Transcriptomic Analysis Suggests a Coordinated Regulation of Carotenoid Metabolism in Ripening Chili Pepper (Capsicum annuum var. conoides) Fruits
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Materials and Growth Condition
2.2. Pigment Extraction and HPLC Analysis
2.3. cDNA Library Construction and Sequencing
2.4. Sequence Annotation
2.5. Analysis of Differentially Expressed Genes
2.6. Correlation Analysis
3. Results and Discussion
3.1. Coordinated Fluctuations of Carotenoid Constituents in Ripening Fruits
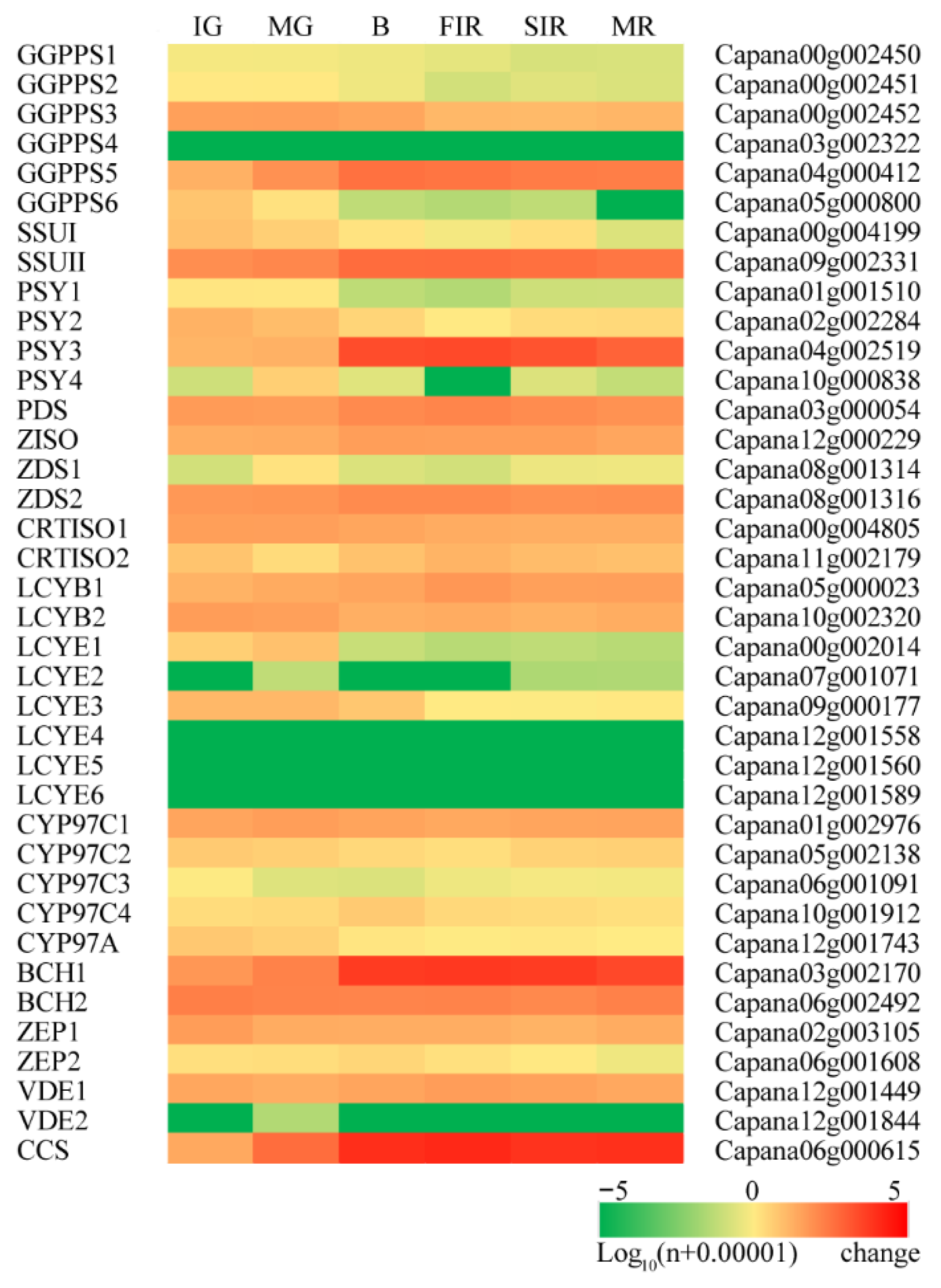
3.2. Concerted Expression of Carotenoid Metabolic Genes
3.3. Genes Co-Expressed with Carotenoid Metabolism in Ripening Fruits
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Giovannoni, J.; Nguyen, C.; Ampofo, B.; Zhong, S.; Fei, Z. The epigenome and transcriptional dynamics of fruit ripening. Annu. Rev. Plant Biol. 2017, 68, 61–84. [Google Scholar] [CrossRef] [PubMed]
- Adams-Phillips, L.; Barry, C.; Giovannoni, J. Signal transduction systems regulating fruit ripening. Trends. Plant Sci. 2004, 9, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Giovannoni, J.J. Fruit ripening mutants yield insights into ripening control. Curr. Opin. Plant Biol. 2007, 10, 283–289. [Google Scholar] [CrossRef]
- Bouvier, F.; Camara, B. The role of plastids in ripening fruits. In The Structure and Function of Plastids; Springer: Dordrecht, The Netherlands, 2006; pp. 419–432. [Google Scholar]
- Tanaka, R.; Tanaka, A. Tetrapyrrole biosynthesis in higher plants. Annu. Rev. Plant Biol. 2007, 58, 321–346. [Google Scholar] [CrossRef] [PubMed]
- Nisar, N.; Li, L.; Lu, S.; Khin, N.C.; Pogson, B.J. Carotenoid metabolism in plants. Mol. Plant 2015, 8, 68–82. [Google Scholar] [CrossRef] [Green Version]
- del Rocío Gómez-García, M.; Ochoa-Alejo, N. Biochemistry and molecular biology of carotenoid biosynthesis in chili peppers (Capsicum spp.). Int. J. Mol. Sci. 2013, 14, 19025–19053. [Google Scholar] [CrossRef] [Green Version]
- Powell, A.L.; Nguyen, C.V.; Hill, T.; Cheng, K.L.; Figueroa-Balderas, R.; Aktas, H.; Ashrafi, H.; Pons, C.; Fernández-Muñoz, R.; Vicente, A.; et al. Uniform ripening encodes a Golden 2-like transcription factor regulating tomato fruit chloroplast development. Science 2012, 336, 1711–1715. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.S.; Roof, S.; Ye, Z.B.; Barry, C.; van Tuinen, A.; Vrebalov, J.; Bowler, C.; Giovannoni, J. Manipulation of light signal transduction as a means of modifying fruit nutritional quality in tomato. Proc. Natl. Acad. Sci. USA 2004, 101, 9897–9902. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mustilli, A.C.; Fenzi, F.; Ciliento, R.; Alfano, F.; Bowler, C. Phenotype of the tomato high pigment-2 mutant is caused by a mutation in the tomato homolog of DEETIOLATED1. Plant Cell 1999, 11, 145–157. [Google Scholar] [CrossRef] [Green Version]
- Lu, S.W.; Zhang, Y.; Zhu, K.; Yang, W.; Ye, J.L.; Chai, L.; Xu, Q.; Deng, X. The citrus transcription factor CsMADS6 modulates carotenoid metabolism by directly regulating carotenogenic genes. Plant Physiol. 2018, 176, 2657–2676. [Google Scholar] [CrossRef]
- Ampomah-Dwamena, C.; Thrimawithana, A.H.; Dejnoprat, S.; Lewis, D.; Espley, R.V.; Allan, A.C. A kiwifruit (Actinidia deliciosa) R2R3-MYB transcription factor modulates chlorophyll and carotenoid accumulation. New Phytol. 2019, 221, 309–325. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zuccarelli, R.; Rodríguez-Ruiz, M.; Lopes-Oliveira, P.J.; Pascoal, G.B.; Andrade, S.C.S.; Furlan, C.M.; Purgatto, E.; Palma, J.M.; Corpas, F.J.; Rossi, M.; et al. Multifaceted roles of nitric oxide in tomato fruit ripening: NO-induced metabolic rewiring and consequences for fruit quality traits. J. Exp. Bot. 2020, 72, 941–958. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.; Li, L. Carotenoid metabolism: Biosynthesis, regulation, and beyond. J. Integr. Plant Biol. 2008, 50, 778–785. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Wang, C.-Y.; Gutensohn, M.; Jiang, L.; Zhang, P.; Zhang, D.; Dudareva, N.; Lu, S. A recruiting protein of geranylgeranyl diphosphate synthase controls metabolic flux toward chlorophyll biosynthesis in rice. Proc. Natl. Acad. Sci. USA 2017, 114, 6866–6871. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Camara, B.; Moneger, R. Carotenoid biosynthesis—In vitro conversion of antheraxanthin to capsanthin by a chromoplast enriched fraction of Capsicum fruits. Biochem. Biophys. Res. Commun. 1981, 99, 1117–1122. [Google Scholar] [CrossRef]
- Furubayashi, M.; Kubo, A.; Takemura, M.; Otani, Y.; Maoka, T.; Terada, Y.; Yaoi, K.; Ohdan, K.; Misawa, N.; Mitani, Y. Capsanthin production in Escherichia coli by overexpression of capsanthin/capsorubin synthase from Capsicum annuum. J. Agric. Food Chem. 2021, 69, 5076–5085. [Google Scholar] [CrossRef]
- Ha, S.-H.; Kim, J.K.; Jeong, Y.S.; You, M.-K.; Lim, S.-H.; Kim, J.-K. Stepwise pathway engineering to the biosynthesis of zeaxanthin, astaxanthin and capsanthin in rice endosperm. Metab. Eng. 2019, 52, 178–189. [Google Scholar] [CrossRef]
- Lefebvre, V.; Kuntz, M.; Camara, B.; Palloix, A. The capsanthin-capsorubin synthase gene: A candidate gene for the y locus controlling the red fruit colour in pepper. Plant Mol. Biol. 1998, 36, 785–789. [Google Scholar] [CrossRef]
- Wang, Q.; Cao, T.-J.; Zheng, H.; Zhou, C.-F.; Wang, Z.; Wang, R.; Lu, S. Manipulation of carotenoid metabolic flux by lycopene cyclization in ripening red pepper (Capsicum annuum var. conoides) fruits. J. Agric. Food Chem. 2019, 67, 4300–4310. [Google Scholar] [CrossRef]
- Wang, Q.; Huang, X.-Q.; Cao, T.J.; Zhuang, Z.; Wang, R.; Lu, S. Heteromeric geranylgeranyl diphosphate synthase contributes to carotenoid biosynthesis in ripening fruits of red pepper (Capsicum annuum var. conoides). J. Agric. Food Chem. 2018, 66, 11691–11700. [Google Scholar] [CrossRef]
- Qin, C.; Yu, C.; Shen, Y.; Fang, X.; Chen, L.; Min, J.; Cheng, J.; Zhao, S.; Xu, M.; Luo, Y.; et al. Whole-genome sequencing of cultivated and wild peppers provides insights into Capsicum domestication and specialization. Proc. Natl. Acad. Sci. USA 2014, 111, 5135–5140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative CT method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Trapnell, C.; Pachter, L.; Salzberg, S.L. TopHat: Discovering splice junctions with RNA-Seq. Bioinformatics 2009, 25, 1105–1111. [Google Scholar] [CrossRef] [PubMed]
- Anders, S.; Pyl, P.T.; Huber, W. HTSeq–a Python framework to work with high-throughput sequencing data. Bioinformatics 2015, 31, 166–169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anders, S.; Huber, W. Differential expression analysis for sequence count data. Genome Biol. 2010, 11, R106. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Jiang, L.; Li, Y.; Chen, Q.; Ye, Y.; Zhang, Y.; Luo, Y.; Sun, B.; Wang, X.; Tang, H. Effect of red and blue light on anthocyanin accumulation and differential gene expression in strawberry (Fragaria × ananassa). Molecules 2018, 23, 820. [Google Scholar] [CrossRef] [Green Version]
- Zhang, R.; Fu, X.; Zhao, C.; Cheng, J.; Liao, H.; Wang, P.; Yao, X.; Duan, X.; Yuan, Y.; Xu, G.; et al. Identification of the key regulatory genes involved in elaborate petal development and specialized character formation in Nigella damascena (Ranunculaceae). Plant Cell 2020, 32, 3095–3112. [Google Scholar] [CrossRef]
- Barja, M.V.; Rodriguez-Concepcion, M. Plant geranylgeranyl diphosphate synthases: Every (gene) family has a story. aBIOTECH 2021, 2, 289–298. [Google Scholar] [CrossRef]
- Coman, D.; Altenhoff, A.; Zoller, S.; Gruissem, W.; Vranová, E. Distinct evolutionary strategies in the GGPPS family from plants. Front. Plant Sci. 2014, 5, 230. [Google Scholar] [CrossRef] [Green Version]
- Chamovitz, D.; Sandmann, G.; Hirschberg, J. Molecular and biochemical characterization of herbicide-resistant mutants of cyanobacteria reveals that phytoene desaturation is a rate-limiting step in carotenoid biosynthesis. J. Biol. Chem. 1993, 268, 17348–17353. [Google Scholar] [CrossRef]
- Cazzonelli, C.I.; Pogson, B.J. Source to sink: Regulation of carotenoid biosynthesis in plants. Trends Plant Sci. 2010, 15, 266–274. [Google Scholar] [CrossRef] [PubMed]
- Layer, G.; Verfürth, K.; Mahlitz, E.; Jahn, D. Oxygen-independent coproporphyrinogen-III oxidase HemN from Escherichia coli. J. Biol. Chem. 2002, 277, 34136–34142. [Google Scholar] [CrossRef] [PubMed]



| Gene ID | Annotation |
|---|---|
| Capana08g001508 | Desiccation-related protein |
| Capana03g001333 | Oxygen-independent coproporphyrinogen-III oxidase-like protein |
| Capana11g000180 | Pheophorbide a oxygenase |
| Capana04g000040 | Dihydroflavonol-4-reductase |
| Capana02g002069 | Protein MOTHER of FT and TF 1 |
| Novel05452 | Metallothionein-like protein type 2 |
| Capana00g003749 | Bifunctional epoxide hydrolase 2 |
| Capana08g000991 | BAG family molecular chaperone regulator 6 |
| Capana08g001915 | Cruciferin PGCRURSE5 |
| Capana02g001627 | Protein SRG1 |
| Capana10g001975 | Zinc finger A20 and AN1 domain-containing stress-associated protein 4 |
| Capana01g004002 | Heat shock 70 kDa protein 7 |
| Capana01g002803 | Probable WRKY transcription factor 23 |
| Capana05g002410 | Receptor-like protein kinase HERK 1 |
| Capana03g003377 | Cytochrome P450 94A1 |
| Capana08g001008 | Desiccation protectant protein LEA14 homolog |
| Capana00g002331 | Phosphopantetheine adenylyltransferase |
| Capana10g000384 | Bifunctional epoxide hydrolase 2 |
| Capana08g001371 | Probable acyl-activating enzyme 6 |
| Capana08g000393 | Nuclear transcription factor Y subunit A-6 |
| Capana10g001621 | Transcription factor HBP-1b (c38) |
| Capana02g001485 | Potassium channel KAT1 |
| Capana07g001105 | UDP-glycosyltransferase 85A1 |
| Capana04g000461 | Cytochrome P450 82A4 |
| Capana06g002950 | S-Norcoclaurine synthase |
| Type | Gene ID | Correlation 1 | ||||||
|---|---|---|---|---|---|---|---|---|
| CaPSY2 | CaLCYE3 | CaPSY3 | CaPDS | CaZISO | CaBCH1 | CaCCS | ||
| β,ε-correlated TFs | ||||||||
| Dof | Capana02g001972 | + | + | - | 0 | 0 | - | 0 |
| G2-like | Capana12g002836 | + | + | - | 0 | 0 | - | 0 |
| bHLH | Capana03g004251 | + | + | - | 0 | - | - | 0 |
| WRKY | Capana03g003085 | + | + | 0 | 0 | - | - | - |
| ERF | Capana04g001803 | + | + | 0 | 0 | 0 | - | - |
| MYB | Capana06g002789 | + | + | 0 | 0 | 0 | - | - |
| B3 | Capana01g004070 | - | - | 0 | + | 0 | + | + |
| NF-YC | Capana11g000539 | - | - | 0 | 0 | 0 | + | + |
| β,β-correlated TFs | ||||||||
| MYB | Capana12g002172 | - | 0 | + | + | + | + | + |
| NAC | Capana04g001537 | 0 | 0 | + | + | + | + | + |
| GRAS | Capana07g001537 | 0 | 0 | + | + | + | + | + |
| MIKC | Capana07g001940 | 0 | 0 | + | + | 0 | + | + |
| MYB | Capana07g001604 | 0 | 0 | - | - | - | - | - |
| bHLH | Capana08g001686 | 0 | 0 | - | - | - | - | - |
| ARF | Capana11g000076 | + | 0 | - | 0 | - | - | - |
| GRF | Capana01g000919 | + | 0 | - | 0 | - | - | - |
| NAC | Capana09g000936 | 0 | 0 | + | 0 | 0 | + | + |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, S.; Song, S.-Y.; Nian, P.; Lv, D.; Jing, Y.; Lu, S.; Wang, Q.; Zhou, F. Transcriptomic Analysis Suggests a Coordinated Regulation of Carotenoid Metabolism in Ripening Chili Pepper (Capsicum annuum var. conoides) Fruits. Antioxidants 2022, 11, 2245. https://doi.org/10.3390/antiox11112245
Song S, Song S-Y, Nian P, Lv D, Jing Y, Lu S, Wang Q, Zhou F. Transcriptomic Analysis Suggests a Coordinated Regulation of Carotenoid Metabolism in Ripening Chili Pepper (Capsicum annuum var. conoides) Fruits. Antioxidants. 2022; 11(11):2245. https://doi.org/10.3390/antiox11112245
Chicago/Turabian StyleSong, Shuyan, Shu-Yuan Song, Peiwen Nian, Dexin Lv, Yunhe Jing, Shan Lu, Qiang Wang, and Fei Zhou. 2022. "Transcriptomic Analysis Suggests a Coordinated Regulation of Carotenoid Metabolism in Ripening Chili Pepper (Capsicum annuum var. conoides) Fruits" Antioxidants 11, no. 11: 2245. https://doi.org/10.3390/antiox11112245
APA StyleSong, S., Song, S.-Y., Nian, P., Lv, D., Jing, Y., Lu, S., Wang, Q., & Zhou, F. (2022). Transcriptomic Analysis Suggests a Coordinated Regulation of Carotenoid Metabolism in Ripening Chili Pepper (Capsicum annuum var. conoides) Fruits. Antioxidants, 11(11), 2245. https://doi.org/10.3390/antiox11112245







