Human Mesenchymal Stem Cells Seeded on the Natural Membrane to Neurospheres for Cholinergic-like Neurons
Abstract
:1. Introduction
2. Materials and Methods
2.1. Umbilical Cord Stem Cells-Ethical Donors
2.2. Isolation of Stem Cells from Wharton Jelly
2.3. Isolation of Stem Cells from Human Umbilical Cord Blood
2.4. Mesenchymal Stem Cells Characterization
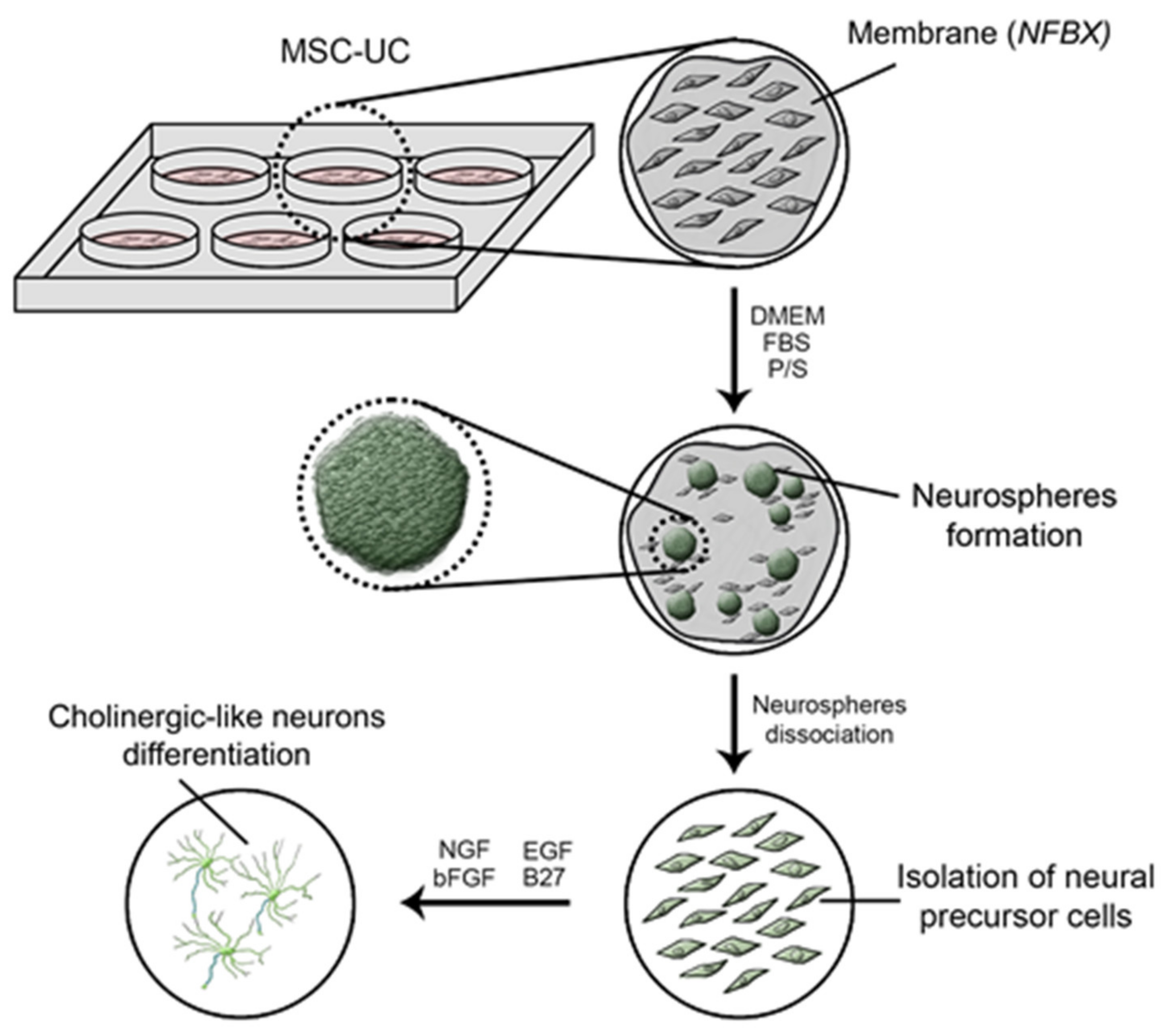
2.5. Preparation of the Polyisoprene-Based Membrane
2.6. Production of Neural Precursor Cells
2.7. Cholinergic-like Neurons Differentiation Protocol
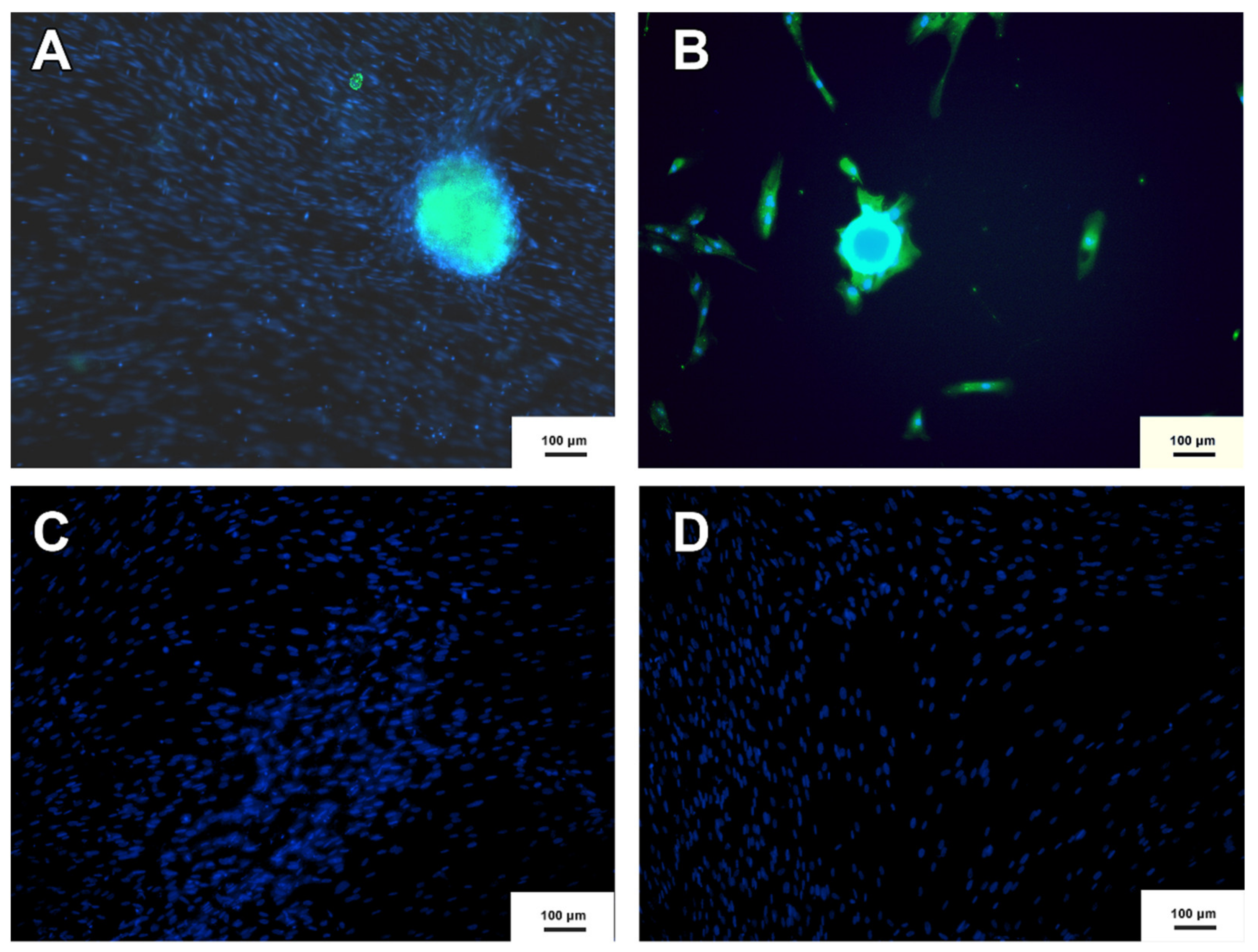
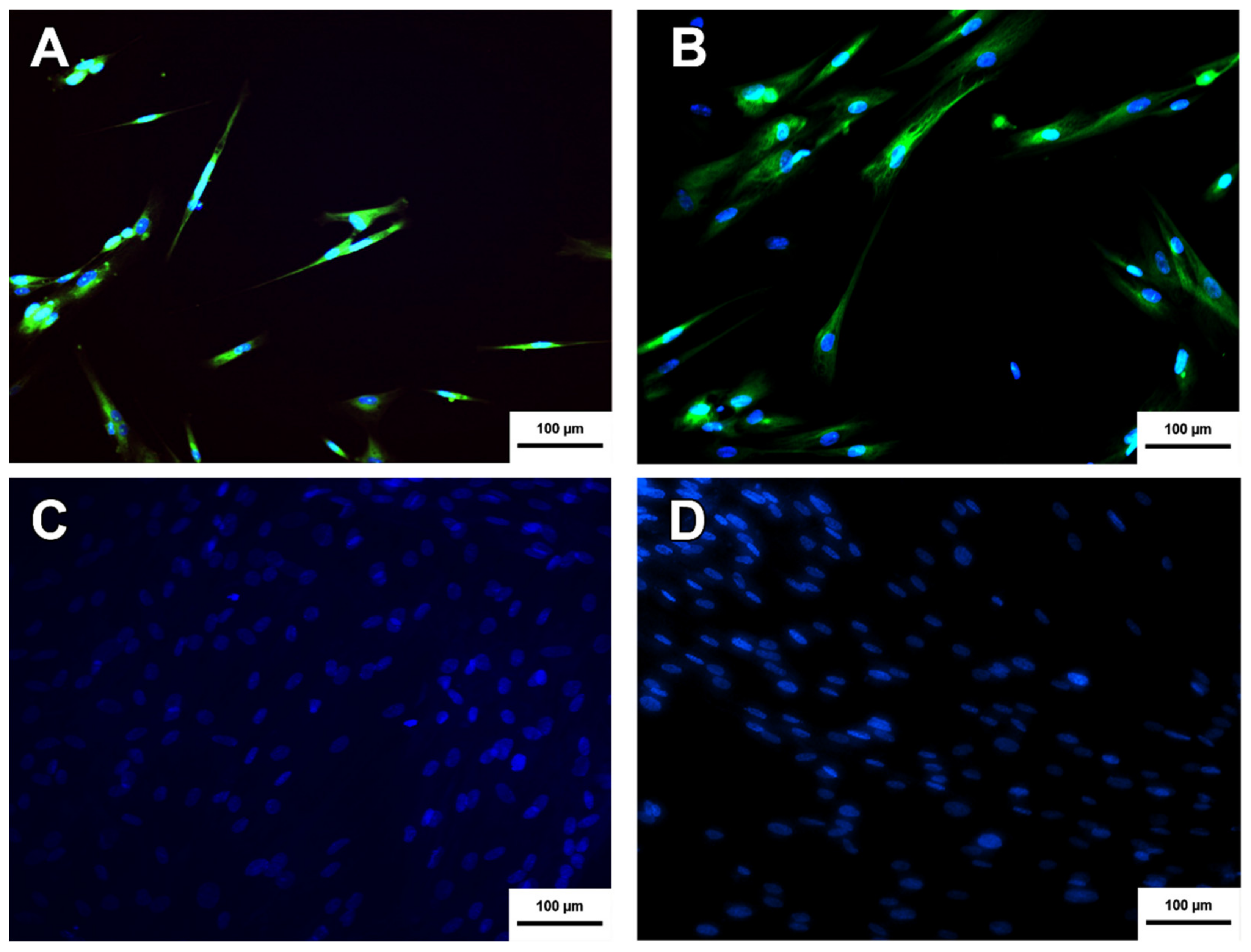
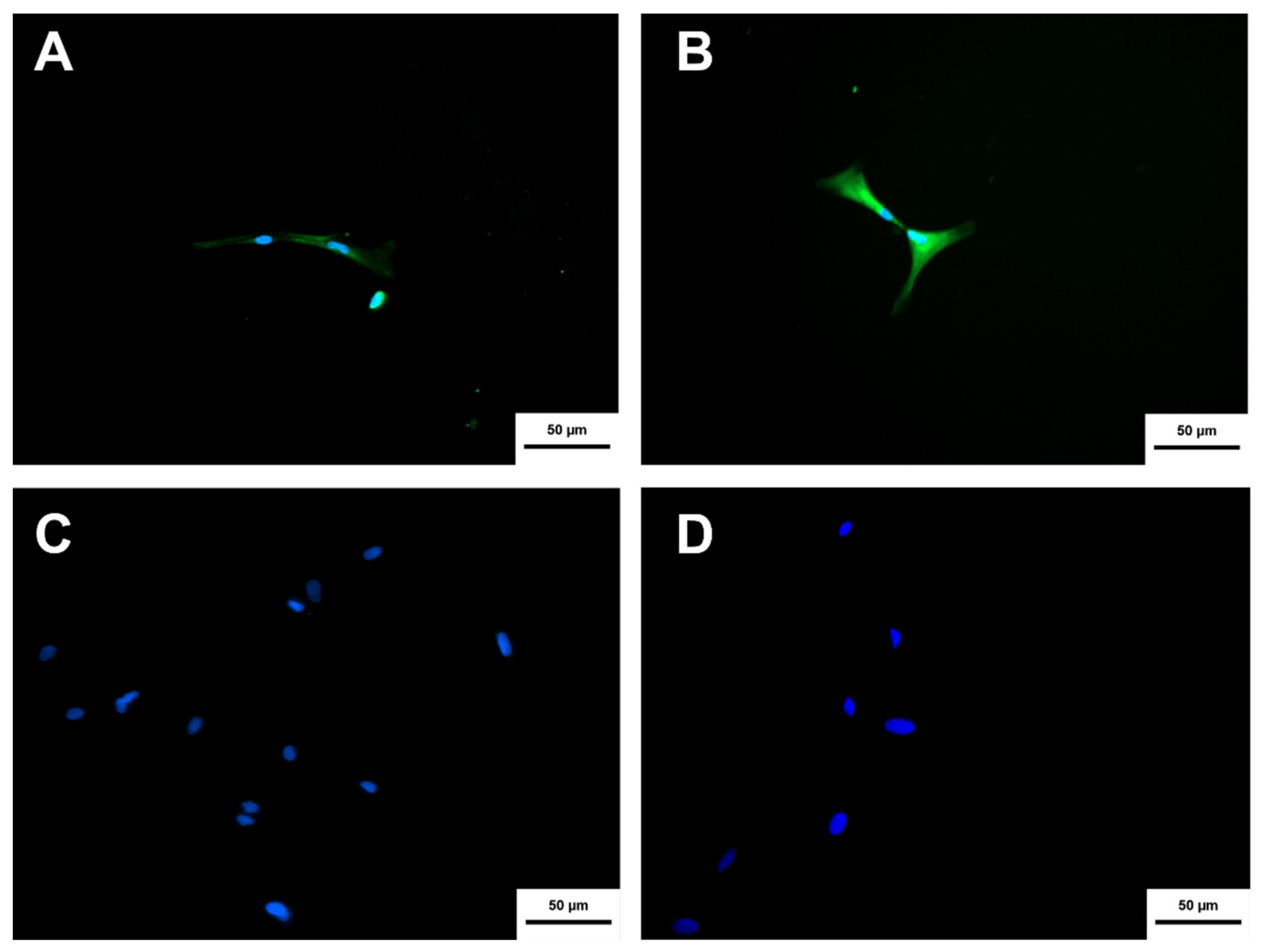
2.8. Immunocytochesmistry
2.9. Scanning Electron Microscopy
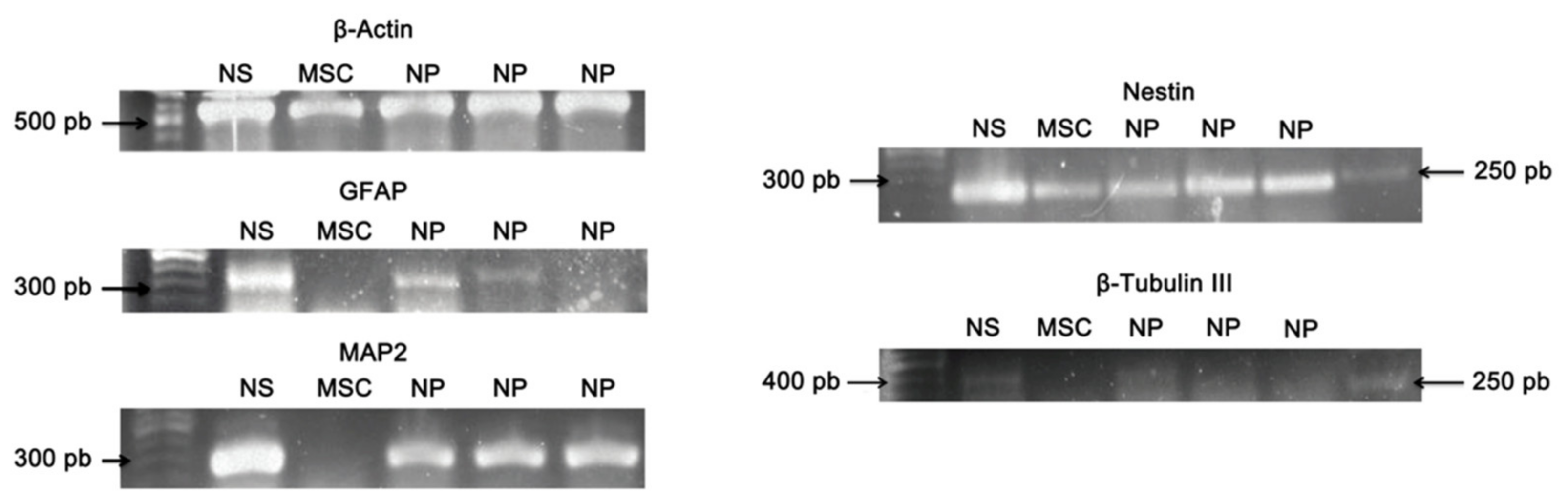
2.10. Qualitative Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
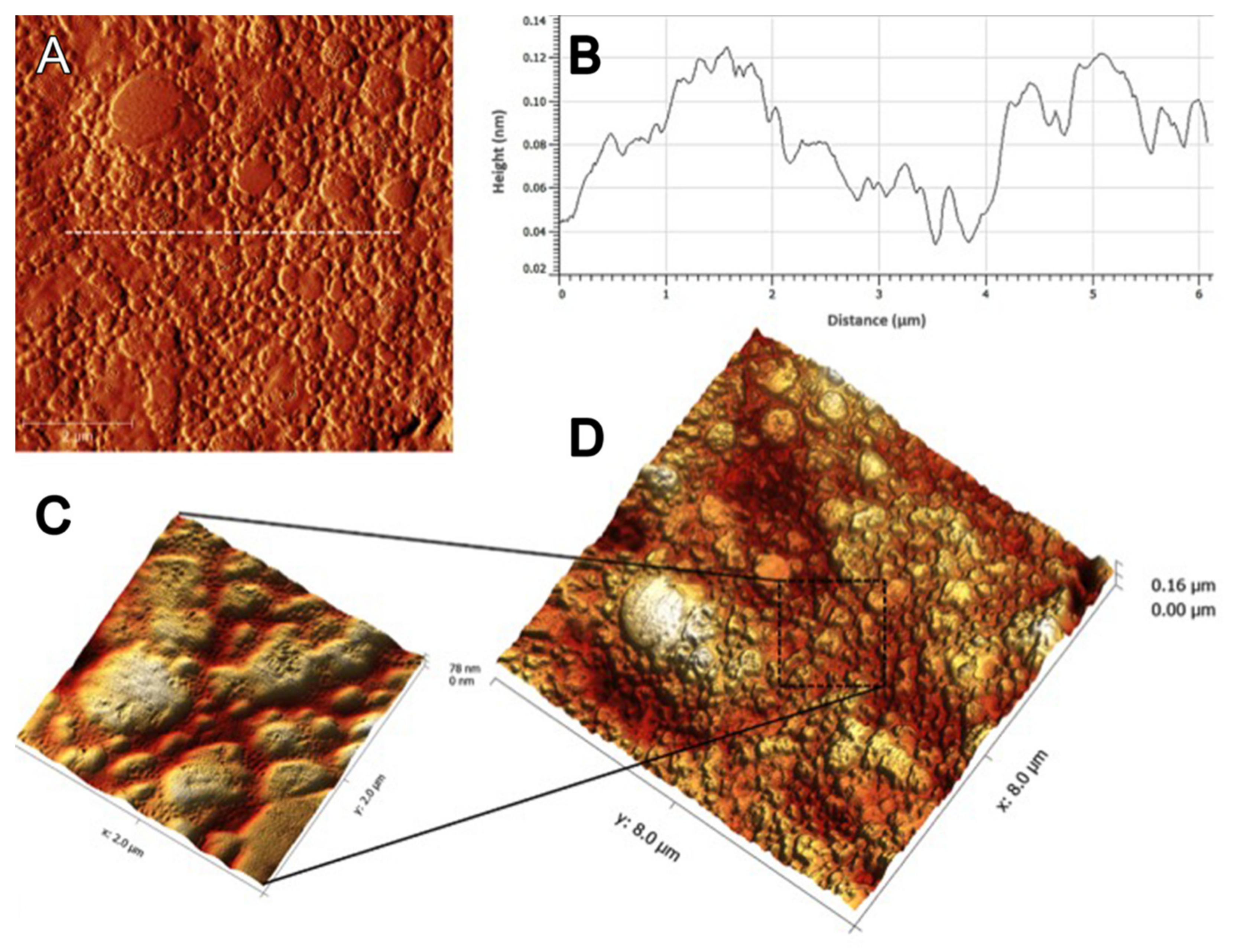
2.11. Atomic Force Microscopy Analysis
3. Results
3.1. Flow Cytometry Analysis
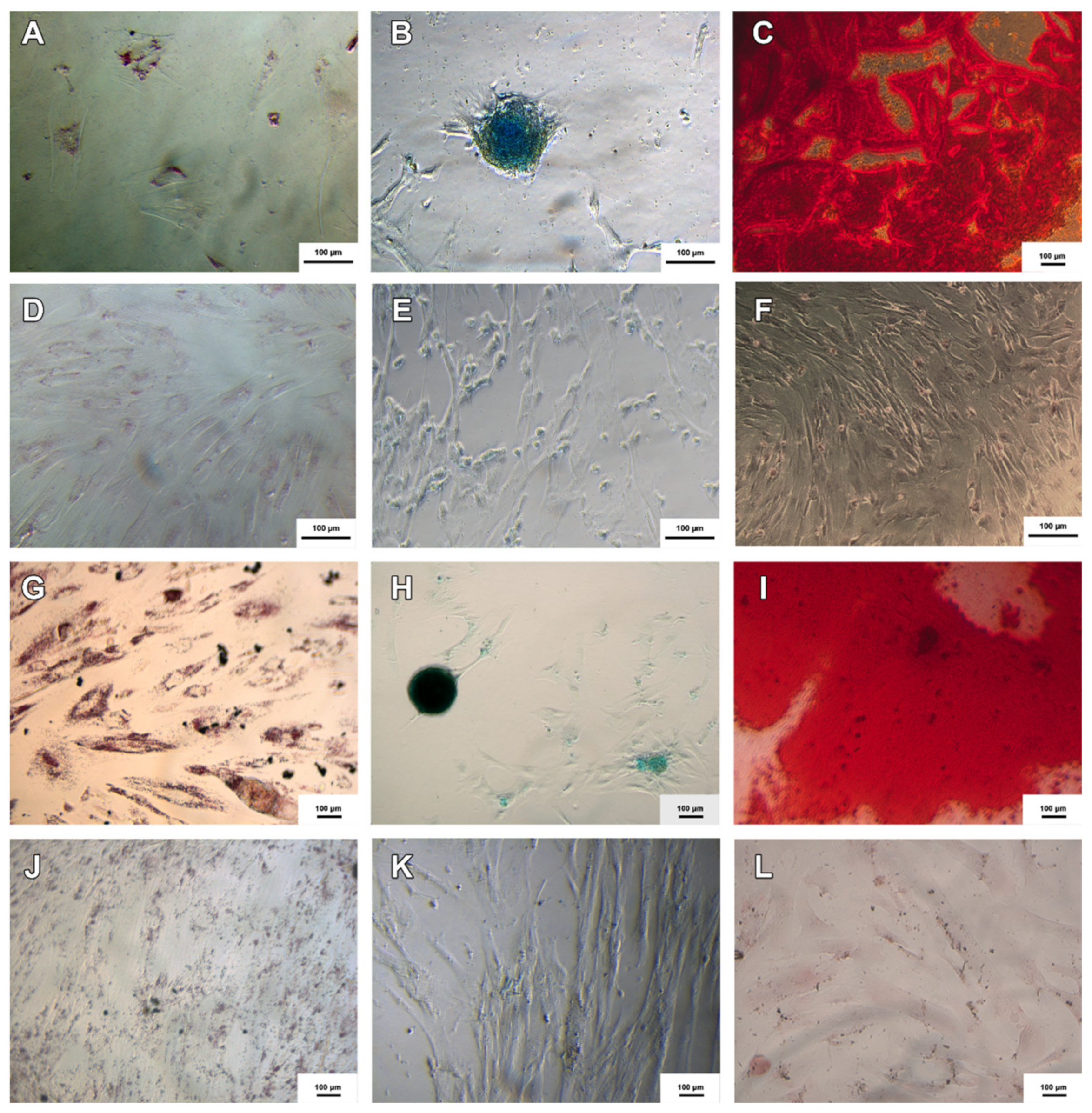
3.2. Trilineage Differentiation Test
3.3. Production of Neural Precursor Cells
3.4. Neurospheres and Neural Precursor Cells
3.5. Scanning Electron Microscopy
3.6. Qualitative Reverse Transcription–Polymerase Chain Reaction (RT-PCR)
3.7. Atomic Force Microscopy Analysis
3.8. Cholinergic Differentiation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mukai, T.; Inoue, T.N.; Shimazu, T.; Mori, Y.; Takahashi, A.; Tsunoda, H.; Yamaguchi, S.; Tojo, A. Neurosphere formation enhances the neurogenic differentiation potential and migratory ability of umbilical cord-mesenchymal stromal cells. Cytotherapy 2016, 18, 229–241. [Google Scholar] [CrossRef]
- Cai, Z.; Xiao, M. Oligodendrocytes and Alzheimer’s disease. Int. J. Neurosci. 2016, 126, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Lee, V.M.; Scientist, S.; Louis, S.A.; Reynolds, B.A. Neural stem cells-mini review. Stem Cell Technol. 2015, 5, 1–6. [Google Scholar]
- Lu, Y.; Yang, Y.; Wang, Z.; Wang, C.; Du, Q.; Wang, Q.; Luan, Z. Isolation and culture of human oligodendrocyte precursor cells from neurospheres. Brain Res. Bull. 2015, 118, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Tsai, P.H.; Chang, Y.C.; Lee, Y.Y.; Ko, Y.L.; Yang, Y.H.; Lin, C.F.; Chang, Y.L.; Yu, W.C.; Shih, Y.H.; Chen, M.T. Differentiation of blood T cells: Reprogramming human induced pluripotent stem cells into neuronal cells. J. Chinese Med. Assoc. 2015, 78, 353–359. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, E.; Liu, N.; Tang, Y.; Hu, Y.; Zhang, P.; Pan, C.; Dong, S.; Zhang, Y.; Tang, Z. Generation of neurospheres from human adipose-derived stem cells. Biomed Res. Int. 2015, 2015, 743714. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, Y.; Mucke, L. Alzheimer mechanisms and therapeutic strategies. Cell 2012, 148, 1204–1222. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.T.; Chen, H.; Zhao, H.; Dai, Y.W.; Xu, R.X. Neural stem cells differentiation ability of human umbilical cord mesenchymal stromal cells is not altered by cryopreservation. Neurosci. Lett. 2011, 487, 118–122. [Google Scholar] [CrossRef]
- Monteiro, B.S.; Neto, N.M.A.; Del Carlo, R.J. Células-tronco mesenquimais. Ciência Rural 2010, 40, 238–245. [Google Scholar] [CrossRef]
- De Carvalho, K.A.T.; Steinhoff, G.; Chachques, J.C. Mesenchymal stem cell therapy in nonhematopoietic diseases. Stem Cells Int. 2015, 2015, 4–6. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Yu, Y.; Chen, L.; Ye, L.; Cui, J.; Sun, Q.; Li, K.; Li, Z.; Liu, L. Human umbilical cord mesenchymal stem cells: A new therapeutic option for tooth regeneration. Stem Cells Int. 2015, 2015, 549432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cho, H.; Seo, Y.K.; Jeon, S.; Yoon, H.H.; Choi, Y.K.; Park, J.K. Neural differentiation of umbilical cord mesenchymal stem cells by sub-sonic vibration. Life Sci. 2012, 90, 591–599. [Google Scholar] [CrossRef]
- Sibov, T.T.; Severino, P.; Marti, L.C.; Pavon, L.F.; Oliveira, D.M.; Tobo, P.R.; Campos, A.H.; Paes, A.T.; Amaro, E.; Gamarra, L.F.; et al. Mesenchymal stem cells from umbilical cord blood: Parameters for isolation, characterization and adipogenic differentiation. Cytotechnology 2012, 64, 511–521. [Google Scholar] [CrossRef] [Green Version]
- Tondreau, T.; Meuleman, N.; Delforge, A.; Dejeneffe, M.; Leroy, R.; Massy, M.; Mortier, C.; Bron, D.; Lagneaux, L. Mesenchymal stem cells derived from CD133-positive cells in mobilized peripheral blood and cord blood: Proliferation, Oct4 expression, and plasticity. Stem Cells 2005, 23, 1105–1112. [Google Scholar] [CrossRef]
- Dominici, M.; Blanc, K.L.; Mueller, I.; Cortenbach, I.S.; Marini, F.C.; Krause, D.S.; Deans, R.J.; Keating, A.; Prockop, D.J.; Horwitz, E.M. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef]
- Devetten, M.; Armitage, J.O. Hematopoietic cell transplantation: Progress and obstacles. Ann. Oncol. 2007, 18, 1450–1456. [Google Scholar] [CrossRef]
- Naghdi, M.; Tiraihi, T.; Mesbah-Namin, S.A.; Arabkheradmand, J. Induction of bone marrow stromal cells into cholinergic-like cells by nerve growth factor. Iran. Biomed. J. 2009, 13, 117–123. [Google Scholar] [PubMed]
- Suzuki, S.; Namiki, J.; Shibata, S.; Mastuzaki, Y.; Okano, H. The neural stem/progenitor cell marker nestin is expressed in proliferative endothelial cells, but not in mature vasculature. J. Histochem. Cytochem. 2010, 58, 721–730. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adib, S.; Tiraihi, T.; Darvishi, M.; Taheri, T.; Kazemi, H. Cholinergic differentiation of neural stem cells generated from cell aggregates-derived from Human Bone marrow stromal cells. Tissue Eng. Regen. Med. 2014, 12, 43–52. [Google Scholar] [CrossRef]
- Karimi, T.; Eslaminejad, M.B.; Aminlari, M.; Shahverdi, A.; Bahmanpour, S. Study of telomerase activity, proliferation and differentiation characteristics in umbilical cord blood mesenchymal stem cells. Iran. J. Vet. Res. 2012, 13, 176–185. [Google Scholar]
- Sarugaser, R.; Lickorish, D.; Baksh, D.; Hosseini, M.M.; Davies, J.E. Human umbilical cord perivascular (HUCPV) cells: A source of mesenchymal progenitors. Stem Cells 2005, 23, 220–229. [Google Scholar] [CrossRef]
- Harichandan, A.; Bühring, H.J. Prospective isolation of human MSC. Best. Pract. Res. Clin. Haematol. 2011, 24, 25–36. [Google Scholar] [CrossRef]
- Jin, H.; Bae, Y.; Kim, M.; Kwon, S.-J.; Jeon, H.; Choi, S.; Kim, S.; Yang, Y.; Oh, W.; Chang, J. Comparative analysis of human mesenchymal Stem cells from bone marrow, adipose tissue, and umbilical cord blood as sources of cell therapy. Int. J. Mol. Sci. 2013, 14, 17986–18001. [Google Scholar] [CrossRef]
- Maslova, O.; Novak, M.; Kruzliak, P. Umbilical cord tissue-derived cells as therapeutic agents. Stem Cells Int. 2015, 2015, 150609. [Google Scholar] [CrossRef] [Green Version]
- Sipahi, R.; Zupanc, G.K.H. Stochastic cellular automata model of neurosphere growth: Roles of proliferative potential, contact inhibition, cell death, and phagocytosis. J. Theor. Biol. 2018, 445, 151–165. [Google Scholar] [CrossRef] [PubMed]
- Erlach, T.C.V.; Bertazzo, S.; Wozniak, M.A.; Horejs, C.M.; Maynard, S.A.; Attwood, S.; Robinson, B.K.; Autefage, H.; Kallepitis, C.; Hernández, A.D.R.; et al. Cell-geometry-dependent changes in plasma membrane order direct stem cell signalling and fate. Nat. Mater. 2018, 17, 237–242. [Google Scholar] [CrossRef] [PubMed]
- Petersen, E.D.; Zenchak, J.R.; Lossia, O.V.; Hochgeschwender, U. Neural stem cells derived directly from adipose tissue. Stem Cells Dev. 2018, 27, 9. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.T.; Fan, J.; Cai, Y.Q.; Zhao, S.J.; Xue, S.; Lin, J.H.; Jiang, X.D.; Xu, R.X. Human Wharton’s jelly cells can be induced to differentiate into growth factor-secreting oligodendrocyte progenitor-like cells. Differentiation 2010, 79, 15–20. [Google Scholar] [CrossRef]
- Park, D.; Xiang, A.P.; Mao, F.F.; Zhang, L.; Di, C.G.; Liu, X.M.; Shao, Y.; Ma, B.F.; Lee, J.H.; Ha, K.S.; et al. Nestin is required for the proper self-renewal of neural stem cells. Stem Cells 2010, 28, 2162–2171. [Google Scholar] [CrossRef]
- Kern, S.; Eichler, H.; Stoeve, J.; Klüter, H.; Bieback, K. Comparative analysis of mesenchymal stem cells from bone marrow, umbilical cord blood, or adipose tissue. Stem Cells 2006, 24, 1294–1301. [Google Scholar] [CrossRef] [PubMed]
- Birenboim, R.; Markus, A.; Goldstein, R.S. Simple generation of neurons from human embryonic stem cells using agarose multiwell dishes. J. Neurosci. Methods 2013, 214, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, S.; Thej, C.; Venugopal, P.; Priya, N. Higher propensity of Wharton’ s jelly derived mesenchymal stromal cells towards neuronal lineage in comparison to those derived from adipose and bone marrow. Cell Biol. Int. 2013, 37, 507–515. [Google Scholar] [CrossRef] [PubMed]
- Chung, D.J.; Wong, A.; Hayashi, K.; Yellowley, C.E. Effect of hypoxia on generation of neurospheres from adipose tissue-derived canine mesenchymal stromal cells. Vet. J. 2014, 199, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Materne, E.M.; Ramme, A.P.; Terrasso, A.P.; Serra, M.; Alves, P.M.; Brito, C.; Sakharov, D.A.; Tonevitsky, A.G.; Lauster, R.; Marx, U. A multi-organ chip co-culture of neurospheres and liver equivalents for long-term substance testing. J. Biotechnol. 2015, 205, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, D.P.; Al-Rubai, A.-j.; Grabowska, A.M.; Pratten, M.K. Separating chemotherapy-related developmental neurotoxicity from cytotoxicity in monolayer and neurosphere cultures of human fetal brain cells. Toxicol. Vitr. 2016, 37, 88–96. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Iwashita, T.; Buchstaller, J.; Molofsky, A.V.; Thomas, D.; Morrison, S.J. Bmi-1 over-expression in neural stem/progenitor cells increases proliferation and neurogenesis in culture but has little effect on these functions in vivo. Dev. Biol. 2009, 328, 257–272. [Google Scholar] [CrossRef] [Green Version]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stricker, P.E.F.; de Souza Dobuchak, D.; Irioda, A.C.; Mogharbel, B.F.; Franco, C.R.C.; de Souza Almeida Leite, J.R.; de Araújo, A.R.; Borges, F.A.; Herculano, R.D.; de Oliveira Graeff, C.F.; et al. Human Mesenchymal Stem Cells Seeded on the Natural Membrane to Neurospheres for Cholinergic-like Neurons. Membranes 2021, 11, 598. https://doi.org/10.3390/membranes11080598
Stricker PEF, de Souza Dobuchak D, Irioda AC, Mogharbel BF, Franco CRC, de Souza Almeida Leite JR, de Araújo AR, Borges FA, Herculano RD, de Oliveira Graeff CF, et al. Human Mesenchymal Stem Cells Seeded on the Natural Membrane to Neurospheres for Cholinergic-like Neurons. Membranes. 2021; 11(8):598. https://doi.org/10.3390/membranes11080598
Chicago/Turabian StyleStricker, Priscila Elias Ferreira, Daiany de Souza Dobuchak, Ana Carolina Irioda, Bassam Felipe Mogharbel, Celia Regina Cavichiolo Franco, José Roberto de Souza Almeida Leite, Alyne Rodrigues de Araújo, Felipe Azevedo Borges, Rondinelli Donizetti Herculano, Carlos Frederico de Oliveira Graeff, and et al. 2021. "Human Mesenchymal Stem Cells Seeded on the Natural Membrane to Neurospheres for Cholinergic-like Neurons" Membranes 11, no. 8: 598. https://doi.org/10.3390/membranes11080598
APA StyleStricker, P. E. F., de Souza Dobuchak, D., Irioda, A. C., Mogharbel, B. F., Franco, C. R. C., de Souza Almeida Leite, J. R., de Araújo, A. R., Borges, F. A., Herculano, R. D., de Oliveira Graeff, C. F., Chachques, J. C., & de Carvalho, K. A. T. (2021). Human Mesenchymal Stem Cells Seeded on the Natural Membrane to Neurospheres for Cholinergic-like Neurons. Membranes, 11(8), 598. https://doi.org/10.3390/membranes11080598






