Effect of Composition and Viscosity of Spinning Solution on Ultrafiltration Properties of Polyphenylene Sulfone Hollow-Fiber Membranes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of PPSUs
2.3. Study of Synthesized PPSUs
2.3.1. Nuclear Magnetic Resonance (NMR) Method
2.3.2. Gel Permeation Chromatography (GPC) Method
2.4. Preparation of Casting Solutions
2.5. Study of Viscosity of Casting Solutions
2.6. Sample Casting of Hollow Fiber Membranes
2.7. Ultrafiltration Properties
3. Results and Discussion
3.1. NMR of Synthesized PPSUs
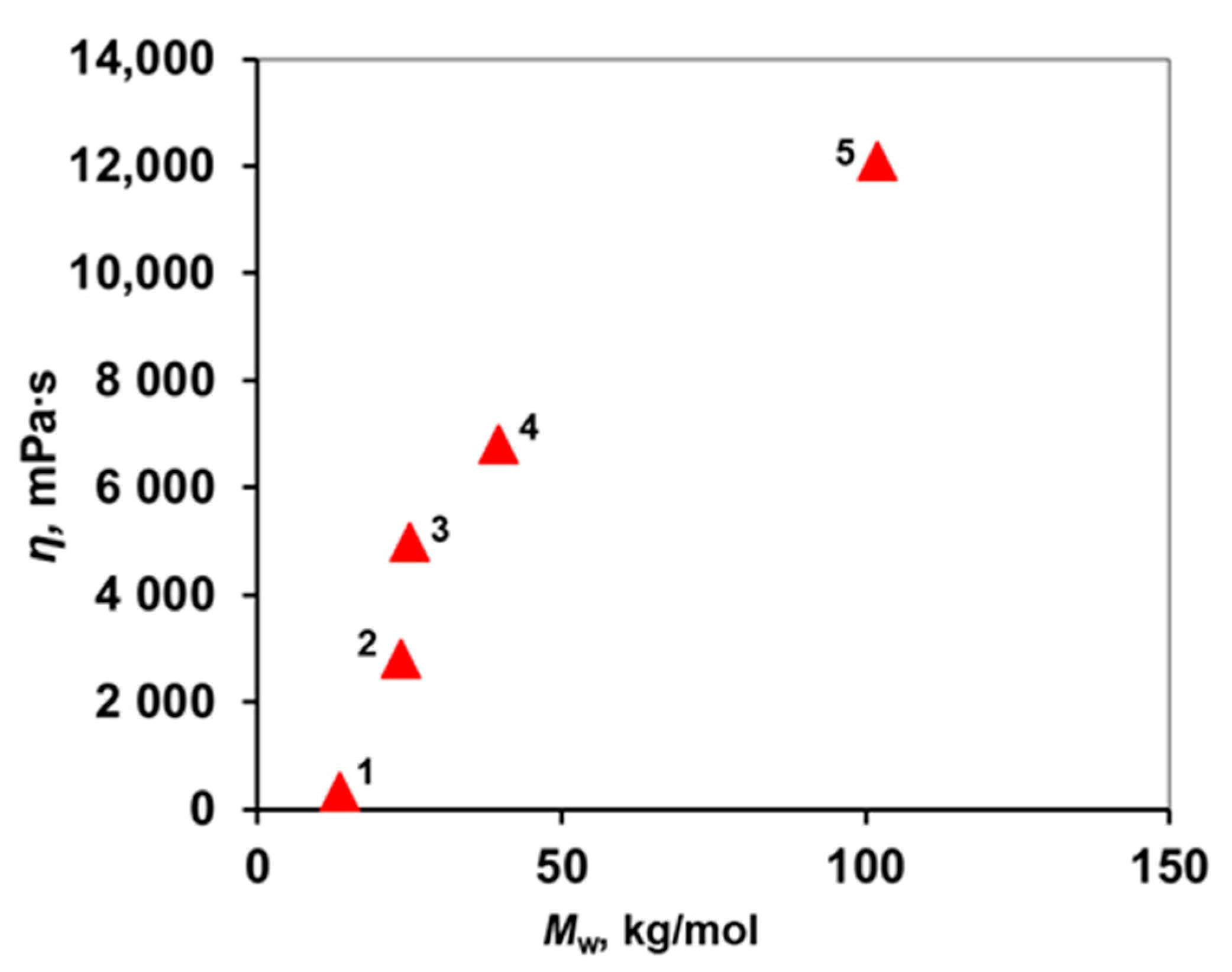
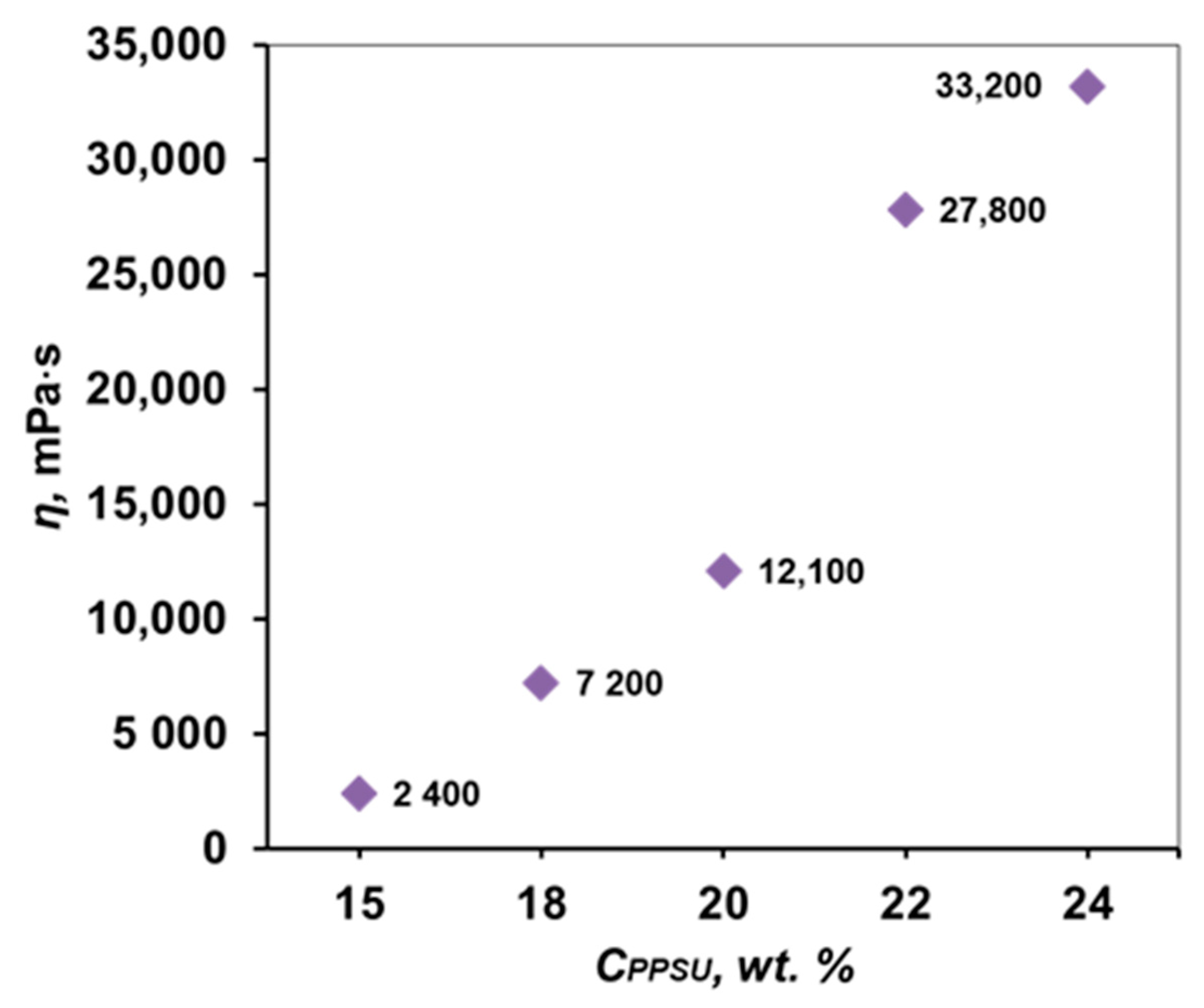
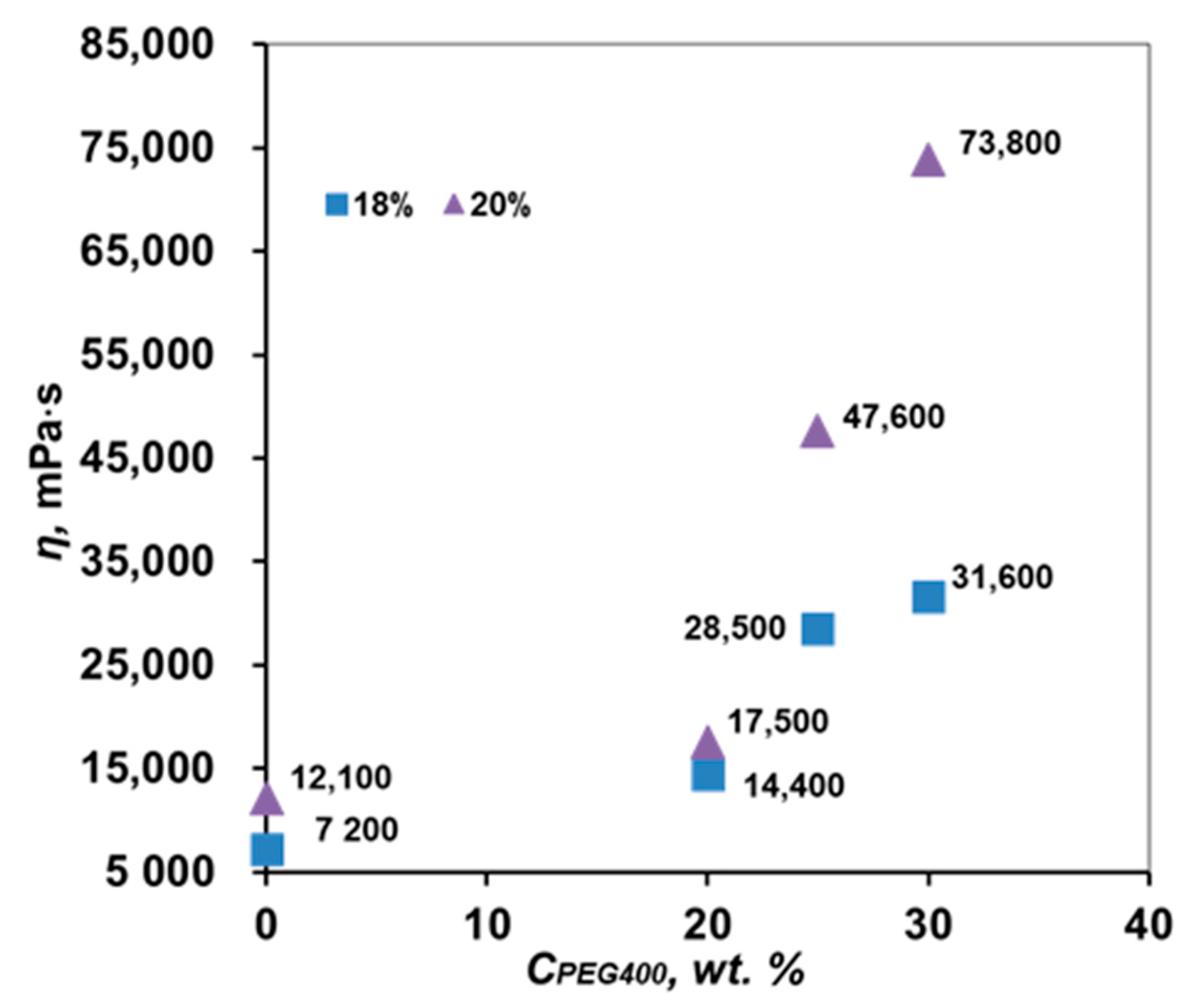
3.2. Study of the Rheological Properties of PPSU Solutions
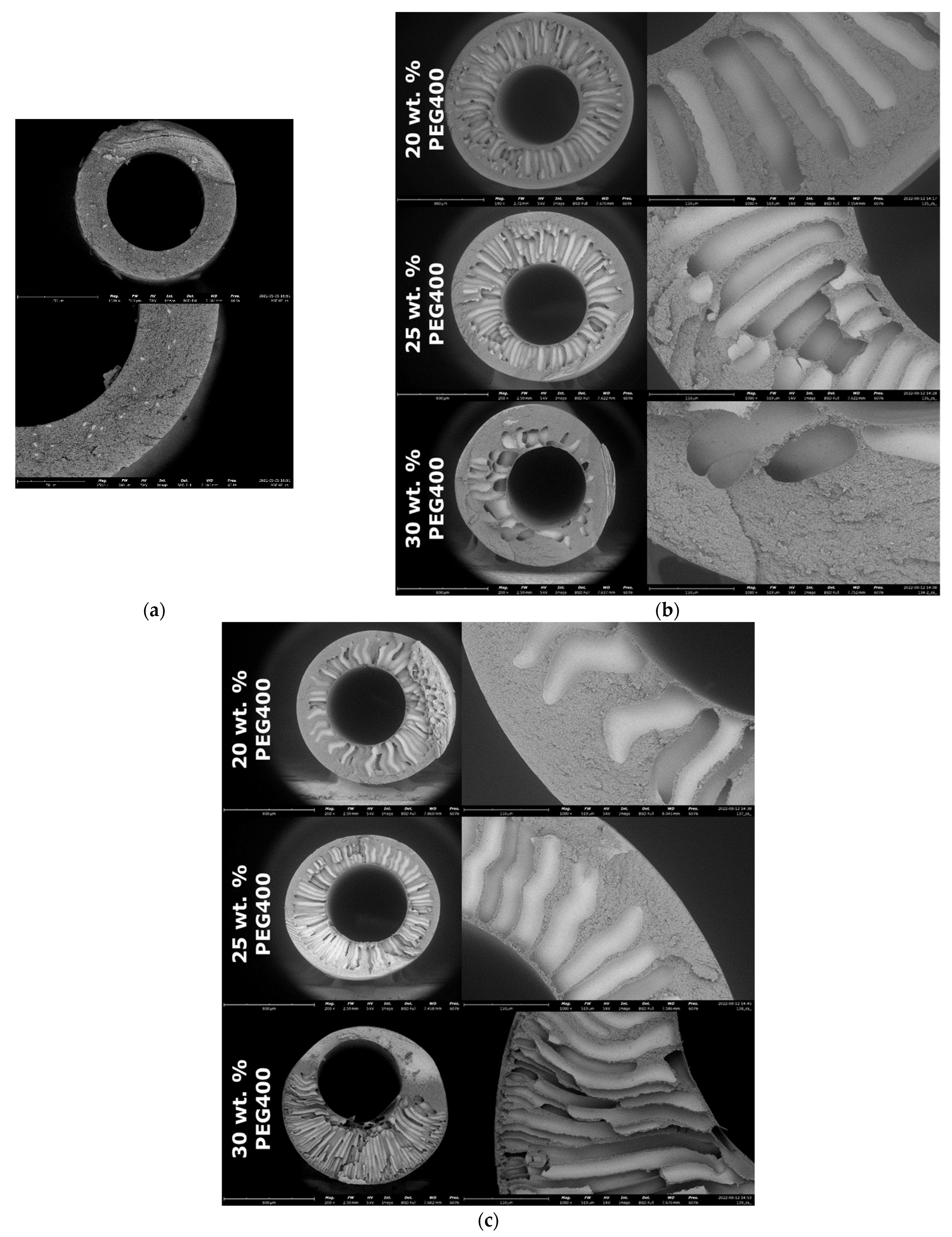
3.3. Porous Structure, Transport and Separation Characteristics of Samples of PPSU-Based Hollow Fiber Membranes Spinning on the Needle-Carrier of the Device
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fahimirad, S.; Fahimirad, Z.; Sillanpää, M. Efficient removal of water bacteria and viruses using electrospun nanofibers. Sci. Total Environ. 2021, 751, 141673. [Google Scholar] [CrossRef]
- Goswami, K.P.; Pugazhenthi, G. Credibility of polymeric and ceramic membrane filtration in the removal of bacteria and virus from water: A review. J. Environ. Manag. 2020, 268, 110583. [Google Scholar] [CrossRef] [PubMed]
- WHO. The World Health Report 2002; World Health Organization: Geneva, Switzerland, 2002. [Google Scholar]
- Fiksdal, L.; Leiknes, T. The effect of coagulation with MF/UF membrane filtration for the removal of virus in drinking water. J. Membr. Sci. 2006, 279, 364–371. [Google Scholar] [CrossRef]
- Landsman, M.R.; Sujanani, R.; Brodfuehrer, S.H.; Cooper, C.M.; Darr, A.G.; Davis, R.J.; Kim, K.; Kum, S.; Nalley, L.K.; Nomaan, S.M.; et al. Water treatment: Are membranes the panacea? Annu. Rev. Chem. Biomol. Eng. 2020, 11, 559–585. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.; Guo, L.; Yang, Y.; Oguma, K.; Hou, L.-A. Comparative effectiveness of membrane technologies and disinfection methods for virus elimination in water: A review. Sci. Total Environ. 2021, 801, 149678. [Google Scholar] [CrossRef] [PubMed]
- Lesimple, A.; Jasim, S.Y.; Johnson, D.J.; Hilal, N. The role of wastewater treatment plants as tools for SARS-CoV-2 early detection and removal. J. Water Process Eng. 2020, 38, 101544. [Google Scholar] [CrossRef]
- Eray, E.; Candelario, V.M.; Boffa, V.; Safafar, H.; Østedgaard-Munck, D.N.; Zahrtmann, N.; Kadrispahic, H.; Jørgensen, M.K. A roadmap for the development and applications of silicon carbide membranes for liquid filtration: Recent advancements, challenges, and perspectives. Chem. Eng. J. 2021, 414, 128826. [Google Scholar] [CrossRef]
- Liu, F.; Hashim, N.A.; Liu, Y.; Moghareh Abed, M.R.; Li, K. Progress in the production and modification of PVDF membranes. J. Membr. Sci. 2011, 375, 1–27. [Google Scholar] [CrossRef]
- Zodrow, K.; Brunet, L.; Mahendra, S.; Li, D.; Zhang, A.; Li, Q.; Alvarez, P.J. Polysulfone ultrafiltration membranes impregnated with silver nanoparticles show improved biofouling resistance and virus removal. Water Res. 2009, 43, 715–723. [Google Scholar] [CrossRef] [Green Version]
- Praneeth, K.; Tardio, J.; Sridhar, S. Design of novel ultrafiltration systems based on robust polyphenylsulfone hollow fiber membranes for treatment of contaminated surface water. Chem. Eng. J. 2014, 248, 297–306. [Google Scholar]
- Darvishmanesh, S.; Jansen, J.; Tasselli, F.; Tocci, E.; Luis, P.; Degrève, J.; Drioli, E.; Van der Bruggen, B. Novel polyphenylsulfone membrane for potential use in solvent nanofiltration. J. Membr. Sci. 2011, 379, 60–68. [Google Scholar] [CrossRef]
- Darvishmanesh, S.; Tasselli, F.; Jansen, J.; Tocci, E.; Bazzarelli, F.; Bernardo, P.; Luis, P.; Degrève, J.; Drioli, E.; Van der Bruggen, B. Preparation of solvent stable polyphenylsulfone hollow fiber nanofiltration membranes. J. Membr. Sci. 2011, 384, 89–96. [Google Scholar] [CrossRef]
- Liu, Z.; Xiang, J.; Hu, X.; Cheng, P.; Zhang, L.; Du, W.; Wang, S.; Tang, N. Effects of coagulation-bath conditions on polyphenylsulfone ultrafiltration membranes. Chin. J. Chem. Eng. 2021, 34, 332–340. [Google Scholar] [CrossRef]
- Plisko, T.V.; Bildyukevich, A.V.; Karslyan, Y.A.; Ovcharova, A.A.; Volkov, V.V. Development of high flux ultrafiltration polyphenylsulfone membranes applying the systems with upper and lower critical solution temperatures: Effect of polyethylene glycol molecular weight and coagulation bath temperature. J. Membr. Sci. 2018, 565, 266–280. [Google Scholar] [CrossRef]
- Liu, Y.; Yue, X.; Zhang, S.; Ren, J.; Yang, L.; Wang, Q.; Wang, G. Synthesis of sulfonated polyphenylsulfone as candidates for antifouling ultrafiltration membrane. Sep. Purif. Technol. 2012, 98, 298–307. [Google Scholar] [CrossRef]
- Yin, Q.; Zhang, Q.; Cui, Z.; Li, W.; Xing, W. Alkali resisting polyphenylsulfone ultrafiltration membrane with tailored microstructure. Polymer 2017, 124, 128–138. [Google Scholar] [CrossRef]
- Xiao, S.; Huo, X.; Fan, S.; Zhao, K.; Yu, S.; Tan, X. Design and synthesis of Al-MOF/PPSU mixed matrix membrane with pollution resistance. Chin. J. Chem. Eng. 2021, 29, 110–120. [Google Scholar] [CrossRef]
- Isloor, A.M.; Nayak, M.C.; Inamuddin; Prabhu, B.; Ismail, N.; Ismail, A.; Asiri, A.M. Novel polyphenylsulfone (PPSU)/nano tin oxide (SnO2) mixed matrix ultrafiltration hollow fiber membranes: Fabrication, characterization and toxic dyes removal from aqueous solutions. React. Funct. Polym. 2019, 139, 170–180. [Google Scholar] [CrossRef]
- Arockiasamy, D.L.; Alhoshan, M.; Alam, J.; Muthumareeswaran; Figoli, A.; Kumar, S.A. Separation of proteins and antifouling properties of polyphenylsulfone based mixed matrix hollow fiber membranes. Sep. Purif. Technol. 2017, 174, 529–543. [Google Scholar] [CrossRef]
- Shalmani, A.K.; ElSherbiny, I.M.; Panglisch, S. Application-oriented mini-plant experiments using non-conventional model foulants to evaluate new hollow fiber membrane materials. Sep. Purif. Technol. 2020, 251, 117345. [Google Scholar] [CrossRef]
- Shalmani, A.K.; ElSherbiny, I.M.; Panglisch, S. Hydrophilic poly (phenylene sulfone) membranes for ultrafiltration. Sep. Purif. Technol. 2020, 250, 117107. [Google Scholar]
- Kolangare, I.M.; Isloor, A.M.; Karim, Z.A.; Kulal, A.; Ismail, A.F.; Asiri, A.M. Antibiofouling hollow-fiber membranes for dye rejection by embedding chitosan and silver-loaded chitosan nanoparticles. Environ. Chem. Lett. 2019, 17, 581–587. [Google Scholar] [CrossRef]
- Bildyukevich, A.V.; Plisko, T.V.; Isaichykova, Y.A.; Ovcharova, A.A. Preparation of high-flux ultrafiltration polyphenylsulfone membranes. Pet. Chem. 2018, 58, 747–759. [Google Scholar] [CrossRef]
- Slonov, A.; Musov, I.; Zhansitov, A.; Kurdanova, Z.; Shakhmurzova, K.; Khashirova, S. Investigation of the Properties of Polyphenylene Sulfone Blends. Materials 2022, 15, 6381. [Google Scholar] [CrossRef] [PubMed]
- Zhansitov, A.A.; Slonov, A.L.; Shetov, R.A.; Baikaziev, A.E.; Shakhmurzova, K.T.; Kurdanova, Z.; Khashirova, I.; Yu, S. Synthesis and properties of polyetheretherketones for 3d printing. Fibre Chem. 2018, 49, 414–419. [Google Scholar] [CrossRef]
- Kurdanova, Z.I. Synthesis and Properties of Polyphenylene Sulfone and Its Copolymers for Use in Additive Technologies. Ph.D. Thesis, Kabardino-Balkarian State University, Nalchik, Russia, 2017. [Google Scholar]
- Korshak, V.V.; Vinogradova, S.V. Nonequilibrium polycondensation. Polym. Sci. USSR 1972, 13, 415–434. [Google Scholar] [CrossRef]
- Anokhina, T.; Raeva, A.; Makaev, S.; Borisov, I.; Vasilevsky, V.; Volkov, A. Express Method of Preparation of Hollow Fiber Membrane Samples for Spinning Solution Optimization: Polysulfone as Example. Membranes 2021, 11, 396. [Google Scholar] [CrossRef]



| Type of Polymer | Manufacture | MWP, g/mol | Type of Membranes | CP, mas. % | Additive | P, L/m2·h bar | Retained Substance | MWS, g/mol | R, % | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| PPSU | Solvay, Alpharetta, GA, USA | 50,000 | HF | 20 | - | 55.8 | BSA | 69,000 | 93.9 | [11] |
| PPSU | BASF, Ludwigshafen, Germany | 51,000 | flat | 16 | - | 36–488.4 | - | - | - | [14] |
| PPSU | BASF, Ludwigshafen, Germany | 48,000 | flat | 20 | PEG6000 | 486 | HSA | 66,400 | 90 | [15] |
| sPPSU | synthesis within the work | the source does not indicate | flat | 17 | PVP | 250 | BSA | 67,000 | 92.2 | [16] |
| PPSU | Solvay, Brussels, Belgium | 50,000 | flat | 20 | PVP | 54 | Dextran | 2100 | >90 | [17] |
| Al-MOF/ PPSU | Solvay, Alpharetta, GA, USA | 65,000 | flat | 20 | Al-MOF | 47.9 | Methyl violet | 394 | 93.8 | [18] |
| PPSU | Solvay, Brussels, Belgium | 50,000 | HF | 16 | nano-SnO2 | 121 | PEG | 20,000 | 90 | [19] |
| sPPSU | Solvay (Bollate (MI)—Italy) | 49,000–55,000 | HF | 25 | nano-TiO2 | 28 | Trypsin | 20,000 | >90 | [20] |
| PPSU | BASF, Ludwigshafen, Germany | 51,000 | HF | - | multiblock copolymers based on Lutensol® AT80, Pluronic® F127 or Pluriol® E8000; PVP | ~520 | - | - | - | [21] |
| PPSU | BASF, Ludwigshafen, Germany | 51,000 | HF | 17.4 | multiblock copolymers based on Lutensol® AT80, Pluronic® F127 and Pluriol® E8000; PVP; 1,2-propanediol | 626 | PEO | 71,400 | >90 | [22] |
| PPSU | Solvay, Brussels, Belgium | 50,000 | HF | 20 | chitosan-based nanoparticles, silver-loaded chitosan nanoparticles | 56.9 | Reactive Black 5 | 991.8 | 89.3 | [23] |
| PPSU | CP, wt. % | Pore-Forming Agent | CA, wt. % |
|---|---|---|---|
| 1 | 20 | - | - |
| 2 | |||
| 3 | |||
| 4 | |||
| 5 | |||
| 5 | 15 | ||
| 18 | |||
| 22 | |||
| 24 | |||
| 18 | PEG400 | 20 | |
| 25 | |||
| 30 | |||
| 20 | 20 | ||
| 25 | |||
| 30 |
| Group | Chemical Shift (ppm) | Multiplicity |
|---|---|---|
| a | 7.89 | d |
| b | 7.07 | d |
| c | 7.12 | d |
| d | 7.58 | d |
| k1 | 7.47 | d |
| k2 | 7.87 | d |
| Sample Number | Estimated Number of Links |
|---|---|
| 1 | 9 |
| 2 | 19 |
| 3 | 30 |
| 4 | 33 |
| 5 | 38 |
| Sample Name PPSUs | Mp × 10−3 | Mw × 10−3 | MN × 10−3 | Mw/MN | MNMR × 10−3 |
|---|---|---|---|---|---|
| 1 | 9.4 | 13.4 | 4.0 | 3.3 | 3.6 |
| 2 | 21.2 | 23.5 | 11.6 | 2.0 | 7.6 |
| 3 | 26.7 | 25.0 | 5.2 | 4.8 | 12.0 |
| 4 | 34.5 | 39.5 | 20.7 | 1.9 | 13.2 |
| 5 | 84 | 102 | 38 | 2.7 | 15.2 |
| CPPSU, wt. % | CPEG400, % Macc. | P, L/m2·h bar | RBlue Dextran 69,000, % |
|---|---|---|---|
| 18 | 20 | 15.7 | 78.6 |
| 18 | 25 | 103.6 | 87.8 |
| 18 | 30 | 6.3 | 99.9 |
| 20 | 0 | 0.2 | 99.9 |
| 20 | 20 | 36.6 | 99.9 |
| 20 | 25 | 59.9 | 99.9 |
| 20 | 30 | 95.7 | 99.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anokhina, T.; Raeva, A.; Sokolov, S.; Storchun, A.; Filatova, M.; Zhansitov, A.; Kurdanova, Z.; Shakhmurzova, K.; Khashirova, S.; Borisov, I. Effect of Composition and Viscosity of Spinning Solution on Ultrafiltration Properties of Polyphenylene Sulfone Hollow-Fiber Membranes. Membranes 2022, 12, 1113. https://doi.org/10.3390/membranes12111113
Anokhina T, Raeva A, Sokolov S, Storchun A, Filatova M, Zhansitov A, Kurdanova Z, Shakhmurzova K, Khashirova S, Borisov I. Effect of Composition and Viscosity of Spinning Solution on Ultrafiltration Properties of Polyphenylene Sulfone Hollow-Fiber Membranes. Membranes. 2022; 12(11):1113. https://doi.org/10.3390/membranes12111113
Chicago/Turabian StyleAnokhina, Tatyana, Alisa Raeva, Stepan Sokolov, Alexandra Storchun, Marina Filatova, Azamat Zhansitov, Zhanna Kurdanova, Kamila Shakhmurzova, Svetlana Khashirova, and Ilya Borisov. 2022. "Effect of Composition and Viscosity of Spinning Solution on Ultrafiltration Properties of Polyphenylene Sulfone Hollow-Fiber Membranes" Membranes 12, no. 11: 1113. https://doi.org/10.3390/membranes12111113








