Extracts of Eryngium foetidum Leaves from the Amazonia Were Efficient Scavengers of ROS and RNS
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Leaves of Eryngium foetidum
2.3. Extracts of Eryngium foetidum Leaves
2.4. Phenolic Compounds Composition by HPLC-DAD
2.5. In Vitro Antioxidant Capacity of E. foetidum Extracts to Scavenge ROS and RNS
2.5.1. Singlet Oxygen (1O2)-Quenching Assay
2.5.2. Superoxide Radical (O2•−)-Scavenging Assay
2.5.3. Hypochlorous Acid (HOCl)-Scavenging Assay
2.5.4. Hydrogen Peroxide (H2O2)-Scavenging Assay
2.5.5. Peroxyl Radicals (ROO•)-Scavenging Assay
2.5.6. Peroxynitrite Anion (ONOO−)-Scavenging Assay
2.6. Statistical Analysis
3. Results and Discussion
3.1. Composition of Phenolic Compounds in the E. foetidum Extracts
3.2. In Vitro Antioxidant Capacity of Eryngium foetidum Extracts to Scavenge ROS and RNS
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Brasil. Ministério da Agricultura, Pecuária e Abastecimento. Manual de Hortaliças não Convencionais; Secretaria de Desenvolvimento Agropecuário e Cooperativismo: Brasília, Brazil, 2010; p. 92. [Google Scholar]
- She, M.; Watson, M. Eryngium Linnaeus, Sp. Pl. 1: 232.1753. Flora China 2005, 14, 23–24. [Google Scholar]
- Paul, J.H.; Seaforth, C.E.; Tikasingh, T. Eryngium foetidum L.: A review. Fitoterapia 2011, 82, 302–308. [Google Scholar] [CrossRef]
- Singh, S.; Singh, D.R.; Salim, K.M.; Srivastava, A.; Singh, L.B.; Srivastava, R.C. Estimation of proximate composition, micronutrients and phytochemical compounds in traditional vegetables from Andaman and Nicobar Islands. Int. J. Food Sci. Nutr. 2011, 62, 765–773. [Google Scholar] [CrossRef] [PubMed]
- Mekhora, C.; Muangnoi, C.; Chingsuwanrote, P.; Dawilai, S.; Svasti, S.; Chasri, K.; Tuntipopipat, S. Eryngium foetidum suppresses inflammatory mediators produced by macrophages. Asian Pac. J. Cancer Prev. 2012, 13, 653–664. [Google Scholar] [CrossRef]
- Dawilai, S.; Muangnoi, C.; Praengamthanachoti, P.; Tuntipopipat, S. Anti-inflammatory activity of bioaccessible fraction from Eryngium foetidum leaves. Biomed. Res. Int. 2013, 2013, 958567. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, T.L.M.; Silva, M.E.P.; Gurgel, E.S.C.; Oliveira, M.S.; Lucas, F.C.A. Eryngium foetidum L. (Apiaceae): A Literature Review of Traditional Uses, Chemical Composition, and Pharmacological Activities. Evid.-Based Complement. Altern. Med. 2022, 2896895. [Google Scholar] [CrossRef]
- Leitão, D.S.T.C.; Siqueira, F.C.; de Sousa, S.H.B.; Mercadante, A.Z.; Chisté, R.C.; Lopes, A.S. Amazonian Eryngium Foetidum Leaves Exhibited Very High Contents of Bioactive Compounds and High Singlet Oxygen Quenching Capacity. Int. J. Food Prop. 2020, 23, 1452–1464. [Google Scholar] [CrossRef]
- Hewage, G.; Hemachandra, T.K.; Thuvaragan, S.; Sanmugarajah, V. Pharmacological screening of Eryngium foetidum Linn—A Review. Borneo J. Pharm. 2021, 4, 248–259. [Google Scholar] [CrossRef]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.D.; Mazur, M.; Telser, J. Free Radicals and Antioxidants in Normal Physiological Functions and Human Disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef]
- Halliwell, B.; Aeschbach, R.; Löliger, J.; Aruoma, O.I. The characterization of antioxidants. Food Chem. Toxicol. 1995, 33, 601–617. [Google Scholar] [CrossRef]
- Okayama, Y. Oxidative stress in allergic and inflame matory skin diseases. Curr. Drug Targets-Inflamm. Allergy 2005, 4, 517–519. [Google Scholar] [CrossRef]
- Lee, J.H.; Johnson, J.V.; Talcott, S.T. Identification of ellagic acid conjugates and other polyphenolics in muscadine grapes by HPLC-ESI-MS. J. Agric. Food Chem. 2005, 53, 6003–6010. [Google Scholar] [CrossRef]
- Carr, A.C.; McCall, M.R.; Frei, B. Oxidation of LDL by myeloperoxidase and reactive nitrogen species: Reaction pathways and antioxidant protection. Arterioscler. Thromb. Vasc. Biol. 2000, 20, 1716–1723. [Google Scholar] [CrossRef]
- Choe, E.; Min, D.B. Chemistry and Reactions of Reactive Oxygen Species in Foods. Crit. Rev. Food Sci. Nutr. 2006, 46, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Brewer, M.S. Natural antioxidants: Sources, compounds, mechanisms of action, and potential applications. Compr. Rev. Food Sci. Food Saf. 2011, 10, 221–247. [Google Scholar] [CrossRef]
- Tajik, N.; Tajik, M.; Mack, I.; Enck, P. The potential effects of chlorogenic acid, the main phenolic components in coffee, on health: A comprehensive review of the literature. Eur. J. Nutr. 2017, 56, 2215–2244. [Google Scholar] [CrossRef]
- Rojas, J.; Buitrago, A. Antioxidant activity of phenolic compound biosynthesized by plants and its relationship with preventio of neurodegenerative diseases. Bioact. Compd. 2019, 3–31. [Google Scholar] [CrossRef]
- Phaniendra, A.; Jestadi, D.B.; Periyasamy, L. Free radicals: Properties, sources, targets, and their implication in various diseases. Indian J. Clin. Biochem. 2015, 30, 11–26. [Google Scholar] [CrossRef]
- Thomas, P.S.; Essien, E.E.; Ntuk, S.J.; Choudhary, M.I. Eryngium foetidum L. essential oils: Chemical composition and antioxidant capacity. Medicines 2017, 4, 24. [Google Scholar] [CrossRef]
- Singh, S.; Singh, D.R.; Banu, S.; Salim, K.M. Determination of bioactives and antioxidant activity in Eryngium foetidum L.: A traditional culinary and medicinal herb. Proc. Natl. Acad. Sci. India Sect. B Biol. Sci. 2013, 83, 453–460. [Google Scholar] [CrossRef]
- Souza, T.C.L.; Silveira, T.F.F.; Rodrigues, M.I.; Ruiz, A.L.T.G.; Neves, D.A.; Duarte, M.C.T.; Cunha-Santos, E.C.M.; Kuhnle, G.; Ribeiro, A.B.; Godoy, H.T. A study of the bioactive potential of seven neglected and underutilized leaves consumed in Brazil. Food Chem. 2021, 364, 130350. [Google Scholar] [CrossRef]
- Chisté, R.C.; Mercadante, A.Z.; Gomes, A.; Fernandes, E.; Lima, J.L.F.C.; Bragagnolo, N. In vitro scavenging capacity of annatto seed extracts against reactive oxygen and nitrogen species. Food Chem. 2011, 127, 419–426. [Google Scholar] [CrossRef] [PubMed]
- Commission Directive 95/45/EC. Laying down specific purity criteria concerning colours for use in foodstuffs. Off. J. Eur. Community 1995, 226, 1–45. [Google Scholar]
- Chisté, R.C.; Mercadante, A.Z. Identification and Quantification, by HPLC-DAD-MS/MS, of Carotenoids and Phenolic Compounds from the Amazonian Fruit Caryocar Villosum. J. Agric. Food Chem. 2012, 60, 5884–5892. [Google Scholar] [CrossRef] [PubMed]
- ICH. International Conference on Harmonization of Technical Requirements for the Registration of Pharmaceuticals for Human Use. Guidance for Industry, Q2B Validation of Analytical Procedures: Methodology; International Conference on Harmonization Secretariat, c/o International Federation of Pharmaceutical Manufacturers and Associations: Geneva, Switzerland, 2005; pp. 7–10. [Google Scholar]
- Rodrigues, E.; Mariutti, L.R.B.; Mercadante, A.Z. Carotenoids and Phenolic Compounds from Solanum Sessiliflorum, an Unexploited Amazonian Fruit, and Their Scavenging Capacities against Reactive Oxygen and Nitrogen Species. J. Agric. Food Chem. 2013, 61, 3022–3029. [Google Scholar] [CrossRef]
- Gomes, A.; Fernandes, E.; Silva, A.M.S.; Santos, C.M.M.; Pinto, D.C.G.A.; Cavaleiro, J.A.S.; Lima, J.L.F.C. 2-Styrylchromones: Novel Strong Scavengers of Reactive Oxygen and Nitrogen Species. Bioorg. Med. Chem. 2007, 15, 6027–6036. [Google Scholar] [CrossRef]
- Santana-Gálvez, J.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A. Chlorogenic acid: Recent advances on its dual role as a food additive and a nutraceutical against metabolic syndrome. Molecules 2017, 22, 358. [Google Scholar] [CrossRef]
- Naveed, M.; Hejazi, V.; Abbas, M.; Kamboh, A.A.; Khan, G.J.; Shumzaid, M.; XiaoHui, Z. Chlorogenic acid (CGA): A pharmacological review and call for further research. Biomed. Pharmacother. 2018, 97, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Guo, D.; Bi, H. Chlorogenic acid attenuates hydrogen peroxide-induced oxidative stress in lens epithelial cells. Int. J. Mol. Med. 2018, 41, 765–772. [Google Scholar] [CrossRef]
- Stefanello, N.; Spanevello, R.M.; Passamonti, S.; Porciúncula, L.; Bonan, C.D.; Olabiyi, A.A.; Schetinger, M.R.C. Coffee, caffeine, chlorogenic acid, and the purinergic system. Food Chem. Toxicol. 2019, 123, 298–313. [Google Scholar] [CrossRef]
- Ghasemzadeh, A.; Jaafar, H.Z.; Juraimi, A.S.; Tayebi-Meigooni, A. Comparative evaluation of different extraction techniques and solvents for the assay of phytochemicals and antioxidant activity of hashemi rice bran. Molecules 2015, 20, 10822–10838. [Google Scholar] [CrossRef]
- Rahman, M.; Hossain, S.; Rahaman, A.; Fatima, N.; Nahar, T.; Uddin, B.; Basunia, M.A. Antioxidant activity of Centella asiatica (Linn.) Urban: Impact of extraction solvent polarity. J. Pharmacogn. Phytochem. 2013, 1, 27–32. [Google Scholar]
- Barchan, A.; Bakkali, M.; Arakrak, A.; Pagán, R.; Laglaoui, A. The effects of solvents polaritiy on the phenolic contents and antioxidant activity of three Mentha species extracts. Int. J. Curr. Microbiol. Appl. Sci. 2014, 3, 399–412. [Google Scholar]
- Chisté, R.C.; Benassi, M.T.; Mercadante, A.Z. Effect of solvent type on the extractability of bioactive compounds, antioxidant capacity and colour properties of natural annatto extracts. Int. J. Food Sci. Technol. 2011, 46, 1863–1870. [Google Scholar] [CrossRef]
- Abdullah, M.Z.; Mohd Ali, J.; Abolmaesoomi, M.; Abdul-Rahman, P.S.; Hashim, O.H. Anti-proliferative, in vitro antioxidant, and cellular antioxidant activities of the leaf extracts from Polygonum minus Huds: Effects of solvent polarity. Int. J. Food Prop. 2017, 20 (Suppl. 1), 846–862. [Google Scholar] [CrossRef]
- Nawaz, H.; Shad, M.A.; Rehman, N.; Andaleeb, H.; Ullah, N. Effect of solvent polarity on extraction yield and antioxidant properties of phytochemicals from bean (Phaseolus vulgaris) seeds. Braz. J. Pharm. Sci. 2020, 56, e17129. [Google Scholar] [CrossRef]
- Shafique, M.; Hussain, S.; Asif, S.; Pradhan, V.; Farooqui, M. Thermodynamic characteristics of solvents: A review. Res. J. Chem. Sci. 2013, 3, 98–104. [Google Scholar]
- Alcântara, M.A.; Polari, I.D.L.B.; de Albuquerque Meireles, B.R.L.; de Lima, A.E.A.; da Silva Junior, J.C.; de Andrade Vieira, É.; de Magalhães Cordeiro, A.M.T. Effect of the solvent composition on the profile of phenolic compounds extracted from chia seeds. Food Chem. 2019, 275, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Salas, P.; Morales-Soto, A.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Phenolic-compound-extraction systems for fruit and vegetable samples. Molecules 2010, 15, 8813–8826. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Correa, H.A.; Bitencourt, R.G.; Kayano, A.C.A.; Magalhães, P.M.; Costa, F.T.; Cabral, F.A. Integrated extraction process to obtain bioactive extracts of Artemisia annua L. leaves using supercritical CO2, ethanol and water. Ind. Crops Prod. 2016, 95, 535–542. [Google Scholar] [CrossRef]
- Kumar, A.; Premoli, M.; Aria, F.; Bonini, S.A.; Maccarinelli, G.; Gianoncelli, A.; Mastinu, A. Cannabimimetic plants: Are they new cannabinoidergic modulators? Planta 2019, 249, 1681–1694. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Rather, M.A.; Kumar Jha, A.; Shashank, A.; Singhal, S.; Sharma, M.; Mastinu, A. Artocarpus lakoocha Roxb. and Artocarpus heterophyllus Lam. Flowers: New Sources of Bioactive Compounds. Plants 2020, 9, 1329. [Google Scholar] [CrossRef]
- Abate, G.; Zhang, L.; Pucci, M.; Morbini, G.; Mac Sweeney, E.; Maccarinelli, G.; Ribaudo, G.; Gianoncelli, A.; Uberti, D.; Memo, M.; et al. Phytochemical Analysis and Anti-Inflammatory Activity of Different Ethanolic Phyto-Extracts of Artemisia annua L. Biomolecules 2021, 11, 975. [Google Scholar] [CrossRef]
- Rashmi, H.B.; Negi, P.S. Phenolic acids from vegetables: A review on processing stability and health benefits. Food Res. Int. 2020, 136, 109298. [Google Scholar] [CrossRef]
- Farhoosh, R.; Johnny, S.; Asnaashari, M.; Molaahmadibahraseman, N.; Sharif, A. Structure–antioxidant activity relationships of o-hydroxyl, o-methoxy, and alkyl ester derivatives of p-hydroxybenzoic acid. Food Chem. 2016, 194, 128–134. [Google Scholar] [CrossRef]
- Ribeiro, A.B.; Chisté, R.C.; Lima, J.L.F.C.; Fernandes, E. Solanum Diploconos Fruits: Profile of bioactive compounds and in vitro antioxidant capacity of different parts of the fruit. Food Funct. 2016, 7, 2249–2257. [Google Scholar] [CrossRef] [PubMed]
- Mandal, S.; Hazra, B.; Sarkar, R.; Biswas, S.; Mandal, N. Assessment of the antioxidant and reactive oxygen species scavenging activity of methanolic extract of Caesalpinia crista leaf. Evidence-Based Complementary and Alternative. Medicine 2011, 2011, 173768. [Google Scholar]
- Pryor, W.A.; Houk, K.N.; Foote, C.S.; Fukuto, J.M.; Ignarro, L.J.; Squadrito, G.L.; Davies, K.J. Free radical biology and medicine: It’s a gas, man! Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2006, 291, R491–R511. [Google Scholar] [CrossRef] [PubMed]
- Kalyanaraman, B. Teaching the basics of redox biology to medical and graduate students: Oxidants, antioxidants and disease mechanisms. Redox Biol. 2013, 1, 244–257. [Google Scholar] [CrossRef]
- Blázquez-Castro, A.; Breitenbach, T.; Ogilby, P.R. Singlet oxygen and ROS in a new light: Low-dose subcellular photodynamic treatment enhances proliferation at the single cell level. Photochem. Photobiol. Sci. 2014, 13, 1235–1240. [Google Scholar] [CrossRef]
- Krumova, K.; Cosa, G. Chapter 1: Overview of Reactive Oxygen Species. Singlet Oxyg. Appl. Biosci. Nanosci. 2016, 1, 1–21. [Google Scholar]
- Sies, H. Hydrogen peroxide as a central redox signaling molecule in physiological oxidative stress: Oxidative eustress. Redox Biol. 2017, 11, 613–619. [Google Scholar] [CrossRef] [PubMed]
- Carrasco, E.; Blázquez-Castro, A.; Calvo, M.I.; Juarranz, Á.; Espada, J. Switching on a transient endogenous ROS production in mammalian cells and tissues. Methods 2016, 109, 180–189. [Google Scholar] [CrossRef]
- Carrasco, E.; Calvo, M.I.; Blázquez-Castro, A.; Vecchio, D.; Zamarrón, A.; De Almeida, I.J.D.; Stockert, J.C.; Hamblin, M.R.; Juarranz, A.; Espada, J. Photoactivation of ROS production in situ transiently activates cell proliferation in mouse skin and in the hair follicle stem cell niche promoting hair growth and wound healing. J. Investig. Dermatol. 2015, 135, 2611–2622. [Google Scholar] [CrossRef]
- Westberg, M.; Bregnhøj, M.; Blázquez-Castro, A.; Breitenbach, T.; Etzerodt, M.; Ogilby, P.R. Control of singlet oxygen production in experiments performed on single mammalian cells. J. Photochem. Photobiol. A Chem. 2016, 321, 297–308. [Google Scholar] [CrossRef]
- Almeida, I.F.; Fernandes, E.; Lima, J.L.F.C.; Costa, P.C.; Fernanda Bahia, M. Walnut (Juglans regia) leaf extracts are strong scavengers of pro-oxidant reactive species. Food Chem. 2008, 106, 1014–1020. [Google Scholar] [CrossRef]
- Ribeiro, A.B.; Berto, A.; Chisté, R.C.; Freitas, M.; Visentainer, J.V.; Fernandes, E. Bioactive compounds and scavenging capacity of extracts from different parts of Vismia cauliflora against reactive oxygen and nitrogen species. Pharm. Biol. 2015, 53, 1267–1276. [Google Scholar] [CrossRef]
- Freitas, M.; Lima, J.L.F.C.; Fernandes, E. Optical Probes for Detection and Quantification of Neutrophils’ Oxidative Burst. A Review. Anal. Chim. Acta 2009, 649, 8–23. [Google Scholar] [CrossRef]
- Aruoma, O.I. Scavenging of hypochlorous acid by carvedilol and ebselen in vitro. Gen. Pharmacol. 1997, 28, 269–272. [Google Scholar] [CrossRef]
- Almeida, I.F.; Fernandes, E. Protective Effect of Castanea Sativa and Quercus Robur Leaf Extracts against Oxygen and Nitrogen Reactive Species. J. Photochem. Photobiol. B Biol. 2008, 91, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Sen, S.; De, B.; Devanna, N.; Chalcraborty, R. Total phenolic, total flavonoid content, and antioxidant capacity of the leaves of Meyna spinosa Roxb., an Indian medicinal plant. Chin. J. Nat. Med. 2013, 11, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Gülçin, I.; Mshvildadze, V.; Gepdiremen, A.; Elias, R. Screening of antiradical and antioxidant activity of monodesmosides and crude extract from Leontice smirnowii tuber. Phytomedicine 2006, 13, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Pistón, M.; Machado, I.; Branco, C.S.; Cesio, V.; Heinzen, H.; Ribeiro, D.; Fernandes, E.; Chisté, R.C.; Freitas, M. Infusion, Decoction and Hydroalcoholic Extracts of Leaves from Artichoke (Cynara Cardunculus L. Subsp. Cardunculus) Are Effective Scavengers of Physiologically Relevant ROS and RNS. Food Res. Int. 2014, 64, 150–156. [Google Scholar] [CrossRef]
- Boeing, J.S.; Ribeiro, D.; Chisté, R.C.; Visentainer, J.V.; Costa, V.M.; Freitas, M.; Fernandes, E. Chemical characterization and protective effect of the Bactris Setosa Mart. fruit against oxidative/nitrosative stress. Food Chem. 2017, 220, 427–437. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Pan, J.H.; Kim, S.H.; Lee, J.H.; Park, J.W. Chlorogenic acid ameliorates alcohol-induced liver injuries through scavenging reactive oxygen species. Biochimie 2018, 150, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Kavi Rajan, R.; Hussein, M.Z.; Fakurazi, S.; Yusoff, K.; Masarudin, M.J. Increased ROS scavenging and antioxidant efficiency of chlorogenic acid compound delivered via a chitosan nanoparticulate system for efficient in vitro visualization and accumulation in human renal adenocarcinoma cells. Int. J. Mol. Sci. 2019, 20, 4667. [Google Scholar] [CrossRef]
- Ferrer-Sueta, G.; Radi, R. Chemical biology of peroxynitrite: Kinetics, diffusion, and radicals. ACS Chem. Biol. 2009, 4, 161–177. [Google Scholar] [CrossRef]
- Koppenol, W.H.; Bounds, P.L.; Nauser, T.; Kissner, R.; Rüegger, H. Peroxynitrous acid: Controversy and consensus surrounding an enigmatic oxidant. Dalton Trans. 2012, 41, 13779–13787. [Google Scholar] [CrossRef]
- Beckman, J.S. The Physiological and Pathological Chemistry of Nitric Oxide. In Nitric Oxide: Principles and Actions; Lancaster, J., Jr., Ed.; Academic Press: Cambridge, MA, USA, 1996; pp. 1–82. [Google Scholar]
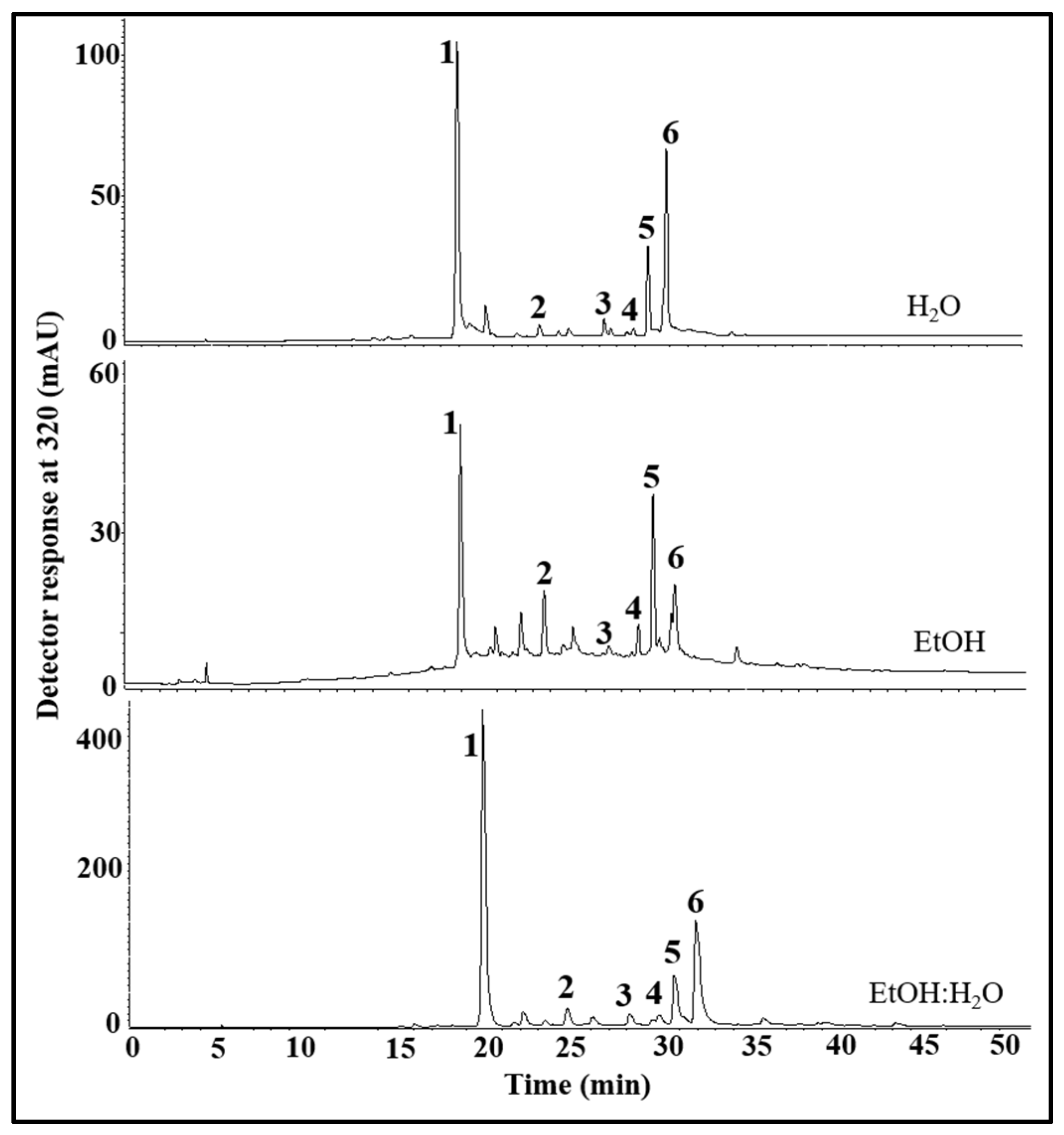
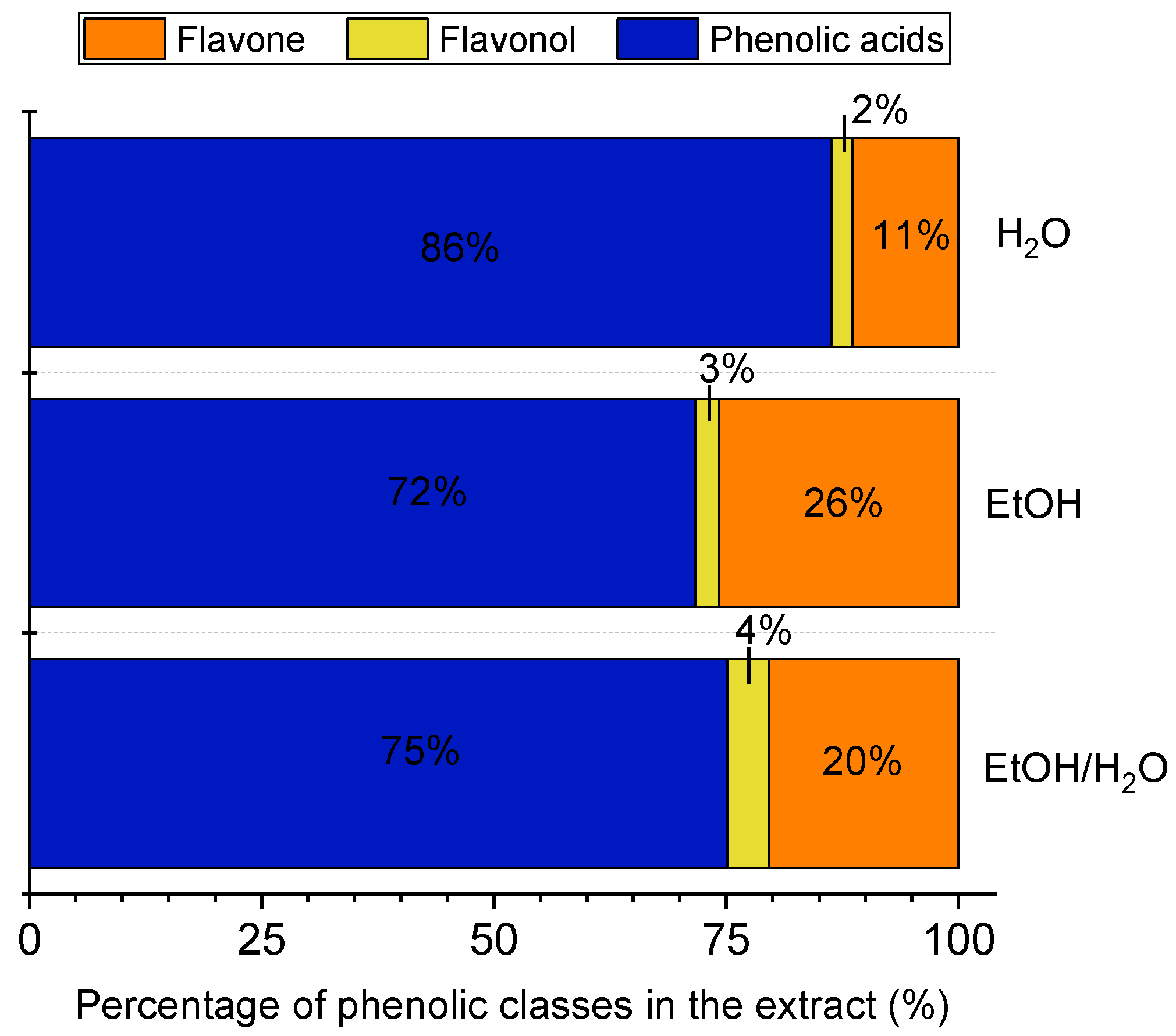
| Peaks | tR (min) 2 | λmax (nm) 3 | Phenolic Compound * | Concentration (μg/g Extract) 1 | ||
|---|---|---|---|---|---|---|
| EtOH/H2O | EtOH | H2O | ||||
| 1 | 18.5–19.1 | 300(sh), 326 | Chlorogenic acid 4 | 2198 ± 38 a | 506 ± 32 b | 1816 ± 160 a |
| 2 | 25.1–25.7 | 265, 326 | Feruloylquinic acid 4 | 169 ± 9 a | 120 ± 10 b | 121 ± 1 b |
| 3 | 27.2–27.7 | 354 | Quercetin glucuronide 5 | 258 ± 5 a | 28 ± 4 c | 77 ± 12 b |
| 4 | 28.6–28.8 | 265,300(sh), 347 | Luteolin hexoside 6 | 225 ± 74 b | 36 ± 3 c | 373 ± 52 a |
| 5 | 29.5–29.7 | 265, 347 | Luteolin glucoronide 6 | 956 ± 20 a | 248 ± 5 b | 17 ± 8 c |
| 6 | 30.9–31.1 | 285(sh), 330 | Ferulic acid derivative 4 | 1976 ± 43 a | 166 ± 5 c | 1012 ± 115 b |
| Sum of compounds | 5781 ± 191 a | 1104 ± 50 c | 3416 ± 348 b | |||
| Extract | IC50 (μg/mL) 1 | ||||
|---|---|---|---|---|---|
| 1O2 | O2•− | HOCl | H2O2 | ROO (Ssample/STrolox) 2 | |
| EtOH/H2O | 76 ± 7 | 45 ± 3 b | 118 ± 9 b | 110 ± 2 c | 0.23 ± <0.01 d |
| EtOH | 50.0 * ± 0.3 | 87 ± 5 a | 222 ± 1 a | 279 ± 12 b | 1.75 ± <0.01 b |
| H2O | NA | 103 ± 3 a | 221 ± 4 a | 400 ± 3 a | 1.00 ± <0.01 c |
| Positive Control | |||||
| Chlorogenic acid | 2.1 ± 0.1 a | 20 ± 2 c | 1.2 ± 0.1 d | 100 ± 4 c | 25.00 ± <0.01 a |
| Quercetin | 1.9 ± 0.1 a | 12.9 ± 0.5 d | 14 ± 1 c | NA | 1.34 ± <0.07 e |
| Ascorbic acid | ND | ND | ND | 40 ± 5 d | ND |
| Trolox | ND | ND | ND | ND | 1 |
| Extract | IC50 (μg/mL) 1 | |
|---|---|---|
| Absence of NaHCO3 | Presence of NaHCO3 | |
| EtOH/H2O | 537 ± 1 b | 596 ± 4 b |
| EtOH | 663 ± 9 a | 704 ± 1 a |
| H2O | 675 ± 6 a | 783 ± 2 a |
| Positive control | ||
| Chlorogenic acid | 0.23 ± 0.01 c | 0.23 ± 0.01 c |
| Quercetin | 0.010 ± <0.002 d | 0.010 ± <0.006 d |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leitão, D.d.S.T.C.; Barbosa-Carvalho, A.P.P.; de Siqueira, F.C.; Sousa, R.P.e.; Lopes, A.S.; Chisté, R.C. Extracts of Eryngium foetidum Leaves from the Amazonia Were Efficient Scavengers of ROS and RNS. Antioxidants 2023, 12, 1112. https://doi.org/10.3390/antiox12051112
Leitão DdSTC, Barbosa-Carvalho APP, de Siqueira FC, Sousa RPe, Lopes AS, Chisté RC. Extracts of Eryngium foetidum Leaves from the Amazonia Were Efficient Scavengers of ROS and RNS. Antioxidants. 2023; 12(5):1112. https://doi.org/10.3390/antiox12051112
Chicago/Turabian StyleLeitão, Deusa do Socorro Teixeira Costa, Anna Paula Pereira Barbosa-Carvalho, Francilia Campos de Siqueira, Railson Pontes e Sousa, Alessandra Santos Lopes, and Renan Campos Chisté. 2023. "Extracts of Eryngium foetidum Leaves from the Amazonia Were Efficient Scavengers of ROS and RNS" Antioxidants 12, no. 5: 1112. https://doi.org/10.3390/antiox12051112
APA StyleLeitão, D. d. S. T. C., Barbosa-Carvalho, A. P. P., de Siqueira, F. C., Sousa, R. P. e., Lopes, A. S., & Chisté, R. C. (2023). Extracts of Eryngium foetidum Leaves from the Amazonia Were Efficient Scavengers of ROS and RNS. Antioxidants, 12(5), 1112. https://doi.org/10.3390/antiox12051112








