Intensification of Polyphenol Extraction from Olive Leaves Using Ired-Irrad®, an Environmentally-Friendly Innovative Technology
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.1.1. Dry Matter
2.1.2. Chemicals
2.2. Experimental Methods
2.2.1. Water Bath Extraction
2.2.2. Infrared-Assisted Extraction
2.2.3. Extraction Procedure
2.2.4. Total Phenolic Compounds
2.3. Experimental Design
2.4. High Performance Liquid Chromatography
2.5. Antioxidant Activity
2.6. Antiradical Activity
2.7. Antifungal Activity
2.8. Fungal Growth Inhibition
2.9. Aflatoxin B1 (AFB1) Inhibition
2.10. Antibacterial Activity
2.10.1. Microorganisms Used
2.10.2. Determination of Minimal Inhibitory Concentration for Extracts
2.11. Statistical Analysis
3. Results and Discussion
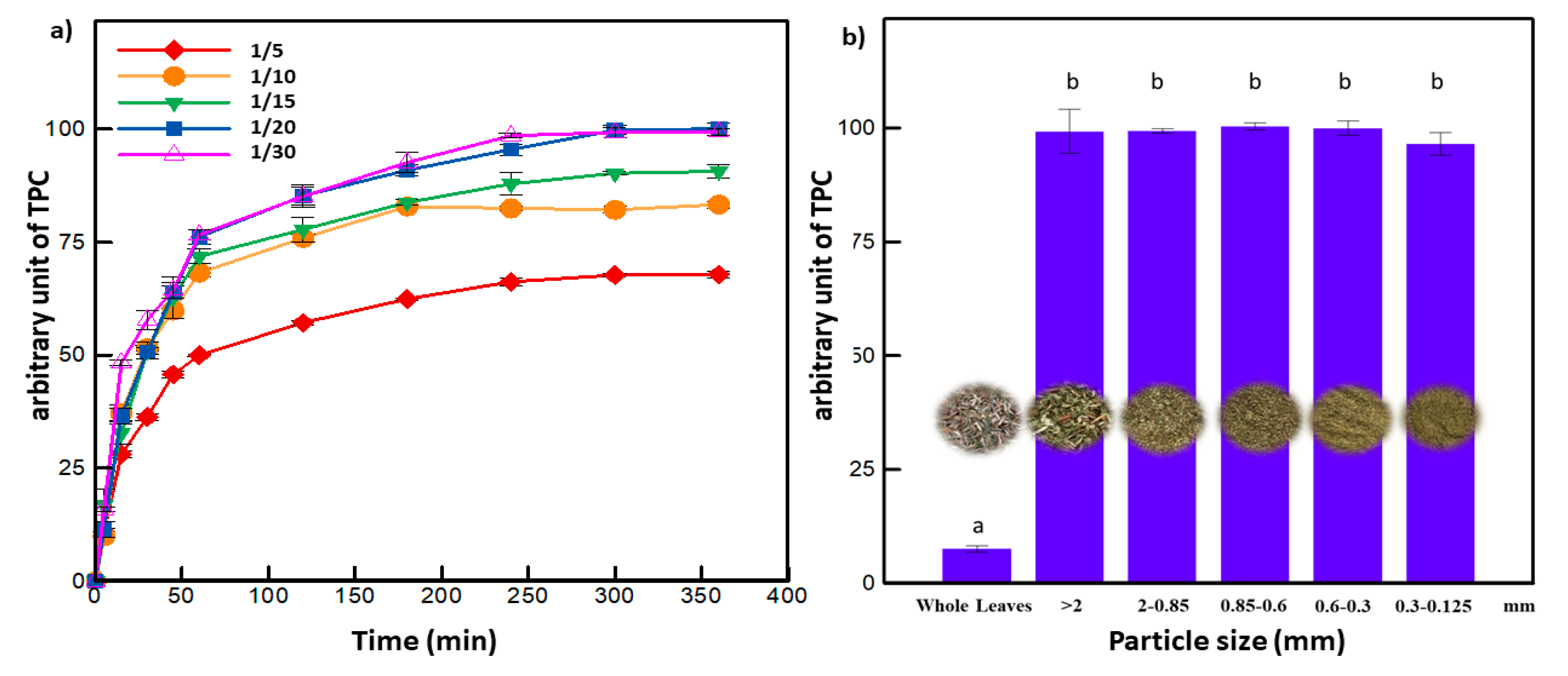
3.1. Solid to Liquid Ratio and Particle Size Selection
3.2. Effect of Solvent Mixture, Time, and Temperature on TPC Extraction
3.3. Optimization of TPC Extraction
3.4. High Performance Liquid Chromatography
3.5. Antioxidant and Antiradical Activity
3.6. Antifungal Activity
3.7. Antibacterial Activity
4. Conclusions
5. Patents
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ahmad-qasem, M.H.; Canovas, J.; Barrajon, E.; Carreres, J.E.; Micol, V.; Perez, J.V.G. Influence of olive leaf processing on the bioaccessibility of bioactive polyphenols. J. Agric. Food Chem. 2014, 62, 6190–6198. [Google Scholar] [CrossRef]
- Lapage, S.P.; Sneath, P.H.A.; Lessel, E.F.; Skerman, V.B.D.; Seeliger, H.P.R.; Clark, W.A. Foreword to the First Edition; FAO: Rome, Italy, 2005. [Google Scholar]
- Guinda, Á.; Castellano, J.M.; Delgado-hervás, T.; Gutiérrez-adánez, P.; Rada, M. Determination of major bioactive compounds from olive leaf. LWT Food Sci. Technol. 2015, 64, 431–438. [Google Scholar] [CrossRef]
- Solarte-Toro, J.C.; Romero-Garcia, J.M.; Lopez-Linares, J.C.; Ramos, E.R.; Castro, E.; Alzate, C.A.C. Simulation Approach Through the Biorefinery Concept of the Antioxidants, Lignin and Ethanol Production using Olive Leaves as Raw Material. Chem. Eng. Trans. 2018, 70, 925–930. [Google Scholar]
- Mkaouar, S.; Bahloul, N.; Gelicus, A.; Allaf, K.; Kechaou, N. Instant controlled pressure drop texturing for intensifying ethanol solvent extraction of olive (Olea europaea) leaf polyphenols. Sep. Purif. Technol. 2015, 145, 139–146. [Google Scholar] [CrossRef]
- Khemakhem, I.; Gargouri, O.D.; Dhouib, A.; Ayadi, M.A.; Bouaziz, M. Oleuropein rich extract from olive leaves by combining microfiltration, ultrafiltration and nanofiltration. Sep. Purif. Technol. 2017, 172, 310–317. [Google Scholar] [CrossRef]
- Romani, A.; Mulas, S.; Heimler, D. Polyphenols and secoiridoids in raw material (Olea europaea L. leaves) and commercial food supplements. Eur. Food Res. Technol. 2017, 243, 429–435. [Google Scholar] [CrossRef]
- Dedousi, M.; Mamoudaki, V.; Grigorakis, S.; Makris, D.P. Ultrasound-Assisted Extraction of Polyphenolic Antioxidants from Olive (Olea europaea) Leaves Using a Novel Glycerol/Sodium-Potassium Tartrate Low-Transition Temperature Mixture (LTTM). Environ. Artic. 2017, 4, 31. [Google Scholar] [CrossRef]
- Talhaoui, N.; Taamalli, A.; Mar, A. Phenolic compounds in olive leaves: Analytical determination, biotic and abiotic influence, and health benefit. Food Res. Int. 2015, 77, 92–108. [Google Scholar] [CrossRef]
- Stamatopoulos, K.; Katsoyannos, E.; Chatzilazarou, A. Antioxidant Activity and Thermal Stability of Oleuropein and Related Phenolic Compounds of Olive Leaf Extract after Separation and Concentration by Salting-Out-Assisted Cloud Point Extraction. Antioxidants 2014, 80, 229–244. [Google Scholar] [CrossRef]
- Mkaouar, S.; Gelicus, A.; Bahloul, N.; Allaf, K.; Kechaou, N. Kinetic study of polyphenols extraction from olive (Olea europaea L.) leaves using instant controlled pressure drop texturing. Sep. Purif. Technol. 2016, 161, 165–171. [Google Scholar] [CrossRef]
- Galanakis, C.M.; Kotsiou, K. Chapter 10—Recovery of Bioactive Compounds from Olive Mill Waste; Elsevier: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Deng, J.; Xu, Z.; Xiang, C.; Liu, J.; Zhou, L.; Li, T.; Yang, Z.; Ding, C. Comparative evaluation of maceration and ultrasonic-assisted extraction of phenolic compounds from fresh olives. Ultrason. Sonochem. 2017, 37, 328–334. [Google Scholar] [CrossRef]
- Rajha, H.N.; Mhanna, T.; El Kantar, S.; El Khoury, A.; Louka, N.; Maroun, R.G. Innovative process of polyphenol recovery from pomegranate peels by combining green deep eutectic solvents and a new infrared technology. Lwt 2019, 111, 138–146. [Google Scholar] [CrossRef]
- Ahmad-Qasem, M.H.; Barrajón-Catalán, E.; Micol, V.; Mulet, A.; García-Pérez, J.V. In fl uence of freezing and dehydration of olive leaves (var. Serrana) on extract composition and antioxidant potential. FRIN 2013, 50, 189–196. [Google Scholar]
- Japón-Luján, R.; Luque-Rodríguez, J.M.; de Castro, M.D.L. Multivariate optimisation of the microwave-assisted extraction of oleuropein and related biophenols from olive leaves. Anal. Bioanal. Chem. 2006, 385, 753–759. [Google Scholar] [CrossRef]
- Şahin, S.; Samli, R.; Tan, A.S.B.; Barba, F.J.; Chemat, F.; Cravotto, G.; Lorenzo, J.M. Solvent-free microwave-assisted extraction of polyphenols from olive tree leaves: Antioxidant and antimicrobial properties. Molecules 2017, 22, 1056. [Google Scholar] [CrossRef]
- Xynos, N.; Papaefstathiou, G.; Gikas, E.; Argyropoulou, A.; Aligiannis, N.; Skaltsounis, A. Design optimization study of the extraction of olive leaves performed with pressurized liquid extraction using response surface methodology. Sep. Purif. Technol. 2014, 122, 323–330. [Google Scholar] [CrossRef]
- de Lucas, A.; De, E.M.; Rinco, J. Supercritical fluid extraction of tocopherol concentrates from olive tree leaves. J. Supercrit. Fluids 2002, 22, 221–228. [Google Scholar] [CrossRef]
- Baldino, A.L.; della Porta, G.; Sesti, L.; Reverchon, E.; Adami, R. Concentrated Oleuropein Powder from Olive Leaves using Alcoholic Extraction. J. Supercrit. Fluids 2017, 133, 65–69. [Google Scholar] [CrossRef]
- Escobedo, R.; Miranda, R.; Martínez, J. Infrared irradiation: Toward green chemistry, a review. Int. J. Mol. Sci. 2016, 17, 453. [Google Scholar] [CrossRef]
- Rajha, H.N.; Debs, E.; Maroun, R.G.; Louka, N. Système d’extraction, de séparation ou de prétraitement assisté par rayonnement infrarouge. Adéquation entre les caractéristiques du rayonnement et celles de la matière traitée. Lebanese Patent 11296, 29 November 2017. [Google Scholar]
- Raafat, K.; El-Darra, N.; Saleh, F.A.; Rajha, H.N.; Maroun, R.G.; Louka, N. Infrared-Assisted Extraction and HPLC-Analysis of Prunus armeniaca L. Pomace and Detoxified-Kernel and their Antidiabetic Effects. Phytochem. Anal. 2018, 29, 156–167. [Google Scholar] [CrossRef]
- Cheaib, D.; El Darra, N.; Rajha, H.N.; El Ghazzawi, I.; Maroun, R.G.; Louka, N. Biological activity of apricot byproducts polyphenols using solid–liquid and infrared-assisted technology. J. Food Biochem. 2018, 42, 1–9. [Google Scholar]
- Cheaib, D.; El Darra, N.; Rajha, H.N.; El-Ghazzawi, I.; Mouneimne, Y.; Jammoul, A.; Maroun, R.G.; Louka, N. Study of the Selectivity and Bioactivity of Polyphenols Using Infrared Assisted Extraction from Apricot Pomace Compared to Conventional Methods. Antioxidants 2018, 7, 174. [Google Scholar] [CrossRef]
- Raafat, K.; El-Darra, N.; Saleh, F.A.; Rajha, H.N.; Louka, N. Optimization of infrared-assisted extraction of bioactive lactones from Saussurea lappa L. and their effects against gestational diabetes. Pharmacogn. Mag. 2019, 15, 208–218. [Google Scholar] [CrossRef]
- Cifá, D.; Skrt, M.; Pittia, P.; di Mattia, C.; Ulrih, N.P. Enhanced yield of oleuropein from olive leaves using ultrasound-assisted extraction. Food Sci. Nutr. 2018, 6, 1128–1137. [Google Scholar] [CrossRef]
- Singleton, V.L.; Lamuela-ravents, R.M. Analysis of Total Phenols and Other Oxidation Substrates and Antioxidants by Means of Folin-Ciocalteu Reagent. METHODS Enzym. 1999, 299, 152–178. [Google Scholar]
- Mehta, D.; Sharma, N.; Bansal, V.; Sangwan, R.S.; Yadav, S.K. Impact of ultrasonication, ultraviolet and atmospheric cold plasma processing on quality parameters of tomato-based beverage in comparison with thermal processing. Innov. Food Sci. Emerg. Technol. 2019, 52, 343–349. [Google Scholar] [CrossRef]
- Selin, C.; Zeynep, I. A novel approach for olive leaf extraction through ultrasound technology: Response surface methodology versus artificial neural networks. Korean J. Chem. Eng. 2014, 31, 1661–1667. [Google Scholar]
- Iban, E.; Zarrouk, M. Optimization of Microwave-Assisted Extraction for the Characterization of Olive Leaf Phenolic Compounds by Using HPLC-ESI-TOF-MS/IT-MS 2. J. Agric. Food Chem. 2012, 60, 791–798. [Google Scholar]
- Prieto, P.; Pineda, M.; Aguilar, M. Spectrophotometric Quantitation of Antioxidant Capacity through the Formation of a Phosphomolybdenum Complex: Specific Application to the Determination of Vitamin E 1. Anal. Biochem. 1999, 341, 337–341. [Google Scholar] [CrossRef]
- Kallithraka, S.; Mohdaly, A.A. Determination of major anthocyanin pigments in Hellenic native grape varieties (Vitis vinifera sp.): Association with antiradical activity. J. Food Compos. Anal. 2005, 18, 375–386. [Google Scholar] [CrossRef]
- Chanioti, S.; Tzia, C. Extraction of phenolic compounds from olive pomace by using natural deep eutectic solvents and innovative extraction techniques. Innov. Food Sci. Emerg. Technol. 2018, 49, 192–201. [Google Scholar] [CrossRef]
- C.L.S. Institute. Performance Standards for Antimicrobial Susceptibility Testing; C.L.S. Institute: Wayne, PA, USA, 2018. [Google Scholar]
- Stamatopoulos, K.; Chatzilazarou, A.; Katsoyannos, E. Optimization of Multistage Extraction of Olive Leaves for Recovery of Phenolic Compounds at Moderated Temperatures and Short Extraction Times. Foods 2014, 3, 66–81. [Google Scholar] [CrossRef]
- Gómez-Caravaca, A.M.; Verardo, V.; Segura-Carretero, A.; Fernández-Gutiérrez, A.; Caboni, M.F. Polyphenols in Plants: Isolation, Purification and Extract Preparation; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Richardson, P. Thermal Technologies in Food Processing; Springer: Berlin, Germany, 2000. [Google Scholar]
- Gorban, A.N.; Sargsyan, H.P.; Wahab, H.A. Quasichemical Models of Multicomponent Nonlinear Diffusion. Math. Model. Nat. Phenom. 2010, 6, 184–262. [Google Scholar] [CrossRef]
- Silva, E.M.; Rogez, H.; Larondelle, Y. Optimization of extraction of phenolics from Inga edulis leaves using response surface methodology. Sep. Purif. Technol. 2007, 55, 381–387. [Google Scholar] [CrossRef]
- Putnik, P.; Barba, F.J.; Španić, I.; Zorić, Z.; Dragović-Uzelac, V.; Kovačević, D.B. Green extraction approach for the recovery of polyphenols from Croatian olive leaves (Olea europaea). Food Bioprod. Process. 2017, 106, 19–28. [Google Scholar] [CrossRef]
- Ahmad-Qasem, M.H.; Cánovas, J.; Barrajón-Catalán, E.; Micol, V.; Cárcel, J.A.; García-Pérez, J.V. Kinetic and compositional study of phenolic extraction from olive leaves (var. Serrana) by using power ultrasound. Innov. Food Sci. Emerg. Technol. 2013, 17, 120–129. [Google Scholar] [CrossRef]
- Mylonaki, S.; Kiassos, E.; Makris, D.P. Optimisation of the extraction of olive (Olea europaea) leaf phenolics using water/ethanol-based solvent systems and response surface methodology. Anal. Bioanal. Chem. 2008, 392, 977–985. [Google Scholar] [CrossRef]
- Gałuszka, A.; Migaszewski, Z.; Namieśnik, J. The 12 principles of green analytical chemistry and the SIGNIFICANCE mnemonic of green analytical practices. TrAC Trends Anal. Chem. 2013, 50, 78–84. [Google Scholar] [CrossRef]
- Abaza, B.L.; Youssef, N.B.; Manai, H.; Haddada, F.M. Chétoui olive leaf extracts: Influence of the solvent type on phenolics and antioxidant activities. Grasas Y Aceites 2011, 62, 96–104. [Google Scholar] [CrossRef]
- Sahin, S.; Samlı, R. Ultrasonics Sonochemistry Optimization of olive leaf extract obtained by ultrasound-assisted extraction with response surface methodology. Ultrason. Sonochem. J. 2013, 20, 595–602. [Google Scholar] [CrossRef]
- Bilgin, M.; Selin, S. Effects of geographical origin and extraction methods on total phenolic yield of olive tree (Olea europaea) leaves. J. Taiwan Inst. Chem. Eng. 2013, 44, 8–12. [Google Scholar] [CrossRef]
- Khemakhem, I.; HussamAhmad-Qasem, M.; Catalán, E.B.; Micol, V.; García-Pérez, J.V.; Ayadi, M.A.; Bouaziz, M. Kinetic improvement of olive leaves’ bioactive compounds extraction by using power ultrasound in a wide temperature range. Ultrason. Sonochem. 2017, 34, 466–473. [Google Scholar] [CrossRef]
- Bouallagui, Z.; Han, J.; Isoda, H.; Sayadi, S. Hydroxytyrosol rich extract from olive leaves modulates cell cycle progression in MCF-7 human breast cancer cells. Food Chem. Toxicol. 2011, 49, 179–184. [Google Scholar] [CrossRef]
- Abaza, L.; Talorete, T.P.; Yamada, P.; Kurita, Y.; Zarrouk, M.; Isoda, H. Induction of Growth Inhibition and Differentiation of Human Leukemia HL-60 Cells by a Tunisian Gerboui Olive Leaf Extract. Biosci. Biotechnol. Biochem. 2007, 71, 1306–1312. [Google Scholar] [CrossRef]
- Xie, P.; Huang, L.; Zhang, C.; Zhang, Y. Phenolic compositions, and antioxidant performance of olive leaf and fruit (Olea europaea L.) extracts and their structure—Activity relationships. J. Funct. Foods 2015, 16, 460–471. [Google Scholar] [CrossRef]
- Korukluoglu, M.; Sahan, Y.; Yigit, A.; Ozer, E.T. Antibacterial activity and chemical constitutions. Food Control 2008, 34, 383–396. [Google Scholar]
- El, S.N.; Karakaya, S. Olive tree (Olea europaea) leaves: Potential beneficial effects on human health. Nutr. Rev. 2009, 67, 632–638. [Google Scholar] [CrossRef]
- Yuan, J.; Wang, C.; Ye, J.; Tao, R.; Zhang, Y. Enzymatic Hydrolysis of Oleauropein from Olea europea (Olive) Leaf Extract and Antioxidant Activities. Molecules 2015, 11, 2903–2921. [Google Scholar] [CrossRef]
- Sahin, S.; Bilgin, M. Olive tree (Olea europaea L.) leaf as a waste by-product of table olive and olive oil industry: A review. J. Sci. Food Agric. 2018, 98, 1271–1279. [Google Scholar] [CrossRef]
- El, A.; Rizk, T.; Lteif, R.; Azouri, H.; Delia, M.; Lebrihi, A. Fungal contamination and Aflatoxin B1 and Ochratoxin A in Lebanese wine—Grapes and musts. Food Chem. Toxicol. J. 2008, 46, 2244–2250. [Google Scholar]
- Klan, A.; Jer, B.; Smole, S. Evaluation of diffusion and dilution methods to determine the antibacterial activity of plant extracts. J. Microbiol. Methods 2010, 81, 121–126. [Google Scholar]
- Gökmen, M.; Kara, R.; Akkaya, L.; Torlak, E.; Önen, A. Evaluation of antimicrobial activity in olive (Olea europaea) leaf extract. Am. J. Microbiol. 2015, 5, 37–40. [Google Scholar]
- Karygianni, L.; Cecere, M.; Skaltsounis, A.L.; Argyropoulou, A.; Hellwig, E.; Aligiannis, N.; Wittmer, A.; Al-Ahmad, A. High-Level Antimicrobial Efficacy of Representative Mediterranean Natural Plant Extracts against Oral Microorganisms. BioMed Res. Int. 2014, 2014, 839019. [Google Scholar] [CrossRef]
- Daoud, Z.; Abdo, E.; Abdel-Massih, R.M. Antibacterial activity of rheum rhaponticum, olea europaea, and viola odorata on esbl producing clinical isolates of escherichia coli and klebsiella pneumoniae. Int. J. Pharm. Sci. Res. 2011, 2, 1669–1678. [Google Scholar]



| Bacterial Strain | Olive Leaves Extract Minimum Inhibitory Concentrations (mg/mL) | |
|---|---|---|
| WB | IR | |
| S. aureus 001 | 12.5 | 12.5 |
| S. aureus 002 | 6.25 | 6.25 |
| S. aureus 003 | 12.5 | 12.5 |
| S. aureus 004 | 3.125 | 3.125 |
| S. aureus 005 | 3.125 | 3.125 |
| S. aureus 006 | 12.5 | 12.5 |
| S. aureus 007 | 12.5 | 12.5 |
| S. aureus 008 | 12.5 | 12.5 |
| S. aureus 009 | 12.5 | 12.5 |
| S. aureus 010 | 12.5 | 12.5 |
| S. aureus 011 | 12.5 | 12.5 |
| S. aureus 012 | 12.5 | 12.5 |
| S. aureus 013 | 12.5 | 12.5 |
| S. aureus 014 | 12.5 | 12.5 |
| S. aureus 015 | 12.5 | 12.5 |
| S. aureus 016 | 12.5 | 12.5 |
| S. aureus 017 | 12.5 | 12.5 |
| S. aureus Newman | 6.25 | 6.25 |
| S. aureus ATCC 29213 | 6.25 | 6.25 |
| S. aureus N315 MRSA | 12.5 | 12.5 |
| Run | Central Composite Design | Variable Levels Uncoded | Phenolic Compounds Yield (mg GAE/g DM) | |||||
|---|---|---|---|---|---|---|---|---|
| Solvent (% Ethanol) | Time (min) | Temperature ( °C) | WB | IR | ||||
| Experimental | Predicted | Experimental | Predicted | |||||
| 1 | Factorial design points | 40 | 60 | 38 | 15.74 | 15.00 | 19.71 | 18.27 |
| 2 | 80 | 60 | 38 | 14.41 | 13.81 | 16.61 | 14.73 | |
| 3 | 40 | 180 | 38 | 18.35 | 18.47 | 23.20 | 22.40 | |
| 4 | 80 | 180 | 38 | 16.12 | 16.65 | 18.39 | 16.63 | |
| 5 | 40 | 60 | 77 | 22.52 | 21.16 | 21.02 | 22.15 | |
| 6 | 80 | 60 | 77 | 23.90 | 22.94 | 23.66 | 23.81 | |
| 7 | 40 | 180 | 77 | 23.81 | 23.58 | 27.51 | 28.75 | |
| 8 | 80 | 180 | 77 | 24.81 | 24.72 | 27.38 | 28.18 | |
| 9 | Star points | 26.36 | 120 | 57.5 | 17.41 | 18.33 | 21.90 | 21.51 |
| 10 | 93.63 | 120 | 57.5 | 18.04 | 18.30 | 16.76 | 18.06 | |
| 11 | 60 | 19.09 | 57.5 | 16.31 | 18.09 | 16.46 | 17.37 | |
| 12 | 60 | 220.9 | 57.5 | 23.11 | 22.51 | 23.86 | 23.86 | |
| 13 | 60 | 120 | 24.7 | 14.31 | 14.31 | 15.01 | 18.20 | |
| 14 | 60 | 120 | 90.3 | 25.11 | 26.28 | 33.47 | 31.18 | |
| 15 | 60 | 120 | 57.5 | 21.20 | 21.22 | 21.77 | 20.81 | |
| 16 | Center points | 60 | 120 | 57.5 | 21.15 | 21.22 | 20.17 | 20.81 |
| 17 | 60 | 120 | 57.5 | 21.43 | 21.22 | 20.32 | 20.81 | |
| 18 | 60 | 120 | 57.5 | 21.09 | 21.22 | 20.70 | 20.81 | |
| 19 | 60 | 120 | 57.5 | 21.20 | 21.22 | 20.45 | 20.81 | |
| 20 | 60 | 120 | 57.5 | 21.49 | 21.22 | 20.51 | 20.81 | |
| 21 | 60 | 120 | 57.5 | 20.47 | 21.22 | 21.29 | 20.81 | |
| 22 | 60 | 120 | 57.5 | 21.89 | 21.22 | 21.45 | 20.81 | |
| Factor | Optimum Conditions | |
|---|---|---|
| WB | IR | |
| Ethanol/Water (%) | 70.16 | 55.35 |
| Time (min) | 193.28 | 220.91 |
| Temperature ( °C) | 90.29 | 90.29 |
| Extraction Technique | R2 (Percent) | Equation |
|---|---|---|
| WB | 90.69 | TPC = 29.1281 − 0.0854381E + 0.0347351t − 0.487806T − 0.000850084E2 − 0.0000128046t2 + 0.00366906T2 − 0.00046598Et + 0.00333862ET + 0.000528357tT |
| IR | 95.23 | TPC = −1.18369 + 0.214169E + 0.0644087t + 0.193729T − 0.0025667E2 − 0.0000897562t2 − 0.000854751T2 − 0.000132978Et + 0.0019014ET − 0.000225784tT |
| Optimum TPC Value (mg GAE/g DM) | Predicted | Experimental |
|---|---|---|
| WB | 27.12 | 26.31a ± 0.3 |
| IR | 36.23 | 34.28b ± 1 |
| Concentration (mg/g DM) | Extraction Technique | |
|---|---|---|
| WB | IR | |
| Oleuropein | 11.84a ± 1.2 | 14.01b ± 0.9 |
| Hydroxytyrosol | 0.33c ± 0.02 | 0.40d ± 0.008 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abi-Khattar, A.-M.; Rajha, H.N.; Abdel-Massih, R.M.; Maroun, R.G.; Louka, N.; Debs, E. Intensification of Polyphenol Extraction from Olive Leaves Using Ired-Irrad®, an Environmentally-Friendly Innovative Technology. Antioxidants 2019, 8, 227. https://doi.org/10.3390/antiox8070227
Abi-Khattar A-M, Rajha HN, Abdel-Massih RM, Maroun RG, Louka N, Debs E. Intensification of Polyphenol Extraction from Olive Leaves Using Ired-Irrad®, an Environmentally-Friendly Innovative Technology. Antioxidants. 2019; 8(7):227. https://doi.org/10.3390/antiox8070227
Chicago/Turabian StyleAbi-Khattar, Anna-Maria, Hiba N. Rajha, Roula M. Abdel-Massih, Richard G. Maroun, Nicolas Louka, and Espérance Debs. 2019. "Intensification of Polyphenol Extraction from Olive Leaves Using Ired-Irrad®, an Environmentally-Friendly Innovative Technology" Antioxidants 8, no. 7: 227. https://doi.org/10.3390/antiox8070227
APA StyleAbi-Khattar, A.-M., Rajha, H. N., Abdel-Massih, R. M., Maroun, R. G., Louka, N., & Debs, E. (2019). Intensification of Polyphenol Extraction from Olive Leaves Using Ired-Irrad®, an Environmentally-Friendly Innovative Technology. Antioxidants, 8(7), 227. https://doi.org/10.3390/antiox8070227









