Upregulation of Tolerogenic Pathways by the Hydrogen Sulfide Donor GYY4137 and Impaired Expression of H2S-Producing Enzymes in Multiple Sclerosis
Abstract
1. Introduction
2. Materials and Methods
2.1. In Vitro, In Vivo and Ex Vivo Studies
2.1.1. Cells and Cell Cultures
2.1.2. Cell Viability Assay
2.1.3. Cytokine Level Measurement
2.1.4. Cytometry
2.1.5. Reverse Transcription—Real-Time Polymerase Chain Reaction
2.2. In Silico Studies
2.2.1. Analysis of the Expression Levels of H2S-Producing Enzymes in Human CD4+ T Cell Subset
2.2.2. Analysis of the Expression Levels of H2S-Producing Enzymes in PBMCs from MS Patients
2.2.3. Computational Deconvolution of Immune Cells Population
2.3. Statistical Analysis
3. Results
3.1. In Vitro, In Vivo and Ex Vivo Studies
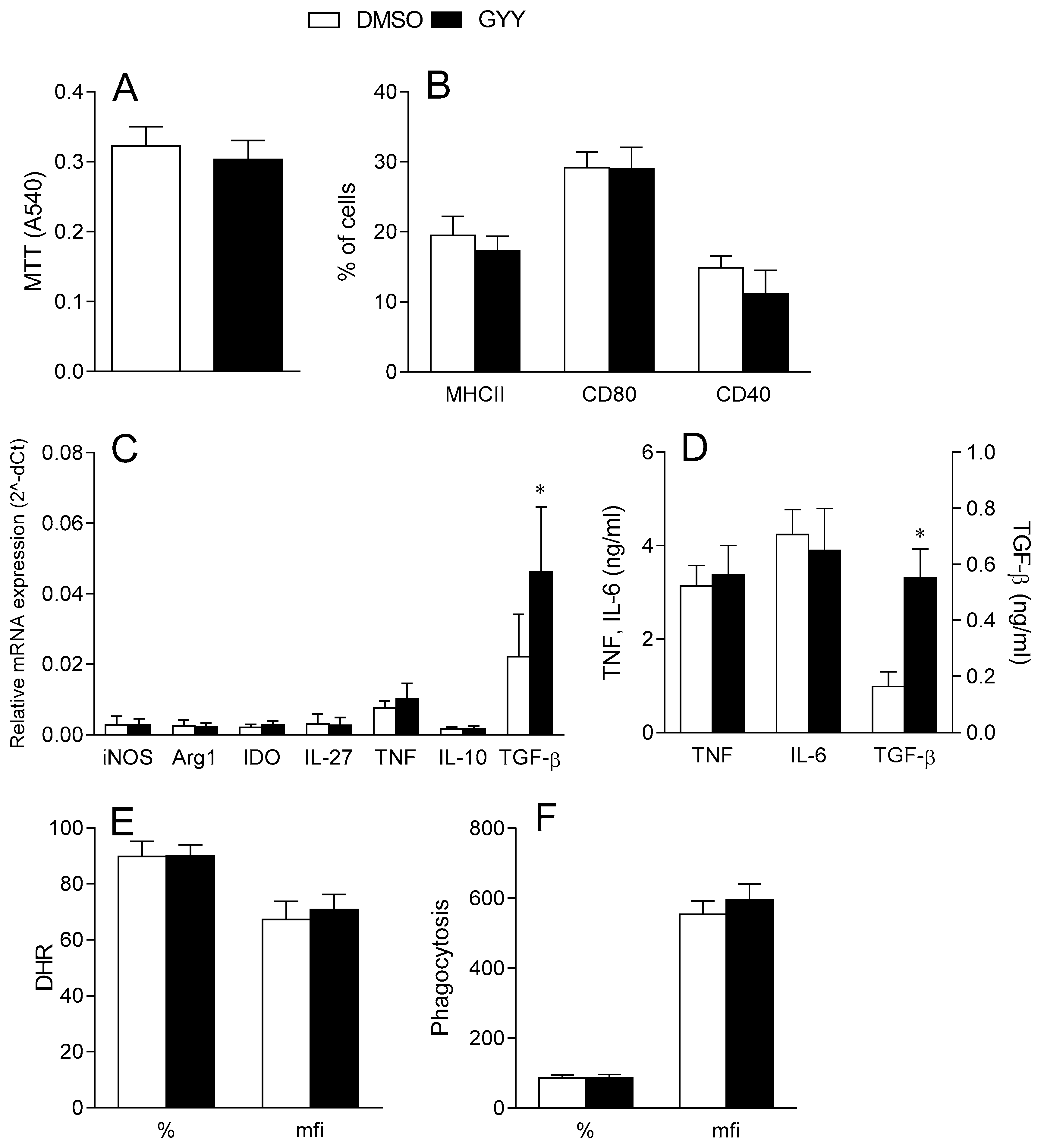
3.1.1. Effects of GYY4137 on Dendritic Cells
3.1.2. Effects of GYY4137 on Mouse Lymph Node T cells
3.1.3. Effects of GYY4137 on the Rat Lymph Node T Cells
3.1.4. Effects of GYY4137 on Spinal Cord T Cells
3.2. In Silico Studies
3.2.1. Expression of H2S-Producing Enzymes in Human CD4+ T Cell Subsets
3.2.2. Expression of H2S-Producing Enzymes in PBMCs from Multiple Sclerosis Patients
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Constantinescu, C.S.; Farooqi, N.; O’Brien, K.; Gran, B. Experimental autoimmune encephalomyelitis (EAE) as a model for multiple sclerosis (MS). Br. J. Pharmacol. 2011, 164, 1079–1106. [Google Scholar] [CrossRef] [PubMed]
- Lo Sasso, B.; Agnello, L.; Bivona, G.; Bellia, C.; Ciaccio, M. Cerebrospinal fluid analysis in multiple sclerosis diagnosis: An update. Medicina 2019, 55, 245. [Google Scholar] [CrossRef] [PubMed]
- Kurschus, F.C. T cell mediated pathogenesis in EAE: Molecular mechanisms. Biomed. J. 2015, 38, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Jafarirad, S.; Siassi, F.; Harirchian, M.-H.; Sahraian, M.-A.; Eshraghian, M.-R.; Shokri, F.; Amani, R.; Bitarafan, S.; Mozafari, S.; Saboor-Yaraghi, A. The effect of vitamin A supplementation on stimulated T-cell proliferation with myelin oligodendrocyte glycoprotein in patients with multiple sclerosis. J. Neurosci. Rural Pract. 2012, 3, 294–298. [Google Scholar] [CrossRef]
- Petermann, F.; Korn, T. Cytokines and effector T cell subsets causing autoimmune CNS disease. FEBS Lett. 2011, 585, 3747–3757. [Google Scholar] [CrossRef]
- Batoulis, H.; Addicks, K.; Kuerten, S. Emerging concepts in autoimmune encephalomyelitis beyond the CD4/TH1 paradigm. Ann. Anat. 2010, 192, 179–193. [Google Scholar] [CrossRef]
- Yamasaki, R.; Kira, J. ichi Multiple Sclerosis. In Advances in Experimental Medicine and Biology; Springer: Berlin/Heidelberg, Germany, 2019; Volume 1190, pp. 217–247. [Google Scholar]
- Danikowski, K.M.; Jayaraman, S.; Prabhakar, B.S. Regulatory T cells in multiple sclerosis and myasthenia gravis. J. Neuroinflamm. 2017, 14, 117. [Google Scholar] [CrossRef]
- Fagone, P.; Mazzon, E.; Bramanti, P.; Bendtzen, K.; Nicoletti, F. Gasotransmitters and the immune system: Mode of action and novel therapeutic targets. Eur. J. Pharmacol. 2018, 834, 92–102. [Google Scholar] [CrossRef]
- Szabo, C. Gasotransmitters in cancer: From pathophysiology to experimental therapy. Nat. Rev. Drug Discov. 2016, 15, 185–203. [Google Scholar] [CrossRef]
- Panthi, S.; Chung, H.J.; Jung, J.; Jeong, N.Y. Physiological Importance of Hydrogen Sulfide: Emerging Potent Neuroprotector and Neuromodulator. Oxid. Med. Cell. Longev. 2016, 2016, 9049782. [Google Scholar] [CrossRef] [PubMed]
- Ineichen, B.V.; Keskitalo, S.; Farkas, M.; Bain, N.; Kallweit, U.; Weller, M.; Klotz, L.; Linnebank, M. Genetic variants of homocysteine metabolism and multiple sclerosis: A case-control study. Neurosci. Lett. 2014, 562, 75–78. [Google Scholar] [CrossRef] [PubMed]
- Role of Gasotransmitters in Oxidative Stresses, Neuroinflammation, and Neuronal Repair—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/28386548/ (accessed on 17 May 2020).
- Fagone, P.; Mangano, K.; Quattrocchi, C.; Motterlini, R.; Di Marco, R.; Magro, G.; Penacho, N.; Romao, C.C.; Nicoletti, F. Prevention of clinical and histological signs of proteolipid protein (PLP)-induced experimental allergic encephalomyelitis (EAE) in mice by the water-soluble carbon monoxide-releasing molecule (CORM)-A1. Clin. Exp. Immunol. 2011, 163, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Fagone, P.; Patti, F.; Mangano, K.; Mammana, S.; Coco, M.; Touil-Boukoffa, C.; Chikovani, T.; Di Marco, R.; Nicoletti, F. Heme oxygenase-1 expression in peripheral blood mononuclear cells correlates with disease activity in multiple sclerosis. J. Neuroimmunol. 2013, 261, 82–86. [Google Scholar] [CrossRef] [PubMed]
- Fagone, P.; Mangano, K.; Coco, M.; Perciavalle, V.; Garotta, G.; Romao, C.C.; Nicoletti, F. Therapeutic potential of carbon monoxide in multiple sclerosis. Clin. Exp. Immunol. 2012, 167, 179–187. [Google Scholar] [CrossRef]
- Maksimovic-Ivanic, D.; Fagone, P.; McCubrey, J.; Bendtzen, K.; Mijatovic, S.; Nicoletti, F. HIV-protease inhibitors for the treatment of cancer: Repositioning HIV protease inhibitors while developing more potent NO-hybridized derivatives? Int. J. Cancer 2017, 140, 1713–1726. [Google Scholar] [CrossRef]
- Paskas, S.; Mazzon, E.; Basile, M.S.; Cavalli, E.; Al-Abed, Y.; He, M.; Rakocevic, S.; Nicoletti, F.; Mijatovic, S.; Maksimovic-Ivanic, D. Lopinavir-NO, a nitric oxide-releasing HIV protease inhibitor, suppresses the growth of melanoma cells in vitro and in vivo. Investig. New Drugs 2019, 37, 1014–1028. [Google Scholar] [CrossRef]
- Sestito, S.; Pruccoli, L.; Runfola, M.; Citi, V.; Martelli, A.; Saccomanni, G.; Calderone, V.; Tarozzi, A.; Rapposelli, S. Design and synthesis of H2S-donor hybrids: A new treatment for Alzheimer’s disease? Eur. J. Med. Chem. 2019, 184, 111745. [Google Scholar] [CrossRef]
- Fagone, P.; Mangano, K.; Quattrocchi, C.; Cavalli, E.; Mammana, S.; Lombardo, G.A.G.; Pennisi, V.; Zocca, M.-B.; He, M.; Al-Abed, Y.; et al. Effects of NO-Hybridization on the Immunomodulatory Properties of the HIV Protease Inhibitors Lopinavir and Ritonavir. Basic Clin. Pharmacol. Toxicol. 2015, 117, 306–315. [Google Scholar] [CrossRef]
- Rothweiler, F.; Michaelis, M.; Brauer, P.; Otte, J.; Weber, K.; Fehse, B.; Doerr, H.W.; Wiese, M.; Kreuter, J.; Al-Abed, Y.; et al. Anticancer effects of the nitric oxide-modified saquinavir derivative saquinavir-NO against multidrug-resistant cancer cells. Neoplasia 2010, 12, 1023–1030. [Google Scholar] [CrossRef]
- Cao, X.; Ding, L.; Xie, Z.-Z.; Yang, Y.; Whiteman, M.; Moore, P.K.; Bian, J.-S. A Review of Hydrogen Sulfide Synthesis, Metabolism, and Measurement: Is Modulation of Hydrogen Sulfide a Novel Therapeutic for Cancer? Antioxid. Redox Signal. 2019, 31, 1–38. [Google Scholar] [CrossRef]
- Pathi, S.S.; Jutooru, I.; Chadalapaka, G.; Sreevalsan, S.; Anand, S.; Thatcher, G.R.J.; Safe, S. GT-094, a NO-NSAID, inhibits colon cancer cell growth by activation of a reactive oxygen species-MicroRNA-27a: ZBTB10-specificity protein pathway. Mol. Cancer Res. 2011, 9, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, N.; Williams, J.L.; Rigas, B. NO-donating aspirin inhibits angiogenesis by suppressing VEGF expression in HT-29 human colon cancer mouse xenografts. Carcinogenesis 2008, 29, 1794–1798. [Google Scholar] [CrossRef] [PubMed]
- Amoruso, A.; Fresu, L.G.; Dalli, J.; Miglietta, D.; Bardelli, C.; Canova, D.F.; Perretti, M.; Brunelleschi, S. Characterization of the anti-inflammatory properties of NCX 429, a dual-acting compound releasing nitric oxide and naproxen. Life Sci. 2015, 126, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Gund, M.; Gaikwad, P.; Borhade, N.; Burhan, A.; Desai, D.C.; Sharma, A.; Dhiman, M.; Patil, M.; Sheikh, J.; Thakre, G.; et al. Gastric-sparing nitric oxide-releasable “true” prodrugs of aspirin and naproxen. Bioorganic Med. Chem. Lett. 2014, 24, 5587–5592. [Google Scholar] [CrossRef] [PubMed]
- Duan, W.; Li, J.; Inks, E.S.; Chou, C.J.; Jia, Y.; Chu, X.; Li, X.; Xu, W.; Zhang, Y. Design, synthesis, and antitumor evaluation of novel histone deacetylase inhibitors equipped with a phenylsulfonylfuroxan module as a nitric oxide donor. J. Med. Chem. 2015, 58, 4325–4338. [Google Scholar] [CrossRef]
- Dillon, K.M.; Carrazzone, R.J.; Matson, J.B.; Kashfi, K. The evolving landscape for cellular nitric oxide and hydrogen sulfide delivery systems: A new era of customized medications. Biochem. Pharmacol. 2020, 176, 113931. [Google Scholar] [CrossRef]
- Ji, J.; Xiang, P.; Li, T.; Lan, L.; Xu, X.; Lu, G.; Ji, H.; Zhang, Y.; Li, Y. NOSH-NBP, a novel nitric oxide and hydrogen Sulfide-Releasing hybrid, attenuates ischemic Stroke-Induced neuroinflammatory injury by modulating microglia polarization. Front. Cell. Neurosci. 2017, 11, 154. [Google Scholar] [CrossRef] [PubMed]
- He, J.T.; Li, H.; Yang, L.; Mao, C.Y. Role of hydrogen sulfide in cognitive deficits: Evidences and mechanisms. Eur. J. Pharmacol. 2019, 849, 146–153. [Google Scholar] [CrossRef]
- De Cicco, P.; Panza, E.; Ercolano, G.; Armogida, C.; Sessa, G.; Pirozzi, G.; Cirino, G.; Wallace, J.L.; Ianaro, A. ATB-346, a novel hydrogen sulfide-releasing anti-inflammatory drug, induces apoptosis of human melanoma cells and inhibits melanoma development in vivo. Pharmacol. Res. 2016, 114, 67–73. [Google Scholar] [CrossRef]
- Szabo, C. A timeline of hydrogen sulfide (H2S) research: From environmental toxin to biological mediator. Biochem. Pharmacol. 2018, 149, 5–19. [Google Scholar] [CrossRef]
- Szabó, C. Hydrogen sulphide and its therapeutic potential. Nat. Rev. Drug Discov. 2007, 6, 917–935. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, M. H2S and inflammation: An overview. Handb. Exp. Pharmacol. 2015, 230, 165–180. [Google Scholar]
- Gadalla, M.M.; Snyder, S.H. Hydrogen sulfide as a gasotransmitter. J. Neurochem. 2010, 113, 14–26. [Google Scholar] [CrossRef] [PubMed]
- Powell, C.R.; Dillon, K.M.; Matson, J.B. A review of hydrogen sulfide (H2S) donors: Chemistry and potential therapeutic applications. Biochem. Pharmacol. 2018, 149, 110–123. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.F.; Lu, M.; Hon Wong, P.T.; Bian, J.S. Hydrogen sulfide: Neurophysiology and neuropathology. Antioxid. Redox Signal. 2011, 15, 405–419. [Google Scholar] [CrossRef]
- Kimura, Y.; Shibuya, N.; Kimura, H. Sulfite protects neurons from oxidative stress. Br. J. Pharmacol. 2019, 176, 571–582. [Google Scholar] [CrossRef]
- Zhou, X.; Cao, Y.; Ao, G.; Hu, L.; Liu, H.; Wu, J.; Wang, X.; Jin, M.; Zheng, S.; Zhen, X.; et al. CaMKKβ-Dependent activation of AMP-activated protein kinase is critical to suppressive effects of hydrogen sulfide on neuroinflammation. Antioxid. Redox Signal. 2014, 21, 1741–1758. [Google Scholar] [CrossRef]
- Kloesch, B.; Liszt, M.; Broell, J. H2S transiently blocks IL-6 expression in rheumatoid arthritic fibroblast-like synoviocytes and deactivates p44/42 mitogen-activated protein kinase. Cell Biol. Int. 2010, 34, 477–484. [Google Scholar] [CrossRef]
- Wallace, J.L. Physiological and Pathophysiological Roles of Hydrogen Sulfide in the Gastrointestinal Tract. Antioxid. Redox Signal. 2010, 12, 1125–1133. [Google Scholar] [CrossRef]
- Oh, G.S.; Pae, H.O.; Lee, B.S.; Kim, B.N.; Kim, J.M.; Kim, H.R.; Jeon, S.B.; Jeon, W.K.; Chae, H.J.; Chung, H.T. Hydrogen sulfide inhibits nitric oxide production and nuclear factor-κB via heme oxygenase-1 expression in RAW264.7 macrophages stimulated with lipopolysaccharide. Free Radic. Biol. Med. 2006, 41, 106–119. [Google Scholar] [CrossRef]
- Muniraj, N.; Stamp, L.K.; Badiei, A.; Hegde, A.; Cameron, V.; Bhatia, M. Hydrogen sulfide acts as a pro-inflammatory mediator in rheumatic disease. Int. J. Rheum. Dis. 2017, 20, 182–189. [Google Scholar] [CrossRef]
- Whiteman, M.; Winyard, P.G. Hydrogen sulfide and inflammation: The good, the bad, the ugly and the promising. Expert Rev. Clin. Pharmacol. 2011, 4, 13–32. [Google Scholar] [CrossRef] [PubMed]
- Powell, N.D.; Papenfuss, T.L.; McClain, M.A.; Gienapp, I.E.; Shawler, T.M.; Satoskar, A.R.; Whitacre, C.C. Cutting edge: Macrophage migration inhibitory factor is necessary for progression of experimental autoimmune encephalomyelitis. J. Immunol. 2005, 175, 5611–5614. [Google Scholar] [CrossRef] [PubMed]
- Rose, P.; Dymock, B.W.; Moore, P.K. GYY4137, a novel water-soluble, H2S-releasing molecule. In Methods in Enzymology; Academic Press Inc.: Cambridge, MA, USA, 2015; Volume 554, pp. 143–167. [Google Scholar]
- Lazarević, M.; Mazzon, E.; Momčilović, M.; Basile, M.; Colletti, G.; Petralia, M.; Bramanti, P.; Nicoletti, F.; Miljković, Đ. The H2S Donor GYY4137 Stimulates Reactive Oxygen Species Generation in BV2 Cells While Suppressing the Secretion of TNF and Nitric Oxide. Molecules 2018, 23, 2966. [Google Scholar] [CrossRef] [PubMed]
- Clough, E.; Barrett, T. The Gene Expression Omnibus database. In Methods in Molecular Biology; Humana Press Inc.: Totowa, NJ, USA, 2016; Volume 1418, pp. 93–110. [Google Scholar]
- Mangano, K.; Cavalli, E.; Mammana, S.; Basile, M.S.; Caltabiano, R.; Pesce, A.; Puleo, S.; Atanasov, A.G.; Magro, G.; Nicoletti, F.; et al. Involvement of the Nrf2/HO-1/CO axis and therapeutic intervention with the CO-releasing molecule CORM-A1, in a murine model of autoimmune hepatitis. J. Cell. Physiol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Mammana, S.; Bramanti, P.; Mazzon, E.; Cavalli, E.; Basile, M.S.; Fagone, P.; Petralia, M.C.; McCubrey, J.A.; Nicoletti, F.; Mangano, K. Preclinical evaluation of the PI3K/Akt/mTOR pathway in animal models of multiple sclerosis. Oncotarget 2018, 9, 8263–8277. [Google Scholar] [CrossRef] [PubMed]
- Fagone, P.; Caltabiano, R.; Russo, A.; Lupo, G.; Anfuso, C.D.; Basile, M.S.; Longo, A.; Nicoletti, F.; De Pasquale, R.; Libra, M.; et al. Identification of novel chemotherapeutic strategies for metastatic uveal melanoma. Sci. Rep. 2017, 7, 44564. [Google Scholar] [CrossRef] [PubMed]
- Basile, M.S.; Mazzon, E.; Russo, A.; Mammana, S.; Longo, A.; Bonfiglio, V.; Fallico, M.; Caltabiano, R.; Fagone, P.; Nicoletti, F.; et al. Differential modulation and prognostic values of immune-escape genes in uveal melanoma. PLoS ONE 2019, 14, e0210276. [Google Scholar] [CrossRef]
- Fagone, P.; Mazzon, E.; Mammana, S.; Di Marco, R.; Spinasanta, F.; Basile, M.; Petralia, M.; Bramanti, P.; Nicoletti, F.; Mangano, K. Identification of CD4+ T cell biomarkers for predicting the response of patients with relapsing-remitting multiple sclerosis to natalizumab treatment. Mol. Med. Rep. 2019, 20, 678–684. [Google Scholar] [CrossRef]
- Zhang, H.; Nestor, C.E.; Zhao, S.; Lentini, A.; Bohle, B.; Benson, M.; Wang, H. Profiling of human CD4+ T-cell subsets identifies the TH2-specific noncoding RNA GATA3-AS1. J. Allergy Clin. Immunol. 2013, 132, 1005–1008. [Google Scholar] [CrossRef]
- Feng, X.; Bao, R.; Li, L.; Deisenhammer, F.; Arnason, B.G.W.; Reder, A.T. Interferon-β corrects massive gene dysregulation in multiple sclerosis: Short-term and long-term effects on immune regulation and neuroprotection. EBioMedicine 2019, 49, 269–283. [Google Scholar] [CrossRef] [PubMed]
- Aran, D.; Hu, Z.; Butte, A.J. xCell: Digitally portraying the tissue cellular heterogeneity landscape. Genome Biol. 2017, 18, 220. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Ding, Y.; Wang, Z.; Kong, Y.; Gao, R.; Chen, G. Hydrogen sulfide therapy in brain diseases: From bench to bedside. Med. Gas Res. 2017, 7, 113. [Google Scholar] [PubMed]
- Shefa, U.; Kim, M.S.; Jeong, N.Y.; Jung, J. Antioxidant and Cell-Signaling Functions of Hydrogen Sulfide in the Central Nervous System. Oxid. Med. Cell. Longev. 2018, 2018, 1873962. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Ray, R.S.; Sandhir, R. Hydrogen sulfide attenuates homocysteine-induced neurotoxicity by preventing mitochondrial dysfunctions and oxidative damage: In vitro and in vivo studies. Neurochem. Int. 2018, 120, 87–98. [Google Scholar] [CrossRef]
- Yang, B.; Bai, Y.; Yin, C.; Qian, H.; Xing, G.; Wang, S.; Li, F.; Bian, J.; Aschner, M.; Lu, R. Activation of autophagic flux and the Nrf2/ARE signaling pathway by hydrogen sulfide protects against acrylonitrile-induced neurotoxicity in primary rat astrocytes. Arch. Toxicol. 2018, 92, 2093–2108. [Google Scholar] [CrossRef]
- Lee, P.W.; Severin, M.E.; Lovett-Racke, A.E. TGF-β regulation of encephalitogenic and regulatory T cells in multiple sclerosis. Eur. J. Immunol. 2017, 47, 446–453. [Google Scholar] [CrossRef]
- Lu, P.; Cao, Y.P.; Wang, M.; Zheng, P.; Hou, J.; Zhu, C.; Hu, J. Mature dendritic cells cause Th17/Treg imbalance by secreting TGF-β1 and IL-6 in the pathogenesis of experimental autoimmune encephalomyelitis. Cent. Eur. J. Immunol. 2016, 41, 143–152. [Google Scholar] [CrossRef]
- Ma, S.; Zhong, D.; Ma, P.; Li, G.; Hua, W.; Sun, Y.; Liu, N.; Zhang, L.; Zhang, W. Exogenous hydrogen sulfide ameliorates diabetes-associated cognitive decline by regulating the mitochondria-mediated apoptotic pathway and IL-23/IL-17 expression in db/db mice. Cell. Physiol. Biochem. 2017, 41, 1838–1850. [Google Scholar] [CrossRef]
- Flügel, T.; Berkowicz, T.; Ritter, T.; Labeur, M.; Jenne, D.E.; Li, Z.; Ellwart, J.W.; Willem, M.; Lassmann, H.; Wekerle, H. Migratory activity and functional changes of green fluorescent effector cells before and during experimental autoimmune encephalomyelitis. Immunity 2001, 14, 547–560. [Google Scholar] [CrossRef]
- Miljković, D.; Stanojević, Ž.; Momcilović, M.; Odoardi, F.; Flügel, A.; Mostarica-Stojković, M. CXCL12 expression within the CNS contributes to the resistance against experimental autoimmune encephalomyelitis in Albino Oxford rats. Immunobiology 2011, 216, 979–987. [Google Scholar] [CrossRef]
- Jevtić, B.; Timotijević, G.; Stanisavljević, S.; Momčilović, M.; Mostarica Stojković, M.; Miljković, D. Micro RNA-155 participates in re-activation of encephalitogenic T cells. Biomed. Pharmacother. 2015, 74, 206–210. [Google Scholar] [CrossRef] [PubMed]
- Stojić-Vukanić, Z.; Pilipović, I.; Vujnović, I.; Nacka-Aleksić, M.; Petrović, R.; Arsenović-Ranin, N.; Dimitrijević, M.; Leposavić, G. GM-CSF-producing Th cells in rats sensitive and resistant to experimental autoimmune encephalomyelitis. PLoS ONE 2016, 11, e0166498. [Google Scholar] [CrossRef]
- Hood, J.D.; Zarnitsyna, V.I.; Zhu, C.; Evavold, B.D. Regulatory and T Effector Cells Have Overlapping Low to High Ranges in TCR Affinities for Self during Demyelinating Disease. J. Immunol. 2015, 195, 4162–4170. [Google Scholar] [CrossRef] [PubMed]
- Mellor, A.L.; Lemos, H.; Huang, L. Indoleamine 2,3-Dioxygenase and tolerance: Where Are We Now? Front. Immunol. 2017, 8, 1360. [Google Scholar] [CrossRef]
- Yan, Z.; Garg, S.K.; Banerjee, R. Regulatory T cells interfere with glutathione metabolism in dendritic cells and T cells. J. Biol. Chem. 2010, 285, 41525–41532. [Google Scholar] [CrossRef]
- Nagahara, N.; Sawada, N. The Mercaptopyruvate Pathway in Cysteine Catabolism: A Physiologic Role and Related Disease of the Multifunctional 3-Mercaptopyruvate Sulfurtransferase. Curr. Med. Chem. 2006, 13, 1219–1230. [Google Scholar] [CrossRef] [PubMed]
- Almeida, L.; Lochner, M.; Berod, L.; Sparwasser, T. Metabolic pathways in T cell activation and lineage differentiation. Semin. Immunol. 2016, 28, 514–524. [Google Scholar] [CrossRef] [PubMed]
- Mikami, Y.; Shibuya, N.; Ogasawara, Y.; Kimura, H. Hydrogen sulfide is produced by cystathionine γ-lyase at the steady-state low intracellular Ca2+ concentrations. Biochem. Biophys. Res. Commun. 2013, 431, 131–135. [Google Scholar] [CrossRef]
- Niu, W.N.; Yadav, P.K.; Adamec, J.; Banerjee, R. S-glutathionylation enhances human cystathionine β-synthase activity under oxidative stress conditions. Antioxid. Redox Signal. 2015, 22, 350–361. [Google Scholar] [CrossRef]
- Renga, B. Hydrogen sulfide generation in mammals: The molecular biology of cystathionine-β-synthase (CBS) and cystathionine-γ-lyase (CSE). Inflamm. Allergy Drug Targets 2011, 10, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Wallace, J.L.; Vaughan, D.; Dicay, M.; Macnaughton, W.K.; De Nucci, G. Hydrogen Sulfide-Releasing Therapeutics: Translation to the Clinic. Antioxid. Redox Signal. 2018, 28, 1533–1540. [Google Scholar] [CrossRef] [PubMed]





| Pearson r | 95% Confidence Interval | R Squared | p Value | |
|---|---|---|---|---|
| MPST vs. TNF | −0.6234 | −0.8112 to −0.3189 | 0.3886 | 0.0005 |
| MPST vs. IL2 | −0.3639 | −0.6536 to 0.01865 | 0.1325 | 0.0620 |
| MPST vs. IL6 | −0.6283 | −0.8140 to −0.3261 | 0.3947 | 0.0004 |
| MPST vs. IL17A | −0.1955 | −0.5357 to 0.1993 | 0.0382 | 0.3285 |
| MPST vs. IFNG | −0.548 | −0.7681 to −0.2122 | 0.3003 | 0.0031 |
| MPST vs. IL12A | 0.3633 | −0.01940 to 0.6531 | 0.1320 | 0.0625 |
| MPST vs. IL23A | −0.492 | −0.7347 to −0.1378 | 0.2421 | 0.0091 |
| MPST vs. IL10 | −0.3544 | −0.6472 to 0.02964 | 0.1256 | 0.0697 |
| MPST vs. CCR7 | −0.6298 | −0.8148 to −0.3284 | 0.3967 | 0.0004 |
| MPST vs. CD40LG | −0.6226 | −0.8108 to −0.3177 | 0.3876 | 0.0005 |
| MPST vs. IL2RA | −0.4755 | −0.7246 to −0.1166 | 0.2261 | 0.0122 |
| MPST vs. SELL | 0.4422 | 0.07476 to 0.7039 | 0.1956 | 0.0209 |
| MPST vs. CD69 | −0.6878 | −0.8465 to −0.4167 | 0.4730 | <0.0001 |
| MPST vs. IDO1 | −0.4533 | −0.7109 to −0.08856 | 0.2055 | 0.0176 |
| MPST vs. HMOX1 | 0.4613 | 0.09860 to 0.7158 | 0.2128 | 0.0154 |
| MPST vs. ARG1 | −0.2147 | −0.5498 to 0.1800 | 0.0461 | 0.2822 |
| MPST vs. NOS2 | −0.1639 | −0.5120 to 0.2305 | 0.0269 | 0.4140 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lazarević, M.; Battaglia, G.; Jevtić, B.; Djedovic, N.; Bruno, V.; Cavalli, E.; Miljković, Đ.; Nicoletti, F.; Momčilović, M.; Fagone, P. Upregulation of Tolerogenic Pathways by the Hydrogen Sulfide Donor GYY4137 and Impaired Expression of H2S-Producing Enzymes in Multiple Sclerosis. Antioxidants 2020, 9, 608. https://doi.org/10.3390/antiox9070608
Lazarević M, Battaglia G, Jevtić B, Djedovic N, Bruno V, Cavalli E, Miljković Đ, Nicoletti F, Momčilović M, Fagone P. Upregulation of Tolerogenic Pathways by the Hydrogen Sulfide Donor GYY4137 and Impaired Expression of H2S-Producing Enzymes in Multiple Sclerosis. Antioxidants. 2020; 9(7):608. https://doi.org/10.3390/antiox9070608
Chicago/Turabian StyleLazarević, Milica, Giuseppe Battaglia, Bojan Jevtić, Neda Djedovic, Valeria Bruno, Eugenio Cavalli, Đorđe Miljković, Ferdinando Nicoletti, Miljana Momčilović, and Paolo Fagone. 2020. "Upregulation of Tolerogenic Pathways by the Hydrogen Sulfide Donor GYY4137 and Impaired Expression of H2S-Producing Enzymes in Multiple Sclerosis" Antioxidants 9, no. 7: 608. https://doi.org/10.3390/antiox9070608
APA StyleLazarević, M., Battaglia, G., Jevtić, B., Djedovic, N., Bruno, V., Cavalli, E., Miljković, Đ., Nicoletti, F., Momčilović, M., & Fagone, P. (2020). Upregulation of Tolerogenic Pathways by the Hydrogen Sulfide Donor GYY4137 and Impaired Expression of H2S-Producing Enzymes in Multiple Sclerosis. Antioxidants, 9(7), 608. https://doi.org/10.3390/antiox9070608







