Effect of pH on Total Volume Membrane Charge Density in the Nanofiltration of Aqueous Solutions of Nitrate Salts of Heavy Metals
Abstract
1. Introduction
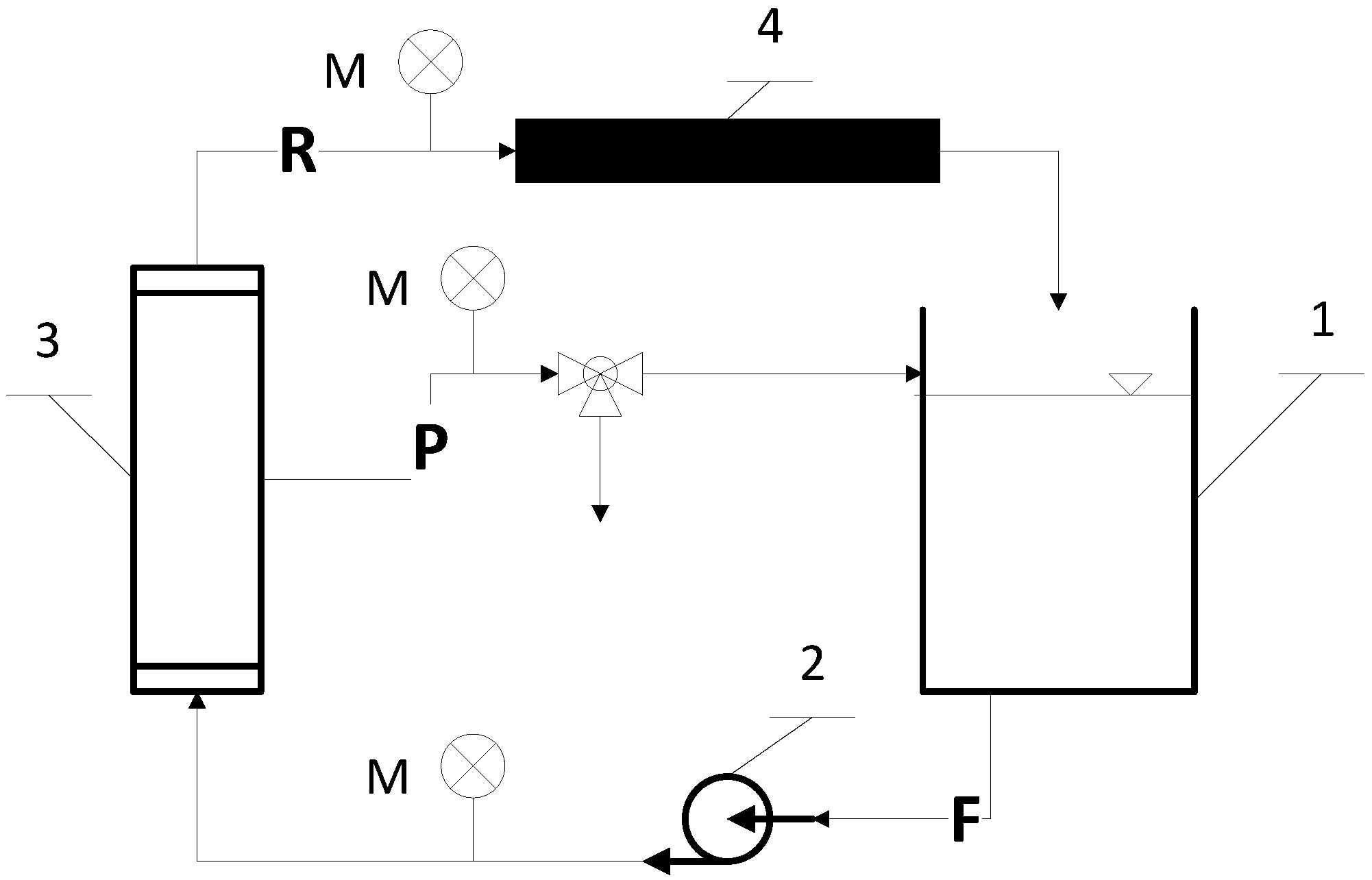
2. Materials and Methods
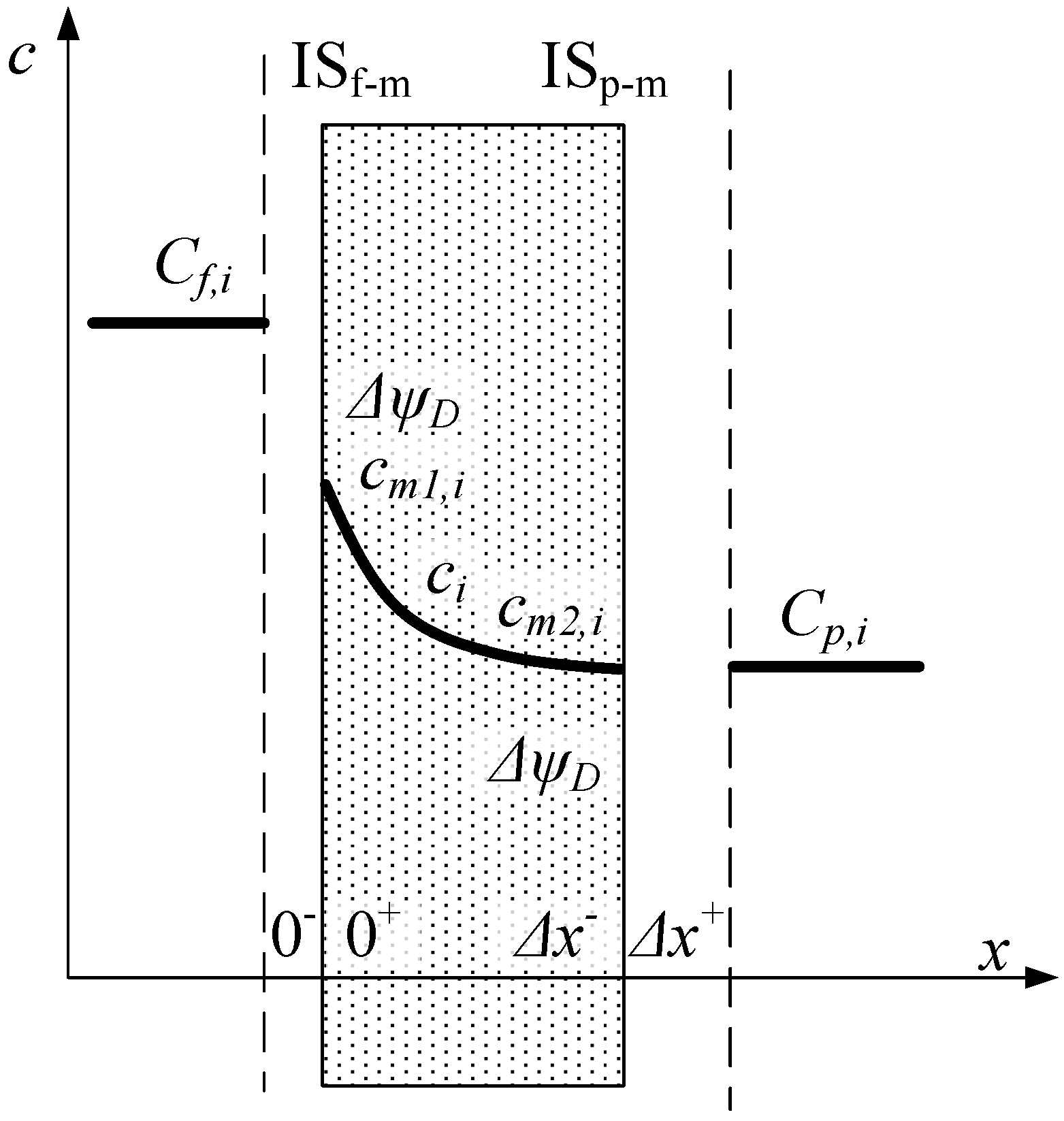
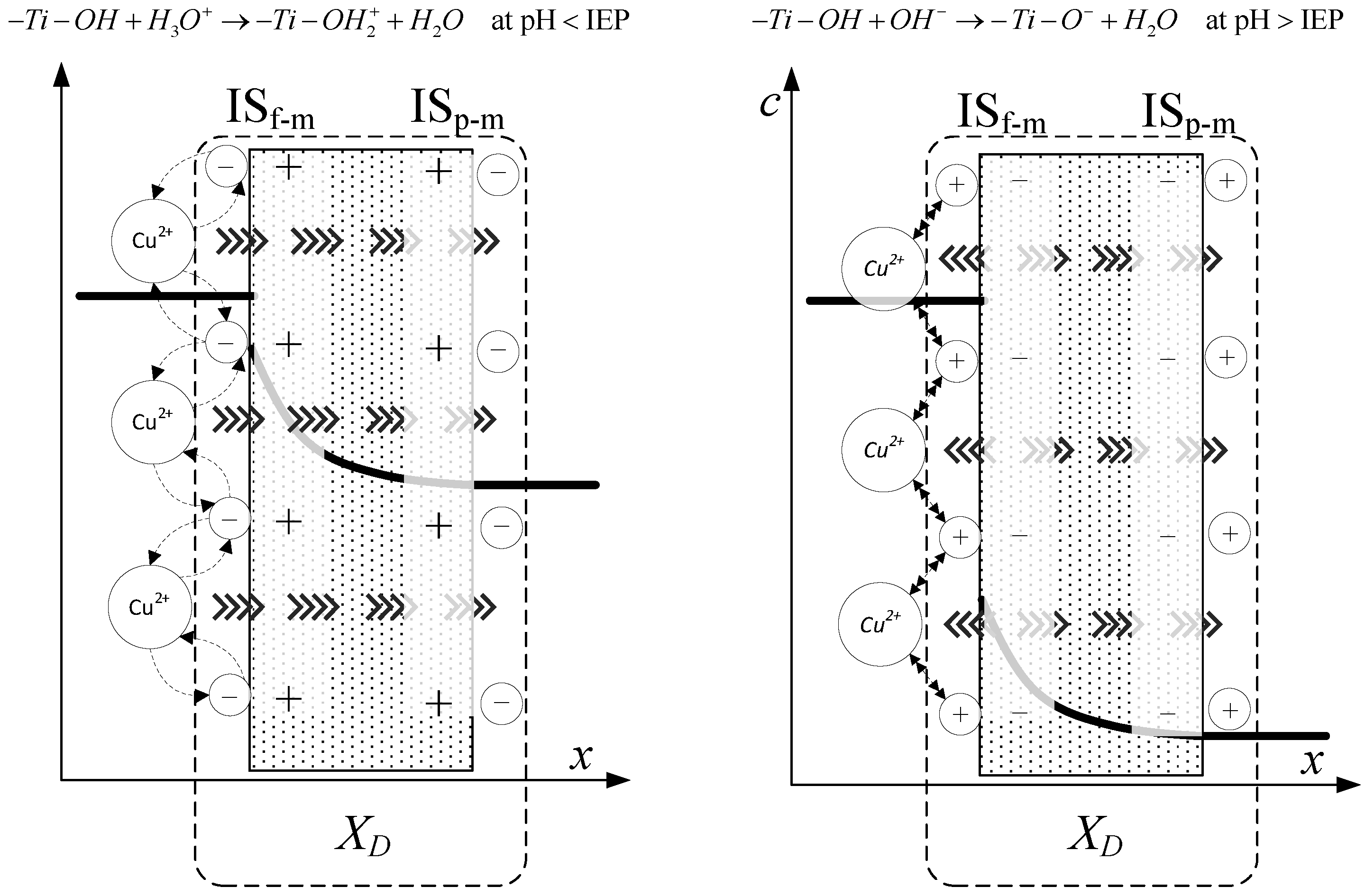
Determination of Effective Membrane Charge Density in Nanofiltration
3. Results and Discussion
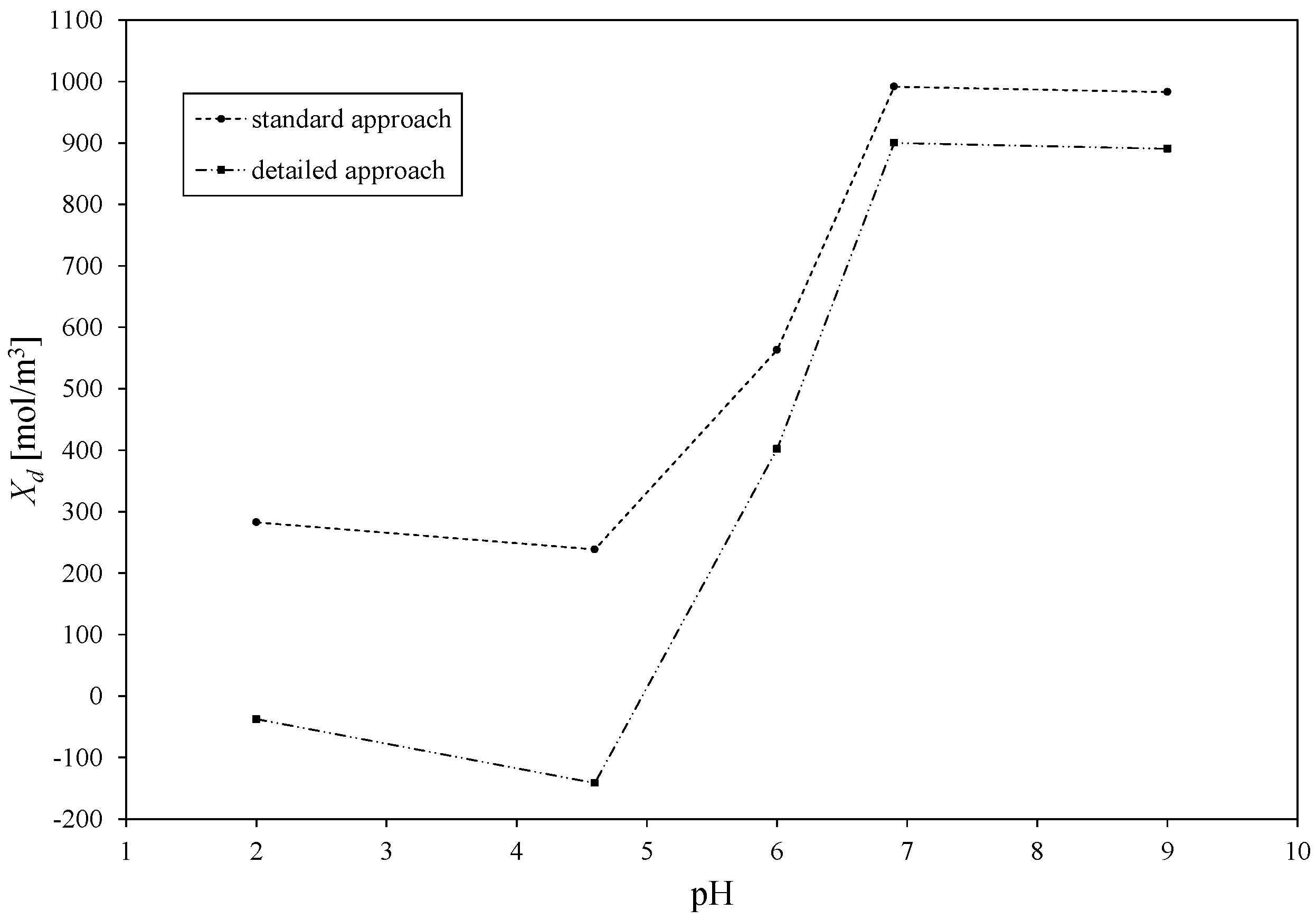
3.1. Comparison of Standard and Detailed DSPM Nanofiltration Modelling
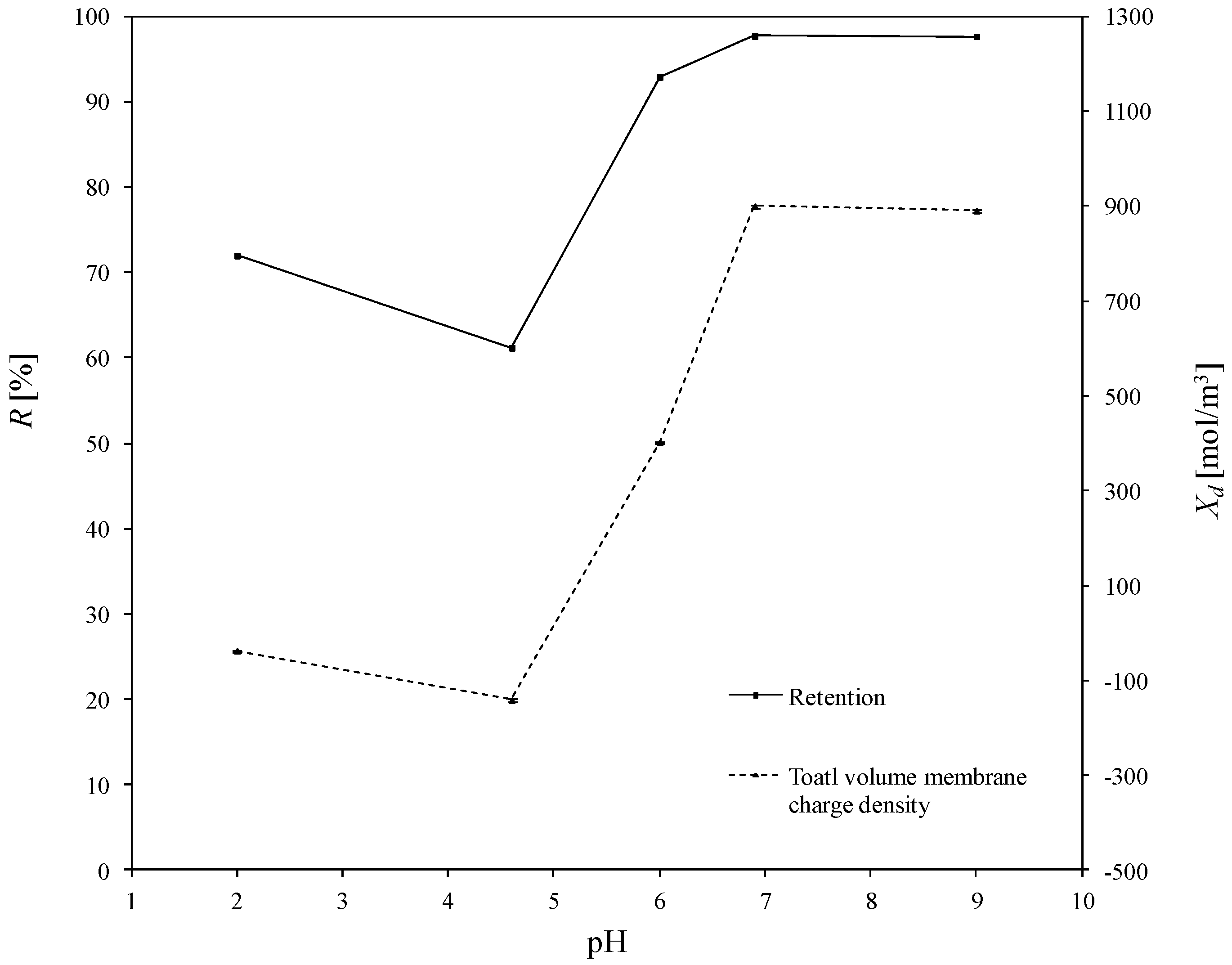
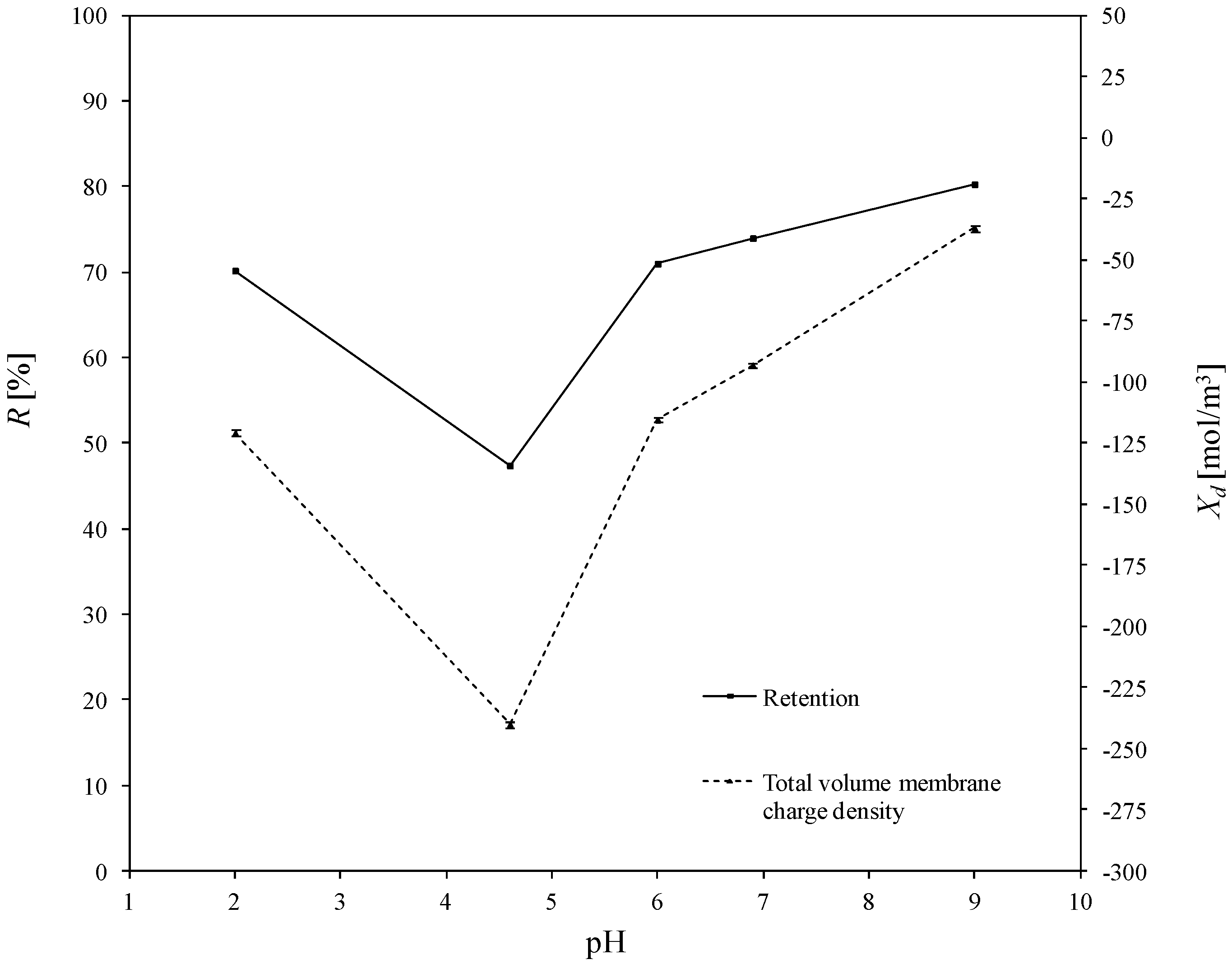
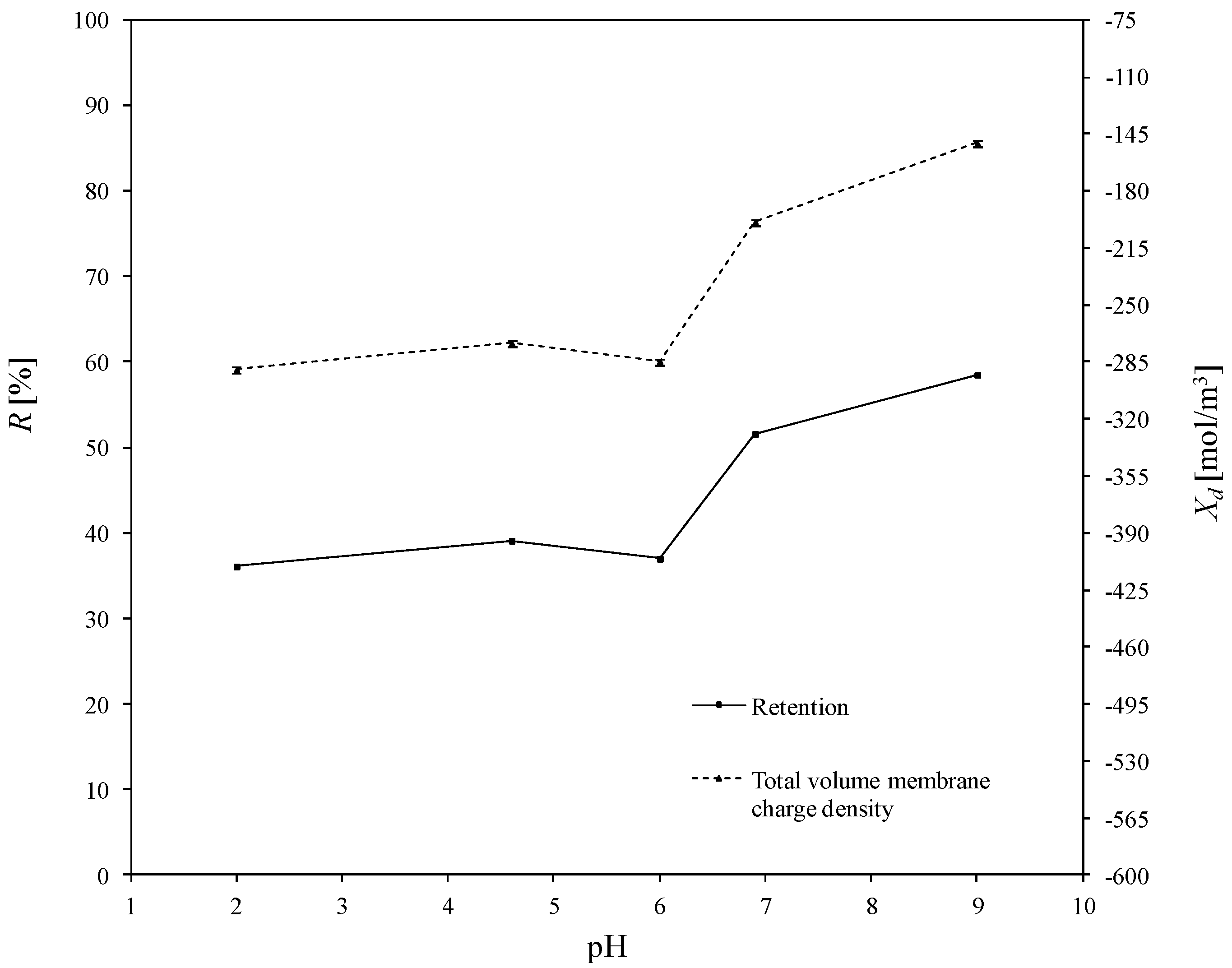
3.2. Estimated Values of Total Volume Membrane Charge Density
3.3. Determination of Corellation of the Total Volume Membrane Charge Density
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wuana, R.A.; Okieimen, F.E. Heavy metals in contaminated soils: A review of sources, chemistry, risks and best available strategies for remediation. ISRN Ecol. 2011, 2011, 402647. [Google Scholar] [CrossRef]
- Barakat, M.A. New trends in removing heavy metals from industrial wastewater. Arab. J. Chem. 2011, 4, 361–377. [Google Scholar] [CrossRef]
- Arbabi, M.; Hemati, S.; Amiri, M. Removal of lead ions from industrial wastewater: A review of removal methods. Int. J. Epidemiol. Res. 2015, 2, 105–109. [Google Scholar]
- National Primary Drinking Water Regulations. Available online: https://www.epa.gov/sites/production/files/2016-06/documents/npwdr_complete_table.pdf (accessed on 18 October 2017).
- Hegazi, H.A. Removal of heavy metals from wastewater using agricultural and industrial wastes as adsorbents. HBRC J. 2013, 9, 276–282. [Google Scholar] [CrossRef]
- Skubal, L.R.; Meshkov, N.K.; Rajh, T.; Thurnauer, M. Cadmium removal from water using thiolactic acid-modified titanium dioxide nanoparticles. J. Photochem. Photobiol. A Chem. 2002, 148, 393–397. [Google Scholar] [CrossRef]
- Prochaska, K.; Woźniak-Budych, M.J. Recovery of fumaric acid from fermentation broth using bipolar electrodialysis. J. Membr. Sci. 2014, 469, 428–435. [Google Scholar] [CrossRef]
- Qdais, H.A.; Moussa, H. Removal of heavy metals from wastewater by membrane processes: A comparative study. Desalination 2004, 164, 105–110. [Google Scholar] [CrossRef]
- Staszak, K.; Woźniak, M.; Sottek, M.; Karaś, Z.; Prochaska, K. Removal of fumaric acid from simulated and real fermentation broth. J. Chem. Technol. Biotechnol. 2015, 90, 432–440. [Google Scholar] [CrossRef]
- Staszak, K.; Woźniak, J.M.; Staniewski, J.; Prochaska, K. Application of nanofiltration in the process of the separation of model fermentation broths components. Polish J. Chem. Technol. 2013, 15, 3–6. [Google Scholar] [CrossRef]
- Chaudhari, L.B.; Murthy, Z.V.P. Treatment of landfill leachates by nanofiltration. J. Environ. Manag. 2010, 91, 1209–1217. [Google Scholar] [CrossRef]
- Religa, P.; Kowalik-Klimczak, A.; Gierycz, P. Study on the behavior of nanofiltration membranes using for chromium(III) recovery from salt mixture solution. Desalination 2013, 315, 115–123. [Google Scholar] [CrossRef]
- Religa, P.; Kowalik, A.; Gierycz, P. Application of nanofiltration for chromium concentration in the tannery wastewater. J. Hazard. Mater. 2011, 186, 288–292. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.I.; Din Dar, M.U.; Bhat, R.A.; Singh, J.P.; Singh, K.; Bhat, S.A. Prospectives and challenges of wastewater treatment technologies to combat contaminants of emerging concerns. Ecol. Eng. 2020, 152, 105882. [Google Scholar] [CrossRef]
- Shen, J.; Huang, J.; Ruan, H.; Wang, J.; Van der Bruggen, B. Techno-economic analysis of resource recovery of glyphosate liquor by membrane technology. Desalination 2014, 342, 118–125. [Google Scholar] [CrossRef]
- Mohammad, A.W.; Teow, Y.H.; Ang, W.L.; Chung, Y.T.; Oatley-Radcliffe, D.L.; Hilal, N. Nanofiltration membranes review: Recent advances and future prospects. Desalination 2014, 356, 226–254. [Google Scholar] [CrossRef]
- Hilal, N.; Al-Zoubi, H.; Darwish, N.A.; Mohamma, A.W.; Abu Arabi, M. A comprehensive review of nanofiltration membranes: Treatment, pretreatment, modelling, and atomic force microscopy. Desalination 2004, 170, 281–308. [Google Scholar] [CrossRef]
- Schmidt, P.; Lutze, P. Characterisation of organic solvent nanofiltration membranes in multi-component mixtures: Phenomena-based modelling and membrane modelling maps. J. Membr. Sci. 2013, 445, 183–199. [Google Scholar] [CrossRef]
- Marchetti, P.; Jimenez Solomon, M.F.; Szekely, G.; Livingston, A.G. Molecular separation with organic solvent nanofiltration: A critical review. Chem. Rev. 2014, 114, 10735–10806. [Google Scholar] [CrossRef]
- Broniarz-Press, L.; Mitkowski, P.T.; Szaferski, W.; Marecka, A. Modelowanie procesu odzysku fumaranu diamonu metodą nanofiltracji. Inżynieria Apar. Chem. 2014, 53, 223–224. [Google Scholar]
- Mulder, M. Basic Principles of Membrane Technology, 2nd ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1996. [Google Scholar]
- Tanninen, J.; Mänttäri, M.; Nyström, M. Nanofiltration of concentrated acidic copper sulphate solutions. Desalination 2006, 189, 92–96. [Google Scholar] [CrossRef]
- Marecka-Migacz, A.; Mitkowski, P.T.; Antczak, J.; Różański, J.; Prochaska, K. Assessment of the Total Volume Membrane Charge Density through Mathematical Modeling for Separation of Succinic Acid Aqueous Solutions on Ceramic Nanofiltration Membrane. Processes 2019, 7, 559. [Google Scholar] [CrossRef]
- Izadpanah, A.A.; Javidnia, A. The ability of a nanofiltration membrane to remove hardness and ions from diluted seawater. Water 2012, 4, 283–294. [Google Scholar] [CrossRef]
- Bernata, X.; Fortuny, A.; Stüber, F.; Bengoa, C.; Fabregat, A.; Font, J. Recovery of iron (III) from aqueous streams by ultrafiltration. Desalination 2008, 221, 413–418. [Google Scholar] [CrossRef]
- Ramadan, Y.; Pátzay, G.; Szabó, G.T. Transport of NaCl, MgSO4, MgCl2 and Na2SO4 across DL type nanofiltration membrane. Period. Polytech. Chem. Eng. 2010, 54, 81–86. [Google Scholar] [CrossRef]
- Van Gestel, T.; Vandecasteele, C.; Buekenhoudt, A.; Dotremont, C.; Luyten, J.; Leysen, R.; Van der Bruggen, B.; Maes, G. Salt retention in nanofiltration with multilayer ceramic TiO2 membranes. J. Membr. Sci. 2002, 209, 379–389. [Google Scholar] [CrossRef]
- Drost, A.; Nedzarek, A.; Tórz, A. Reduction of proteins and products of their hydrolysis in process of cleaning post-production herring (Clupea harengus) marinating brines by using membranes. Membr. Water Treat. 2016, 7, 451–462. [Google Scholar] [CrossRef]
- Drost, A.; Nȩdzarek, A.; Bogusławska-Wa̧s, E.; Tõrz, A.; Bonisławska, M. UF application for innovative reuse of fish brine: Product quality, CCP management and the HACCP system. J. Food Process Eng. 2014, 37, 396–401. [Google Scholar] [CrossRef]
- Nędzarek, A.; Drost, A.; Harasimiuk, F.; Tórz, A.; Bonisławska, M. Application of ceramic membranes for microalgal biomass accumulation and recovery of the permeate to be reused in algae cultivation. J. Photochem. Photobiol. B Biol. 2015, 153, 367–372. [Google Scholar] [CrossRef]
- Kujawski, W.; Kujawa, J.; Wierzbowska, E.; Cerneaux, S.; Bryjak, M.; Kujawski, J. Influence of hydrophobization conditions and ceramic membranes pore size on their properties in vacuum membrane distillation of water-organic solvent mixtures. J. Membr. Sci. 2016, 499, 442–451. [Google Scholar] [CrossRef]
- Braeken, L.; Bettens, B.; Boussu, K.; Van der Meeren, P.; Cocquyt, J.; Vermant, J.; Van der Bruggen, B. Transport mechanisms of dissolved organic compounds in aqueous solution during nanofiltration. J. Membr. Sci. 2006, 279, 311–319. [Google Scholar] [CrossRef]
- Jun, B.M.; Cho, J.; Jang, A.; Chon, K.; Westerhoff, P.; Yoon, Y.; Rho, H. Charge characteristics (surface charge vs. zeta potential) of membrane surfaces to assess the salt rejection behavior of nanofiltration membranes. Sep. Purif. Technol. 2020, 247. [Google Scholar] [CrossRef]
- Oatley, D.L.; Llenas, L.; Pérez, R.; Williams, P.M.; Martínez-Lladó, X.; Rovira, M. Review of the dielectric properties of nanofiltration membranes and verification of the single oriented layer approximation. Adv. Colloid Interface Sci. 2012, 173, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, M.; Rosa, M.; Nystrom, M. The role of membrane charge on nanofiltration performance. J. Membr. Sci. 2005, 265, 160–166. [Google Scholar] [CrossRef]
- Puhlfürß, P.; Voigt, A.; Weber, R.; Morbé, M. Microporous TiO2 membranes with a cut off <500 Da. J. Membr. Sci. 2000, 174, 123–133. [Google Scholar]
- Su, B.; Duan, X.; Dou, M.; Gao, X.; Gao, C. Charge characteristics of nanofiltration membrane by streaming potential method. Adv. Mater. Res. 2011, 396–398, 547–551. [Google Scholar] [CrossRef]
- Peeters, J.M.M.; Mulder, M.H.V.; Strathmann, H. Streaming potential measurements as a characterization method for nanofiltration membranes. Colloids Surf. A Physicochem. Eng. Asp. 1999, 150, 247–259. [Google Scholar] [CrossRef]
- Pérez-González, A.; Ibáñez, R.; Gómez, P.; Urtiaga, A.M.; Ortiz, I.; Irabien, J.A. Nanofiltration separation of polyvalent and monovalent anions in desalination brines. J. Membr. Sci. 2015, 473, 16–27. [Google Scholar]
- Darvishmanesh, S.; Buekenhoudt, A.; Degrève, J.; Van der Bruggen, B. General model for prediction of solvent permeation through organic and inorganic solvent resistant nanofiltration membranes. J. Membr. Sci. 2009, 334, 43–49. [Google Scholar] [CrossRef]
- Tanimura, S.; Nakao, S.-I.; Kimura, S. Transport equation for a membrane based on a frictional model. J. Membr. Sci. 1993, 84, 79–91. [Google Scholar] [CrossRef]
- Nair, R.R.; Protasova, E.; Strand, S.; Bilstad, T. Implementation of spiegler–Kedem and steric hindrance pore models for analyzing nanofiltration membrane performance for smart water production. Membranes 2018, 8, 78. [Google Scholar] [CrossRef]
- Nair, R.R.; Protasova, E.; Strand, S.; Bilstad, T. Effect of ph on produced water treatment using nanofiltration membranes: Artificial neural network for performance assessment and steric hindrance pore model for flux variation evaluation. Desalin. Water Treat. 2019, 146, 120–130. [Google Scholar] [CrossRef]
- Bowen, W.R.; Mukhtar, H. Characterisation and prediction of separation performance of nanofiltration membranes. J. Membr. Sci. 1996, 112, 263–274. [Google Scholar] [CrossRef]
- Bowen, W.R.; Mohammad, A.W. Diafiltration by nanofiltration: Prediction and optimization. AIChE J. 1998, 44, 1799–1812. [Google Scholar] [CrossRef]
- Bowen, W.R.; Mohammad, A.W. A theoretical basis for specifying nanofiltration membranes—Dye/salt/water streams. Desalination 1998, 117, 257–264. [Google Scholar] [CrossRef]
- Bowen, W.R.; Welfoot, J.S. Predictive modelling of nanofiltration: Membrane specification and process optimisation. Desalination 2002, 147, 197–203. [Google Scholar] [CrossRef]
- Déon, S.; Dutournié, P.; Bourseau, P. Modeling nanofiltration with Nernst-Planck approach and polarization layer. AIChE J. 2007, 53, 1952–1969. [Google Scholar] [CrossRef]
- Santafé-Moros, A.; Gozálvez-Zafrilla, J.M.; Lora-García, J. Applicability of the DSPM with dielectric exclusion to a high rejection nanofiltration membrane in the separation of nitrate solutions. Desalination 2008, 221, 268–276. [Google Scholar] [CrossRef]
- Fadaei, F.; Shirazian, S.; Ashrafizadeh, S.N. Mass transfer modeling of ion transport through nanoporous media. Desalination 2011, 281, 325–333. [Google Scholar] [CrossRef]
- Szymczyk, A.; Lanteri, Y.; Fievet, P. Modelling the transport of asymmetric electrolytes through nanofiltration membranes. Desalination 2009, 245, 396–407. [Google Scholar] [CrossRef]
- Szymczyk, A.; Sbaï, M.; Fievet, P.; Vidonne, A. Transport properties and electrokinetic characterization of an amphoteric nanofilter. Langmuir 2006, 22, 3910–3919. [Google Scholar] [CrossRef]
- Kowalik-Klimczak, A.; Zalewski, M.; GIERYCZ, P. Prediction of the Chromium (Iii) Separation From Acidic Salt Solutions on Nanofiltration Membranes Using Donnan and Steric Partitioning Pore (Dsp) Model. Archit. Civ. Eng. Environ. 2016, 9, 135–140. [Google Scholar] [CrossRef]
- Kowalik-Klimczak, A.; Zalewski, M.; Gierycz, P. Removal of Cr(III) ions from salt solution by nanofiltration: Experimental and modelling analysis. Polish J. Chem. Technol. 2016, 18, 10–16. [Google Scholar] [CrossRef][Green Version]
- Xu, R.; Zhou, M.; Wang, H.; Wang, X.; Wen, X. Influences of temperature on the retention of PPCPs by nanofiltration membranes: Experiments and modeling assessment. J. Membr. Sci. 2020, 599, 117817. [Google Scholar] [CrossRef]
- Kingsbury, R.S.; Wang, J.; Coronell, O. Comparison of water and salt transport properties of ion exchange, reverse osmosis, and nanofiltration membranes for desalination and energy applications. J. Membr. Sci. 2020, 604, 117998. [Google Scholar] [CrossRef]
- Rall, D.; Schweidtmann, A.M.; Kruse, M.; Evdochenko, E.; Mitsos, A.; Wessling, M. Multi-scale membrane process optimization with high-fidelity ion transport models through machine learning. J. Membr. Sci. 2020, 608, 118208. [Google Scholar] [CrossRef]
- Farsi, A.; Boffa, V.; Christensen, M.L. Electroviscous effects in ceramic nanofiltration membranes. ChemPhysChem 2015, 16, 3397–3407. [Google Scholar] [CrossRef]
- Van Gestel, T.; Vandecasteele, C.; Buekenhoudt, A.; Dotremont, C.; Luyten, J.; Leysen, R.; Van Der Bruggen, B.; Maes, G. Alumina and titania multilayer membranes for nanofiltration: Preparation, characterization and chemical stability. J. Membr.Sci. 2002, 207, 73–89. [Google Scholar] [CrossRef]
- Nędzarek, A.; Drost, A.; Harasimiuk, F.B.; Tórz, A. The influence of pH and BSA on the retention of selected heavy metals in the nanofiltration process using ceramic membrane. Desalination 2015, 369, 62–67. [Google Scholar] [CrossRef]
- Bonisławska, M.; Nędzarek, A.; Rybczyk, A. Assessment of the use of precipitating agents and ceramic membranes for treatment of effluents with high concentrations of nitrogen and phosphorus from recirculating aquaculture systems. Aquac. Res. 2019, 50, 1248–1256. [Google Scholar] [CrossRef]
- Labbez, C.; Fievet, P.; Szymczyk, A.; Vidonne, A.; Foissy, A.; Pagetti, J. Analysis of the salt retention of a titania membrane using the “DSPM” model: Effect of pH, salt concentration and nature. J. Membr. Sci. 2002, 208, 315–329. [Google Scholar] [CrossRef]
- Ribeiro, A.C.F.; Esteso, M.A.; Lobo, V.M.M.; Valente, A.J.M.; Simões, S.M.N.; Sobral, A.J.F.N.; Burrows, H.D. Diffusion coefficients of copper chloride in aqueous solutions at 298.15 K and 310.15 K. J. Chem. Eng. Data 2005, 50, 1986–1990. [Google Scholar] [CrossRef]
- Atomic and Ionic Radii. Available online: http://www.wiredchemist.com/chemistry/data/atomic-and-ionic-radii (accessed on 7 July 2018).
- Kariuki, S.; Dewald, H.D. Evaluation of diffusion coefficients of metallic ions in aqueous solutions. Electroanalysis 2005, 8, 307–313. [Google Scholar] [CrossRef]
- Perry, R.; Green, D.; Maloney, J. Perry’s Chemical Engineers’ Handbook; McGraw-Hill: New York, NY, USA, 1997; ISBN 0-07-115448-5. [Google Scholar]
- Jenkins, H.D.B.; Thakur, K.P. Reappraisal of thermochemical radii for complex ions. J. Chem. Educ. 1979, 56, 576. [Google Scholar] [CrossRef]
- Krishna, R.; Wesselingh, J.A. The Maxwell-Stefan approach to mass transfer. Chem. Eng. Sci. 1997, 52, 861–911. [Google Scholar] [CrossRef]
- Samson, E.; Marchand, J.; Snuder, K.A. Calculation of ionic diffusion coefficients on the basis of migration test results. Mater. Struct. 2003, 36, 156–165. [Google Scholar] [CrossRef]
- Process Systems Enterprise Limited. gPROMS Model Builder Documentation 2014; Release 4.0.0; Process Systems Enterprise Limited: London, UK, April 2014. [Google Scholar]
- Peeters, J.M.M.; Boom, J.P.; Mulder, M.H.V.; Strathmann, H. Retention measurements of nanofiltration membranes with electrolyte solutions. J. Membr. Sci. 1998, 145, 199–209. [Google Scholar] [CrossRef]
- Takagi, R.; Larbot, A.; Cot, L.; Nakagaki, M. Effect of Al2O3 support on electrical properties of TiO2/Al2O3 membrane formed by sol–gel method. J. Membr. Sci. 2000, 177, 33–40. [Google Scholar] [CrossRef]
- Mazzoni, C.; Bruni, L.; Bandini, S. Nanofiltration: Role of the electrolyte and pH on Desal DK performances. Ind. Eng. Chem. Res. 2007, 46, 2254–2262. [Google Scholar] [CrossRef]
- Elimelech, M.; Chen, W.H.; Waypa, J.J. Measuring the zeta (electrokinetic) potential of reverse osmosis membranes by a streaming potential analyzer. Desalination 1994, 95, 269–286. [Google Scholar] [CrossRef]
- Schaep, J.; Vandecasteele, C. Evaluating the charge of nanofiltration membranes. J. Membr. Sci. 2001, 188, 129–136. [Google Scholar] [CrossRef]
- Mullet, M.; Fievet, P.; Reggiani, J.; Pagetti, J. Surface electrochemical properties of mixed oxide ceramic membranes: Zeta-potential and surface charge density. J. Membr. Sci. 1997, 123, 255–265. [Google Scholar] [CrossRef]


| Differential Variables | Number | |
| Concentration of ion in the membrane [mol/m3] | ci | NC |
| Algebraic and Implicit Variables | Number | |
| Potential gradient inside membrane pore [V] | ψ | 1 |
| Ratio of solute to pore radius [-] | λi | NC |
| Steric term [-] | φi | NC |
| Hindrance factor for diffusion [-] | Kd,i | NC |
| Hindrance factor for convection [-] | Kc,i | NC |
| Ion concentration in the permeate [mol/m3] | Cp.i | NC |
| Retention coefficient [-] | Ri | NC |
| Solvent velocity [m3/m2/s] | Vs | 1 |
| Donnan potential [V] | ψD | 1 |
| Osmotic pressure difference [Pa] | Δπ | 1 |
| Osmotic pressure on the feed side [Pa] | πfeed | 1 |
| Osmotic pressure on the permeate side [Pa] | πpermeate | 1 |
| Parameters and Known Variable | Number | |
| Effective membrane charge density [mol/m3] | Xd | 1 |
| Molar fraction on the feed side [mol/mol] | xf,i | NC |
| Molar fraction on the permeate side [mol/mol] | xp,i | NC |
| Pore radii [m] | rp | 1 |
| Ion radii [m] | rs,i | NC |
| Transmembrane pressure [Pa] | ΔP | 1 |
| Ideal gas constant [J/(mol⋅K] | R | 1 |
| Faraday constant [C/mol] | F | 1 |
| Temperature [K] | T | 1 |
| Solvent viscosity [Pa⋅s] | ηs | 1 |
| Thickness of membrane active layer [m] | Δx | 1 |
| Molar volume of water [m3/mol] | Ṽw | 1 |
| Diffusion coefficient of ion [m2/s] | Di | NC |
| Charge of individual ion [e] | zi | NC |
| Ion concentration in the feed [mol/m3] | Cf,i | NC |
| Ion concentration in the membrane in the surface directly contacting with the feed [mol/m3] | cm1,i | NC |
| Ion concentration in the membrane in the surface directly contacting with the permeate [mol/m3] | cm2,i | NC |
| Total number of variables: | ||
| Equation Description | Equations | Number of Equations |
|---|---|---|
| Solvent velocity based on Hagen–Poiseuille-type relationship | (2) | 1 |
| Osmotic pressure difference across the membrane | (3) | 1 |
| Osmotic pressure at the feed side | (4) | 1 |
| Osmotic pressure at the permeate side | (5) | 1 |
| Ratio of the solute radii to the pore radii | (6) | NC |
| Steric partitioning coefficient | (7) | NC |
| Hindrance factor for diffusion | (8) | NC |
| Hindrance factor for convection | (9) | NC |
| Concentration gradient inside the membrane pore | (10) | NC |
| Potential gradient inside the membrane pore | (11) | 1 |
| Electroneutrality conditions in the membrane | (12) | 1 |
| Electroneutrality conditions in the permeate | (13) | 1 |
| Donnan–Steric partitioning | (14) | NC |
| Retention coefficient | (15) | NC |
| Total Number of Equations: 7 + 7·NC | ||
| Ion | Diffusion Coefficient, Di [m2/s] | Size of Ion/Molecule, rs,i [m] |
|---|---|---|
| Cu2+ | 1.24 × 10−9 [63] | 7.7 × 10−11 [64] |
| Fe3+ | 7.19 × 10−9 [65] | 6.0 × 10−11 [64] |
| Zn2+ | 5.18 × 10−8 [65] | 7.4 × 10−11 [64] |
| Pb2+ | 8.45 × 10−9 [65] | 11.9 × 10−11 [64] |
| 1.25 × 10−9 [66] | 1.79 × 10−10 [67] | |
| H+ | 4.50 × 10−9 [68] | 0.9 × 10−9 [64] |
| Na+ | 1.33 × 10−9 [69] | 0.1 × 10−9 [64] |
| OH− | 5.27 × 10−9 [69] | 1.33 × 10−10 [67] |
| Variant | Heavy Metal Ion * | Heavy Metal Ion Concentration [mol/m3] | pH | ||||
|---|---|---|---|---|---|---|---|
| 1-Set | 2-Set | 3-Set | 4-Set | 5-Set | |||
| 1. | Cu2+ | 7.87 × 10−3 | 2.0 | 4.6 | 6.0 | 6.9 | 9.0 |
| 2. | Fe3+ | 8.95 × 10−3 | 2.0 | 4.6 | 6.0 | 6.9 | 9.0 |
| 3. | Zn2+ | 7.69 × 10−3 | 2.0 | 4.6 | 6.0 | 6.9 | 9.0 |
| 4. | Pb2+ | 2.41 × 10−3 | 2.0 | 4.6 | 6.0 | 6.9 | 9.0 |
| pH | Cu2+ | Fe3+ | Zn2+ | Pb2+ |
|---|---|---|---|---|
| 2.0 | −0.00082 | −0.00262 | −0.00628 | −0.00532 |
| 4.6 | −0.00307 | −0.00521 | −0.00594 | −0.00981 |
| 6.0 | 0.00873 | −0.00250 | −0.00618 | −0.00207 |
| 6.9 | 0.01954 | −0.00202 | −0.00432 | 0.00461 |
| 9.0 | 0.01933 | −0.00080 | −0.00328 | 0.00229 |
| Set of Parameters | Parameters | Fe3+ | Cu2+ | Zn2+ | Pb2+ |
|---|---|---|---|---|---|
| First set of parameters | a | 4.44 | 342.07 | −76.42 | −247.56 |
| b | −199.89 | −2251.68 | 1557.69 | 3754.86 | |
| c | −40.6 | 2369.91 | −6684.97 | −14,589.5 | |
| d | 1 | 1 | 1 | 1 | |
| e | −6.55 | −12.30 | −11.72 | −14.27 | |
| f | 12.59 | 40.73 | 35.28 | 53.18 | |
| Se | 8.03 | 0.549 | 14.276 | 0.408 | |
| Second set of parameters | a | - | 277.57 | −92.68 | −223.51 |
| b | - | −1795.30 | 2263.50 | 3104.14 | |
| c | - | 1630.37 | −10,754.65 | −11,272.98 | |
| d | - | 1.00 | 1.00 | 1.00 | |
| e | - | −12.77 | −12.77 | −12.77 | |
| f | - | 43.06 | 43.06 | 43.06 | |
| Se | - | 3.19 | 106.74 | 24.85 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marecka-Migacz, A.; Mitkowski, P.T.; Nędzarek, A.; Różański, J.; Szaferski, W. Effect of pH on Total Volume Membrane Charge Density in the Nanofiltration of Aqueous Solutions of Nitrate Salts of Heavy Metals. Membranes 2020, 10, 235. https://doi.org/10.3390/membranes10090235
Marecka-Migacz A, Mitkowski PT, Nędzarek A, Różański J, Szaferski W. Effect of pH on Total Volume Membrane Charge Density in the Nanofiltration of Aqueous Solutions of Nitrate Salts of Heavy Metals. Membranes. 2020; 10(9):235. https://doi.org/10.3390/membranes10090235
Chicago/Turabian StyleMarecka-Migacz, Agata, Piotr Tomasz Mitkowski, Arkadiusz Nędzarek, Jacek Różański, and Waldemar Szaferski. 2020. "Effect of pH on Total Volume Membrane Charge Density in the Nanofiltration of Aqueous Solutions of Nitrate Salts of Heavy Metals" Membranes 10, no. 9: 235. https://doi.org/10.3390/membranes10090235
APA StyleMarecka-Migacz, A., Mitkowski, P. T., Nędzarek, A., Różański, J., & Szaferski, W. (2020). Effect of pH on Total Volume Membrane Charge Density in the Nanofiltration of Aqueous Solutions of Nitrate Salts of Heavy Metals. Membranes, 10(9), 235. https://doi.org/10.3390/membranes10090235







