Taxonomic Revision of the Genus Croaker Johnius (Perciformes: Sciaenidae) in Taiwanese Waters
Abstract
:1. Introduction
2. Materials and Methods
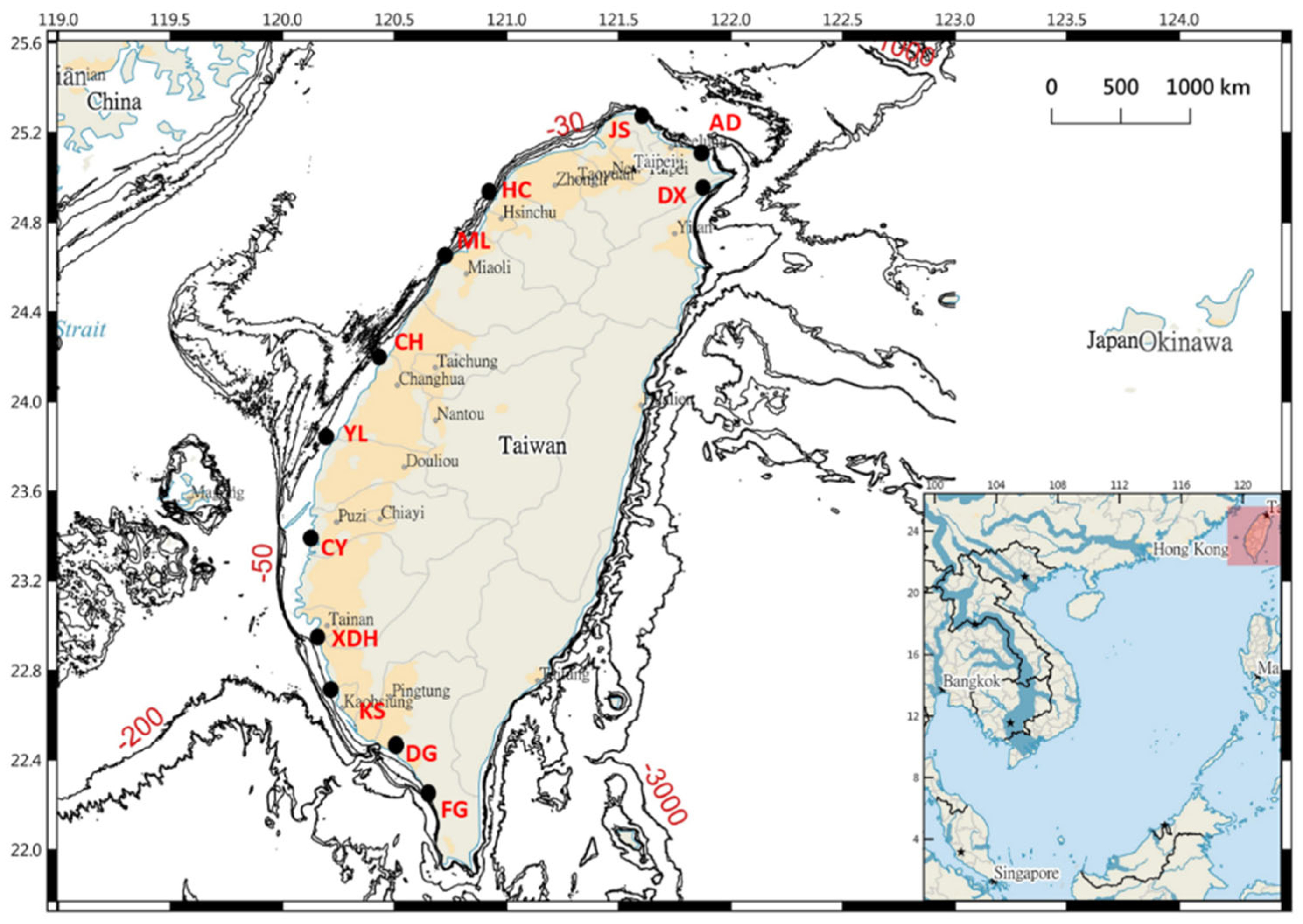
2.1. Sampling and Examination of Specimens
2.2. Integrated Approach to Verifying Johnius Species
3. Results
3.1. Genus Wak Lin, 1938, Accepted as a Junior Synonym of Johnius Bloch, 1793
3.2. Synonyms and Invalid Names of Nominal Species of Johnius Bloch, 1793
3.3.Integrated Identification of Taiwan Johnius species
4. Systematics
- Johnius Bloch, 1793. Naturgesch. auslaänd. Fische, Part 7. J. Morino & Comp., Berlin: 132 (type species: Johnius carutta Bloch).
- Bola Hamilton-Buchanan, 1822. Fishes of Ganges: 73 (type species: Bola coitor Hamilton-Buchanan; junior synonym); Talwar, 1970, Proc. Indian Acad. Sci., 72B: 266.
- Apeches Gistel, 1848. Naturges. Thierr.: ix (type species: Johnius carutta Bloch; substitute name for Johnius Bloch).
- Pseudomycterus Ogibly, 1908. Proc. Roy. Soc. Qld.,21: 94 (type species: Pseudomycterus maccullochii Ogilby).
- Wak Lin, 1938. Lingnan Sci. J., 17(2): 35, 378 (type species: Bola coitor Hamilton-Buchanan; substitute name for Bola Hamilton-Buchanan, 1822, unnecessary substitute name preoccupied by Bola Gunther).
- Blythia Talwar, 1971. (non-Blythia Theobald, 1868), J. Inland Fish. Soc. India, 3: 23 (type species: Umbrina dussumieri Valenciennes; junior synonym).
Key to the Species of Johnius Occurring in Taiwan Waters
- 1a.
- Chin with barbel…………………………………... J. amblycephalus
- 1b.
- Chin without barbel. …………………………………………….... 2
- 2a.
- Inner row of lower jaw teeth enlarged…………………………... 3
- 2b.
- Inner row of lower jaw teeth uniform in size…………………... 4
- 3a.
- Lateral line with distinct white streak; spinous dorsal fin with distinct black blotch on upper 1/3. .…………………. J. distinctus
- 3b.
- Lateral line without white streak; spinous dorsal fins are often dark-tipped or grayish .. .……………………………..J. borneensis
- 4a.
- Scales small, seven to eight scale rows above lateral line from dorsal fin origin; lower fins often dark gray to black…………..J. belangerii
- 4b.
- Scales large, five to six scale rows above lateral line from origin dorsal fin . . . . . . ….………………………………………………….. . … 5
- 5a.
- Caudal fin pointed; gill rakers slender, comprising more than 40% of gill filaments; prepelvic to the isthmus region ctenoid scaled, rarely cycloid scales…………………………………………….. . J. grypotus
- 5b.
- Caudal fin rhomboidal; gill rakers short, comprising less than 40% of gill filaments; prepelvic to the isthmus region ctenoid scaled, gradually turning cycloid scales…….…………………………………..……6
- 6a.
- Gill rakers very short, 6.3−7.9% of eye diameter; outer gill rakers of first arch 12−13 ……………………………………..J. trewavasae
- 6b.
- Gill rakers short, 7.9−13.0% of eye diameter; outer gill rakers of first arch 14−15. . . . ……………................................. .J. taiwanensis
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mok, H.K.; Yu, H.Y.; Ueng, J.P.; Wei, R.C. Characterization of sounds of the blackspotted croaker Protonibea diacanthus (Sciaenidae) and localization of its spawning sites in estuarine coastal waters of Taiwan. Zool. Stud. 2009, 48, 325–333. [Google Scholar]
- Zhang, L.L.; Liu, M.; Fang, L.P.; Xu, Q.; Lin, J.J. Reproductive Biology of Johnius taiwanensis (Perciformes: Sciaenidae) in Fujian Waters, Southern China. Zool. Stud. 2019, 58, e38. [Google Scholar] [CrossRef] [PubMed]
- Lo, P.C.; Liu, S.H.; Chao, N.L.; Nunoo, F.K.; Mok, H.K.; Chen, W.J. A multi-gene dataset reveals a tropical New World origin and Early Miocene diversification of croakers (Perciformes: Sciaenidae). Mol. Phylogenet. Evol. 2015, 88, 132–143. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, K. Sciaenidae. In FAO Species Identification Guide for Fishery Purposes; The Living Marine Resources of the Western Central Pacific. Volume 5: Bony Fishes Part 3 (Menidae to Pomacentridae); Carpenter, K.E., Niem, V.H., Eds.; FAO: Rome, Italy, 2001; pp. 3117–3174. [Google Scholar]
- Chao, N.L.; Chang, C.W.; Chen, M.H.; Guo, C.C.; Lin, B.A.; Liou, Y.Y.; Shen, K.N.; Liu, M. Johnius taiwanensis, a new species of Sciaenidae from the Taiwan Strait, with a key to Johnius species from Chinese waters. Zootaxa 2019, 4651, 259–270. [Google Scholar] [CrossRef] [PubMed]
- Lo, P.C.; Liu, S.H.; Nor, S.A.M.; Chen, W.J. Molecular exploration of hidden diversity in the Indo-West Pacific sciaenid clade. PLoS ONE 2017, 12, e0176623. [Google Scholar] [CrossRef] [Green Version]
- Fricke, R.; Eschmeyer, W.N.; Van der Laan, R. Catalog of Fishes: Genera, Species, References; California Academy of Sciences: San Francisco, CA, USA, 2022. [Google Scholar]
- Chu, Y.T.; Lo, Y.L.; Wu, H.L. A Study on the Classification of the Sciaenoid Fishes of China, with Description of New Genera and Species; Shanghai Science and Technology Press: Shanghai, China, 1963; 140p. [Google Scholar]
- Lal Mohan, R.S. A synopsis of the Indian genera of the fishes of the family Sciaenidae. Indian J. Fish. 1972, 16, 82–98. [Google Scholar]
- Trewavas, E. The sciaenid fishes (croakers or drums) of the Indo-West-Pacific. Trans. Zool. Soc. Lond. 1977, 33, 253–541. [Google Scholar] [CrossRef]
- Talwar, P.K. Fauna of India and the Adjacent Countries: Pisces. Perciformes, Sciaenidae; Zoological Survey of India: Kolkata, India, 1995; 144p. [Google Scholar]
- Yu, L.C.; Shen, S.C. Study on sciaenoid fishes from the adjacent waters around Taiwan. Ann. Taiwan. Mus. 1987, 30, 65–133, (In Chinese with English Abstract). [Google Scholar]
- Shen, S.C. Fishes of Taiwan; National Taiwan University: New Taipei, Taiwan, 1993; 960p. [Google Scholar]
- Lin, Y.C.; Mok, H.K.; Huang, B.Q. Sound characteristics of big-snout croaker Johnius macrorhynus (Sciaenidae). J. Acoustic. Soc. Am. 2007, 121, 586–593. [Google Scholar] [CrossRef]
- Shao, K.T.; Lin, J.; Wu, C.H.; Yeh, H.M.; Cheng, T.Y. A dataset from bottom trawl survey around Taiwan. ZooKeys 2012, 198, 103–109. [Google Scholar] [CrossRef] [Green Version]
- Liou, Y.Y. Taxonomic Revision of Taiwan Sciaenidae with a Note on Seasonal Variation along Yun-Lin Coast. Master’s Dissertation, Department of Marine Biotechnology and Resources, National Sun Yat-sen University, Kaohsiung, Taiwan, 2016; 118p. (In Chinese). [Google Scholar]
- Seah, Y.G.; Hanafi, N.; Mazlan, A.G.; Chao, N.L. A new species of Larimichthys from Terengganu, east coast of Peninsular Malaysia (Perciformes: Sciaenidae). Zootaxa 2015, 3956, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Hubbs, C.L.; Lagler, K.F. Fishes of the Great Lakes Region. Smith, G.R., Ed.; University of Michigan Press: Ann Arbor, MI, USA, 2004; 332p. [Google Scholar] [CrossRef]
- Hanafi, N.; Chen, M.H.; Seah, Y.G.; Chang, C.W.; Liu, S.Y.V.; Chao, N.L. Johnius sasakii, a new species of croaker (Perciformes: Sciaenidae) with a key to Johnius from East Malaysia, Borneo. Zootaxa 2022, 5116, 393–409. [Google Scholar] [CrossRef] [PubMed]
- Hammer, Ø.; Harper, D.A.; Ryan, P.D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 9. [Google Scholar]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547. [Google Scholar] [CrossRef]
- Kimura, M. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J. Mol. Evol. 1980, 16, 111–120. [Google Scholar] [CrossRef]
- Guindon, S.; Dufayard, J.F.; Lefort, V.; Anisimova, M.; Hordijk, W.; Gascuel, O. New algorithms and methods to estimate maximum-likelihood phylogenies: Assessing the performance of PhyML 3.0. Syst. Biol. 2010, 59, 307–321. [Google Scholar] [CrossRef] [Green Version]
- Lefort, V.; Longueville, J.E.; Gascuel, O. SMS: Smart model selection in PhyML. Mol. Biol. Evol. 2017, 34, 2422–2424. [Google Scholar] [CrossRef] [Green Version]
- Zhou, J.; Liu, X.; Stones, D.S.; Xie, Q.; Wang, G. MrBayes on a graphics processing unit. Bioinformatics 2011, 27, 1255–1261. [Google Scholar] [CrossRef] [Green Version]
- Posada, D. JModelTest: Phylogenetic model averaging. Mol. Biol. Evol. 2008, 25, 1253–1256. [Google Scholar] [CrossRef]
- Drummond, A.J.; Rambaut, A. BEAST: Bayesian evolutionary analysis by sampling trees. BMC Evol. Biol. 2007, 7, 214. [Google Scholar] [CrossRef] [Green Version]
- Fowler, H.W. Contribution to the biology of the Philippine Archipelago and adjacent regions. The fishes of the families Banjosidae, Lethrinidae, Sparidae, Girellidae, Kyphosidae, Oplegnathidae, Gerridae, Mullidae, Emmelichthyidae, Sciaenidae, Sillaginidae, Arripidae, and Enoplosidae collected by the United States Bureau of Fisheries Steamer “Albatross”, chiefly in Philippine seas and adjacent waters. Bull. US Nat. Mus. 1933, 100, 1–465. [Google Scholar]
- Lin, S.Y. Further notes on sciaenid fishes of China. Lingnan Sci. J. 1938, 17, 378–381. [Google Scholar]
- Hamilton, F. An Account of the Fishes Found in the River Ganges and Its Branches; Atlas of 39 pls; A. Constable: London, UK, 1822; p. vii-405p. [Google Scholar]
- Trewavas, E. The Sciaenid Fishes with a Single Mental Barbel; American Society of Ichthyologists and Herpetologists: Lawrence, KS, USA, 1964; pp. 107–117. [Google Scholar]
- Talwar, P.K. On the Generic Relationship of Bola coitor Hamilton (Pisces: Sciaenidae) with a Redescription of the Species. Master’s Dissertation, Indian Academy of Sciences, Bengaluru, India, 1969. [Google Scholar]
- Shen, S.C. Coastal Fishes of Taiwan; National Taiwan University: New Taipei, Taiwan, 1984; 190p. [Google Scholar]
- Shao, K.T.; Chen, J.Y. The Fish Illustrations—Taiwan More Than Seven Hundred Kinds of Common Fish Illustrations; Yuan-Liou Publishing Company: Taipei, Taiwan, 2003; 431p. [Google Scholar]
- Shen, S.C.; Wu, R.K. Fishes of Taiwan; National Museum of Marine Biology & Aquarium: Pingtung County, Taiwan, 2011; 896p. [Google Scholar]
- Shao, K.T. The Fishes of Matsu; Lienchiang County Government: Nangan, Taiwan, 2012; 240p. [Google Scholar]
- Chen, M.H.; Chen, K.S.; Chen, H.S.; Chen, C.Y. The Marine Fishes of Taijiang National Park; Taijiang National Park: Tainan, Taiwan, 2019; 359p. (In Chinese) [Google Scholar]
- Koeda, K.; Ho, H.C. Fishes of Southern Taiwan II; National Museum of Marine Biology and Aquarium: Pingtung, Taiwan, 2020; 650p.
- Shao, K.T. The Fish Database of Taiwan; WWW Web Electronic Publication. Available online: http://fishdb.sinica.edu.tw (accessed on 25 May 2022).
- Lal Mohan, R.S.; Trewavas, E.; Whitehead, P.J.P. Sciaenidae. In FAO Species Identification Sheets for Fishery Purposes; Volume 4. Western Indian Ocean (Fishing Area 51); Fishers, W., Bianchi, G., Eds.; FAO: Rome, Italy, 1984. [Google Scholar]
- Sasaki, K. Two new and two resurrected species of the sciaenid genus Johnius (Johnius) from the West Pacific. Jpn. J. Ichthyol. 1992, 39, 191–199. [Google Scholar] [CrossRef]
- Sasaki, K. Johnius (Johnieops) philippinus, a new species sciaenid from the Philippines, with a synopsis of species included in the subgenus a Johnieops. Ichthyol. Res. 1999, 46, 271–279. [Google Scholar] [CrossRef]
- Sasaki, K. Sciaenid fishes of the Indian Ocean (Teleostei, Perciformes). Mem. Fac. Kochi Univ. Ser. D (Biol.) 1996, 16, 83–96. [Google Scholar]
- Sasaki, K.; Amaoka, K. Johnius distinctus (Tanaka, 1916), a senior synonym of J. tingi (Tang, 1937) (Perciformes, Sciaenidae). Jpn. J. Ichthyol. 1989, 35, 466–468. [Google Scholar] [CrossRef]
- Sasaki, K. Phylogeny of the Family Sciaenidae, with notes on its zoogeography (Teleostei, Perciformes). Mem. Fac. Fish. Sci. Hokkaido Univ. 1989, 36, 1–137. [Google Scholar] [CrossRef]
- Sasaki, K. Johnius grypotus (Richardson, 1846), resurrection of a Chinese sciaenid species. Jpn. J. Ichthyol. 1990, 37, 224–229. [Google Scholar]
- Lin, B.A.; Guo, C.C.; Fang, L.P.; Yang, W.D.; Liu, M. Complete mitochondrial genome and the phylogenetic position of a new species, Johnius taiwanensis (Perciformes: Sciaenidae) from Chinese waters. Mitochondrial DNA Part B 2020, 5, 920–921. [Google Scholar] [CrossRef] [Green Version]
- Aguirre, W.E.; Shervette, V.R. Morphological diversity of the Cynoscion group (Perciformes: Sciaenidae) in the Gulf of Guayaquil region, Ecuador: A comparative approach. Environ. Biol. Fishes 2005, 73, 403–413. [Google Scholar] [CrossRef]
- Khaironizam, M.Z.; Akaria-Ismail, M.; Armbruster, J.W. Cyprinid fishes of the genus Neolissochilus in Peninsular Malaysia. Zootaxa 2015, 3962, 139–157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walton, S.E.; Gan, H.M.; Raghavan, R.; Pinder, A.C.; Ahmad, A. Disentangling the taxonomy of the mahseers (Tor spp.) of Malaysia: An integrated approach using morphology, genetics and historical records. Rev. Fish. Sci. Aquacult. 2017, 25, 171–183. [Google Scholar] [CrossRef] [Green Version]
- Ciccotto, P.J.; Page, L.M. Revision of the genus Henicorhynchus, with a revised diagnosis of Gymnostomus (Cyprinidae: Labeoninae). Copeia 2020, 108, 485–502. [Google Scholar] [CrossRef]
- Parenti, P. An annotated checklist of fishes of the family Sciaenidae. J. Anim. Diversit. 2020, 2, 1–92. [Google Scholar] [CrossRef]
- Tseng, H.C.; You, W.L.; Huang, W.; Chung, C.C.; Tsai, A.Y.; Chen, T.Y.; Lan, K.W.; Gong, G.C. Seasonal variations of marine environment and primary production in the Taiwan Strait. Front. Mar. Sci. 2020, 7, 38. [Google Scholar] [CrossRef]
- Chen, C.H. Studies on the Species Composition and Abundance Fluctuation of Set-Net Fisheries in the Coastal Waters of Northern Taiwan. Master’s Dissertation, Department of Environmental Biology and Fisheries Science, National Taiwan Ocean University, Keelung, Taiwan, 2006; 99p. (In Chinese). [Google Scholar]





| Character | Description | Acronym | Landmark | |
|---|---|---|---|---|
| 28 | Morphometric measurements | |||
| Total length | Tip of snout to tip of caudal | TL | 1–2 | |
| Standard length | Tip of snout to end of hypural | SL | 1–3 | |
| Head length | Tip of snout to posterior edge of operculum | HL | 1–4 | |
| Snout length | Tip of snout to anterior edge of orbit | SnL | 1–5 | |
| Upper jaw length | Tip of snout to the posterior edge of maxilla | UJL | 1–6 | |
| Tip of upper jaw to mouth hinge length | Tip of snout to hinge of mouth | UJHL | 1–7 | |
| Tip of lower jaw to mouth hinge length | Tip of mandible to hinge of mouth | LJHL | 8–7 | |
| Eye diameter | The greatest bony diameter of orbit | ED | 5–9 | |
| Interorbital width | The smallest width between both orbits | IOW | 10–10 | |
| Pectoral fin length | Insertion of pectoral fin to distal of pectoral fin | P1L | 11–12 | |
| Pelvic fin length | Insertion of pelvic fin to the distal of pelvic fin | P2L | 13–14 | |
| Body depth | Origin of first dorsal fin spine to insertion of pelvic fin | BD | 13–15 | |
| Body width | The greatest width between pectoral fin insertion | BW | 11–11 | |
| 1st dorsal fin base length | Origin to end of first dorsal fin | D1BL | 15–16 | |
| Length of 4th spine of 1st dorsal fin | Proximal to distal of fourth spine at first dorsal fin | 4D1L | 17–18 | |
| 2nd dorsal fin base length | Origin to end of second dorsal fin | D2BL | 16–19 | |
| Length of 5th ray of 2nd dorsal fin length | Proximal to distal of fifth ray at second dorsal fin | 5RD2L | 20–21 | |
| Anal fin base length | Origin to end of anal fin | ABL | 22–23 | |
| Length of 2nd anal fin spine | Proximal to distal of second anal spine | 2AL | 24–25 | |
| Length of 1st anal fin ray | Proximal to distal of first anal ray | 1RAL | 26–27 | |
| Predorsal length | Tip of snout to origin of first dorsal fin | PD1L | 1–15 | |
| Distance between snout to origin of 2nd dorsal fin | Tip of snout to origin of second dorsal fin | PD2L | 1–16 | |
| Prepectoral length | Tip of snout to insertion of pectoral fin | PP1L | 1–11 | |
| Prepelvic length | Tip of snout to insertion of pelvic fin | PP2L | 1–13 | |
| Preanal length | Tip of snout to origin of the anal fin | PAL | 1–22 | |
| Caudal peduncle depth | The smallest depth of the tail base | CPD | 28–29 | |
| Gill raker length | Proximal to distal of gill raker at the first gill arch junction | GRL | - | |
| Gill filament length | Proximal to distal of gill filament at the first gill arch junction | GFL | - | |
| 15 | Meristic counts | |||
| Dorsal fin spine | Number of spines in dorsal fin | DFS | - | |
| Dorsal fin ray | Number of soft dorsal fin rays in dorsal fin | DFR | - | |
| Anal fin spine | Number of spines in anal fin | AFS | - | |
| Anal fin ray | Number of soft dorsal fin rays in anal fin | AFR | - | |
| Pectoral fin ray | Number of soft dorsal fin rays in pectoral fin | PFR | - | |
| Outer gill raker of 1st arch | Number of total gill rakers at outer arch | OGR | - | |
| Upper limb at outer gill raker | Number of upper limbs in gill raker at outer arch | UOGR | - | |
| Lower limb at outer gill raker | Number of lower limbs in gill raker at outer arch | LOGR | - | |
| Inner gill raker of 1st arch | Number of total gill rakers at inner arch | IGR | - | |
| Upper limb at inner gill raker | Number of upper limbs in gill raker at inner arch | UIGR | - | |
| Lower limb at inner gill raker | Number of lower limbs in gill raker at inner arch | LIGR | - | |
| Lateral line pored scale | Number of scales along lateral line | LLS | - | |
| Circumpeduncular scales | Number of scales at the circumference caudal peduncle | CPS | - | |
| Scales above lateral line | Number of scales from origin of dorsal fin to the lateral line scales | SALL | - | |
| Scales below lateral line | Number of scales from origin of anal fin to the lateral line scales | SBLL | - |
| Taxon | Voucher Specimen (NMMB-P) | GSCN Specimen | Localition Sites | Country | Number of Individuals | GenBank No. |
|---|---|---|---|---|---|---|
| Dendrophysa russelii | - | - | - | Taiwan | 2 | NC017606, JQ728562 |
| J. amblycephalus | 37066 | - | - | - | 3 | KX777910, KX777912, KX777913 |
| J. belangerii | - | 869, 868 870, 871 | - | Taiwan | 4 | - |
| J. belangerii | 37067 | 2137, 2105, 2214, 2216 | Miaoli | Taiwan | 1 | OK253026 |
| J. belangerii | - | - | - | Taiwan | 1 | MG917695 |
| J. borneensis | - | - | - | Taiwan | 1 | MG917696 |
| J. borneensis | - | - | - | - | 1 | NC041308 |
| J. borneensis | 37068 | - | - | - | 2 | KX777940-KX777941 |
| J. distinctus | 37069 | TSI02 | Miaoli | Taiwan | 4 | OK326790-OK326800 |
| J. taiwanensis | 37073 | YLN0202 | Yunlin | Taiwan | 1 | OK355352 |
| J. taiwanensis | 37074 | 2175, 2021 | Miaoli | Taiwan | 1 | OK355356 |
| J. taiwanensis | - | - | - | Taiwan | 3 | MG917694, NC022464, KF211426 |
| J. trewavasae | 37071 | 2252 | Miaoli | Taiwan | 1 | OK253997 |
| J. trewavasae | - | 2251 | Miaoli | Taiwan | 1 | - |
| J. trewavasae | - | - | - | Taiwan | 5 | KX777980-KX777984 |
| J. trewavasae | - | - | - | - | 3 | KP722729, MF083700, MF004324 |
| J. grypotus | 23015 | - | Shangdong | China | 8 | NC021130, KC491206 |
| Species | This Study | [33] | [12] | [13] | [4] | [34] | [35] | [36] | [37] | [38] | [39] | Remarks |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| J. amblycephalus | + | v | v | v | v | v | v | v | v | v | v | |
| J. belangerii | + | x Atrobucca nibe | v | v | v | v | v | v | v | v | ||
| J. borneensis | + | v | v | |||||||||
| J. carutta | x J. distinctus | v | Indian Ocean | |||||||||
| J. distinctus | + | v | v | v | v | v | v | |||||
| J. dussumieri | x J. belangerii | x J. borneensis | x J.borneensis | Indian Ocean | ||||||||
| J. fasciatus | n J. belangerii | |||||||||||
| J. grypotus | + | x J. taiwanensis | x J. taiwanensis | x J. belangerii | ||||||||
| J. macrorhynus | x J. grypotus | x J. trewavasae | v | x J. trewavasae | x J. trewavasae | x J. taiwanensis | x J. trewavasae | Indian Ocean to South China Sea | ||||
| J. sina | x J. borneensis | x J. borneensis | Indian Ocean | |||||||||
| J. taiwanensis | + | v | x J. trewavasae | v | ||||||||
| J. tingi | n J. distinctus | |||||||||||
| J. trewavasae | + | v | ||||||||||
| Wak tingi | s J. distinctus |
| Characters Acronym | PC 1 | PC 2 |
|---|---|---|
| HL | −0.06 | 0.11 |
| P1L | −0.04 | 0.12 |
| P2L | −0.01 | 0.05 |
| BD | 0.00 | −0.08 |
| BW | −0.12 | −0.01 |
| D1BL | −0.03 | −0.07 |
| 4D1L | −0.17 | −0.03 |
| D2BL | 0.14 | −0.24 |
| 5RD2L | 0.05 | −0.08 |
| ABL | 0.05 | −0.14 |
| PAL | −0.09 | 0.20 |
| PD1L | −0.10 | 0.08 |
| PD2L | −0.14 | 0.07 |
| PP1L | −0.07 | 0.13 |
| PP2L | −0.06 | 0.03 |
| CPD | −0.06 | −0.03 |
| SnL/HL | −0.13 | 0.17 |
| UJL | 0.13 | 0.13 |
| UJHL | 0.27 | 0.45 |
| LJHL | 0.26 | 0.64 |
| ED/HL | 0.10 | −0.09 |
| IOW/HL | 0.12 | 0.01 |
| 2AL/HL | 0.24 | −0.34 |
| 1RAL/HL | 0.16 | −0.11 |
| GRL/HL | 0.19 | −0.04 |
| GFL/HL | 0.06 | −0.07 |
| GRL/ED | 0.74 | −0.09 |
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | |
|---|---|---|---|---|---|---|---|
| J. amblycephalus (1) | |||||||
| J. belangerii (2) | 0.38 | ||||||
| J. borneensis (3) | 0.35 | 0.18 | |||||
| J. distinctus (4) | 0.34 | 0.21 | 0.12 | ||||
| J. grypotus (5) | 0.36 | 0.17 | 0.17 | 0.19 | |||
| J. taiwanensis (6) | 0.37 | 0.17 | 0.19 | 0.21 | 0.16 | ||
| J. trewavasae (7) | 0.37 | 0.15 | 0.19 | 0.21 | 0.17 | 0.21 | |
| D. russelli (8) | 0.26 | 0.4 | 0.33 | 0.35 | 0.36 | 0.33 | 0.37 |
| Species | J. amblycephalus (n = 5) | J. belangerii (n = 5) | J. borneensis (n = 7) | J. distinctus (n = 6) | J. grypotus (n = 6) | J. taiwanensis (n = 12) | J. trewavasae (n = 5) |
|---|---|---|---|---|---|---|---|
| Standard length (mm) | 121–163 | 139–175 | 100–147 | 87–172 | 79–130 | 120–142 | 140–147 |
| Meristic counts | |||||||
| 1st dorsal fin spine | 10 | 10 | 10 | 10 | 10 | 10 | 10 |
| 2nd dorsal fin spine | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 2nd dorsal fin soft rays | 23–25 | 27–29 | 27–29 | 28–29 | 24–26 | 23–26 | 24–26 |
| Anal fin spine | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
| Anal fin soft rays | 7 | 7 | 7 | 7 | 7–8 | 7 | 7 |
| Pectoral rays | 16–18 | 16–17 | 16–18 | 16–18 | 16–17 | 14–17 | 16–17 |
| Outer gill rakers of 1st arch | 13–14 | 13–15 | 15–18 | 17–18 | 15–18 | 14–15 | 12–13 |
| Upper limb | 4 | 4 | 5–6 | 6–7 | 4–6 | 4–5 | 4–5 |
| Lower limb | 8–9 | 9–11 | 10–12 | 11–13 | 11–12 | 8–10 | 7–8 |
| Inner gill rakers of 1st arch | 9–10 | 10–12 | 11–14 | 12–13 | 11–12 | 10–11 | 11–12 |
| Upper limb | 3 | 3 | 3–4 | 3–4 | 3–4 | 3 | 4 |
| Lower limb | 6–7 | 7–9 | 8–10 | 8–9 | 7–9 | 7–8 | 7–8 |
| Lateral line pored scales | 46–50 | 47–49 | 47–50 | 47–48 | 44–48 | 46–49 | 44–47 |
| Circumpeduncular scales | 14–17 | 14–16 | 14–17 | 13–15 | 12–14 | 14–15 | 12–13 |
| Scales above lateral line | 7–8 | 7 | 5–6 | 5–6 | 5–6 | 5–6 | 4–5 |
| Scales below lateral line | 12–13 | 11–12 | 10–12 | 10–11 | 9–12 | 10–11 | 8–10 |
| Scales on body | Cycloid | Ctenoid | Ctenoid | Ctenoid | Ctenoid | Ctenoid | Ctenoid |
| Scales on head | Cycloid | Ctenoid | Cycloid | Cycloid | Cycloid | Ctenoid and Cycloid | Cycloid |
| Morphometric measurements | |||||||
| As a percentage of SL | |||||||
| Head length | 29.9–31.8 (31 ± 0.8) | 28.2–31.6 (30 ± 1.3) | 320.5–31.8 (31 ± 0.5) | 30.1–32.0 (31 ± 0.9) | 27.8–29.7 (29 ± 0.7) | 29.1–32.0 (30 ± 0.9) | 30.1–31.6 (31 ± 0.7) |
| Pectoral fin length | 20.3–21.8 (21 ± 0.6) | 18.0–19.6 (19 ± 0.7) | 16.7–22.2 (20 ± 2.3) | 19.2–22.7 (20 ± 1.5) | 17.4–20.0 (19 ± 0.9) | 16.3–23.3 (21 ± 1.8) | 15.2–21.6 (18 ± 2.4) |
| Pelvic fin length | 16.2–17.9 (17 ± 0.7) | 16.3–17.8 (17 ± 0.6) | 14.6–17.6 (17 ± 1.0) | 15.0–18.0 (17 ± 1.3) | 14.5–17.3 (16 ± 1.2) | 14.9–19.3 (17 ± 1.2) | 15.1–16.2 (16 ± 0.5) |
| Body depth | 25.0–28.5 (27 ± 1.4) | 28.6–31.2 (30 ± 1.1) | 27.0–29.3 (29 ± 0.9) | 23.2–28.5 (27 ± 2.3) | 26.8–29.4 (28 ± 0.9) | 26.5–30.1 (28 ± 1.1) | 25.6–27.9 (27 ± 0.9) |
| Body width | 16.3–19.1 (18 ± 1.2) | 14.9–16.2 (15 ± 0.5) | 14.3–16.7 (15 ± 0.8) | 13.7–15.6 (15 ± 0.8) | 13.4–15.5 (14 ± 0.7) | 15.2–16.8 (16 ± 0.6) | 15.6–18.3 (17 ± 1.1) |
| 1st dorsal fin base length | 20.6–22.0 (21 ± 0.5) | 20.0–23.0 (21 ± 1.1) | 19.5–22.0 (21 ± 0.9) | 16.6–19.9 (18 ± 1.5) | 16.3–21.2 (19 ± 1.6) | 17.3–21.5 (20 ± 1.4) | 18.3–20.4 (19 ± 0.8) |
| Length of 4th spine of 1st dorsal fin | 18.3–22.9 (21 ± 2.2) | 13.4–16.1 (15 ± 1.1) | 12.7–15.7 (15 ± 1.1) | 12.3–13.0 (13 ± 0.3) | 9.7–15.8 (13 ± 2.2) | 11.9–16.3 (14 ± 1.4) | 12.5–16.3 (15 ± 1.5) |
| 2nd dorsal fin base length | 34.3–38.7 (37 ± 1.7) | 39.6–43.6 (42 ± 1.5) | 36.8–40.7 (39 ± 1.4) | 37.7–41.4 (39 ± 1.7) | 39.3–43.1 (41 ± 1.2) | 34.5–42.4 (38 ± 2.5) | 35.1–38.7 (37 ± 1.5) |
| Length of 5th ray of 2nd dorsal fin | 6.2–10.9 (8 ± 1.8) | 8.1–9.0 (8 ± 0.4) | 7.2–8.5 (7 ± 0.5) | 6.0–7.7 (7 ± 0.7) | 9.9–13.2 (11 ± 1.1) | 6.8–12.0 (10 ± 1.8) | 7.0–7.9 (8 ± 0.4) |
| Anal fin base length | 9.7–11.0 (10 ± 0.5) | 10.7–11.7 (11 ± 0.4) | 8.7–10.2 (9 ± 0.55) | 10.5–11.4(11 ± 0.4) | 10.4–12.3 (12 ± 0.7) | 7.9–10.4 (9 ± 1.0) | 9.1–11.4 (10 ± 0.9) |
| Length of 2nd anal fin spine | 7.2–9.5 (8 ± 1.0) | 9.4–10.4 (10 ± 0.4) | 7.7–10.2 (9 ± 0.8) | 7.3–12.2 (9 ± 2.1) | 8.6–10.8 (9 ± 0.9) | 7.6–9.5 (9 ± 0.6) | 8.4–8.7 (9 ± 0.1) |
| Length of 1st anal fin ray | 10.6–12.8 (12 ± 0.9) | 12.7–13.3 (13 ± 0.3) | 11.6–13.6 (12 ± 0.7) | 10.9–15.2 (13 ± 1.8) | 11.0–13.7 (12 ± 1.1) | 10.4–13.9 (13 ± 1.1) | 11.6–12.5 (12 ± 0.4) |
| Preanal length | 66.9–70.4 (69 ± 1.3) | 66.0–70.1 (68 ± 1.5) | 68.2–72.6 (70 ± 1.5) | 68.6–72.0 (71 ± 1.6) | 65.4–68.4 (67 ± 1.0) | 66.7–71.3 (69 ± 1.5) | 70.8–74.8 (72 ± 1.6) |
| Predorsal length | 32.9–36.0 (35 ± 1.2) | 33.9–35.4 (35 ± 0.7) | 33.2–34.5 (33 ± 1.2) | 33.5–35.3 (35 ± 0.8) | 31.4–33.6 (33 ± 0.8) | 32.3–37.1 (35 ± 1.5) | 34.5–37.5 (35 ± 1.3) |
| Distance from snout to origin of 2nd dorsal fin | 54.0–55.7 (55 ± 0.7) | 51.2–55.3 (53 ± 1.5) | 52.1–54.1 (53 ± 0.6) | 49.3–51.8 (51 ± 1.2) | 47.1–51.9 (49 ± 1.7) | 50.7–55.2 (53 ± 1.4) | 51.8–53.2 (52 ± 0.5) |
| Prepectoral length | 30.9–32.6 (32 ± 0.7) | 29.8–30.9 (30 ± 0.5) | 30.5–34.5 (33 ± 1.2) | 31.1–34.2 (32 ± 1.4) | 27.6–30.2 (29 ± 0.9) | 30.2–32.8 (31 ± 0.8) | 30.5–33.2 (32 ± 1.1) |
| Prepelvic length | 33.5–35.2 (34 ± 0.7) | 33.5–35.2 (34 ± 0.8) | 32.0–36.9 (35 ± 1.6) | 33.7–35.9 (35 ± 1.0) | 32.5–34.0 (33 ± 0.5) | 33.5–36.6 (35 ± 1.0) | 36.2–38.0 (37 ± 0.7) |
| Caudal peduncle depth | 9.1–10.0 (10 ± 0.4) | 8.9–10.2 (10 ± 0.5) | 8.5–10.5 (10 ± 0.6) | 8.2–9.7 (9 ± 0.7) | 8.0–9.5 (9 ± 0.5) | 8.8–14.3 (10 ± 1.7) | 9.6–10.2 (10 ± 0.3) |
| As a percentage of HL | |||||||
| Snout length | 27.0–29.2 (28 ± 0.8) | 24.6–30.0 (28 ± 2.0) | 26.3–30.9 (28 ± 1.8) | 25.8–30.9 (28 ± 2.1) | 26.0–27.4 (27 ± 0.6) | 25.5–32.1 (29 ± 2.5) | 28.9–35.1 (32 ± 2.7) |
| Upper jaw length | 41.0–45.7 (43 ± 1.8) | 39.1–43.5 (42 ± 1.7) | 43.5–47.4 (45 ± 1.5) | 42.4–46.8 (45 ± 1.9) | 39.4–48.5 (45 ± 3.1) | 41.1–45.9 (43 ± 1.5) | 41.1–43.4 (42 ± 1.1) |
| Tip upper jaw to hinge length | 25.0–28.0 (26 ± 1.3) | 25.9–30.9 (29 ± 1.8) | 34.3–38.0 (36 ± 1.3) | 32.1–34.9 (34 ± 1.2) | 30.4–34.1 (32 ± 1.4) | 25.9–31.4 (29 ± 2.1) | 27.0–33.6 (29 ± 2.6) |
| Tip lower jaw to hinge length | 18.8–23.2 (21 ± 2.0) | 18.8–25.8 (23 ± 2.5) | 27.4–32.0 (29 ± 1.9) | 27.2–30.7 (29 ± 1.5) | 23.5–30.0 (26 ± 2.5) | 19.9–32.3 (26 ± 4.3) | 20.1–27.6 (22.5 ± 3.0) |
| Eye diameter | 19.0–23.4 (21 ± 1.6) | 21.9–25.4 (23 ± 1.4) | 21.4–25.2 (24 ± 1.4) | 21.1–25.8 (22 ± 2.3) | 21.9–26.4 (25 ± 1.5) | 20.3–24.1 (22 ± 1.1) | 22.9–25.2 (24 ± 0.9) |
| Interorbital width | 27.5–29.3 (28 ± 0.7) | 21.8–24.3 (23 ± 1.1) | 24.3–26.3 (25 ± 0.8) | 25.2–28.6 (27 ± 1.4) | 27.3–30.1 (29 ± 1.1) | 24.0–27.5 (26 ± 0.9) | 22.4–23.8 (23 ± 0.6) |
| 2nd spine of anal fin length | 22.6–31.9 (27 ± 4.0) | 32.2–36.5 (34 ± 2.2) | 25.0–33.5 (30 ± 2.6) | 26.4–31.1 (28 ± 2.1) | 29.8–37.4 (33 ± 3.1) | 25.4–31.6 (28 ± 1.8) | 27.0–28.9 (28 ± 0.7) |
| 1st ray of anal fin length | 34.5–40.4 (38 ± 2.4) | 41.7–46.9 (44 ± 2.5) | 36.7–43.7 (40 ± 2.4) | 36.9–41.8 (40 ± 2.3) | 37.5–47.4 (44 ± 3.9) | 35.7–45.4 (41 ± 3.0) | 37.3–41.3 (39 ± 1.8) |
| Gill raker length | 1.8–2.6 (2 ± 0.3) | 2.4–3.5 (3 ± 0.5) | 3.5–5.5 (5 ± 0.8) | 4.0–5.3 (5 ± 0.5) | 5.8–7.6 (6 ± 0.6) | 1.6–3.0 (3 ± 0.2) | 1.4–2.0 (2 ± 0.2) |
| Gill filament length | 9.5–11.6 (11 ± 1.0) | 8.6–10.2 (9 ± 0.6) | 7.7–11.1 (9 ± 1.2) | 8.3–10.9 (9 ± 1.1) | 9.6–14.9 (12 ± 1.8) | 8.4–11.4 (10 ± 0.9) | 8.5–10.8 (10 ± 0.9) |
| As a percentage of ED | |||||||
| Gill raker length | 9.5–12.4 (11 ± 1.2) | 9.6–14.9 (13 ± 2.2) | 15.4–22.8 (19 ± 3.1) | 18.4–22.8 (21 ± 1.6) | 23.2–28.7 (26 ± 1.7) | 7.9–13.0 (11 ± 1.7) | 6.3–7.9 (7 ± 0.6) |
| Gill filament length | 44.1–60.9 (51 ± 6.8) | 34.3–43.7 (40 ± 3.9) | 11–12 (42 ± 0.4) | 36.3–50.8 (42 ± 5.9) | 39.4–62.6 (49 ± 8.8) | 39.7–52.7 (45 ± 4.2) | 36.0–44.8 (40 ± 3.4) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hanafi, N.; Chen, M.-H.; Seah, Y.G.; Chang, C.-W.; Liu, S.Y.V.; Lai, C.-C.; Chao, N.L. Taxonomic Revision of the Genus Croaker Johnius (Perciformes: Sciaenidae) in Taiwanese Waters. J. Mar. Sci. Eng. 2023, 11, 471. https://doi.org/10.3390/jmse11030471
Hanafi N, Chen M-H, Seah YG, Chang C-W, Liu SYV, Lai C-C, Chao NL. Taxonomic Revision of the Genus Croaker Johnius (Perciformes: Sciaenidae) in Taiwanese Waters. Journal of Marine Science and Engineering. 2023; 11(3):471. https://doi.org/10.3390/jmse11030471
Chicago/Turabian StyleHanafi, Norhafiz, Meng-Hsien Chen, Ying Giat Seah, Chih-Wei Chang, Shang Yin Vanson Liu, Chien-Cheng Lai, and Ning Labbish Chao. 2023. "Taxonomic Revision of the Genus Croaker Johnius (Perciformes: Sciaenidae) in Taiwanese Waters" Journal of Marine Science and Engineering 11, no. 3: 471. https://doi.org/10.3390/jmse11030471
APA StyleHanafi, N., Chen, M.-H., Seah, Y. G., Chang, C.-W., Liu, S. Y. V., Lai, C.-C., & Chao, N. L. (2023). Taxonomic Revision of the Genus Croaker Johnius (Perciformes: Sciaenidae) in Taiwanese Waters. Journal of Marine Science and Engineering, 11(3), 471. https://doi.org/10.3390/jmse11030471






