Reactive Oxygen Species and the Redox-Regulatory Network in Cold Stress Acclimation
Abstract
1. Plant Response to Cold Temperature
2. Cold Stress Experiments in the Laboratory
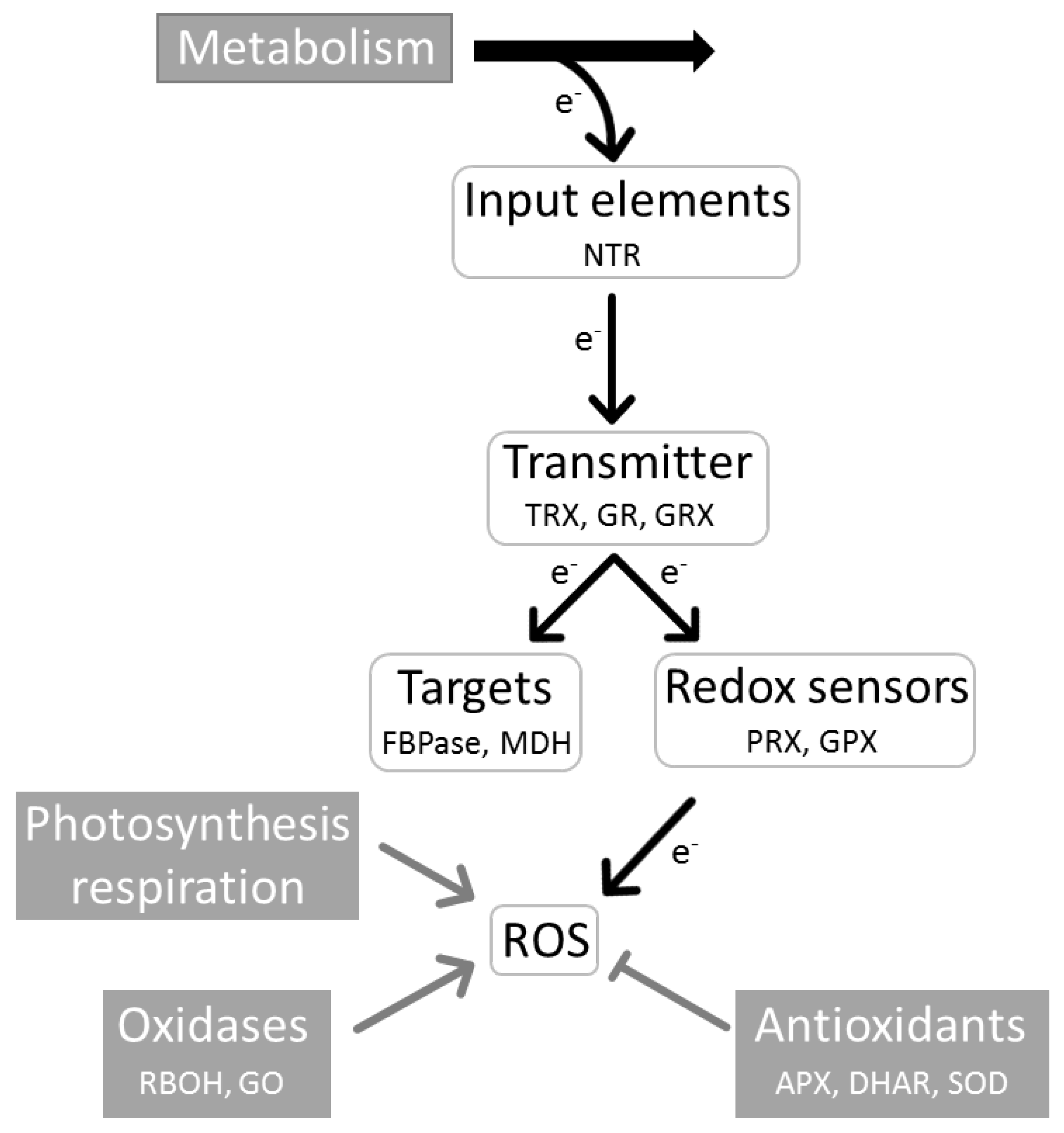
3. Central Role of the Redox Regulatory Network in Stress Acclimation
4. Variability of Cold Response Between Species
5. The Compartment-Specific Response of the Components of the Redox-Network to Cold in A. thaliana
6. Improvement of Cold Tolerance by Modulating the Redox-Network
7. Conclusions
- (1)
- Cold stress acclimation experiments often focus on leaves and photosynthetic metabolism. Response heterogeneity of different cell types has scarcely been addressed. Cell type-specific transcriptome, proteome, and metabolome analyses should reveal how other cell types respond to cold stress. But these approaches remain challenging and laborious.
- (2)
- Only a few methods allow researchers to address subcellular compartments. Transcriptome data provide easy access due to the predicted and often proven subcellular localization of the encoded gene products. This approach is straightforward and was applied here to the redox regulatory network. It would be interesting to see this type of data processing more frequently. However, the transcript amount is poorly linked to protein amount and activity. For a full understanding, we need compartment-specific proteomics and enzyme activity tests.
- (3)
- Metabolite-profiling of non-aqueous tissue fractions is another method which provides access to the major subcellular compartments. Non-aqueous fractions reflect the metabolic state of the compartments in vivo and are obtained from previously frozen and freeze-dried plant material like leaves [91]. This method was recently applied to cold-stress A. thaliana [92]. The latter study did not include metabolites with direct significance in the redox regulatory network.
- (4)
- Subcellular and cellular specificity can be addressed by imaging technologies detecting specific physicochemical properties such as Ca2+-activity, specific compounds or the redox state of the glutathione system by using roGFP coupled to GRX [93]. The roGFP:GRX sensor can be targeted to different cell compartments and should be used to explore the glutathione redox state in dependence on cold stress intensity and duration.
- (5)
- To describe the state of the redox network in subcellular compartments, mathematical modeling and simulation combined with redox-proteomics for validation will be required. A pioneering modeling study presented a simulation of the fluxes through the ascorbate-dependent water-water cycle [94] and most recently, the thioredoxin oxidase-dependent inactivation of chloroplast enzymes was simulated [23]. Conceptually, cold stress appears to be an interesting target for this kind of simulation and prediction.
- (6)
- The question of acclimation and damage during the cold period is certainly of significant interest. However, the costs of priming and the speed of recovery likely play a major role when it comes to fitness and competitiveness. Thus, the report by Juszczak et al. [6] deserves attention as it provides clues on the advantages and disadvantages of expressing a strong antioxidant system.
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Oakley, C.G.; Savage, L.; Lotz, S.; Larson, G.R.; Thomashow, M.F.; Kramer, D.M.; Schemske, D.W. Genetic basis of photosynthetic responses to cold in two locally adapted populations of Arabidopsis thaliana. J. Exp. Bot. 2018, 69, 699–709. [Google Scholar] [CrossRef] [PubMed]
- Thomashow, M.F. PLANT COLD ACCLIMATION: Freezing Tolerance Genes and Regulatory Mechanisms. Ann. Rev. Plant Physiol. Plant Mol. Biol. 1999, 50, 571–599. [Google Scholar] [CrossRef] [PubMed]
- Hayward, S.A.L.; Murray, P.A.; Gracey, A.Y.; Cossins, A.R. Beyond the Lipid Hypothesis. In Molecular Aspects of the Stress Response: Chaperones, Membranes and Networks; Csermely, P., Vígh, L., Eds.; Springer: New York, NY, USA, 2007; pp. 132–142. [Google Scholar]
- Li, F.; Bian, C.S.; Xu, J.F.; Pang, W.F.; Liu, J.; Duan, S.G.; Lei, Z.-G.; Jiwan, P.; Jin, L.-P. Cloning and functional characterization of SAD genes in potato. PloS ONE 2015, 10, e0122036. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.-G.; Yuan, L.-X.; Wang, Q.-L.; Ding, Z.-L.; Dong, C.-Y. Combined action of antioxidant defense system and osmolytes in chilling shock-induced chilling tolerance in Jatropha curcas seedlings. Acta Physiol. Plantarum 2013, 35, 2127–2136. [Google Scholar] [CrossRef]
- Juszczak, I.; Cvetkovic, J.; Zuther, E.; Hincha, D.K.; Baier, M. Natural Variation of Cold Deacclimation Correlates with Variation of Cold-Acclimation of the Plastid Antioxidant System in Arabidopsis thaliana Accessions. Front. Plant Sci. 2016, 7, 305. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Cai, J.; Liu, F.; Dai, T.; Cao, W.; Jiang, D. Cold priming drives the sub-cellular antioxidant systems to protect photosynthetic electron transport against subsequent low temperature stress in winter wheat. Plant Physiol. Biochem. PPB 2014, 82, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Kilian, J.; Whitehead, D.; Horak, J.; Wanke, D.; Weinl, S.; Batistic, O.; D’Angelo, C.; Bornberg-Bauer, E.; Kudla, J.; Harter, K. The AtGenExpress global stress expression data set: Protocols, evaluation and model data analysis of UV-B light, drought and cold stress responses. Plant J. 2007, 50, 347–363. [Google Scholar] [CrossRef] [PubMed]
- Wani, M.A.; Jan, N.; Qazi, H.A.; Andrabi, K.I.; John, R. Cold stress induces biochemical changes, fatty acid profile, antioxidant system and gene expression in Capsella bursa pastoris L. Acta Physiol. Plantarum 2018, 40, 167. [Google Scholar] [CrossRef]
- Jan, N.; Majeed, U.; Andrabi, K.I.; John, R. Cold stress modulates osmolytes and antioxidant system in Calendula officinalis. Acta Physiol. Plantarum 2018, 40, 73. [Google Scholar] [CrossRef]
- Spanò, C.; Bottega, S.; Ruffini Castiglione, M.; Pedranzani, H.E. Antioxidant response to cold stress in two oil plants of the genus Jatropha. Plant Soil Environ. 2017, 63, 271–276. [Google Scholar] [CrossRef]
- Han, Q.-H.; Huang, B.; Ding, C.-B.; Zhang, Z.-W.; Chen, Y.-E.; Hu, C.; Zhou, L.-J.; Huang, Y.; Liao, J.-Q.; Yuan, S.; et al. Effects of Melatonin on Anti-oxidative Systems and Photosystem II in Cold-Stressed Rice Seedlings. Front. Plant Sci. 2017, 8, 785. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, D.; Hu, J.; Salas Fernandez, M.G. Genetic architecture of photosynthesis in Sorghum bicolor under non-stress and cold stress conditions. J. Exp. Bot. 2017, 68, 4545–4557. [Google Scholar] [CrossRef] [PubMed]
- Dietz, K.-J. Redox signal integration: From stimulus to networks and genes. Physiol. Plant 2008, 133, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Buchanan, B.B. The Path to Thioredoxin and Redox Regulation in Chloroplasts. Ann. Rev. Plant Biol. 2016, 67, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Reichheld, J.-P.; Khafif, M.; Riondet, C.; Droux, M.; Bonnard, G.; Meyer, Y. Inactivation of thioredoxin reductases reveals a complex interplay between thioredoxin and glutathione pathways in Arabidopsis development. Plant Cell 2007, 19, 1851–1865. [Google Scholar] [CrossRef] [PubMed]
- Serrato, A.J.; Pérez-Ruiz, J.M.; Spínola, M.C.; Cejudo, F.J. A novel NADPH thioredoxin reductase, localized in the chloroplast, which deficiency causes hypersensitivity to abiotic stress in Arabidopsis thaliana. J. Biol. Chem. 2004, 279, 43821–43827. [Google Scholar] [CrossRef] [PubMed]
- Keryer, E.; Collin, V.; Lavergne, D.; Lemaire, S.; Issakidis-Bourguet, E. Characterization of Arabidopsis Mutants for the Variable Subunit of Ferredoxin:thioredoxin Reductase. Photosynthes. Res. 2004, 79, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Okegawa, Y.; Motohashi, K. Chloroplastic thioredoxin m functions as a major regulator of Calvin cycle enzymes during photosynthesis in vivo. Plant J. 2015, 84, 900–913. [Google Scholar] [CrossRef] [PubMed]
- Raines, C.A. The Calvin cycle revisited. Photosynthes. Res. 2003, 75, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Flohe, L. The impact of thiol peroxidases on redox regulation. Free Rad. Res. 2016, 50, 126–142. [Google Scholar] [CrossRef] [PubMed]
- Dietz, K.-J. Thiol-Based Peroxidases and Ascorbate Peroxidases: Why Plants Rely on Multiple Peroxidase Systems in the Photosynthesizing Chloroplast? Mol. Cells 2016, 39, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Vaseghi, M.-J.; Chibani, K.; Telman, W.; Liebthal, M.F.; Gerken, M.; Schnitzer, H.; Mueller, S.M.; Dietz, K.-J. The chloroplast 2-cysteine peroxiredoxin functions as thioredoxin oxidase in redox regulation of chloroplast metabolism. eLife 2018, 7. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.S.; Dietz, K.-J. Tuning of Redox Regulatory Mechanisms, Reactive Oxygen Species and Redox Homeostasis under Salinity Stress. Front. Plant Sci. 2016, 7, 548. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Ye, T.; Zhong, B.; Liu, X.; Chan, Z. Comparative proteomic and metabolomic analyses reveal mechanisms of improved cold stress tolerance in bermudagrass (Cynodon dactylon (L.) Pers.) by exogenous calcium. J. Integrat. Plant Biol. 2014, 56, 1064–1079. [Google Scholar] [CrossRef] [PubMed]
- Asada, K. THE WATER-WATER CYCLE IN CHLOROPLASTS: Scavenging of Active Oxygens and Dissipation of Excess Photons. Ann. Rev. Plant Physiol. Plant Mol. Biol. 1999, 50, 601–639. [Google Scholar] [CrossRef] [PubMed]
- Foyer, C.H.; Shigeoka, S. Understanding oxidative stress and antioxidant functions to enhance photosynthesis. Plant Physiol. 2011, 155, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.S.; ElSayed, A.I.; Moore, M.; Dietz, K.-J. Redox and Reactive Oxygen Species Network in Acclimation for Salinity Tolerance in Sugar Beet. J. Exp. Bot. 2017, 68, 1283–1298. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.-P.; Zhang, Q.-Y.; Tang, Z.-C.; Su, W.-A.; Sun, W.-N. Comparative proteomic analysis provides new insights into chilling stress responses in rice. Mol. Cell. Proteom. MCP 2006, 5, 484–496. [Google Scholar] [CrossRef] [PubMed]
- Soliman, M.H.; Alayafi, A.A.M.; El Kelish, A.A.; Abu-Elsaoud, A.M. Acetylsalicylic acid enhance tolerance of Phaseolus vulgaris L. to chilling stress, improving photosynthesis, antioxidants and expression of cold stress responsive genes. Botan. Stud. 2018, 59, 6. [Google Scholar] [CrossRef] [PubMed]
- Mir, B.A.; Mir, S.A.; Khazir, J.; Tonfack, L.B.; Cowan, D.A.; Vyas, D.; Koul, S. Cold stress affects antioxidative response and accumulation of medicinally important withanolides in Withania somnifera (L.) Dunal. Ind. Crops Prod. 2015, 74, 1008–1016. [Google Scholar] [CrossRef]
- Wang, X.; Fang, G.; Li, Y.; Ding, M.; Gong, H.; Li, Y. Differential antioxidant responses to cold stress in cell suspension cultures of two subspecies of rice. Plant Cell Tiss Organ Cult 2013, 113, 353–361. [Google Scholar] [CrossRef]
- Glaszmann, J.C.; Kaw, R.N.; Khush, G.S. Genetic divergence among cold tolerant rices (Oryza sativa L.). Euphytica 1990, 45, 95–104. [Google Scholar] [CrossRef]
- Cheng, F.; Lu, J.; Gao, M.; Shi, K.; Kong, Q.; Huang, Y.; Bie, Z. Redox Signaling and CBF-Responsive Pathway Are Involved in Salicylic Acid-Improved Photosynthesis and Growth under Chilling Stress in Watermelon. Front. Plant Sci. 2016, 7, 1519. [Google Scholar] [CrossRef] [PubMed]
- Dietz, K.J.; Horling, F.; Konig, J.; Baier, M. The function of the chloroplast 2-cysteine peroxiredoxin in peroxide detoxification and its regulation. J. Exp. Bot. 2002, 53, 1321–1329. [Google Scholar]
- Dietz, K.-J. Peroxiredoxins in plants and cyanobacteria. Antioxid. Redox Signal. 2011, 15, 1129–1159. [Google Scholar] [CrossRef] [PubMed]
- Dat, J.; Vandenabeele, S.; Vranova, E.; van Montagu, M.; Inze, D.; van Breusegem, F. Dual action of the active oxygen species during plant stress responses. Cell. Mol. Life Sci. 2000, 57, 779–795. [Google Scholar] [CrossRef] [PubMed]
- Cavalcanti, F.R.; Oliveira, J.T.A.; Martins-Miranda, A.S.; Viégas, R.A.; Silveira, J.A.G. Superoxide dismutase, catalase and peroxidase activities do not confer protection against oxidative damage in salt-stressed cowpea leaves. New Phytol. 2004, 163, 563–571. [Google Scholar] [CrossRef]
- Baier, M.; Pitsch, N.T.; Mellenthin, M.; Guo, W. Regulation of Genes Encoding Chloroplast Antioxidant Enzymes in Comparison to Regulation of the Extra-plastidic Antioxidant Defense System. In Ascorbate-Glutathione Pathway and Stress Tolerance in Plants; Anjum, N.A., Chan, M.-T., Umar, S., Eds.; Springer Netherlands: Dordrecht, The Netherland, 2010. [Google Scholar]
- Muthuramalingam, M.; Matros, A.; Scheibe, R.; Mock, H.-P.; Dietz, K.-J. The hydrogen peroxide-sensitive proteome of the chloroplast in vitro and in vivo. Front Plant Sci. 2013, 4, 54. [Google Scholar] [CrossRef] [PubMed]
- Koç, I.; Yuksel, I.; Caetano-Anollés, G. Metabolite-Centric Reporter Pathway and Tripartite Network Analysis of Arabidopsis Under Cold Stress. Front Bioeng. Biotechnol. 2018, 6, 121. [Google Scholar] [CrossRef] [PubMed]
- Kliebenstein, D.J.; Monde, R.-A.; Last, R.L. Superoxide Dismutase in Arabidopsis: An Eclectic Enzyme Family with Disparate Regulation and Protein Localization. Plant Physiol. 1998, 118, 637–650. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.-H.; Kuo, W.-Y.; Weiss, C.; Jinn, T.-L. Copper Chaperone-Dependent and -Independent Activation of Three Copper-Zinc Superoxide Dismutase Homologs Localized in Different Cellular Compartments in Arabidopsis. Plant Physiol. 2012, 158, 737–746. [Google Scholar] [CrossRef] [PubMed]
- Panchuk, I.I.; Volkov, R.A.; Schöffl, F. Heat Stress- and Heat Shock Transcription Factor-Dependent Expression and Activity of Ascorbate Peroxidase in Arabidopsis. Plant Physiol. 2002, 129, 838–853. [Google Scholar] [CrossRef] [PubMed]
- Mittler, R.; Poulos, T.L. Ascorbate Peroxidase. In Antioxidants and Reactive Oxygen Species in Plants; Wiley-Blackwell: Hoboken, NJ, USA, 2007; pp. 87–100. [Google Scholar]
- Lisenbee, C.S.; Lingard, M.J.; Trelease, R.N. Arabidopsis peroxisomes possess functionally redundant membrane and matrix isoforms of monodehydroascorbate reductase. Plant J. 2005, 43, 900–914. [Google Scholar] [CrossRef] [PubMed]
- Obara, K.; Sumi, K.; Fukuda, H. The Use of Multiple Transcription Starts Causes the Dual Targeting of Arabidopsis Putative Monodehydroascorbate Reductase to Both Mitochondria and Chloroplasts. Plant Cell Physiol. 2002, 43, 697–705. [Google Scholar] [CrossRef] [PubMed]
- Chew, O.; Whelan, J.; Millar, A.H. Molecular definition of the ascorbate-glutathione cycle in Arabidopsis mitochondria reveals dual targeting of antioxidant defenses in plants. J. Biol. Chem. 2003, 278, 46869–46877. [Google Scholar] [CrossRef] [PubMed]
- Reumann, S.; Quan, S.; Aung, K.; Yang, P.; Manandhar-Shrestha, K.; Holbrook, D.; Linka, N.; Switzenberg, R.; Wilkerson, C.G.; Weber, A.P.M.; et al. In-depth proteome analysis of Arabidopsis leaf peroxisomes combined with in vivo subcellular targeting verification indicates novel metabolic and regulatory functions of peroxisomes. Plant Physiol. 2009, 150, 125–143. [Google Scholar] [CrossRef] [PubMed]
- Dixon, D.P.; Davis, B.G.; Edwards, R. Functional divergence in the glutathione transferase superfamily in plants. Identification of two classes with putative functions in redox homeostasis in Arabidopsis thaliana. J. Biol. Chem. 2002, 277, 30859–30869. [Google Scholar] [CrossRef] [PubMed]
- Trivedi, D.K.; Gill, S.S.; Yadav, S.; Tuteja, N. Genome-wide analysis of glutathione reductase (GR) genes from rice and Arabidopsis. Plant Signal. Behav. 2012, 8, e23021. [Google Scholar] [CrossRef] [PubMed]
- Mhamdi, A.; Queval, G.; Chaouch, S.; Vanderauwera, S.; van Breusegem, F.; Noctor, G. Catalase function in plants: A focus on Arabidopsis mutants as stress-mimic models. J. Exp. Bot. 2010, 61, 4197–4220. [Google Scholar] [CrossRef] [PubMed]
- Milla, M.A.R.; Maurer, A.; Huete, A.R.; Gustafson, J.P. Glutathione peroxidase genes in Arabidopsis are ubiquitous and regulated by abiotic stresses through diverse signaling pathways. Plant J. 2003, 36, 602–615. [Google Scholar] [CrossRef]
- Sagi, M.; Fluhr, R. Superoxide Production by Plant Homologues of the gp91phox NADPH Oxidase. Modulation of Activity by Calcium and by Tobacco Mosaic Virus Infection. Plant Physiol. 2001, 126, 1281–1290. [Google Scholar] [CrossRef] [PubMed]
- Heazlewood, J.L.; Tonti-Filippini, J.S.; Gout, A.M.; Day, D.A.; Whelan, J.; Millar, A.H. Experimental analysis of the Arabidopsis mitochondrial proteome highlights signaling and regulatory components, provides assessment of targeting prediction programs, and indicates plant-specific mitochondrial proteins. Plant Cell Online 2004, 16, 241–256. [Google Scholar] [CrossRef] [PubMed]
- Konert, G.; Trotta, A.; Kouvonen, P.; Rahikainen, M.; Durian, G.; Blokhina, O.; Fagerstedt, K.; Muth, D.; Corthals, G.L.; Kangasjärvi, S. Protein phosphatase 2A (PP2A) regulatory subunit B′γ interacts with cytoplasmic ACONITASE 3 and modulates the abundance of AOX1A and AOX1D in Arabidopsis thaliana. New Phytol. 2014, 205, 1250–1263. [Google Scholar] [CrossRef] [PubMed]
- Saisho, D.; Nakazono, M.; Lee, K.-H.; Tsutsumi, N.; Akita, S.; Hirai, A. The gene for alternative oxidase-2 (AOX2) from Arabidopsis thaliana consists of five exons unlike other AOX genes and is transcribed at an early stage during germination. Genes Genet. Syst. 2001, 76, 89–97. [Google Scholar] [CrossRef]
- Lennon, A.M.; Prommeenate, P.; Nixon, P.J. Location, expression and orientation of the putative chlororespiratory enzymes, Ndh and IMMUTANS, in higher-plant plastids. Planta 2003, 218, 254–260. [Google Scholar] [CrossRef] [PubMed]
- Haslekås, C.; Viken, M.K.; Grini, P.E.; Nygaard, V.; Nordgard, S.H.; Meza, T.J.; Aalen, R.B. Seed 1-Cysteine Peroxiredoxin Antioxidants Are Not Involved in Dormancy, But Contribute to Inhibition of Germination during Stress. Plant Physiol. 2003, 133, 1148–1157. [Google Scholar] [CrossRef] [PubMed]
- König, J.; Baier, M.; Horling, F.; Kahmann, U.; Harris, G.; Schürmann, P.; Dietz, K.-J. The plant-specific function of 2-Cys peroxiredoxin-mediated detoxification of peroxides in the redox-hierarchy of photosynthetic electron flux. Proc. Natl. Acad. Sci. USA 2002, 99, 5738–5743. [Google Scholar] [CrossRef] [PubMed]
- Lamkemeyer, P.; Laxa, M.; Collin, V.; Li, W.; Finkemeier, I.; Schöttler, M.A.; Holtkamp, V.; Tognetti, V.B.; Issakidis-Bourguet, E.; Kandlbinder, A.; et al. Peroxiredoxin Q of Arabidopsis thaliana is attached to the thylakoids and functions in context of photosynthesis†. Plant J. 2006, 45, 968–981. [Google Scholar] [CrossRef] [PubMed]
- Bréhélin, C.; Meyer, E.H.; de Souris, J.-P.; Bonnard, G.; Meyer, Y. Resemblance and Dissemblance of Arabidopsis Type II Peroxiredoxins: Similar Sequences for Divergent Gene Expression, Protein Localization, and Activity. Plant Physiol. 2003, 132, 2045–2057. [Google Scholar] [CrossRef] [PubMed]
- Finkemeier, I.; Goodman, M.; Lamkemeyer, P.; Kandlbinder, A.; Sweetlove, L.J.; Dietz, K.-J. The mitochondrial type II peroxiredoxin F is essential for redox homeostasis and root growth of Arabidopsis thaliana under stress. J. Biol. Chem. 2005, 280, 12168–12180. [Google Scholar] [CrossRef] [PubMed]
- Meyer, Y.; Siala, W.; Bashandy, T.; Riondet, C.; Vignols, F.; Reichheld, J.P. Glutaredoxins and thioredoxins in plants. Biochim. Biophys. Acta 2008, 1783, 589–600. [Google Scholar] [CrossRef] [PubMed]
- Reichheld, J.-P.; Meyer, E.; Khafif, M.; Bonnard, G.; Meyer, Y. AtNTRB is the major mitochondrial thioredoxin reductase in Arabidopsis thaliana. FEBS Lett. 2005, 579, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Dangoor, I.; Peled-Zehavi, H.; Levitan, A.; Pasand, O.; Danon, A. A small family of chloroplast atypical thioredoxins. Plant Physiol. 2009, 149, 1240–1250. [Google Scholar] [CrossRef] [PubMed]
- Lemaire, S.D. The Glutaredoxin Family in Oxygenic Photosynthetic Organisms. Photosynthes. Res. 2004, 79, 305–318. [Google Scholar] [CrossRef] [PubMed]
- Cheng, N.-H. AtGRX4, an Arabidopsis chloroplastic monothiol glutaredoxin, is able to suppress yeast grx5 mutant phenotypes and respond to oxidative stress. FEBS Lett. 2008, 582, 848–854. [Google Scholar] [CrossRef] [PubMed]
- Cheng, N.-H.; Liu, J.-Z.; Brock, A.; Nelson, R.S.; Hirschi, K.D. AtGRXcp, an Arabidopsis chloroplastic glutaredoxin, is critical for protection against protein oxidative damage. J. Biol. Chem. 2006, 281, 26280–26288. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Liu, S.; Feng, Y.; Liu, J.-Z.; Chen, Y.; Pham, K.; Deng, H.; Hirschi, K.D.; Wang, X.; Cheng, N. Structural insights into the N-terminal GIY-YIG endonuclease activity of Arabidopsis glutaredoxin AtGRXS16 in chloroplasts. Proc. Natl. Acad. Sci. USA 2013, 110, 9565–9570. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Lauri, A.; Ziemann, M.; Busch, A.; Bhave, M.; Zachgo, S. Nuclear activity of ROXY1, a glutaredoxin interacting with TGA factors, is required for petal development in Arabidopsis thaliana. Plant Cell Online 2009, 21, 429–441. [Google Scholar] [CrossRef] [PubMed]
- Murmu, J.; Bush, M.J.; DeLong, C.; Li, S.; Xu, M.; Khan, M.; Malcolmson, C.; Fobert, P.R.; Zachgo, S.; Hepworth, S.R. Arabidopsis basic leucine-zipper transcription factors TGA9 and TGA10 interact with floral glutaredoxins ROXY1 and ROXY2 and are redundantly required for anther development. Plant Physiol. 2010, 154, 1492–1504. [Google Scholar] [CrossRef] [PubMed]
- König, J.; Muthuramalingam, M.; Dietz, K.-J. Mechanisms and dynamics in the thiol/disulfide redox regulatory network: Transmitters, sensors and targets. Curr. Opin. Plant Biol. 2012, 15, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Meyer, Y.; Buchanan, B.B.; Vignols, F.; Reichheld, J.-P. Thioredoxins and Glutaredoxins: Unifying Elements in Redox Biology. Ann. Rev. Genet. 2009, 43, 335–367. [Google Scholar] [CrossRef] [PubMed]
- Shafi, A.; Dogra, V.; Gill, T.; Ahuja, P.S.; Sreenivasulu, Y. Simultaneous over-expression of PaSOD and RaAPX in transgenic Arabidopsis thaliana confers cold stress tolerance through increase in vascular lignifications. PloS one 2014, 9, e110302. [Google Scholar] [CrossRef] [PubMed]
- Schwanhäusser, B.; Busse, D.; Li, N.; Dittmar, G.; Schuchhardt, J.; Wolf, J.; Chen, W.; Selbach, M. Global quantification of mammalian gene expression control. Nature 2011, 473, 337. [Google Scholar] [CrossRef] [PubMed]
- Mittler, R.; Vanderauwera, S.; Gollery, M.; van Breusegem, F. Reactive oxygen gene network of plants. Trends Plant Sci. 2004, 9, 490–498. [Google Scholar] [CrossRef] [PubMed]
- Apel, K.; Hirt, H. REACTIVE OXYGEN SPECIES: Metabolism, Oxidative Stress, and Signal Transduction. Ann. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef] [PubMed]
- Miyake, C.; Michihata, F.; Asada, K. Scavenging of Hydrogen Peroxide in Prokaryotic and Eukaryotic Algae: Acquisition of Ascorbate Peroxidase during the Evolution of Cyanobacteria. Plant Cell Physiol. 1991, 32, 33–43. [Google Scholar] [CrossRef]
- Juszczak, I.; Rudnik, R.; Pietzenuk, B.; Baier, M. Natural genetic variation in the expression regulation of the chloroplast antioxidant system among Arabidopsis thaliana accessions. Physiol. Plantarum 2012, 146, 53–70. [Google Scholar] [CrossRef] [PubMed]
- Driever, S.M.; Baker, N.R. The water-water cycle in leaves is not a major alternative electron sink for dissipation of excess excitation energy when CO(2) assimilation is restricted. Plant Cell Environ. 2011, 34, 837–846. [Google Scholar] [CrossRef] [PubMed]
- Liebthal, M.; Maynard, D.; Dietz, K.-J. Peroxiredoxins and Redox Signaling in Plants. Antioxid. Redox Signal. 2018, 28, 609–624. [Google Scholar] [CrossRef] [PubMed]
- Karpinski, S.; Reynolds, H.; Karpinska, B.; Wingsle, G.; Creissen, G.; Mullineaux, P. Systemic Signaling and Acclimation in Response to Excess Excitation Energy in Arabidopsis. Science 1999, 284, 654–657. [Google Scholar] [CrossRef] [PubMed]
- Krause, G.H.; Somersalo, S.; Osmond, C.B.; Briantais, J.-M.; Schreiber, U. Fluorescence as a Tool in Photosynthesis Research: Application in Studies of Photoinhibition, Cold Acclimation and Freezing Stress [and Discussion]. Philosoph. Trans. Royal Soc. London B: Biol. Sci. 1989, 323, 281–293. [Google Scholar] [CrossRef]
- Duan, M.; Feng, H.-L.; Wang, L.-Y.; Li, D.; Meng, Q.-W. Overexpression of thylakoidal ascorbate peroxidase shows enhanced resistance to chilling stress in tomato. J. Plant Physiol. 2012, 169, 867–877. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Wu, Q.; Sprague, S.A.; Park, J.; Oh, M.; Rajashekar, C.B.; Koiwa, H.; Nakata, P.A.; Cheng, N.; Hirschi, K.D.; et al. Tomato expressing Arabidopsis glutaredoxin gene AtGRXS17 confers tolerance to chilling stress via modulating cold responsive components. Horticul. Res. 2015, 2, 15051. [Google Scholar] [CrossRef] [PubMed]
- Moon, J.C.; Lee, S.; Shin, S.Y.; Chae, H.B.; Jung, Y.J.; Jung, H.S.; Lee, K.O.; Lee, J.R.; Lee, S.Y. Overexpression of Arabidopsis NADPH-dependent thioredoxin reductase C (AtNTRC) confers freezing and cold shock tolerance to plants. Biochem. Biophys. Res. Commun. 2015, 463, 1225–1229. [Google Scholar] [CrossRef] [PubMed]
- Maruta, T.; Tanouchi, A.; Tamoi, M.; Yabuta, Y.; Yoshimura, K.; Ishikawa, T.; Shigeoka, S. Arabidopsis Chloroplastic Ascorbate Peroxidase Isoenzymes Play a Dual Role in Photoprotection and Gene Regulation under Photooxidative Stress. Plant Cell Physiol. 2010, 51, 190–200. [Google Scholar] [CrossRef] [PubMed]
- Cheng, N.-H.; Liu, J.-Z.; Liu, X.; Wu, Q.; Thompson, S.M.; Lin, J.; Chang, J.; Whitham, S.A.; Park, S.; Cohen, J.D.; et al. Arabidopsis monothiol glutaredoxin, AtGRXS17, is critical for temperature-dependent postembryonic growth and development via modulating auxin response. J. Biol. Chem. 2011, 286, 20398–20406. [Google Scholar] [CrossRef] [PubMed]
- Baier, M.; Bittner, A.; Prescher, A.; van Buer, J. Preparing plants for improved cold tolerance by priming. Plant Cell Environ. 2018. [Google Scholar] [CrossRef] [PubMed]
- Dietz, K.-J. Subcellular metabolomics: The choice of method depends on the aim of the study. J. Exp. Bot. 2017, 68, 5695–5698. [Google Scholar] [CrossRef] [PubMed]
- Hoermiller, I.I.; Naegele, T.; Augustin, H.; Stutz, S.; Weckwerth, W.; Heyer, A.G. Subcellular reprogramming of metabolism during cold acclimation in Arabidopsis thaliana. Plant Cell Environ. 2017, 40, 602–610. [Google Scholar] [CrossRef] [PubMed]
- Meyer, A.J.; Brach, T.; Marty, L.; Kreye, S.; Rouhier, N.; Jacquot, J.-P.; Hell, R. Redox-sensitive GFP in Arabidopsis thaliana is a quantitative biosensor for the redox potential of the cellular glutathione redox buffer. Plant J. 2007, 52, 973–986. [Google Scholar] [CrossRef] [PubMed]
- Polle, A. Dissecting the Superoxide Dismutase-Ascorbate-Glutathione-Pathway in Chloroplasts by Metabolic Modeling. Computer Simulations as a Step towards Flux Analysis. Plant Physiol. 2001, 126, 445–462. [Google Scholar] [CrossRef] [PubMed]

| Species | Plant Age | Growth Condition | Cold Treatment | Other Comments | Reference | |
|---|---|---|---|---|---|---|
| duration | temperature | |||||
| Arabidopsis thaliana | 16 days | 16/8 h light/dark 24 °C | 0; 0.5; 1; 3; 6; 12; 24 h | 4 °C | Decreased light intensity during cold stress treatment | [8] |
| Arabidopsis thaliana | 42 days | 16/8 h light/dark 20 °C/18 °C | 14 days | 4 °C | Decreased light intensity during cold stress Additional 1,2, and 3 days of deacclimation | [6] |
| Capsella bursa pastoris L. | 30 days | 16/8 h light/dark 25 °C | 24; 48; 72; 96; 120 h | 10 °C | [9] | |
| Calendula officinalis | 14 days | 16/8 h light/dark 25 ± 2 °C | 24; 48; 72; 96; 120 h | 4 °C | [10] | |
| Jatropha curcas | 45 days | 14/10 h light/dark 28 °C | 48 h | 4 °C | Partially pretreated at 15 °C for five days Cold sensitive Jatropha | [11] |
| Jatropha macrocarpa | 45 days | 14/10 h light/dark 28 °C | 48 h | 4 °C | Partially pretreated at 15 °C for 5 days, cold tolerant Jatropha | [11] |
| Oryza sativa | 14 days | 14/10 h light/dark 28 °C/22 °C | 6 days | 12 °C | 2 days pretreatment with melatonin | [12] |
| Sorghum bicolor | 30 days | 16/8 h light/dark 28 °C/24 °C | 6 days | 15 °C | 5 days recovery | [13] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dreyer, A.; Dietz, K.-J. Reactive Oxygen Species and the Redox-Regulatory Network in Cold Stress Acclimation. Antioxidants 2018, 7, 169. https://doi.org/10.3390/antiox7110169
Dreyer A, Dietz K-J. Reactive Oxygen Species and the Redox-Regulatory Network in Cold Stress Acclimation. Antioxidants. 2018; 7(11):169. https://doi.org/10.3390/antiox7110169
Chicago/Turabian StyleDreyer, Anna, and Karl-Josef Dietz. 2018. "Reactive Oxygen Species and the Redox-Regulatory Network in Cold Stress Acclimation" Antioxidants 7, no. 11: 169. https://doi.org/10.3390/antiox7110169
APA StyleDreyer, A., & Dietz, K.-J. (2018). Reactive Oxygen Species and the Redox-Regulatory Network in Cold Stress Acclimation. Antioxidants, 7(11), 169. https://doi.org/10.3390/antiox7110169





