Recent Advances in Continuous MOF Membranes for Gas Separation and Pervaporation
Abstract
:1. Introduction
2. Developments of the MOF-Based Membrane
2.1. Continuous MOF-Based Membranes
2.1.1. In Situ Growth
2.1.2. Secondary Growth

2.1.3. Interfacial Synthesis
Static Interfacial Synthesis
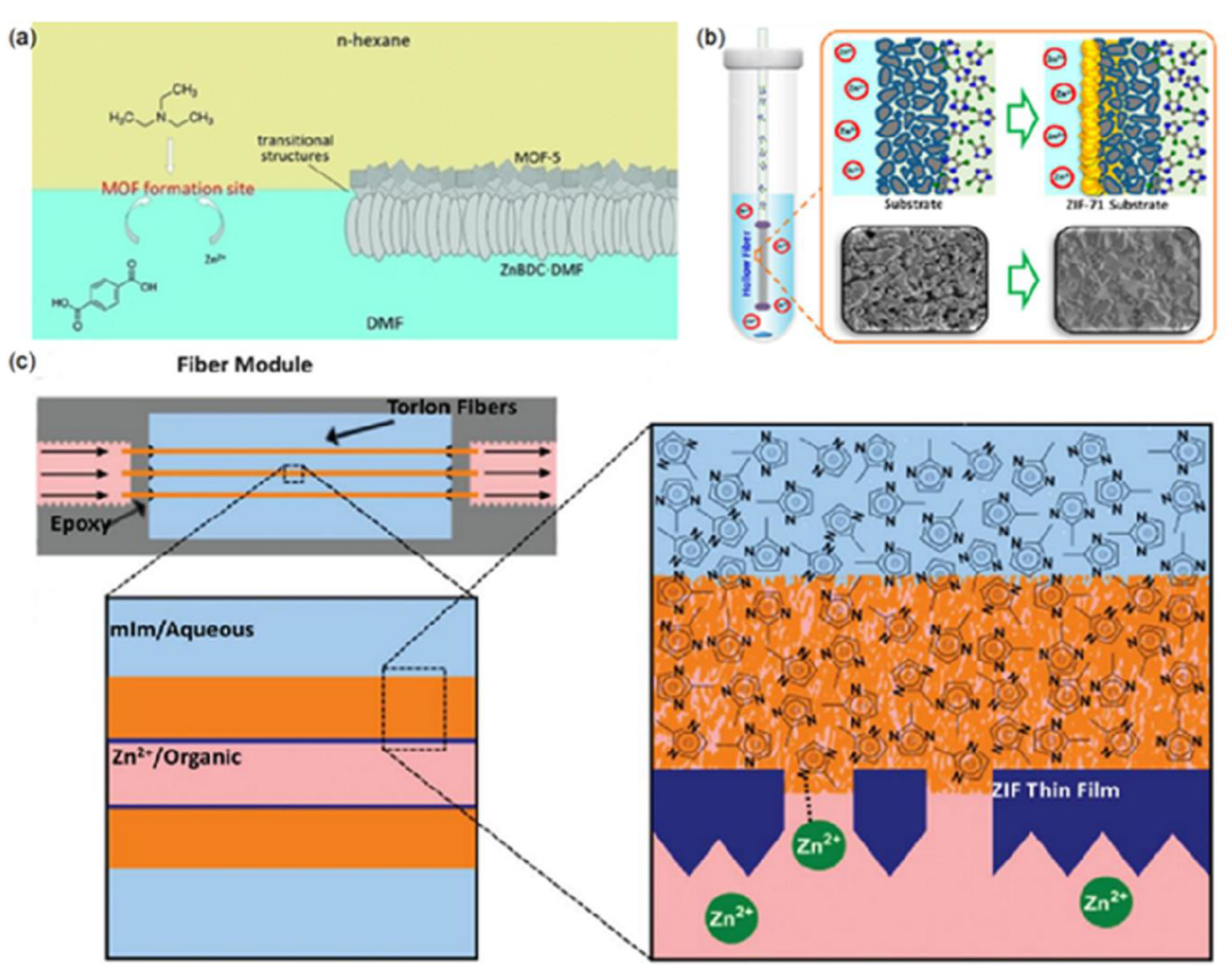
Dynamic Interfacial Synthesis
2.1.4. Liquid Phase Epitaxy (LPE)

2.1.5. Vapor Deposition

2.1.6. Electrodeposition

2.1.7. Other Methods
3. Application of Continuous MOF Membranes
3.1. Mechanisms for Gas Separation and Pervaporation
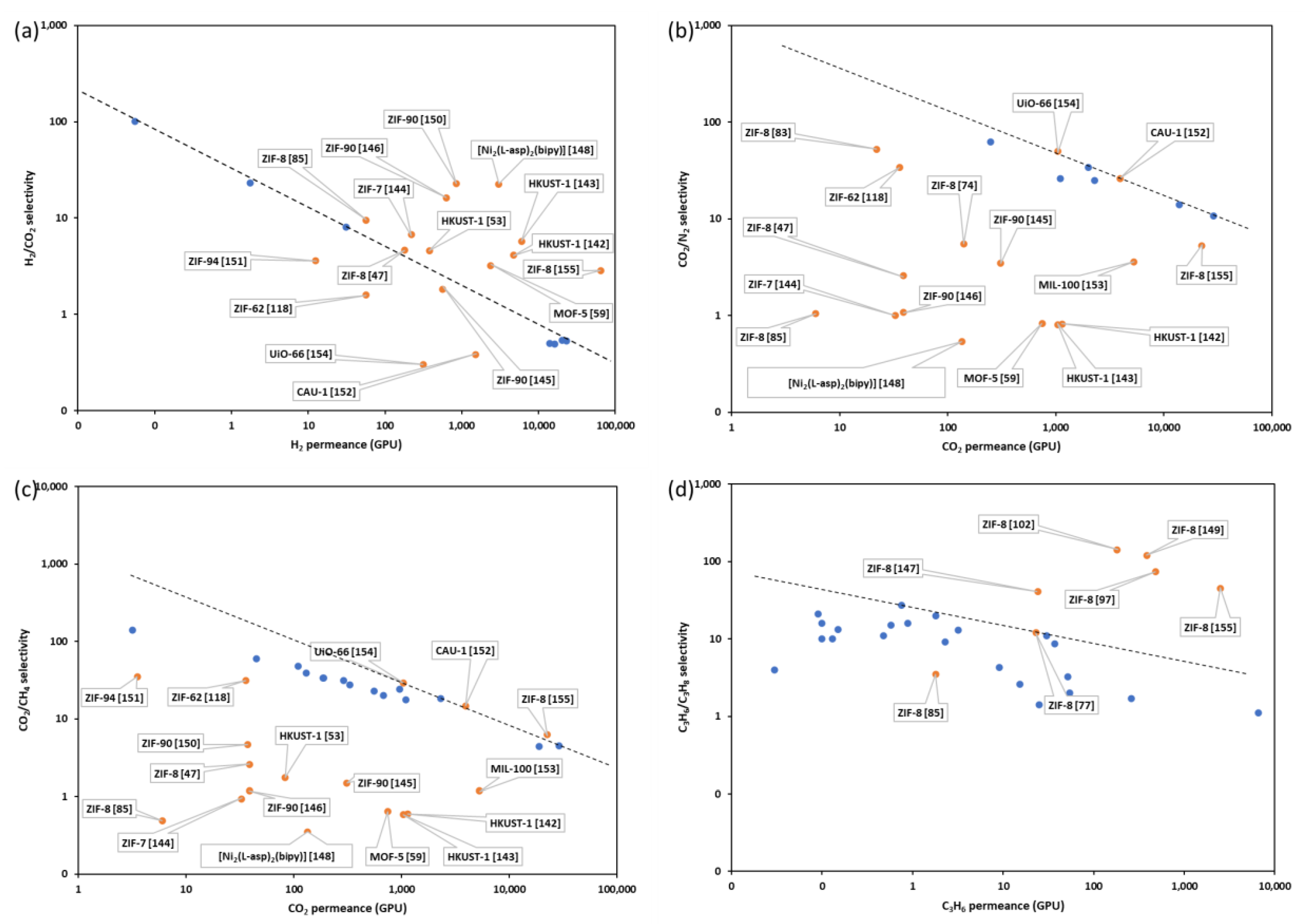
3.2. Continuous MOF Membranes in Gas Separation
| MOF | Support | Method | Permeance (GPU) | Selectivity | Ref. | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| H2 | CO2 | CH4 | N2 | C3H6 | C3H8 | H2/CO2 | CO2/N2 | CO2/CH4 | C3H6/C3H8 | ||||
| HKUST-1 | N/A | in situ | 4716.42 | 1149.25 | 1901.49 | 1402.99 | N/A | N/A | 4.10 | 0.82 | 0.60 | N/A | [142] |
| HKUST-1 | PVDF | in situ | 6000 | 1044.78 | 1761.19 | 1313.43 | N/A | N/A | 5.74 | 0.80 | 0.59 | N/A | [143] |
| MOF-5 | alumina | secondary growth | 2388.06 | 746.27 | 1164.18 | 895.52 | N/A | N/A | 3.20 | 0.83 | 0.64 | N/A | [59] |
| ZIF-8 | TiO2 | in situ | 179.1 | 38.81 | 14.93 | 14.93 | N/A | N/A | 4.61 | 2.60 | 2.60 | N/A | [47] |
| ZIF-7 | alumina | secondary growth | 220.9 | 32.84 | 35.22 | 32.84 | N/A | N/A | 6.73 | 1.00 | 0.93 | N/A | [144] |
| ZIF-90 | Torlon® fiber | secondary growth | 567.16 | 310.45 | 208.96 | 89.55 | N/A | N/A | 1.83 | 3.47 | 1.49 | N/A | [145] |
| ZIF-90 | alumina | in situ | 626.87 | 38.81 | 32.84 | 35.82 | N/A | N/A | 16.15 | 1.08 | 1.18 | N/A | [146] |
| HKUST-1 | Cu net | in situ | 379.1 | 83.58 | 47.76 | 83.58 | N/A | N/A | 4.54 | 1.00 | 1.75 | N/A | [53] |
| ZIF-8 | alumina | in situ | N/A | N/A | N/A | N/A | 24.15 | 0.59 | N/A | N/A | N/A | 41.00 | [147] |
| [Ni2(L-asp)2(bipy)] | nickel mesh | secondary growth | 3044.78 | 134.93 | 385.07 | 249.85 | N/A | N/A | 22.57 | 0.54 | 0.35 | N/A | [148] |
| ZIF-8 | Torlon | interfacial synthesis | 1710.8 | N/A | N/A | N/A | 22.9 | 2.5 | N/A | N/A | N/A | 12.00 | [77] |
| ZIF-8 | alumina | Electrodeposition | 5133 | N/A | N/A | N/A | 386 | 3.22 | N/A | N/A | N/A | 120.00 | [149] |
| ZIF-90 | alumina | in situ | 850.75 | 37.61 | 8 | N/A | N/A | N/A | 22.62 | N/A | 4.70 | N/A | [150] |
| ZIF-94 | P84® | in situ | 12.54 | 3.5 | 0.1 | N/A | N/A | N/A | 3.58 | N/A | 35.00 | N/A | [151] |
| CAU-1 | alumina | secondary growth | 1515.5 | 3940.3 | 266.24 | 150.39 | N/A | N/A | 0.38 | 26.20 | 14.80 | N/A | [152] |
| MIL-100 | alumina | in situ | N/A | 5283.58 | 4477.61 | 1462.69 | N/A | N/A | N/A | 3.61 | 1.18 | N/A | [153] |
| UiO-66 | alumina | Liquid phase epitaxy | 313.43 | 1047.76 | 35.82 | 20.9 | N/A | N/A | 0.30 | 50.13 | 29.25 | N/A | [154] |
| ZIF-62 | alumina | melt-quenching | 56.72 | 35.82 | 1.14 | 1.05 | N/A | N/A | 1.58 | 34.11 | 31.42 | N/A | [118] |
| ZIF-8 | alumina | interfacial synthesis | N/A | 140.3 | N/A | 25.56 | N/A | N/A | N/A | 5.49 | N/A | N/A | [74] |
| ZIF-8 | nylon | interfacial synthesis | 3373.13 | N/A | N/A | 746.27 | N/A | N/A | N/A | N/A | N/A | N/A | [75] |
| ZIF-8 | torlon | interfacial synthesis | N/A | 22 | N/A | 0.42 | N/A | N/A | N/A | 52.38 | N/A | N/A | [83] |
| ZIF-8 | alumina | liquid-phase epitaxy | 56.72 | 5.97 | 12.24 | 5.67 | 1.79 | 0.51 | 9.50 | 1.05 | 0.49 | 3.51 | [85] |
| UiO-66-NH2 | alumina | liquid-phase epitaxy | 35,671.64 | N/A | N/A | 8268.66 | N/A | N/A | N/A | N/A | N/A | N/A | [91] |
| ZIF-8 | alumina | vapor deposition | N/A | N/A | N/A | N/A | 479.4 | 6.48 | N/A | N/A | N/A | 73.98 | [97] |
| ZIF-8 | PVDF | vapor deposition | 64,298.51 | 22,568.35 | 3592.1 | 4258.18 | 2498.94 | 56.16 | 2.85 | 5.30 | 6.28 | 44.50 | [155] |
| ZIF-8 | alumina | Electrodeposition | N/A | N/A | N/A | N/A | 182 | 1.28 | N/A | N/A | N/A | 142.19 | [102] |
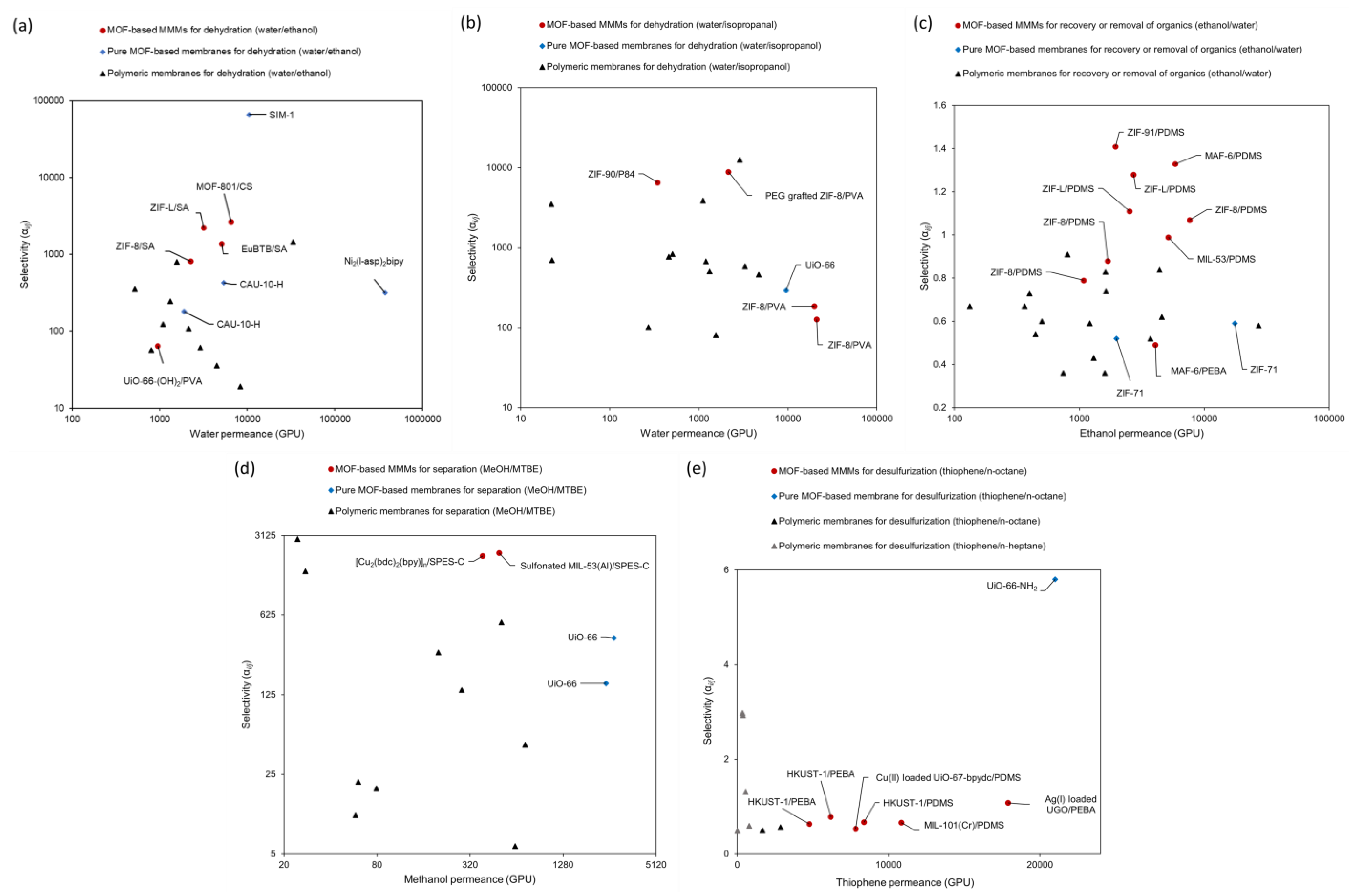
3.3. Continuous MOF Membranes in Pervaporation
4. Conclusions and Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Yong, W.F.; Zhang, H. Recent advances in polymer blend membranes for gas separation and pervaporation. Prog. Mater. Sci. 2021, 116, 100713. [Google Scholar] [CrossRef]
- Baker, R.W. Membrane Technology and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Luis, P. Fundamental Modeling of Membrane Systems: Membrane and Process Performance; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Zeng, H.; He, S.; Hosseini, S.S.; Zhu, B.; Shao, L. Emerging nanomaterial incorporated membranes for gas separation and pervaporation towards energetic-efficient applications. Adv. Membr. 2022, 2, 100015. [Google Scholar] [CrossRef]
- Pulyalina, A.; Rostovtseva, V.; Faykov, I.; Tataurov, M.; Dubovenko, R.; Shugurov, S. Development of Novel Polyamide-Imide/DES Composites and Their Application for Pervaporation and Gas Separation. Molecules 2021, 26, 990. [Google Scholar] [CrossRef] [PubMed]
- Pandey, P.; Chauhan, R. Membranes for gas separation. Prog. Polym. Sci. 2001, 26, 853–893. [Google Scholar] [CrossRef]
- Li, W.; Galiano, F.; Estager, J.; Monbaliu, J.-C.M.; Debecker, D.P.; Figoli, A.; Luis, P. Sorption and pervaporation study of methanol/dimethyl carbonate mixture with poly (etheretherketone)(PEEK-WC) membrane. J. Membr. Sci. 2018, 567, 303–310. [Google Scholar] [CrossRef]
- Qian, Q.; Asinger, P.A.; Lee, M.J.; Han, G.; Mizrahi Rodriguez, K.; Lin, S.; Benedetti, F.M.; Wu, A.X.; Chi, W.S.; Smith, Z.P. MOF-based membranes for gas separations. Chem. Rev. 2020, 120, 8161–8266. [Google Scholar] [CrossRef]
- Xu, X.; Nikolaeva, D.; Hartanto, Y.; Luis, P. MOF-based membranes for pervaporation. Sep. Purif. Technol. 2021, 278, 119233. [Google Scholar]
- Dai, F.; Luo, J.; Zhou, S.; Qin, X.; Liu, D.; Qi, H. Porous hafnium-containing acid/base bifunctional catalysts for efficient upgrading of bio-derived aldehydes. J. Bioresour. Bioprod. 2021, 6, 243–253. [Google Scholar] [CrossRef]
- Wang, J.; Sun, Y.; Zhao, X.; Chen, L.; Peng, S.; Ma, C.; Duan, G.; Liu, Z.; Wang, H.; Yuan, Y. A poly (amidoxime)-modified MOF macroporous membrane for high-efficient uranium extraction from seawater. e-Polymers 2022, 22, 399–410. [Google Scholar] [CrossRef]
- Jian, S.; Ma, X.; Wang, Q.; Wu, J.; Wang, Y.; Jiang, S.; Xu, W.; Yang, W. Hierarchical porous Co3O4 nanocages with elaborate microstructures derived from ZIF-67 toward lithium storage. Vacuum 2021, 184, 109879. [Google Scholar] [CrossRef]
- Caro, J. Are MOF membranes better in gas separation than those made of zeolites? Curr. Opin. Chem. Eng. 2011, 1, 77–83. [Google Scholar] [CrossRef]
- Shekhah, O.; Chernikova, V.; Belmabkhout, Y.; Eddaoudi, M. Metal–organic framework membranes: From fabrication to gas separation. Crystals 2018, 8, 412. [Google Scholar] [CrossRef] [Green Version]
- Kang, Z.; Fan, L.; Sun, D. Recent advances and challenges of metal–organic framework membranes for gas separation. J. Mater. Chem. A 2017, 5, 10073–10091. [Google Scholar] [CrossRef]
- Yang, L.; Qian, S.; Wang, X.; Cui, X.; Chen, B.; Xing, H. Energy-efficient separation alternatives: Metal–organic frameworks and membranes for hydrocarbon separation. Chem. Soc. Rev. 2020, 49, 5359–5406. [Google Scholar] [CrossRef] [PubMed]
- Xue, D.-X.; Cadiau, A.; Weseliński, Ł.J.; Jiang, H.; Bhatt, P.M.; Shkurenko, A.; Wojtas, L.; Chen, Z.; Belmabkhout, Y.; Adil, K. Topology meets MOF chemistry for pore-aperture fine tuning: Ftw-MOF platform for energy-efficient separations via adsorption kinetics or molecular sieving. Chem. Commun. 2018, 54, 6404–6407. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Zhao, S.; Tian, Z.; Duan, G.; Pan, H.; Yue, Y.; Li, S.; Jian, S.; Yang, W.; Liu, K. MOFs meet wood: Reusable magnetic hydrophilic composites toward efficient water treatment with super-high dye adsorption capacity at high dye concentration. Chem. Eng. J. 2022, 446, 136851. [Google Scholar] [CrossRef]
- Ma, X.; Xiong, Y.; Liu, Y.; Han, J.; Duan, G.; Chen, Y.; He, S.; Mei, C.; Jiang, S.; Zhang, K. When MOFs meet wood: From opportunities toward applications. Chem 2022, 8, 2342–2361. [Google Scholar] [CrossRef]
- Hermes, S.; Schröder, F.; Chelmowski, R.; Wöll, C.; Fischer, R.A. Selective nucleation and growth of metal- organic open framework thin films on patterned COOH/CF3-terminated self-assembled monolayers on Au (111). J. Am. Chem. Soc. 2005, 127, 13744–13745. [Google Scholar] [CrossRef]
- Wang, X.-P.; Hou, J.; Chen, F.-S.; Meng, X.-M. In-situ growth of metal-organic framework film on a polydopamine-modified flexible substrate for antibacterial and forward osmosis membranes. Sep. Purif. Technol. 2020, 236, 116239. [Google Scholar] [CrossRef]
- Kwon, H.T.; Jeong, H.-K. In situ synthesis of thin zeolitic–imidazolate framework ZIF-8 membranes exhibiting exceptionally high propylene/propane separation. J. Am. Chem. Soc. 2013, 135, 10763–10768. [Google Scholar] [CrossRef]
- Tuergong, M.; Nizamidin, P.; Yimit, A.; Simayi, R. In situ growth and optical gas adsorption performance of Zn (II) metal–organic framework membranes at room temperature. Analyst 2019, 144, 4887–4896. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Xia, T.; Zhao, D.; Cui, Y.; Yang, Y.; Qian, G. In situ secondary growth of Eu (III)-organic framework film for fluorescence sensing of sulfur dioxide. Sens. Actuators B Chem. 2018, 260, 63–69. [Google Scholar] [CrossRef]
- Wu, X.; Yang, Y.; Lu, X.; Wang, Z. Seeded growth of high-performance ZIF-8 membranes in thick wall autoclaves assisted by modulator. J. Membr. Sci. 2020, 613, 118518. [Google Scholar] [CrossRef]
- Ma, X.; Li, Y.; Huang, A. Synthesis of nano-sheets seeds for secondary growth of highly hydrogen permselective ZIF-95 membranes. J. Membr. Sci. 2020, 597, 117629. [Google Scholar] [CrossRef]
- Wu, W.; Jia, M.; Su, J.; Li, Z.; Li, W. Air–water interfacial synthesis of metal–organic framework hollow fiber membranes for water purification. AIChE J. 2020, 66, e16238. [Google Scholar] [CrossRef]
- Vatanpour, V.; Khorshidi, S. Surface modification of polyvinylidene fluoride membranes with ZIF-8 nanoparticles layer using interfacial method for BSA separation and dye removal. Mater. Chem. Phys. 2020, 241, 122400. [Google Scholar] [CrossRef]
- Yao, J.; Wang, H. Zeolitic imidazolate framework composite membranes and thin films: Synthesis and applications. Chem. Soc. Rev. 2014, 43, 4470–4493. [Google Scholar] [CrossRef]
- Yeo, Z.Y.; Chai, S.-P.; Zhu, P.W.; Mohamed, A.R. An overview: Synthesis of thin films/membranes of metal organic frameworks and its gas separation performances. RSC Adv. 2014, 4, 54322–54334. [Google Scholar] [CrossRef]
- Zhang, C.; Wu, B.-H.; Ma, M.-Q.; Wang, Z.; Xu, Z.-K. Ultrathin metal/covalent–organic framework membranes towards ultimate separation. Chem. Soc. Rev. 2019, 48, 3811–3841. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.-J.; Xian, Y.-M.; Wang, C.; Mao, H.-L.; Kind, M.; Abu-Husein, T.; Chen, Z.; Zhu, S.-B.; Ren, B.; Terfort, A. Liquid-phase epitaxial growth of highly oriented and multivariate surface-attached metal–organic frameworks. J. Am. Chem. Soc. 2019, 141, 18984–18993. [Google Scholar] [CrossRef]
- Liu, J.; Wöll, C. Surface-supported metal–organic framework thin films: Fabrication methods, applications, and challenges. Chem. Soc. Rev. 2017, 46, 5730–5770. [Google Scholar] [CrossRef] [PubMed]
- Eddaoudi, M.; Shekhah, O.; Belmabkhout, Y. Fabrication of Highly CO2 Selective Metal-Organic Framework Membrane using Liquid Phase Epitaxy Approach. U.S. Patents US20170203261A1, 15 October 2019. [Google Scholar]
- Chernikova, V.; Shekhah, O.; Belmabkhout, Y.; Eddaoudi, M. Nanoporous fluorinated metal–organic framework-based membranes for CO2 capture. ACS Appl. Nano Mater. 2020, 3, 6432–6439. [Google Scholar] [CrossRef]
- Boscher, N.D.; Wang, M.; Perrotta, A.; Heinze, K.; Creatore, M.; Gleason, K.K. Metal–organic covalent network chemical vapor deposition for gas separation. Adv. Mater. 2016, 28, 7479–7485. [Google Scholar] [CrossRef] [Green Version]
- Li, W. Metal–organic framework membranes: Production, modification, and applications. Prog. Mater. Sci. 2019, 100, 21–63. [Google Scholar] [CrossRef]
- Betard, A.; Fischer, R.A. Metal–organic framework thin films: From fundamentals to applications. Chem. Rev. 2012, 112, 1055–1083. [Google Scholar] [CrossRef]
- Stassen, I.; Styles, M.; Grenci, G.; Gorp, H.V.; Vanderlinden, W.; Feyter, S.D.; Falcaro, P.; Vos, D.D.; Vereecken, P.; Ameloot, R. Chemical vapour deposition of zeolitic imidazolate framework thin films. Nat. Mater. 2016, 15, 304–310. [Google Scholar] [CrossRef] [Green Version]
- Liu, X. Metal-organic framework UiO-66 membranes. Front. Chem. Sci. Eng. 2020, 14, 216–232. [Google Scholar] [CrossRef] [Green Version]
- Cruz, A.J.; Stassen, I.; Krishtab, M.; Marcoen, K.; Stassin, T.; Rodríguez-Hermida, S.; Teyssandier, J.; Pletincx, S.; Verbeke, R.; Rubio-Giménez, V. Integrated cleanroom process for the vapor-phase deposition of large-area zeolitic imidazolate framework thin films. Chem. Mater. 2019, 31, 9462–9471. [Google Scholar] [CrossRef]
- Alizadeh, S.; Nematollahi, D. Convergent and divergent paired electrodeposition of metal-organic framework thin films. Sci. Rep. 2019, 9, 14325. [Google Scholar] [CrossRef] [Green Version]
- Li, M.; Dincă, M. Selective formation of biphasic thin films of metal–organic frameworks by potential-controlled cathodic electrodeposition. Chem. Sci. 2014, 5, 107–111. [Google Scholar] [CrossRef] [Green Version]
- Liu, H.; Wang, H.; Chu, T.; Yu, M.; Yang, Y. An electrodeposited lanthanide MOF thin film as a luminescent sensor for carbonate detection in aqueous solution. J. Mater. Chem. C 2014, 2, 8683–8690. [Google Scholar] [CrossRef]
- Lin, Y.S. Metal organic framework membranes for separation applications. Curr. Opin. Chem. Eng. 2015, 8, 21–28. [Google Scholar] [CrossRef]
- Liu, Y.; Ng, Z.; Khan, E.A.; Jeong, H.-K.; Ching, C.-B.; Lai, Z. Synthesis of continuous MOF-5 membranes on porous α-alumina substrates. Microporous Mesoporous Mater. 2009, 118, 296–301. [Google Scholar] [CrossRef]
- Bux, H.; Liang, F.; Li, Y.; Cravillon, J.; Wiebcke, M.; Caro, J.R. Zeolitic imidazolate framework membrane with molecular sieving properties by microwave-assisted solvothermal synthesis. J. Am. Chem. Soc. 2009, 131, 16000–16001. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.; Yao, J.; Wang, K.; He, L.; Webley, P.A.; Chen, C.-S.; Wang, H. Preparation of ZIF-8 membranes supported on ceramic hollow fibers from a concentrated synthesis gel. J. Membr. Sci. 2011, 385, 187–193. [Google Scholar] [CrossRef]
- Kang, Z.; Xue, M.; Fan, L.; Ding, J.; Guo, L.; Gao, L.; Qiu, S. “Single nickel source” in situ fabrication of a stable homochiral MOF membrane with chiral resolution properties. Chem. Commun. 2013, 49, 10569–10571. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, Y.; Kong, L.; Liu, H.; Qiu, J.; Han, W.; Weng, L.-T.; Yeung, K.L.; Zhu, W. A simple and scalable method for preparing low-defect ZIF-8 tubular membranes. J. Mater. Chem. A 2013, 1, 10635–10638. [Google Scholar] [CrossRef]
- Ma, L.; Svec, F.; Tan, T.; Lv, Y. In-situ growth of highly permeable zeolite imidazolate framework membranes on porous polymer substrate using metal chelated polyaniline as interface layer. J. Membr. Sci. 2019, 576, 1–8. [Google Scholar] [CrossRef]
- Wu, X.; Wei, W.; Jiang, J.; Caro, J.; Huang, A. High-Flux High-Selectivity Metal–Organic Framework MIL-160 Membrane for Xylene Isomer Separation by Pervaporation. Angew. Chem. Int. Ed. 2018, 57, 15354–15358. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Zhu, G.; Hewitt, I.J.; Qiu, S. “Twin copper source” growth of metal-organic framework membrane: Cu3(BTC)2 with high permeability and selectivity for recycling H2. J. Am. Chem. Soc. 2009, 131, 1646–1647. [Google Scholar] [CrossRef]
- Algieri, C.; Golemme, G.; Kallus, S.; Ramsay, J. Preparation of thin supported MFI membranes by in situ nucleation and secondary growth. Microporous Mesoporous Mater. 2001, 47, 127–134. [Google Scholar] [CrossRef]
- Snyder, M.A.; Tsapatsis, M. Hierarchical nanomanufacturing: From shaped zeolite nanoparticles to high-performance separation membranes. Angew. Chem. Int. Ed. 2007, 46, 7560–7573. [Google Scholar] [CrossRef] [PubMed]
- Bohrman, J.A.; Carreon, M.A. Synthesis and CO2/CH4 separation performance of Bio-MOF-1 membranes. Chem. Commun. 2012, 48, 5130–5132. [Google Scholar] [CrossRef] [Green Version]
- Pan, Y.; Li, T.; Lestari, G.; Lai, Z. Effective separation of propylene/propane binary mixtures by ZIF-8 membranes. J. Membr. Sci. 2012, 390, 93–98. [Google Scholar] [CrossRef]
- Wu, Y.-N.; Li, F.; Liu, H.; Zhu, W.; Teng, M.; Jiang, Y.; Li, W.; Xu, D.; He, D.; Hannam, P. Electrospun fibrous mats as skeletons to produce free-standing MOF membranes. J. Mater. Chem. 2012, 22, 16971–16978. [Google Scholar] [CrossRef]
- Yoo, Y.; Lai, Z.; Jeong, H.-K. Fabrication of MOF-5 membranes using microwave-induced rapid seeding and solvothermal secondary growth. Microporous Mesoporous Mater. 2009, 123, 100–106. [Google Scholar] [CrossRef]
- Hu, Y.; Wei, J.; Liang, Y.; Zhang, H.; Zhang, X.; Shen, W.; Wang, H. Zeolitic imidazolate framework/graphene oxide hybrid nanosheets as seeds for the growth of ultrathin molecular sieving membranes. Angew. Chem. 2016, 128, 2088–2092. [Google Scholar] [CrossRef]
- Hu, Y.; Dong, X.; Nan, J.; Jin, W.; Ren, X.; Xu, N.; Lee, Y.M. Metal–organic framework membranes fabricated via reactive seeding. Chem. Commun. 2011, 47, 737–739. [Google Scholar] [CrossRef]
- Guerrero, V.V.; Yoo, Y.; McCarthy, M.C.; Jeong, H.-K. HKUST-1 membranes on porous supports using secondary growth. J. Mater. Chem. 2010, 20, 3938–3943. [Google Scholar] [CrossRef]
- Ranjan, R.; Tsapatsis, M. Microporous metal organic framework membrane on porous support using the seeded growth method. Chem. Mater. 2009, 21, 4920–4924. [Google Scholar] [CrossRef]
- Pan, Y.; Wang, B.; Lai, Z. Synthesis of ceramic hollow fiber supported zeolitic imidazolate framework-8 (ZIF-8) membranes with high hydrogen permeability. J. Membr. Sci. 2012, 421, 292–298. [Google Scholar] [CrossRef]
- Wang, S.; Kang, Z.; Xu, B.; Fan, L.; Li, G.; Wen, L.; Xin, X.; Xiao, Z.; Pang, J.; Du, X. Wettability switchable metal-organic framework membranes for pervaporation of water/ethanol mixtures. Inorg. Chem. Commun. 2017, 82, 64–67. [Google Scholar] [CrossRef]
- Diestel, L.; Bux, H.; Wachsmuth, D.; Caro, J. Pervaporation studies of n-hexane, benzene, mesitylene and their mixtures on zeolitic imidazolate framework-8 membranes. Microporous Mesoporous Mater. 2012, 164, 288–293. [Google Scholar] [CrossRef]
- Tao, K.; Kong, C.; Chen, L. High performance ZIF-8 molecular sieve membrane on hollow ceramic fiber via crystallizing-rubbing seed deposition. Chem. Eng. J. 2013, 220, 1–5. [Google Scholar] [CrossRef]
- Lee, M.J.; Hamid, M.R.A.; Lee, J.; Kim, J.S.; Lee, Y.M.; Jeong, H.-K. Ultrathin zeolitic-imidazolate framework ZIF-8 membranes on polymeric hollow fibers for propylene/propane separation. J. Membr. Sci. 2018, 559, 28–34. [Google Scholar] [CrossRef]
- Zhao, X.; Zhang, H.; Xu, S.; Wang, Y. ZIF-8 membrane synthesized via covalent-assisted seeding on polyimide substrate for pervaporation dehydration. AIChE J. 2019, 65, e16620. [Google Scholar] [CrossRef]
- Liu, Y.; Ban, Y.; Yang, W. Microstructural engineering and architectural design of metal–organic framework membranes. Adv. Mater. 2017, 29, 1606949. [Google Scholar] [CrossRef] [PubMed]
- Ameloot, R.; Vermoortele, F.; Vanhove, W.; Roeffaers, M.B.; Sels, B.F.; De Vos, D.E. Interfacial synthesis of hollow metal–organic framework capsules demonstrating selective permeability. Nat. Chem. 2011, 3, 382–387. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Dong, D.; Li, D.; He, L.; Xu, G.; Wang, H. Contra-diffusion synthesis of ZIF-8 films on a polymer substrate. Chem. Commun. 2011, 47, 2559–2561. [Google Scholar] [CrossRef]
- Lu, H.; Zhu, S. Interfacial Synthesis of Free-Standing Metal–Organic Framework Membranes. Eur. J. Inorg. Chem. 2013, 2013, 1294–1300. [Google Scholar] [CrossRef]
- Chen, S.S.; Yang, Z.-J.; Chang, C.-H.; Koh, H.-U.; Al-Saeedi, S.I.; Tung, K.-L.; Wu, K.C.-W. Interfacial nanoarchitectonics for ZIF-8 membranes with enhanced gas separation. Beilstein J. Nanotechnol. 2022, 13, 313–324. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Yao, J.; Li, L.; Zhong, Z.; Chen, F.; Wang, H. Aqueous solution synthesis of ZIF-8 films on a porous nylon substrate by contra-diffusion method. Microporous Mesoporous Mater. 2013, 179, 10–16. [Google Scholar] [CrossRef]
- Huang, K.; Li, Q.; Liu, G.; Shen, J.; Guan, K.; Jin, W. A ZIF-71 hollow fiber membrane fabricated by contra-diffusion. ACS Appl. Mater. Interfaces 2015, 7, 16157–16160. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.J.; Brunelli, N.A.; Eum, K.; Rashidi, F.; Johnson, J.; Koros, W.J.; Jones, C.W.; Nair, S. Interfacial microfluidic processing of metal-organic framework hollow fiber membranes. Science 2014, 345, 72–75. [Google Scholar] [CrossRef]
- Peng, Y.; Li, Y.; Ban, Y.; Jin, H.; Jiao, W.; Liu, X.; Yang, W. Metal-organic framework nanosheets as building blocks for molecular sieving membranes. Science 2014, 346, 1356–1359. [Google Scholar] [CrossRef]
- Shen, J.; Liu, G.; Huang, K.; Chu, Z.; Jin, W.; Xu, N. Subnanometer two-dimensional graphene oxide channels for ultrafast gas sieving. ACS Nano 2016, 10, 3398–3409. [Google Scholar] [CrossRef]
- Zhang, S.; Wang, Z.; Ren, H.; Zhang, F.; Jin, J. Nanoporous film-mediated growth of ultrathin and continuous metal–organic framework membranes for high-performance hydrogen separation. J. Mater. Chem. A 2017, 5, 1962–1966. [Google Scholar] [CrossRef]
- Huang, K.; Dong, Z.; Li, Q.; Jin, W. Growth of a ZIF-8 membrane on the inner-surface of a ceramic hollow fiber via cycling precursors. Chem. Commun. 2013, 49, 10326–10328. [Google Scholar] [CrossRef]
- Cacho-Bailo, F.; Caro, G.; Etxeberría-Benavides, M.; Karvan, O.; Téllez, C.; Coronas, J. High selectivity ZIF-93 hollow fiber membranes for gas separation. Chem. Commun. 2015, 51, 11283–11285. [Google Scholar] [CrossRef]
- Marti, A.M.; Wickramanayake, W.; Dahe, G.; Sekizkardes, A.; Bank, T.L.; Hopkinson, D.P.; Venna, S.R. Continuous flow processing of ZIF-8 membranes on polymeric porous hollow fiber supports for CO2 capture. ACS Appl. Mater. Interfaces 2017, 9, 5678–5682. [Google Scholar] [CrossRef]
- Shekhah, O.; Wang, H.; Strunskus, T.; Cyganik, P.; Zacher, D.; Fischer, R.; Wöll, C. Layer-by-layer growth of oriented metal organic polymers on a functionalized organic surface. Langmuir 2007, 23, 7440–7442. [Google Scholar] [CrossRef] [PubMed]
- Shekhah, O.; Swaidan, R.; Belmabkhout, Y.; Du Plessis, M.; Jacobs, T.; Barbour, L.J.; Pinnau, I.; Eddaoudi, M. The liquid phase epitaxy approach for the successful construction of ultra-thin and defect-free ZIF-8 membranes: Pure and mixed gas transport study. Chem. Commun. 2014, 50, 2089–2092. [Google Scholar] [CrossRef] [PubMed]
- Arslan, H.K.; Shekhah, O.; Wohlgemuth, J.; Franzreb, M.; Fischer, R.A.; Wöll, C. High-Throughput Fabrication of Uniform and Homogenous MOF Coatings. Adv. Funct. Mater. 2011, 21, 4228–4231. [Google Scholar] [CrossRef]
- Gu, Z.-G.; Pfriem, A.; Hamsch, S.; Breitwieser, H.; Wohlgemuth, J.; Heinke, L.; Gliemann, H.; Wöll, C. Transparent films of metal-organic frameworks for optical applications. Microporous Mesoporous Mater. 2015, 211, 82–87. [Google Scholar] [CrossRef]
- Shekhah, O.; Wang, H.; Kowarik, S.; Schreiber, F.; Paulus, M.; Tolan, M.; Sternemann, C.; Evers, F.; Zacher, D.; Fischer, R.A. Step-by-step route for the synthesis of metal-organic frameworks. J. Am. Chem. Soc. 2007, 129, 15118–15119. [Google Scholar] [CrossRef] [PubMed]
- Valadez Sánchez, E.P.; Gliemann, H.; Haas-Santo, K.; Wöll, C.; Dittmeyer, R. ZIF-8 SURMOF Membranes Synthesized by Au-Assisted Liquid Phase Epitaxy for Application in Gas Separation. Chem. Ing. Tech. 2016, 88, 1798–1805. [Google Scholar] [CrossRef]
- Hashem, T.; Valadez Sánchez, E.P.; Weidler, P.G.; Gliemann, H.; Alkordi, M.H.; Wöll, C. Liquid-Phase Quasi-Epitaxial Growth of Highly Stable, Monolithic UiO-66-NH2 MOF thin Films on Solid Substrates. ChemistryOpen 2020, 9, 524–527. [Google Scholar] [CrossRef] [Green Version]
- Micero, A.; Hashem, T.; Gliemann, H.; Léon, A. Hydrogen Separation Performance of UiO-66-NH2 Membranes Grown via Liquid-Phase Epitaxy Layer-by-Layer Deposition and One-Pot Synthesis. Membranes 2021, 11, 735. [Google Scholar] [CrossRef]
- Stassen, I.; De Vos, D.; Ameloot, R. Vapor-Phase Deposition and Modification of Metal–Organic Frameworks: State-of-the-Art and Future Directions. Chem. A Eur. J. 2016, 22, 14452–14460. [Google Scholar] [CrossRef]
- Bennett, T.D.; Cheetham, A.K. Amorphous metal–organic frameworks. Acc. Chem. Res. 2014, 47, 1555–1562. [Google Scholar] [CrossRef]
- Fischer, D.; von Mankowski, A.; Ranft, A.; Vasa, S.K.; Linser, R.; Mannhart, J.; Lotsch, B.V. ZIF-8 films prepared by femtosecond pulsed-laser deposition. Chem. Mater. 2017, 29, 5148–5155. [Google Scholar] [CrossRef]
- Ritala, M.; Parala, H.; Kanjolia, R.; Dupuis, R.D.; Alexandrov, S.; Irvine, S.J.; Palgrave, R.; Parkin, I.P.; Niinisto, J.; Krumdieck, S. Chemical Vapour Deposition: Precursors, Processes and Applications; Royal Society of Chemistry: London, UK, 2008. [Google Scholar]
- Salmi, L.D.; Heikkilä, M.J.; Puukilainen, E.; Sajavaara, T.; Grosso, D.; Ritala, M. Studies on atomic layer deposition of MOF-5 thin films. Microporous Mesoporous Mater. 2013, 182, 147–154. [Google Scholar] [CrossRef]
- Ma, X.; Kumar, P.; Mittal, N.; Khlyustova, A.; Daoutidis, P.; Mkhoyan, K.A.; Tsapatsis, M. Zeolitic imidazolate framework membranes made by ligand-induced permselectivation. Science 2018, 361, 1008–1011. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lausund, K.B.; Petrovic, V.; Nilsen, O. All-gas-phase synthesis of amino-functionalized UiO-66 thin films. Dalton Trans. 2017, 46, 16983–16992. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lausund, K.B.; Olsen, M.S.; Hansen, P.-A.; Valen, H.; Nilsen, O. MOF thin films with bi-aromatic linkers grown by molecular layer deposition. J. Mater. Chem. A 2020, 8, 2539–2548. [Google Scholar] [CrossRef] [Green Version]
- Multia, J.; Karppinen, M. Atomic/Molecular Layer Deposition for Designer′s Functional Metal–Organic Materials. Adv. Mater. Interfaces 2022, 9, 2200210. [Google Scholar] [CrossRef]
- Li, W.; Su, P.; Li, Z.; Xu, Z.; Wang, F.; Ou, H.; Zhang, J.; Zhang, G.; Zeng, E. Ultrathin metal–organic framework membrane production by gel–vapour deposition. Nat. Commun. 2017, 8, 406. [Google Scholar] [CrossRef] [Green Version]
- Wei, R.; Chi, H.Y.; Li, X.; Lu, D.; Wan, Y.; Yang, C.W.; Lai, Z. Aqueously cathodic deposition of ZIF-8 membranes for superior propylene/propane separation. Adv. Funct. Mater. 2020, 30, 1907089. [Google Scholar] [CrossRef]
- Van de Voorde, B.; Ameloot, R.; Stassen, I.; Everaert, M.; De Vos, D.; Tan, J.-C. Mechanical properties of electrochemically synthesised metal–organic framework thin films. J. Mater. Chem. C 2013, 1, 7716–7724. [Google Scholar] [CrossRef]
- Li, W.-J.; Tu, M.; Cao, R.; Fischer, R.A. Metal–organic framework thin films: Electrochemical fabrication techniques and corresponding applications & perspectives. J. Mater. Chem. A 2016, 4, 12356–12369. [Google Scholar]
- Hod, I.; Bury, W.; Karlin, D.M.; Deria, P.; Kung, C.W.; Katz, M.J.; So, M.; Klahr, B.; Jin, D.; Chung, Y.W. Directed growth of electroactive metal-organic framework thin films using electrophoretic deposition. Adv. Mater. 2014, 26, 6295–6300. [Google Scholar] [CrossRef] [PubMed]
- Czaja, A.U.; Trukhan, N.; Müller, U. Industrial applications of metal–organic frameworks. Chem. Soc. Rev. 2009, 38, 1284–1293. [Google Scholar] [CrossRef] [PubMed]
- Mueller, U.; Puetter, H.; Hesse, M.; Schubert, M.; Wessel, H.; Huff, J.; Guzmann, M. Method for Electrochemical Production of a Crystalline Porous Metal Organic Skeleton Material. U.S. Patents US7968739B2, 28 June 2011. [Google Scholar]
- Campagnol, N.; Souza, E.R.; De Vos, D.E.; Binnemans, K.; Fransaer, J. Luminescent terbium-containing metal–organic framework films: New approaches for the electrochemical synthesis and application as detectors for explosives. Chem. Commun. 2014, 50, 12545–12547. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, K.-Y.; Wang, J.-C.; Lin, C.-Y.; Lin, W.-R.; Chen, Y.-A.; Tsai, F.-J.; Chuang, Y.-C.; Lin, G.-Y.; Ni, C.-W.; Zeng, Y.-T. Electrochemical synthesis, characterization of Ir–Zn containing coordination polymer, and application in oxygen and glucose sensing. Dalton Trans. 2014, 43, 6536–6547. [Google Scholar] [CrossRef]
- Worrall, S.D.; Mann, H.; Rogers, A.; Bissett, M.A.; Attfield, M.P.; Dryfe, R.A. Electrochemical deposition of zeolitic imidazolate framework electrode coatings for supercapacitor electrodes. Electrochim. Acta 2016, 197, 228–240. [Google Scholar] [CrossRef]
- Suttipat, D.; Butcha, S.; Assavapanumat, S.; Maihom, T.; Gupta, B.; Perro, A.; Sojic, N.; Kuhn, A.; Wattanakit, C. Chiral macroporous MOF surfaces for electroassisted enantioselective adsorption and separation. ACS Appl. Mater. Interfaces 2020, 12, 36548–36557. [Google Scholar] [CrossRef]
- Van Assche, T.R.; Denayer, J.F. Fabrication and separation performance evaluation of a metal–organic framework based microseparator device. Chem. Eng. Sci. 2013, 95, 65–72. [Google Scholar] [CrossRef]
- Campagnol, N.; Van Assche, T.R.; Li, M.; Stappers, L.; Dincă, M.; Denayer, J.F.; Binnemans, K.; De Vos, D.E.; Fransaer, J. On the electrochemical deposition of metal–organic frameworks. J. Mater. Chem. A 2016, 4, 3914–3925. [Google Scholar] [CrossRef] [Green Version]
- Hauser, J.L.; Tso, M.; Fitchmun, K.; Oliver, S.R. Anodic electrodeposition of several metal organic framework thin films on indium tin oxide glass. Cryst. Growth Des. 2019, 19, 2358–2365. [Google Scholar] [CrossRef]
- Ameloot, R.; Stappers, L.; Fransaer, J.; Alaerts, L.; Sels, B.F.; De Vos, D.E. Patterned growth of metal-organic framework coatings by electrochemical synthesis. Chem. Mater. 2009, 21, 2580–2582. [Google Scholar] [CrossRef]
- Li, M.; Dinca, M. Reductive electrosynthesis of crystalline metal–organic frameworks. J. Am. Chem. Soc. 2011, 133, 12926–12929. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, H.; Liu, H.; Zhitomirsky, I.; Zhu, S. Preparation of metal–organic framework films by electrophoretic deposition method. Mater. Lett. 2015, 142, 19–22. [Google Scholar] [CrossRef]
- Wang, Y.; Jin, H.; Ma, Q.; Mo, K.; Mao, H.; Feldhoff, A.; Cao, X.; Li, Y.; Pan, F.; Jiang, Z. A MOF glass membrane for gas separation. Angew. Chem. 2020, 132, 4395–4399. [Google Scholar] [CrossRef]
- Marti, A.M.; Tran, D.; Balkus, K.J. Fabrication of a Substituted Imidazolate Material 1 (SIM-1) membrane using post synthetic modification (PSM) for pervaporation of water/ethanol mixtures. J. Porous Mater. 2015, 22, 1275–1284. [Google Scholar] [CrossRef]
- Nath, K.; Ahmed, A.; Siegel, D.J.; Matzger, A.J. Microscale Determination of Binary Gas Adsorption Isotherms in MOFs. J. Am. Chem. Soc. 2022, 144, 20939–20946. [Google Scholar] [CrossRef] [PubMed]
- Kundu, A.; Sillar, K.; Sauer, J. Ab initio prediction of adsorption isotherms for gas mixtures by Grand Canonical Monte Carlo simulations on a lattice of sites. J. Phys. Chem. Lett. 2017, 8, 2713–2718. [Google Scholar] [CrossRef] [Green Version]
- Kundu, A.; Sillar, K.; Sauer, J. Predicting adsorption selectivities from pure gas isotherms for gas mixtures in metal–organic frameworks. Chem. Sci. 2020, 11, 643–655. [Google Scholar] [CrossRef] [Green Version]
- Hou, Q.; Zhou, S.; Wei, Y.; Caro, J.R.; Wang, H. Balancing the grain boundary structure and the framework flexibility through bimetallic Metal–Organic Framework (MOF) membranes for gas separation. J. Am. Chem. Soc. 2020, 142, 9582–9586. [Google Scholar] [CrossRef]
- Wiersum, A.D.; Chang, J.-S.; Serre, C.; Llewellyn, P.L. An adsorbent performance indicator as a first step evaluation of novel sorbents for gas separations: Application to metal–organic frameworks. Langmuir 2013, 29, 3301–3309. [Google Scholar] [CrossRef]
- Furukawa, H.; Ko, N.; Go, Y.B.; Aratani, N.; Choi, S.B.; Choi, E.; Yazaydin, A.Ö.; Snurr, R.Q.; O’Keeffe, M.; Kim, J. Ultrahigh porosity in metal-organic frameworks. Science 2010, 329, 424–428. [Google Scholar] [CrossRef] [Green Version]
- Thornton, A.W.; Dubbeldam, D.; Liu, M.S.; Ladewig, B.P.; Hill, A.J.; Hill, M.R. Feasibility of zeolitic imidazolate framework membranes for clean energy applications. Energy Environ. Sci. 2012, 5, 7637–7646. [Google Scholar] [CrossRef]
- Reinsch, H.; van der Veen, M.A.; Gil, B.; Marszalek, B.; Verbiest, T.; De Vos, D.; Stock, N. Structures, sorption characteristics, and nonlinear optical properties of a new series of highly stable aluminum MOFs. Chem. Mater. 2013, 25, 17–26. [Google Scholar] [CrossRef]
- Cuadrado-Collados, C.; Fernández-Català, J.; Fauth, F.; Cheng, Y.Q.; Daemen, L.L.; Ramirez-Cuesta, A.J.; Silvestre-Albero, J. Understanding the breathing phenomena in nano-ZIF-7 upon gas adsorption. J. Mater. Chem. A 2017, 5, 20938–20946. [Google Scholar] [CrossRef] [Green Version]
- Zhai, L.; Yu, X.; Wang, Y.; Zhang, J.; Ying, Y.; Cheng, Y.; Peh, S.B.; Liu, G.; Wang, X.; Cai, Y. Polycrystalline rare-earth metal-organic framework membranes with in-situ healing ability for efficient alcohol dehydration. J. Membr. Sci. 2020, 610, 118239. [Google Scholar] [CrossRef]
- Marti, A.M.; Van, M.; Balkus, K.J. Tuning the crystal size and morphology of the substituted imidazole material, SIM-1. J. Porous Mater. 2014, 21, 889–902. [Google Scholar] [CrossRef]
- Sabetghadam, A.; Liu, X.; Benzaqui, M.; Gkaniatsou, E.; Orsi, A.; Lozinska, M.M.; Sicard, C.; Johnson, T.; Steunou, N.; Wright, P.A. Influence of Filler Pore Structure and Polymer on the Performance of MOF-Based Mixed-Matrix Membranes for CO2 Capture. Chem. A Eur. J. 2018, 24, 7949–7956. [Google Scholar] [CrossRef] [Green Version]
- Song, S.; Liu, C.; Ding, R.; Gao, X.; Wang, M.; Li, Z.; Zhao, X. Isomerous Al-BDC-NH2 metal-organic frameworks for metronidazole removal: Effect of topology structure. J. Solid State Chem. 2022, 314, 123376. [Google Scholar] [CrossRef]
- Zhang, F.; Zou, X.; Feng, W.; Zhao, X.; Jing, X.; Sun, F.; Ren, H.; Zhu, G. Microwave-assisted crystallization inclusion of spiropyran molecules in indium trimesate films with antidromic reversible photochromism. J. Mater. Chem. 2012, 22, 25019–25026. [Google Scholar] [CrossRef]
- Frentzel-Beyme, L.; Kolodzeiski, P.; Weiß, J.-B.; Schneemann, A.; Henke, S. Quantification of gas-accessible microporosity in metal-organic framework glasses. ChemRxiv 2022. [Google Scholar] [CrossRef]
- Gandara-Loe, J.; Bueno-Perez, R.; Missyul, A.; Fairen-Jimenez, D.; Silvestre-Albero, J. Molecular Sieving Properties of Nanoporous Mixed-Linker ZIF-62: Associated Structural Changes upon Gas Adsorption Application. ACS Appl. Nano Mater. 2021, 4, 3519–3528. [Google Scholar] [CrossRef]
- Vaidhyanathan, R.; Bradshaw, D.; Rebilly, J.N.; Barrio, J.P.; Gould, J.A.; Berry, N.G.; Rosseinsky, M.J. A family of nanoporous materials based on an amino acid backbone. Angew. Chem. Int. Ed. 2006, 45, 6495–6499. [Google Scholar] [CrossRef] [PubMed]
- Akpinar, I.; Drout, R.J.; Islamoglu, T.; Kato, S.; Lyu, J.; Farha, O.K. Exploiting π–π interactions to design an efficient sorbent for atrazine removal from water. ACS Appl. Mater. Interfaces 2019, 11, 6097–6103. [Google Scholar] [CrossRef] [PubMed]
- DMello, M.E.; Sundaram, N.G.; Singh, A.; Singh, A.K.; Kalidindi, S.B. An amine functionalized zirconium metal–organic framework as an effective chemiresistive sensor for acidic gases. Chem. Commun. 2019, 55, 349–352. [Google Scholar] [CrossRef] [PubMed]
- Abid, H.R.; Rada, Z.H.; Li, Y.; Mohammed, H.A.; Wang, Y.; Wang, S.; Arandiyan, H.; Tan, X.; Liu, S. Boosting CO2 adsorption and selectivity in metal–organic frameworks of MIL-96 (Al) via second metal Ca coordination. RSC Adv. 2020, 10, 8130–8139. [Google Scholar] [CrossRef] [Green Version]
- Benzaqui, M.; Pillai, R.S.; Sabetghadam, A.; Benoit, V.; Normand, P.; Marrot, J.; Menguy, N.; Montero, D.; Shepard, W.; Tissot, A. Revisiting the aluminum trimesate-based MOF (MIL-96): From structure determination to the processing of mixed matrix membranes for CO2 capture. Chem. Mater. 2017, 29, 10326–10338. [Google Scholar] [CrossRef] [Green Version]
- Chowdhury, P.; Mekala, S.; Dreisbach, F.; Gumma, S. Adsorption of CO, CO2 and CH4 on Cu-BTC and MIL-101 metal organic frameworks: Effect of open metal sites and adsorbate polarity. Microporous Mesoporous Mater. 2012, 152, 246–252. [Google Scholar] [CrossRef]
- Mao, Y.; Huang, H.; Cao, W.; Li, J.; Sun, L.; Jin, X.; Peng, X. Room temperature synthesis of free-standing HKUST-1 membranes from copper hydroxide nanostrands for gas separation. Chem. Commun. 2013, 49, 5666–5668. [Google Scholar] [CrossRef]
- Mao, Y.; Li, J.; Cao, W.; Ying, Y.; Sun, L.; Peng, X. Pressure-assisted synthesis of HKUST-1 thin film on polymer hollow fiber at room temperature toward gas separation. ACS Appl. Mater. Interfaces 2014, 6, 4473–4479. [Google Scholar] [CrossRef]
- Li, Y.S.; Liang, F.Y.; Bux, H.; Feldhoff, A.; Yang, W.S.; Caro, J. Molecular sieve membrane: Supported metal–organic framework with high hydrogen selectivity. Angew. Chem. 2010, 122, 558–561. [Google Scholar] [CrossRef]
- Huang, A.; Wang, N.; Kong, C.; Caro, J. Organosilica-functionalized zeolitic imidazolate framework ZIF-90 membrane with high gas-separation performance. Angew. Chem. 2012, 124, 10703–10707. [Google Scholar] [CrossRef]
- Huang, A.; Caro, J. Covalent post-functionalization of zeolitic imidazolate framework ZIF-90 membrane for enhanced hydrogen selectivity. Angew. Chem. Int. Ed. 2011, 50, 4979–4982. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.N.; Gonzalez, M.A.; McCarthy, M.C.; Jeong, H.-K. An unconventional rapid synthesis of high performance metal–organic framework membranes. Langmuir 2013, 29, 7896–7902. [Google Scholar] [CrossRef] [PubMed]
- Kang, Z.; Fan, L.; Wang, S.; Sun, D.; Xue, M.; Qiu, S. In situ confinement of free linkers within a stable MOF membrane for highly improved gas separation properties. CrystEngComm 2017, 19, 1601–1606. [Google Scholar] [CrossRef]
- Wang, J.; Wang, Y.; Liu, Y.; Wu, H.; Zhao, M.; Ren, Y.; Pu, Y.; Li, W.; Wang, S.; Song, S. Ultrathin ZIF-8 Membrane through Inhibited Ostwald Ripening for High-Flux C3H6/C3H8 Separation. Adv. Funct. Mater. 2022, 32, 2208064. [Google Scholar] [CrossRef]
- Huang, A.; Liu, Q.; Wang, N.; Caro, J. Organosilica functionalized zeolitic imidazolate framework ZIF-90 membrane for CO2/CH4 separation. Microporous Mesoporous Mater. 2014, 192, 18–22. [Google Scholar] [CrossRef]
- Cacho-Bailo, F.; Etxeberría-Benavides, M.; Karvan, O.; Téllez, C.; Coronas, J. Sequential amine functionalization inducing structural transition in an aldehyde-containing zeolitic imidazolate framework: Application to gas separation membranes. CrystEngComm 2017, 19, 1545–1554. [Google Scholar] [CrossRef] [Green Version]
- Yin, H.; Wang, J.; Xie, Z.; Yang, J.; Bai, J.; Lu, J.; Zhang, Y.; Yin, D.; Lin, J.Y. A highly permeable and selective amino-functionalized MOF CAU-1 membrane for CO2–N2 separation. Chem. Commun. 2014, 50, 3699–3701. [Google Scholar] [CrossRef]
- Dou, Z.; Cai, J.; Cui, Y.; Yu, J.; Xia, T.; Yang, Y.; Qian, G. Preparation and Gas Separation Properties of Metal-Organic Framework Membranes. Z. Anorg. Allg. Chem. 2015, 641, 792–796. [Google Scholar] [CrossRef]
- Wu, W.; Li, Z.; Chen, Y.; Li, W. Polydopamine-modified metal–organic framework membrane with enhanced selectivity for carbon capture. Environ. Sci. Technol. 2019, 53, 3764–3772. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, S.; Liu, H.; Peng, L.-M. Carbon nanotube-based three-dimensional monolithic optoelectronic integrated system. Nat. Commun. 2017, 8, 15649. [Google Scholar] [CrossRef] [Green Version]
- Robeson, L.M. The upper bound revisited. J. Membr. Sci. 2008, 320, 390–400. [Google Scholar] [CrossRef]
- Burns, R.L.; Koros, W.J. Defining the challenges for C3H6/C3H8 separation using polymeric membranes. J. Membr. Sci. 2003, 211, 299–309. [Google Scholar] [CrossRef]
- Xu, X.; Hartanto, Y.; Nikolaeva, D.; He, Z.; Chergaoui, S.; Luis, P. High-performance ZIF-8/biopolymer chitosan mixed-matrix pervaporation membrane for methanol/dimethyl carbonate separation. Sep. Purif. Technol. 2022, 293, 121085. [Google Scholar] [CrossRef]
- Mahdipoor, H.R.; Halladj, R.; Babakhani, E.G.; Amjad-Iranagh, S.; Ahari, J.S. Synthesis, characterization, and CO2 adsorption properties of metal organic framework Fe-BDC. RSC Adv. 2021, 11, 5192–5203. [Google Scholar] [CrossRef]
- Tovar, T.M.; Zhao, J.; Nunn, W.T.; Barton, H.F.; Peterson, G.W.; Parsons, G.N.; LeVan, M.D. Diffusion of CO2 in large crystals of Cu-BTC MOF. J. Am. Chem. Soc. 2016, 138, 11449–11452. [Google Scholar] [CrossRef]
- Yan, T.; Tong, M.; Yang, Q.; Liu, D.; Guo, Y.; Zhong, C. Large-scale simulations of CO2 diffusion in metal–organic frameworks with open Cu sites. Chin. J. Chem. Eng. 2022, 42, 1–9. [Google Scholar] [CrossRef]
- Jin, H.; Mo, K.; Wen, F.; Li, Y. Preparation and pervaporation performance of CAU-10-H MOF membranes. J. Membr. Sci. 2019, 577, 129–136. [Google Scholar] [CrossRef]
- Liu, X.; Wang, C.; Wang, B.; Li, K. Novel organic-dehydration membranes prepared from zirconium metal-organic frameworks. Adv. Funct. Mater. 2017, 27, 1604311. [Google Scholar] [CrossRef]
- Miyamoto, M.; Hori, K.; Goshima, T.; Takaya, N.; Oumi, Y.; Uemiya, S. An Organoselective Zirconium-Based Metal–Organic-Framework UiO-66 Membrane for Pervaporation. Eur. J. Inorg. Chem. 2017, 2017, 2094–2099. [Google Scholar] [CrossRef]
- Dong, X.; Lin, Y. Synthesis of an organophilic ZIF-71 membrane for pervaporation solvent separation. Chem. Commun. 2013, 49, 1196–1198. [Google Scholar] [CrossRef]
- Wu, F.; Lin, L.; Liu, H.; Wang, H.; Qiu, J.; Zhang, X. Synthesis of stable UiO-66 membranes for pervaporation separation of methanol/methyl tert-butyl ether mixtures by secondary growth. J. Membr. Sci. 2017, 544, 342–350. [Google Scholar] [CrossRef]
- Wu, F.; Cao, Y.; Liu, H.; Zhang, X. High-performance UiO-66-NH2 tubular membranes by zirconia-induced synthesis for desulfurization of model gasoline via pervaporation. J. Membr. Sci. 2018, 556, 54–65. [Google Scholar] [CrossRef]
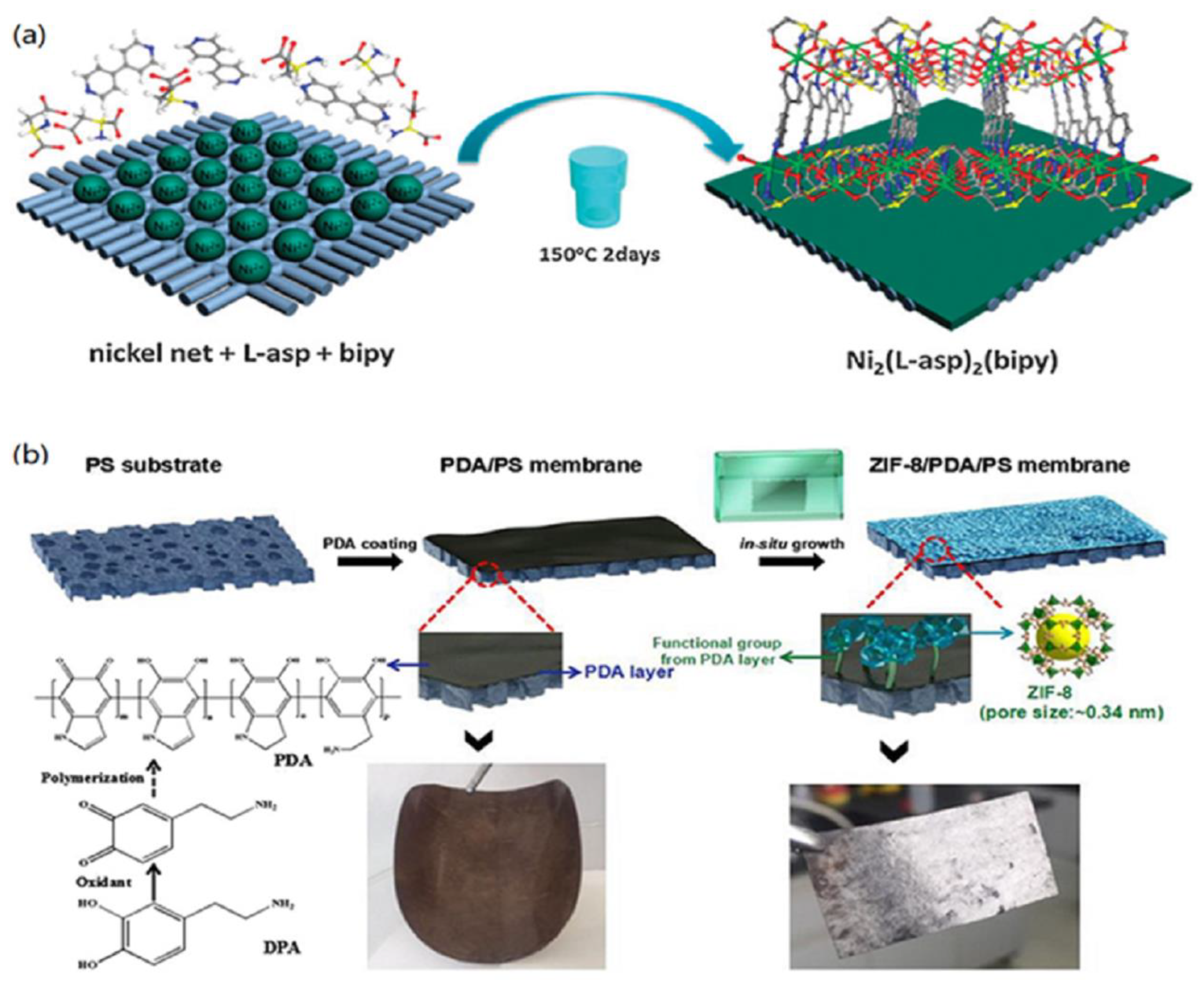
| MOF Name | Metal | Ligand | Formula Composition | Pore Volume (cm3/g) | BET Surface Area (N2) (m2/g) | Window Size (Å) | Ref. |
|---|---|---|---|---|---|---|---|
| UiO-66 | Zr | 1,4-benzenedicarboxylate (BDC) | Zr6O4(OH)4(BDC)6 | 0.36 | 970 | 6 | [124] |
| MOF-5 (Zn) | Zn | BDC | Zn4O(BDC)3 | 1.4 | 3800 | 12 | [125] |
| ZIF-71 | Zn | 4,5-dichloroimidazole (dcIm) | Zn(dcIm)2 | 0.452 | 1007 | 4.8 | [126] |
| CAU-10-H | Al | benzene-1,3-dicarboxylate (1,3-H2BDC) | [Al(OH)(benzene-1,3-dicarboxylate)]·nH2O | 0.43 | 635 | 7 | [127] |
| ZIF-7 | Zn | Benzimidazole (H-bIM) | Zn(bIM)2 | 0.207 | 380 | 3 | [128] |
| ZIF-8 | Zn | 2-Methylimidazole (Hmim) | Zn(mim)2 | 0.554 | 1344 | 3.4 | [126] |
| Sm-DOBDC | Sm | 2,5-dihydroxy-1,4-benzenedicarboxylate (DOBDC) | Sm6(OH)8(DOBDC)6 | 0.263 | 520 | N/A | [129] |
| SIM-1(ZIF-94) | Zn | 4,5-imidazolecarboxaldehyde | Zn(4-methyl-5-imidazolcarboxaldehyde)2 | 0.19 | 471 | 2.6 | [130,131] |
| CAU-1 | Al | 2-amino-1,4-benzene dicarboxylic acid (H2BDC-NH2) | Al8(OH)4(OCH3)8 | 0.59 | 1021.7 | 4 | [132] |
| MIL-100 | In | 1,3,5-benzenetricarboxylic acid (H3BTC) | In3O(H2O)2OH(BTC)2 | 0.636 | 1456 | 4.6 and 8.2 | [133] |
| ZIF-62 | Zn | Imidazole (Im) and benzimidazole (Bim) | Zn(Im)1.75(Bim)0.25 | 0.20 | 257 | 1.4 | [134,135] |
| Ni2(L-asp)2(bipy) | Ni | L-aspartic acid and 4,4′-bipyridine (L-asp and bipy) | Ni2(L-asp)2(bipy) | N/A | 247 (CO2) | 3.8 Å × 4.7 Å | [136] |
| ZIF-90 | Zn | imidazolate-2-carboxyaldehyde (ICA) | Zn(ICA)2 | 0.561 | 1360 | 2.86 | [126] |
| UiO-66-NH2 | Zr | 2-amino-1,4-benzenedicarboxylic acid (H2BDC-NH2) | Zr6O4(OH)4(BDC-NH2)6 | 0.48 | 1073 | 7.5 | [137,138] |
| MIL-96 | Al | H3BTC | Al12O(OH)18(H2O)3(Al2(OH)4) [BTC]6·24H2O | 0.24 | 629.98 | 3.6 Å × 4.5 Å | [139,140] |
| HKUST-1 | Cu | benzene 1,3,5-tricarboxylate (BTC) | Cu3(BTC)2(H2O)3 | 0.75 | 1663 | 9 | [141] |
| Molecule | Kinetic Diameter (Å) |
|---|---|
| H2 | 2.89 |
| CO2 | 3.30 |
| N2 | 3.64 |
| CH4 | 3.80 |
| C3H6 | 3.90 |
| C3H8 | 4.30 |
| MOF | Support | Method | Mixture (i/j) | Mass Ratio (i/j) | T (°C) | Flux (g/m2·h) | i Permeance (GPU) | j Permeance (GPU) | Separation Factor (βi/j) | Selectivity (αi/j) | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|
| CAU-10-H | alumina | secondary growth | H2O/EtOH | 10–90 | 40 | 397 | 5391.77 | 12.52 | 324 | 430.61 | [162] |
| CAU-10-H | alumina | secondary growth | H2O/EtOH | 10–91 | 65 | 493 | 1914.48 | 10.63 | 148 | 180.09 | [162] |
| Ni2(l-asp)2bipy | SiO2 | secondary growth | H2O/EtOH | 50/50 | 30 | 28,100 | 373,279.57 | 1169.2 | 73.6 | 319.26 | [65] |
| Sm-DOBDC | alumina | secondary growth | H2O/EtOH | 5/95 | 50 | 786.4 | N/A | N/A | >9481 | N/A | [129] |
| SIM-1 | glass | mechanical press | H2O/EtOH | 55.82/44.18 | 25 | 460 | 10,493.15 | 0.1589 | >10,000 | >66,036.24 | [119] |
| UiO-66 | Yttria-Stabilized Zirconia | in situ | H2O/EtOH | 5/95 | 50 | 2550 | 8776.12 | 276.85 | 32.9 | 31.7 | [163] |
| MIL-96 | alumina | secondary growth | H2O/Ethyl acetate | 4.4/95.6 | 60 | 70 | N/A | N/A | >1279 | N/A | [61] |
| UiO-66 | alumina | liquid-phase epitaxy | EtOH/H2O | 10/90 | 50 | 1490 | N/A | N/A | 4.9 | N/A | [164] |
| ZIF-71 | alumina | interfacial synthesis | EtOH/H2O | 5–95 | 25 | 2601 | 17,619.83 | 29,951.78 | 6.88 | 0.59 | [76] |
| ZIF-71 | ZnO | secondary growth | EtOH/H2O | 5–95 | 25 | 322.18 | 1980.52 | 3833.32 | 6.07 | 0.52 | [165] |
| UiO-66 | alumina | secondary growth | MeOH/MTBE | 5–95 | 40 | 1210 | 2733.36 | 6.92 | 597 | 395.06 | [166] |
| UiO-66 | alumina | secondary growth | MeOH/MTBE | 15/85 | 40 | 1920 | 2423.83 | 15.35 | 147 | 157.9 | [166] |
| UiO-66-NH2 | alumina | in situ | Thiophene/n-octane | 0.13/99.87 | 40 | 2160 | ~21,000 | ~4000 | 17.96 (enrichment factor) | ~5.8 | [167] |
| Solvent | Kinetic Diameter/Å |
|---|---|
| Water | 2.65 |
| Methanol (MeOH) | 3.8 |
| Ethanol (EtOH) | 4.3 |
| Isopropanol | 4.6 |
| Thiophene | 4.6 |
| Methyl tert-butyl ether (MTBE) | 6.2 |
| Ethyl acetate | 5.2 |
| N-octane | 4.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, X.; Hartanto, Y.; Zheng, J.; Luis, P. Recent Advances in Continuous MOF Membranes for Gas Separation and Pervaporation. Membranes 2022, 12, 1205. https://doi.org/10.3390/membranes12121205
Xu X, Hartanto Y, Zheng J, Luis P. Recent Advances in Continuous MOF Membranes for Gas Separation and Pervaporation. Membranes. 2022; 12(12):1205. https://doi.org/10.3390/membranes12121205
Chicago/Turabian StyleXu, Xiao, Yusak Hartanto, Jie Zheng, and Patricia Luis. 2022. "Recent Advances in Continuous MOF Membranes for Gas Separation and Pervaporation" Membranes 12, no. 12: 1205. https://doi.org/10.3390/membranes12121205
APA StyleXu, X., Hartanto, Y., Zheng, J., & Luis, P. (2022). Recent Advances in Continuous MOF Membranes for Gas Separation and Pervaporation. Membranes, 12(12), 1205. https://doi.org/10.3390/membranes12121205







