Antioxidant and Anti-Inflammatory Properties of Phytochemicals Found in the Yucca Genus
Abstract
1. Introduction
2. Yucca Genus
3. Ethnobotanical Use
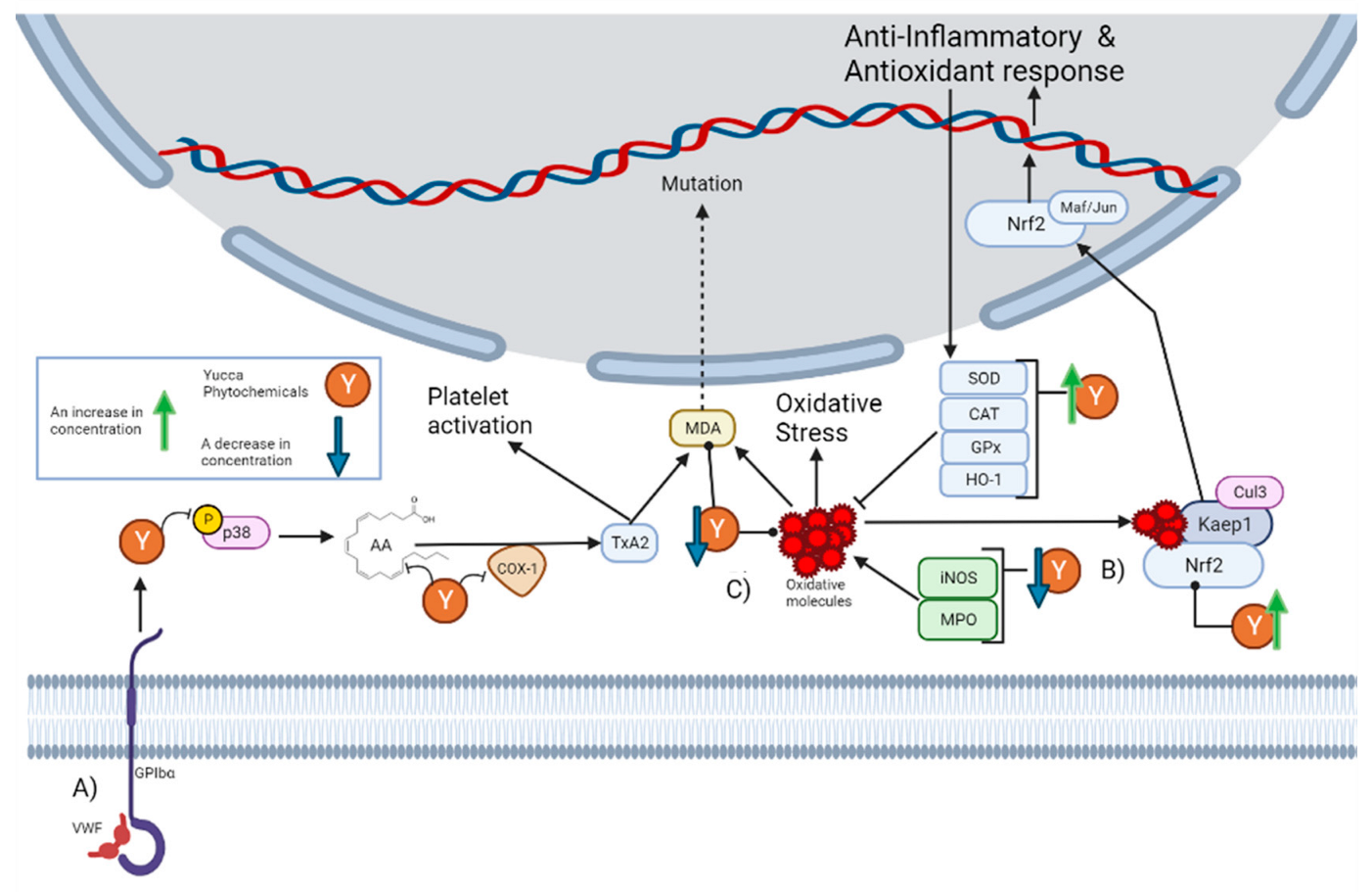
4. Antioxidant and Anti-Inflammatory Activities
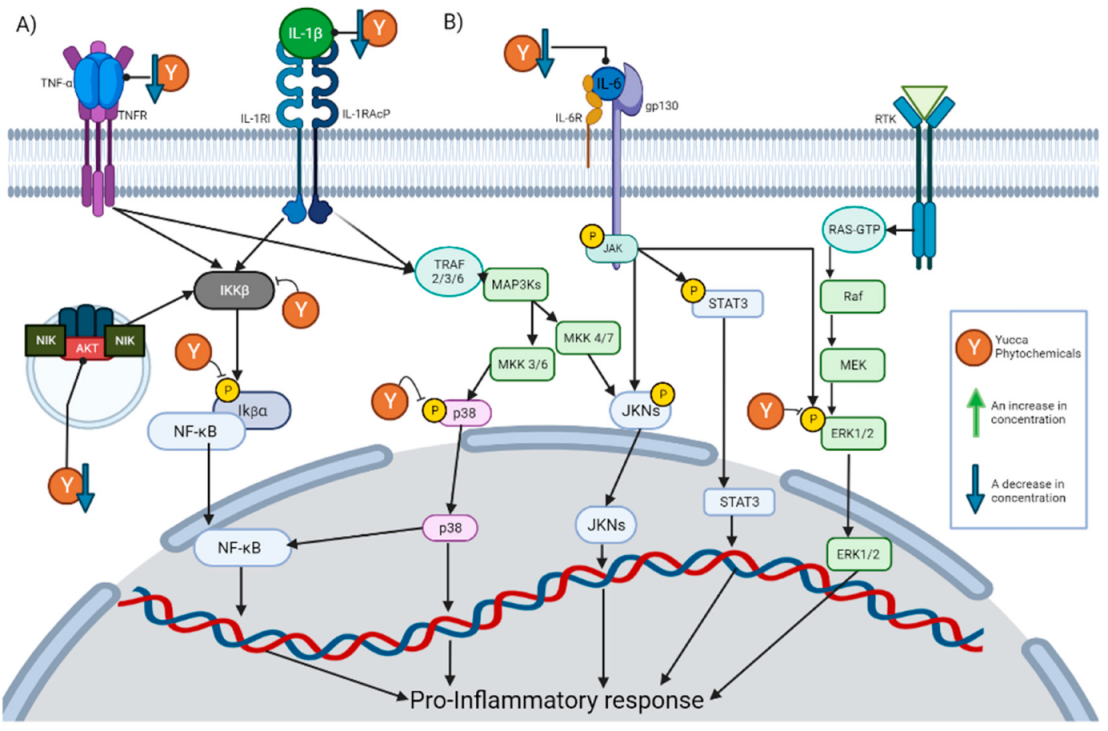
4.1. Inhibition of NF-κB Signaling Pathway
4.2. Inhibition of the MAPK Signaling Pathway
4.3. Activation of the Nrf2 Signaling Pathway
4.4. Free Radical Scavenging Activity
5. Phytochemistry
5.1. Phenolic Compounds
5.2. Saponins
5.3. Glycosides
5.4. Fatty Acids
5.5. Other Phytochemicals
5.6. Availability of Reported Phytochemicals
6. Future Perspectives
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| TNF-α | Tumor necrosis factor-α |
| IL-6 | Interleukin-6 |
| iNOS | Inducible nitric oxide synthase |
| IL-1β | Interleukin 1β |
| GPx | Glutathione peroxidase |
| CAT | Catalase |
| SOD | Superoxide dismutase |
| MAPK | Mitogen-Activated Protein Kinase |
| NF-κB | Nuclear factor kappa B |
| Nrf2 | Nuclear factor erythroid 2–related factor |
| FDA | Food and Drug Administration |
| ROS | Reactive oxygen species |
| RNS | Reactive nitrogen species |
| MDA | Malonaldehyde |
| SIRT1 | Sirtuin 1 |
| PGE2 | Prostaglandin E2 |
| COX-2 | Cyclooxygenase-2 |
| IC50 | Half maximal inhibitory concentration |
| 8-oxo-dG | 8-Oxo-2’-deoxyguanosine |
| MMP-9 | Matrix metalloproteinase 9 |
| TEAC | Trolox equivalent antioxidant capacity |
| NLRP3 | Nucleotide-binding oligomerization domain-like receptor containing domain 3 of pyrin |
| PDE4 | Phosphodiesterase 4 |
| NO | Nitric oxide |
| ERK | Extracellular signal-regulated kinase |
| LPO | Lipid Peroxidation |
| TLR4 | Toll-like receptor 4 |
| MCP-1 | Monocyte chemoattractant protein 1 |
| MPO | Myeloperoxidase |
| GSH | Glutathione |
| HO-1 | Heme oxygenase 1 |
| IκBα | Inhibitor of nuclear factor kappa B |
| LPS | Lipopolysaccharide |
| MIP-2 | Macrophage inflammatory protein 2 |
| IKK | IkB kinase |
| JNK | c-Jun amino terminal kinase |
| ICAM-1 | Intercellular adhesion molecule |
| VEGF | Vascular endothelial growth factor |
| GST | Glutathione-S-transferase |
| CRP | Reactive C-protein |
| PAR-1 | Protease-activated receptor 1 |
| TXB2 | Thromboxane B2 |
| IgE | Immunoglobulin E |
| TBARS | Thiobarbituric acid-reactive substances |
| PKA | Protein kinase A |
| mTOR | Mammalian target of rapamycin |
| EGFR | Epidermal growth factor receptor |
| Keap1 | Kelch-like ECH-associated protein 1 |
| COX-1 | Cyclooxygenase-1 |
| VCAM1 | Vascular cell adhesion protein 1 |
| VWF | von Willebrand factor |
| GPIbα | platelet adhesion receptor glycoprotein |
| AA | arachidonic acid |
| TxA2 | Thromboxane A2 |
| IL-13 | Interleukin-13 |
| NOS | Nitric oxide synthase |
| PUFAs | Polyunsaturated fatty acids |
| PEs | Phosphatidylethanolamines |
| IL-8 | Interleukin 8 |
| IL-1RI | IL-1 type I receptor |
| IL-1RAcP | IL-1R accessory protein |
| H2O2 | Hydrogen peroxide |
| NIK | NF-kB inducing kinase |
| TRAF | TNF receptor-associated factor |
| RTK | Receptor tyrosine kinase |
| Cul3 | Cullin 3 |
| NQO1 | NADP(H) quinone oxidoreductase |
| ORAC | Oxygen radical absorbance capacity |
| DPPH | 1,1-diphenyl-2-picrylhydrazyl |
References
- Karamova, N.; Gumerova, S.; Hassan, G.O.; Abdul-Hafeez, E.Y.; Ibrahim, O.H.M.; Orabi, M.A.A.; Ilinskaya, O. Antioxidant and Antimutagenic Potential of Extracts of Some Agavaceae Family Plants. Bionanoscience 2016, 6, 591–593. [Google Scholar] [CrossRef]
- Patel, S. Yucca: A medicinally significant genus with manifold therapeutic attributes. Nat. Prod. Bioprospecting 2012, 2, 231–234. [Google Scholar] [CrossRef]
- Sahoo, S.; Kaur, D.; Sethi, A.; Chandrahas; Saini, A.; Chandra, M. Effect of Dietary Supplementation of Yucca schidigera Extract on the Performance and Litter Quality of Broilers in Winter Season. Anim. Nutr. Feed. Technol. 2016, 16, 475–484. [Google Scholar] [CrossRef]
- Borges, R.S.; Ortiz, B.L.S.; Pereira, A.C.M.; Keita, H.; Carvalho, J.C.T. Rosmarinus officinalis essential oil: A review of its phytochemistry, anti-inflammatory activity, and mechanisms of action involved. J. Ethnopharmacol. 2018, 229, 29–45. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Deng, H.; Cui, H.; Fang, J.; Zuo, Z.; Deng, J.; Li, Y.; Wang, X.; Zhao, L. Inflammatory Responses and Inflamma-tion-Associated Diseases in Organs. Oncotarget 2018, 9, 7204. [Google Scholar] [CrossRef]
- Garcia, E.I.C.; Elghandour, M.M.M.Y.; Khusro, A.; Alcala-Canto, Y.; Tirado-González, D.N.; Barbabosa-Pliego, A.; Salem, A.Z.M. Dietary Supplements of Vitamins E, C, and β-Carotene to Reduce Oxidative Stress in Horses: An Overview. J. Equine Vet. Sci. 2022, 110, 103863. [Google Scholar] [CrossRef]
- Debnath, T.; Kim, D.H.; Lim, B.O. Natural Products as a Source of Anti-Inflammatory Agents Associated with Inflammatory Bowel Disease. Molecules 2013, 18, 7253–7270. [Google Scholar] [CrossRef]
- López-Ramírez, Y.; Palomeque-Carlín, A.; Chávez Ortiz, L.I.; de la Rosa-Carrillo, M.D.L.; Pérez-Molphe-Balch, E. Micropropagation of Yucca Species. Plant Cell Cult. Protoc. 2018, 1815, 171–177. [Google Scholar]
- Pellmyr, O. Yuccas, Yucca Moths, and Coevolution: A Review. Ann. Mo. Bot. Gard. 2003, 90, 35–55. [Google Scholar] [CrossRef]
- Eguiarte, L.E.; Leebens-Mack, J.; Heyduk, K. Editorial: Recent Advances and Future Perspectives for Agavoideae Research: Agave, Yucca and Related Taxa. Front. Plant Sci. 2021, 12, 687596. [Google Scholar] [CrossRef]
- The Plant List. Available online: http://www.theplantlist.org/ (accessed on 5 October 2022).
- Anderson, M.K.; Keeley, J.E. Native Peoples’ Relationship to the California Chaparral. In Valuing Chaparral; Springer: Berlin/Heidelberg, Germany, 2018; pp. 79–121. [Google Scholar]
- Corrales, V.M.S. Léxico Relativo al Ámbito Del Tamal En Costa Rica. Káñina Rev. Artes Letras 2006, 30, 191–197. [Google Scholar]
- Bernier, G.R. Ethnobotany of the Northern Cheyenne: Medicinal Plants. Masters Thesis, University of Montana, Missoula, MT, USA, 2004. [Google Scholar]
- Lim, T.K.; Lim, T.K. Yucca Filamentosa. Edible Medicinal and Non-Medicinal Plants. Flowers 2014, 7, 134–137. [Google Scholar]
- Gupta, S.; Duraiswamy, B.; Muthureddy Nataraj, S.K.; Rama Satyanarayana Raju, K.; Babu, U.V. Inhibitory Potential of Yucca Gloriosa, L. Extract and Isolated Gloriosaol Isomeric Mixture on Ovalbumin Induced Airway Hyperresponsiveness in Balb/C Mice. Clin. Pharm. Biopharm. 2014, 2, 2. [Google Scholar] [CrossRef]
- Bingham, R.; Bellew, B.A.; Bellew, J.G. Yucca Plant Saponin in the Management of Arthritis. J. Appl. Nutr. 1975, 17, 45–51. [Google Scholar]
- Kim, S.-W.; Park, S.-K.; Kang, S.; Kang, H.-C.; Oh, H.-J.; Bae, C.-Y.; Bae, D.-H. Hypocholesterolemic Property OfYucca Schidigera AndQuillaja Saponaria Extracts in Human Body. Arch. Pharm. Res. 2003, 26, 1042–1046. [Google Scholar] [CrossRef]
- Gutiérrez-García, G.J.; Quintana-Romero, L.A.; Morales-Figueroa, G.G.; Esparza-Romero, J.; Pérez-Morales, R.; López-Mata, M.A.; Juárez, J.; Sánchez-Escalante, J.J.; Peralta, E.; Quihui-Cota, L. Effect of Yucca Baccata Butanolic Extract on the Shelf Life of Chicken and Development of an Antimicrobial Packaging for Beef. Food Control. 2021, 127, 108142. [Google Scholar] [CrossRef]
- Del Trujillo, R.C.L. Characterization and Evaluation of yucca baccata saponins against giardia intestinalis trophozoites in vitro. Doctoral Thesis, Centro de Investigación en Alimentación y Desarrollo, Mazatlán Mexico, 2017. [Google Scholar]
- Mokbli, S.; Nehdi, I.A.; Sbihi, H.M.; Tan, C.P.; Al-Resayes, S.I.; Rashid, U. Yucca Aloifolia Seed Oil: A New Source of Bioactive Compounds. Waste Biomass Valorization 2018, 9, 1087–1093. [Google Scholar] [CrossRef]
- Thomas, M.R.; Storey, R.F. The Role of Platelets in Inflammation. Thromb. Haemost. 2015, 114, 449–458. [Google Scholar]
- Olas, B.; Wachowicz, B.; Stochmal, A.; Oleszek, W. Inhibition of Blood Platelet Adhesion and Secretion by Different Phenolics from Yucca Schidigera Roezl. Bark. Nutrition 2005, 21, 199–206. [Google Scholar] [CrossRef]
- Driver, B.; Marks, D.C.; van der Wal, D.E. Not All (N) SAID and Done: Effects of Nonsteroidal Anti-inflammatory Drugs and Paracetamol Intake on Platelets. Res. Pr. Thromb. Haemost. 2020, 4, 36–45. [Google Scholar] [CrossRef]
- Marion-Letellier, R.; Savoye, G.; Ghosh, S. Polyunsaturated Fatty Acids and Inflammation. IUBMB Life 2015, 67, 659–667. [Google Scholar] [CrossRef] [PubMed]
- Wenzig, E.M.; Oleszek, W.; Stochmal, A.; Kunert, O.; Bauer, R. Influence of Phenolic Constituents from Yucca Schidigera Bark on Arachidonate Metabolism in Vitro. J. Agric. Food Chem. 2008, 56, 8885–8890. [Google Scholar] [CrossRef] [PubMed]
- Casaro, M.; Souza, V.R.; Oliveira, F.A.; Ferreira, C.M. OVA-Induced Allergic Airway Inflammation Mouse Model. In Pre-Clinical Models: Techniques and Protocols; Guest, P.C., Ed.; Springer New York: New York, NY, USA, 2019; Volume 1916, pp. 297–301. [Google Scholar]
- Iqbal, M.; Verpoorte, R.; Korthout, H.A.A.J.; Mustafa, N.R. Phytochemicals as a Potential Source for TNF-α Inhibitors. Phytochem. Rev. 2013, 12, 65–93. [Google Scholar] [CrossRef]
- Israël, A. The IKK Complex: An Integrator of All Signals That Activate NF-ΚB? Trends Cell. Biol. 2000, 10, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.E.; O’Keefe, R.A.; Grandis, J.R. Targeting the IL-6/JAK/STAT3 Signalling Axis in Cancer. Nat. Rev. Clin. Oncol. 2018, 15, 234–248. [Google Scholar] [CrossRef]
- Korhonen, R.; Lahti, A.; Kankaanranta, H.; Moilanen, E. Nitric Oxide Production and Signaling in Inflammation. Curr. Drug Targets Inflamm. Allergy 2005, 4, 471–479. [Google Scholar] [CrossRef]
- Pautz, A.; Art, J.; Hahn, S.; Nowag, S.; Voss, C.; Kleinert, H. Regulation of the expression of inducible nitric oxide synthase. Nitric Oxide 2010, 23, 75–93. [Google Scholar] [CrossRef]
- Rochette, L.; Lorin, J.; Zeller, M.; Guilland, J.-C.; Lorgis, L.; Cottin, Y.; Vergely, C. Nitric Oxide Synthase Inhibition and Oxi-dative Stress in Cardiovascular Diseases: Possible Therapeutic Targets? Pharmacol. Ther. 2013, 140, 239–257. [Google Scholar] [CrossRef]
- Marzocco, S.; Piacente, S.; Pizza, C.; Oleszek, W.; Stochmal, A.; Pinto, A.; Sorrentino, R.; Autore, G. Inhibition of inducible nitric oxide synthase expression by yuccaol C from Yucca schidigera roezl. Life Sci. 2004, 75, 1491–1501. [Google Scholar] [CrossRef]
- Hawkins, C.L.; Davies, M.J. Role of Myeloperoxidase and Oxidant Formation in the Extracellular Environment in Inflamma-tion-Induced Tissue Damage. Free. Radic. Biol. Med. 2021, 172, 633–651. [Google Scholar] [CrossRef]
- Malle, E.; Furtmüller, P.G.; Sattler, W.; Obinger, C. Myeloperoxidase: A Target for New Drug Development? Br. J. Pharmacol. 2007, 152, 838–854. [Google Scholar] [CrossRef]
- Kawai, Y.; Kiyokawa, H.; Kimura, Y.; Kato, Y.; Tsuchiya, K.; Terao, J. Hypochlorous Acid-Derived Modification of Phospho-lipids: Characterization of Aminophospholipids as Regulatory Molecules for Lipid Peroxidation. Biochemistry 2006, 45, 14201–14211. [Google Scholar] [CrossRef]
- Ayala, A.; Muñoz, M.F.; Argüelles, S. Lipid Peroxidation: Production, Metabolism, and Signaling Mechanisms of Malondial-dehyde and 4-Hydroxy-2-Nonenal. Oxidative Med. Cell. Longev. 2014, 2014, 360438. [Google Scholar] [CrossRef]
- Singh, Z.; Karthigesu, I.P.; Singh, P.; Rupinder, K. Use of Malondialdehyde as a Biomarker for Assessing Oxidative Stress in Different Disease Pathologies: A Review. Iran J. Public Health 2014, 43, 7–16. [Google Scholar]
- Marnett, L.J. Oxy radicals, lipid peroxidation and DNA damage. Toxicology 2002, 181, 219–222. [Google Scholar] [CrossRef]
- Cigerci, H.; Fidan, A.F.; Konuk, M.; Yuksel, H.; Kucukkurt, I.; Eryavuz, A.; Sozbilir, N.B. The protective potential of Yucca schidigera (Sarsaponin 30®) against nitrite-induced oxidative stress in rats. J. Nat. Med. 2009, 63, 311–317. [Google Scholar] [CrossRef]
- Li, S.; Wang, R.; Dai, Z.; Wang, C.; Wu, Z. Dietary supplementation with Yucca schidigera extract alleviated heat stress-induced unfolded protein response and oxidative stress in the intestine of Nile tilapia (Oreochromis niloticus). Ecotoxicol. Environ. Saf. 2021, 219, 112299. [Google Scholar] [CrossRef]
- Gabay, C.; Lamacchia, C.; Palmer, G. IL-1 pathways in inflammation and human diseases. Nat. Rev. Rheumatol. 2010, 6, 232–241. [Google Scholar] [CrossRef]
- Weber, A.; Wasiliew, P.; Kracht, M. Interleukin-1β (IL-1β) Processing Pathway. Sci. Signal 2010, 3, cm2. [Google Scholar] [CrossRef]
- Wang, L.; Wu, D.; Fan, Z.; Li, H.; Li, J.; Zhang, Y.; Xu, Q.; Wang, G.; Zhu, Z. Effect of Yucca schidigera extract on the growth performance, intestinal antioxidant status, immune response, and tight junctions of mirror carp (Cyprinus carpio). Fish Shellfish. Immunol. 2020, 103, 211–219. [Google Scholar] [CrossRef]
- Culhuac, E.B.; Elghandour, M.M.M.Y.; Adegbeye, M.J.; Barbabosa-Pliego, A.; Salem, A.Z.M. Influence of Dietary Selenium on the Oxidative Stress in Horses. Biol. Trace Element Res. 2022, 201, 1695–1703. [Google Scholar] [CrossRef] [PubMed]
- Smale, S.T. Dimer-specific Regulatory Mechanisms within the NF-κB Family of Transcription Factors. Immunol. Rev. 2012, 246, 193–204. [Google Scholar] [CrossRef]
- Yu, H.; Lin, L.; Zhang, Z.; Zhang, H.; Hu, H. Targeting NF-ΚB Pathway for the Therapy of Diseases: Mechanism and Clinical Study. Signal Transduct. Target 2020, 5, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, T. The Nuclear Factor NF-ΚB Pathway in Inflammation. Cold Spring Harb. Perspect. Biol. 2009, 1, a001651. [Google Scholar] [CrossRef] [PubMed]
- Arbabi, S.; Maier, R. v Mitogen-Activated Protein Kinases. Crit. Care Med. 2002, 30, S74–S79. [Google Scholar] [CrossRef]
- Kaminska, B. MAPK signalling pathways as molecular targets for anti-inflammatory therapy—from molecular mechanisms to therapeutic benefits. Biochim. Biophys. Acta 2005, 1754, 253–262. [Google Scholar] [CrossRef]
- Soares-Silva, M.; Diniz, F.F.; Gomes, G.N.; Bahia, D. The Mitogen-Activated Protein Kinase (MAPK) Pathway: Role in Immune Evasion by Trypanosomatids. Front. Microbiol. 2016, 7, 183. [Google Scholar] [CrossRef]
- Kong, T.; Liu, M.; Ji, B.; Bai, B.; Cheng, B.; Wang, C. Role of the Extracellular Signal-Regulated Kinase 1/2 Signaling Pathway in Ischemia-Reperfusion Injury. Front. Physiol. 2019, 10, 1038. [Google Scholar] [CrossRef]
- Beamer, E.; Corrêa, S.A.L. The P38MAPK-MK2 Signaling Axis as a Critical Link Between Inflammation and Synaptic Trans-mission. Front. Cell. Dev. Biol. 2021, 9, 635636. [Google Scholar] [CrossRef]
- Wagner, E.F.; Nebreda, Á.R. Signal integration by JNK and p38 MAPK pathways in cancer development. Nat. Rev. Cancer 2009, 9, 537–549. [Google Scholar] [CrossRef]
- Park, J.G.; Aziz, N.; Cho, J.Y. MKK7, the essential regulator of JNK signaling involved in cancer cell survival: A newly emerging anticancer therapeutic target. Ther. Adv. Med Oncol. 2019, 11. [Google Scholar] [CrossRef]
- Roy, P.K.; Rashid, F.; Bragg, J.; A Ibdah, J. Role of the JNK signal transduction pathway in inflammatory bowel disease. World J. Gastroenterol. 2008, 14, 200–202. [Google Scholar] [CrossRef]
- Loboda, A.; Damulewicz, M.; Pyza, E.; Jozkowicz, A.; Dulak, J. Role of Nrf2/HO-1 system in development, oxidative stress response and diseases: An evolutionarily conserved mechanism. Cell. Mol. Life Sci. 2016, 73, 3221–3247. [Google Scholar] [CrossRef]
- Svensson, G.P.; Pellmyr, O.; Raguso, R.A. Strong Conservation of Floral Scent Composition in Two Allopatric Yuccas. J. Chem. Ecol. 2006, 32, 2657–2665. [Google Scholar] [CrossRef]
- Cuadrado, A.; Manda, G.; Hassan, A.; Alcaraz, M.J.; Barbas, C.; Daiber, A.; Ghezzi, P.; León, R.; López, M.G.; Oliva, B.; et al. Transcription Factor NRF2 as a Therapeutic Target for Chronic Diseases: A Systems Medicine Approach. Pharmacol. Rev. 2018, 70, 348–383. [Google Scholar] [CrossRef]
- Liu, Q.; Gao, Y.; Ci, X. Role of Nrf2 and Its Activators in Respiratory Diseases. Oxidative Med. Cell. Longev. 2019, 2019, 7090534. [Google Scholar] [CrossRef]
- Huang, Y.; Li, W.; Su, Z.; Kong, A.-N.T. The Complexity of the Nrf2 Pathway: Beyond the Antioxidant Response. J. Nutr. Biochem. 2015, 26, 1401–1413. [Google Scholar] [CrossRef]
- Akhter, S.; Irfan, H.M.; Jahan, S.; Shahzad, M.; Latif, M.B. Nerolidol: A Potential Approach in Rheumatoid Arthritis through Reduction of TNF-α, IL-1β, IL-6, NF-KB, COX-2 and Antioxidant Effect in CFA-Induced Arthritic Model. Inflammopharmacology 2022, 30, 537–548. [Google Scholar] [CrossRef]
- Silva-Islas, C.A.; Maldonado, P.D. Canonical and Non-Canonical Mechanisms of Nrf2 Activation. Pharm. Res. 2018, 134, 92–99. [Google Scholar] [CrossRef]
- Campbell, N.K.; Fitzgerald, H.K.; Dunne, A. Regulation of Inflammation by the Antioxidant Haem Oxygenase. Nat. Rev. Immunol. 2021, 21, 411–425. [Google Scholar] [CrossRef]
- Muhammad, Z.; Nasir, R.; Asim, M.; Fozia, A.; Munawar, I.; Muhammad, M.; Muhammad, S. Antioxidant, Antibacterial, Antifungal Activities and Phytochemical Analysis of Dagger (Yucca Aloifolia) Leaves Extracts. J. Med. Plants Res. 2013, 7, 243–249. [Google Scholar]
- Piacente, S.; Pizza, C.; Oleszek, W. Saponins and Phenolics of Yucca Schidigera Roezl: Chemistry and Bioactivity. Phytochem. Rev. 2005, 4, 177–190. [Google Scholar] [CrossRef]
- Piacente, S.; Montoro, P.; Oleszek, W.; Pizza, C. Yucca s Chidigera Bark: Phenolic Constituents and Antioxidant Activity. J. Nat. Prod. 2004, 67, 882–885. [Google Scholar] [CrossRef] [PubMed]
- Bassarello, C.; Bifulco, G.; Montoro, P.; Skhirtladze, A.; Benidze, M.; Kemertelidze, E.; Pizza, C.; Piacente, S. Yucca Gloriosa: A Source of Phenolic Derivatives with Strong Antioxidant Activity. J. Agric. Food Chem. 2007, 55, 6636–6642. [Google Scholar] [CrossRef] [PubMed]
- Morales-Figueroa, G.-G.; Pereo-Vega, G.D.; Reyna-Murrieta, M.E.; Pérez-Morales, R.; López-Mata, M.A.; Sánchez-Escalante, J.J.; Tapia-Rodriguez, M.R.; Ayala-Zavala, J.F.; Juárez, J.; Quihui-Cota, L. Antibacterial and Antioxidant Properties of Extracts of Yucca Baccata, a Plant of Northwestern Mexico, against Pathogenic Bacteria. Biomed. Res. Int. 2022, 2022, 9158836. [Google Scholar] [CrossRef]
- Cordell, G.A. Phytochemistry and Traditional Medicine–A Revolution in Process. Phytochem. Lett. 2011, 4, 391–398. [Google Scholar] [CrossRef]
- Xiao, L.; Sun, Y.; Tsao, R. Paradigm Shift in Phytochemicals Research: Evolution from Antioxidant Capacity to Anti-Inflammatory Effect and to Roles in Gut Health and Metabolic Syndrome. J. Agric. Food Chem. 2022, 70, 8551–8568. [Google Scholar] [CrossRef]
- Vuolo, M.M.; Lima, V.S.; Junior, M.R.M. Phenolic Compounds: Structure, Classification, and Antioxidant Power. In Bioactive Compounds; Elsevier: Amsterdam, The Netherlands, 2019; pp. 33–50. [Google Scholar]
- Kumar, S.; Abedin, M.M.; Singh, A.K.; Das, S. Role of Phenolic Compounds in Plant-Defensive Mechanisms. Plant Phenolics Sustain. Agric. 2020, 1, 517–532. [Google Scholar]
- Ambriz-Pérez, D.L.; Leyva-López, N.; Gutierrez-Grijalva, E.P.; Heredia, J.B. Phenolic Compounds: Natural Alternative in Inflammation Treatment. A Review. Cogent Food Agric. 2016, 2, 1131412. [Google Scholar]
- Ememe, M.U.; Mshelia, W.P.; Ayo, J.O. Ameliorative Effects of Resveratrol on Oxidative Stress Biomarkers in Horses. J. Equine Vet. Sci. 2015, 35, 518–523. [Google Scholar] [CrossRef]
- Torres, P.; Avila, J.G.; de Vivar, A.R.; Garcı́a, A.M.; Marı́n, J.C.; Aranda, E.; Céspedes, C.L. Antioxidant and Insect Growth Regulatory Activities of Stilbenes and Extracts from Yucca Periculosa. Phytochemistry 2003, 64, 463–473. [Google Scholar] [CrossRef]
- Montoro, P.; Skhirtladze, A.; Bassarello, C.; Perrone, A.; Kemertelidze, E.; Pizza, C.; Piacente, S. Determination of Phenolic Compounds in Yucca Gloriosa Bark and Root by LC–MS/MS. J. Pharm. Biomed. Anal. 2008, 47, 854–859. [Google Scholar] [CrossRef]
- Juarez-Trujillo, N.; Monribot-Villanueva, J.L.; Jiménez-Fernández, V.M.; Suárez-Montaño, R.; Aguilar-Colorado, Á.S.; Guerrero-Analco, J.A.; Jiménez, M. Phytochemical Characterization of Izote (Yucca Elephantipes) Flowers. J. Appl. Bot. Food Qual. 2018, 91, 202–210. [Google Scholar]
- Pan, W.; Yu, H.; Huang, S.; Zhu, P. Resveratrol Protects against TNF-α-Induced Injury in Human Umbilical Endothelial Cells through Promoting Sirtuin-1-Induced Repression of NF-KB and P38 MAPK. PLoS ONE 2016, 11, e0147034. [Google Scholar] [CrossRef]
- Zykova, T.A.; Zhu, F.; Zhai, X.; Ma, W.; Ermakova, S.P.; Lee, K.W.; Bode, A.M.; Dong, Z. Resveratrol Directly Targets COX-2 to Inhibit Carcinogenesis. Molecular Carcinogenesis: Published in cooperation with the University of Texas MD Anderson Cancer Center 2008, 47, 797–805. [Google Scholar] [CrossRef]
- Wang, T.; Dai, F.; Li, G.-H.; Chen, X.-M.; Li, Y.-R.; Wang, S.-Q.; Ren, D.-M.; Wang, X.-N.; Lou, H.-X.; Zhou, B. Trans-4, 4′-Dihydroxystilbene Ameliorates Cigarette Smoke-Induced Progression of Chronic Obstructive Pulmonary Disease via Inhibiting Oxidative Stress and Inflammatory Response. Free. Radic Biol. Med. 2020, 152, 525–539. [Google Scholar] [CrossRef]
- Fan, G.-J.; Liu, X.-D.; Qian, Y.-P.; Shang, Y.-J.; Li, X.-Z.; Dai, F.; Fang, J.-G.; Jin, X.-L.; Zhou, B. 4, 4′-Dihydroxy-Trans-Stilbene, a Resveratrol Analogue, Exhibited Enhanced Antioxidant Activity and Cytotoxicity. Bioorg. Med. Chem. 2009, 17, 2360–2365. [Google Scholar] [CrossRef]
- Olas, B.; Wachowicz, B.; Nowak, P.; Stochmal, A.; Oleszek, W.; Glowacki, R.; Bald, E. Comparative Studies of the Antioxidant Effects of a Naturally Occurring Resveratrol Analogue–Trans-3, 3′, 5, 5′-Tetrahydroxy-4′-Methoxystilbene and Resveratrol–against Oxidation and Nitration of Biomolecules in Blood Platelets. Cell. Biol. Toxicol. 2008, 24, 331–340. [Google Scholar] [CrossRef]
- Lin, Y.; Luo, T.; Weng, A.; Huang, X.; Yao, Y.; Fu, Z.; Li, Y.; Liu, A.; Li, X.; Chen, D. Gallic Acid Alleviates Gouty Arthritis by Inhibiting NLRP3 Inflammasome Activation and Pyroptosis through Enhancing Nrf2 Signaling. Front. Immunol. 2020, 11, 580593. [Google Scholar] [CrossRef]
- Attallah, N.G.M.; El-Sherbeni, S.A.; El-Kadem, A.H.; Elekhnawy, E.; El-Masry, T.A.; Elmongy, E.I.; Altwaijry, N.; Negm, W.A. Elucidation of the Metabolite Profile of Yucca Gigantea and Assessment of Its Cytotoxic, Antimicrobial, and Anti-Inflammatory Activities. Molecules 2022, 27, 1329. [Google Scholar] [CrossRef]
- El Sayed, A.M.; Basam, S.M.; Marzouk, H.S.; El-Hawary, S. LC–MS/MS and GC–MS Profiling as Well as the Antimicrobial Effect of Leaves of Selected Yucca Species Introduced to Egypt. Sci. Rep. 2020, 10, 1–15. [Google Scholar] [CrossRef] [PubMed]
- JIANG, D.; ZHANG, M.; ZHANG, Q.; CHEN, Y.; WU, W.; Xiang, M.U.; Wu, C. Influence of Gallic Acid on Porcine Neutrophils Phosphodiesterase 4, IL-6, TNF-α and Rat Arthritis Model. J. Integr. Agric. 2015, 14, 758–764. [Google Scholar] [CrossRef]
- Fu, X.; Lyu, X.; Liu, H.; Zhong, D.; Xu, Z.; He, F.; Huang, G. Chlorogenic Acid Inhibits BAFF Expression in Collagen-Induced Arthritis and Human Synoviocyte MH7A Cells by Modulating the Activation of the NF-ΚB Signaling Pathway. J. Immunol. Res. 2019, 2019, 8042097. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Raouf, O.M.; El-Sayed, E.M.; Manie, M.F. Cinnamic Acid and Cinnamaldehyde Ameliorate Cisplatin-induced Splenotoxicity in Rats. J. Biochem. Mol. Toxicol. 2015, 29, 426–431. [Google Scholar] [CrossRef] [PubMed]
- Ikram, M.; Muhammad, T.; Rehman, S.U.; Khan, A.; Jo, M.G.; Ali, T.; Kim, M.O. Hesperetin Confers Neuroprotection by Regulating Nrf2/TLR4/NF-ΚB Signaling in an Aβ Mouse Model. Mol. Neurobiol. 2019, 56, 6293–6309. [Google Scholar] [CrossRef]
- Jo, S.H.; Kim, M.E.; Cho, J.H.; Lee, Y.; Lee, J.; Park, Y.-D.; Lee, J.S. Hesperetin Inhibits Neuroinflammation on Microglia by Suppressing Inflammatory Cytokines and MAPK Pathways. Arch. Pharm. Res. 2019, 42, 695–703. [Google Scholar] [CrossRef]
- Hajizadeh, A.; Azizi, S. Effects of Naringenin on Experimentally Induced Rheumatoid Arthritis in Wistar Rats. Arch. Razi. Inst. 2021, 76, 903. [Google Scholar]
- Park, H.Y.; Kim, G.-Y.; Choi, Y.H. Naringenin Attenuates the Release of Pro-Inflammatory Mediators from Lipopolysaccharide-Stimulated BV2 Microglia by Inactivating Nuclear Factor-ΚB and Inhibiting Mitogen-Activated Protein Kinases. Int. J. Mol. Med. 2012, 30, 204–210. [Google Scholar]
- Tsilioni, I.; Taliou, A.; Francis, K.; Theoharides, T.C. Children with Autism Spectrum Disorders, Who Improved with a Luteolin-Containing Dietary Formulation, Show Reduced Serum Levels of TNF and IL. Transl. Psychiatry 2015, 5, e647. [Google Scholar] [CrossRef]
- Oyagbemi, A.A.; Omobowale, T.O.; Ola-Davies, O.E.; Asenuga, E.R.; Ajibade, T.O.; Adejumobi, O.A.; Afolabi, J.M.; Ogunpolu, B.S.; Falayi, O.O.; Saba, A.B. Luteolin-mediated Kim-1/NF-kB/Nrf2 Signaling Pathways Protects Sodium Fluoride-induced Hypertension and Cardiovascular Complications. Biofactors 2018, 44, 518–531. [Google Scholar] [CrossRef]
- Paciello, F.; di Pino, A.; Rolesi, R.; Troiani, D.; Paludetti, G.; Grassi, C.; Fetoni, A.R. Anti-Oxidant and Anti-Inflammatory Effects of Caffeic Acid: In Vivo Evidences in a Model of Noise-Induced Hearing Loss. Food Chem. Toxicol. 2020, 143, 111555. [Google Scholar] [CrossRef]
- Gülçin, İ. Antioxidant Activity of Caffeic Acid (3, 4-Dihydroxycinnamic Acid). Toxicology 2006, 217, 213–220. [Google Scholar] [CrossRef]
- Sun, L.-C.; Zhang, H.-B.; Gu, C.-D.; Guo, S.-D.; Li, G.; Lian, R.; Yao, Y.; Zhang, G.-Q. Protective Effect of Acacetin on Sepsis-Induced Acute Lung Injury via Its Anti-Inflammatory and Antioxidative Activity. Arch. Pharm. Res. 2018, 41, 1199–1210. [Google Scholar] [CrossRef]
- Zhang, H.; Li, X.; Wu, K.; Wang, M.; Liu, P.; Wang, X.; Deng, R. Antioxidant Activities and Chemical Constituents of Flavonoids from the Flower of Paeonia Ostii. Molecules 2016, 22, 5. [Google Scholar] [CrossRef]
- Yilmaz, Y.; Toledo, R.T. Major Flavonoids in Grape Seeds and Skins: Antioxidant Capacity of Catechin, Epicatechin, and Gallic. Acid. J. Agric. Food Chem. 2004, 52, 255–260. [Google Scholar] [CrossRef]
- Li, X.; Wang, X.; Chen, D.; Chen, S. Antioxidant Activity and Mechanism of Protocatechuic Acid in Vitro. Funct. Foods Health Dis. 2011, 1, 232–244. [Google Scholar] [CrossRef]
- Lende, A.B.; Kshirsagar, A.D.; Deshpande, A.D.; Muley, M.M.; Patil, R.R.; Bafna, P.A.; Naik, S.R. Anti-Inflammatory and Analgesic Activity of Protocatechuic Acid in Rats and Mice. Inflammopharmacology 2011, 19, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Rabelo, T.K.; Guimaraes, A.G.; Oliveira, M.A.; Gasparotto, J.; Serafini, M.R.; de Souza Araújo, A.A.; Quintans-Junior, L.J.; Moreira, J.C.F.; Gelain, D.P. Shikimic Acid Inhibits LPS-Induced Cellular pro-Inflammatory Cytokines and Attenuates Mechanical Hyperalgesia in Mice. Int. Immunopharmacol. 2016, 39, 97–105. [Google Scholar] [CrossRef] [PubMed]
- López-Ramírez, Y.; Cabañas-García, E.; Areche, C.; Trejo-Tapia, G.; Pérez-Molphe-Balch, E.; Gómez-Aguirre, Y.A. Callus Induction and Phytochemical Profiling of Yucca Carnerosana (Trel.) McKelvey Obtained from in Vitro Cultures. Rev. Mex. Ing. Quim. 2021, 20, 823–837. [Google Scholar] [CrossRef]
- Mokdad-Bzeouich, I.; Mustapha, N.; Sassi, A.; Bedoui, A.; Ghoul, M.; Ghedira, K.; Chekir-Ghedira, L. Investigation of Immunomodulatory and Anti-Inflammatory Effects of Eriodictyol through Its Cellular Anti-Oxidant Activity. Cell Stress Chaperones 2016, 21, 773–781. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; Guo, H.; Huang, Y.A.N.; Wu, C.; Zhang, X. Eriodictyol, a Plant Flavonoid, Attenuates LPS induced Acute Lung Injury through Its Antioxidative and Anti inflammatory Activity. Exp. Ther. Med. 2015, 10, 2259–2266. [Google Scholar] [CrossRef]
- Shaw, C.; Chen, C.; Hsu, C.; Chen, C.; Tsai, Y. Antioxidant Properties of Scopoletin Isolated from Sinomonium Acutum. Phytother. Res. 2003, 17, 823–825. [Google Scholar] [CrossRef]
- Ding, Z.; Dai, Y.; Hao, H.; Pan, R.; Yao, X.; Wang, Z. Anti-Inflammatory Effects of Scopoletin and Underlying Mechanisms. Pharm. Biol. 2008, 46, 854–860. [Google Scholar] [CrossRef]
- Sancho, R.; Lucena, C.; Macho, A.; Calzado, M.A.; Blanco-Molina, M.; Minassi, A.; Appendino, G.; Muñoz, E. Immunosuppressive Activity of Capsaicinoids: Capsiate Derived from Sweet Peppers Inhibits NF-κB Activation and Is a Potent Antiinflammatory Compound in Vivo. Eur. J. Immunol. 2002, 32, 1753–1763. [Google Scholar] [CrossRef]
- Wang, Z.H.; Kang, K.A.; Zhang, R.; Piao, M.J.; Jo, S.H.; Kim, J.S.; Kang, S.S.; Lee, J.S.; Park, D.H.; Hyun, J.W. Myricetin Suppresses Oxidative Stress-Induced Cell Damage via Both Direct and Indirect Antioxidant Action. Env. Toxicol. Pharm. 2010, 29, 12–18. [Google Scholar] [CrossRef]
- Hou, W.; Hu, S.; Su, Z.; Wang, Q.; Meng, G.; Guo, T.; Zhang, J.; Gao, P. Myricetin Attenuates LPS-Induced Inflammation in RAW 264.7 Macrophages and Mouse Models. Future Med. Chem. 2018, 10, 2253–2264. [Google Scholar] [CrossRef]
- Han, D.H.; Lee, M.J.; Kim, J.H. Antioxidant and Apoptosis-Inducing Activities of Ellagic Acid. Anticancer. Res. 2006, 26, 3601–3606. [Google Scholar]
- Chao, P.; Hsu, C.; Yin, M. Anti-Inflammatory and Anti-Coagulatory Activities of Caffeic Acid and Ellagic Acid in Cardiac Tissue of Diabetic Mice. Nutr. Metab. 2009, 6, 1–8. [Google Scholar] [CrossRef]
- Guzmán-Beltrán, S.; Espada, S.; Orozco-Ibarra, M.; Pedraza-Chaverri, J.; Cuadrado, A. Nordihydroguaiaretic Acid Activates the Antioxidant Pathway Nrf2/HO-1 and Protects Cerebellar Granule Neurons against Oxidative Stress. Neurosci. Lett. 2008, 447, 167–171. [Google Scholar] [CrossRef]
- Li, Y.-J.; Kukita, A.; Watanabe, T.; Takano, T.; Qu, P.; Sanematsu, K.; Ninomiya, Y.; Kukita, T. Nordihydroguaiaretic Acid Inhibition of NFATc1 Suppresses Osteoclastogenesis and Arthritis Bone Destruction in Rats. Lab. Investig. 2012, 92, 1777–1787. [Google Scholar] [CrossRef]
- Gaspar, A.; Martins, M.; Silva, P.; Garrido, E.M.; Garrido, J.; Firuzi, O.; Miri, R.; Saso, L.; Borges, F. Dietary Phenolic Acids and Derivatives. Evaluation of the Antioxidant Activity of Sinapic Acid and Its Alkyl Esters. J. Agric. Food Chem. 2010, 58, 11273–11280. [Google Scholar] [CrossRef] [PubMed]
- Yun, K.-J.; Koh, D.-J.; Kim, S.-H.; Park, S.J.; Ryu, J.H.; Kim, D.-G.; Lee, J.-Y.; Lee, K.-T. Anti-Inflammatory Effects of Sinapic Acid through the Suppression of Inducible Nitric Oxide Synthase, Cyclooxygase-2, and Proinflammatory Cytokines Expressions via Nuclear Factor-ΚB Inactivation. J. Agric. Food Chem. 2008, 56, 10265–10272. [Google Scholar] [CrossRef] [PubMed]
- Soulage, C.; Soares, A.F.; Girotti, C.; Zarrouki, B.; Demarne, F.E.; Lagarde, M.; Géloën, A. Antioxidant Effect of Cirsimarin, a Flavonoid Extracted from Microtea Debilis. Phytopharm. Ther. Values II 2008, 20, 55–63. [Google Scholar]
- Zhao, H.; Jiang, Z.; Chang, X.; Xue, H.; Yahefu, W.; Zhang, X. 4-Hydroxyphenylacetic Acid Prevents Acute APAP-Induced Liver Injury by Increasing Phase II and Antioxidant Enzymes in Mice. Front. Pharmacol. 2018, 9, 653. [Google Scholar] [CrossRef]
- Qiao, S.; Li, W.; Tsubouchi, R.; Haneda, M.; Murakami, K.; Takeuchi, F.; Nisimoto, Y.; Yoshino, M. Rosmarinic Acid Inhibits the Formation of Reactive Oxygen and Nitrogen Species in RAW264. 7 Macrophages. Free Radic. Res. 2005, 39, 995–1003. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, X.; Yang, L.; Zu, Y.; Lu, Q. Effects of Rosmarinic Acid on Liver and Kidney Antioxidant Enzymes, Lipid Peroxidation and Tissue Ultrastructure in Aging Mice. Food Funct. 2015, 6, 927–931. [Google Scholar] [CrossRef]
- Li, H.; Xu, J.; Li, X.; Hu, Y.; Liao, Y.; Zhou, W.; Song, Z. Anti-Inflammatory Activity of Psoralen in Human Periodontal Ligament Cells via Estrogen Receptor Signaling Pathway. Sci. Rep. 2021, 11, 1–12. [Google Scholar] [CrossRef]
- Wang, C.; Al-Ani, M.K.; Sha, Y.; Chi, Q.; Dong, N.; Yang, L.; Xu, K. Psoralen Protects Chondrocytes, Exhibits Anti-Inflammatory Effects on Synoviocytes, and Attenuates Monosodium Iodoacetate-Induced Osteoarthritis. Int. J. Biol. Sci. 2019, 15, 229. [Google Scholar] [CrossRef]
- Wei, D.; Guo, X.; Lin, L.; Lin, M.; Gong, Y.; Ying, B.; Huang, M. Effects of Angelicin on Ovalbumin (OVA)-Induced Airway Inflammation in a Mouse Model of Asthma. Inflammation 2016, 39, 1876–1882. [Google Scholar] [CrossRef]
- Liu, F.; Sun, G.; Gao, H.; Li, R.; Soromou, L.-W.; Chen, N.; Deng, Y.-H.; Feng, H. Angelicin Regulates LPS-Induced Inflammation via Inhibiting MAPK/NF-ΚB Pathways. J. Surg. Res. 2013, 185, 300–309. [Google Scholar] [CrossRef]
- Cheng, C.-Y.; Ho, T.-Y.; Lee, E.-J.; Su, S.-Y.; Tang, N.-Y.; Hsieh, C.-L. Ferulic Acid Reduces Cerebral Infarct through Its Antioxidative and Anti-Inflammatory Effects Following Transient Focal Cerebral Ischemia in Rats. Am. J. Chin. Med. 2008, 36, 1105–1119. [Google Scholar] [CrossRef]
- Kang, J.-K.; Chung, Y.-C.; Hyun, C.-G. Anti-Inflammatory Effects of 6-Methylcoumarin in LPS-Stimulated RAW 264.7 Macrophages via Regulation of MAPK and NF-ΚB Signaling Pathways. Molecules 2021, 26, 5351. [Google Scholar] [CrossRef]
- Lee, H.-S.; Kim, E.-N.; Jeong, G.-S. Ameliorative Effect of Citropten Isolated from Citrus Aurantifolia Peel Extract as a Modulator of T Cell and Intestinal Epithelial Cell Activity in DSS-Induced Colitis. Molecules 2022, 27, 4633. [Google Scholar] [CrossRef]
- Xia, S.; Ni, Y.; Zhou, Q.; Liu, H.; Xiang, H.; Sui, H.; Shang, D. Emodin Attenuates Severe Acute Pancreatitis via Antioxidant and Anti-Inflammatory Activity. Inflammation 2019, 42, 2129–2138. [Google Scholar] [CrossRef]
- Ding, Y.; Zhao, L.; Mei, H.; Zhang, S.-L.; Huang, Z.-H.; Duan, Y.-Y.; Ye, P. Exploration of Emodin to Treat Alpha-Naphthylisothiocyanate-Induced Cholestatic Hepatitis via Anti-Inflammatory Pathway. Eur. J. Pharmacol. 2008, 590, 377–386. [Google Scholar] [CrossRef]
- Jiang, Y.; Gong, F.-L.; Zhao, G.-B.; Li, J. Chrysin Suppressed Inflammatory Responses and the Inducible Nitric Oxide Synthase Pathway after Spinal Cord Injury in Rats. Int. J. Mol. Sci. 2014, 15, 12270–12279. [Google Scholar] [CrossRef]
- Pushpavalli, G.; Kalaiarasi, P.; Veeramani, C.; Pugalendi, K.V. Effect of Chrysin on Hepatoprotective and Antioxidant Status in D-Galactosamine-Induced Hepatitis in Rats. Eur. J. Pharmacol. 2010, 631, 36–41. [Google Scholar] [CrossRef]
- Zhu, Z.; Hu, R.; Li, J.; Xing, X.; Chen, J.; Zhou, Q.; Sun, J. Alpinetin Exerts Anti-Inflammatory, Anti-Oxidative and Anti-Angiogenic Effects through Activating the Nrf2 Pathway and Inhibiting NLRP3 Pathway in Carbon Tetrachloride-Induced Liver Fibrosis. Int. Immunopharmacol. 2021, 96, 107660. [Google Scholar] [CrossRef]
- Gao, Y.; Wang, S.; He, L.; Wang, C.; Yang, L. Alpinetin Protects Chondrocytes and Exhibits Anti-Inflammatory Effects via the NF-ΚB/ERK Pathway for Alleviating Osteoarthritis. Inflammation 2020, 43, 1742–1750. [Google Scholar] [CrossRef]
- Tai, A.; Sawano, T.; Yazama, F.; Ito, H. Evaluation of Antioxidant Activity of Vanillin by Using Multiple Antioxidant Assays. Biochim. Biophys. Acta 2011, 1810, 170–177. [Google Scholar] [CrossRef]
- Makni, M.; Chtourou, Y.; Fetoui, H.; Garoui, E.M.; Boudawara, T.; Zeghal, N. Evaluation of the Antioxidant, Anti-Inflammatory and Hepatoprotective Properties of Vanillin in Carbon Tetrachloride-Treated Rats. Eur. J. Pharmacol. 2011, 668, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Zhang, X.; Zhu, G.; Liu, H.; Chen, J.; Wang, Y.; He, X. Quercetin Inhibits TNF-α Induced HUVECs Apoptosis and Inflammation via Downregulating NF-KB and AP-1 Signaling Pathway in Vitro. Medicine 2020, 99, e22241. [Google Scholar] [CrossRef] [PubMed]
- Abdelmoaty, M.A.; Ibrahim, M.A.; Ahmed, N.S.; Abdelaziz, M.A. Confirmatory Studies on the Antioxidant and Antidiabetic Effect of Quercetin in Rats. Indian J. Clin. Biochem. 2010, 25, 188–192. [Google Scholar] [CrossRef] [PubMed]
- Chekalina, N.; Burmak, Y.; Petrov, Y.; Borisova, Z.; Manusha, Y.; Kazakov, Y.; Kaidashev, I. Quercetin Reduces the Transcriptional Activity of NF-KB in Stable Coronary Artery Disease. Indian Heart J. 2018, 70, 593–597. [Google Scholar] [CrossRef]
- Wang, Z.; Sun, W.; Sun, X.; Wang, Y.; Zhou, M. Kaempferol Ameliorates Cisplatin Induced Nephrotoxicity by Modulating Oxidative Stress, Inflammation and Apoptosis via ERK and NF-ΚB Pathways. AMB Express 2020, 10, 1–11. [Google Scholar] [CrossRef]
- Martinez-Micaelo, N.; González-Abuín, N.; Pinent, M.; Ardévol, A.; Blay, M. Procyanidin B2 Inhibits Inflammasome-mediated IL-1β Production in Lipopolysaccharide-stimulated Macrophages. Mol. Nutr. Food Res. 2015, 59, 262–269. [Google Scholar] [CrossRef]
- Wang, W.; Chen, R.; Wang, J. Procyanidin B2 Ameliorates Carrageenan-Induced Chronic Nonbacterial Prostatitis in Rats via Anti-Inflammatory and Activation of the Nrf2 Pathway. Biochem. Biophys. Res. Commun. 2017, 493, 794–799. [Google Scholar] [CrossRef]
- Ramesh, B.; Pugalendi, K. v Antioxidant Role of Umbelliferone in STZ-Diabetic Rats. Life Sci. 2006, 79, 306–310. [Google Scholar] [CrossRef]
- Wu, G.; Nie, W.; Wang, Q.; Hao, Y.; Gong, S.; Zheng, Y.; Lv, H. Umbelliferone Ameliorates Complete Freund Adjuvant–Induced Arthritis via Reduction of NF-ΚB Signaling Pathway in Osteoclast Differentiation. Inflammation 2021, 44, 1315–1329. [Google Scholar] [CrossRef]
- Li, T.; Li, F.; Liu, X.; Liu, J.; Li, D. Synergistic Anti-inflammatory Effects of Quercetin and Catechin via Inhibiting Activation of TLR4–MyD88-mediated NF-κB and MAPK Signaling Pathways. Phytother. Res. 2019, 33, 756–767. [Google Scholar] [CrossRef]
- Samarghandian, S.; Azimi-Nezhad, M.; Farkhondeh, T. Catechin Treatment Ameliorates Diabetes and Its Complications in Streptozotocin-Induced Diabetic Rats. Dose Response 2017, 15, 1559325817691158. [Google Scholar] [CrossRef]
- Pan, R.; Dai, Y.; Gao, X.; Xia, Y. Scopolin Isolated from Erycibe Obtusifolia Benth Stems Suppresses Adjuvant-Induced Rat Arthritis by Inhibiting Inflammation and Angiogenesis. Int. Immunopharmacol. 2009, 9, 859–869. [Google Scholar] [CrossRef]
- García-Mediavilla, V.; Crespo, I.; Collado, P.S.; Esteller, A.; Sánchez-Campos, S.; Tuñón, M.J.; González-Gallego, J. The Anti-Inflammatory Flavones Quercetin and Kaempferol Cause Inhibition of Inducible Nitric Oxide Synthase, Cyclooxygenase-2 and Reactive C-Protein, and down-Regulation of the Nuclear Factor KappaB Pathway in Chang Liver Cells. Eur. J. Pharmacol. 2007, 557, 221–229. [Google Scholar] [CrossRef]
- Liao, W.; Chen, L.; Ma, X.; Jiao, R.; Li, X.; Wang, Y. Protective Effects of Kaempferol against Reactive Oxygen Species-Induced Hemolysis and Its Antiproliferative Activity on Human Cancer Cells. Eur. J. Med. Chem. 2016, 114, 24–32. [Google Scholar] [CrossRef]
- Omar, H.A.; Mohamed, W.R.; Arafa, E.-S.A.; Shehata, B.A.; el Sherbiny, G.A.; Arab, H.H.; Elgendy, A.N.A.M. Hesperidin Alleviates Cisplatin-Induced Hepatotoxicity in Rats without Inhibiting Its Antitumor Activity. Pharmacol. Rep. 2016, 68, 349–356. [Google Scholar] [CrossRef]
- Wilmsen, P.K.; Spada, D.S.; Salvador, M. Antioxidant Activity of the Flavonoid Hesperidin in Chemical and Biological Systems. J. Agric. Food Chem. 2005, 53, 4757–4761. [Google Scholar] [CrossRef]
- Wang, J.; Liu, Y.-T.; Xiao, L.; Zhu, L.; Wang, Q.; Yan, T. Anti-Inflammatory Effects of Apigenin in Lipopolysaccharide-Induced Inflammatory in Acute Lung Injury by Suppressing COX-2 and NF-KB Pathway. Inflammation 2014, 37, 2085–2090. [Google Scholar] [CrossRef]
- Singh, J.P.V.; Selvendiran, K.; Banu, S.M.; Padmavathi, R.; Sakthisekaran, D. Protective Role of Apigenin on the Status of Lipid Peroxidation and Antioxidant Defense against Hepatocarcinogenesis in Wistar Albino Rats. Phytomedicine 2004, 11, 309–314. [Google Scholar] [CrossRef]
- Calixto-Campos, C.; Carvalho, T.T.; Hohmann, M.S.N.; Pinho-Ribeiro, F.A.; Fattori, V.; Manchope, M.F.; Zarpelon, A.C.; Baracat, M.M.; Georgetti, S.R.; Casagrande, R. Vanillic Acid Inhibits Inflammatory Pain by Inhibiting Neutrophil Recruitment, Oxidative Stress, Cytokine Production, and NFκB Activation in Mice. J. Nat. Prod. 2015, 78, 1799–1808. [Google Scholar] [CrossRef]
- Prince, P.S.M.; Rajakumar, S.; Dhanasekar, K. Protective Effects of Vanillic Acid on Electrocardiogram, Lipid Peroxidation, Antioxidants, Proinflammatory Markers and Histopathology in Isoproterenol Induced Cardiotoxic Rats. Eur. J. Pharmacol. 2011, 668, 233–240. [Google Scholar] [CrossRef]
- Singh, B.; Singh, J.P.; Singh, N.; Kaur, A. Saponins in Pulses and Their Health Promoting Activities: A Review. Food Chem. 2017, 233, 540–549. [Google Scholar] [CrossRef]
- Faizal, A.; Geelen, D. Saponins and Their Role in Biological Processes in Plants. Phytochem. Rev. 2013, 12, 877–893. [Google Scholar] [CrossRef]
- Passos, F.R.S.; Araújo-Filho, H.G.; Monteiro, B.S.; Shanmugam, S.; de Souza Araújo, A.A.; da Silva Almeida, J.R.G.; Thangaraj, P.; Júnior, L.J.Q.; Quintans, J. de S.S. Anti-Inflammatory and Modulatory Effects of Steroidal Saponins and Sapogenins on Cytokines: A Review of Pre-Clinical Research. Phytomedicine 2021, 96, 153842. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Zhong, S.; Li, T.; Zhang, J. Saponins as Modulators of Nuclear Receptors. Crit. Rev. Food Sci. Nutr. 2020, 60, 94–107. [Google Scholar] [CrossRef] [PubMed]
- Tenon, M.; Feuillère, N.; Roller, M.; Birtić, S. Rapid, Cost-Effective and Accurate Quantification of Yucca Schidigera Roezl. Steroidal Saponins Using HPLC-ELSD Method. Food Chem. 2017, 221, 1245–1252. [Google Scholar] [CrossRef]
- Qu, L.; Wang, J.; Ruan, J.; Yao, X.; Huang, P.; Wang, Y.; Yu, H.; Han, L.; Zhang, Y.; Wang, T. Spirostane-Type Saponins Obtained from Yucca Schidigera. Molecules 2018, 23, 167. [Google Scholar] [CrossRef]
- Tang, Z.-Z.; Zhang, Y.-M.; Zheng, T.; Huang, T.-T.; Ma, T.-F.; Liu, Y.-W. Sarsasapogenin Alleviates Diabetic Nephropathy through Suppression of Chronic Inflammation by Down-Regulating PAR-1: In Vivo and in Vitro Study. Phytomedicine 2020, 78, 153314. [Google Scholar] [CrossRef]
- Ingawale, D.K.; Mandlik, S.K.; Patel, S.S. Combination of Sarsasapogenin and Fluticasone Attenuates Ovalbumin-Induced Airway Inflammation in a Mouse Asthma Model. Immunopharmacol. Immunotoxicol. 2020, 42, 128–137. [Google Scholar] [CrossRef]
- Pei, R.; Duan, C.; Vijayalakshmi, A. Hecogenin Attenuates Isoproterenol-Induced Myocardial Infarction through Nuclear Factor-Kappa B-Mediated Signaling Pathway in Rats. Pharmacogn. Mag. 2022, 18, 463. [Google Scholar]
- Ingawale, D.K.; Mandlik, S.K.; Patel, S.S. Hecogenin and Fluticasone Combination Attenuates TNBS-Induced Ulcerative Colitis in Rats via Downregulation of pro-Inflammatory Mediators and Oxidative Stress. Immunopharmacol. Immunotoxicol. 2021, 43, 160–170. [Google Scholar] [CrossRef]
- Ingawale, D.K.; Patel, S.S. Hecogenin Exhibits Anti-Arthritic Activity in Rats through Suppression of pro-Inflammatory Cytokines in Complete Freund’s Adjuvant-Induced Arthritis. Immunopharmacol. Immunotoxicol. 2018, 40, 59–71. [Google Scholar] [CrossRef]
- Chen, C.-T.; Wang, Z.-H.; Hsu, C.-C.; Lin, H.-H.; Chen, J.-H. In Vivo Protective Effects of Diosgenin against Doxorubicin-Induced Cardiotoxicity. Nutrients 2015, 7, 4938–4954. [Google Scholar] [CrossRef]
- Deng, X.-Y.; Chen, J.-J.; Li, H.-Y.; Ma, Z.-Q.; Ma, S.-P.; Fu, Q. Cardioprotective Effects of Timosaponin B II from Anemarrhenae Asphodeloides Bge on Isoproterenol-Induced Myocardial Infarction in Rats. Chem. Biol. Interact. 2015, 240, 22–28. [Google Scholar] [CrossRef]
- Jiménez, G.G.; Durán, A.G.; Macías, F.A.; Simonet, A.M. Structure, Bioactivity and Analytical Methods for the Determination of Yucca Saponins. Molecules 2021, 26, 5251. [Google Scholar] [CrossRef]
- Wang, N.; Xu, P.; Wu, R.; Wang, X.; Wang, Y.; Shou, D.; Zhang, Y. Timosaponin BII Improved Osteoporosis Caused by Hyperglycemia through Promoting Autophagy of Osteoblasts via Suppressing the MTOR/NFκB Signaling Pathway. Free Radic. Biol. Med. 2021, 171, 112–123. [Google Scholar] [CrossRef]
- Lim, S.-M.; Jeong, J.-J.; Kang, G.-D.; Kim, K.-A.; Choi, H.-S.; Kim, D.-H. Timosaponin AIII and Its Metabolite Sarsasapogenin Ameliorate Colitis in Mice by Inhibiting NF-ΚB and MAPK Activation and Restoring Th17/Treg Cell Balance. Int. Immunopharmacol. 2015, 25, 493–503. [Google Scholar] [CrossRef]
- Tuan Anh, H.L.; Tran, P.T.; Thao, D.T.; Trang, D.T.; Dang, N.H.; van Cuong, P.; van Kiem, P.; van Minh, C.; Lee, J.-H. Degalactotigonin, a Steroidal Glycoside from Solanum Nigrum, Induces Apoptosis and Cell Cycle Arrest via Inhibiting the EGFR Signaling Pathways in Pancreatic Cancer Cells. Biomed. Res. Int. 2018, 2018, 3120972. [Google Scholar] [CrossRef]
- Li, J.; Zhao, C.; Zhu, Q.; Wang, Y.; Li, G.; Li, X.; Li, Y.; Wu, N.; Ma, C. Sweroside Protects Against Myocardial Ischemia–Reperfusion Injury by Inhibiting Oxidative Stress and Pyroptosis Partially via Modulation of the Keap1/Nrf2 Axis. Front. Cardiovasc. Med. 2021, 8, 650368. [Google Scholar] [CrossRef]
- Wang, R.; Dong, Z.; Lan, X.; Liao, Z.; Chen, M. Sweroside Alleviated LPS-Induced Inflammation via SIRT1 Mediating NF-ΚB and FOXO1 Signaling Pathways in RAW264. 7 Cells. Molecules 2019, 24, 872. [Google Scholar] [CrossRef]
- Bartnik, M.; Facey, P.C. Glycosides. In Pharmacognosy; Elsevier: Amsterdam, The Netherlands, 2017; pp. 101–161. [Google Scholar]
- Kytidou, K.; Artola, M.; Overkleeft, H.S.; Aerts, J.M.F.G. Plant Glycosides and Glycosidases: A Treasure-Trove for Therapeutics. Front. Plant. Sci. 2020, 11, 357. [Google Scholar] [CrossRef]
- Guardia, T.; Rotelli, A.E.; Juarez, A.O.; Pelzer, L.E. Anti-Inflammatory Properties of Plant Flavonoids. Effects of Rutin, Quercetin and Hesperidin on Adjuvant Arthritis in Rat. Il Farm. 2001, 56, 683–687. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Liu, H.; Yang, J.; Gupta, V.K.; Jiang, Y. New Insights on Bioactivities and Biosynthesis of Flavonoid Glycosides. Trends Food Sci. Technol. 2018, 79, 116–124. [Google Scholar] [CrossRef]
- Ying, X.; Li, H.; Xiong, Z.; Sun, Z.; Cai, S.; Zhu, W.; Bi, Y.; Li, F. LC Determination of Malondialdehyde Concentrations in the Human Umbilical Vein Endothelial Cell Culture Medium: Application to the Antioxidant Effect of Vitexin-2 ″-O-Rhamnoside. Chromatographia 2008, 67, 679–686. [Google Scholar] [CrossRef]
- Ullah, H.; Khan, A.; Baig, M.W.; Ullah, N.; Ahmed, N.; Tipu, M.K.; Ali, H.; Khan, S. Poncirin Attenuates CCL4-Induced Liver Injury through Inhibition of Oxidative Stress and Inflammatory Cytokines in Mice. BMC Complement Med. 2020, 20, 1–14. [Google Scholar] [CrossRef]
- Ullah, H.; Khan, A.; Bibi, T.; Ahmad, S.; Shehzad, O.; Ali, H.; Seo, E.K.; Khan, S. Comprehensive in Vivo and in Silico Approaches to Explore the Hepatoprotective Activity of Poncirin against Paracetamol Toxicity. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2022, 395, 195–215. [Google Scholar] [CrossRef] [PubMed]
- Domitrović, R.; Rashed, K.; Cvijanović, O.; Vladimir-Knežević, S.; Škoda, M.; Višnić, A. Myricitrin Exhibits Antioxidant, Anti-Inflammatory and Antifibrotic Activity in Carbon Tetrachloride-Intoxicated Mice. Chem. Biol. Interact. 2015, 230, 21–29. [Google Scholar] [CrossRef]
- Chen, W.; Feng, L.; Shen, Y.; Su, H.; Li, Y.; Zhuang, J.; Zhang, L.; Zheng, X. Myricitrin Inhibits Acrylamide-Mediated Cytotoxicity in Human Caco-2 Cells by Preventing Oxidative Stress. Biomed. Res. Int. 2013, 2013, 724183. [Google Scholar] [CrossRef]
- De Sousa, E.; Zanatta, L.; Seifriz, I.; Creczynski-Pasa, T.B.; Pizzolatti, M.G.; Szpoganicz, B.; Silva, F.R.M.B. Hypoglycemic Effect and Antioxidant Potential of Kaempferol-3, 7-O-(α)-Dirhamnoside from Bauhinia f Orficata Leaves. J. Nat. Prod. 2004, 67, 829–832. [Google Scholar] [CrossRef]
- Kamdi, S.P.; Raval, A.; Nakhate, K.T. Phloridzin Attenuates Lipopolysaccharide-Induced Cognitive Impairment via Antioxidant, Anti-Inflammatory and Neuromodulatory Activities. Cytokine 2021, 139, 155408. [Google Scholar] [CrossRef]
- Taiwo, F.O.; Oyedeji, O.; Osundahunsi, M.T. Antimicrobial and Antioxidant Properties of Kaempferol-3-O-Glucoside and 1-(4-Hydroxyphenyl)-3-Phenylpropan-1-One Isolated from the Leaves of Annona Muricata (Linn.). J. Pharm. Res. Int. 2019, 26, 1–13. [Google Scholar] [CrossRef]
- Babujanarthanam, R.; Kavitha, P.; Mahadeva Rao, U.S.; Pandian, M.R. Quercitrin a Bioflavonoid Improves the Antioxidant Status in Streptozotocin: Induced Diabetic Rat Tissues. Mol. Cell. Biochem. 2011, 358, 121–129. [Google Scholar] [CrossRef]
- Camuesco, D.; Comalada, M.; Rodríguez-Cabezas, M.E.; Nieto, A.; Lorente, M.D.; Concha, A.; Zarzuelo, A.; Gálvez, J. The Intestinal Anti-inflammatory Effect of Quercitrin Is Associated with an Inhibition in INOS Expression. Br. J. Pharmacol. 2004, 143, 908–918. [Google Scholar] [CrossRef]
- Comalada, M.; Camuesco, D.; Sierra, S.; Ballester, I.; Xaus, J.; Gálvez, J.; Zarzuelo, A. In Vivo Quercitrin Anti-inflammatory Effect Involves Release of Quercetin, Which Inhibits Inflammation through Down-regulation of the NF-κB Pathway. Eur. J. Immunol. 2005, 35, 584–592. [Google Scholar] [CrossRef]
- Mittal, R.; Kumar, A.; Singh, D.P.; Bishnoi, M.; Nag, T.C. Ameliorative Potential of Rutin in Combination with Nimesulide in STZ Model of Diabetic Neuropathy: Targeting Nrf2/HO-1/NF-KB and COX Signalling Pathway. Inflammopharmacology 2018, 26, 755–768. [Google Scholar] [CrossRef]
- Yang, J.; Guo, J.; Yuan, J. In Vitro Antioxidant Properties of Rutin. LWT Food Sci. Technol. 2008, 41, 1060–1066. [Google Scholar] [CrossRef]
- Dou, W.; Zhang, J.; Sun, A.; Zhang, E.; Ding, L.; Mukherjee, S.; Wei, X.; Chou, G.; Wang, Z.-T.; Mani, S. Protective Effect of Naringenin against Experimental Colitis via Suppression of Toll-like Receptor 4/NF-ΚB Signalling. Br. J. Nutr. 2013, 110, 599–608. [Google Scholar] [CrossRef] [PubMed]
- Jung, U.J.; Kim, H.J.; Lee, J.S.; Lee, M.K.; Kim, H.O.; Park, E.J.; Kim, H.K.; Jeong, T.S.; Choi, M.S. Naringin Supplementation Lowers Plasma Lipids and Enhances Erythrocyte Antioxidant Enzyme Activities in Hypercholesterolemic Subjects. Clin. Nutr. 2003, 22, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-J.; Um, J.-Y.; Hong, S.-H.; Lee, J.-Y. Anti-Inflammatory Activity of Hyperoside through the Suppression of Nuclear Factor-ΚB Activation in Mouse Peritoneal Macrophages. Am. J. Chin. Med. 2011, 39, 171–181. [Google Scholar] [CrossRef]
- Ku, S.-K.; Zhou, W.; Lee, W.; Han, M.-S.; Na, M.; Bae, J.-S. Anti-Inflammatory Effects of Hyperoside in Human Endothelial Cells and in Mice. Inflammation 2015, 38, 784–799. [Google Scholar] [CrossRef]
- Piao, M.J.; Kang, K.A.; Zhang, R.; Ko, D.O.; Wang, Z.H.; You, H.J.; Kim, H.S.; Kim, J.S.; Kang, S.S.; Hyun, J.W. Hyperoside Prevents Oxidative Damage Induced by Hydrogen Peroxide in Lung Fibroblast Cells via an Antioxidant Effect. Biochim. Biophys. Acta 2008, 1780, 1448–1457. [Google Scholar] [CrossRef]
- Song, Y.; Wang, X.; Qin, S.; Zhou, S.; Li, J.; Gao, Y. Esculin Ameliorates Cognitive Impairment in Experimental Diabetic Nephropathy and Induces Anti-Oxidative Stress and Anti-Inflammatory Effects via the MAPK Pathway. Mol. Med. Rep. 2018, 17, 7395–7402. [Google Scholar] [CrossRef]
- Rios, E.R.V.; Rocha, N.F.M.; Venâncio, E.T.; Moura, B.A.; Feitosa, M.L.; Cerqueira, G.S.; Soares, P.M.G.; Woods, D.J.; de Sousa, F.C.F.; Leal, L.K.A.M. Mechanisms Involved in the Gastroprotective Activity of Esculin on Acute Gastric Lesions in Mice. Chem. Biol. Interact. 2010, 188, 246–254. [Google Scholar] [CrossRef]
- Chung, M.J.; Pandey, R.P.; Choi, J.W.; Sohng, J.K.; Choi, D.J.; Park, Y. il Inhibitory Effects of Kaempferol-3-O-Rhamnoside on Ovalbumin-Induced Lung Inflammation in a Mouse Model of Allergic Asthma. Int. Immunopharmacol. 2015, 25, 302–310. [Google Scholar] [CrossRef]
- Lund, J.; Rustan, A.C. Fatty Acids: Structures and Properties. In eLS; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2022. [Google Scholar]
- Weber, H. Fatty Acid-Derived Signals in Plants. Trends Plant Sci. 2002, 7, 217–224. [Google Scholar] [CrossRef]
- He, M.; Ding, N.-Z. Plant Unsaturated Fatty Acids: Multiple Roles in Stress Response. Front. Plant Sci. 2020, 11, 562785. [Google Scholar] [CrossRef]
- Munsterman, A.S.; Bertone, A.L.; Zachos, T.A.; Weisbrode, S.E. Effects of the Omega-3 Fatty Acid, α-Linolenic Acid, on Lipopolysaccharide-Challenged Synovial Explants from Horses. Am. J. Vet. Res. 2005, 66, 1503–1508. [Google Scholar] [CrossRef]
- Youn, K.; Lee, S.; Jun, M. Gamma-Linolenic Acid Ameliorates Aβ-Induced Neuroinflammation through NF-ΚB and MAPK Signalling Pathways. J. Funct. Foods 2018, 42, 30–37. [Google Scholar] [CrossRef]
- Huang, W.-C.; Tsai, T.-H.; Chuang, L.-T.; Li, Y.-Y.; Zouboulis, C.C.; Tsai, P.-J. Anti-Bacterial and Anti-Inflammatory Properties of Capric Acid against Propionibacterium Acnes: A Comparative Study with Lauric. Acid. J. Dermatol. Sci. 2014, 73, 232–240. [Google Scholar] [CrossRef]
- Henry, G.E.; Momin, R.A.; Nair, M.G.; Dewitt, D.L. Antioxidant and Cyclooxygenase Activities of Fatty Acids Found in Food. J. Agric. Food Chem. 2002, 50, 2231–2234. [Google Scholar] [CrossRef] [PubMed]
- Venn-Watson, S.; Lumpkin, R.; Dennis, E.A. Efficacy of Dietary Odd-Chain Saturated Fatty Acid Pentadecanoic Acid Parallels Broad Associated Health Benefits in Humans: Could It Be Essential? Sci. Rep. 2020, 10, 1–14. [Google Scholar] [CrossRef]
- Saiki, P.; Kawano, Y.; van Griensven, L.J.L.D.; Miyazaki, K. The Anti-Inflammatory Effect of Agaricus Brasiliensis Is Partly Due to Its Linoleic Acid Content. Food Funct. 2017, 8, 4150–4158. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Liang, C.; Li, G.; Yu, C.; Yin, M. Stearic Acid Protects Primary Cultured Cortical Neurons against Oxidative Stress. Acta Pharmacol. Sin. 2007, 28, 315–326. [Google Scholar] [CrossRef] [PubMed]
- Pan, P.-H.; Lin, S.-Y.; Ou, Y.-C.; Chen, W.-Y.; Chuang, Y.-H.; Yen, Y.-J.; Liao, S.-L.; Raung, S.-L.; Chen, C.-J. Stearic Acid Attenuates Cholestasis-Induced Liver Injury. Biochem. Biophys. Res. Commun. 2010, 391, 1537–1542. [Google Scholar] [CrossRef]
- Bhattacharjee, B.; Pal, P.K.; Chattopadhyay, A.; Bandyopadhyay, D. Oleic Acid Protects against Cadmium Induced Cardiac and Hepatic Tissue Injury in Male Wistar Rats: A Mechanistic Study. Life Sci. 2020, 244, 117324. [Google Scholar] [CrossRef]
- Zhao, Y.; Joshi-Barve, S.; Barve, S.; Chen, L.H. Eicosapentaenoic Acid Prevents LPS-Induced TNF-α Expression by Preventing NF-ΚB Activation. J. Am. Coll. Nutr. 2004, 23, 71–78. [Google Scholar] [CrossRef]
- Mathur, A.; Verma, S.K.; Singh, S.K.; Prasad, G.; Dua, V.K. Investigation of the Antimicrobial, Antioxidant and Anti-Inflammatory Activity of Compound Isolated from Murraya Koenigii. Int. J. Appl. Biol. Pharm. 2011, 2, 470–477. [Google Scholar]
- Oldfield, E.; Lin, F. Terpene Biosynthesis: Modularity Rules. Angew. Chem. Int. Ed. 2012, 51, 1124–1137. [Google Scholar] [CrossRef]
- Munné-Bosch, S.; Falk, J. New Insights into the Function of Tocopherols in Plants. Planta 2004, 218, 323–326. [Google Scholar] [CrossRef]
- Niki, E.; Traber, M.G. A History of Vitamin, E. Ann. Nutr. Metab. 2012, 61, 207–212. [Google Scholar] [CrossRef]
- Gabay, O.; Sanchez, C.; Salvat, C.; Chevy, F.; Breton, M.; Nourissat, G.; Wolf, C.; Jacques, C.; Berenbaum, F. Stigmasterol: A Phytosterol with Potential Anti-Osteoarthritic Properties. Osteoarthr. Cartil. 2010, 18, 106–116. [Google Scholar] [CrossRef]
- Khan, M.A.; Sarwar, A.H.M.G.; Rahat, R.; Ahmed, R.S.; Umar, S. Stigmasterol Protects Rats from Collagen Induced Arthritis by Inhibiting Proinflammatory Cytokines. Int. Immunopharmacol. 2020, 85, 106642. [Google Scholar] [CrossRef] [PubMed]
- Liang, Q.; Yang, J.; He, J.; Chen, X.; Zhang, H.; Jia, M.; Liu, K.; Jia, C.; Pan, Y.; Wei, J. Stigmasterol Alleviates Cerebral Ischemia/Reperfusion Injury by Attenuating Inflammation and Improving Antioxidant Defenses in Rats. Biosci. Rep. 2020, 40, BSR20192133. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, A.M.S.; Heimfarth, L.; Pereira, E.W.M.; Oliveira, F.S.; Menezes, I.R.A.; Coutinho, H.D.M.; Picot, L.; Antoniolli, A.R.; Quintans, J.S.S.; Quintans-Júnior, L.J. Phytol, a Chlorophyll Component, Produces Antihyperalgesic, Anti-Inflammatory, and Antiarthritic Effects: Possible NFκB Pathway Involvement and Reduced Levels of the Proinflammatory Cytokines TNF-α and IL. J. Nat. Prod. 2020, 83, 1107–1117. [Google Scholar] [CrossRef]
- Islam, M.T.; Ayatollahi, S.A.; Zihad, S.M.N.K.; Sifat, N.; Khan, M.R.; Paul, A.; Salehi, B.; Islam, T.; Mubarak, M.S.; Martins, N. Phytol Anti-Inflammatory Activity: Pre-Clinical Assessment and Possible Mechanism of Action Elucidation. Cell. Mol. Biol. 2020, 66, 264–269. [Google Scholar] [CrossRef]
- Santos, C.C.D.M.P.; Salvadori, M.S.; Mota, V.G.; Costa, L.M.; de Almeida, A.A.C.; de Oliveira, G.A.L.; Costa, J.P.; de Sousa, D.P.; de Freitas, R.M.; de Almeida, R.N. Antinociceptive and Antioxidant Activities of Phytol in Vivo and in Vitro Models. Neurosci. J. 2013, 2013, 949452. [Google Scholar] [CrossRef]
- Tang, X.; Liu, J.; Dong, W.; Li, P.; Li, L.; Lin, C.; Zheng, Y.; Hou, J.; Li, D. The Cardioprotective Effects of Citric Acid and L-Malic Acid on Myocardial Ischemia/Reperfusion Injury. Evid. Based Complement. Altern. Med. 2013, 2013, 820695. [Google Scholar] [CrossRef]
- Yang, X.; Kang, M.-C.; Lee, K.-W.; Kang, S.-M.; Lee, W.-W.; Jeon, Y.-J. Antioxidant Activity and Cell Protective Effect of Loliolide Isolated from Sargassum Ringgoldianum Subsp. Coreanum. Algae 2011, 26, 201–208. [Google Scholar] [CrossRef]
- Silva, J.; Alves, C.; Martins, A.; Susano, P.; Simões, M.; Guedes, M.; Rehfeldt, S.; Pinteus, S.; Gaspar, H.; Rodrigues, A. Loliolide, a New Therapeutic Option for Neurological Diseases? In Vitro Neuroprotective and Anti-Inflammatory Activities of a Monoterpenoid Lactone Isolated from Codium Tomentosum. Int. J. Mol. Sci. 2021, 22, 1888. [Google Scholar] [CrossRef]
- Jayawardena, T.U.; Kim, H.-S.; Sanjeewa, K.K.A.; Han, E.J.; Jee, Y.; Ahn, G.; Rho, J.-R.; Jeon, Y.-J. Loliolide, Isolated from Sargassum Horneri; Abate LPS-Induced Inflammation via TLR Mediated NF-ΚB, MAPK Pathways in Macrophages. Algal Res. 2021, 56, 102297. [Google Scholar] [CrossRef]
- Banudevi, S.; Krishnamoorthy, G.; Venkataraman, P.; Vignesh, C.; Aruldhas, M.M.; Arunakaran, J. Role of α-Tocopherol on Antioxidant Status in Liver, Lung and Kidney of PCB Exposed Male Albino Rats. Food Chem. Toxicol. 2006, 44, 2040–2046. [Google Scholar] [CrossRef]
- Sharma, R.; Vinayak, M. α-Tocopherol Attenuates NF-ΚB Activation and pro-Inflammatory Cytokine IL-6 Secretion in Cancer-Bearing Mice. Biosci. Rep. 2011, 31, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Park, N.-Y.; Jang, Y.; Ma, A.; Jiang, Q. Vitamin E γ-Tocotrienol Inhibits Cytokine-Stimulated NF-ΚB Activation by Induction of Anti-Inflammatory A20 via Stress Adaptive Response Due to Modulation of Sphingolipids. J. Immunol. 2015, 195, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, T.; Shoji, A.; Gu, N.; Joo, E.; Li, S.; Adachi, T.; Yamazaki, H.; Yasuda, K.; Kondoh, T.; Tsuda, K. γ-Tocotrienol Attenuates TNF-α-Induced Changes in Secretion and Gene Expression of MCP-1, IL-6 and Adiponectin in 3T3-L1 Adipocytes. Mol. Med. Rep. 2012, 5, 905–909. [Google Scholar] [CrossRef] [PubMed]
- Radhakrishnan, A.; Tudawe, D.; Chakravarthi, S.; Chiew, G.S.; Haleagrahara, N. Effect of Γ tocotrienol in Counteracting Oxidative Stress and Joint Damage in Collagen induced Arthritis in Rats. Exp. Ther. Med. 2014, 7, 1408–1414. [Google Scholar] [CrossRef]
- Qureshi, A.A.; Tan, X.; Reis, J.C.; Badr, M.Z.; Papasian, C.J.; Morrison, D.C.; Qureshi, N. Inhibition of Nitric Oxide in LPS-Stimulated Macrophages of Young and Senescent Mice by δ-Tocotrienol and Quercetin. Lipids Health Dis. 2011, 10, 1–22. [Google Scholar] [CrossRef]
- Zarubina, I.V.; Lukk, M.V.; Shabanov, P.D. Antihypoxic and Antioxidant Effects of Exogenous Succinic Acid and Aminothiol Succinate-Containing Antihypoxants. Bull. Exp. Biol. Med. 2012, 153, 336–339. [Google Scholar] [CrossRef]
- Bhardwaj, M.; Sali, V.K.; Mani, S.; Vasanthi, H.R. Neophytadiene from Turbinaria Ornata Suppresses LPS-Induced Inflammatory Response in RAW 264.7 Macrophages and Sprague Dawley Rats. Inflammation 2020, 43, 937–950. [Google Scholar] [CrossRef]
- Prabhu, S.; Jainu, M.; Sabitha, K.E.; Devi, C.S.S. Role of Mangiferin on Biochemical Alterations and Antioxidant Status in Isoproterenol-Induced Myocardial Infarction in Rats. J. Ethnopharmacol. 2006, 107, 126–133. [Google Scholar] [CrossRef]
- He, L.; Peng, X.; Zhu, J.; Chen, X.; Liu, H.; Tang, C.; Dong, Z.; Liu, F.; Peng, Y. Mangiferin Attenuate Sepsis-Induced Acute Kidney Injury via Antioxidant and Anti-Inflammatory Effects. Am. J. Nephrol. 2014, 40, 441–450. [Google Scholar] [CrossRef]
- Gong, X.; Zhang, L.; Jiang, R.; Ye, M.; Yin, X.; Wan, J. Anti-Inflammatory Effects of Mangiferin on Sepsis-Induced Lung Injury in Mice via up-Regulation of Heme Oxygenase. J. Nutr. Biochem. 2013, 24, 1173–1181. [Google Scholar] [CrossRef]
- Gowder, S.J.T.; Devaraj, H. Effect of the Food Flavour Cinnamaldehyde on the Antioxidant Status of Rat Kidney. Basic Clin. Pharm. Toxicol. 2006, 99, 379–382. [Google Scholar] [CrossRef]
- Mateen, S.; Rehman, M.T.; Shahzad, S.; Naeem, S.S.; Faizy, A.F.; Khan, A.Q.; Khan, M.S.; Husain, F.M.; Moin, S. Anti-Oxidant and Anti-Inflammatory Effects of Cinnamaldehyde and Eugenol on Mononuclear Cells of Rheumatoid Arthritis Patients. Eur. J. Pharmacol. 2019, 852, 14–24. [Google Scholar] [CrossRef]
- Ni, Y.-L.; Shen, H.-T.; Su, C.-H.; Chen, W.-Y.; Huang-Liu, R.; Chen, C.-J.; Chen, S.-P.; Kuan, Y.-H. Nerolidol Suppresses the Inflammatory Response during Lipopolysaccharide-Induced Acute Lung Injury via the Modulation of Antioxidant Enzymes and the AMPK/Nrf-2/HO-1 Pathway. Oxidative Med. Cell. Longev. 2019, 2019, 9605980. [Google Scholar] [CrossRef]
- Wen, Q.; Lau, N.; Weng, H.; Ye, P.; Du, S.; Li, C.; Lv, J.; Li, H. Chrysophanol Exerts Anti-Inflammatory Activity by Targeting Histone Deacetylase 3 through the High Mobility Group Protein 1-Nuclear Transcription Factor-Kappa B Signaling Pathway in Vivo and in Vitro. Front. Bioeng. Biotechnol. 2021, 8, 623866. [Google Scholar] [CrossRef]
- Lian, Y.; Xia, X.; Zhao, H.; Zhu, Y. The Potential of Chrysophanol in Protecting against High Fat-Induced Cardiac Injury through Nrf2-Regulated Anti-Inflammation, Anti-Oxidant and Anti-Fibrosis in Nrf2 Knockout Mice. Biomed. Pharmacother. 2017, 93, 1175–1189. [Google Scholar] [CrossRef]
- Sivakumar, D.; Sultanbawa, Y.; Cooperstone, J.L.; Ziv, C. Editorial: Phytochemical changes in vegetables during post-harvest storage and processing, and implications for consumer benefits. Front. Nutr. 2022, 9, 1025361. [Google Scholar] [CrossRef]
- Xu, D.P.; Li, Y.; Meng, X.; Zhou, T.; Zhou, Y.; Zheng, J.; Li, H.B. Natural antioxidants in foods and medicinal plants: Extraction, assessment and resources. Int. J. Mol. Sci. 2017, 18, 96. [Google Scholar] [CrossRef]
- Dey, P. Gut microbiota in phytopharmacology: A comprehensive overview of concepts, reciprocal interactions, biotransformations and mode of actions. Pharmacol. Res. 2019, 147, 104367. [Google Scholar] [CrossRef]
- Chen, F.; Wen, Q.; Jiang, J.; Li, H.L.; Tan, Y.F.; Li, Y.H.; Zeng, N.K. Could the gut microbiota reconcile the oral bioavailability conundrum of traditional herbs? J. Ethnopharmacol. 2016, 179, 253–264. [Google Scholar] [CrossRef]
- Saeedi, B.J.; Liu, K.H.; Owens, J.A.; Hunter-Chang, S.; Camacho, M.C.; Eboka, R.U.; Neish, A.S. Gut-resident lactobacilli activate hepatic Nrf2 and protect against oxidative liver injury. Cell Metab. 2020, 31, 956–968. [Google Scholar] [CrossRef]
- Aboulgheit, A.; Karbasiafshar, C.; Zhang, Z.; Sabra, M.; Shi, G.; Tucker, A.; Sellke, F.W. Lactobacillus plantarum probiotic induces Nrf2-mediated antioxidant signaling and eNOS expression resulting in improvement of myocardial diastolic function. Am. J. Physiol. Heart Circ. Physiol. 2021, 321, H839–H849. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Li, J.; Ma, Y.; Khan, I.; Yang, Y.; Li, Y.; Zhang, C. Fermented Angelica sinensis activates Nrf2 signaling and modulates the gut microbiota composition and metabolism to attenuate d-gal induced liver aging. Food Funct. 2023, 14, 215–230. [Google Scholar] [CrossRef] [PubMed]
- Martucci, M.E.P.; De Vos, R.C.; Carollo, C.A.; Gobbo-Neto, L. Metabolomics as a potential chemotaxonomical tool: Application in the genus Vernonia Schreb. PLoS ONE 2014, 9, e93149. [Google Scholar] [CrossRef] [PubMed]
- Zoete, V.; Daina, A.; Bovigny, C.; Michielin, O. SwissSimilarity: A web tool for low to ultra-high throughput ligand-based virtual screening. J. Chem. Inf. Model. 2016, 56, 1399–1404. [Google Scholar] [CrossRef]
- Leonti, M.; Casu, L. Traditional medicines and globalization: Current and future perspectives in ethnopharmacology. Front. Pharmacol. 2013, 4, 92. [Google Scholar] [CrossRef]
| Metabolite | Species Where It Has Been Founded | Effect | Administrated Doses | References | |
|---|---|---|---|---|---|
| Effect | Metabolite Screening | ||||
| Resveratrol | Yucca schidigera Yucca gloriosa Yucca periculosa Yucca elephantipes | A Resveratrol supplement significantly reduced the concentrations of MDA and increased the levels of GPx, CAT, and SOD in horses | Horses were fed with 30 g (2 g of resveratrol) for 10 days and then 15 g (1 g of resveratrol) for 18 days. | [76] | [67,77,78,79] |
| Resveratrol increased the expression of SIRT1 (Sirtuin 1) and decreased ROS and p38 levels in human umbilical endothelial cells with TNF-α-induced injury. By inhibiting the MAPK/NF-κB pathway. | The cells were treated with doses of 0, 5, 10, and 20 μM. | [80] | |||
| Resveratrol inhibited PGE2 (Prostaglandin E2) synthesis in murine embryonic fibroblasts ex vivo. This was found to be due to resveratrol’s ability to bind directly with COX-2. | The IC50 (Half maximal inhibitory concentration) was 60 μM. | [81] | |||
| 4,4’-Dihydroxystilbene (DHS) | Yucca periculosa | DHS decreased levels of MDA and 8-oxo-dG (8-Oxo-2’-deoxyguanosine), TNF-α, COX-2, MMP-9 (Matrix metalloproteinase 9), and p65 in mice with cigarette smoke-induced lung failure. It also activates Nrf2 by suppressing its ubiquitylation. | Dose of 2 and 4 mg/kg. | [82] | [77] |
| DHS showed good antioxidant activity in vitro against the galvinoxyl radical, Cu (II) ions, and Fe2+/VC-induced lipid peroxidation in human erythrocyte ghosts. | Doses of 1, 2, and 3 μM were used for Fe2+/VC-induced lipid peroxidation. | [83] | |||
| 3,3’,5,5’-tetrahydroxy-4-methoxystilbene | Yucca periculosa Yucca schidigera Yucca gloriosa | The TEAC assay showed that 3,3’,5,5’-tetrahydroxy-4-methoxystilbene has the highest scavenging activity of all phenols in Yucca schidigera. It also reduced the effects of oxidative stress on blood platelets. | TEAC assay value was 2.252 mM | [67] | [67,77,78] |
| It inhibited the carbonylation, nitration, and oxidation of proteins. Reduced peroxynitrite-induced platelet lipid peroxidation. | A concentration of 0.1 mM was used | [84] | |||
| Yuccaol A | Yucca schidigera Yucca gloriosa | Yuccaol A significantly decreases the NO release in LPS-stimulated J774.A1 macrophages. | At a concentration of 100 µM | [34,78] | |
| Yuccaol C | Yucca schidigera Yucca gloriosa | Yuccaol C reduces iNOS expression by inhibiting the transcription factor NF-kB. | From 0.1 µM it reduces the expression of iNOS in J774.A1 macrophages induced by LPS (Lipopolysaccharide). | [34,78] | |
| Gloriosaol A | Yucca gloriosa | Showed an Antioxidant Activity higher than quercetin with the TEAC assay. | TEAC assay value was 5.55 mM | [69,78] | |
| Gloriosaol B | Yucca gloriosa | Showed an Antioxidant Activity higher than quercetin with the TEAC assay. | TEAC assay value was 3 mM | [69,78] | |
| Gloriosaol C | Yucca gloriosa | Showed an Antioxidant Activity higher than quercetin with the TEAC assay. | TEAC assay value was 5.6 mM | [69,78] | |
| Gloriosaol D | Yucca gloriosa | A combination of Gloriosaol D and E Showed an Antioxidant Activity higher than quercetin with the TEAC assay. | TEAC assay value was 4.91 mM | [69,78] | |
| Gloriosaol E | Yucca gloriosa | ||||
| Gallic acid | Yucca elephantipes Yucca aloifolia variegata Yucca aloifolia | Gallic acid increased Nrf2 expression, which suppresses ROS and IL-1β levels and blocked the activation of the inflammasome NLRP3 (Nucleotide-binding oligomerization domain-like receptor containing domain 3 of pyrin) in mice. | A dose of 100 mg/kg was injected intra-articular. | [85] | [86,87] |
| Gallic acid significantly inhibited inflammation in a rat model of arthritis in a dose-dependent manner. This could be due to an inhibition of PDE4 (Phosphodiesterase 4) activity, and a decrease in TNF-α concentration. | Doses of 1, 5, and 10 μg/g. | [88] | |||
| Chlorogenic acid | Yucca elephantipes Yucca aloifolia variegata Yucca aloifolia | Chlorogenic acid inhibited the production of TNF-α in and attenuated arthritis progression in collagen-induced arthritis mice. All of these are probably possible through the suppression of NF-κB pathways. | Doses of 30 mg/kg and 60 mg/kg for the attenuation of arthritis. | [89] | [79,86,87] |
| Cinnamic acid | Yucca elephantipes Yucca aloifolia variegata Yucca aloifolia | Cinnamic acid reduced the concentrations of MDA, TNF-α, and increased CAT activity in cisplatin-induced splenotoxicity in rats. | A dose of 50 mg/kg per 7 days | [90] | [86,87] |
| Hesperetin | Yucca elephantipes Yucca aloifolia variegata | Hesperetin decreased the levels of ROS, LPO (Lipid Peroxidation), TLR4 Toll-like receptor 4, p-NF-κB, and TNF-α and IL-1β, and increased the level of Nrf2 and HO-1 in mice with Aβ-induced neurodegeneration. | Doses of 50 mg/kg were injected for 6 weeks | [91] | [79,86,87] |
| Hesperetin decreased the expression of NO, IL-1β, IL-6, and TNF-α in LPS-stimulated BV-2 microglial cells. This is done by inhibiting the activation of the ERK pathway. | Cells were pretreated with doses of 0, 5, 25, 50, 100, and 200 µM | [92] | |||
| Naringenin | Yucca elephantipes Yucca aloifolia variegata | Mice with induced rheumatoid arthritis treated with naringenin significantly reduced inflammation and arthritis. It also decreased MPO and NO levels even more than those treated with methotrexate, a common treatment for arthritis. | The treatment against Rheumatoid Arthritis lasted three weeks with a dose of 40 mg/kg daily and orally. | [93] | [79,86,87] |
| Naringenin pretreatment reduced the concentration of NO, PGE2, iNOS, COX-2, IL-1β, TNF-α, and MCP-1 (Monocyte chemoattractant protein 1) in BV2 microglial cells treated with LPS. Attenuation could be due to the suppression of NF-κB and MAPK pathways. | Cells were treated with 80 µM | [94] | |||
| Luteolin | Yucca elephantipes Yucca aloifolia variegata | A clinical trial with a dietary formulation containing luteolin was administered to children with autism spectrum disorders (ASD) and it was found that this formulation significantly decreased IL-6 and TNF-α. | The dietary formulation contained 100 mg of luteolin, 70 mg of quercetin, and 30 mg of quercetin glycoside rutin, for 26 weeks. | [95] | [79,86,87] |
| Luteolin decreased MPO, H2O2, MDA, and NF-κB levels, while increasing GPx, SOD, CAT activity, and GSH (Glutathione), and vitamin C levels in rats exposed to sodium fluoride. | Doses of 100 and 200 mg/kg/day orally for one week. | [96] | |||
| Caffeic acid | Yucca elephantipes Yucca aloifolia variegata | Treatment with caffeic acid decreased the expression of NF-κB and IL-1β in Wistar rats with hearing loss | A dose of 30 mg/kg | [97] | [79,86,87] |
| Caffeic acid showed effective antioxidant properties in vitro compared to standard antioxidants. | At 10 and 30 μg/mL, caffeic acid showed 68.2 and 75.8% inhibition of lipid peroxidation. | [98] | |||
| Acacetin | Yucca elephantipes Yucca aloifolia variegata | Acacetin blocked the activation of NF-κB, decreased the activity of MPO, the expression of iNOS and COX-2, and increased the expression of SOD and HO-1 in sepsis-induced Acute lung injury mice. | Oral administration of 80 mg/kg showed the best results. | [99] | [86,87] |
| Epicatechin | Yucca elephantipes | Epicatechin decreased the levels of TNF-a, IL-6, NO, MPO, and MDA and inhibited NF-kB activation in mice with induced ulcerative colitis. | Doses of 100, 200, or 300 mg/kg were administered orally. | [100] | [79] |
| Epicatechin has peroxyl radical scavenging activity. | At a concentration of 20.53 µmol. | [101] | |||
| Protocatechuic acid | Yucca elephantipes | Protocatechuic acid showed higher antioxidant activity than Trolox in DPPH, ABTS, reducing power (Fe3+), reducing power (Cu2+), superoxide anion radical scavenging, hydroxyl radical scavenging, chelating capacity (Fe2+), and chelating capacity (Cu2+). | The ratio of IC50 Trolox/Protocatechuic acid was: 2.8, 2.3, 3.7, 6.1, 4.2, 1.0, 2.7, and 1.5, respectively. | [102] | [79] |
| Protocatechial acid treatment decreased the levels of NO and LPO and increased GSH, CAT, and SOD in different rat models. | Doses of 25, 50, and 100 mg/kg orally | [103] | |||
| Shikimic acid | Yucca carnerosana Yucca elephantipes | Shikimic acid treatment decreased TNF-α and IL-1β levels and inhibited of ERK 1/2 and p38 phosphorylation in LPS-stimulated RAW 264.7 macrophages. | Cells were cultured with 10 mM | [104] | [79,105] |
| Eriodictyol | Yucca carnerosana | Eriodictyol decreased NO production, prevented induced oxidation, and increased CAT activity in murine macrophages. | The mean effective concentrations of antioxidant activity were 14 μM. | [106] | [105] |
| Pre-treatment with eriodictyol induced the Nrf2 pathway in mice with acute lung injury. This decreased the levels of TNF-α, IL-6, IL-1β, MIP-2 (Macrophage inflammatory protein 2), H2O2, and MDA | 30 mg/kg was given orally 2 days before the acute lung injury. | [107] | |||
| Scopoletin | Yucca elephantipes | Scopoletin had scavenging activity against superoxide anion in the xanthine/xanthine oxidase reaction system. | At concentration of 12.5, 25, 50, and 100 µm. | [108] | [79] |
| Scopoletin reduced PGE2 and TNF-α expression, MPO activity, and MDA levels in carrageenan-induced mice. | A dose of 100 mg/kg | [109] | |||
| Nordihydrocapsiate | Yucca carnerosana | Nordihydrocapsiate inhibits NF-κB activation by inhibiting IκBα degradation and IKK activation. | At a concentration of 100 μM | [110] | [105] |
| Myricetin | Yucca elephantipes | Myricetin protects cells against H2O2-induced cell damage by scavenging radicals and restoring the expression of SOD, CAT, and GPx. | At a concentration of 10μg/ml | [111] | [79] |
| Myricetin suppresses activation of the NF-κB pathway; by inhibiting AKT and NF-κB p65 phosphorylation; and the MAPK signaling pathway; by inhibiting the phosphorylation of JNK, p-ERK, and p38. | At a concentration of 12.5 and 25 μM | [112] | |||
| Ellagic acid | Yucca elephantipes | Ellagic acid showed high radical scavenging capacity against DPPH, inhibits lipid peroxidation, and increases SOD, CAT, and GPX activity in V79-4 cells. | 4, 20, and 100 µg/ml | [113] | [79] |
| Ellagic acid intake decreased the expression of IL-1beta, IL-6, TNF-α, and MCP-1 and increased that of GPx, SOD, and CAT in diabetic mice. | Ellagic acid at 2% | [114] | |||
| Nordihydroguaiaretic acid | Yucca elephantipes | Nordihydroguaiaretic acid activates the Nrf2/HO-1 antioxidant pathway in cerebellar granule neurons. | At a concentration of 20 µM | [115] | [79] |
| Nordihydroguaiaretic acid suppressed ERK activation in RANKL-treated mouse RAW-D and BMM cells. | At a concentration of 5 µM | [116] | |||
| Sinapic acid | Yucca carnerosana Yucca elephantipes | Sinapic acid has a good antioxidant capacity in vitro. | It had an IC50 of 32.2 μM in the DPPH and for the Ferric Reducing Antioxidant Power assay of 482.6 μM/mol | [117] | [79,105] |
| Sinapic acid inhibited the expression of iNOS, COX-2, TNF-α, and IL-1β in RAW 264.7 cells induced with LPS. These observations are due to the suppression of the nuclear translocation of the p65 and p50 NF-κB subunits and the degradation of IκBα. | The reported inhibition was with pretreatment of 30 mg/kg orally 1 h before the induction of paw edema. | [118] | |||
| Cirsimarin | Yucca elephantipes | Cirsimarin in vitro inhibited lipid peroxidation and induced a 36% reduction in H2O2 production in adipose tissue. | IC50 = 370 µM in lipid peroxidation and 15 µM in H2O2 production. | [119] | [79] |
| Rosmarinic acid | Yucca elephantipes Yucca aloifolia variegata | Rosmarinic acid reduced the levels of MPO, NO, IL-6, IL-1β, TNF-α, iNOS, and COX-2 in mice with dextran sodium sulfate-induced colitis. | The dose range of 25–100 mg/kg | [120] | [79,87] |
| Rosmarinic acid showed protective activity against peroxynitrite-mediated damage and inhibited NO and iNOS in RAW264.7. This is done by inhibiting the serine phosphorylation of IκBα. | At a concentration of 10, 50 mm | [121] | |||
| Rosmarinic increased the activity of SOD, CAT, and GPx in aged mice. | Results were seen at a dose of 200 mg/kg once daily for 30 days. | [122] | |||
| Psoralen | Yucca elephantipes | Psoralens inhibit the expression of TNF-α, IL-1β, IL-6, and IL-8, and proteins involved in the TLR4-IRAK4-NF-κb pathway in LPS-stimulated cells. The anti-inflammatory activity could be due to an agonist effect on the estrogen receptor. | At a concentration of 12.5 micrograms/mL | [123] | [79] |
| Psoralen inhibits the expression of MMP-1, -2, -3, -9, -12, -13 and IL-1β, -6, -12 in human synoviocytes cultured with TNF-α. | At a concentration of 1,10 and 20 µM | [124] | |||
| 4-Hydroxyphenylacetic acid | Yucca elephantipes | 4-Hydroxyphenylacetic acid increased Nrf2 translocation to the nucleus, which enhanced antioxidant enzymes in mice with induced acute liver failure. | The mice were treated with 4-hydroxyphenylacetic acid at doses of 6, 12, or 25 mg/kg for 3 days. | [120] | [79] |
| Angelicin | Yucca elephantipes | Angelicin inhibited IL-4, IL-5, and IL-13 production and NF-kB activation in mice with ovalbumin-induced asthma. | Pretreatment of 2.5, 5, and 10 mg/kg was administered intraperitoneally | [125] | [79] |
| Angelicin blocked the phosphorylation of IκB-α, NF-κBp65, p38 MAPK, and JNK in mice with LPS-induced acute lung injury. | Doses of 1, 5, and 10 mg/kg were injected intraperitoneally. | [126] | |||
| Ferulic acid | Yucca carnerosana Yucca elephantipes Yucca aloifolia variegata | Ferulic acid demonstrated an anti-inflammatory effect and reduced IL-1β and TNF-α levels in mice with collagen-induced arthritis. | 1.28 μg/g/d were administered intragastrically twice a day. | [107] | [79,87,105] |
| Ferulic acid reduced superoxide radicals and ICAM-1 (Intercellular adhesion molecule) and NF-κB expression in mice with cerebral artery occlusion. | Dose of 100 mg/kg, intravenously | [127] | |||
| 6-Methylcoumarin | Yucca aloifolia variegata | 6-methylcoumarin reduced NO, PGE2, iNOS, COX-2 levels, and MAPK phosphorylation and degradation of RAW 264.7 cells stimulated by IκB-α LPS | Cells were pretreated with doses of 300, 400, and 500 μM for 1 h | [128] | [87] |
| Citropten | Yucca elephantipes | Citropten Reduces NFκB and MAPK Signaling Pathway, Alleviates Colon Inflammation and Effector T Cell Activity in Mice with DSS-Induced Colitis. | Oral administration of 10 mg/kg and 40 mg/kg | [129] | [79] |
| Emodin | Yucca elephantipes | Emodin decreased levels of amylase, lipase, TNF-α, IL-18, and ROS in rats with severe acute pancreatitis. | At a concentration of 6 mg/mL | [130] | [79] |
| Emodin reduced the levels of TNF-α, IL-6, MPO, MDA, Cytokine-induced neutrophil chemoattractant, MIP-2, ICAM-1, and NF-κB nuclear translocation in cholestatic-induced hepatitis rats. | 20 mg/kg/d were administered intragastrically for 4 days. | [131] | |||
| Chrysin | Yucca elephantipes | Chrysin treatment inhibits p65 NF-κB unit, TNF-α, IL-1β, and IL-6 levels, where 100 mg/kg had the same effect as 100 mg/kg methylprednisolone in rats with spinal cord injury. It also reduced the concentration of iNOS and NO. | Doses of 30 and 100 mg/kg/day for 26 days. | [132] | [79] |
| Chrysin treatment decreased lipid peroxidation and increased the activities of SOD, CAT GPx, GSH, vitamin C, and vitamin E in hepatitis-induced rats. | Doses of 25, 50, and 100 mg/kg/day orally for 6 days. | [133] | |||
| Alpinetin | Yucca carnerosana | Alpinetin decreased levels of ROS, MDA, NLRP3 inflammasome activation, and IL-1β, IL-6, TNF-α, COX-2, and iNOS in mice with induced liver fibrosis. This could be because the nuclear expression of Nrf2 increased. | Mice were injected intraperitoneally with doses of 15 and 60 mg/kg. | [134] | [105] |
| Alpinetin repressed p65 nuclear translocation, and IκB phosphorylation, and stimulated ERK phosphorylation in TNF-α-stimulated rat chondrocytes for 48 h. | The chondrocytes were treated with doses of 10 μM and 20 μM, | [135] | |||
| Vanillin | Yucca elephantipes | Vanillin treatment in ICR mice increased plasma antioxidant activity as demonstrated by the ORAC assay. It also showed good antioxidant capacity in vitro ith: DPPH radical, galvinoxyl radical, ABTS• scavenging assays, and Oxidative Hemolysis Inhibition Assay. | Doses of 25 mg/mL dissolved were administered orally. | [136] | [79] |
| Vanillin pretreatment inhibited hepatic lipid peroxidation, protein carbonyl formation, expression of TNF-α, IL-6, IL-1β, and increased CAT, SOD, and GSH levels in rats with induced hepatotoxicity. | Doses of 150 mg/kg were injected intraperitoneally for 3 days before induction of hepatotoxicity. | [137] | |||
| Quercetin | Yucca aloifolia variegata Yucca elephantipes | Quercetin pretreatment decreased the expression of NF-κB, VCAM-1, ICAM-1, E-selectin, and AP-1 in human umbilical vein endothelial cells induced with TNF-α. | The cells were cultured with quercetin for 18 h at a dose of 30 μg/mL. | [138] | [79,87] |
| Quercetin significantly increased CAT, SOD, and GPx activity in rats with induced diabetes. | A dose of 15 mg/kg/day was injected intraperitoneally. | [139] | |||
| Quercetin decreased IL-1β, TNF-a levels, and IκBα expression in patients with coronary artery disease. | A daily dose of 120 mg for 2 months. | [140] | |||
| Kaempferide | Yucca elephantipes | Kaempferol decreased levels of TNF-α, iNOS, IL-12, p38, and IκBα phosphorylation, p65 nuclear translocation, and increased HO-1 and Nrf2 levels in mice with nephrotoxicity. | Doses of 50, 100, 150 and 200 mg/kg. | [141] | [79] |
| Procyanidin B2 | Yucca aloifolia variegata Yucca elephantipes | Procyanidin B2 decreased the expression of p65, COX2, iNOS, IL-6, IL-1β, NO production, and blocked NLRP3 inflammasome activation in LPS-treated THP-1 macrophages. | Cells were treated with 10 μM. | [142] | [79,87] |
| Procyanidin B2 decreased PSA, TNF-α, IL-6, and IL-10 levels and increased SOD, CAT, GSH, Nrf2, and HO-1 levels in rats with chronic induced prostatitis. | Doses of 20 and 40 mg/kg were administered for 4 weeks. | [143] | |||
| Umbelliferone | Yucca elephantipes | Umbelliferone decreased lipid peroxidation and increased vitamin C, vitamin E, GSH levels, and SOD, CAT, and GPx activity in rats with induced diabetes. | Dose of 30 mg/kg for 45 days. | [144] | [79] |
| Umbelliferone reduced the levels of MPO, MDA, COX-2, PGE2, NF-kB, VEGF (Vascular endothelial growth factor), TNF-α, IL-1β, IL-6, IL-10, and IL-17 in rats treated with complete Freund’s adjuvant. | Doses of 10, 20, 40 mg/kg for 28 days | [145] | |||
| Catechin | Yucca elephantipes | Catechin decreased NO production and iNOS, COX2, TNF-α, and IL-1β expression in LPS-stimulated RAW 264.7 cells. Combined treatment with quercetin and catechin was also shown to have synergistic anti-inflammatory effects. | At a concentration of 150μM | [146] | [79] |
| Catechin decreased MDA level and increased SOD, GST (Glutathione-S-transferase), and CAT activities in diabetic-induced rats. | Doses of 20, 40 or 80 mg/kg/day | [147] | |||
| Scopolin | Yucca elephantipes | Scopolin reduced IL-6, VEGF, and FGF-2 in rats with adjuvant-induced arthritis. | Doses of 50 or 100 mg/kg were injected intraperitoneally for 10 days | [148] | [79] |
| Kaempferol | Yucca aloifolia variegata Yucca elephantipes | Kaempferol reduced iNOS, COX-2, and CRP (Reactive C-protein) expression and inhibits NF-κB activation by inhibiting IκBα degradation and IKK activation in the human hepatocyte-derived Chang Liver cell line. | Concentration from 5 to 200 μmol/L | [149] | [79,87] |
| Kaempferol pretreatment decreased ROS excess and increased SOD, CAT, and GPx levels in human erythrocytes with ROS-induced hemolysis. | A dose of 100 μg/mL suppressed 87.4% of hemolysis | [150] | |||
| Hesperidin | Yucca elephantipesYucca aloifolia variegata Yucca aloifolia Yucca flamentosa | Hesperidin decreased the levels of MDA, NO, NF-kB and p-Akt, while it increased the levels of GSH in rats with acute cisplatin-induced hepatotoxicity. | Dose of 100 or 200 mg/kg/day for 7 days | [151] | [79,87] |
| Hesperidin showed good scavenging potential against DPPH and protected yeast cells against stressors. | 25 and 50 µM were used in all tests | [152] | |||
| Apigenin | Yucca elephantipesYucca aloifolia variegata | Apigenin pretreatment decreased NF-kB and COX-2 expression, and IL-6, IL-1β, and TNF-α levels in ALI mice with LPS-induced inflammation. | Doses of 10 or 20 mg/kg were injected intraperitoneally. | [153] | [79,87] |
| Apigenin decreased LPO levels and increased SOD, CAT, GPx, vitamin C, vitamin E, and GSH levels in rats with oxidative stress caused by the carcinogen. | A dose of 25 mg/kg/day was administered intraperitoneally for 14 days. | [154] | |||
| Vanillic acid | Yucca elephantipes | Vanillic acid pretreatment inhibited oxidative stress, IL1β, TNF-α, and IL-33 expression and suppressed NF-κB activation in inflammation-induced mice. | Doses of 3–30 mg/kg injected intraperitoneally. | [155] | [79] |
| Vanillic acid pretreatment reduced lipid peroxidation and decreased IL-1β, IL-6, and TNF-α expression in rats with induced cardiotoxicity. It also showed good radical scavenging ability in vitro. | Dose of 5 mg or 10 mg/kg/day for 10 days. | [156] | |||
| Metabolite | Species Where It Has Been Founded | Effect | Administrated Doses | References | |
|---|---|---|---|---|---|
| Effect | Metabolite Screening | ||||
| Sarsasapogenin | Yucca schidigera Yucca aloifolia variegata | Sarsasapogenin suppressed NLRP3 inflammasome activation and the NF-κB pathway by downregulating PAR-1 (Protease-activated receptor 1) expression in rats with induced diabetes. | Dose 20 or 60 mg/kg/day by intragastric tube for 10 weeks. | [163] | [87] |
| Sarsapogenin decreases the levels of MDA, MPO, NO, TNF-α, IL-12, IL-6, TXB2 (Thromboxane B2), and IgE (Immunoglobulin E) in mice with induced asthma. | A dose of 50 µg/mouse | [164] | |||
| Hecogenin | Yucca elephantipes Yucca aloifolia variegata Yucca glauca | Hecogenin decreased NF-κB and p53 expression levels, decreased lipid peroxidation, and increased SOD, GPx, GST, CAT, vitamin E, vitamin C, and GSH levels in rats with induced myocardial infarction. | 50 µg/kg dose orally for 21 days | [165] | [86,87] |
| Hecogenin reduced the levels of MDA, MPO, TNF-α, IL-6, IL12, TXB2, IgE and NO, and increased SOD, GSH in rats with induced colitis | Dose of 50 µg/rat transrectally at 72, 48, 24 and 2 h before colitis induction. | [166] | |||
| Hecogenin decreases the levels of MPO, IL-6, IL-2, TBX2, and COX-2 expression in rats treated with complete Freund’s adjuvant. | At a concentration of 50 µg/kg dose, orally | [167] | |||
| Diosgenin | Yucca aloifolia variegata | Diosgenin increased SOD, GPx levels, and decreased ROS concentration, TBARS (Thiobarbituric acid-reactive substances), caspase-3 activation, NF-κB expression, and modulated PKA (Protein kinase A) and p38 activation. | Oral feeding at a dose of 130 mg/kg daily for 4 weeks. | [168] | [87] |
| Timosaponin BII | Yucca glauca | Timosaponin BII decreased IL-6, TNF-α, MDA levels and increased SOD, HO-1 and Nrf-2 levels in rats with induced myocardial infarction. | Dose of 50 or 100 mg/kg/day by gastric tube for 5 days. | [169] | [170] |
| Timosaponin BII decreased phosphorylation of mTOR (Mammalian target of rapamycin) and NF-κB. | 100, 300, and 500 mg/kg/day orally for 3 months | [171] | |||
| Timosaponin AIII | Yucca gloriosa Yucca macrocarpa | Thymosaponin AIII decreased IL-1β, TNF-α, IL-6, MPO levels, NF-κB activation, and increased IL-10 concentration in mice with induced colonic shortening. It also inhibited the binding of LPS to the Toll-like receptor 4 of macrophages and could be metabolized to sarsasapogenin by intestinal microbiota, a molecule that has also shown anti-inflammatory activity. | Oral administration of 5 or 10 mg/kg/day for 3 days. | [172] | [170] |
| Degalactotigonin | Yucca gloriosa | Degalactotigonin inhibited the phosphorylation of the EGFR (Epidermal growth factor receptor) in human pancreatic cancer cell lines PANC1. | At a concentration of 100 ng/mL for 24 h | [173] | [78,170] |
| Sweroside | Yucca aloifolia variegata | Sweroside pretreatment inhibited Keap1, induced Nrf2 nuclear translocation, inhibited ROS, and increased SOD and GPx activity in hypoxia/reoxygenation-induced H9c2 cells. | At a concentration of 50 μM for 24 h | [174] | [87] |
| Sweroside activated SIRT1, suppressed NF-κB, and promoted Forkhead O1 transcription factor signaling pathways in LPS-induced RAW264.7 cells. 20, | At a concentration of 40 and 80 µM | [175] | |||
| Metabolite | Species Where It Has Been Founded | Effect | Administrated Doses | References | |
|---|---|---|---|---|---|
| Effect | Metabolite Screening | ||||
| Vitexin-2″-O-rhamnoside | Yucca elephantipes | Vitexin-2″-O-rhamnoside had an antioxidant effect against tert-butyl-hydroperoxide-induced lipid peroxidation in ECV304 cell culture. | The optimal concentration in ECV304 cell culture was 62.5 μmol/L | [180] | [86] |
| Isosakuranetin-7-O-neohesperidoside (Poncirin) | Yucca elephantipes | A treatment with poncirin showed improvement in the liver in male albino mice with Carbon tetra chloride-induced liver injury. This is done by neutralizing nitric oxide production, enhancing the concentration of GST, GSH, CAT, SOD, and attenuating the activity of lipid peroxidase, MPO, and the levels of IL-1β, IL-6 and TNF-α. | At a concentration of 30 mg/kg. | [181] | [86] |
| Poncirin improved symptoms associated in mice with liver injury induced by paracetamol. This is done by decreasing the expression of NF-κB, JNK, and COX-2. | At a concentration of 30 mg/kg. | [182] | |||
| Apigenin-7-O-β-d-glucoside | Yucca elephantipes | Apigenin-7-O-β-d-glucoside showed antioxidant activity in vitro. | Where its IC50 (μg/mL) for ABTS was 36.5 ± 0.76, O•2− 86.4 ± 0.21, DPPH• 34.2 ± 0.98 and •OH 155.2 ± 0.25. | [100] | [86] |
| Myricitrin | Yucca elephantipes | Myricitrin treatment increased GSH level and Cytochrome P450 2E1 expression, and decreased lipid peroxidation, and COX-2 and TNF-a expression in mice with tetrachloride intoxication. carbon (CCl4) | Doses of 10, 30 and 100 mg/kg by oral tube, once a day for 2 days after poisoning. | [183] | [79] |
| Myricitrin can effectively scavenge free radicals in vitro and decrease acrylamide toxicity by inhibiting ROS generation in human gastrointestinal Caco-2 cells. | At a concentration of 2.5–10 μg | [184] | |||
| Kaempferol dirhamnoside | Yucca aloifolia variegata | In vitro kaempferol dirhamnoside showed high reactivity with DPPH, inhibited MPO activity and decreased ascorbyl radical-induced lipid peroxidation. | The IC50s were: 84.0 ± 7.8 μM, 86 ± 9.9 μM, 320 ± 14.1 μM, respectively | [185] | [87] |
| Phloridzin | Yucca elephantipes | Phloridzin increased SOD, GSH levels, and decreased TNF-α, IL-6, and MDA levels in LPS-treated mice. | 10–20 mg/kg, orally | [186] | [79] |
| Sweroside | Yucca aloifolia variegata | Sweroside pretreatment inhibited Keap1, induced Nrf2 nuclear translocation, inhibited ROS, and increased SOD and GPx activity in hypoxia/reoxygenation-induced H9c2 cells. | At a concentration of 50 μM for 24 h | [174] | [87] |
| Sweroside activated SIRT1, suppressed NF-κB, and promoted Forkhead O1 transcription factor signaling pathways in LPS-induced RAW264.7 cells. 20, | At a concentration of 40 and 80 µM | [175] | |||
| Kaempferol-3-O-glucoside | Yucca elephantipes | Kaempferol-3-O-glucoside had a good antioxidant activity with the DPPH radical scavenging method. | The mean IC50 of 1.25 ± 0.09 µg/mL | [187] | [79] |
| Quercitrin | Yucca aloifolia variegata Yucca elephantipes | Quercitrin decreased lipid peroxidative products and increased SOD, CAT, GPx, activity in rats with induced diabetes. | Oral administration of 30 mg/kg for 30 days | [188] | [79,87] |
| Quercitrin decreased iNOS expression in rats with induced colitis. This was due to the inhibition of NF-κB activity. | Oral administration of 1 or 5 mg/kg/day | [189] | |||
| Quercetin reduced MPO activity, TNF-α and IL-1β expression, iNOS, and reduced NF-κB activation in rats with colitis. | Oral administration of 1 mg/kg/day | [190] | |||
| Rutin | Yucca aloifolia variegata Yucca elephantipes | Rutin decreased MDA, ROS, NF-kBp65, TNF-α, and caspase-3 levels, as well as increased GSH levels, CAT activity, and Nrf-2 and HO-1 expression in rats with diabetic neuropathy. | Doses of 100 or 200 mg/kg were injected intraperitoneally for 8 weeks. | [191] | [79,87] |
| Rutin showed good radical scavenging activity in DPPH assay, total antioxidant activity, reducing power, hydroxyl radical scavenging assay, superoxide radical scavenging assay and lipid peroxidation assay. | At the concentration of 0.05 mg/mL it showed an inhibition of 90.4% in DPPH and the IC50 was 24 mg/mL for the lipid peroxidation assay. | [192] | |||
| Naringin | Yucca aloifolia variegata Yucca elephantipes | Naringenin decreased the levels of iNOS, ICAM-1, COX-2, MCP-1, TNF-α, TLR4, IL-6, p65, and p-IkBa in mice with induced colitis. | Dosage of 50 mg/kg/day orally for 10 days. | [193] | [79,87] |
| Naringenin supplementation increased SOD and CAT activities in hypercholesterolemic patients. | 400 mg/capsule/day for 8 weeks | [194] | |||
| Hyperoside | Yucca aloifolia variegata | Hyperoside decreased levels of TNF-α, IL-6, NO, NF-κB, and IκB-α degradation in lipopolysaccharide-stimulated mouse peritoneal macrophages. | The maximum rate of inhibition by hyperoside 5 μM. | [195] | [87] |
| Hyperoside decreased the levels of TNF-α, and the activation of AKT, NF-κB and ERK 1/2 in HUVEC. | Cells were treated with doses of 5, 10, 20 or 50 µM | [196] | |||
| Hyperoside scavenged ROS, which prevented lipid peroxidation, protein carbonyl, and cellular DNA damage in cells with H2O2-induced damage. | Cells were treated with 5 µM. | [197] | |||
| Esculin | Yucca aloifolia variegata | Esculin decreased levels of TNF-α, IL-6, MDA, MCP-1, Intracellular adhesion molecule 1, and p38, and increased SOD activity in C57BL/6J mice. | Dose of 5, 10, or 20 mg/kg for 2 weeks. | [198] | [87] |
| Esculin decreased NO, MPO, and MDA levels while increasing SOD activity in mice with acute gastric injury. | Doses of 12.5, 25 and 50 mg/kg | [199] | |||
| Kaempferol-3-O-rhamnoside | Yucca aloifolia variegata | Kaempferol-3-O-rhamnoside decreased the levels of IL-4, IL-5 and IL-13, TNF-α, IgE, this through the inhibition of Akt phosphorylation. | Doses of 26 mg/kg/day were administered orally for 6 days. | [200] | [87] |
| Metabolite | Species Where It Has Been Founded | Effect | Administrated Doses | References | |
|---|---|---|---|---|---|
| Effect | Metabolite Screening | ||||
| Gamma-Linolenic acid | Yucca elephantipes Yucca aloifolia variegata | Treatment with α-linolenic acid inhibited the production of PGE2 in on synovial membrane explant cultures with LPS-induced inflammation. | At a concentration of 300 µg/mL | [204] | [86,87] |
| α-linolenic acid inhibited PGE2 production in Aβ-stimulated PC12 cells. It also suppressed IκB-α degradation, which blocked NF-κB activation. | At a concentration of 100 µM | [205] | |||
| Caproic acid | Yucca aloifolia variegata | Capric acid suppressed MAPK phosphorylation and NF-kB activation in THP-1 cells treated with P. acnes. | 100 μM | [206] | [87] |
| Lauric acid | Yucca aloifolia variegata | Lauric acid suppressed MAPK phosphorylation and NF-kB activation in THP-1 cells treated with P. acnes. | 100 μM | [206] | [87] |
| Lauric acid had inhibitory activity against COX-1 and COX-2 | Inhibitory activity against COX-1 and COX-2 at 100 µg/mL. | [207] | |||
| Pentadecanoic acid | Yucca elephantipes Yucca aloifolia variegata | Pentadecanoic acid decreased the levels of MCP-1 and IL-6 in C57BL/6 J mice | 5 mg/kg for 12 weeks | [208] | [79,87] |
| Gama-linoleic acid | Yucca aloifolia variegata | Linoleic acid inhibited NO production and decreased p50 subunit expression of NF-κB, TNF-α, IL-6, IL-1β, and nitric oxide synthase 2 in LPS-treated RAW 264.7 cells. This could be due to the activation of the peroxisome proliferator-activated receptor-α. | 150 µM | [209] | [21,87] |
| Stearic acid | Yucca aloifolia variegata | Stearic acid inhibits lipid peroxidation and increases SOD and CAT activity in cultured rat cortical neurons. | 3,10 and 30 µmol/L | [210] | [21,87] |
| Stearic acid attenuated NF-κB activation in rats with cholestasis and leukocyte accumulation. | Intraperitoneal administration of 1000 nmol/kg/day was started 2 weeks before injury and continued for 5 weeks. | [211] | |||
| Oleic acid | Yucca aloifolia variegata | Oleic acid decreases the concentration of IL-1β, IL-6, and TNF-α and NF-κβ in rats with cadmium-induced lesions in heart and liver tissues. | 10 mg/kg, orally | [212] | [21,87] |
| Cis-5,8,11,14,17-Eicosapentaenoic acid | Yucca aloifolia variegata | Pretreatment with 5,8,11,14,17-eicosapentaenoic acid decreased TNF-α expression by preventing IκB-α phosphorylation in LPS-induced THP-1 cells. | Cells were incubated with 60 μM | [213] | [87] |
| 9,12-Octadecadienoic acid | Yucca elephantipes | 9,12-octadecadienoic acid had antioxidant activity with an IC50 value of 45.65 µg/mL by the DPPH Radical Scavenging Method. Anti-inflammatory activity was also tested in rats with carrageenan-induced edema, where treatment with 9,12-Octadecadienoic acid showed a significant reduction in edema paw volume. | 150μg/kg | [214] | [79] |
| Metabolite | Group | Species Where It Has Been Founded | Effect | Administrated Doses | References | |
|---|---|---|---|---|---|---|
| Effect | Metabolite Screening | |||||
| Stigmasterol | Phytosterol | Yucca aloifolia variegata | In chondrocytes from newborn mice or human osteoarthritis treated with IL-1β, preincubation with stigmasterol decreased the expression of Matrix Metallopeptidase 3, Matrix Metallopeptidase 13, ADAMTS-4 and PGE2, this by counteracting the effect of IL-1β on the NF-κB pathway. | Cells were pre-incubated for 48 h with 20 μg/ml | [218] | [87] |
| Stigmasterol treatment improved clinical severity by reducing joint destruction and decreasing the expression of TNF-α, IL-6, IL-1β, iNOS and COX-2, p65 and p38 by inhibiting the activation of p- IκBα in collagen-induced arthritic rats. | 200 mg/kg orally daily for 20 days | [219] | ||||
| In rats with ischemia/reperfusion brain injury, stigmasterol treatment decreased COX-2 and p65 expressions. In addition to significantly increasing the expression of Nrf2, HO-1, SOD, CAT, and GPx. | Dosis de 20, 40 y 80 mg/kg | [220] | ||||
| Phytol | Terpenes | Yucca aloifolia variegata | Phytol treatment reduced MPO activity and the concentration of TNF-α, IL-6 and COX-2. By downregulating p38 and NF-κB signaling pathways in a mouse model of arthritis induced by complete Freund’s adjuvant. | Oral administration of 50 mg/kg | [221] | [87] |
| Phytol exhibits a dose-dependent anti-inflammatory effect in formalin-induced paw edema by decreasing the levels of COX-1, COX-2, NF-κB and IL-1 β. | 100 mg/kg of phytol | [222] | ||||
| Phytol demonstrated a strong antioxidant effect in vitro. Eliminating hydroxyl radicals and nitric oxide and preventing the formation of TBARS | The concentration of 0.9, 1.8, 3.6, 5.4 and 7.2 ng/mL was used in the in vitro tests. | [223] | ||||
| Malic acid | Dicarboxylic acid | Yucca aloifolia variegata | Malic acid decreases TNF-α levels and inhibits platelet aggregation in rats with myocardial ischemia/reperfusion injury. | Doses of 500 mg/kg and 250 mg/kg | [224] | [87] |
| Loliolide | Terpenes | Yucca aloifolia variegata | Loliolide was found to exert dose-dependent positive effects on the protective effects against peroxide-induced cell damage. | Cells were pretreated with 250 and 500 μM of loliode | [225] | [87] |
| Loliolide treatment in LPS-stimulated RAW 264.7 macrophages decreased TNF-α and IL-6 production. | 100 µM | [226] | ||||
| Loliolide treatment in LPS-stimulated RAW 264.7 macrophages downregulated IκBα and p65 in a dose-dependent manner, thereby inhibiting NF-κB nuclear translocation. In addition, it downregulates the proteins involved in the MAPK pathway (p38, ERK, and JNK), thus inhibiting MAPK phosphorylation. | 15.6, 31.2, 62.5 µm/mL | [227] | ||||
| α-Tocopherol | Tocopherol | Yucca aloifolia | Oral α-tocopherol supplementation maintains cellular redox status in polychlorinated biphenyl-induced toxicity in rat liver, lung, and kidney. Therefore, the activities of SOD, CAT, GPx, and glutathione reductase are maintained, and the levels of lipid peroxides, hydroxyl radicals, and hydrogen peroxides are kept low. | 50 mg/kg body weight/day | [228] | [21] |
| α-Tocopherol treatment significantly reduced IL-6 levels, thereby inhibiting NF-κB-mediated gene transcription in cancerous mice. | 150 mg/kg per day orally for 15 consecutive days | [229] | ||||
| γ-Tocotrienol | Tocotrienols | Yucca aloifolia | γ-tocotrienol an inhibits NF-κB pathaway in RAW 264.7 macrophages. | 20 µM | [230] | [21] |
| γ-tocotrienol treatment in TNF-α-induced inflammation in adipocytes suppressed IκB-α phosphorylation and NF-κB activation. | Cells were pretreated with 0.024, 0.24, and 2.4 μM γ-tocotrienol for 6 h. | [231] | ||||
| In rats with induced arthritis, treatment with γ-tocotrienol reduced arthritis-induced changes in the C-reactive protein, TNF-α, SOD, and GSH levels. | The rats were treated orally with 5 mg/kg body weight of γ-tocotrienol between days 21 and 45. | [232] | ||||
| δ-Tocotrienol | Tocotrienols | Yucca aloifolia | δ-tocotrienol inhibits NO production and expression of IL-1β, IL-1α, IL-6, TNF-α, IL-12, iNOS, VCAM1 (Vascular cell adhesion protein 1), ICAM1, COX2, IL-1RA, TRAF1 and CD40 in mice. | Mice were fed 100 ppm tocotrienol | [233] | [21] |
| Succinic acid | Dicarboxylic acid | Yucca carnerosana | The injection of succinic acid in Wistar rats showed a high inhibitory capacity of LPO. | 25 mg/kg intraperitoneally injected into rats | [234] | [105] |
| Neophytadiene | Terpenes | Yucca aloifolia variegata | Neophytadiene was treated with LPS-stimulated RAW 264.7 and pretreated in rats injected with 10 mg/kg LPS. Neophytadiene inhibited NO production and the expression of TNF-α, IL-6, and IL-10, | 25, 50, 100 μM/mL in vitro 12, 25, 50 mg/kg pretreated for 7 days in vivo | [235] | [87] |
| Mangiferin | Xanthones | Yucca elephantipes | Mangiferin pretreatment increases GST, GPx, SOD, and CAT levels in rats with induced myocardial infarction. | 100 mg/kg suspended in 2 mL dimethyl sulfoxide administered intraperitoneally for 28 days | [236] | [79] |
| Mangiferin pretreatment prevented apoptosis and decreased levels of MDA, IL-1F, and IL-18 in mice subjected to cecal ligation and puncture. This is due to the inhibition of the NLRP3 inflammasome and the upregulation of Nrf2. | 15 mg/kg | [237] | ||||
| Mangiferin pretreatment increases HO-1 expression and activity in sepsis-induced mice with acute lung injury. This inhibited MAPK and NF-κB signaling. | 10, 30, and 100 mg/kg once daily for 7 days before cecal ligation and puncture. | [238] | ||||
| Cinnamaldehyde | Terpenes | Yucca aloifolia variegata | Cinnamaldehyde increased antioxidant enzymes such as SOD, GPx, and GST. | Rats were administered orally by gavage at dose levels of 2.14, 6.96, 22.62, and 73.5 mg/kg body weight/day for a period of 10, 30, and 90 days. | [239] | [87] |
| Cinnamaldehyde reduced levels of proinflammatory cytokines and increased levels of antioxidant enzymes in patients with rheumatoid arthritis. Molecular docking indicated that cinnamaldehyde interacts with the key residues of TNF-α and IL-6. | 20 or 40 µM cinnamaldehyde | [240] | ||||
| Nerolidol | Terpenes | Yucca elata Yucca filamentosa | Nerolidol pretreatment increases SOD, CAT, and GPx, activity by enhancing MP-activated protein kinase phosphorylation. | A significant effect started at 30 μmol/kg | [241] | [59] |
| Nerolidol decreased the expression of TNF-α, IL-1β, IL-6, NF-kB, PGE-2, and COX-2 and increased the level of IL-10, IL-4, and serum, antioxidant activity in rats with arthritis induced. | Doses of 200, 400, and 800 m,g/kg were administered for 28 days. | [242] | ||||
| Chrysophanol | Anthraquinone | Yucca elephantipes | Chrysophanol reduced protein expression of NF-κBp65, p-NF-κB p65, IκBα, p-IκBα, TNF-α, and IL-1β, decreased MPO and MDA levels, and increased SOD activity in mice with acute lung injury (LPS). This by regulating HMGB1/NF-κB signaling through histone deacetylase 3. | Doses of 7.5, 15 and 30 mg/kg | [243] | [79] |
| Chrysophanol increased Nrf2 levels and decreased HO-1, glutamate-cysteine ligase, glutamate cysteine ligase, IKKa, IkBa, NF-kB, and ROS generation in H9C2 cells stimulated with LPS. Chrysophanol is suggested to act as an activator of Nrf2. | Cells were cultured at doses of 0, 10, 20, 40, 80, 160, and 320 uM. | [159] | ||||
| Metabolite | Yucca Specie | Plant Material | Extraction | Concentration | Reference |
|---|---|---|---|---|---|
| Resveratrol | Yucca schidigera | Bark was obtained from Desert King Int., Chula Vista, CA, USA | Ultrasonic | 9.11 ± 2.11 mg/g plant | [77] |
| Yucca schidigera | Yucca bark was collected in October 2001 in San Diego, CA. | Soxhlet | 2.5 mg/300 g of Yucca powder | [68] | |
| 3,3’,5,5’-tetrahydroxy-4-methoxystilbene | Yucca schidigera | 5.1 mg/300 g of Yucca powder | |||
| Yucca schidigera | Bark was obtained from Desert King Int., Chula Vista, CA, USA | Ultrasonic | 9.10 ± 1.10 mg/g plant | [68] | |
| Yuccaol A | Yucca schidigera | 14.72 ± 2.10 mg/g plant | |||
| Yucca gloriosa roots | The roots and the bark were collected in December, in Tbilisi, Georgia. | MeOH at room temperature | 0.02 ± 0.01 mg/g plant | ||
| Yucca gloriosa | 2.64 ± 0.80 mg/g plant | ||||
| Yucca schidigera | Yucca bark was collected in October 2001 in San Diego, CA. | Soxhlet | 3.1 mg/300 g of Yucca powder | [68] | |
| Yuccaol C | Yucca schidigera | 8.0 mg/300 g of Yucca powder | |||
| Yucca gloriosa roots | The roots and the bark were collected in December, in Tbilisi, Georgia. | MeOH at room temperature | 10.68 ± 1.01 | [77] | |
| Yucca gloriosa | 13.85 ± 1.21 mg/g plant | ||||
| Yucca schidigera | Bark was obtained from Desert King Int., Chula Vista, CA, USA | Ultrasonic | 12.02 ± 2.28 mg/g plant | ||
| Gloriosaol A | Yucca gloriosa roots | The roots and the bark were collected in December, in Tbilisi, Georgia | MeOH at room temperature | 3.02 ± 1.50 mg/g plant | [77] |
| Yucca gloriosa | 11.87 ± 1.32 mg/g plant | ||||
| Gloriosaol B | Yucca gloriosa roots | 5.30 ± 1.80 mg/g plant | |||
| Yucca gloriosa | 29.73 ± 2.15 mg/g plant | ||||
| Gloriosaol C | Yucca gloriosa roots | 3.33 ± 0.9 mg/g plant | |||
| Yucca gloriosa | 4.53 ± 1.42 mg/g plant | ||||
| Gloriosaol D & Gloriosaol E | Yucca gloriosa roots | 4.96 ± 1.71 mg/g plant | |||
| Yucca gloriosa | 10.85 ± 1.58 mg/g plant | ||||
| Gallic acid | Yucca elephantipes flowers | Collected during summer in the city of Xalapa, Veracruz, México | MeOH at room temperature | 5.28 μg/g of dry sample | [79] |
| Protocatechuic acid | 21.14 μg/g of dry sample | ||||
| Vanillic acid | 205.28 μg/g of dry sample | ||||
| Chlorogenic acid | 47.52 μg/g of dry sample | ||||
| Caffeic acid | 74.1 μg/g of dry sample | ||||
| Ferulic acid | 140.59 μg/g of dry sample | ||||
| Rutin | 393.2 μg/g of dry sample | ||||
| Quercetin | 9.54 μg/g of dry sample | ||||
| Kaempferol | 37.16 μg/g of dry sample |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Culhuac, E.B.; Maggiolino, A.; Elghandour, M.M.M.Y.; De Palo, P.; Salem, A.Z.M. Antioxidant and Anti-Inflammatory Properties of Phytochemicals Found in the Yucca Genus. Antioxidants 2023, 12, 574. https://doi.org/10.3390/antiox12030574
Culhuac EB, Maggiolino A, Elghandour MMMY, De Palo P, Salem AZM. Antioxidant and Anti-Inflammatory Properties of Phytochemicals Found in the Yucca Genus. Antioxidants. 2023; 12(3):574. https://doi.org/10.3390/antiox12030574
Chicago/Turabian StyleCulhuac, Erick Bahena, Aristide Maggiolino, Mona M. M. Y. Elghandour, Pasquale De Palo, and Abdelfattah Z. M. Salem. 2023. "Antioxidant and Anti-Inflammatory Properties of Phytochemicals Found in the Yucca Genus" Antioxidants 12, no. 3: 574. https://doi.org/10.3390/antiox12030574
APA StyleCulhuac, E. B., Maggiolino, A., Elghandour, M. M. M. Y., De Palo, P., & Salem, A. Z. M. (2023). Antioxidant and Anti-Inflammatory Properties of Phytochemicals Found in the Yucca Genus. Antioxidants, 12(3), 574. https://doi.org/10.3390/antiox12030574








