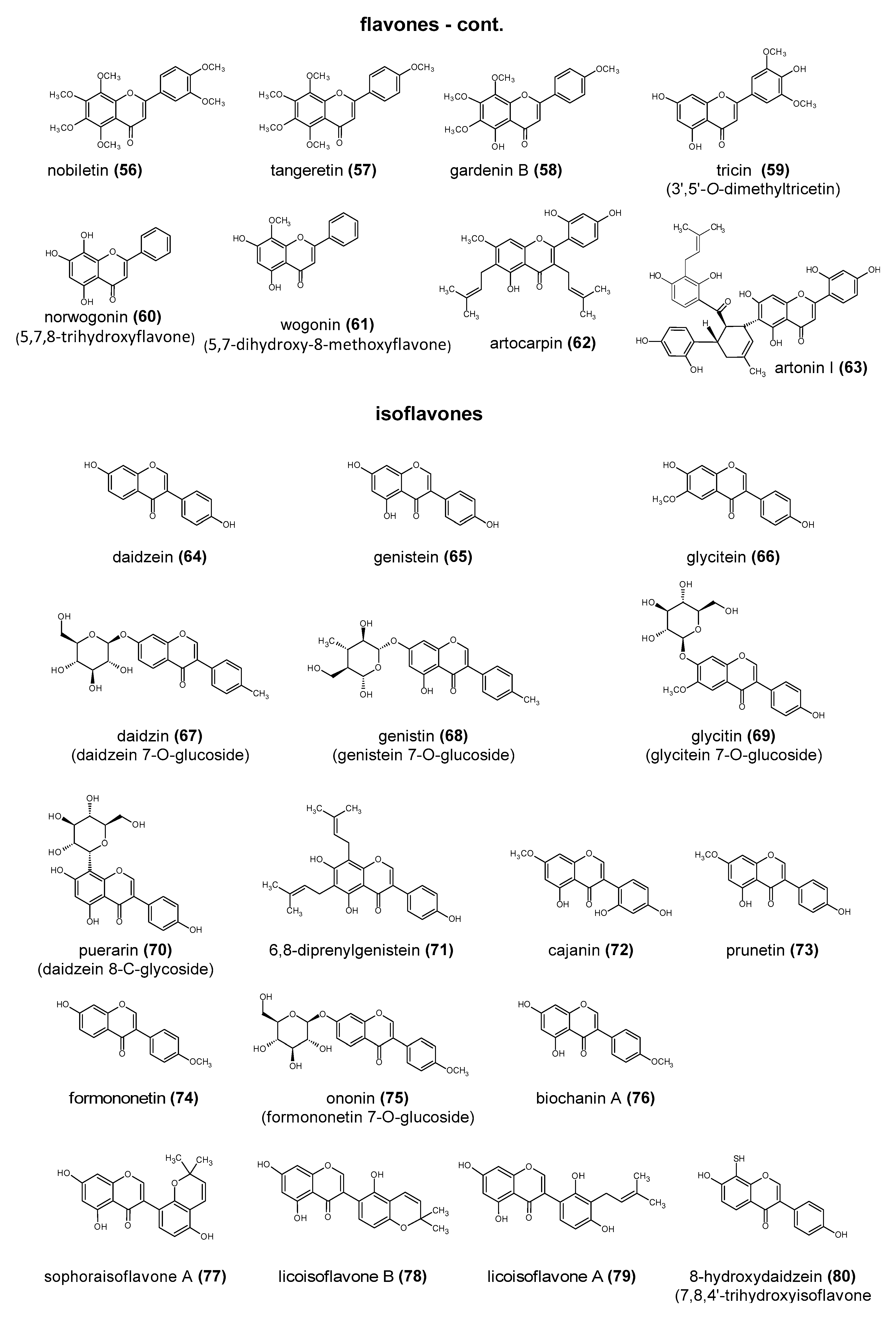
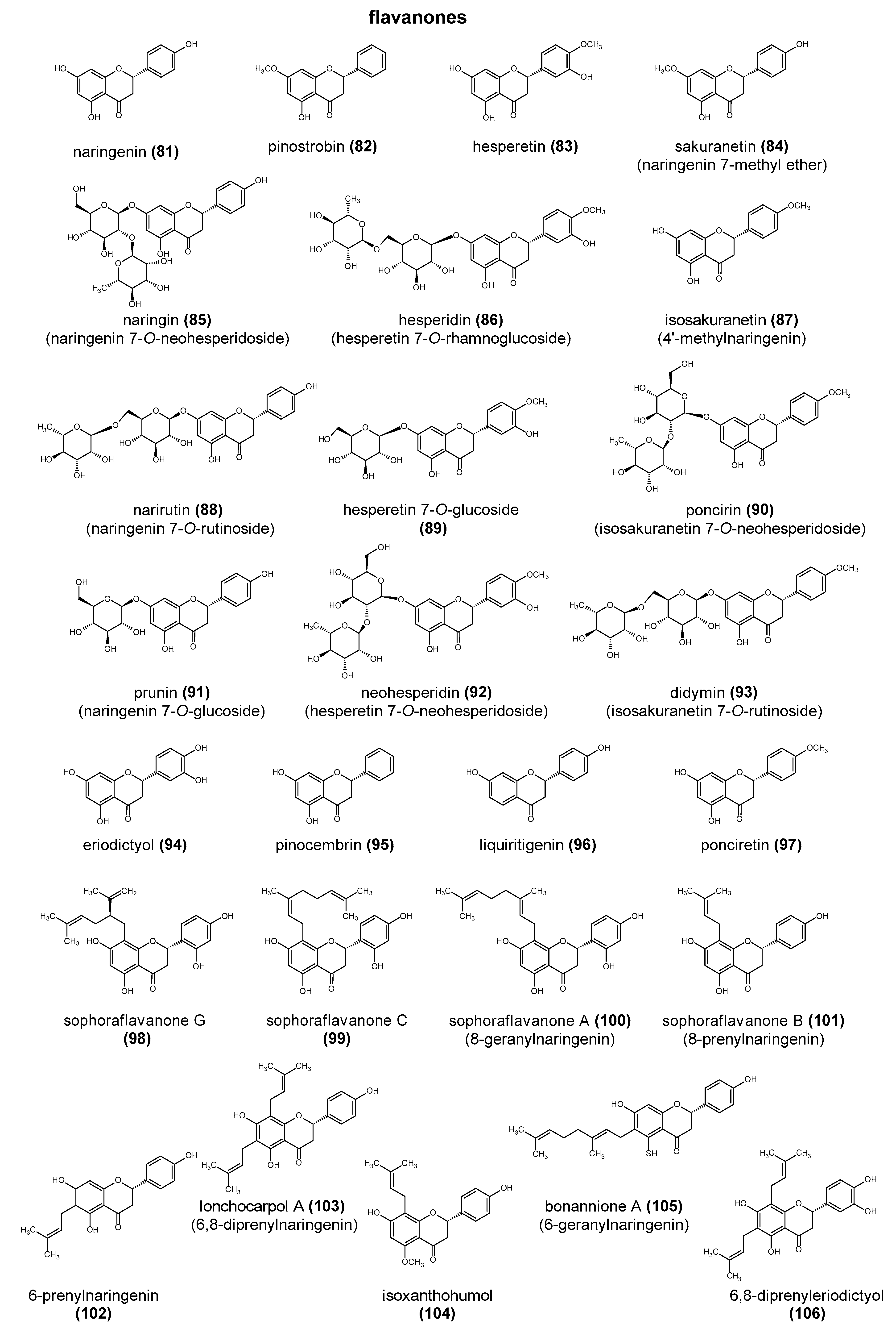

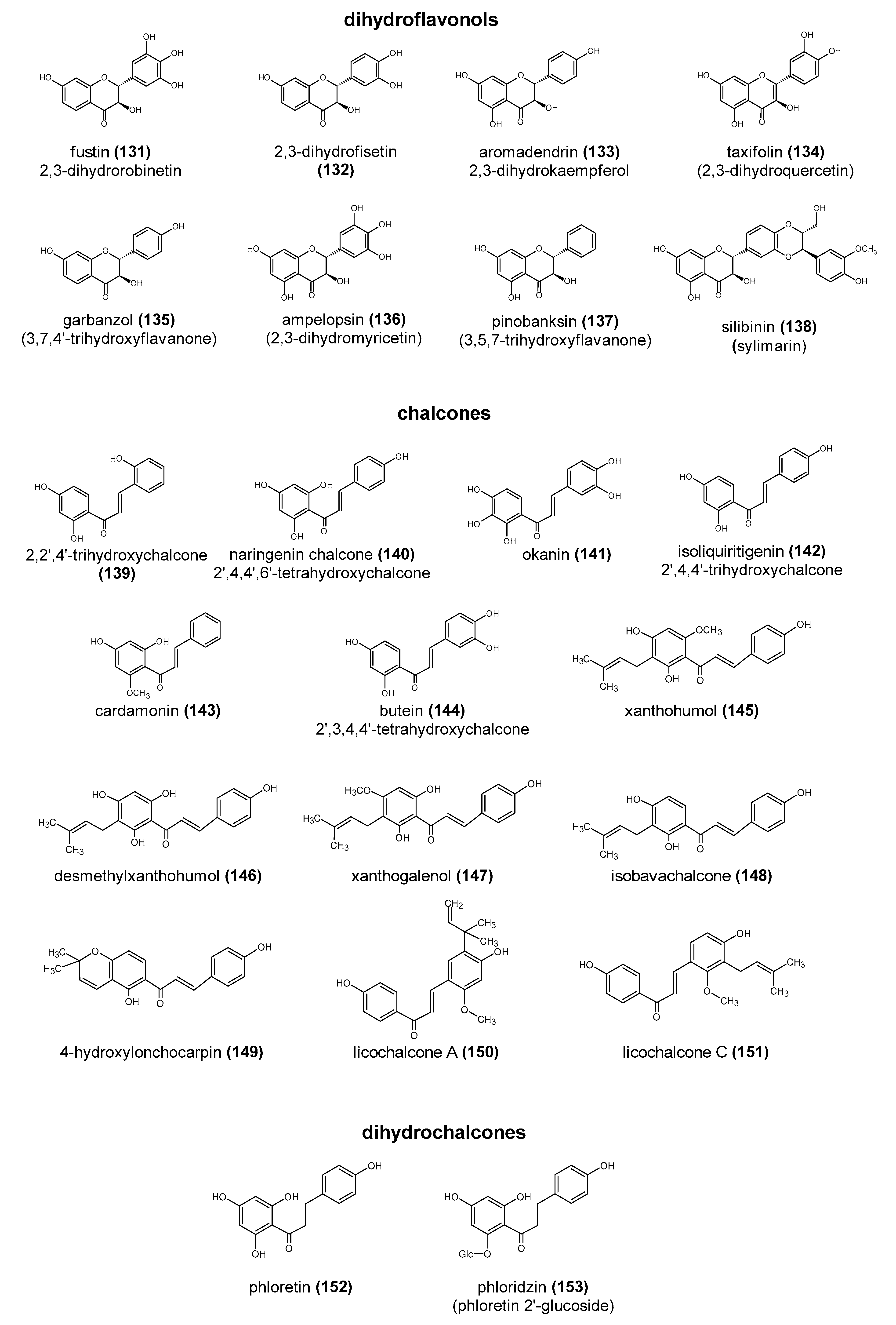
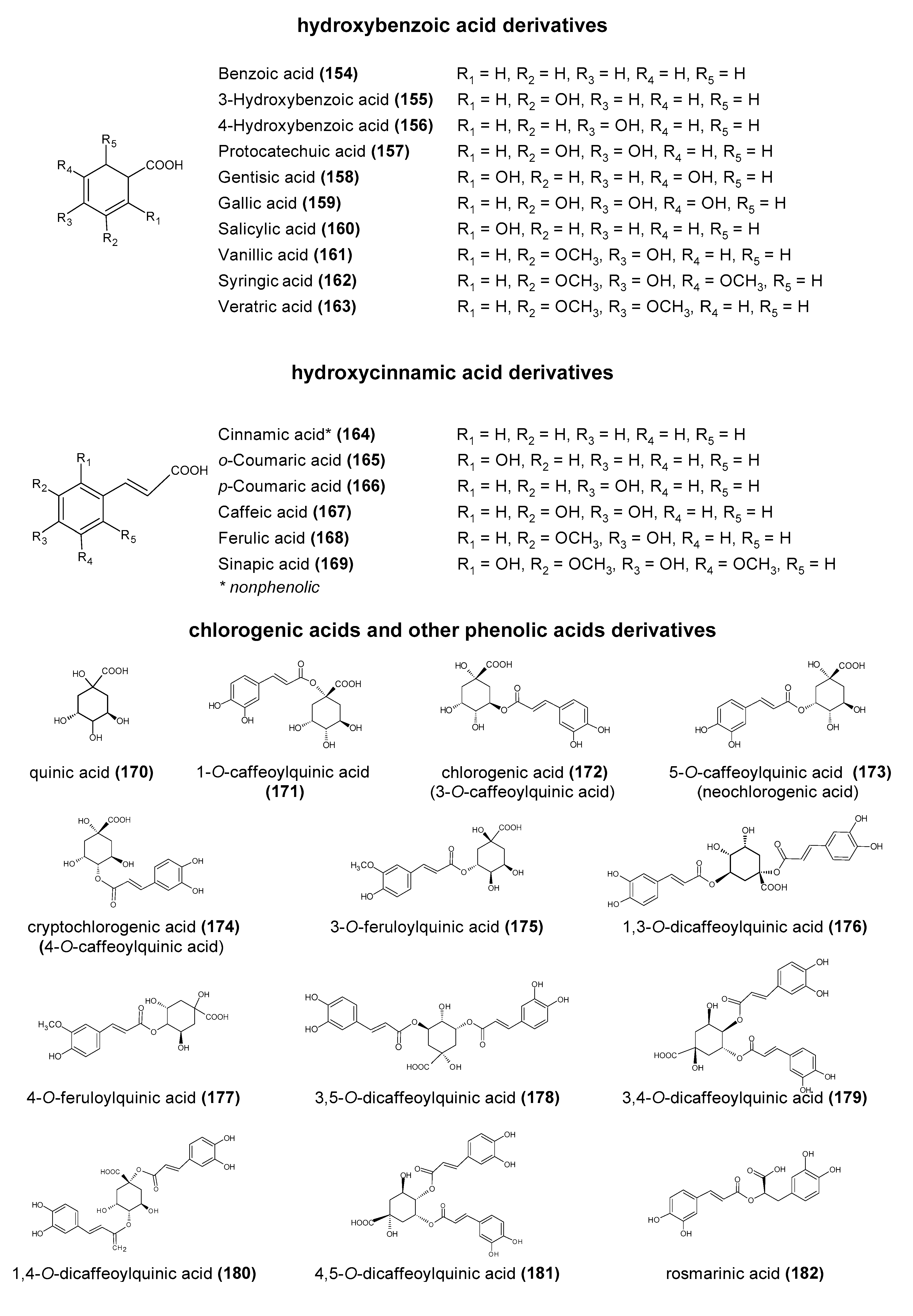
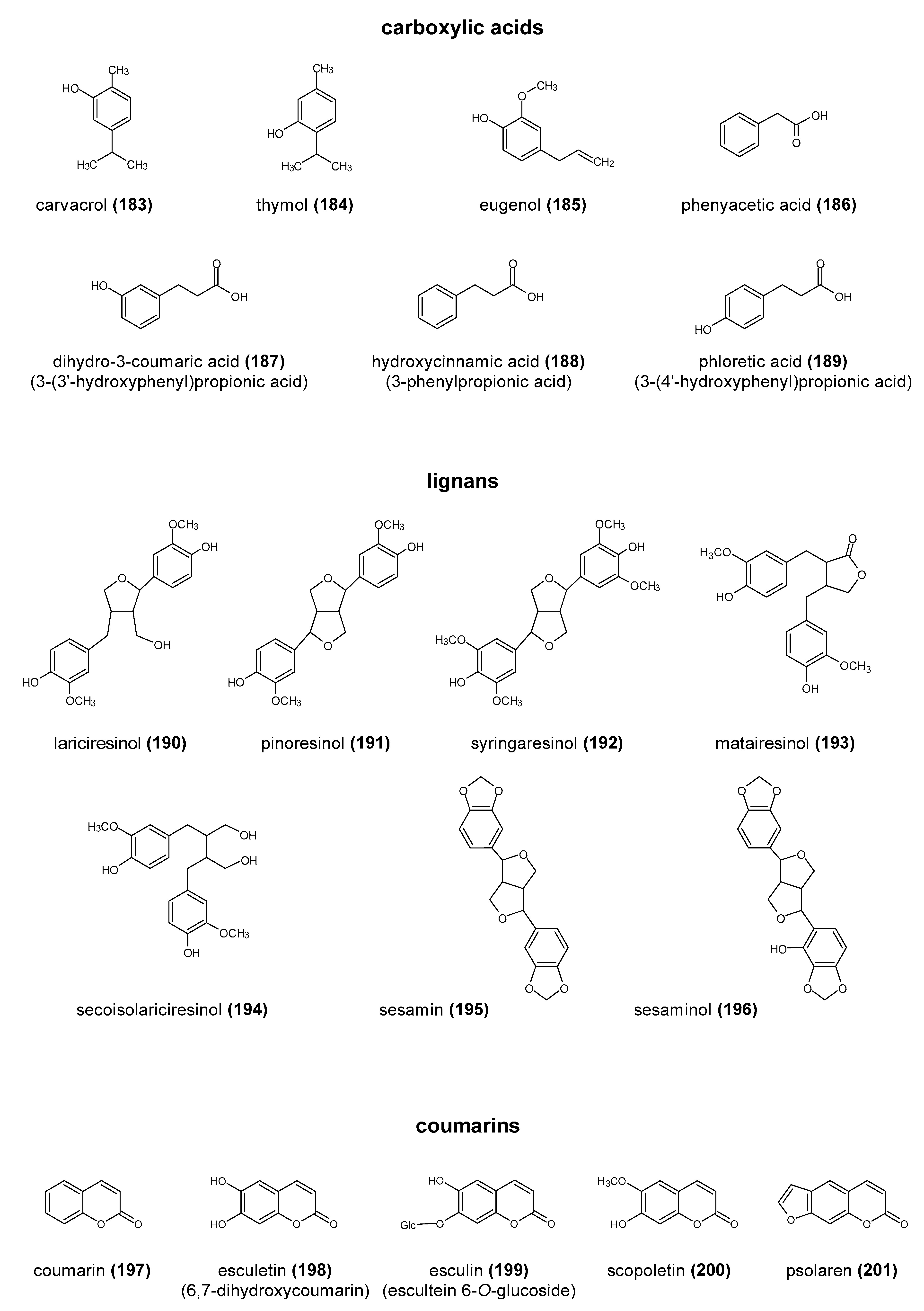
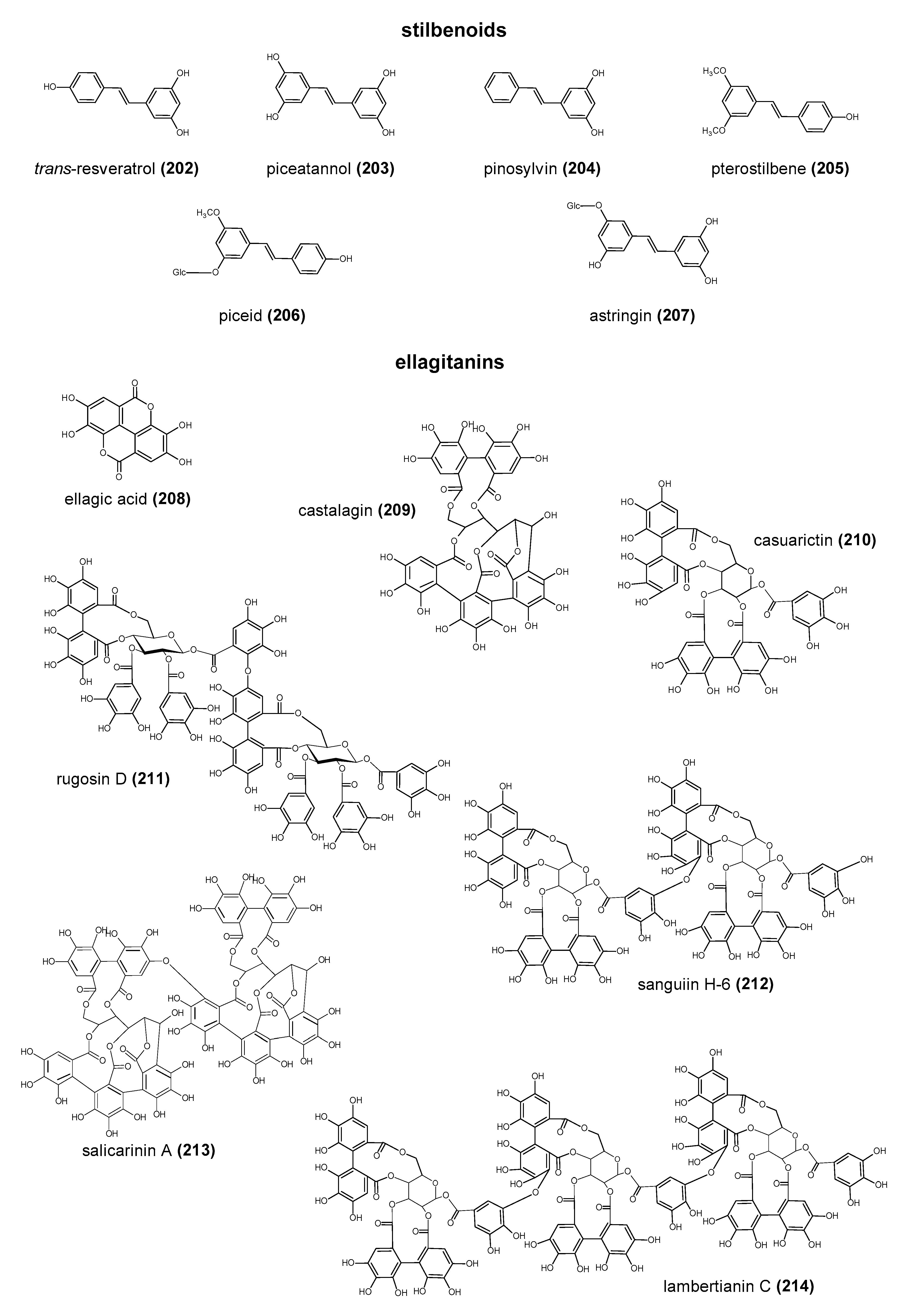
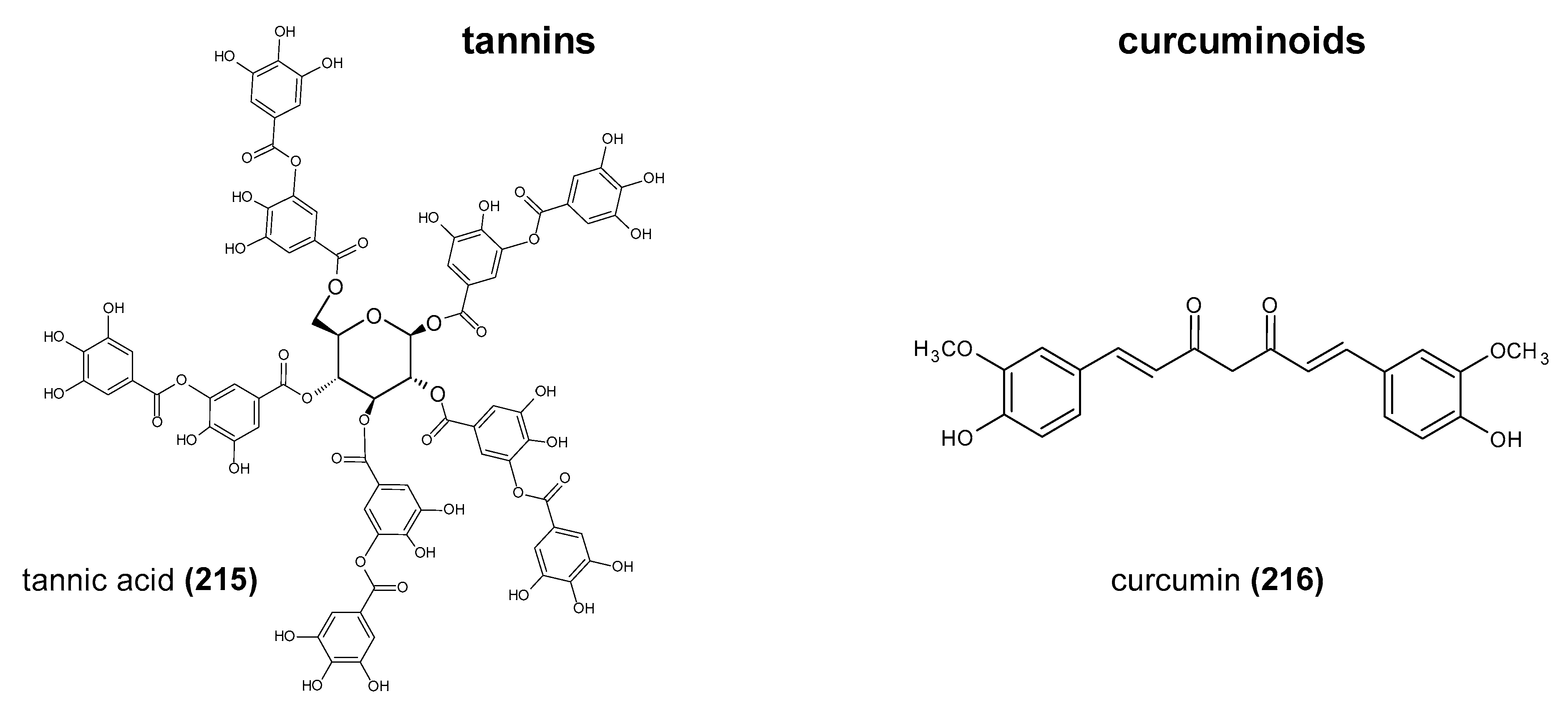


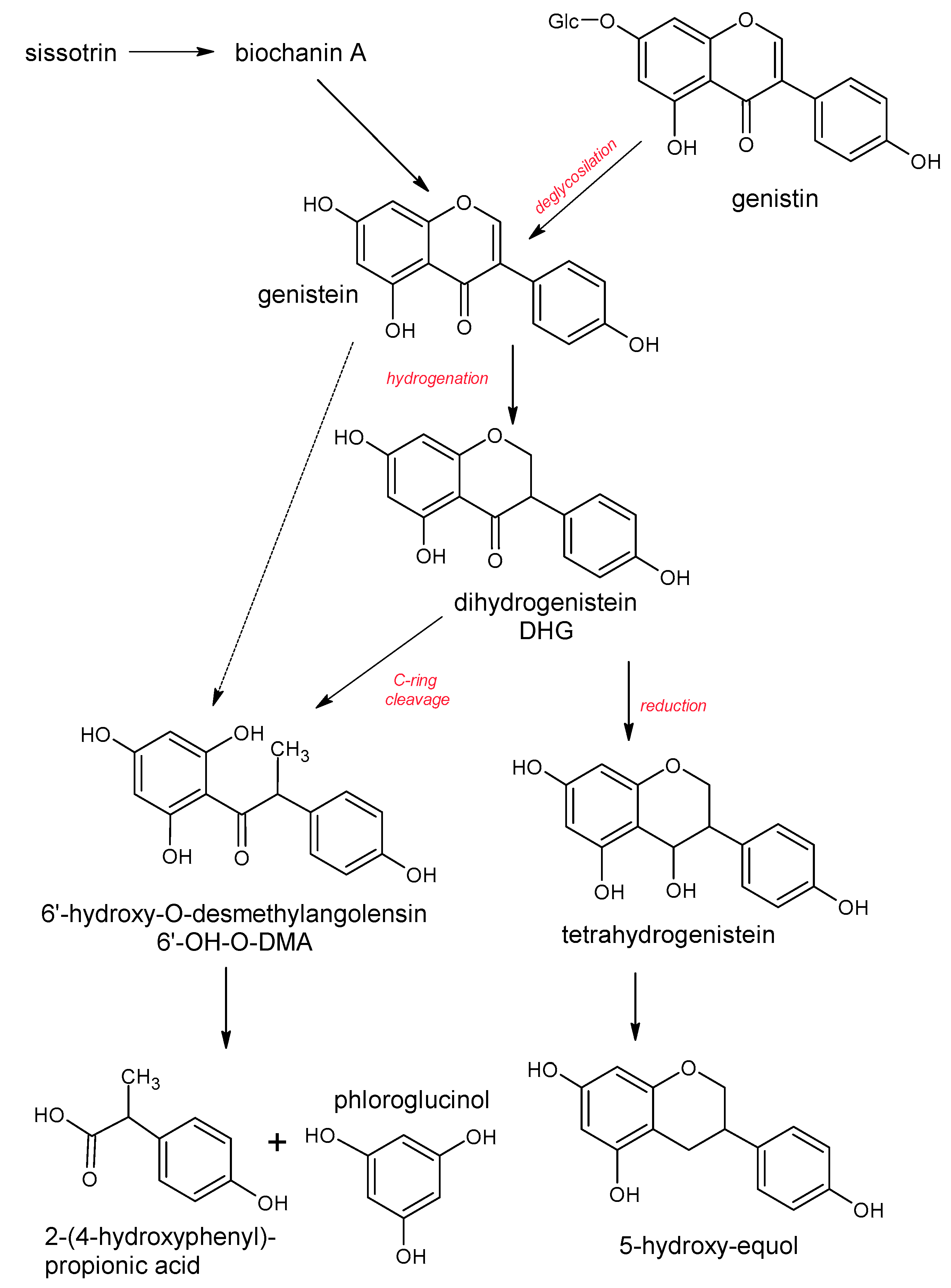

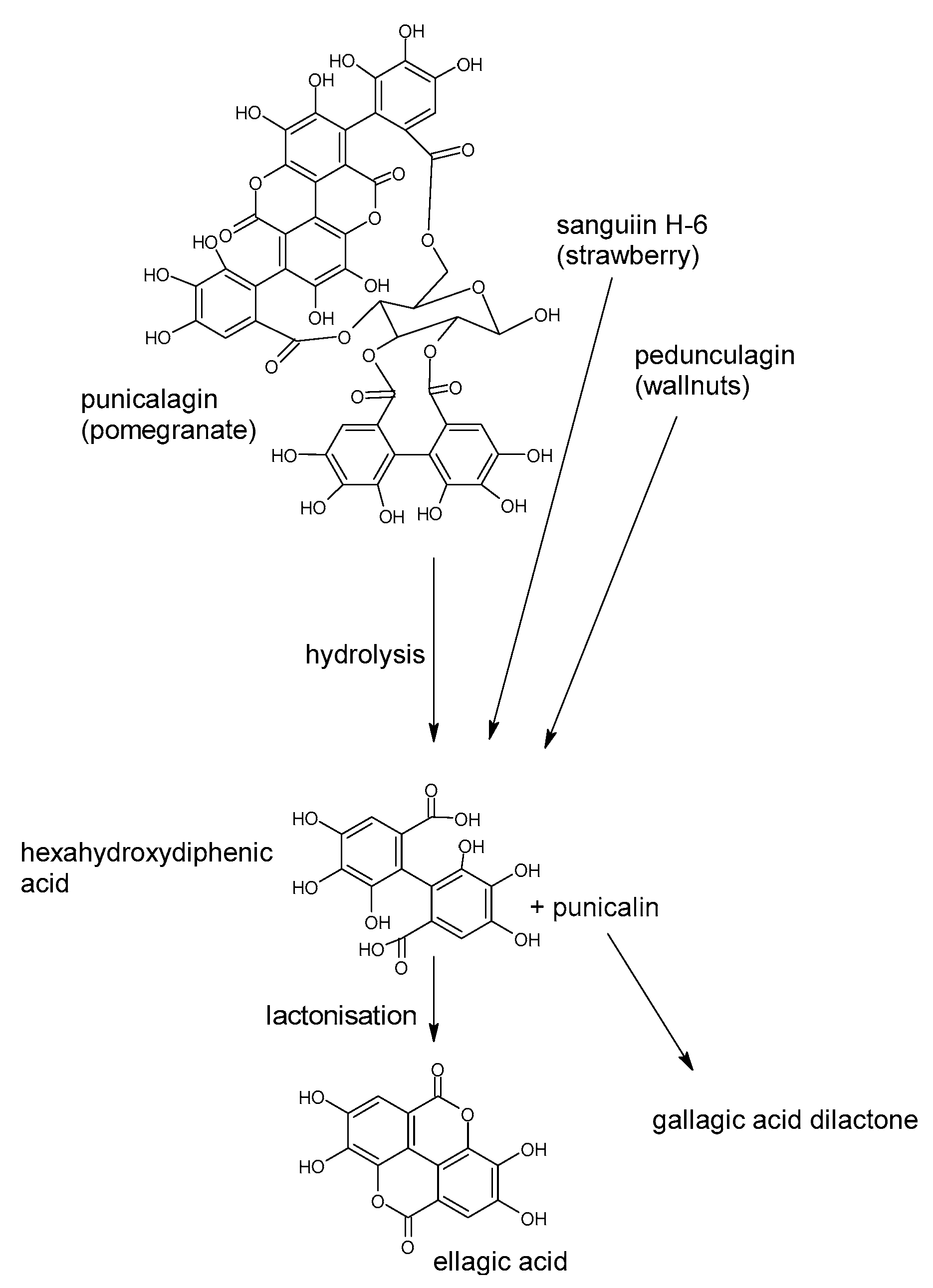

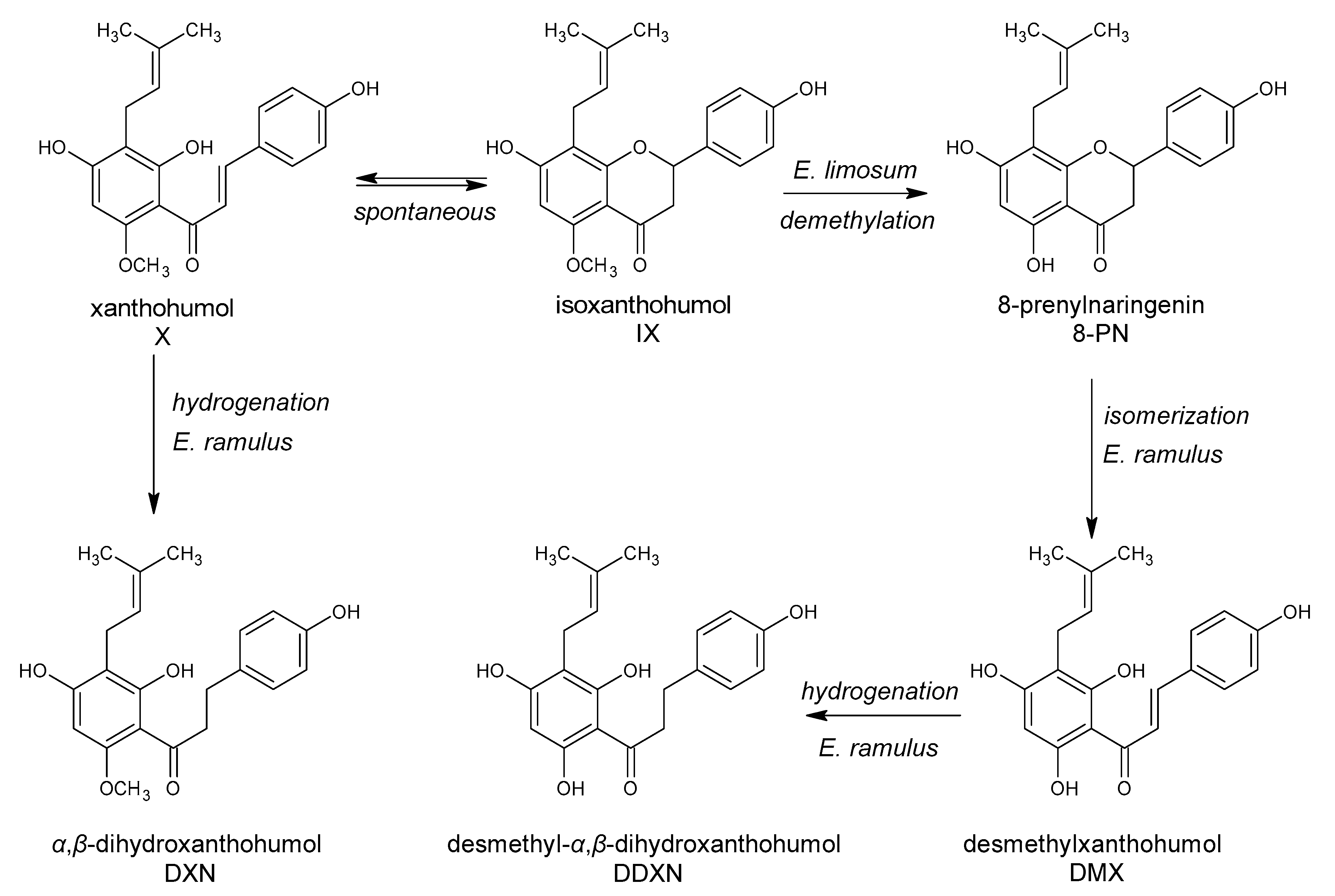
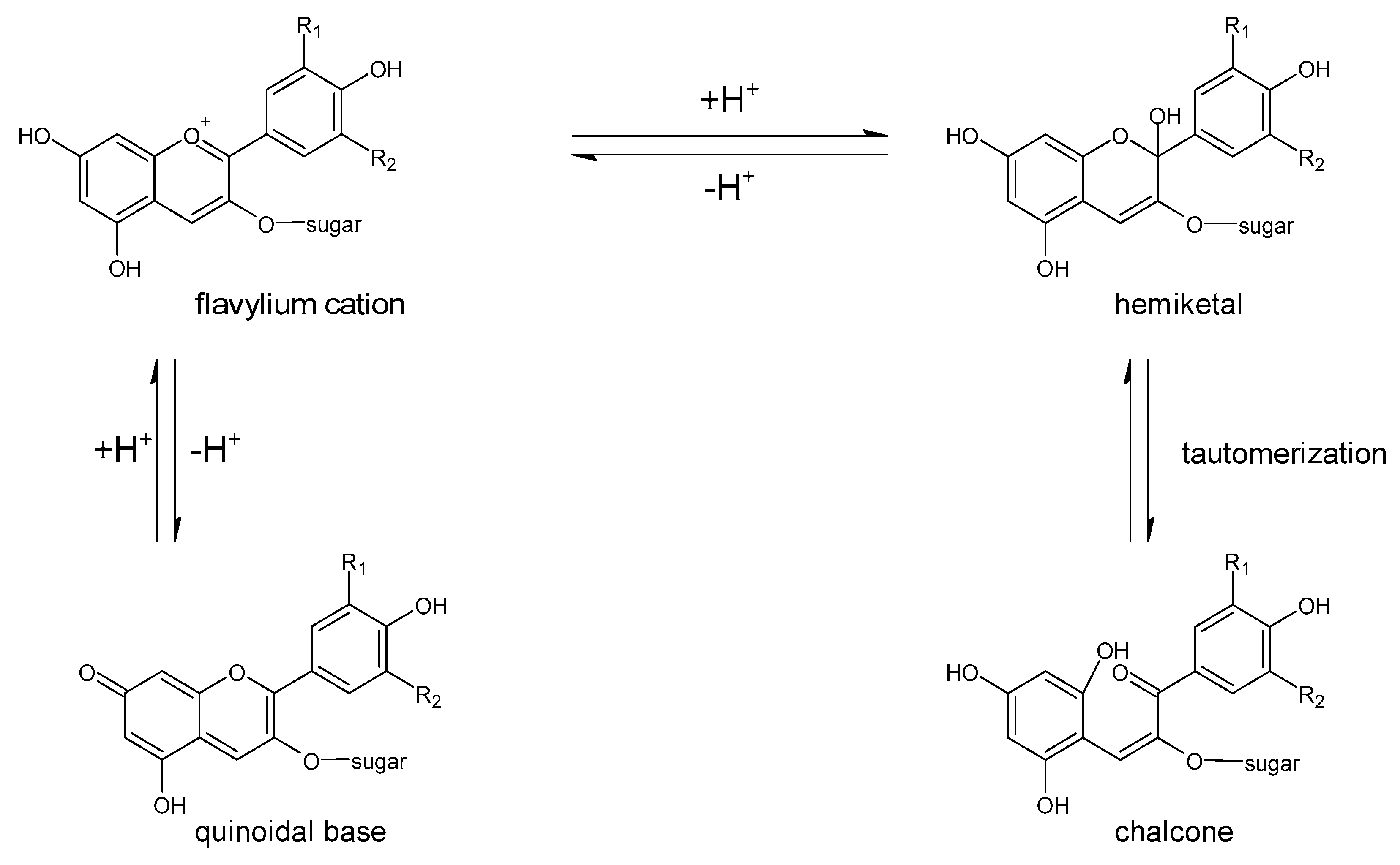

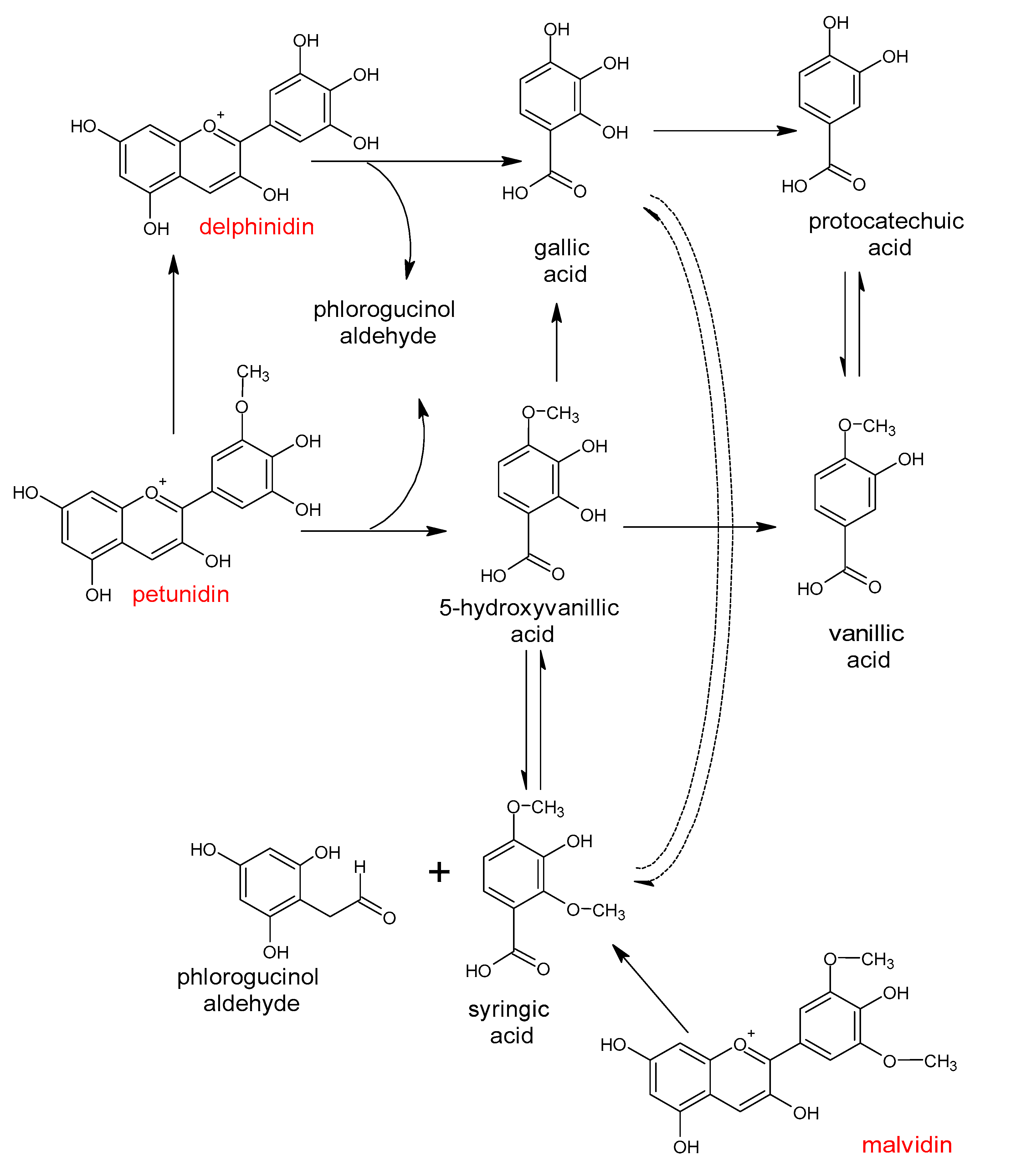

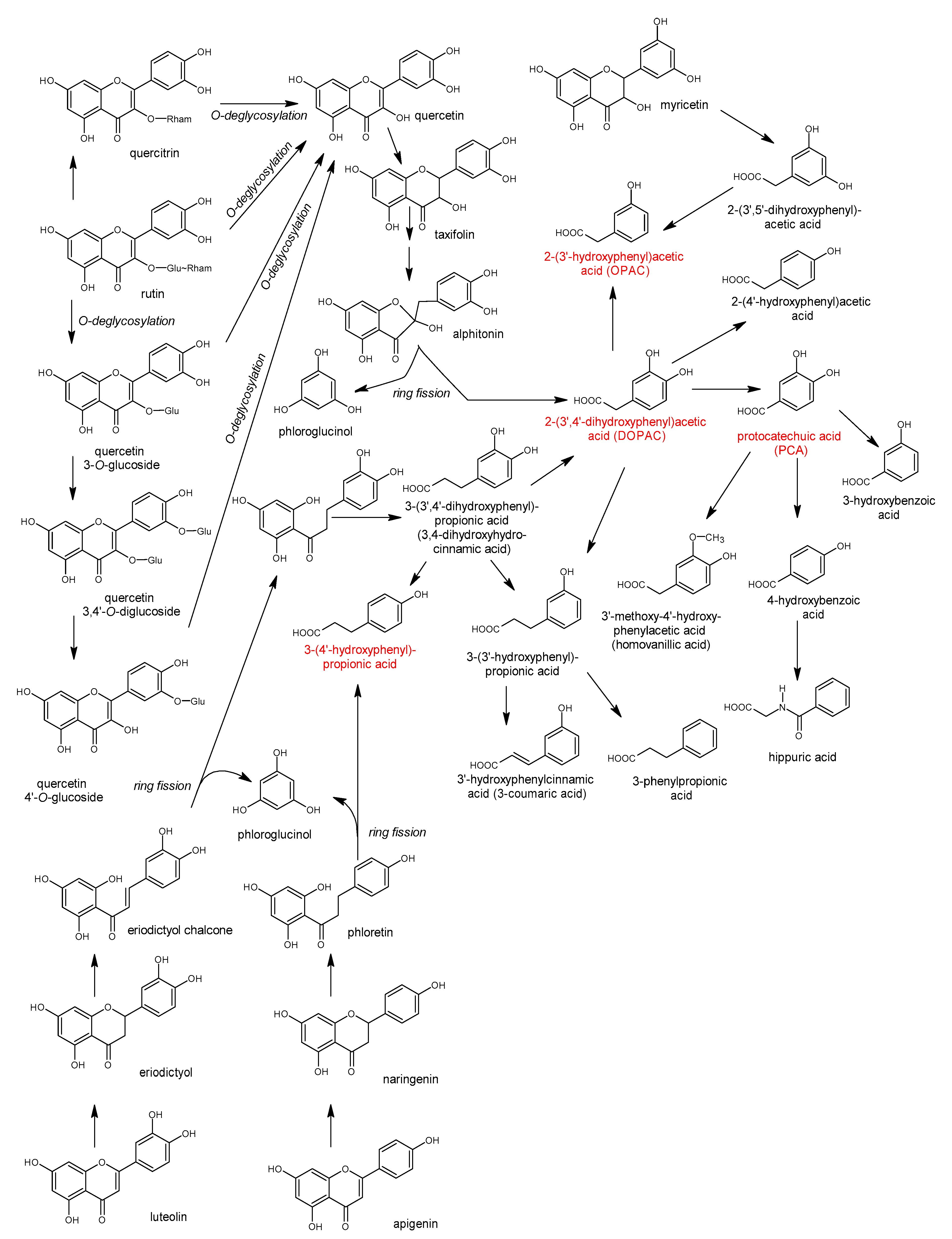

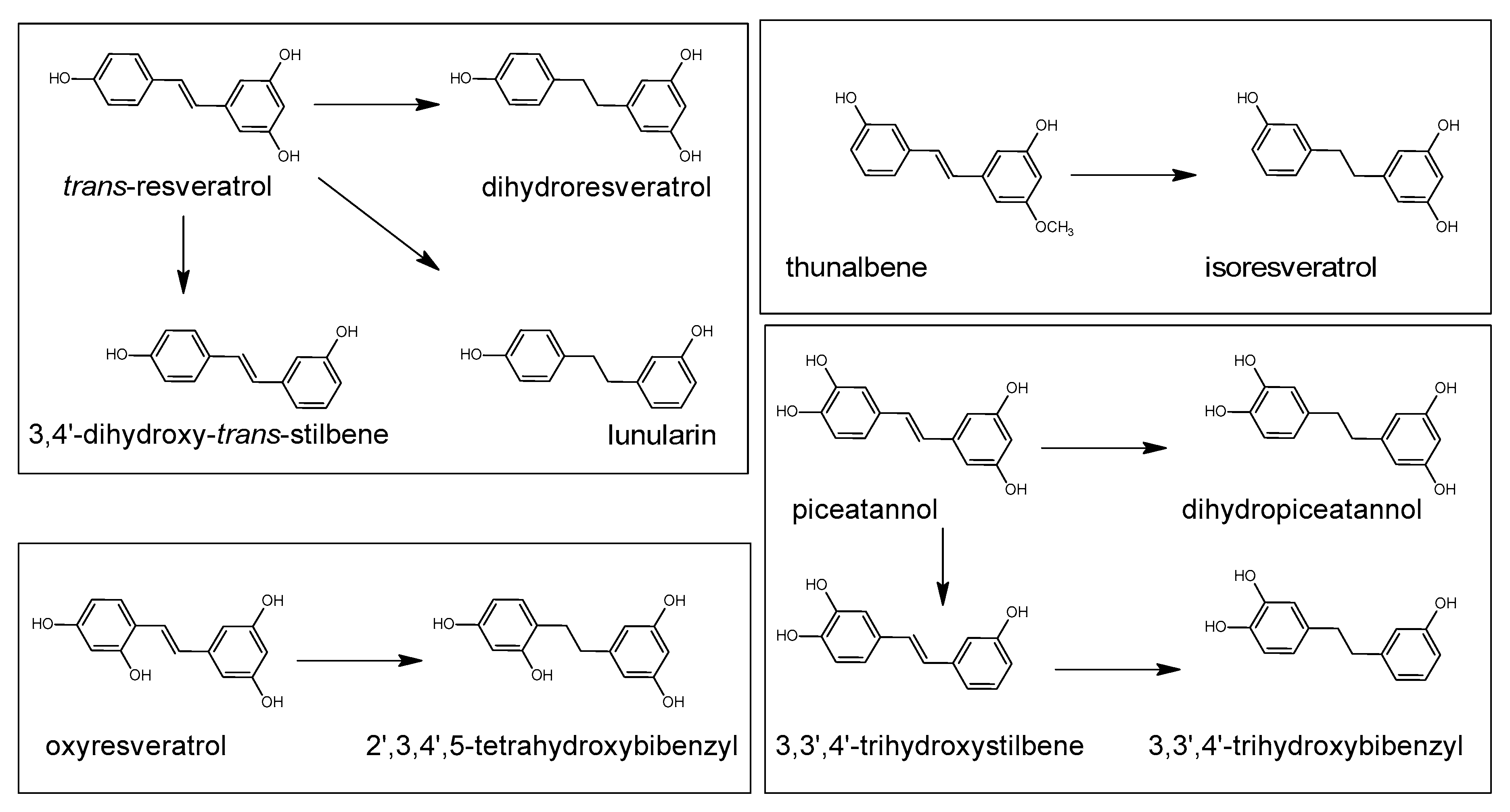
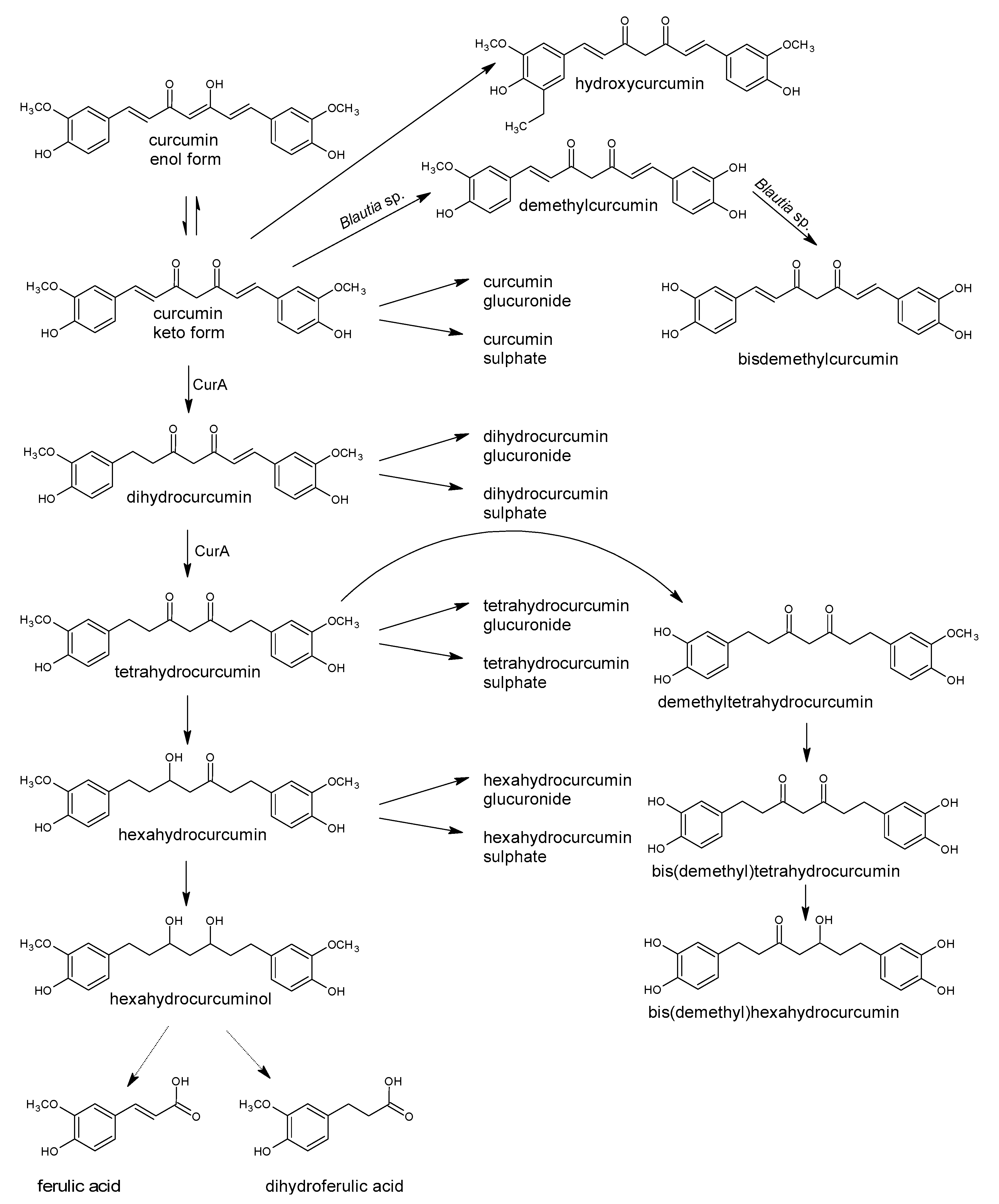
| Plant Material, Preparation or Source | Pure Bioactive Compounds or Polyphenols Identified in Plant Material | Impact on Microorganisms | Reference |
|---|---|---|---|
| Ethanolic extracts of: rosemary, pomegranate peel, grape seed | Antimicrobial activity of rosemary extract against S. aureus, E. coli, P. aeruginosa, K. pneumonia, B. subtilis, M. luteus and C. albicans. Pomegranate peel extract inhibited all mentioned microorganism except P. aeruginosa. Inhibitory impact on S. aureus, E. coli, B. subtilis, M. luteus and C. albicans. | [41] | |
| Ethanolic extract of sage (Salvia officinalis) flower, leaf and stem and essential oils | Manool, α-pinene, camphene, camphor, limonene, bornyl, 1,8-cineole, linalool, cis- and trans-thujone, acetate, α-humulene | Antibacterial activity against B. subtilis, S. aureus, S. enteritidis, P. aeruginosa, E. coli. Extracts had stronger antibacterial activity that the essential oils. | [42] |
| Ethanolic extracts of Heliotropum europaeum | Antimicrobial activity against B. subtilis, E. coli, P. aeruginosa, P. fluorescens, S. aureus, MRSA, S. epidermidis, S. odorifera P. vulgaris, K. pneumoniae, E. faecium and clinic isolate of E. faecalis. No impact on E. aerogenes, multidrug resistant strains of E. coli and K. pneumoniae, E. faecalis ATCC 29212, L. monocytogenes, S. haemolyticus, S. boydi, A. baumannii, and S. flexneri. | [43] | |
| Ethanolic extracts from thyme (Thymus vulgaris) and sage (Salvia officinalis) leaves | Flavonoids tannins rosmarinic acid (182), caffeic acid (167), chlorogenic acid (172), carnosol | Inhibited growth of S. aureus, Vibrio tubiashii, L. pneumophila, M. luteus, Streptococcus sp., B. cereus. | [44] |
| Aqueous and methanol extracts of Salvadora persica L. | Aqueous extract inhibited in dose-dependent manner all tested microorganisms, especially Streptococcus mutans, S. faecalis, S. pyogenes, as well as S. aureus, L. acidophilus, P. aeruginosa, and C. albicans. Methanol extract was inactive against L. acidophilus and P. aeruginosa. | [45] | |
| Hydrodistilled essential oils from Mentha piperita and Rosmarinus officinalis | Mint: α-terpinene, piperitenone oxide, isomenthone, trans-carveol and β-caryophyllene. Rosemary: piperitone, α-pinene, linalool, camphor, 1,8-cineole | Inhibition of the growth of Streptococcus mutans and S. pyogenes. Inhibition of biofilm formation by S. mutans. | [46] |
| Phenolic extracts from blueberry, lingonberry, blackcurrant, raspberry, cloudberry, cranberry and strawberry Pure polyphenols | Extracts contained anthocyanins, flavonols, flavan-3-ols, hydroxycinnamate. myricetin (114), luteolin (49), apigenin (38), kaempferol (202), quercetin (111) rutin (121) isoquercitrin (126) (+)-catechin (7) phenolic acids: trans-cinnamic acid (164), m-coumaric acid, caffeic acid (167), ferulic acid (168), chlorogenic acid (172) | Strong antimicrobial activity against Salmonella enterica ser. Thyphimurium SH5014, E. coli CM871 and E. coli 50. Inhibition of Lactobacillus rhamnosus and L. rhamnosus GG growth by cloudberry, raspberry and strawberry extracts at higher concentrations. Strawberry extract was effective inhibitor against E. faecalis and Bifidobacterium lactis. Strong inhibition of the growth of all lactic acid bacteria derived from the human gut, and E. coli growth by myricetin; but no impact on Salmonella Typhimurium and Lactobacillus plantarum from beer. Bacteriostatic impact of luteolin on some Lactobacillus species, Bifidobacterium lactis and Enterococcus faecalis, no impact on Gram-negative bacteria. No impact of (+)-catechin, kaempferol, apigenin, isoquercitrin, and rutin. Phenolic acids at concentration 0.5 mg/well phenolic acids inhibited the growth of E. coli, and S. enterica. | [47] |
| Methanolic extracts of six species of Hypericum | tannin, flavonoid and phenolic acids, among them quercitrin (123), hyperoside (120), isoquercitrin (126), chlorogenic acid (172) were identified | H. caprifoliatum – the most active, inhibition of S. aureus. H. polyanthemum and H. ternum extracts antibacterial activity against B. subtilis. No activity of extracts against activity against S. epidermidis and E. coli. | [48] |
| Extracts of meadowsweet, willow herb, cloudberry, raspberry, bilberry and crowberry Extracts of white birch, pine, potato | Extract contained flavonoids and phenolic acid Pure compounds: quercetin (111), morin (110), rutin (121), naringenin (81), naringin (85), kaempferol (202) | E. coli and S. aureus growth inhibition. Only S. aureus growth inhibition. Quercetin, morin, naringenin inhibited S. aureus, S. epidermidis, E. coli, B. subtilis, M. luteous, P. aeruginosa. Glycosides rutin and naringin were inactive. Kaempferol inhibited only S. aureus. | [49] |
| Peppermint essential oil and various extracts | Strong inhibition of S. aureus and S. pyogenes growth, less impact on of E. coli and Klebsiella pneumoniae. Petroleum ether, chloroform and ethyl acetate extracts were more effective antibacterial agents than ethanol and aqueous extracts. The strength of inhibition by extracts: S. aureus > K. pneumoniae > S. pyogenes > E. coli. | [50] | |
| Peppermint oil Green tea polyphenols (GTP) | 53 constituents of oil, e.g., menthol, menthone, neomenthol, menthofuran, (+)-limonene, piperiton, 3-octanol, cis-jasmone, mint lactone, (−)-myrtenol, piperitol, eugenol (185), carvacrol (183), 2-ethylfuran, ocimene (−)-epigallocatechin (14), (−)-epigallocatechin-3-gallate (18) | Mentioned compounds had strong antibacterial activity against non-pathogenic E. coli. Oil, menthol, menthone and neomenthol killed the enterohemorrhagic strain E. coli O157:H7 at concentrations 400 µg/mL within 1 h. GTP inhibited E. coli O157:O7 growth at concentration of 800 µg/mL within 18 h. The synergistic effect was reported for peppermint oil + GTP and menthol + GTP. | [51] |
| Cranberry, blueberry and grape seed extracts and their synergy mixtures | Dose-dependent inhibitory activity against Helicobacter pylori. The synergy mixtures with higher concentrations of polymeric phenolics (procyanidins and tannins) had higher antimicrobial activity against H. pylori. | [52] | |
| Chinese green tea extract | (−)-epicatechin gallate (ECG) (17), (−)-epigallocatechin-3-gallate (EGCG) (18) | Crude green tea extract caused growth inhibition (by 44–100%) of food-borne pathogens Escherichia coli O157:H7, Salmonella Typhimurium DT104, L. monocytogenes, S. aureus, a diarrhoea food-poisoning pathogen Bacillus cereus. The lowest MIC90 values had EGCG: against S. aureus MSSA was 58 µg/mL, while against MRSA 37 µg/mL. ECG had MIC 309 and 105 µg/mL, respectively. | [53,54,55,56,57,58,59] |
| Green tea, Chung tea, Black tea | (−)-epicatechin (EC) (12), (−)-epigallocatechin (EGC) (14), (−)-epigallocatechin-3-gallate (EGCG) (18), (−)-epicatechin gallate (ECG) (17), Teaflavins, quercetin (111) | EC and EGC strongly inhibited S. aureus ATCC 29213 (MIC 12.5 µg/mL), while ECG had MIC 50 µg/mL, ECG and EGCG strongly inhibited E. coli ATCC 25922 (MIC 12.5 µg/mL). ECG inhibited Enterobacter cloacae 1321E (MIC 12.5 µg/mL) and S. pyogenes (MIC 25 µg/mL), EC inhibited the growth of P. aeruginosa (MIC 25 µg/mL) | [54] |
| 9 Hot water extracts of various tea (oolong, green, black, white) | gallic acid (159), quercetin (111), caffeine, (+)-catechin (7), (−)-epicatechin (12), (−)-epigallocatechin (14) | 5-min extracts inhibited the growth of Helicobacter pylori, but growth of probiotics Bifidobacterium longum and Lactobacillus acidophilus was not affected. | [55] |
| (−)-epigallocatechin-3-gallate (18) | EGCG inhibited intracellular groth of Listeria monocytogenes in macrophages | [56] | |
| (−)-epigallocatechin-3-gallate (18) | EGCG inhibits major functions of cellular and surface proteins, leading to growth inhibition of Bacillus subtilis | [57] | |
| (−)-epigallocatechin-3-gallate (18), (−)-epigallocatechin (EGC) (14) | EGCG inhibits S. aureus, MRSA, S. mutans, E. coli, P. aeruginosa, K. pneumoniae. EGC inhibits E. coli | [58] | |
| (+)-catechin (7), (−)-epicatechin (12) | Catechin caused significant decrease in the growth of the Clostridium histolyticum group and a marked increase in the growth of the beneficial bacterial group of C. coccoides–Eubacterium rectale, Lactobacillus spp. and Bifidobacterium spp. Epicatechin caused a significant increase in the growth of the Eubacterium rectale–C. coccoides. | [59] | |
| Pure polyphenols, inter alia: baicalein (45), quercetin (111), myricetin (114), naringenin (81), naringin (85), hesperetin (83), hesperidin (86), resveratrol (202), gallic acid (159) | Aglycones quercetin, naringenin, hesperetin inhibited growth of tested bacteria, while their glycoside did not. The lowest MIC were reported for: baicalein, myricetin, hesperetin and kaempferol against E. coli O157; baicalein nad myricetin against S. aureus; and baicalein against Salmonella Typhimurium, Enterobacter sakazakii and Vibrio parahemolyticus. | [60] | |
| Citrus fruit | hesperetin (83), naringenin (81), poncirin (90), diosmetin (50) | Inhibition of the growth of H. pylori. | [61] |
| rutin (121), quercetin (111) | Inhibition of L. monocytogenes growth. | [62] | |
| (+)-catechin (7), quercetin (111), naringenin (81), hesperetin (83), rutin (121), naringin (85), hesperidin (86) | Aglycones naringenin and hesperetin, inhibited growth of almost all analysed bacteria (MIC ≥ 250 μg/mL). Catechin, and glycosides naringin, hesperidin, and rutin had no impact on tested intestinal bacteria. Quercetin had strong inhibitory impact (MIC 20–50 μg/mL) on Ruminococcus gauvreauii, Bacteroides galacturonicus and Lactobacillus sp. growth. | [63] | |
| 13 phenolic acids: benzoic acid (154), 3-hydroxybenzoic acid (155), 4-hydroxybenzoic acid (156), 4-hydroxy-3-methoxy-benzoic acid, 3,4-dihydroxybenzoic acid, phenylpropionic acid (188), 3-hydroxyphenylpropionic acid (187), 4-hydroxyphenylpropionic acid (189), 3,4-dihydroxyphenylpro-pionic acids, phenylacetic acid (186), 3-hydroxyphenylacetic acid, 4-hydroxyphenylacetic acid, 3,4-dihydroxyphenylacetic acid | No impact on P. aeruginosa PAO1. Impact on E. coli was strain dependent. E. coli ATCC 25922 inhibition by benzoic and 4-hydroxy-3-methoxy-benzoic, phenylacetic and phenylpropionic acids at a concentration of 1000 mg/mL. E. coli O157:H7 (CECT 5947) was susceptible to benzoic, 3-hydroxybenzoic, 4-hydroxybenzoic, 4-hydroxy-3-methoxy-benzoic acids, phenylacetic, 3-hydroxy-phenylacetic, 4-hydroxyphenylacetic and 3,4-dihydroxyphenylacetic acids, phenylpropionic, 3-hydroxyphenylpropionic acid and 4-hydroxyphenylpropionic acids. Lactobacillus paraplantarum LCH7 was the most susceptible to the action of phenolic acids, while L. fermentum LPH1 was the most resistant. The susceptibility was strain-dependent. The most active compound were: 4-hydroxybenzoic acid for L. fermentum CECT 5716, L. fermentum LPH1, L. brevis LCH23, and L. plantarum LCH17; 4-hydroxybenzoic acid.and phenylpropionic acid for L. paraplantarum LCH7 and L. coryniformis CECT 5711, while 3-hydroxyphenylpropionic acid for L. fermentum CECT 5716. | [64] | |
| Extracts from 3 Eucalyptus species | 2’,6’-dihydroxy-3’-methyl-4’-methoxy-dihydrochalcone, eucalyptin (48), 8-desmethyl-eucalyptin | Significantly inhibited growth of Gram-positive bacteria: S. aureus, MRSA, B. cereus, E. faecalis, Alicyclobacillus acidoterrestris, Propionibacterium acnes. Not show strong antibacterial activity against Gram-negative E. coli and P. putida. | [65] |
| Essential oils from Origanum vulgare and Thymus vulgaris Ocinum basilicum oil | Minimal Bactericidal Concentration /MBC/ ≤ 5 mg/mL against pathogens (E. coli, Salmonella Enteritidis, and S. Typhimurium), and beneficial bacteria (Lactobacillus acidophilus and Bifidobacterium breve). Higher activity against pathogenic bacteria (MBCs ≤ 10 mg/mL) than beneficial bacteria (MBCs of 80 mg/mL). | [66] | |
| 13 common flavonoids (flavones, flavonols, flavanones) and 6 organic acids (aliphatic and aromatic acids) kaempferol (108), quercetin (111), chlorogenic acid (172), salicylic acid (160) | Antimicrobial activity against Gram-negative bacteria E. coli and P. aeruginosa, higher than impact on Gram-positive ones: E. faecalis and S. aureus. Kaempferol, quercetin and chlorogenic acid had no significant influence on P. aeruginosa. Salicylic acid – the highest inhibitory activity against all tested bacterial species (MIC = 250–500 μg/mL). | [67] | |
| A flavan-3-ol enriched grape seed extract | Inhibited growth of Streptococcus thermophilus, Bifidobacterium lactis BB12, Lactobacillus fermentum, L. acidophilus and L. vaginalis. Stimulated growth of some Lactobacillus plantarum, L. casei, and L. bulgaricus strains. No impact on Bifidobacterium breve 26M2 and B. bifidum HDD541 growth | [68] | |
| naringenin (85), hes-peridin (86), rutin (121), quercetin (111), gal-lic acid (159), caffeic acid (167), p-coumaric acid (166), ferulic acid (168), chlorogenic acid (172), vanillic acid (161), sinapic acid (169), hesperidin (86), quercetin (111) | All polyphenols influenced the growth of Bifidobacterium adolescentis and B. bifidum was assessed. Both the stimulatory and inhibitory effects of polyphenols were observed. Coumaric acid had strongest stimulatory effect on B. bifidum, while vanillic and caffeic acid stimulated B. adolescentis. Inhibitory dose-dependent impact of hesperidin and quercetin, on B. bifidum and B. adolescentis. | [69] | |
| Tea phenolics and their derivatives | 3-phenylpropionic acid (188), caffeic acid (167), gallic acid (159) | Inhibition of pathogenic Clostridium perfringens, C. difficile and Bacteroides spp. Less effect on commensal Clostridium spp., Bifidobacterium spp. and probiotic Lactobacillus sp. The growth of Lactobacillus ssp. and Bifidobacterium spp. inhibited by caffeic acid, 3-phenylpropionic acid and to the lesser extent by gallic acid, while Lactobacillus casei Shirota growth inhibited only by 3-phenylpropionic acid. | [70] |
| Red wine polyphenols | Stimulated growth of Bifidobacterium and Lactobacillus. | [71] | |
| Tannin-rich diet | Significantly decreased growth of Clostridium spp. with a corresponding shift toward Entero-bacteriaceae and Bacteroides | [72] | |
| Pomegranate extract | ellagitannins | Enhancing the growth of beneficial bacteria Akkermansia muciniphila. | [73] |
| resveratrol (202) | Enhancing the growth of beneficial bacteria Akkermansia muciniphila, Lactobacillus and Bifidobacterium. | [74] [75] | |
| polymeric procyanidins | Enhancing the growth of beneficial bacteria Akkermansia muciniphila. Markedly decreased the Firmicutes/Bacteroidetes ratio | [76] | |
| Grape polyphenols | Enhancing the growth of beneficial bacteria Akkermansia muciniphila, and decreasing the proportion of Firmicutes to Bacteroidetes. | [77] | |
| rutin (121) | The beneficial impact on Lactobacillus and Bifidobacterium | [63] | |
| Cocoa | polyphenols | The beneficial impact on Lactobacillus and Bifidobacterium | [78] |
| Red wine polyphenols | Significantly increased the number of Enterococcus, Prevotella, Bacteroides, Bifidobacterium, Bacteroides uniformis, Eggerthella lenta, and Blautia coccoides–Eubacterium rectale groups | [79] | |
| Blueberry | anthocyanidins | The beneficial impact on Lactobacillus acidophilus and Bifidobacterium spp | [80] |
| Red wine | polyphenols | Significantly increase of the number of faecal bifidobacteria and Lactobacillus (intestinal barrier protectors), Faecalibacterium prausnitzii and Roseburia (butyrate-producing bacteria) | [81] |
| flavan-3-ols epigallocatechin gallate (18), (−)-epigallocatechin (14), procyanidin B1 (19), procyanidin B2 (20) | Significant inhibition of L. acidophilus LA-5 and L. plantarum IFPL379 adhesion, except 4 compounds. Enhanced L. acidophilus LA-5 adhesion to Caco-2 cells. Increased L. casei LC115 adhesion to Caco-2 cells. Increased the adhesion of Lactobacillus casei LC115 to HT-29 cells. | [82] | |
| Extract from apples | procyanidin B2 (20), chlorogenic acid (172) | The increase of adhesion of Lactobacillus gasseri and Lactobacillus casei to intestinal epithelial cells. | [83] |
| Cranberry | proanthocyanidins with A-type linkages | Inhibition of the adhesion of both antibiotic-susceptible and antibiotic-resistant strains of uropathogenic P-fimbriated E. coli. | [84,85] |
| anthocyanidins: pelargonidin (25), cyanidin (26), delphinidin (27), cyanidin-3-glucoside (36) | Inhibition of the growth of E. coli strain CM871. No effect on E. faecalis strain E-203, S. enterica SH-5014 and strains of the Lactobacillus and Bifidobacterium. | [47] | |
| Wine extract | quercetin (111), flavan-3-ols, anthocyanins | No impact on species belonging to the Lactobacillus, Enterococcus, Bacteroides, and Bifidobacterium genera. | [86] |
| Grape seeds and pomace | tannic acid (215) | Potent growth-promoting effects on L. acidophilus. | [87] |
| gallic acid (159), and free anthocyanins, vanillic acid (161), protocatechuic acid (157) | Activated cell growth and the rate malolactic fermentation of Leuconostoc oenos. Vanillic acid showed a slight inhibiting effect, while protocatechuic acid had no effect. | [88] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Makarewicz, M.; Drożdż, I.; Tarko, T.; Duda-Chodak, A. The Interactions between Polyphenols and Microorganisms, Especially Gut Microbiota. Antioxidants 2021, 10, 188. https://doi.org/10.3390/antiox10020188
Makarewicz M, Drożdż I, Tarko T, Duda-Chodak A. The Interactions between Polyphenols and Microorganisms, Especially Gut Microbiota. Antioxidants. 2021; 10(2):188. https://doi.org/10.3390/antiox10020188
Chicago/Turabian StyleMakarewicz, Małgorzata, Iwona Drożdż, Tomasz Tarko, and Aleksandra Duda-Chodak. 2021. "The Interactions between Polyphenols and Microorganisms, Especially Gut Microbiota" Antioxidants 10, no. 2: 188. https://doi.org/10.3390/antiox10020188
APA StyleMakarewicz, M., Drożdż, I., Tarko, T., & Duda-Chodak, A. (2021). The Interactions between Polyphenols and Microorganisms, Especially Gut Microbiota. Antioxidants, 10(2), 188. https://doi.org/10.3390/antiox10020188