The Roles of Membrane Technology in Artificial Organs: Current Challenges and Perspectives
Abstract
1. Introduction
2. Membrane-Based Artificial Organs
2.1. Artificial Lung (Blood Oxygenation Membranes)
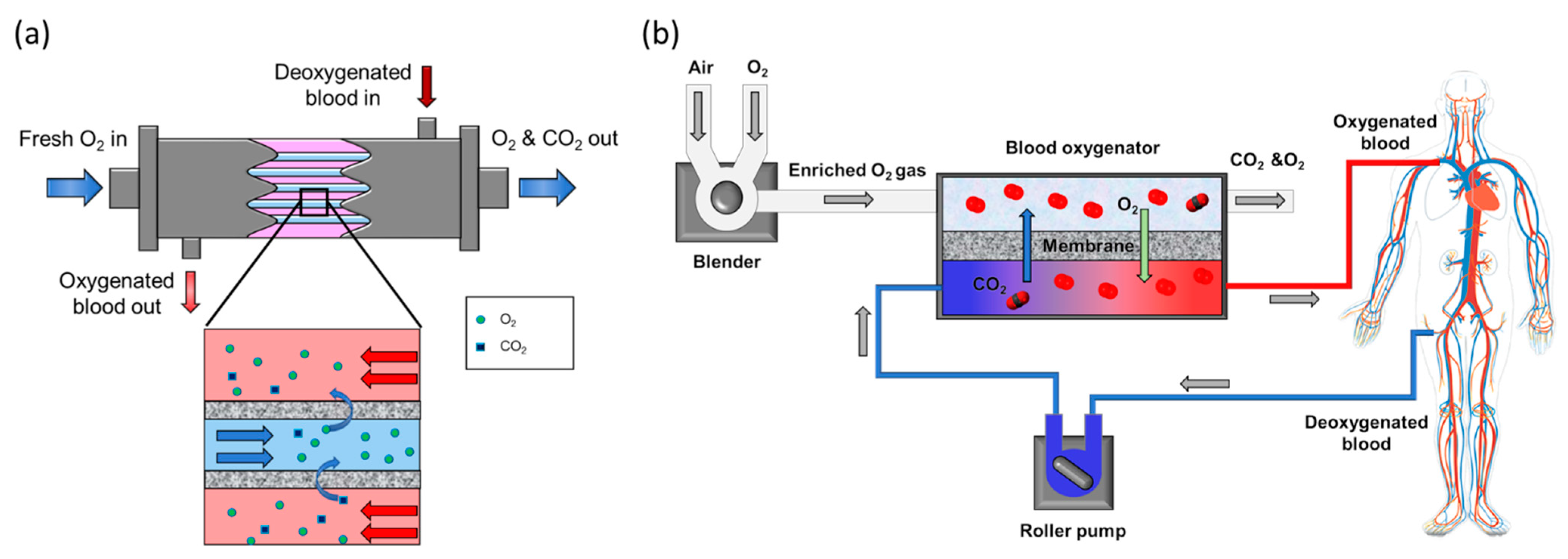
2.1.1. Blood Oxygenation Physiology and the Role of Membrane
2.1.2. Membranes for Artificial Lung
2.1.3. Current Challenges and Research Direction
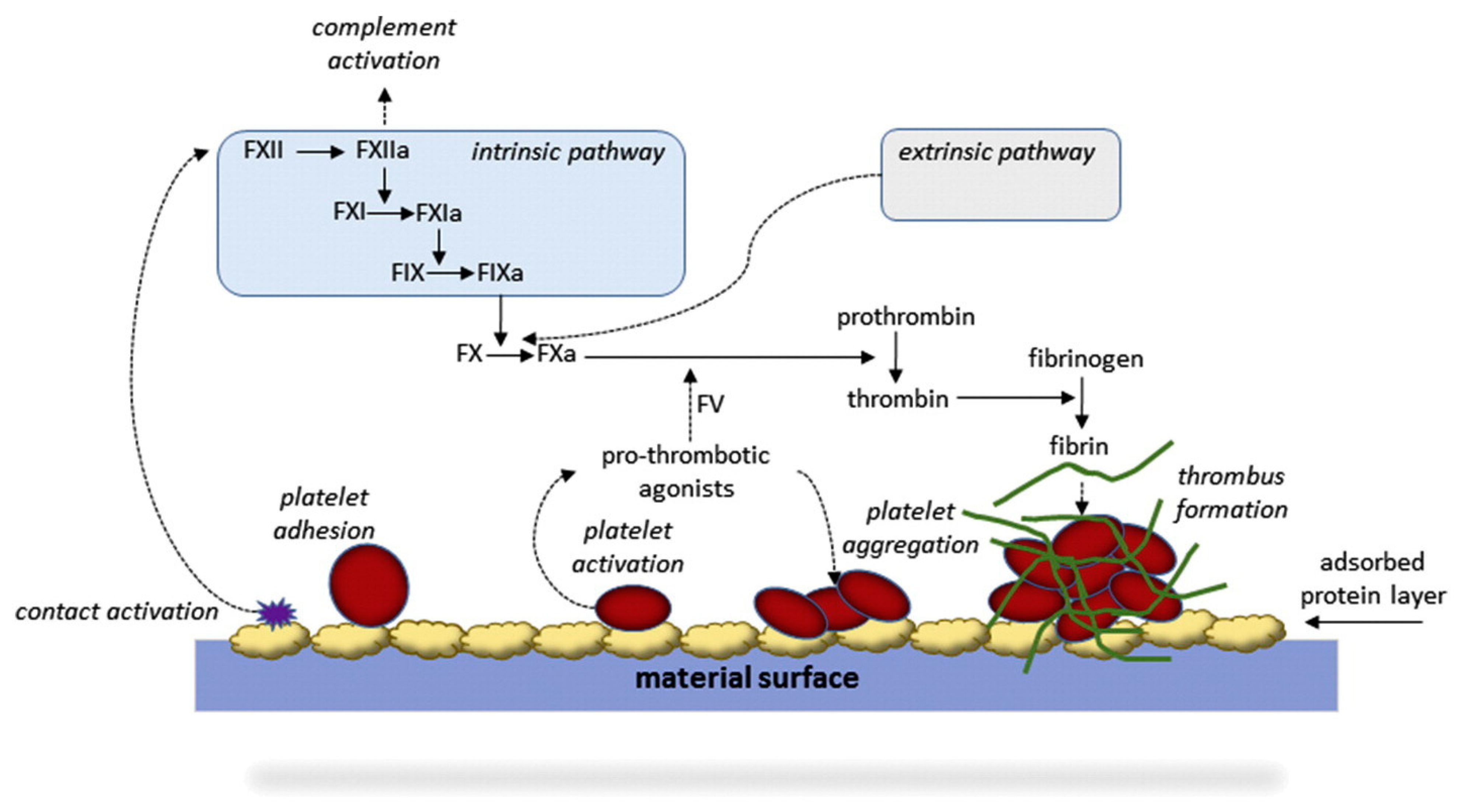
Blood Compatibility (Hemocompatibility)
Improving Blood Oxygenation Efficiency
Wearable and Implantable Artificial Lung
2.2. Artificial Kidney and Blood Purification
2.3. Artificial Liver
2.4. Bioartificial Pancreas
3. Summary and Conclusions
Funding
Conflicts of Interest
Abbreviations
| Abbreviation | Full Name |
| AI | Artificial intelligence |
| AN62 | Acrylonitrile |
| AO | Artificial organ |
| BAK | Bioartificial kidney |
| BAL | Bioartificial liver |
| BAP | Bioartificial pancreas |
| BLSS | Bioartificial liver support system |
| CFD | Computational fluid dynamic |
| CIT | Clinical islet transplantation |
| CPB | Cardiopulmonary bypass system |
| CTA | Cellulose-triacetate |
| cTAL | Compliant thoracic artificial lung |
| ECMO | Extracorporeal membrane oxygenation |
| ELAP | Extracorporeal liver assist device |
| EVAL | Ethyl-vinyl-acetate copolymer |
| FPSA | Fractionated plasma separation and adsorption |
| GPU | Gas permeation unit |
| HD | Hemodialysis |
| HDF | Hemodiafiltration |
| HF | Hemofiltration |
| HF | Hollow fiber |
| HFP | Hexafluoropropylene |
| IVOX | Intravenacaval oxygenator and carbon dioxide removal device |
| MARS | Molecular adsorbent recycling system |
| MW | Molecular weight |
| MWCO | Molecular weight cut-off |
| NIPS | Nonsolvent induced phase separation |
| N-TIPS | Nonsolvent-thermally induced phase separation |
| P(VDF-CO-HFP) | Poly(vinylidene fluoride)-co-hexafluoropropylene |
| PAAL | Paracorporeal ambulatory assist lung |
| PAES | Polyarylethersulfones |
| PAN | Polyacrylontrile |
| PAN-PVC | Polyacrylontrile-polyvinylchloride |
| PC | Phosphorylcholine |
| PDMS | Polydimethylsiloxane |
| PES | Polyethersulfone |
| PMEA | Poly(2-methoxyethylacrylate) |
| PMMA | Polymethylmethacrylate |
| PMP | Poly-4-methylpentence |
| POSS | Polyhedral oligomeric silsesquioxanes |
| PP | Polypropylene |
| Psu | Polysulfone |
| PTFE | Polytetrafluoroethylene |
| PVDF | Poly(vinylidene fluoride) |
| PVP | Polyvinylpyrrolidone |
| RAD | Renal tubule assist device |
| SEPET | Selective plasma filtration therapy |
| SPAD | Single-pass albumin dialysis |
| TIPS | Thermally induced phase separation |
| WAK | Wearable artificial kidney |
References
- Cascalho, M.; Platt, J.L. The future of organ replacement: Needs, potential applications, and obstacles to application. Transplant. Proc. 2006, 38, 362–364. [Google Scholar] [CrossRef]
- Zhang, J.; Wei, X.; Zeng, R.; Xu, F.; Li, X. Stem cell culture and differentiation in microfluidic devices toward organ-on-a-chip. Future Sci. OA 2017, 3, FSO187. [Google Scholar] [CrossRef]
- Marcheque, J.; Bussolati, B.; Csete, M.; Perin, L. Concise reviews: Stem cells and kidney regeneration: An update. Stem Cells Transl. Med. 2019, 8, 82–92. [Google Scholar] [CrossRef] [PubMed]
- Pellegrini, G.; Rama, P.; Di Rocco, A.; Panaras, A.; De Luca, M. Concise review: Hurdles in a successful example of limbal stem cell-based regenerative medicine. Stem Cells 2014, 32, 26–34. [Google Scholar] [CrossRef] [PubMed]
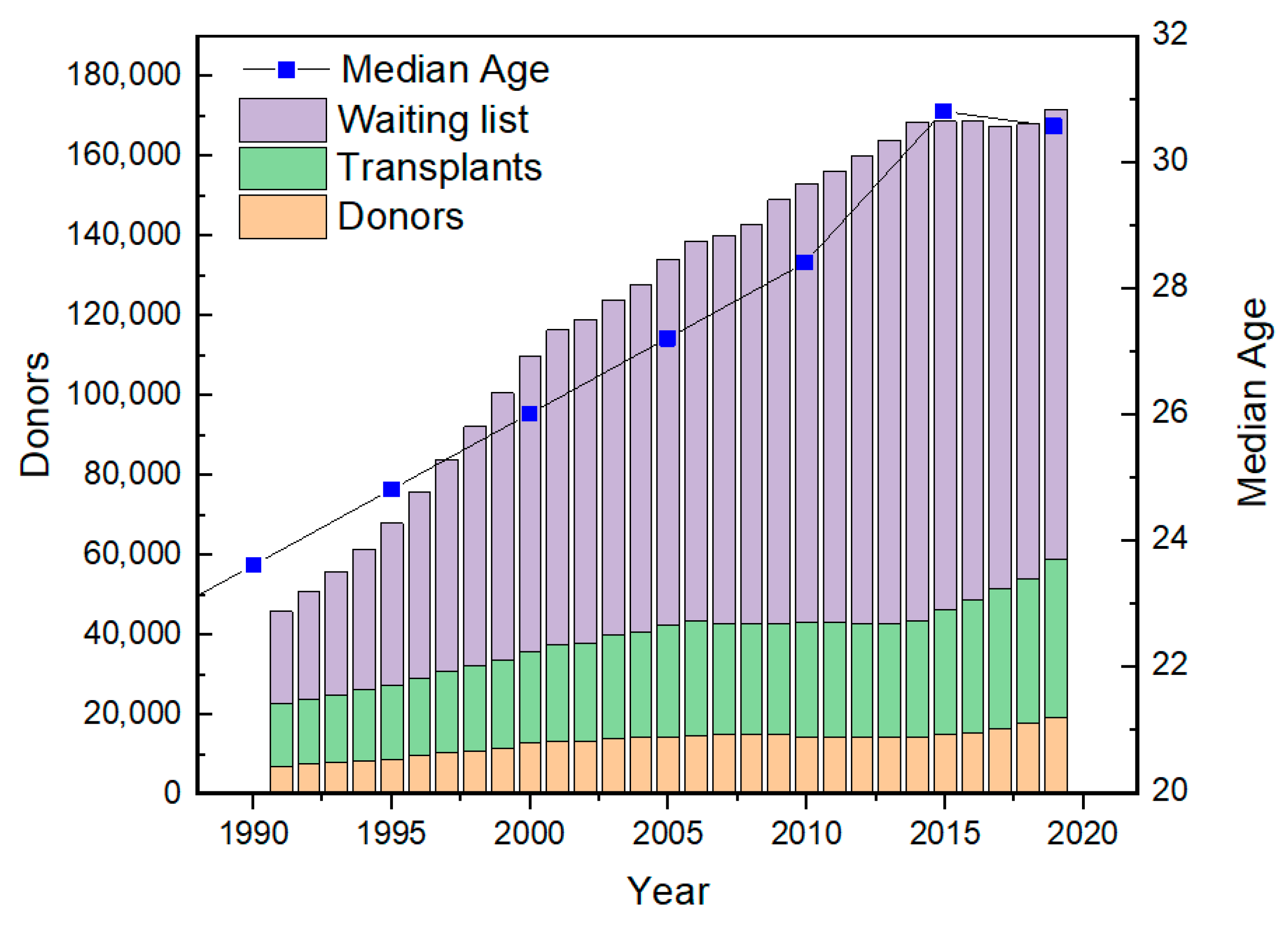
- Health Services & Resources Administration. Available online: https://www.organdonor.gov/statistics-stories/statistics.html (accessed on 15 January 2021).
- Worldometers. Available online: https://www.worldometers.info/ (accessed on 15 January 2021).
- Lattemann, S.; Kennedy, M.D.; Schippers, J.C.; Amy, G. Global desalination situation. Sustain. Sci. Eng. 2010, 2, 7–39. [Google Scholar]
- Pera-Titus, M. Porous inorganic membranes for CO2 capture: Present and prospects. Chem. Rev. 2014, 114, 1413–1492. [Google Scholar] [CrossRef] [PubMed]
- Dimitrios, S. Biomedical Membranes and (Bio) Artificial Organs; World Scientific: Singapore, 2017; Volume 2. [Google Scholar]
- Marketsandmarkets. Hemodialysis and Peritoneal Dialysis Market by Product (Machines, Dialyzers, Concentrates, Dialysates, Catheters) Services, Modality (Conventional, CAPD, Nocturnal), End User (Hospital, Independent Dialysis Center, Home Dialysis)—Global Forecasts to 2024; MarktsandMarkts: Northbrook, IL, USA, 2019. [Google Scholar]
- Researchandmarkets. Extracorporeal Membrane Oxygenation Machine Market by Modality, Application and Region—Analysis & Forecasts to 2021; MarktsandMarkts: Northbrook, IL, USA, 2017. [Google Scholar]
- Lillehei, C.W. Controlled cross circulation for direct-vision intracardiac surgery: Correction of ventricular septal defects, atrioventricularis communis, and tetralogy of Fallot. Postgrad. Med. 1955, 17, 388–396. [Google Scholar] [CrossRef] [PubMed]
- DeWall, R.; Warden, H.; Varco, R.; Lillehei, C. The helix reservoir pump-oxygenator. Surg. Gynecol. Obstet. 1957, 104, 699. [Google Scholar]
- Clowes, G., Jr. An artificial lung dependent upon diffusion of oxygen and carbon dioxide through plastic membrane. J. Thorac. Surg 1956, 32, 630–637. [Google Scholar] [CrossRef]
- Gassmann, C.J.; Galbraith, G.D.; Smith, R.G. Evaluation of three types of membrane oxygenators and their suitability for use with pulsatile flow. J. Extracorpor. Technol. 1987, 19, 297–304. [Google Scholar]
- Stamatialis, D.F.; Papenburg, B.J.; Girones, M.; Saiful, S.; Bettahalli, S.N.; Schmitmeier, S.; Wessling, M. Medical applications of membranes: Drug delivery, artificial organs and tissue engineering. J. Membr. Sci. 2008, 308, 1–34. [Google Scholar] [CrossRef]
- Supady, A.; DellaVolpe, J.; Taccone, F.S.; Scharpf, D.; Ulmer, M.; Lepper, P.M.; Halbe, M.; Ziegeler, S.; Vogt, A.; Ramanan, R.; et al. Outcome Prediction in Patients with Severe COVID-19 Requiring Extracorporeal Membrane Oxygenation—A Retrospective International Multicenter Study. Membranes 2021, 11, 170. [Google Scholar] [CrossRef] [PubMed]
- Punjabi, P.P.; Taylor, K. The science and practice of cardiopulmonary bypass: From cross circulation to ECMO and SIRS. Glob. Cardiol. Sci. Pract. 2013, 2013, 249. [Google Scholar] [CrossRef] [PubMed]
- Hemmila, M.R.; Rowe, S.A.; Boules, T.N.; Miskulin, J.; McGillicuddy, J.W.; Schuerer, D.J.; Haft, J.W.; Swaniker, F.; Arbabi, S.; Hirschl, R.B.; et al. Extracorporeal life support for severe acute respiratory distress syndrome in adults. Ann. Surg. 2004, 240, 595–605. [Google Scholar] [CrossRef]
- Peek, G.J.; Moore, H.M.; Moore, N.; Sosnowski, A.W.; Firmin, R.K. Extracorporeal membrane oxygenation for adult respiratory failure. Chest 1997, 112, 759–764. [Google Scholar] [CrossRef]
- Lewandowski, K.; Rossaint, R.; Pappert, D.; Gerlach, H.; Slama, K.J.; Weidemann, H.; Frey, D.J.; Hoffmann, O.; Keske, U.; Falke, K.J. High survival rate in 122 ARDS patients managed according to a clinical algorithm including extracorporeal membrane oxygenation. Intensive Care Med. 1997, 23, 819–835. [Google Scholar] [CrossRef] [PubMed]
- Ullrich, R.; Lorber, C.; Röder, G.; Urak, G.; Faryniak, B.; Sladen, R.N.; Germann, P. Controlled airway pressure therapy, nitric oxide inhalation, prone position, and extracorporeal membrane oxygenation (ECMO) as components of an integrated approach to ARDS. Anesthesiology 1999, 91, 1577–1586. [Google Scholar] [CrossRef] [PubMed]
- Rich, P.B.; Awad, S.S.; Kolla, S.; Annich, G.; Schreiner, R.J.; Hirschl, R.B.; Bartlett, R.H. An approach to the treatment of severe adult respiratory failure. J. Crit. Care 1998, 13, 26–36. [Google Scholar] [CrossRef]
- Kolla, S.; Awad, S.S.; Rich, P.B.; Schreiner, R.J.; Hirschl, R.B.; Bartlett, R.H. Extracorporeal life support for 100 adult patients with severe respiratory failure. Ann. Surg. 1997, 226, 544–564. [Google Scholar] [CrossRef] [PubMed]
- Davies, A.; Jones, D.; Bailey, M.; Beca, J.; Bellomo, R.; Blackwell, N.; Forrest, P.; Gattas, D.; Granger, E.; Herkes, R.; et al. Extracorporeal Membrane Oxygenation for 2009 Influenza A(H1N1) Acute Respiratory Distress Syndrome. JAMA 2009, 302, 1888–1895. [Google Scholar] [CrossRef] [PubMed]
- Posluszny, J.; Rycus, P.T.; Bartlett, R.H.; Engoren, M.; Haft, J.W.; Lynch, W.R.; Park, P.K.; Raghavendran, K.; Napolitano, L.M. Outcome of Adult Respiratory Failure Patients Receiving Prolonged (≥14 Days) ECMO. Ann. Surg. 2016, 263, 573–581. [Google Scholar] [CrossRef] [PubMed]
- Robba, C.; Ortu, A.; Bilotta, F.; Lombardo, A.; Sekhon, M.S.; Gallo, F.; Matta, B.F. Extracorporeal membrane oxygenation for adult respiratory distress syndrome in trauma patients: A case series and systematic literature review. J. Trauma Acute Care Surg. 2017, 82, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Boissier, F.; Bagate, F.; Schmidt, M.; Labbé, V.; Kimmoun, A.; Fartoukh, M.; Mekontso Dessap, A. Extracorporeal Life Support for Severe Acute Chest Syndrome in Adult Sickle Cell Disease: A Preliminary Report. Crit. Care Med. 2019, 47, e263–e265. [Google Scholar] [CrossRef]
- Ochs, M.; Nyengaard, J.R.; Jung, A.; Knudsen, L.; Voigt, M.; Wahlers, T.; Richter, J.; Gundersen, H.J.G. The number of alveoli in the human lung. Am. J. Respir. Crit. Care Med. 2004, 169, 120–124. [Google Scholar] [CrossRef]
- Johnson, R.; Hsia, C. Anatomy and Physiology of the Human Respiratory System; WIT Press: Boston, MA, USA, 2006; pp. 1–29. [Google Scholar]
- Goutelle, S.; Maurin, M.; Rougier, F.; Barbaut, X.; Bourguignon, L.; Ducher, M.; Maire, P. The Hill equation: A review of its capabilities in pharmacological modelling. Fundam. Clin. Pharmacol. 2008, 22, 633–648. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.V. The possible effects of the aggregation of the molecules of haemoglobin on its dissociation curves. J. Physiol. 1910, 40, 4–7. [Google Scholar]
- Des Jardins, T. Cardiopulmonary Anatomy & Physiology: Essentials of Respiratory Care; Nelson Education: Scarborough, ON, Canada, 2012. [Google Scholar]
- Annesini, M.C.; Marrelli, L.; Piemonte, V.; Turchetti, L. Artificial Organ Engineering; Springer: Berlin/Heidelberg, Germany, 2017. [Google Scholar]
- Tsakiridis, K.; Mpakas, A.; Kesisis, G.; Arikas, S.; Argyriou, M.; Siminelakis, S.; Zarogoulidis, P.; Katsikogiannis, N.; Kougioumtzi, I.; Tsiouda, T. Lung inflammatory response syndrome after cardiac-operations and treatment of lornoxicam. J. Thorac. Dis. 2014, 6, S78. [Google Scholar] [PubMed]
- Nagase, K.; Kohori, F.; Sakai, K. Oxygen transfer performance of a membrane oxygenator composed of crossed and parallel hollow fibers. Biochem. Eng. J. 2005, 24, 105–113. [Google Scholar] [CrossRef]
- Low, K.W.; Van Loon, R.; Rolland, S.A.; Sienz, J. Formulation of generalized mass transfer correlations for blood oxygenator design. J. Biomech. Eng. 2017, 139, 031007. [Google Scholar] [CrossRef] [PubMed]
- Wickramasinghe, S.; Goerke, A.; Garcia, J.; Han, B. Designing blood oxygenators. Ann. New York Acad. Sci. 2003, 984, 502–514. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, N.; Sakai, K. Blood flow and oxygen transfer rate of an outside blood flow membrane oxygenator. J. Membr. Sci. 2000, 170, 153–158. [Google Scholar] [CrossRef]
- Zierenberg, J.R.; Fujioka, H.; Cook, K.E.; Grotberg, J.B. Pulsatile flow and oxygen transport past cylindrical fiber arrays for an artificial lung: Computational and experimental studies. J. Biomech. Eng. 2008, 130, 031019. [Google Scholar] [CrossRef] [PubMed]
- Federspiel, W.J.; Henchir, K.A. Lung, artificial: Basic principles and current applications. Encycl. Biomater. Biomed. Eng. 2004, 9, 910. [Google Scholar]
- Awad, J.A.; Cloutier, R.; Fournier, L.; Major, D.; Martin, L.; Masson, M.; Guidoin, R. Pumpless respiratory assistance using a membrane oxygenator as an artificial placenta: A preliminary study in newborn and preterm lambs. J. Investig. Surg. 1995, 8, 21–30. [Google Scholar] [CrossRef]
- Kim, J.F.; Kim, J.H.; Lee, Y.M.; Drioli, E. Thermally induced phase separation and electrospinning methods for emerging membrane applications: A review. AIChE J. 2016, 62, 461–490. [Google Scholar] [CrossRef]
- Wieses, F.; Paul, D.; Possart, W.; Malsch, G.; Bossin, E. Grenzfläuchenenergie gequollener Polymermembranen. Acta Polym. 1990, 41, 95–98. [Google Scholar] [CrossRef]
- Wiese, F. Membranes for artificial lungs. Membr. Life Sci. 2007, 1, 49–68. [Google Scholar]
- LaFayette, N.G.; Schewe, R.E.; Montoya, J.P.; Cook, K.E. Performance of a MedArray silicone hollow fiber oxygenator. ASAIO J. 2009, 55, 382–387. [Google Scholar] [CrossRef]
- Toomasian, J.M.; Schreiner, R.J.; Meyer, D.E.; Schmidt, M.E.; Hagan, S.E.; Griffith, G.W.; Bartlett, R.H.; Cook, K.E. A Polymethylpentene Fiber Gas Exchanger for Long-Term Extracorporeal Life Support. ASAIO J. 2005, 51, 390–397. [Google Scholar] [CrossRef] [PubMed]
- Khoshbin, E.; Roberts, N.; Harvey, C.; Machin, D.; Killer, H.; Peek, G.J.; Sosnowski, A.W.; Firmin, R.K. Poly-Methyl Pentene Oxygenators Have Improved Gas Exchange Capability and Reduced Transfusion Requirements in Adult Extracorporeal Membrane Oxygenation. ASAIO J. 2005, 51, 281–287. [Google Scholar] [CrossRef]
- Jung, J.T.; Kim, J.F.; Wang, H.H.; Di Nicolo, E.; Drioli, E.; Lee, Y.M. Understanding the non-solvent induced phase separation (NIPS) effect during the fabrication of microporous PVDF membranes via thermally induced phase separation (TIPS). J. Membr. Sci. 2016, 514, 250–263. [Google Scholar] [CrossRef]
- Park, A.; Song, Y.; Yi, E.; Duy Nguyen, B.T.; Han, D.; Sohn, E.; Park, Y.; Jung, J.; Lee, Y.M.; Cho, Y.H. Blood Oxygenation Using Fluoropolymer-Based Artificial Lung Membranes. ACS Biomater. Sci. Eng. 2020, 6, 6424–6434. [Google Scholar] [CrossRef]
- Guisheng, F.; Incarnato, L.; Di Maio, L.; Acierno, D. Discussion about the use of relative values of permeabilities between two gases for high molecular weight polymers. Polymer 1995, 36, 4345–4346. [Google Scholar] [CrossRef]
- Allen, S.; Fujii, M.; Stannett, V.; Hopfenberg, H.; Williams, J. The barrier properties of polyacrylonitrile. J. Membr. Sci. 1977, 2, 153–163. [Google Scholar] [CrossRef]
- Lewis, J. Gas Separation Membranes: Polymeric and Inorganic by AF Ismail, KC Khulbe, and T. Matsuura. Chem. Eng. Educ. 2018, 52, 223. [Google Scholar]
- Toyoda, Y.; Bhama, J.K.; Shigemura, N.; Zaldonis, D.; Pilewski, J.; Crespo, M.; Bermudez, C. Efficacy of extracorporeal membrane oxygenation as a bridge to lung transplantation. J. Thorac. Cardiovasc. Surg. 2013, 145, 1065–1071. [Google Scholar] [CrossRef] [PubMed]
- Ratner, B.D. The Blood Compatibility Catastrophe; Wiley Online Library: Hoboken, NJ, USA, 1993. [Google Scholar]
- Davies, M.; Hagen, P.O. Systemic inflammatory response syndrome. Br. J. Surg. 1997, 84, 920–935. [Google Scholar] [CrossRef]
- Valipour Motlagh, N.; Birjandi, F.C.; Sargolzaei, J.; Shahtahmassebi, N. Durable, superhydrophobic, superoleophobic and corrosion resistant coating on the stainless steel surface using a scalable method. Appl. Surf. Sci. 2013, 283, 636–647. [Google Scholar] [CrossRef]
- Zhou, G.; Groth, T. Host Responses to Biomaterials and Anti-Inflammatory Design—a Brief Review. Macromol. Biosci. 2018, 18, 1800112. [Google Scholar] [CrossRef] [PubMed]
- Marasco, S.F.; Lukas, G.; McDonald, M.; McMillan, J.; Ihle, B. Review of ECMO (extra corporeal membrane oxygenation) support in critically ill adult patients. Heartlung Circ. 2008, 17, S41–S47. [Google Scholar] [CrossRef] [PubMed]
- Bennett, M.; Horton, S.; Thuys, C.; Augustin, S.; Rosenberg, M.; Brizard, C. Pump-induced haemolysis: A comparison of short-term ventricular assist devices. Perfusion 2004, 19, 107–111. [Google Scholar] [CrossRef]
- Philipp, A.; Foltan, M.; Gietl, M.; Reng, M.; Liebold, A.; Kobuch, R.; Keyl, C.; Bein, T.; Müller, T.; Schmid, F. Interventionelle extrakorporale Lungenunterstützung (ILA) mittels arterio-venösem Shunt und einem neu entwickelten Low Resistance Lung Assist Device (LAD). Kardiotechnik 2003, 1, 7–13. [Google Scholar]
- Biran, R.; Pond, D. Heparin coatings for improving blood compatibility of medical devices. Adv. Drug Deliv. Rev. 2017, 112, 12–23. [Google Scholar] [CrossRef]
- Cheung, P.-Y.; Sawicki, G.; Salas, E.; Etches, P.C.; Schulz, R.; Radomski, M.W. The mechanisms of platelet dysfunction during extracorporeal membrane oxygenation in critically ill neonates. Crit. Care Med. 2000, 28, 2584–2590. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, I.; Pangule, R.C.; Kane, R.S. Antifouling coatings: Recent developments in the design of surfaces that prevent fouling by proteins, bacteria, and marine organisms. Adv. Mater. 2011, 23, 690–718. [Google Scholar] [CrossRef] [PubMed]
- Mérian, T.; Goddard, J.M. Advances in Nonfouling Materials: Perspectives for the Food Industry. J. Agric. Food Chem. 2012, 60, 2943–2957. [Google Scholar] [CrossRef]
- Koc, Y.; de Mello, A.J.; McHale, G.; Newton, M.I.; Roach, P.; Shirtcliffe, N.J. Nano-scale superhydrophobicity: Suppression of protein adsorption and promotion of flow-induced detachment. Lab A Chip 2008, 8, 582–586. [Google Scholar] [CrossRef]
- Deng, X.; Mammen, L.; Butt, H.-J.; Vollmer, D. Candle Soot as a Template for a Transparent Robust Superamphiphobic Coating. Science 2012, 335, 67–70. [Google Scholar] [CrossRef] [PubMed]
- Paven, M.; Papadopoulos, P.; Schöttler, S.; Deng, X.; Mailänder, V.; Vollmer, D.; Butt, H.-J. Super liquid-repellent gas membranes for carbon dioxide capture and heart–lung machines. Nat. Commun. 2013, 4, 2512. [Google Scholar] [CrossRef] [PubMed]
- Hsu, L.-C. Biocompatibility in cardiopulmonary bypass. J. Cardiothorac. Vasc. Anesth. 1997, 11, 376–382. [Google Scholar] [CrossRef]
- Janvier, G.; Baquey, C.; Roth, C.; Benillan, N.; Belisle, S.; Hardy, J.-F. Extracorporeal circulation, hemocompatibility, and biomaterials. Ann. Thorac. Surg. 1996, 62, 1926–1934. [Google Scholar] [CrossRef]
- Aulin, C.; Yun, S.H.; Wågberg, L.; Lindström, T. Design of Highly Oleophobic Cellulose Surfaces from Structured Silicon Templates. ACS Appl. Mater. Interfaces 2009, 1, 2443–2452. [Google Scholar] [CrossRef] [PubMed]
- Ruthven, D.M. Principles of Adsorption and Adsorption Processes; John Wiley & Sons: Hoboken, NJ, USA, 1984. [Google Scholar]
- Rabinovich, Y.I.; Adler, J.J.; Esayanur, M.S.; Ata, A.; Singh, R.K.; Moudgil, B.M. Capillary forces between surfaces with nanoscale roughness. Adv. Colloid Interface Sci. 2002, 96, 213–230. [Google Scholar] [CrossRef]
- Hsieh, C.-T.; Wu, F.-L.; Chen, W.-Y. Superhydrophobicity and superoleophobicity from hierarchical silica sphere stacking layers. Mater. Chem. Phys. 2010, 121, 14–21. [Google Scholar] [CrossRef]
- Srinivasan, S.; Chhatre, S.S.; Mabry, J.M.; Cohen, R.E.; McKinley, G.H. Solution spraying of poly(methyl methacrylate) blends to fabricate microtextured, superoleophobic surfaces. Polymer 2011, 52, 3209–3218. [Google Scholar] [CrossRef]
- Tuteja, A.; Choi, W.; Ma, M.; Mabry, J.M.; Mazzella, S.A.; Rutledge, G.C.; McKinley, G.H.; Cohen, R.E. Designing Superoleophobic Surfaces. Science 2007, 318, 1618–1622. [Google Scholar] [CrossRef]
- Zhao, H.; Law, K.-Y.; Sambhy, V. Fabrication, Surface Properties, and Origin of Superoleophobicity for a Model Textured Surface. Langmuir 2011, 27, 5927–5935. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, C.-T.; Chen, J.-M.; Kuo, R.-R.; Lin, T.-S.; Wu, C.-F. Influence of surface roughness on water- and oil-repellent surfaces coated with nanoparticles. Appl. Surf. Sci. 2005, 240, 318–326. [Google Scholar] [CrossRef]
- Bico, J.; Thiele, U.; Quéré, D. Wetting of textured surfaces. Colloids Surf. A Physicochem. Eng. Asp. 2002, 206, 41–46. [Google Scholar] [CrossRef]
- Tuteja, A.; Choi, W.; Mabry, J.M.; McKinley, G.H.; Cohen, R.E. Robust omniphobic surfaces. Proc. Natl. Acad. Sci. USA 2008, 105, 18200–18205. [Google Scholar] [CrossRef]
- Steele, A.; Bayer, I.; Loth, E. Inherently Superoleophobic Nanocomposite Coatings by Spray Atomization. Nano Lett. 2009, 9, 501–505. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Tian, Y.; Jiang, L. Bio-inspired superoleophobic and smart materials: Design, fabrication, and application. Prog. Mater. Sci. 2013, 58, 503–564. [Google Scholar] [CrossRef]
- Choudhury, A.; Barve, S.; Chutia, J.; Pal, A.; Kishore, R.; Pande, M.; Patil, D. RF-PACVD of water repellent and protective HMDSO coatings on bell metal surfaces: Correlation between discharge parameters and film properties. Appl. Surf. Sci. 2011, 257, 8469–8477. [Google Scholar] [CrossRef]
- Hsieh, C.-T.; Wu, F.-L.; Chen, W.-Y. Super water- and oil-repellencies from silica-based nanocoatings. Surf. Coat. Technol. 2009, 203, 3377–3384. [Google Scholar] [CrossRef]
- Yuan, Z.; Xiao, J.; Wang, C.; Zeng, J.; Xing, S.; Liu, J. Preparation of a superamphiphobic surface on a common cast iron substrate. J. Coat. Technol. Res. 2011, 8, 773. [Google Scholar] [CrossRef]
- Ellinas, K.; Tserepi, A.; Gogolides, E. From Superamphiphobic to Amphiphilic Polymeric Surfaces with Ordered Hierarchical Roughness Fabricated with Colloidal Lithography and Plasma Nanotexturing. Langmuir 2011, 27, 3960–3969. [Google Scholar] [CrossRef]
- Wang, D.; Wang, X.; Liu, X.; Zhou, F. Engineering a Titanium Surface with Controllable Oleophobicity and Switchable Oil Adhesion. J. Phys. Chem. C 2010, 114, 9938–9944. [Google Scholar] [CrossRef]
- Zhang, J.; Seeger, S. Superoleophobic Coatings with Ultralow Sliding Angles Based on Silicone Nanofilaments. Angew. Chem. Int. Ed. 2011, 50, 6652–6656. [Google Scholar] [CrossRef] [PubMed]
- Ceria, A.; Hauser, P.J. Atmospheric plasma treatment to improve durability of a water and oil repellent finishing for acrylic fabrics. Surf. Coat. Technol. 2010, 204, 1535–1541. [Google Scholar] [CrossRef]
- Guo, F.; Su, X.; Hou, G.; Li, P. Bioinspired fabrication of stable and robust superhydrophobic steel surface with hierarchical flowerlike structure. Colloids Surf. A Physicochem. Eng. Asp. 2012, 401, 61–67. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhu, X.; Yang, J.; Xu, X.; Men, X.; Zhou, X. Facile fabrication of superoleophobic surfaces with enhanced corrosion resistance and easy repairability. Appl. Phys. A 2012, 108, 601–606. [Google Scholar] [CrossRef]
- Holmquist, H.; Schellenberger, S.; van Der Veen, I.; Peters, G.; Leonards, P.; Cousins, I.T. Properties, performance and associated hazards of state-of-the-art durable water repellent (DWR) chemistry for textile finishing. Environ. Int. 2016, 91, 251–264. [Google Scholar] [CrossRef]
- Kettwich, S.C.; Pierson, S.N.; Peloquin, A.J.; Mabry, J.M.; Iacono, S.T. Anomalous macromolecular assembly of partially fluorinated polyhedral oligomeric silsesquioxanes. New J. Chem. 2012, 36, 941–946. [Google Scholar] [CrossRef]
- Thompson, A.J.; Ma, L.J.; Major, T.; Jeakle, M.; Lautner-Csorba, O.; Goudie, M.J.; Handa, H.; Rojas-Peña, A.; Potkay, J.A. Assessing and improving the biocompatibility of microfluidic artificial lungs. Acta Biomater. 2020, 112, 190–201. [Google Scholar] [CrossRef]
- Weber, N.; Wendel, H.P.; Ziemer, G. Hemocompatibility of heparin-coated surfaces and the role of selective plasma protein adsorption. Biomaterials 2002, 23, 429–439. [Google Scholar] [CrossRef]
- De Somer, F.; Van Belleghem, Y.; Caes, F.; Francois, K.; Arnout, J.; Bossuyt, X.; Taeymans, Y.; Van Nooten, G. Phosphorylcholine coating offers natural platelet preservation during cardiopulmonary bypass. Perfusion 2002, 17, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Ranucci, M.; Isgro, G.; Soro, G.; Canziani, A.; Menicanti, L.; Frigiola, A. Reduced systemic heparin dose with phosphorylcholine coated closed circuit in coronary operations. Int. J. Artif. Organs 2004, 27, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Ranucci, M.; Pazzaglia, A.; Isgro, G.; Cazzaniga, A.; Ditta, A.; Boncilli, A.; Cotza, M.; Carboni, G.; Brozzi, S.; Bonifazi, C. Closed, phosphorylcholine-coated circuit and reduction of systemic heparinization for cardiopulmonary bypass: The intraoperative ECMO concept. Int. J. Artif. Organs 2002, 25, 875–881. [Google Scholar] [CrossRef]
- Onishi, M.; Shimura, K.; Seita, Y.; Yamashita, S. Design of a new plasma separation membrane by graft copolymerization. Radiat. Phys. Chem. 1995, 46, 219–223. [Google Scholar] [CrossRef]
- Ikuta, T.; Fujii, H.; Shibata, T.; Hattori, K.; Hirai, H.; Kumano, H.; Suehiro, S. A new poly-2-methoxyethylacrylate-coated cardiopulmonary bypass circuit possesses superior platelet preservation and inflammatory suppression efficacy. Ann. Thorac. Surg. 2004, 77, 1678–1683. [Google Scholar] [CrossRef] [PubMed]
- McGuigan, A.P.; Sefton, M.V. The influence of biomaterials on endothelial cell thrombogenicity. Biomaterials 2007, 28, 2547–2571. [Google Scholar] [CrossRef] [PubMed]
- Inoue, T.; Takagl, M.; Sawa, Y.; Ohata, T.; Masai, T.; Shirakura, R.; Matsuda, H.; Yoshida, T. Oxygen transfer rate of a hybrid artificial lung model. ASAIO J. 1996, 42, 93. [Google Scholar] [CrossRef]
- Takagi, M.; Haraguchi, T.; Kawai, H.; Shiwaku, K.; Inoue, T.; Sawa, Y.; Matsuda, H.; Yoshida, T. Enhanced adhesion of endothelial cells onto a polypropylene hollow-fiber membrane by plasma discharge treatment and high inoculum cell density. J. Artif. Organs 2001, 4, 220–225. [Google Scholar] [CrossRef]
- Sawa, Y.; Ohata, T.; Takagi, M.; Suhara, H.; Matsuda, H. Development of hybrid artificial lung with gene transfected biological cells. J. Artif. Organs 2000, 3, 1–4. [Google Scholar] [CrossRef]
- Pflaum, M.; Kühn-Kauffeldt, M.; Schmeckebier, S.; Dipresa, D.; Chauhan, K.; Wiegmann, B.; Haug, R.J.; Schein, J.; Haverich, A.; Korossis, S. Endothelialization and characterization of titanium dioxide-coated gas-exchange membranes for application in the bioartificial lung. Acta Biomater. 2017, 50, 510–521. [Google Scholar] [CrossRef] [PubMed]
- Polk, A.A.; Maul, T.M.; McKeel, D.T.; Snyder, T.A.; Lehocky, C.A.; Pitt, B.; Stolz, D.B.; Federspiel, W.J.; Wagner, W.R. A biohybrid artificial lung prototype with active mixing of endothelialized microporous hollow fibers. Biotechnol. Bioeng. 2010, 106, 490–500. [Google Scholar] [CrossRef]
- Wiegmann, B.; von Seggern, H.; Höffler, K.; Korossis, S.; Dipresa, D.; Pflaum, M.; Schmeckebier, S.; Seume, J.; Haverich, A. Developing a biohybrid lung–sufficient endothelialization of poly-4-methly-1-pentene gas exchange hollow-fiber membranes. J. Mech. Behav. Biomed. Mater. 2016, 60, 301–311. [Google Scholar] [CrossRef] [PubMed]
- Sakai, K. Comparison of oxygenation and flow characteristics of inside and outside blood flow membrane oxygenators. Artif. Organs Today 1993, 3, 57–80. [Google Scholar]
- Matsuda, N.; Sakai, K.; Yamamoto, K.-I.; Iwasaki, H. Effects of hollow fiber packing fraction on blood flow pattern and gas transfer rate of an intravascular oxygenator (IVOX). J. Membr. Sci. 2000, 179, 231–241. [Google Scholar] [CrossRef]
- Funakubo, A.; Taga, I.; McGillicuddy, J.W.; Fukui, Y.; Hirschl, R.B.; Bartlett, R.H. Flow vectorial analysis in an artificial implantable lung. ASAIO J. 2003, 49, 383–387. [Google Scholar] [PubMed]
- Catapano, G.; Hornscheidt, R.; Wodetzki, A.; Baurmeister, U. Turbulent flow technique for the estimation of oxygen diffusive permeability of membranes for the oxygenation of blood and other cell suspensions. J. Membr. Sci. 2004, 230, 131–139. [Google Scholar] [CrossRef]
- Zhang, J.; Nolan, T.D.; Zhang, T.; Griffith, B.P.; Wu, Z.J. Characterization of membrane blood oxygenation devices using computational fluid dynamics. J. Membr. Sci. 2007, 288, 268–279. [Google Scholar] [CrossRef]
- Yang, M.C.; Lin, C.C. In vitro characterization of the occurrence of hemolysis during extracorporeal blood circulation using a mini hemodialyzer. ASAIO J. 2000, 46, 293–297. [Google Scholar] [CrossRef] [PubMed]
- Tabesh, H.; Amoabediny, G.; Poorkhalil, A.; Khachab, A.; Kashefi, A.; Mottaghy, K. A theoretical model for evaluation of the design of a hollow-fiber membrane oxygenator. J. Artif. Organs 2012, 15, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Hirano, A.; Yamamoto, K.-i.; Matsuda, M.; Inoue, M.; Nagao, S.; Kuwana, K.; Kamiya, M.; Sakai, K. Flow uniformity in oxygenators with different outlet port design. ASAIO J. 2009, 55, 209–212. [Google Scholar] [CrossRef] [PubMed]
- Nolan, H.; Wang, D.; Zwischenberger, J.B. Artificial lung basics: Fundamental challenges, alternative designs and future innovations. Organogenesis 2011, 7, 23–27. [Google Scholar] [CrossRef][Green Version]
- Chambers, S.; Merz, S.; Mcgillicuddy, J.; Bartlett, R. Development of the MC3 BioLung/spl trade. In Proceedings of the Second Joint 24th Annual Conference and the Annual Fall Meeting of the Biomedical Engineering Society: Engineering in Medicine and Biology, Houston, TX, USA, 23–26 October 2002; pp. 1581–1582. [Google Scholar]
- Guzmán, A.M.; Escobar, R.A.; Amon, C.H. Flow mixing enhancement from balloon pulsations in an intravenous oxygenator. J. Biomech. Eng. 2005, 127, 400–415. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hattler, B.G.; Reeder, G.D.; Sawzik, P.J.; Lund, L.W.; Walters, F.R.; Shah, A.S.; Rawleigh, J.; Goode, J.S.; Klain, M.; Borovetz, H.S. Development of an intravenous membrane oxygenator: Enhanced intravenous gas exchange through convective mixing of blood around hollow fiber membranes. Artif. Organs 1994, 18, 806–812. [Google Scholar] [CrossRef]
- Conrad, S.A.; Bagley, A.; Bagley, B.; Schaap, R.N. Major findings from the clinical trials of the intravascular oxygenator. Artif. Organs 1994, 18, 846–863. [Google Scholar] [CrossRef]
- Kolobow, T.; Bowman, R.L. Construction and evaluation of an alveolar membrane artificial heart-lung. ASAIO J. 1963, 9, 238–243. [Google Scholar]
- Hung, T.-K.; Borovetz, H.S.; Weissman, M.H. Transport and flow phenomena in a microchannel membrane oxygenator. Ann. Biomed. Eng. 1977, 5, 343–361. [Google Scholar] [CrossRef]
- Federspiel, W.J.; Svitek, R.G. Lung, Artificial: Current Research and Future Directions; CRC Press: Boca Raton, FL, USA, 2004; pp. 922–931. [Google Scholar]
- Lee, J.K.; Kung, H.H.; Mockros, L.F. Microchannel technologies for artificial lungs: (1) theory. ASAIO J. 2008, 54, 372–382. [Google Scholar] [CrossRef] [PubMed]
- Kung, M.; Lee, J.; Kung, H.H.; Mockros, L. Microchannel technologies for artificial lungs: (2) screen-filled wide rectangular channels. ASAIO J. (Am. Soc. Artif. Intern. Organs 1992) 2008, 54, 383. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Kung, M.; Kung, H.; Mockros, L. Microchannel technologies for artificial lungs: (3) open rectangular channels. ASAIO J. (Am. Soc. Artif. Intern. Organs 1992) 2008, 54, 390. [Google Scholar] [CrossRef]
- Burgess, K.A.; Hu, H.-H.; Wagner, W.R.; Federspiel, W.J. Towards microfabricated biohybrid artificial lung modules for chronic respiratory support. Biomed. Microdev. 2009, 11, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Potkay, J.A. The promise of microfluidic artificial lungs. Lab. A Chip 2014, 14, 4122–4138. [Google Scholar] [CrossRef]
- Kniazeva, T.; Epshteyn, A.A.; Hsiao, J.C.; Kim, E.S.; Kolachalama, V.B.; Charest, J.L.; Borenstein, J.T. Performance and scaling effects in a multilayer microfluidic extracorporeal lung oxygenation device. Lab. A Chip 2012, 12, 1686–1695. [Google Scholar] [CrossRef]
- Shin, J.; Ko, J.; Jeong, S.; Won, P.; Lee, Y.; Kim, J.; Hong, S.; Jeon, N.L.; Ko, S.H. Monolithic digital patterning of polydimethylsiloxane with successive laser pyrolysis. Nat. Mater. 2021, 20, 100–107. [Google Scholar] [CrossRef]
- Borenstein, J.T.; Vunjak-Novakovic, G. Engineering tissue with BioMEMS. IEEE Pulse 2011, 2, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.J.; Zhang, T.; Bianchi, G.; Wei, X.; Son, H.-S.; Zhou, K.; Sanchez, P.G.; Garcia, J.; Griffith, B.P. Thirty-day in-vivo performance of a wearable artificial pump-lung for ambulatory respiratory support. Ann. Thorac. Surg. 2012, 93, 274–281. [Google Scholar] [CrossRef]
- Madhani, S.P.; Frankowski, B.J.; Burgreen, G.W.; Antaki, J.F.; Kormos, R.; D’Cunha, J.; Federspiel, W.J. In vitro and in vivo evaluation of a novel integrated wearable artificial lung. J. Heart Lung Transplant. 2017, 36, 806–811. [Google Scholar] [CrossRef]
- Madhani, S.P.; Frankowski, B.J.; Ye, S.-H.; Burgreen, G.W.; Wagner, W.R.; Kormos, R.; D’Cunha, J.; Federspiel, W.J. In Vivo 5 Day Animal Studies of a Compact, Wearable Pumping Artificial Lung. ASAIO J. 2019, 65, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Schewe, R.E.; Khanafer, K.M.; Arab, A.; Mitchell, J.A.; Skoog, D.J.; Cook, K.E. Design and in vitro assessment of an improved, low-resistance compliant thoracic artificial lung. ASAIO J. 2012, 58, 583–589. [Google Scholar] [CrossRef] [PubMed]
- Zwischenberger, J.B.; Jr, V.J.C.; Tao, W.; Niranjan, S.C.; Clark, J.W.; Bidani, A. Intravascular Membrane Oxygenation and Carbon Dioxide Removal with IVOX: Can Improved Design and Permissive Hypercapnia Achieve Adequate Respiratory Support During Severe Respiratory Failure? Artif. Organs 1994, 18, 833–839. [Google Scholar] [CrossRef] [PubMed]
- Hattler, B.G.; Lund, L.W.; Golob, J.; Russian, H.; Lann, M.F.; Merrill, T.L.; Frankowski, B.; Federspiel, W.J. A respiratory gas exchange catheter: In vitro and in vivo tests in large animals. J. Thorac. Cardiovasc. Surg. 2002, 124, 520–530. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Graham, T. VII. The Bakerian lecture.—On osmotic force. Philos. Trans. R. Soc. Lond. 1854, 144, 177–228. [Google Scholar]
- Sakai, K. Dialysis membranes for blood purification. Front. Med. Biol. Eng. 2000, 10, 117–129. [Google Scholar] [CrossRef]
- Azar, A.T.; Canaud, B. Hemodialysis system. In Modelling and Control of Dialysis Systems; Springer: Berlin/Heidelberg, Germany, 2013; pp. 99–166. [Google Scholar]
- Curtis, J. Splitting fibers: Understanding how dialyzer differences can impact adequacy. Nephrol. News Issues 2001, 15, 36–39. [Google Scholar] [PubMed]
- Karkar, A. Advances in hemodialysis techniques. In Hemodialysis; IntechOpen: London, UK, 2013. [Google Scholar]
- Bello, G.; Di, F.M.; Maviglia, R.; Antonelli, M. New membranes for extracorporeal blood purification in septic conditions. Minerva Anestesiol. 2012, 78, 1265–1281. [Google Scholar] [PubMed]
- Eloot, S.; Ledebo, I.; Ward, R.A. Extracorporeal removal of uremic toxins: Can we still do better? Semin. Nephrol. 2014, 34, 209–227. [Google Scholar] [CrossRef]
- Henderson, L.W.; Chenoweth, D.E. Cellulose membranes—Time for a change. In Disputed Issues in Renal Failure Therapy; Karger Publishers: Basel, Switzerland, 1985; Volume 44, pp. 112–126. [Google Scholar]
- Johnson, R.J.; Lelah, M.D.; Sutliff, T.M.; Boggs, D.R. A modification of cellulose that facilitates the control of complement activation. Blood Purif. 1990, 8, 318–328. [Google Scholar] [CrossRef]
- Sunohara, T.; Masuda, T. Cellulose triacetate as a high-performance membrane. In High-Performance Membrane Dialyzers; Karger Publishers: Basel, Switzerland, 2011; Volume 173, pp. 156–163. [Google Scholar]
- Streicher, E.; Schneider, H. Polysulphone membrane mimicking human glomerular basement membrane. Lancet 1983, 322, 1136. [Google Scholar] [CrossRef]
- Thomas, M.; Moriyama, K.; Ledebo, I. AN69: Evolution of the world’s first high permeability membrane. In High-Performance Membrane Dialyzers; Karger Publishers: Basel, Switzerland, 2011; Volume 173, pp. 119–129. [Google Scholar]
- Fabbrini, P.; Sirtori, S.; Casiraghi, E.; Pieruzzi, F.; Genovesi, S.; Corti, D.; Brivio, R.; Gregorini, G.; Como, G.; Carati, M. Polymethylmethacrylate membrane and serum free light chain removal: Enhancing adsorption properties. Blood Purif. 2013, 35, 52–58. [Google Scholar] [CrossRef]
- Nakano, A. Ethylene vinyl alcohol co-polymer as a high-performance membrane: An EVOH membrane with excellent biocompatibility. In High-Performance Membrane Dialyzers; Karger Publishers: Basel, Switzerland, 2011; Volume 173, pp. 164–171. [Google Scholar]
- Aebischer, P.; Ip, T.K.; Panol, G.; Galletti, P.M. The bioartificial kidney: Progress towards an ultrafiltration device with renal epithelial cells processing. Life Support Syst. 1987, 5, 159–168. [Google Scholar]
- Humes, H.D.; Fissell, W.H.; Weitzel, W.F.; Buffington, D.A.; Westover, A.J.; MacKay, S.M.; Gutierrez, J.M. Metabolic replacement of kidney function in uremic animals with a bioartificial kidney containing human cells. Am. J. Kidney Dis. 2002, 39, 1078–1087. [Google Scholar] [CrossRef]
- Oo, Z.Y.; Kandasamy, K.; Tasnim, F.; Zink, D. A novel design of bioartificial kidneys with improved cell performance and haemocompatibility. J. Cell. Mol. Med. 2013, 17, 497–507. [Google Scholar] [CrossRef] [PubMed]
- Chevtchik, N.V.; Fedecostante, M.; Jansen, J.; Mihajlovic, M.; Wilmer, M.; Rüth, M.; Masereeuw, R.; Stamatialis, D. Upscaling of a living membrane for bioartificial kidney device. Eur. J. Pharmacol. 2016, 790, 28–35. [Google Scholar] [CrossRef]
- Ip, T.K.; Aebischer, P.; Calletti, P.M. Cellular control of membrane permeability: Implications for a bioartificial renal tubule. ASAIO J. 1988, 34, 351–355. [Google Scholar]
- Nielsen, R.; Birn, H.; Moestrup, S.K.; Nielsen, M.; Verroust, P.; Christensen, E.I. Characterization of a kidney proximal tubule cell line, LLC-PK1, expressing endocytotic active megalin. J. Am. Soc. Nephrol. 1998, 9, 1767–1776. [Google Scholar]
- Dukes, J.D.; Whitley, P.; Chalmers, A.D. The MDCK variety pack: Choosing the right strain. BMC Cell Biol. 2011, 12, 43. [Google Scholar] [CrossRef]
- Derakhshanfar, S.; Mbeleck, R.; Xu, K.; Zhang, X.; Zhong, W.; Xing, M. 3D bioprinting for biomedical devices and tissue engineering: A review of recent trends and advances. Bioact. Mater. 2018, 3, 144–156. [Google Scholar] [CrossRef] [PubMed]
- Sochol, R.D.; Gupta, N.R.; Bonventre, J.V. A role for 3D printing in kidney-on-a-chip platforms. Curr. Transplant. Rep. 2016, 3, 82–92. [Google Scholar] [CrossRef] [PubMed]
- Gura, V.; Rivara, M.B.; Bieber, S.; Munshi, R.; Smith, N.C.; Linke, L.; Kundzins, J.; Beizai, M.; Ezon, C.; Kessler, L. A wearable artificial kidney for patients with end-stage renal disease. JCI Insight 2016, 1, e86397. [Google Scholar] [CrossRef]
- Kooman, J.P.; Joles, J.A.; Gerritsen, K.G. Creating a Wearable Artificial Kidney: Where Are We Now? Taylor & Francis: Abingdon, UK, 2015. [Google Scholar]
- Ronco, C.; Davenport, A.; Gura, V. A wearable artificial kidney: Dream or reality? Nat. Clin. Pract. Nephrol. 2008, 4, 604–605. [Google Scholar] [CrossRef]
- Himmelfarb, J.; Ratner, B. Wearable artificial kidney: Problems, progress and prospects. Nat. Rev. Nephrol. 2020, 16, 558–559. [Google Scholar] [CrossRef]
- Meng, F.; Seredych, M.; Chen, C.; Gura, V.; Mikhalovsky, S.; Sandeman, S.; Ingavle, G.; Ozulumba, T.; Miao, L.; Anasori, B. MXene sorbents for removal of urea from dialysate: A step toward the wearable artificial kidney. ACS Nano 2018, 12, 10518–10528. [Google Scholar] [CrossRef]
- Shao, G.; Zang, Y.; Hinds, B.J. TiO2 Nanowires Based System for Urea Photodecomposition and Dialysate Regeneration. ACS Appl. Nano Mater. 2019, 2, 6116–6123. [Google Scholar] [CrossRef]
- van Gelder, M.K.; Jong, J.A.W.; Folkertsma, L.; Guo, Y.; Blüchel, C.; Verhaar, M.C.; Odijk, M.; Van Nostrum, C.F.; Hennink, W.E.; Gerritsen, K.G.F. Urea removal strategies for dialysate regeneration in a wearable artificial kidney. Biomaterials 2020, 234, 119735. [Google Scholar] [CrossRef]
- Hueso, M.; Navarro, E.; Sandoval, D.; Cruzado, J.M. Progress in the development and challenges for the use of artificial kidneys and wearable dialysis devices. Kidney Dis. 2019, 5, 3–10. [Google Scholar] [CrossRef]
- Venuthurupalli, S.K.; Hoy, W.E.; Healy, H.G.; Cameron, A.; Fassett, R.G. CKD screening and surveillance in Australia: Past, present, and future. Kidney Int. Rep. 2018, 3, 36–46. [Google Scholar] [CrossRef]
- Underhill, G.H.; Khetani, S.R.; Chen, A.A.; Bhatia, S.N. Chapter 48–Liver. In Principles of Tissue Engineering, 3rd ed.; Vacanti, R., Lanza, R., Langer, J., Eds.; Academic Press: Burlington, MA, USA, 2007; pp. 707–731. [Google Scholar]
- Wlodzimirow, K.A.; Eslami, S.; Abu-Hanna, A.; Nieuwoudt, M.; Chamuleau, R.A. A systematic review on prognostic indicators of acute on chronic liver failure and their predictive value for mortality. Liver Int. 2013, 33, 40–52. [Google Scholar] [CrossRef]
- Siami, G.A.; Siami, F.S. Membrane plasmapheresis in the United States: A review over the last 20 years. Ther. Apher. 2001, 5, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Winchester, J.F.; Ronco, C. Sorbent hemoperfusion in end-stage renal disease: An in-depth review. Adv. Ren. Replace. Ther. 2002, 9, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Di Paola, L. Chapter 1—Artificial kidney: A chemical engineering challenge. In Current Trends and Future Developments on (Bio-) Membranes; Basile, A., Annesini, M.C., Piemonte, V., Charcosset, C., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 1–20. [Google Scholar] [CrossRef]
- Annesini, M.C.; Piemonte, V.; Turchetti, L. Albumin-bound toxin removal in liver support devices: Case study of tryptophan adsorption and dialysis. Chem. Eng. Trans. 2008, 14, 365–372. [Google Scholar]
- McMenamy, R.H.; Oncley, J. The specific binding of L-tryptophan to serum albumin. J. Biol. Chem. 1958, 233, 1436–1447. [Google Scholar] [CrossRef]
- Falkenhagen, D.; Strobl, W.; Vogt, G.; Schrefl, A.; Linsberger, I.; Gerner, F.J.; Schoenhofen, M. Fractionated plasma separation and adsorption system: A novel system for blood purification to remove albumin bound substances. Artif. Organs 1999, 23, 81–86. [Google Scholar] [CrossRef]
- Campli, C.D.; Gaspari, R.; Mignani, V.; Stifano, G.; Santoliquido, A.; Verme, L.Z.D.; Proietti, R.; Pola, P.; Silveri, N.G.; Gasbarrini, G. Successful MARS treatment in severe cholestatic patients with acute on chronic liver failure. Artif. Organs 2003, 27, 565–569. [Google Scholar] [CrossRef]
- Kribben, A.; Gerken, G.; Haag, S.; Herget-Rosenthal, S.; Treichel, U.; Betz, C.; Sarrazin, C.; Hoste, E.; Van Vlierberghe, H.; Escorsell, À.; et al. Effects of Fractionated Plasma Separation and Adsorption on Survival in Patients With Acute-on-Chronic Liver Failure. Gastroenterology 2012, 142, 782–789.e3. [Google Scholar] [CrossRef]
- Pless, G.; Sauer, I.M. Bioartificial Liver: Current Status. Transplant. Proc. 2005, 37, 3893–3895. [Google Scholar] [CrossRef]
- Sivaraman, B.; Latour, R.A. The relationship between platelet adhesion on surfaces and the structure versus the amount of adsorbed fibrinogen. Biomaterials 2010, 31, 832–839. [Google Scholar] [CrossRef]
- Sperling, C.; Fischer, M.; Maitz, M.F.; Werner, C. Blood coagulation on biomaterials requires the combination of distinct activation processes. Biomaterials 2009, 30, 4447–4456. [Google Scholar] [CrossRef]
- Sussman, N.L.; Gislason, G.T.; Conlin, C.A.; Kelly, J.H. The Hepatix extracorporeal liver assist device: Initial clinical experience. Artif. Organs 1994, 18, 390–396. [Google Scholar] [CrossRef]
- Patzer, J.F.; Mazariegos, G.V.; Lopez, R.; Molmenti, E.; Gerber, D.; Riddervold, F.; Khanna, A.; YIN, W.Y.; Chen, Y.; Scott, V.L. Novel bioartificial liver support system: Preclinical evaluation. Ann. N. Y. Acad. Sci. 1999, 875, 340–352. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Ding, Y.; Xu, Q.; Yu, D. Bioartificial liver inoculated with porcine hepatocyte spheroids for treatment of canine acute liver failure model. Artif. Organs 2003, 27, 613–622. [Google Scholar] [CrossRef] [PubMed]
- De Bartolo, L.; Catapano, G.; Volpe, C.D.; Drioli, E. The effect of surface roughness of microporous membranes on the kinetics of oxygen consumption and ammonia elimination by adherent hepatocytes. J. Biomater. Sci. Polym. Ed. 1999, 10, 641–655. [Google Scholar] [CrossRef]
- De Bartolo, L.; Morelli, S.; Bader, A.; Drioli, E. Evaluation of cell behaviour related to physico-chemical properties of polymeric membranes to be used in bioartificial organs. Biomaterials 2002, 23, 2485–2497. [Google Scholar] [CrossRef]
- Demetriou, A.; Rozga, J.; Podesta, L.; Lepage, E.; Morsiani, E.; Moscioni, A.; Hoffman, A.; McGrath, M.; Kong, L.; Rosen, H. Early clinical experience with a hybrid bioartificial liver. Scand. J. Gastroenterol. 1995, 30, 111–117. [Google Scholar] [CrossRef]
- Gerlach, J.C.; Encke, J.; Hole, O.; Müller, C.; Ryan, C.; Neuhaus, P. Bioreactor for a larger scale hepatocyte in vitro perfusion. Transplantation 1994, 58, 984–988. [Google Scholar] [CrossRef]
- Shiraha, H.; Koide, N.; Hada, H.; Ujike, K.; Nakamura, M.; Shinji, T.; Gotoh, S.; Tsuji, T. Improvement of serum amino acid profile in hepatic failure with the bioartificial liver using multicellular hepatocyte spheroids. Biotechnol. Bioeng. 1996, 50, 416–421. [Google Scholar] [CrossRef]
- De Bartolo, L.; Salerno, S.; Curcio, E.; Piscioneri, A.; Rende, M.; Morelli, S.; Tasselli, F.; Bader, A.; Drioli, E. Human hepatocyte functions in a crossed hollow fiber membrane bioreactor. Biomaterials 2009, 30, 2531–2543. [Google Scholar] [CrossRef]
- Matsumura, K.N.; Guevara, G.R.; Huston, H.; Hamilton, W.; Rikimaru, M.; Yamasaki, G.; Matsumura, M. Hybrid bioartificial liver in hepatic failure: Preliminary clinical report. Surgery 1987, 101, 99–103. [Google Scholar] [PubMed]
- Sussman, N.L.; Chong, M.G.; Koussayer, T.; He, D.E.; Shang, T.A.; Whisennand, H.H.; Kelly, J.H. Reversal of fulminant hepatic failure using an extracorporeal liver assist device. Hepatology 1992, 16, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Ye, S.H.; Watanabe, J.; Takai, M.; Iwasaki, Y.; Ishihara, K. High functional hollow fiber membrane modified with phospholipid polymers for a liver assist bioreactor. Biomaterials 2006, 27, 1955–1962. [Google Scholar] [CrossRef] [PubMed]
- Berney, T.; Johnson, P.R. Donor pancreata: Evolving approaches to organ allocation for whole pancreas versus islet transplantation. Transplantation 2010, 90, 238–243. [Google Scholar] [CrossRef]
- Shapiro, A.J.; Ricordi, C.; Hering, B.J.; Auchincloss, H.; Lindblad, R.; Robertson, R.P.; Secchi, A.; Brendel, M.D.; Berney, T.; Brennan, D.C. International trial of the Edmonton protocol for islet transplantation. New Engl. J. Med. 2006, 355, 1318–1330. [Google Scholar] [CrossRef]
- Grundfest-Broniatowski, S.F.; Tellioglu, G.; Rosenthal, K.S.; Kang, J.; Erdodi, G.; Yalcin, B.; Cakmak, M.; Drazba, J.; Bennett, A.; Lu, L. A new bioartificial pancreas utilizing amphiphilic membranes for the immunoisolation of porcine islets: A pilot study in the canine. ASAIO J. 2009, 55, 400–405. [Google Scholar] [CrossRef]
- Barton, F.B.; Rickels, M.R.; Alejandro, R.; Hering, B.J.; Wease, S.; Naziruddin, B.; Oberholzer, J.; Odorico, J.S.; Garfinkel, M.R.; Levy, M. Improvement in outcomes of clinical islet transplantation: 1999–2010. Diabetes Care 2012, 35, 1436–1445. [Google Scholar] [CrossRef]
- McCall, M.; Shapiro, A.J. Update on islet transplantation. Cold Spring Harb. Perspect. Med. 2012, 2, a007823. [Google Scholar] [CrossRef]
- Schweicher, J.; Nyitray, C.; Desai, T.A. Membranes to achieve immunoprotection of transplanted islets. Front. Biosci. (Landmark Ed.) 2014, 19, 49. [Google Scholar] [CrossRef]
- Iacovacci, V.; Ricotti, L.; Menciassi, A.; Dario, P. The bioartificial pancreas (BAP): Biological, chemical and engineering challenges. Biochem. Pharmacol. 2016, 100, 12–27. [Google Scholar] [CrossRef]
- Li, R.H. Materials for immunoisolated cell transplantation. Adv. Drug Deliv. Rev. 1998, 33, 87–109. [Google Scholar] [CrossRef]
- Sun, A.M.; Parisius, W.; Healy, G.M.; Vacek, I.; Macmorine, H.G. The use, in diabetic rats and monkeys, of artificial capillary units containing cultured islets of Langerhans (artificial endocrine pancreas). Diabetes 1977, 26, 1136–1139. [Google Scholar] [CrossRef]
- Scharp, D.W.; Mason, N.S.; Sparks, R.E. Islet immuno-isolation: The use of hybrid artificial organs to prevent islet tissue rejection. World J. Surg. 1984, 8, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, H.; Kobayashi, N.; Tanaka, Y.; Nakaji, S.; Yong, C.; Okitsu, T.; Oshita, M.; Matsumoto, S.; Noguchi, H.; Narushima, M. A newly developed bioartificial pancreas successfully controls blood glucose in totally pancreatectomized diabetic pigs. Tissue Eng. 2006, 12, 1799–1809. [Google Scholar] [CrossRef]
- Prochorov, A.; Tretjak, S.; Goranov, V.; Glinnik, A.; Goltsev, M. Treatment of insulin dependent diabetes mellitus with intravascular transplantation of pancreatic islet cells without immunosuppressive therapy. Adv. Med. Sci. 2008, 53, 240. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Desai, T.A.; Hansford, D.J.; Ferrari, M. Micromachined interfaces: New approaches in cell immunoisolation and biomolecular separation. Biomol. Eng. 2000, 17, 23–36. [Google Scholar] [CrossRef]
- La Flamme, K.E.; Popat, K.C.; Leoni, L.; Markiewicz, E.; La Tempa, T.J.; Roman, B.B.; Grimes, C.A.; Desai, T.A. Biocompatibility of nanoporous alumina membranes for immunoisolation. Biomaterials 2007, 28, 2638–2645. [Google Scholar] [CrossRef]
- Ainslie, K.M.; Tao, S.L.; Popat, K.C.; Daniels, H.; Hardev, V.; Grimes, C.A.; Desai, T.A. In vitro inflammatory response of nanostructured titania, silicon oxide, and polycaprolactone. J. Biomed. Mater. Res. Part A Off. J. Soc. Biomater. Jpn. Soc. Biomater. Aust. Soc. Biomater. Korean Soc. Biomater. 2009, 91, 647–655. [Google Scholar]
- Strautz, R. Studies of hereditary-obese mice (obob) after implantation of pancreatic islets in Millipore filter capsules. Diabetologia 1970, 6, 306–312. [Google Scholar] [CrossRef] [PubMed]
- Klomp, G.; Ronel, S.; Hashiguchi, H.; D’Andrea, M.; Dobelle, W. Hydrogels for encapsulation of pancreatic islet cells. ASAIO J. 1979, 25, 74–76. [Google Scholar] [CrossRef]
- Altman, J.; Houlbert, D.; Chollier, A.; Leduc, A.; McMillan, P.; Galletti, P. Encapsulated human islet transplants in diabetic rats. ASAIO J. 1984, 30, 382–386. [Google Scholar]
- Lacy, P.E.; Hegre, O.D.; Gerasimidi-Vazeou, A.; Gentile, F.T.; Dionne, K.E. Maintenance of normoglycemia in diabetic mice by subcutaneous xenografts of encapsulated islets. Science 1991, 254, 1782–1784. [Google Scholar] [CrossRef] [PubMed]
- Kessler, L.; Pinget, M.; Aprahamian, M.; Dejardin, P.; Damge, C. In vitro and in vivo studies of the properties of an artificial membrane for pancreatic islet encapsulation. Horm. Metab. Res. 1991, 23, 312–317. [Google Scholar] [CrossRef] [PubMed]
- Lembert, N.; Petersen, P.; Wesche, J.; Zschocke, P.; Enderle, A.; Doser, M.; Planck, H.; Becker, H.; Ammon, H. In vitro test of new biomaterials for the development of a bioartificial pancreas. Ann. N. Y. Acad. Sci. 2001, 944, 271–276. [Google Scholar] [CrossRef]













| Parameters | Lung | Oxygenator Membrane | Ref. |
|---|---|---|---|
| Exchange surface area | 70 m2 | 1–3 m2 | [34,35] |
| Surface characteristics | Hydrophilic | Hydrophobic | [34] |
| Membrane thickness | 1–2 μm | 50–100 μm | [34] |
| Gas permeability | High | Low | [34] |
| Blood contact time | <1 s | 5–15 s | [34] |
| Type of gas | Air | Enriched air | [34] |
| O2 quantity added into blood | 2 L/min | >470 mL/min | [35] |
| CO2 quantity removed from blood | 1.6 L/min | >370 mL/min | [35] |
| Type of Membrane | Blood Flow Pattern | Correlation for Blood Side Mass Transfer Coefficient | Ref. |
|---|---|---|---|
| PP/Hollow fibers | Across hollow fiber mat | [38] | |
| PP/Flat sheet | In thin channel | [38] | |
| PP /Hollow fibers | Across a 1 to 6 perpendicular hollow fibers | is the void fraction) | [39] |
| PP /Hollow fibers | Across a parallel hollow fibers | [36] | |
| PP /Hollow fibers | Across a perpendicular hollow fibers | [36] | |
| PMP /Hollow fibers | Across a parallel hollow fibers | [40] |
| Flow Direction | Gas Type | Fiber Bundles Arrangement | |
|---|---|---|---|
| Square | Staggered | ||
| Parallel flow | O2 | ||
| CO2 | |||
| Transverse flow | O2 | ||
| CO2 | |||
| Polymer | P(O2) | P(CO2) | Ref. |
|---|---|---|---|
| Polypropylene (PP) | 2.2 | 9.2 | [51,52] |
| Poly 4-methyl pentene-1 (PMP) | 32.3 | 92.6 | [52] |
| Polydimethylsiloxane (PDMS) | 605 | 3240 | [52] |
| Teflon AF2400 | 1600 | 3900 | [53] |
| Teflon AF1600 | 270 | 520 | [53] |
| Hyflon AD80 | 67 | 150 | [53] |
| Hyflon AD60 | 57 | 130 | [53] |
| Natural rubber | 23.3 | 153 | [52] |
| Polyethylene (dens. 0.922) | 6.9 | 28 | [52] |
| Polytetrafluoroethylene (PTFE) | 4.9 | 12.7 | [52] |
| Neoprene | 4 | 25.8 | [52] |
| Polystyrene | 2.63 | 10.5 | [51,52] |
| Polycarbonate | 1.4 | 8 | [52] |
| Butyl rubber | 1.3 | 5.18 | [52] |
| Cellulose acetate | 0.8 | 2.4 | [52] |
| Polyvinyl chloride (unplasticized) | 0.045 | 0.16 | [52] |
| Nylon 6 | 0.038 | 0.16 | [51,52] |
| Polyethylene terephthalate | 0.035 | 0.17 | [52] |
| Polyvinylidene chloride | 0.0053 | 0.029 | [52] |
| Polymethacrylonitrile | 0.0012 | 0.0032 | [52] |
| Polyacrylonitrile | 0.0003 | 0.0018 | [52] |
| Device | Capiox FX | Affinity NT | Quadrox | Vision | Vital | Eurosets | Hilite | Hilite LT |
|---|---|---|---|---|---|---|---|---|
| Company | Terumo | Medtronic | Maquet | Gish Biomedical | NIPRO | Eurosets | Medos | Medos |
| Membrane materials | PP | PP | PP | PP | PP | PMP | PP | PMP |
| Membrane area, m2 | 0.5–2.5 | 2.5 | 1.8 | 2.45 | 2 | 0.69–1.81 | 0.39–1.9 | 0.32–1.9 |
| Priming volume, ml | 43–260 | 270 | 250 | 280 | 180 | 90–225 | 57–275 | 55–275 |
| Blood flow, L/min | 0.1–7 | 1–7 | 0.5–7 | 1–8 | 0.5–7 | 0.2–7 | 1–7 | 0.8–7 |
| O2 transfer rate, mL/min | 50–500 | 50–400 | Max 425 | 400 | _ | 90–350 | _ | 100–550 |
| CO2 transfer rate, mL/min | 50–500 | 50–400 | Max 320 | 200–500 | _ | 70–300 | _ | 75–350 |
| Coating materials | X coating (nonheparin) | Balance biosurface (heparin-free) | SOFTLINE coating | GBS coating (nonleaching heparin coating) | _ | Phosphorylcholine | Uncoated/ x.eed/ rheoparin | Uncoated/ rheoparin |
 |  |  |  |  |  |  | ||
| Type of devices | Material | Islet source | Model for | Ref. |
|---|---|---|---|---|
| Intravascular | PAN-PVC | Rat, Monkey | Rat, Monkey | [203] |
| Polycarbonate | Rat | Dog | [204] | |
| EVAL fibers | Porcine | Pig | [205] | |
| Poly-animo-urethane-coated | ||||
| Nonwoven PTFE fabric | ||||
| Nylon | Rabbit fetuses | Human | [206] | |
| Extravascular | Nitrocellulose acetate | Mice | Mice | [210] |
| 2-Hydroxyethyl methacrylate | Rat, Rabbit | Rat | [211] | |
| Cellulose acetate | Human | Rat | [212] | |
| Acrylic copolymer | Rat | Mice | [213] | |
| Acrylonitrile (AN62) | Rat | Rat | [214] | |
| Polysulfone | Rat | In vitro | [215] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duy Nguyen, B.T.; Nguyen Thi, H.Y.; Nguyen Thi, B.P.; Kang, D.-K.; Kim, J.F. The Roles of Membrane Technology in Artificial Organs: Current Challenges and Perspectives. Membranes 2021, 11, 239. https://doi.org/10.3390/membranes11040239
Duy Nguyen BT, Nguyen Thi HY, Nguyen Thi BP, Kang D-K, Kim JF. The Roles of Membrane Technology in Artificial Organs: Current Challenges and Perspectives. Membranes. 2021; 11(4):239. https://doi.org/10.3390/membranes11040239
Chicago/Turabian StyleDuy Nguyen, Bao Tran, Hai Yen Nguyen Thi, Bich Phuong Nguyen Thi, Dong-Ku Kang, and Jeong F. Kim. 2021. "The Roles of Membrane Technology in Artificial Organs: Current Challenges and Perspectives" Membranes 11, no. 4: 239. https://doi.org/10.3390/membranes11040239
APA StyleDuy Nguyen, B. T., Nguyen Thi, H. Y., Nguyen Thi, B. P., Kang, D.-K., & Kim, J. F. (2021). The Roles of Membrane Technology in Artificial Organs: Current Challenges and Perspectives. Membranes, 11(4), 239. https://doi.org/10.3390/membranes11040239






